Abstract
Peptide-loaded poly(lactide-co-glycolide) (PLGA) nanocarriers represent a transformative approach to addressing the challenges of peptide-based therapies. These systems offer solutions to peptide instability, enzymatic degradation, and limited bioavailability by providing controlled release, targeted delivery, and improved stability. The versatility of PLGA nanocarriers extends across therapeutic domains, including cancer therapy, neurodegenerative diseases, vaccine development, and regenerative medicine. Innovations in polymer chemistry, surface functionalization, and advanced manufacturing techniques, such as microfluidics and electrospraying, have further enhanced the efficacy and scalability of these systems. This review highlights the key physicochemical properties, preparation strategies, and proven benefits of peptide-loaded PLGA systems, emphasizing their role in sustained drug release, immune activation, and tissue regeneration. Despite remarkable progress, challenges such as production scalability, cost, and regulatory hurdles remain.
1. Peptide-Loaded PLGA Therapeutics
Peptide-based therapeutics are advancing rapidly due to their high specificity, potency, and wide applicability across various diseases, including cancer, infectious diseases, and neurodegenerative disorders. Peptides are short chains of amino acids connected by peptide bonds, typically comprising two to fifty amino acids. They serve as versatile bioactive molecules in various therapeutic applications, including antimicrobial therapy, immunomodulation, tissue regeneration, and hormonal analogs. Their high specificity and biocompatibility make them attractive therapeutic candidates, but their clinical utility is limited by inherent challenges such as poor bioavailability, susceptibility to enzymatic degradation, and difficulties in targeted delivery [1,2,3,4,5,6,7,8]. These limitations compromise therapeutic efficacy and emphasize the critical need for innovative delivery systems to protect peptides and ensure their functional stability.
Poly(lactide-co-glycolide) (PLGA) has emerged as a leading material for drug delivery due to its biodegradability, biocompatibility, and flexibility in modulating drug release profiles [9,10,11]. These properties make PLGA ideal for peptide encapsulation and delivery. By offering solutions to peptide instability, enzymatic degradation, and limited bioavailability, PLGA-based systems enable controlled release, protection against degradation, and enhanced biodistribution. For instance, sustained and localized drug delivery provided by PLGA nanocarriers mitigates systemic side effects while preserving peptide bioactivity [12,13,14]. Such systems have demonstrated their ability to overcome peptide-specific challenges, including ensuring consistent therapeutic concentrations and mitigating rapid clearance or degradation in vivo [9,15,16,17,18,19]. This strategic integration of peptide therapeutics into advanced delivery platforms enhances their clinical application, offering a robust response to the limitations of traditional peptide therapies.
Peptides can be classified based on their origin into natural, synthetic, or recombinant categories. Natural peptides are derived from the enzymatic hydrolysis of proteins and include examples like the antihypertensive peptide KGYGGVSLPEW obtained from whey protein [16]. Microbial sources contribute antimicrobial peptides such as Dermaseptin-PP, which is used for wound-healing applications [20]. Synthetic peptides, on the other hand, are designed and engineered for specific purposes, such as G17 and G19 peptides for combating resistant bacterial infections like Methicillin-resistant Staphylococcus aureus (MRSA) and E. coli [21]. Recombinant DNA technology enables the large-scale production of therapeutic peptides, exemplified by recombinant Interleukin-10 (IL-10) and myelin oligodendrocyte glycoprotein (MOG) peptides for treating autoimmune encephalomyelitis [15].
Structurally, peptides may be linear, such as antimicrobial peptides like OH-CATH30 used for bacterial keratitis treatment [17], or cyclic, such as somatostatin analogs for long-term cancer therapy [22]. Functionally, peptides serve various roles. Antimicrobial peptides, such as LL37, are known for their ability to enhance wound healing [23], while immunomodulatory peptides, such as ESAT-6(1-20), are employed in vaccine development for tuberculosis [24]. Additionally, hormonal peptides like leuprolide acetate are used for treating prostate cancer and other hormone-related conditions [1]. Peptides also play a pivotal role in regenerative medicine, as seen with BMP-2-derived P24 peptides for bone tissue repair [18].
The versatility of peptide-loaded PLGA systems spans diverse therapeutic domains. For neurological disorders, PLGA nanocarriers facilitate the delivery of therapeutic agents across the blood/brain barrier (BBB), a significant challenge in treating conditions such as Alzheimer’s disease and glioblastoma [25,26,27,28,29]. The incorporation of peptides such as cyclic beta-hairpin BSBP8 for inhibiting amyloid beta aggregation further underscores their potential in addressing neurodegenerative diseases [27].
In vaccine delivery, sustained antigen release and immune system activation achieved through PLGA systems play a pivotal role in eliciting robust and durable responses [24,30,31,32,33]. The use of antigenic peptides, such as ESAT-6(1-20) for tuberculosis vaccines, highlights the ability of PLGA carriers to enhance vaccine efficacy [24]. In cancer therapy, peptide-functionalized PLGA nanoparticles, such as those targeting hepatocellular carcinoma with SP94 peptides, offer precise delivery to tumor sites, minimizing off-target effects and improving therapeutic outcomes [34,35].
Regenerative medicine represents another frontier for peptide-loaded PLGA systems. By leveraging their ability for controlled peptide release, these systems support critical processes such as bone regeneration, wound healing, and nerve repair. Hybrid formulations and functionalized scaffolds further enhance functionality by combining therapeutic and structural roles, extending their utility in tissue engineering applications [18,19,36,37,38]. For example, PLGA systems functionalized with BMP-2-derived peptides have been shown to support osteointegration and enhance the healing of bone defects [19,39].
Despite these advancements, peptide-loaded PLGA systems face challenges in scalability, reproducibility, and cost-effectiveness in large-scale production [40,41,42]. Addressing these hurdles requires continued innovation in polymer chemistry, nanotechnology, and pharmaceutical engineering to optimize these platforms for broader applications and clinical accessibility [14,43,44]. By combining peptide engineering with advancements in nanotechnology, future research can further improve their stability, efficacy, and patient-specific customization. These developments are expected to drive significant progress in personalized medicine and open new avenues for peptide-based therapies [9,15,20].
2. Therapeutic Roles of Peptides and Drugs in PLGA Systems
PLGA-based systems are redefining therapeutic strategies across various medical domains by enabling the targeted delivery and controlled release of bioactive agents. Their applications span cancer treatment, neurodegenerative disease management, tissue regeneration, infectious disease control, and advanced gene delivery systems.
2.1. Cancer Therapy
PLGA nanocarriers have transformed cancer therapy by improving the delivery and effectiveness of therapeutic agents. For instance, leuprolide acetate, encapsulated in PLGA, provides sustained treatment for prostate cancer by modulating hormonal pathways [1]. Functionalized nanoparticles, such as T7 peptide-functionalized carmustine systems, enhance blood/brain barrier (BBB) penetration, addressing challenges in glioma therapy [25].
Immunotherapeutic strategies have also advanced with synthetic long peptides (SLPs) and antigenic peptides, which activate cytotoxic T-cell responses, offering promising pathways for cancer immunotherapy [33,45]. Dual-drug delivery systems, including doxorubicin paired with anti-PD-L1 peptides, combine chemotherapy and immunotherapy for enhanced tumor suppression [46]. Similarly, Trametinib-loaded nanoparticles, cloaked with melanoma-specific T-cell membranes, demonstrate superior tumor-targeting capabilities (Figure 1) [47].
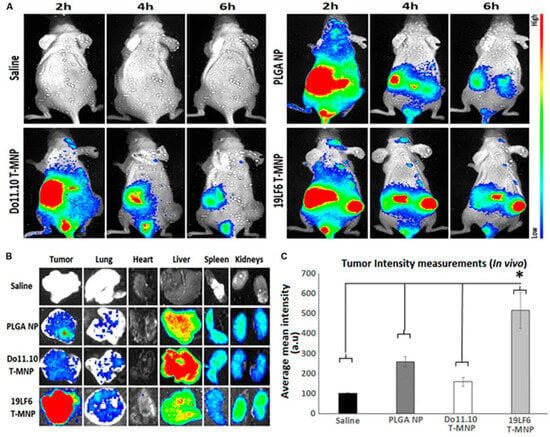
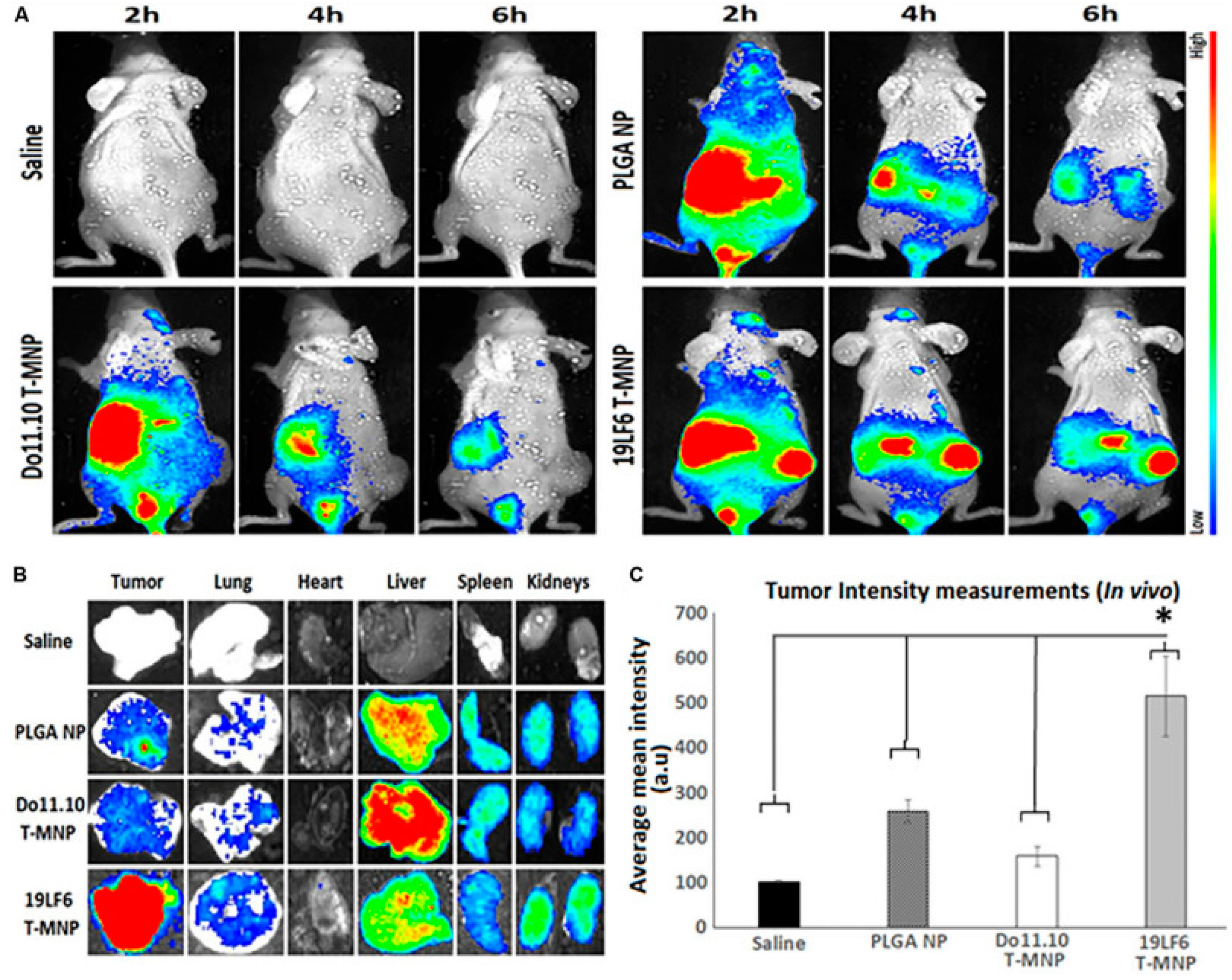
Figure 1.
In vivo and ex vivo analysis of T-MNP biodistribution. (A) Characteristics of the IV-injected NPs on melanoma tumor models with real-time tumor targeting at 2 h, 4 h, and 6 h time intervals. (B) Images of the ex vivo organ biodistribution in different study groups. (C) Intensity of in vivo biodistribution study groups in tissue homogenates measured via fluorescent (n = 6 per group). * Statistically significant with p < 0.05. Poly-lactide-co-glycolide (PLGA); Nanoparticle (NP); T-cell membrane-coated PLGA NPs (T-MNPs); DO10.11 membrane-coated PLGA NPs (D-MNP’s); Coumarin-6 (C-6); A549 membrane-coated PLGA NPs (A-MNP’s); Naked nanoparticle (NNP); Optical Density (OD); Inhibitory Concentration (IC); Reverse Transcriptase Polymerase Chain Reaction (RT-PCR); Ultraviolet–Visible (UV–Vis); 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS). Adopted with permission [47].
2.2. Neurodegenerative Disease Management
Peptide-loaded PLGA systems have demonstrated significant potential in neurodegenerative disease therapy. For Alzheimer’s disease, curcumin functionalized with Tet-1 or B6 peptides reduces amyloid plaques, oxidative stress, and neuroinflammation [3,5]. Insulin encapsulated in CPP-modified PLGA nanoparticles shows promise for treating cognitive dysfunction via intranasal delivery, bypassing the BBB [29]. Innovations like berberine conjugated with Tet-1 to reduce Tau phosphorylation [8] and phytol-loaded nanoparticles for disrupting amyloid aggregates [48] further enhance therapeutic options for complex neurological disorders.
2.3. Tissue Regeneration and Bone Repair
PLGA-based systems incorporating bioactive peptides significantly advance bone regeneration and tissue repair. For example, BMP-2 delivered via PLGA-RADA16 hydrogels or collagen-mimetic peptides enhances osteogenesis in bone defect models [19,38]. Additionally, adrenomedullin and PTHrP1-34 peptides promote osteoblast proliferation and angiogenesis, accelerating tissue repair [49,50]. In neural tissue repair, RADA16-I-BMHP1 peptides embedded in nanofibers support Schwann cell differentiation, fostering peripheral nerve regeneration [51,52]. Figure 2 shows in vivo experiments with the use of 3D-printed PLGA scaffolds with BMP-9 and P-15 peptide hydrogel in the treatment of bone defects in rabbits [38].
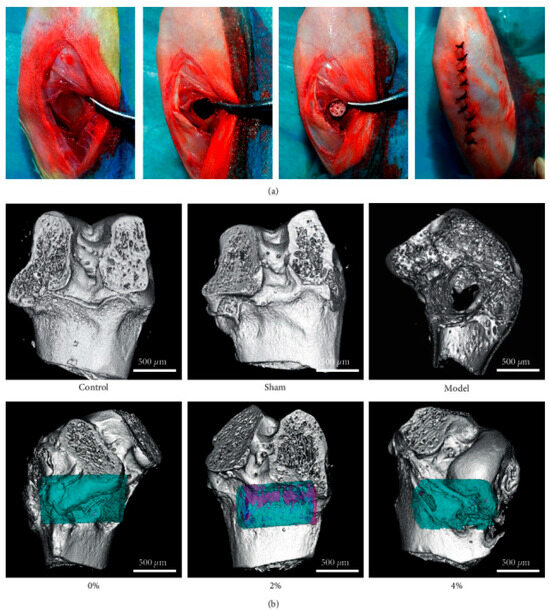
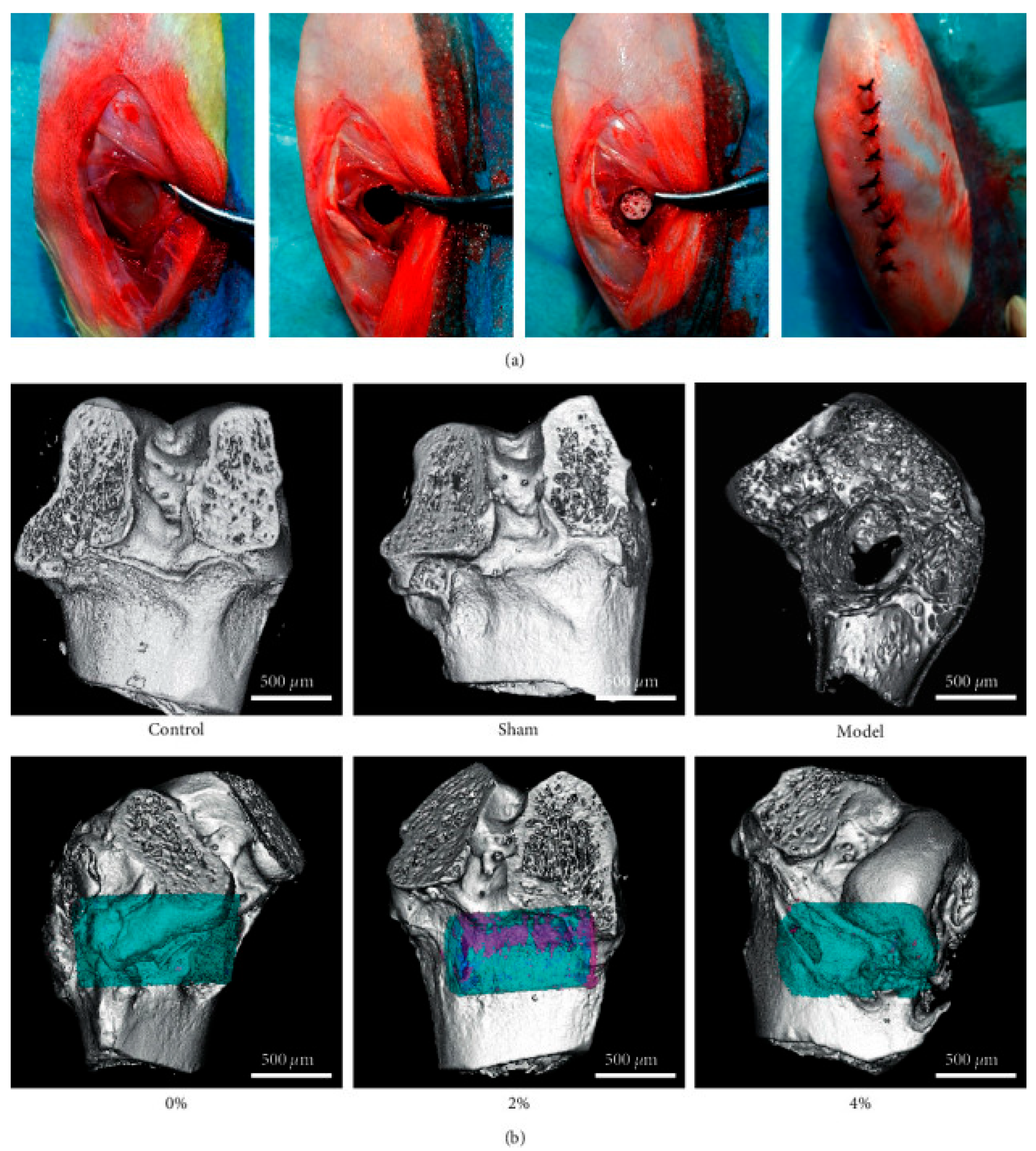
Figure 2.
A 3D-printed PLGA scaffold composite peptide hydrogel implant with BMP-9 and P-15 in the treatment of bone defects in rabbits. (a) Implantation in rabbit bone process. (b) MicroCT test used for the detection of new bone formation and tissue-related protein expressions (blue, scaffold; pink, new bone). The MicroCT detection of 2% polypeptide scaffold showed good bone repair, promoting the expression of ALP, COL-1, OCN, RUNX2, and Sp7. Adopted with permission [38].
2.4. Infectious Diseases and Vaccines
PLGA systems have shown immense potential in combating infectious diseases and advancing vaccine technology. Antimicrobial peptides like SAAP-148 and OH-CATH30 effectively target multidrug-resistant pathogens and bacterial keratitis [9,17]. Vaccine candidates, including W-1 L19 peptides for canine parvovirus and multi-epitope peptides for influenza, elicit robust immune responses [53]. In response to COVID-19, oseltamivir phosphate encapsulated with SBP1 peptides targets the SARS-CoV-2 spike protein, offering potential therapeutic benefits [54].
2.5. Wound Healing
Peptide-functionalized PLGA nanoparticles accelerate wound healing by promoting angiogenesis, granulation, and collagen deposition. LL37 peptides enhance wound closure while providing antimicrobial benefits [23]. BMP-2-loaded PLGA nanoparticles combined with RADA16 hydrogels support osteogenesis and tissue regeneration in wound models [19]. Additionally, MSI-78(4-20), a potent antimicrobial peptide, combats infections and expedites recovery [55].
2.6. Gene Delivery and Genetic Therapy
PLGA nanoparticles functionalized with cell-penetrating peptides or RGD ligands enhance the intracellular delivery of genetic materials. DNA-loaded nanoparticles targeting lung epithelial cells enable effective therapeutic gene expression, with implications for treating genetic disorders [56,57]. Furthermore, short cationic peptide nucleic acids (PNAs) encapsulated in PLGA nanoparticles provide a promising platform for antisense and gene-editing applications [58].
2.7. Combating Infections
Incorporating antimicrobial peptides into PLGA systems offers effective strategies against bacterial infections. Peptides like G17 and G19 target resistant strains, including MRSA and E. coli, while ponericin G1 and bFGF contribute to wound healing and antibacterial action [21,59]. Fusion peptides that combine antimicrobial, osteogenic, and angiogenic properties address bone defect repair and infection control [60].
2.8. Additional Applications
Peptide-loaded PLGA systems have shown versatility in addressing unique medical needs. For example, Asiatic acid functionalized with renal-targeting peptides enhances kidney-specific drug delivery for chronic kidney disease [61]. In glioma treatment, dual-peptide systems such as Euphorbia factor L1 improve tumor targeting by crossing the BBB [62]. SDF-1alpha and VEGF peptides co-loaded in PLGA scaffolds promote angiogenesis and skin regeneration, supporting diabetic wound healing [37].
Table 1 explores the diverse therapeutic applications of peptide-loaded PLGA systems, categorizing active ingredients based on their clinical use, such as antimicrobial, hormonal, and anticancer therapies. These systems address critical challenges, including instability and poor bioavailability, while enabling targeted delivery and immune activation. Patterns reveal their extensive utility in combating antimicrobial resistance, facilitating vaccine development, and advancing regenerative medicine. The data highlight PLGA’s capacity to stabilize peptides, improve targeting precision, and reduce dosing frequency, showcasing its pivotal role in modern therapeutic strategies.

Table 1.
Therapeutic applications and functional enhancements of peptide-loaded PLGA systems.
3. Essential Polymers and Additives in Peptide-Loaded PLGA Systems
PLGA polymers serve as the foundation for numerous peptide-loaded delivery systems due to their excellent biodegradability and ability to provide controlled drug release. Adjustments to PLGA, such as incorporating acid- or ester-terminated variants, enable precise manipulation of its degradation properties and peptide release kinetics in specific environments [10,43]. To enhance solubility and biocompatibility, PLGA is frequently combined with polyethylene glycol (PEG) or its grafted derivatives, resulting in PEG/PLGA polymers that improve both peptide stability and targeting capabilities [35,74,82]. Moreover, hybrid composites blending PLGA with polymers like polycaprolactone (PCL) or gelatin-based hydrogels further expand their use, particularly in neural and skeletal tissue engineering applications [37,85,86]. Figure 3 illustrates the use of co-axial PLGA–gelatin fibers containing stromal cell-derived factor-1α (SDF-1α) and angiogenic signals for enhancing cutaneous wound healing [37]. Additionally, porous PLGA has been adopted in vaccine formulations and tissue-targeted delivery systems to enhance payload encapsulation and enable site-specific release [87].
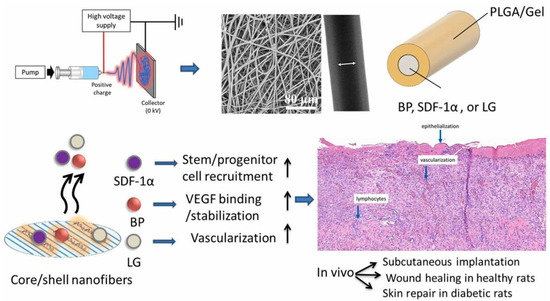
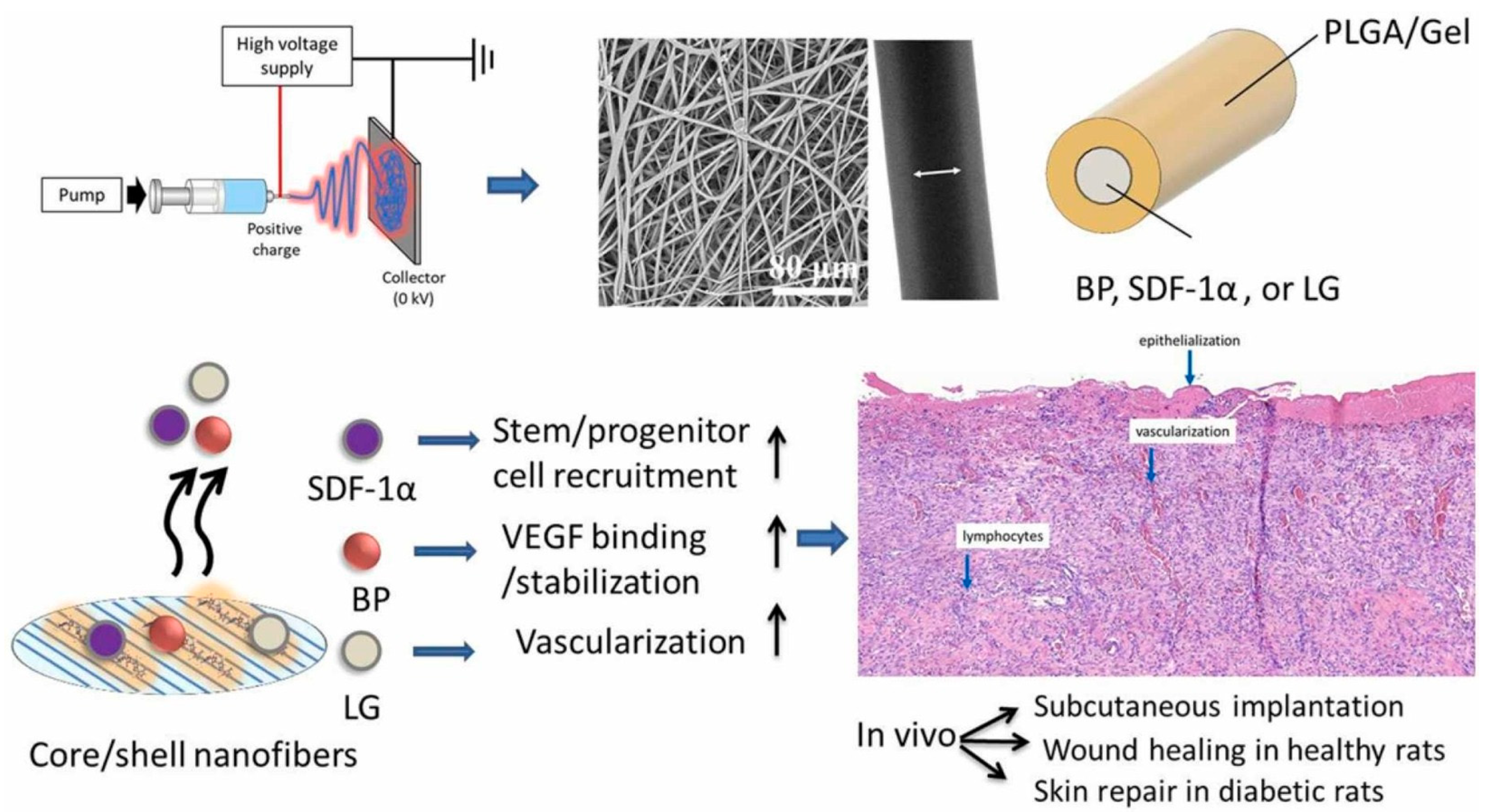
Figure 3.
Shows stromal cell-derived factor-1α (SDF-1), vascular endothelial growth factor (VEGF)-binding peptide (BP), glucagon-like peptide-1 analog (GLP), and liraglutide (LG) in core/shell poly(L-lactide-co-glycolide)/gelatin fibers used to harness synergistic effects for skin repair in healthy and diabetic wound models in rats. Adopted with permission [37].
The integration of peptides into PLGA-based systems offers distinct therapeutic advantages and facilitates precise tissue targeting. For example, cell-penetrating peptides (CPPs) such as TAT, R8, and SBP1 enable effective drug delivery across challenging biological barriers like the blood/brain barrier or intestinal epithelium, significantly improving bioavailability for diseases such as Alzheimer’s and diabetes [28,29,39]. Bioactive peptides, including RGD and YIGSR, play a vital role in promoting skeletal muscle differentiation, while BMP-2 and P-15 peptides are crucial for bone regeneration and repair [19,38,88]. Other specialized peptides, such as OH-CATH30 and MSI-78(4-20), exhibit antimicrobial properties, whereas angiogenic peptides like VEGF and Apelin contribute to wound healing and tissue regeneration [55,59,89].
Surface modifications of PLGA nanoparticles are essential for enhancing stability, targeting, and bioactivity. Coatings with PEG or polyethyleneimine (PEI) effectively prevent aggregation, extend circulation times, and enable applications such as gene delivery [3,90]. Polydopamine coatings promote osteogenic differentiation and improve mechanical integration for bone-related applications, while graphene oxide enhances hydrophilicity and mechanical strength for skeletal tissue engineering [59,91,92]. Functionalization with erythrocyte membranes or maleimide/PEG facilitates immune evasion and ensures stable conjugation, critical attributes for cancer therapeutics [62,82]. These modifications not only extend the circulation lifespan of nanoparticles but also optimize their interaction with target tissues.
Additives are integral to preserving peptide bioactivity and stabilizing PLGA formulations. Materials such as carboxymethyl chitosan (CMCS) and calcium phosphate (Ca3(PO4)2) are commonly used to prevent peptide acylation, a process that can degrade peptide integrity during PLGA breakdown [93,94]. Stabilizers, such as hydroxypropyl-beta-cyclodextrin, protect sensitive peptides like peptide-24, ensuring sustained therapeutic efficacy in bone regeneration applications [95]. Superparamagnetic iron oxide nanoparticles (SPIONs) are incorporated into PLGA systems to enable simultaneous imaging and therapeutic delivery, exemplifying the multifunctionality of these nanoparticles [96]. Fluorapatite further enhances the bioactivity of PLGA formulations, particularly in dental and bone-related applications [97].
Innovative materials also contribute to the versatility of PLGA-based systems. Dopamine ad-layers enhance scaffold adhesion, promoting osteointegration and cellular proliferation [91,92]. Self-immolative-protecting groups safeguard peptides during formulation processes, thereby maintaining their functional stability under challenging conditions [98]. Additionally, fluorescent dyes like Rhodamine-B are used to enable the tracking and imaging of nanoparticles in cellular delivery studies [28,99].
Table 2 highlights the pivotal role of PLGA as a versatile nanocarrier, enhanced by additives like PEG, CPPs, and adjuvants to address specific therapeutic challenges. PEG improves circulation and biocompatibility, while CPPs enable intracellular and barrier-crossing delivery, crucial for CNS and cancer treatments. Adjuvants like CpG amplify vaccine responses, and hydroxyapatite supports bone-targeted delivery. Innovations in self-assembling peptides (e.g., RADA16) bolster regenerative applications. Sustained-release strategies, leveraging stabilizers and ion-pairing agents, extend peptide bioavailability. The integration of these excipients maximizes efficacy in applications like antimicrobials, gene therapy, and vaccines, offering a robust blueprint for tailored peptide-loaded PLGA systems targeting a wide range of diseases.

Table 2.
PLGA compositions and their roles in PLGA-based systems.
4. Methodologies and Optimization Strategies for Peptide-Loaded PLGA Products
Peptide-loaded PLGA systems are critical for advanced drug delivery and tissue engineering applications. This section consolidates methodologies and optimization strategies and details techniques and guidelines for producing high-performance systems.
4.1. Emulsion Solvent Evaporation Techniques
The emulsification solvent evaporation method is a cornerstone for fabricating peptide-loaded PLGA particles, offering flexibility and scalability for various therapeutic applications. Variants like the double-emulsion (water–oil–water) technique are particularly advantageous for encapsulating hydrophilic peptides, such as MOG and plasmid DNA, as they protect the peptides during preparation and improve encapsulation efficiency [15,43,56]. This method’s adaptability extends to large-scale production using microfluidic flow-focusing systems, which ensure particle uniformity and scalability for drugs like exenatide (Figure 4) [80].
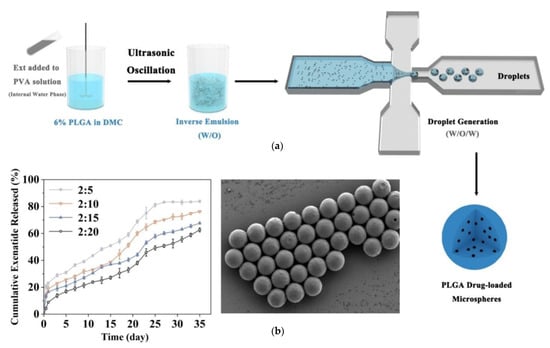
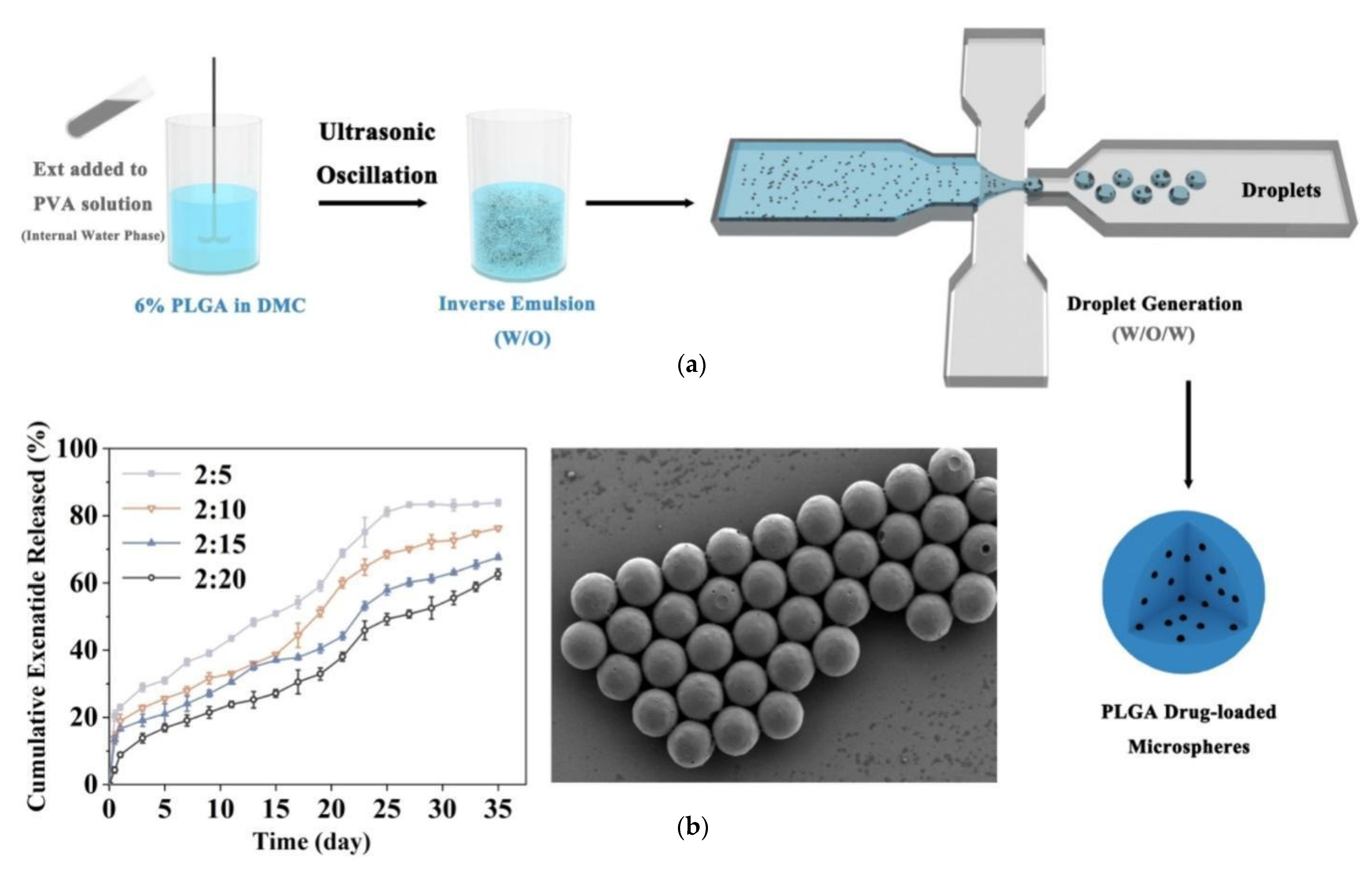
Figure 4.
(a) The preparation of poly(lactic-co-glycolic acid) (PLGA) exenatide-loaded microspheres via water/oil/water (W/O/W) emulsion method with the use of microfluidic device. (b) The design of multi-channel droplet microfluidic devices [80].
Optimizing process parameters, such as homogenization speed and solvent evaporation rate, allows for precise control over particle size, enhancing cellular uptake and distribution [1,9]. Burst release, a common challenge, can be minimized by incorporating stabilizers like polyvinyl alcohol (PVA) or hydrophilic agents like HP-beta-CD, which also support sustained release profiles [50,78]. Moreover, maintaining an alkaline pH in the inner aqueous phase reduces peptide degradation, preserving bioactivity [45]. Surface functionalization with targeting ligands, such as TAT peptides, enhances specificity and uptake for applications like brain delivery [3,107]. The integration of PLGA nanoparticles into hydrogels has also proven effective for achieving controlled and prolonged drug release, particularly for immune modulation applications [36,108]. However, challenges such as residual solvent removal and peptide acylation during PLGA degradation highlight the need for meticulous post-processing and stabilization [94].
4.2. Surface Functionalization and Advanced Coating Techniques
Surface functionalization plays an essential role in enhancing the bioactivity, specificity, and therapeutic potential of peptide-loaded PLGA systems. Functionalization strategies such as thiol–maleimide chemistry and maleimide–thiolether conjugation stabilize peptide attachment, enabling precise targeting and improved therapeutic outcomes [55,82]. Coatings like polydopamine have been shown to enhance cell adhesion and osteogenic activity, making them highly effective for bone regeneration [69]. Similarly, graphene oxide coatings improve mechanical strength and hydrophilicity, broadening the applications of PLGA in wound healing and tissue engineering [59]. Functionalization with cell-penetrating peptides (CPPs) such as transactivator of transcription (TAT) or erythrocyte membranes expands the potential for crossing biological barriers like the blood brain barrier [107,109]. These modifications not only increase cellular uptake but also enable targeted delivery, making them critical for applications in neurology, oncology, and tissue regeneration. By integrating advanced coating techniques, researchers can significantly enhance the therapeutic impact of PLGA-based systems across diverse applications.
4.3. Alternative Fabrication Techniques
Alternative methods like nanoprecipitation, electrospinning, and 3D printing offer unique advantages for specific applications, complementing traditional emulsion techniques. Nanoprecipitation, for example, provides an environmentally friendly, solvent-efficient approach ideal for hydrophobic drug delivery. This method is simple and rapid, requiring minimal solvent use, but may necessitate stabilizers or co-solvents to encapsulate hydrophilic peptides effectively [6,94,110]. Electrospinning, on the other hand, produces nanofibers functionalized with bioactive peptides such as RADA16 or BMP-2, offering a high surface-area-to-volume ratio and precise control over scaffold architecture, making it ideal for neural repair and wound healing [38,51]. Similarly, 3D printing allows for unparalleled control over the scaffold structure, incorporating peptides like BMP-9 to enhance mechanical properties and mineralization for bone defect repair [49,111]. These advanced fabrication techniques continue to expand the horizons of PLGA applications, offering tailored solutions for regenerative medicine and specialized drug delivery.
4.4. Guidelines for Optimizing Key Parameters
Optimizing preparation methods for peptide-loaded PLGA products is crucial for achieving the desired therapeutic outcomes. In emulsion-based systems, adjustments to solvent systems, stabilizer concentrations, and homogenization speeds ensure uniform particle size, high encapsulation efficiency, and controlled release profiles [1,9,78]. Surface coatings such as polydopamine or graphene oxide further enhance mechanical properties and biological interactions, particularly in tissue engineering [59,69]. Release kinetics can be refined through techniques like pH neutralization and the use of stabilizers, such as calcium phosphate salts, to inhibit peptide acylation and maintain bioactivity over extended periods [93,98,112]. Mechanistic modeling, including tri-phasic release analysis, allows for the precise tailoring of burst, erosion, and terminal phases to match specific therapeutic needs [22]. These optimization strategies enable researchers to align PLGA system design with the requirements of various applications, ensuring efficient and effective delivery.
4.5. Integration with Complementary Materials
The integration of PLGA with complementary materials significantly enhances its functionality, broadening its application across diverse therapeutic areas. Chitosan/PLGA blends, for instance, extend release durations in vaccine formulations, particularly for combating multidrug-resistant pathogens (Figure 5) [30]. Composites like PLGA/nano-hydroxyapatite improve bioactivity and osteointegration, making them ideal for bone regeneration applications [60,92]. Similarly, ocular and pulmonary delivery systems benefit from composite designs that enhance drug retention and bioavailability, as seen in formulations incorporating agents like Licochalcone-A [76,108]. These integrations highlight the versatility of PLGA systems and their ability to address complex therapeutic challenges, paving the way for innovative solutions in drug delivery and regenerative medicine.
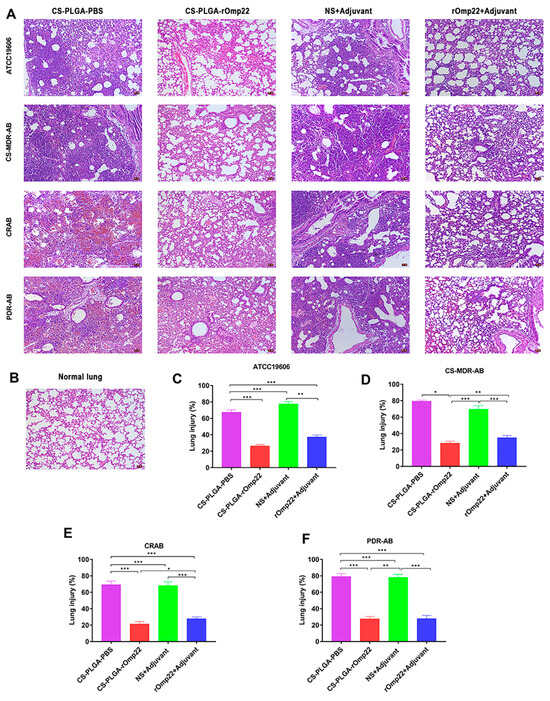
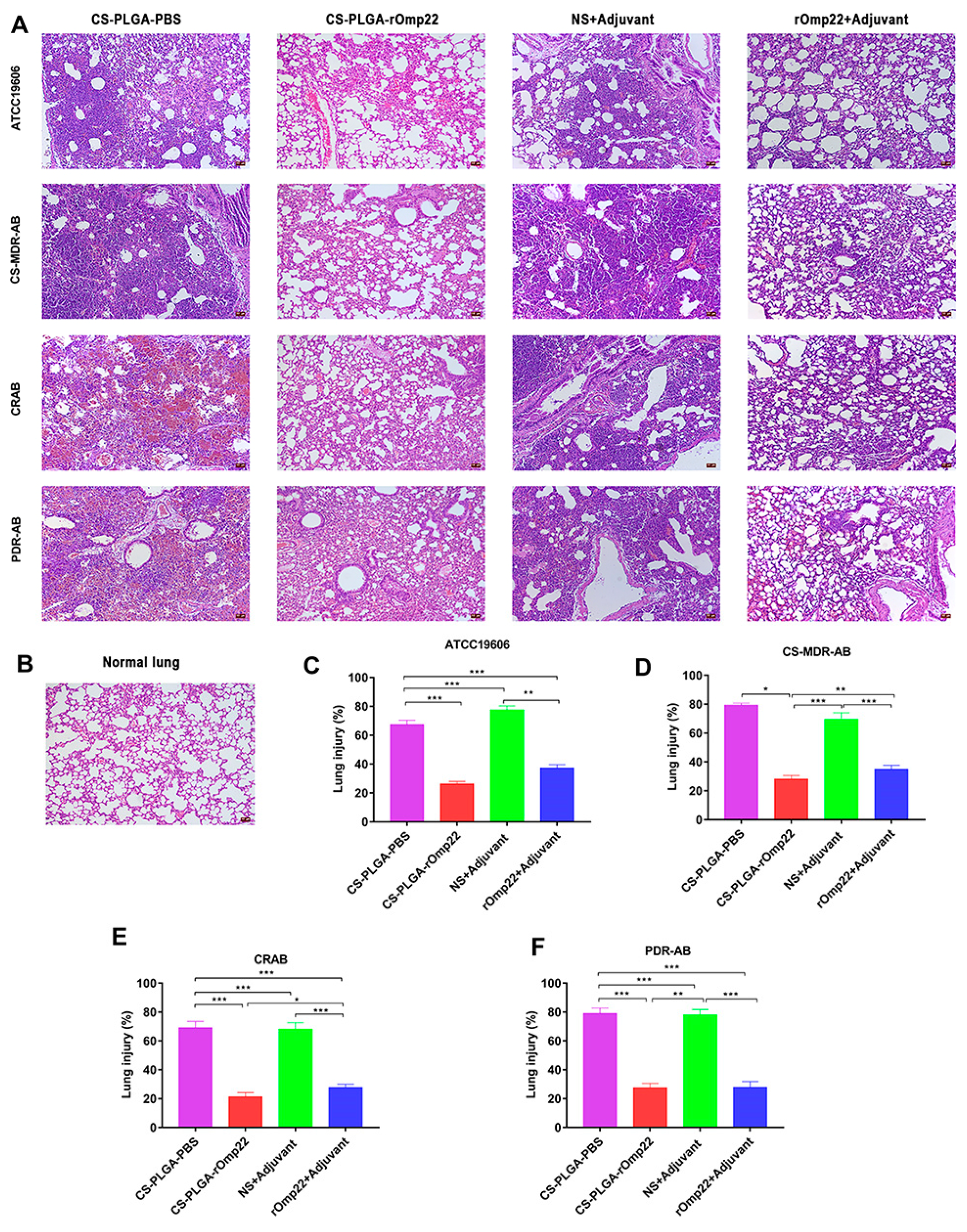
Figure 5.
Histopathology of lung tissue specimens stained with haematoxylin-eosin and observed under a microscope (200×) taken from six mice with A. baumannii from each group at 24 h post-challenge. (A) Lung tissue from different immunized groups subjected to A. baumannii ATCC 19606 and three clinical A. baumannii strains (scale bar, 50 μm). (B) Lung tissue from unimmunized uninfected normal BALB/c mice with normal histological characteristics (scale bar, 50 μm). (C–F) Semiquantitative analysis of the inflammatory area in the lung tissue (n = 6). The histograms with mean percentage of lesion area within the total lung. Data are presented as the means ± SD (n = 6). * p < 0.05, ** p < 0.01, *** p < 0.001. Adopted with permission [30].
4.6. Optimization Strategies
Optimization strategies are critical for refining peptide-loaded PLGA systems to achieve maximum therapeutic efficacy. Techniques such as factorial design and the Box–Behnken Design (BBD) are used to optimize formulations for ocular and pulmonary delivery [108,110]. Simulated accelerated release methods allow for the prediction of long-term performance, facilitating the creation of customized release profiles for drugs like somatostatin analogs [13,22]. Adjusting osmotic pressure and encapsulation parameters further improves the therapeutic potential of peptides such as OH-CATH30 [17].
4.7. Stabilization Techniques
Ensuring effective stabilization is essential for maintaining peptide functionality within PLGA formulations. Approaches like incorporating carboxymethyl chitosan and inorganic cations reduce peptide acylation, preserving stability during degradation [44,94]. Additionally, freeze-drying and nebulization techniques maintain bioactivity and enable efficient pulmonary delivery for respiratory therapies [113]. Intrinsic porogens are often employed to optimize release kinetics, ensuring consistent therapeutic effects [44].
Table 3 highlights the variety of methodologies used in preparing peptide-loaded PLGA systems, from traditional emulsion techniques to advanced 3D printing and surface functionalization. Each method is aligned with specific therapeutic goals, ensuring precision in encapsulation, stability, and delivery. Patterns emphasize how innovations like nanoprecipitation, hydrophobic ion pairing, and functionalized scaffolds enhance encapsulation efficiency, control release, and optimize bioactivity. These methodologies reflect a balance between scalable production and tailored functionality, supporting applications in cancer therapy, tissue regeneration, and immunomodulation.

Table 3.
Methodologies to prepare peptide-loaded PLGA products.
5. Key Physicochemical Features in PLGA Nanocarriers
Peptide-loaded PLGA systems possess a range of physicochemical properties that make them highly adaptable for various medical applications. Particle size is a critical factor, varying from 27 nm to 558 nm for nanoparticles, which are used for targeted drug delivery in conditions such as Alzheimer’s disease and bacterial infections [2,82,103]. Larger structures, such as microspheres designed for Triptorelin acetate, reach sizes up to 35.3 µm, enabling sustained drug release over extended periods [78]. Advanced designs, such as grooved PLGA films with 800 nm ridges, enhance cellular alignment and are particularly suitable for skeletal muscle engineering [88].
5.1. Surface Stability and Charge
The surface charge and stability of PLGA nanocarriers significantly influence their bioactivity and interaction with biological systems. Zeta potentials for these systems range from −46.0 mV to +1.65 mV, depending on surface modifications [9,110]. Specific coatings, such as chitosan (+4.39 mV) and Pep5 modifications (+20.01 mV), enhance colloidal stability and cellular uptake, which are crucial for effective drug delivery in cancer therapies and wound-healing applications [30,119].
5.2. Encapsulation Efficiency and Release Profiles
Encapsulation efficiency in peptide-loaded PLGA products is consistently high, typically exceeding 70%, and reaching as much as 96.56% in systems like BAMLET and octreotide acetate [93,120]. These systems offer tailored release profiles to meet diverse therapeutic requirements, from rapid initial release to sustained delivery over extended periods. For example, BMP-2 systems retain their activity for weeks, supporting bone regeneration [19], while self-immolative-protecting groups ensure a controlled release for up to 50 days [98].
5.3. Biocompatibility and Bioactivity
PLGA systems are highly biocompatible and bioactive, making them suitable for a variety of medical applications. For instance, systems designed for insulin delivery and oral vaccines demonstrate non-cytotoxicity and safety [75,102]. Poly(dopamine)-coated PLGA scaffolds improve osteogenic differentiation and tissue integration, proving effective in bone regeneration [92]. Additionally, RADA16-I-BMHP1 nanofibers facilitate Schwann cell proliferation and gene expression, making them instrumental in neural repair [51,52].
5.4. Therapeutic Efficacy Across Applications
The therapeutic efficacy of peptide-loaded PLGA systems spans a wide range of medical fields. Functionalized nanoparticles, such as those incorporating TNF-alpha-mimicking peptides, activate dendritic cells and prime T-cells, demonstrating promise for vaccine applications (Figure 6) [121]. In Alzheimer’s therapy, BSBP8 nanoparticles reduce amyloid beta levels by 82%, significantly improving cognitive function [27]. For antimicrobial applications, peptides like MSI-78(4-20) and KSL-W provide sustained antibacterial activity and effectively disrupt biofilms, addressing infections such as periodontitis [55,64].
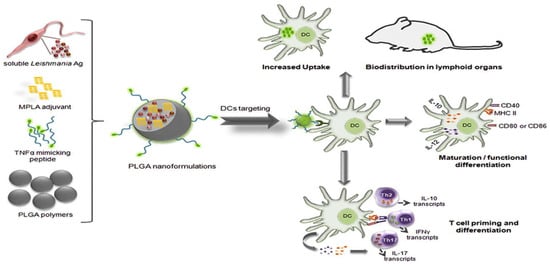
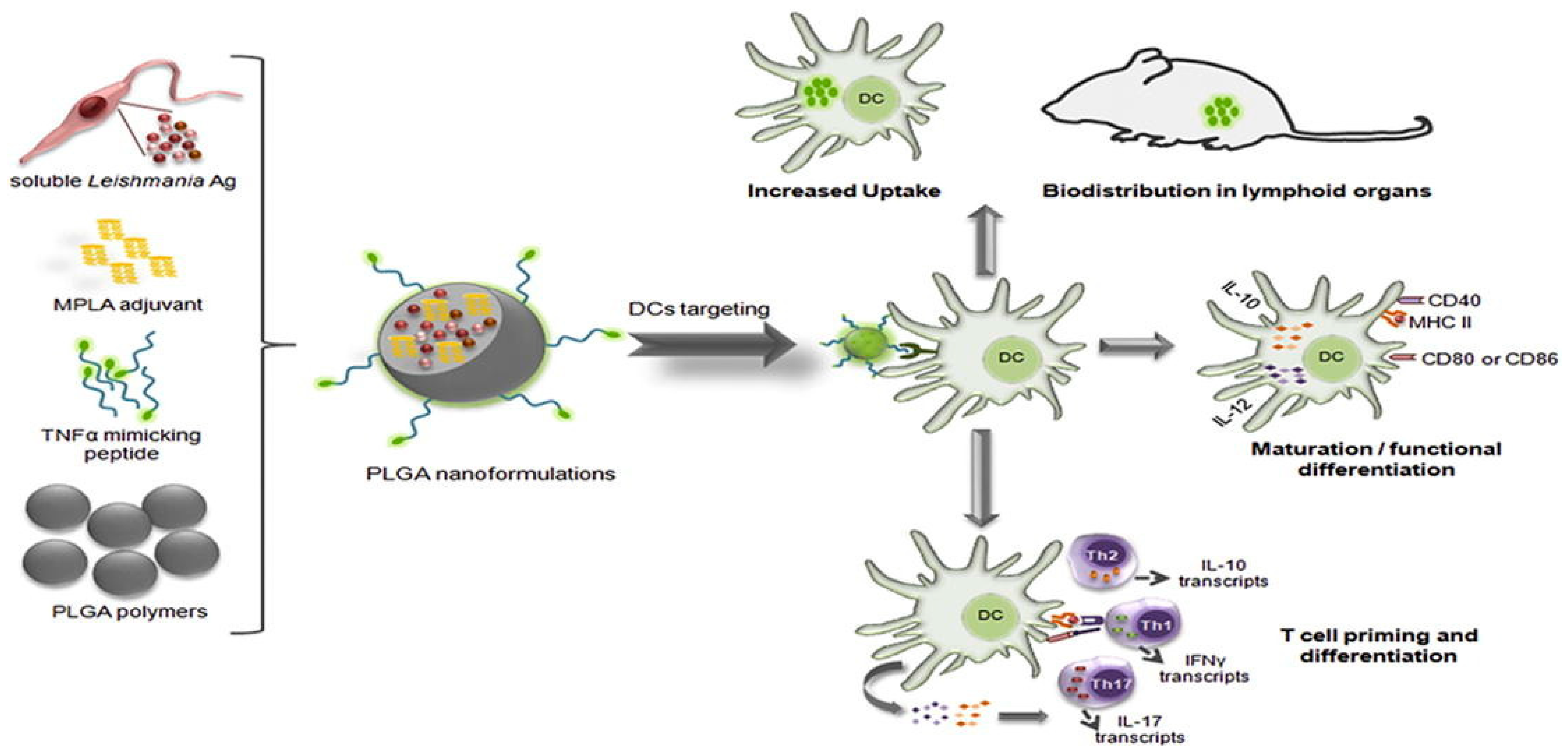
Figure 6.
PLGA NPs surface-modified with a TNF-α-mimicking peptide encapsulated with soluble Leishmania antigens (sLiAg) and MPLA adjuvant. The synthesized PLGA NPs exhibited low cytotoxicity levels, efficient uptake by dendritic cells (DCs), induced maturation and functional differentiation, and increased stimulation of IL-12 and IL-10 production. Adopted with permission [121].
5.5. Advanced Applications and Complex Challenges
PLGA nanocarriers are increasingly utilized in advanced and complex medical applications. BMP-9 and GFOGER peptide-functionalized PLGA scaffolds have shown significant improvements in bone density and mineralization, making them highly effective in bone repair [111]. Graphene oxide and RGD peptide-functionalized fibers support tissue regeneration, particularly in promoting smooth muscle and myoblast differentiation [103]. Cutting-edge designs, such as curcumin/cisplatin nanoparticles and BMP-2-loaded fibers, have demonstrated precise targeting capabilities, achieving superior outcomes in glioblastoma treatment and chronic wound healing [37,109].
Table 4 describes the physicochemical properties critical to the design and performance of peptide-loaded PLGA systems, including size, surface charge, and encapsulation efficiency. Patterns indicate that precise size control enhances tissue penetration, while surface modifications optimize cellular uptake and colloidal stability. Tri-phasic release profiles align with therapeutic needs for chronic conditions. The data highlight the customization potential of PLGA systems in tailoring bioactivity, release profiles, and structural compatibility for applications like bone regeneration, cancer therapy, and neuroprotection.

Table 4.
Key physicochemical features in PLGA nanocarriers.
6. Evaluation Metrics and Methods for Peptide-Loaded PLGA Nanocarriers
Peptide-loaded PLGA systems undergo thorough evaluation to ensure their safety, efficacy, and suitability for a wide array of therapeutic applications. These evaluations focus on physicochemical properties, structural integrity, biological interactions, and in vivo performance.
6.1. Physicochemical and Structural Characterization
Characterizing the physicochemical properties of peptide-loaded PLGA systems is the first step in their development. Analytical techniques such as scanning electron microscopy (SEM), atomic force microscopy (AFM), and transmission electron microscopy (TEM) are employed to assess particle size, zeta potential, and morphology [1,36,57,124,125].
Encapsulation efficiency and stability are evaluated using methods like ALP assays, MALDI-TOF-MS, and high-performance liquid chromatography (HPLC) to confirm peptide bioactivity [94,95,98]. Material interactions and thermal stability are further investigated using spectroscopic techniques (FTIR, NMR) and circular dichroism [27,93,116]. Advanced functionalization methods, including polydopamine-assisted modifications, and fabrication techniques like electrospinning facilitate the creation of innovative material designs [91,111,114].
6.2. Controlled Release Profiles
The ability to control the release of therapeutic peptides is crucial to their effectiveness. In vitro studies evaluate burst and sustained release profiles under varying conditions, including pH [11,14,108,124]. For instance, BMP-2-loaded PLGA systems deliver peptides slowly, enhancing osteoinductive properties in bone regeneration [19,115]. Techniques such as mathematical modeling and flow-through systems are used to optimize polymer degradation and release kinetics, ensuring consistency [14,42,112].
6.3. Cellular and Molecular Bioactivity
The cellular uptake and bioactivity of PLGA nanoparticles are explored through mechanisms like macropinocytosis and endosomal escape, which optimize peptide delivery [29,57,118]. Additional studies, including immune activation assays and cytokine production analyses, validate their suitability for vaccine applications [33,45,126]. In regenerative medicine, osteoblast development, cellular differentiation, and apoptosis induction are monitored to evaluate therapeutic potential [6,50,92].
6.4. Immune-Modulatory Properties
Peptide-loaded PLGA formulations play a critical role in immunotherapy and vaccine development. Evaluations include T-cell response assays, cytokine profiling, and tests for antigen-specific immune activation [32,121,127]. Studies such as immunogenic cell death assays and PD-L1 blockade experiments assess their potential in cancer immunotherapy [46]. Computational and experimental epitope design methodologies further refine vaccine candidates [32,126].
6.5. Therapeutic Efficacy and Safety
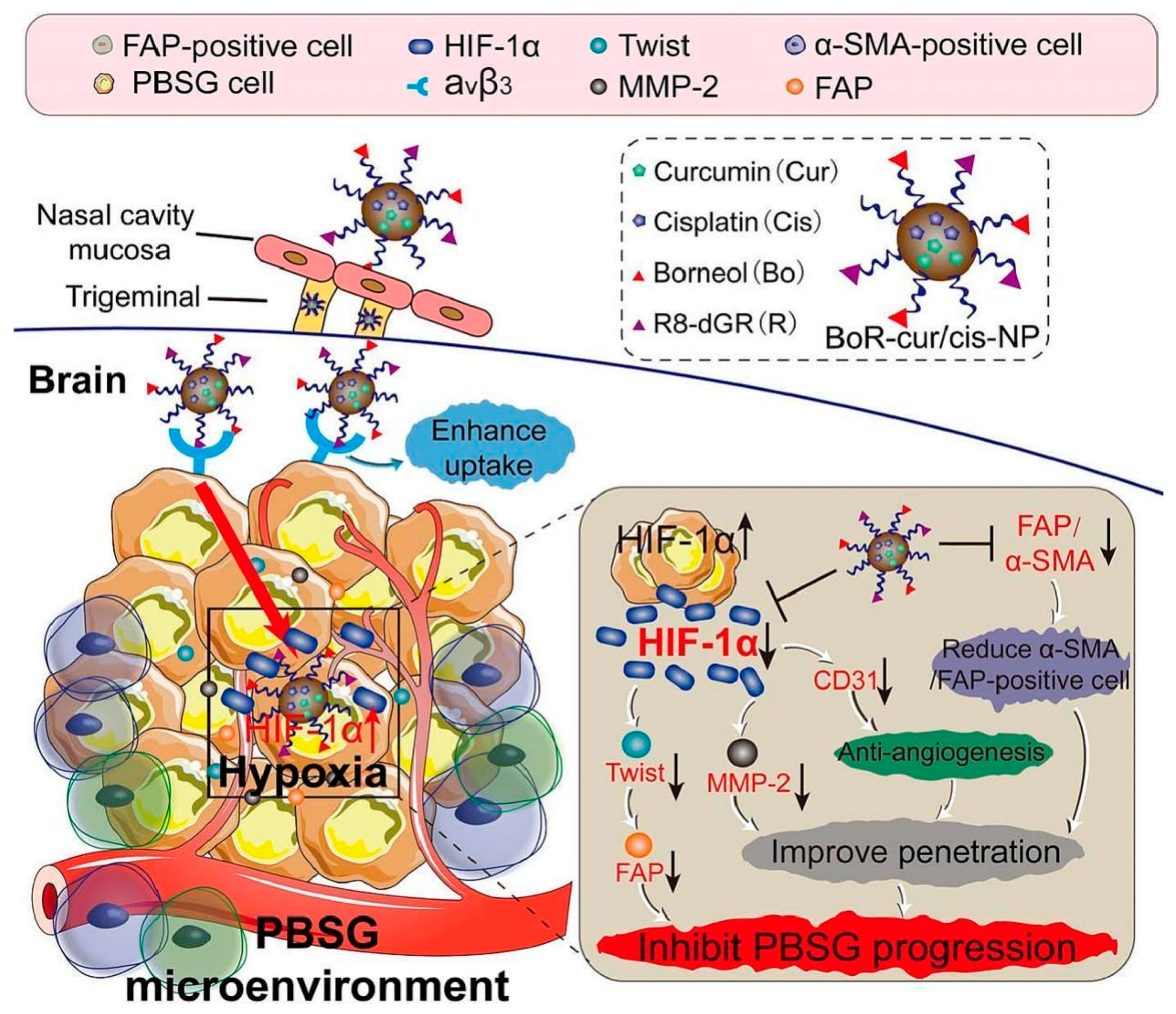
Therapeutic efficacy and safety are validated through extensive in vitro and in vivo testing. Tumor-targeting capabilities are assessed via tumor retention, survival analyses, and cancer cell inhibition assays [47,109,125]. For instance, Figure 7 demonstrates the intranasal delivery of Borneol/R8dGR peptide-modified PLGA nanoparticles co-loaded with curcumin and cisplatin for pediatric brainstem glioma treatment [109].
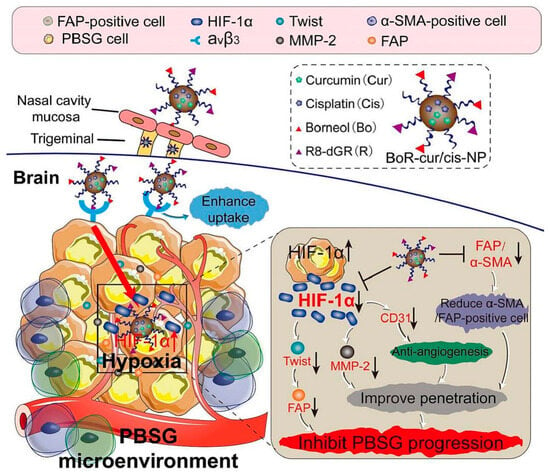
Figure 7.
Intranasal administration of borneol (Bo)/R8dGR peptide-modified PLGA-based nanoparticles (NP) co-loaded with curcumin and cisplatin (cur/cis). This nano-formulation improved the brain penetration via reduction of the expression of ZO-1 and occluding in nasal mucosa, while the R8dGR peptide modification improved the targeting of the NP by binding to integrin αvβ3 receptors and reduced hypoxia and angiogenesis in the PBSG microenvironment. Adopted with permission [109].
6.6. Applications in Regenerative Medicine
In regenerative medicine, evaluations include testing for bone and cartilage repair using 3D-printed scaffolds, monitoring osteogenic marker expression, and conducting in vivo mineralization studies [38,50,111]. Skin regeneration studies focus on epithelialization, angiogenesis, and the formation of granulation tissue [59,97]. Similarly, applications in nerve and muscle repair involve neurite alignment studies, Schwann cell behavior analysis, and myoblast differentiation assessments [52,88,103]. Neurotherapeutic applications include assessments of cognitive improvements and biochemical markers in Alzheimer’s disease models [5,8,48].
6.7. Antimicrobial Properties
Antimicrobial properties are demonstrated through tests for biofilm inhibition, reductions in bacterial burden, and antimicrobial peptide activity [21,55,128]. Sustained antifungal efficacy is also evaluated, including effects against Cryptococcus neoformans, showing potential for managing infections and promoting wound healing [20,30,123].
6.8. Advanced Functional Studies
Advanced studies investigate targeted delivery and tumor penetration. For example, integrin receptor targeting and hypoxia mitigation are evaluated for improved tumor-specific delivery [109,125]. Apoptosis pathways are analyzed using methods such as caspase-3 staining and mitochondrial function assays [7,119]. Neurotherapeutic studies address challenges like blood brain barrier (BBB) targeting and amyloid plaque reduction for Alzheimer’s disease [8,27,48].
6.9. Process Optimization
Scalable production methods are a key focus. Comparisons between microfluidic techniques and bulk production methods help evaluate efficiency [41,80]. Quality by Design (QbD) methodologies optimize critical process parameters, ensuring reproducibility and performance [42,78]. Statistical tools such as response surface methodology and precision testing further enhance formulation reliability [22,90,110].
Table 5 outlines the comprehensive evaluation metrics used to validate the safety, efficacy, and performance of peptide-loaded PLGA systems. These include physicochemical characterization, release kinetics, and in vivo assessments of biocompatibility and therapeutic efficacy. Patterns emphasize the integration of multidisciplinary testing, such as imaging techniques, immune response assays, and mechanical property evaluations. The table showcases a rigorous approach to ensuring product stability, optimized release, and targeted delivery, highlighting their potential in advancing therapies for cancer, tissue regeneration, and infectious diseases.

Table 5.
Evaluation metrics and methods for peptide-loaded PLGA nanocarriers.
7. Milestones and Multifunctional Capabilities of PLGA Nanocarriers
Peptide-loaded PLGA systems represent a significant advancement in sustained drug delivery, effectively reducing dosing frequency while enhancing therapeutic outcomes. These nanocarriers demonstrate long-term efficacy across a wide range of applications, including bone regeneration, infection treatment, and the management of chronic systemic diseases. For instance, prolonged drug retention has been observed in ischemic injury and Alzheimer’s disease models, highlighting their potential for addressing complex medical challenges [3,9,36,63,77,83]. Functionalized PLGA nanoparticles enhance targeting and selectivity, as seen with T7 peptide-functionalized micelles for glioblastoma and renal-targeting peptides for kidney-specific delivery, both of which improve therapeutic precision [2,25,61,62].
7.1. Versatile Applications
The versatility of peptide-loaded PLGA systems spans neural repair, autoimmune disease management, and infectious disease prevention. Their applications range from glioblastoma therapies to nanovaccines for zoonotic infections, demonstrating their broad applicability and effectiveness [15,30,53,56,104]. Targeted delivery minimizes systemic toxicity and enhances bioavailability, as observed in therapies like radionuclide treatment and thrombolysis, which reduce side effects such as renal toxicity and bleeding risk [12,16,132].
7.2. Innovative Delivery Platforms
Cutting-edge delivery platforms, such as composite hydrogels, supramolecular nanofibers, and hybrid scaffolds, are transforming drug delivery and tissue regeneration. These advanced materials improve drug retention and bioavailability in applications like mucosal delivery and bone repair [36,92,113]. Additionally, PLGA systems have become integral to vaccine development, enabling prolonged antigen release and eliciting robust immune responses in vaccines targeting multidrug-resistant infections, influenza, and cancer [24,30,31,66].
7.3. Clinical Translation
Peptide-loaded PLGA systems are progressing toward clinical translation, supported by predictive IVIVC models and successful outcomes in treatments for Alzheimer’s disease and glioblastoma. These advances underscore the systems’ readiness for broader therapeutic adoption [1,25,39]. Enhanced intracellular and endosomal delivery capabilities, facilitated by cell-penetrating peptide-functionalized nanoparticles, ensure effective gene delivery and sustained expression in pulmonary therapies [26,56,57].
7.4. Controlled Release and Antimicrobial Properties
Controlled release mechanisms ensure long-term effects while minimizing drug instability. Examples include tri-phasic release profiles for somatostatin analogs and steady peptide delivery in antifungal therapies, addressing diverse therapeutic needs [13,22,116,123]. The antimicrobial capabilities of these systems are particularly notable, with formulations targeting MRSA, bacterial keratitis, and periodontitis, as well as photothermal therapy providing synergistic effects [17,20,21,64].
7.5. Biocompatibility and Safety
The consistent demonstration of biocompatibility and safety makes peptide-loaded PLGA systems suitable for managing chronic diseases, preventing allergies, and advancing tissue regeneration [70,75,129]. In neurotherapeutics, these systems effectively cross the blood brain barrier, reducing amyloid-beta aggregation in Alzheimer’s disease therapies and suppressing tumor growth in glioblastoma [4,27,116].
7.6. Advances in Vaccine Development
PLGA-based platforms play a pivotal role in vaccine development, offering enhanced immune responses, multi-epitope formulations, and extended antigen release. Applications include cross-reactive influenza vaccines and mucosal immunity for diseases like swine dysentery, showcasing their impact on global health challenges [31,87,121].
7.7. Tissue Engineering and Regenerative Medicine
In regenerative medicine, peptide-functionalized PLGA scaffolds demonstrate significant potential. Functional peptides such as BMP-2 and RADA16-I improve outcomes in bone, cartilage, and nerve repair, as well as promote angiogenesis in ischemic models [18,60,91,114].
7.8. Cancer Therapy Innovations
Innovations in cancer therapy have led to improved outcomes by enhancing tumor-specific delivery while reducing systemic toxicity. For example, SP94-functionalized nanoparticles for hepatocellular carcinoma achieve selective tumor targeting and high therapeutic efficacy [34,46,119].
7.9. Scalable Manufacturing and Quality Control
Efficient manufacturing techniques, including microfluidics and co-axial electrospraying, enhance scalability and reproducibility. These approaches are complemented by accelerated in vitro release testing, which ensures robust quality control and supports widespread application [13,42,80].
Table 6 summarizes the proven benefits of peptide-loaded PLGA systems, including sustained drug release, enhanced stability, and multifunctionality. These systems address critical therapeutic challenges like peptide instability, inefficient targeting, and dosing frequency. Patterns emphasize their versatility across applications such as neurotherapeutics, oncology, and vaccine delivery. The table underscores PLGA’s role in enabling advanced diagnostics, immune response activation, and tissue engineering, paving the way for scalable, personalized medicine solutions.

Table 6.
Proven benefits of peptide-loaded PLGA products.
8. Overcoming Developmental and Translational Challenges in Peptide-Loaded PLGA Nanocarriers
Peptide-loaded PLGA systems face significant challenges in formulation design and stability. Peptide acylation and degradation in acidic microenvironments are persistent issues, with positively charged peptides posing additional challenges due to strong interactions with the negatively charged PLGA. Strategies such as calcium phosphate depots, hydrophobic ion-pairing, self-immolative-protecting groups, and dynamic surface coatings have shown promise, but universal solutions are still required to ensure stability across diverse physiological conditions [11,22,40,77,93,98,112,116].
To provide a comprehensive overview of these challenges, Table 7 summarizes the key applications of PLGA-based systems, highlighting their advantages and associated challenges across diverse fields such as cancer therapy, vaccine development, and tissue engineering. Insights include the potential of PLGA systems for targeted delivery, improved stability, and sustained release while addressing hurdles like scalability, cost, and variability in therapeutic outcomes. The table provides a comprehensive overview to guide strategic improvements in the formulation, functionalization, and clinical translation of PLGA-based technologies.

Table 7.
Comparative analysis of applications, advantages, and challenges of PLGA-based systems.
Manufacturing Scalability, Cost, and Regulatory Barriers: Implications for Clinical Use
The challenges associated with manufacturing scalability, cost, and regulatory hurdles critically impact the clinical translation of peptide-loaded PLGA nanocarriers. Large-scale production requires high reproducibility, cost-effectiveness, and compliance with stringent regulatory standards, which are often difficult to achieve with current methodologies.
Manufacturing Scalability: Techniques such as 3D printing, electrospinning, and microfluidic technologies, while promising in controlled laboratory settings, struggle with scalability for industrial production [37,40,41,51,69,80,86,91]. The precision required for incorporating multifunctional elements like targeting ligands or dual-drug systems complicates automation and increases production time. For example, microfluidic methods require costly modifications to achieve the throughput needed for large-scale applications, which hinders the transition from laboratory prototypes to market-ready products.
Cost Constraints: Advanced materials and processes used in peptide-loaded PLGA systems, such as PEGylation, gold nanoparticle incorporation, and hydrophobic ion-pairing, significantly increase production costs [3,20,59,108,115]. While these components enhance therapeutic outcomes, their high costs make large-scale production economically prohibitive. Furthermore, resource-intensive strategies, such as T-cell membrane-coated nanoparticles, further inflate costs, limiting accessibility and affordability for widespread clinical use.
Regulatory Barriers: The regulatory requirements for complex peptide-loaded PLGA systems present significant obstacles [7,8,27,28,38,47,109,118]. Each added functionality, such as targeting ligands or dual-drug formulations, necessitates rigorous safety and efficacy evaluations. Regulatory approval processes demand consistent product quality and compliance with Good Manufacturing Practice (GMP) standards. However, the inherent variability in PLGA systems due to batch-to-batch differences in polymer characteristics complicates reproducibility and standardization, further slowing clinical translation.
Clinical Impact: These challenges collectively limit the deployment of peptide-loaded PLGA nanocarriers in clinical settings. Despite demonstrating significant promise in preclinical studies, such as sustained drug release and enhanced targeting for cancer therapy and neurodegenerative diseases, many systems fail to transition to clinical trials [3,27,62,74,106,107]. The inability to achieve large-scale, cost-effective production undermines the feasibility of widespread application, while regulatory delays and high costs reduce the commercial appeal of these technologies.
9. Next Generation of Peptide-Loaded PLGA Systems
Future developments for peptide-loaded PLGA systems focus on enhancing customization and scalability, addressing challenges in mass production while maintaining precision. Techniques such as 3D printing and microfluidics must evolve to support the synthesis of complex systems, including dual-drug hydrogels and composite scaffolds. Incorporating Quality by Design (QbD) principles will help ensure consistent production and regulatory compliance, making these systems more accessible [42,69,80,100].
Integrating multifunctional systems capable of therapeutic, diagnostic, and regenerative applications is a key direction. These platforms will combine properties such as antimicrobial, osteogenic, and angiogenic functions, which are critical for wound healing and tissue engineering. Stimuli-responsive components, such as light-activated nanoparticles, offer promising applications in precision oncology [23,68,91,125].
9.1. Patient-Specific Applications and Personalized Medicine
Emerging trends in personalized medicine highlight the need for peptide-loaded PLGA systems tailored to individual patient profiles. These systems can be customized to align with specific disease pathologies, genetic markers, or immune responses. For example, patient-derived biomolecules could be integrated into PLGA scaffolds, ensuring compatibility and enhancing therapeutic outcomes in tissue engineering [25,51,59,91,111].
In oncology, personalized nanoparticles targeting specific tumor markers, such as HER2-positive breast cancers or EGFR-overexpressing glioblastomas, demonstrate potential for precision drug delivery. Ligand-functionalized PLGA systems tailored to these markers can improve therapeutic outcomes and reduce off-target effects [28,34,61,84]. Similarly, for neurodegenerative disorders like Alzheimer’s and Parkinson’s, tailored brain-targeting platforms incorporating disease-specific ligands could enhance drug distribution across the blood/brain barrier, accommodating variability across patient populations [29,106,129].
Personalized vaccine platforms also represent a transformative application of peptide-loaded PLGA systems. Multi-epitope nanoparticle systems, developed using tumor-specific neoantigens, offer the potential for individualized cancer immunotherapies. Self-adjuvant designs enhance immune activation, providing significant benefits in vaccine efficacy for infectious diseases and emerging zoonotic pathogens [7,30,31,32,118].
In regenerative medicine, patient-specific bioengineered scaffolds incorporating individualized growth factors or extracellular matrix components could offer customized solutions for complex tissue defects. Applications include scaffolds for bone regeneration infused with patient-specific osteoinductive molecules or neural repair systems optimized for the localized delivery of neuroprotective peptides [19,91,111].
9.2. Enhancing Precision and Stability
Advancements in targeting mechanisms and peptide stabilization techniques further support patient-specific applications. Ligands designed to target renal, endothelial, or bone tissues can be tailored to the unique physiological conditions of individual patients, enhancing precision and therapeutic efficacy [29,34,61,84]. Stabilization strategies, including hydrophobic ion-pairing and calcium phosphate depots, address the variability in peptide degradation and clearance, ensuring consistent therapeutic effects across diverse populations [11,22,93,98].
9.3. Clinical Translation and Future Directions
Transitioning these technologies to clinical practice will require extensive validation in patient-specific contexts. Clinical trials targeting diseases such as glioblastoma and Alzheimer’s, as well as chronic wounds must incorporate diverse patient populations to evaluate efficacy and safety comprehensively [5,25,27,115,125]. For example, personalized treatments for glioblastoma can leverage tumor-specific biomarkers to refine targeting and improve therapeutic responses.
Incorporating artificial intelligence and machine learning may further advance patient-specific applications by optimizing nanoparticle design and predicting release profiles based on individual patient data. These innovations will enable peptide-loaded PLGA systems to align with the growing emphasis on precision medicine, offering highly tailored therapeutic solutions [2,86,118].
10. Conclusions
Peptide-loaded PLGA nanocarriers have emerged as pivotal tools in addressing the challenges associated with peptide therapeutics, offering transformative potential in various biomedical applications. Their inherent biodegradability, biocompatibility, and capacity for controlled and targeted peptide delivery make them highly effective for addressing therapeutic needs in areas such as cancer treatment, neurodegenerative disorders, and infectious diseases. Advanced surface functionalization and hybrid polymer designs have further expanded their applications to regenerative medicine and vaccine development, enhancing immune responses and promoting tissue repair.
Despite these advances, significant hurdles remain, particularly in scalability, cost-effectiveness, and achieving regulatory approval. Future research should focus on specific areas within polymer chemistry to address these challenges effectively. For instance, the development of novel copolymer systems could improve the mechanical stability, degradation rates, and drug-release profiles of PLGA carriers, enabling more reliable therapeutic delivery. Refining surface modification strategies, such as grafting ligands or functional groups onto PLGA surfaces, may enhance cellular targeting and bioavailability, while responsive polymer chemistry could facilitate site-specific peptide release through stimuli-responsive designs. Additionally, investigating advanced polymer architectures, such as block polymers or star-shaped polymers, could further enhance control over degradation and release kinetics. Emphasis on green chemistry approaches in polymer synthesis would also align innovation with sustainability goals, addressing environmental concerns during material development.
In a clinical context, these advancements could directly support the development of cancer immunotherapies, peptide-based treatments for neurodegenerative conditions, and targeted vaccine platforms for infectious diseases. Collaboration across disciplines, including nanotechnology, pharmaceutical engineering, and clinical sciences, will be essential to optimize production processes, enhance scalability, and expedite regulatory pathways. By addressing these focused areas, peptide-loaded PLGA nanocarriers hold the potential to drive significant progress in therapeutic delivery and play a central role in the evolution of personalized medicine.
Author Contributions
The authors confirm contribution to the paper as follows: Conceptualization, writing, review and editing, H.O.; Investigation, review and editing, R.L.W. and A.M.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments
The authors partly used an OpenAI Large-Scale Language Model to maximize accuracy, clarity, and organization.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Andhariya, J.V.; Jog, R.; Shen, J.; Choi, S.; Wang, Y.; Zou, Y.; Burgess, D.J. Development of Level A in vitro-in vivo correlations for peptide loaded PLGA microspheres. J. Control. Release 2019, 308, 1–13. [Google Scholar] [CrossRef]
- Bhowmik, A.; Chakravarti, S.; Ghosh, A.; Shaw, R.; Bhandary, S.; Bhattacharyya, S.; Sen, P.C.; Ghosh, M.K. Anti-SSTR2 peptide based targeted delivery of potent PLGA encapsulated 3,3′-diindolylmethane nanoparticles through blood brain barrier prevents glioma progression. Oncotarget 2017, 8, 65339–65358. [Google Scholar] [CrossRef] [PubMed]
- Fan, S.; Zheng, Y.; Liu, X.; Fang, W.; Chen, X.; Liao, W.; Jing, X.; Lei, M.; Tao, E.; Ma, Q.; et al. Curcumin-loaded PLGA-PEG nanoparticles conjugated with B6 peptide for potential use in Alzheimer’s disease. Drug Deliv. 2018, 25, 1091–1102. [Google Scholar] [CrossRef]
- Huang, N.; Lu, S.; Liu, X.G.; Zhu, J.; Wang, Y.J.; Liu, R.T. PLGA nanoparticles modified with a BBB-penetrating peptide co-delivering Abeta generation inhibitor and curcumin attenuate memory deficits and neuropathology in Alzheimer’s disease mice. Oncotarget 2017, 8, 81001–81013. [Google Scholar] [CrossRef] [PubMed]
- Mathew, A.; Fukuda, T.; Nagaoka, Y.; Hasumura, T.; Morimoto, H.; Yoshida, Y.; Maekawa, T.; Venugopal, K.; Kumar, D.S. Curcumin loaded-PLGA nanoparticles conjugated with Tet-1 peptide for potential use in Alzheimer’s disease. PLoS ONE 2012, 7, e32616. [Google Scholar] [CrossRef] [PubMed]
- Panda, P.K.; Jain, S.K. Doxorubicin bearing peptide anchored PEGylated PLGA nanoparticles for the effective delivery to prostate cancer cells. J. Drug Deliv. Sci. Technol. 2023, 86, 21. [Google Scholar] [CrossRef]
- Priwitaningrum, D.L.; Jentsch, J.; Bansal, R.; Rahimian, S.; Storm, G.; Hennink, W.E.; Prakash, J. Apoptosis-inducing peptide loaded in PLGA nanoparticles induces anti-tumor effects in vivo. Int. J. Pharm. 2020, 585, 119535. [Google Scholar] [CrossRef] [PubMed]
- Saleh, S.R.; Abd-Elmegied, A.; Aly Madhy, S.; Khattab, S.N.; Sheta, E.; Elnozahy, F.Y.; Mehanna, R.A.; Ghareeb, D.A.; Abd-Elmonem, N.M. Brain-targeted Tet-1 peptide-PLGA nanoparticles for berberine delivery against STZ-induced Alzheimer’s disease in a rat model: Alleviation of hippocampal synaptic dysfunction, Tau pathology, and amyloidogenesis. Int. J. Pharm. 2024, 658, 124218. [Google Scholar] [CrossRef]
- Ali, M.; van Gent, M.E.; de Waal, A.M.; van Doodewaerd, B.R.; Bos, E.; Koning, R.I.; Cordfunke, R.A.; Drijfhout, J.W.; Nibbering, P.H. Physical and Functional Characterization of PLGA Nanoparticles Containing the Antimicrobial Peptide SAAP-148. Int. J. Mol. Sci. 2023, 24, 2867. [Google Scholar] [CrossRef] [PubMed]
- Encinas-Basurto, D.; Konhilas, J.P.; Polt, R.; Hay, M.; Mansour, H.M. Glycosylated Ang-(1-7) MasR Agonist Peptide Poly Lactic-co-Glycolic Acid (PLGA) Nanoparticles and Microparticles in Cognitive Impairment: Design, Particle Preparation, Physicochemical Characterization, and In Vitro Release. Pharmaceutics 2022, 14, 587. [Google Scholar] [CrossRef]
- Liu, J.; Xu, Y.; Liu, Z.; Ren, H.; Meng, Z.; Liu, K.; Liu, Z.; Yong, J.; Wang, Y.; Li, X. A modified hydrophobic ion-pairing complex strategy for long-term peptide delivery with high drug encapsulation and reduced burst release from PLGA microspheres. Eur. J. Pharm. Biopharm. 2019, 144, 217–229. [Google Scholar] [CrossRef]
- Arora, G.; Shukla, J.; Ghosh, S.; Maulik, S.K.; Malhotra, A.; Bandopadhyaya, G. PLGA nanoparticles for peptide receptor radionuclide therapy of neuroendocrine tumors: A novel approach towards reduction of renal radiation dose. PLoS ONE 2012, 7, e34019. [Google Scholar] [CrossRef]
- Goel, M.; Leung, D.; Famili, A.; Chang, D.; Nayak, P.; Al-Sayah, M. Accelerated in vitro release testing method for a long-acting peptide-PLGA formulation. Eur. J. Pharm. Biopharm. 2021, 165, 185–192. [Google Scholar] [CrossRef]
- Tomic, I.; Vidis-Millward, A.; Mueller-Zsigmondy, M.; Cardot, J.M. Setting accelerated dissolution test for PLGA microspheres containing peptide, investigation of critical parameters affecting drug release rate and mechanism. Int. J. Pharm. 2016, 505, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Cappellano, G.; Woldetsadik, A.D.; Orilieri, E.; Shivakumar, Y.; Rizzi, M.; Carniato, F.; Gigliotti, C.L.; Boggio, E.; Clemente, N.; Comi, C.; et al. Subcutaneous inverse vaccination with PLGA particles loaded with a MOG peptide and IL-10 decreases the severity of experimental autoimmune encephalomyelitis. Vaccine 2014, 32, 5681–5689. [Google Scholar] [CrossRef]
- Castro, P.M.; Baptista, P.; Madureira, A.R.; Sarmento, B.; Pintado, M.E. Combination of PLGA nanoparticles with mucoadhesive guar-gum films for buccal delivery of antihypertensive peptide. Int. J. Pharm. 2018, 547, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Jiao, X.; Dong, X.; Shan, H.; Qin, Z. Assessing the Efficacy of PLGA-Loaded Antimicrobial Peptide OH-CATH30 Microspheres for the Treatment of Bacterial Keratitis: A Promising Approach. Biomolecules 2023, 13, 1244. [Google Scholar] [CrossRef]
- Lin, Z.Y.; Duan, Z.X.; Guo, X.D.; Li, J.F.; Lu, H.W.; Zheng, Q.X.; Quan, D.P.; Yang, S.H. Bone induction by biomimetic PLGA-(PEG-ASP)n copolymer loaded with a novel synthetic BMP-2-related peptide in vitro and in vivo. J. Control. Release 2010, 144, 190–195. [Google Scholar] [CrossRef]
- Wu, G.; Cao, Z.Z.; Luo, X.L.; Wang, X.X.; Wang, S.H.; Wang, D.L. Fabrication and Characterization of a PDLSCs/BMP-2-PLGA-NP/RADA Peptide Hydrogel Composite for Bone Repair. J. Biomater. Tissue Eng. 2017, 7, 379–385. [Google Scholar] [CrossRef]
- Guo, M.; Ruan, M.; Wu, J.; Ye, J.; Wang, C.; Guo, Z.; Chen, W.; Wang, L.; Wu, K.; Du, S.; et al. Poly-tannic acid coated PLGA nanoparticle decorated with antimicrobial peptide for synergistic bacteria treatment and infectious wound healing promotion. Colloids Surf. B Biointerfaces 2025, 245, 114217. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Sequeda, N.; Ruiz, J.; Ortiz, C.; Urquiza, M.; Torres, R. Potent and Specific Antibacterial Activity against Escherichia coli O157:H7 and Methicillin Resistant Staphylococcus aureus (MRSA) of G17 and G19 Peptides Encapsulated into Poly-Lactic-Co-Glycolic Acid (PLGA) Nanoparticles. Antibiotics 2020, 9, 384. [Google Scholar] [CrossRef] [PubMed]
- Tomic, I.; Mueller-Zsigmondy, M.; Vidis-Millward, A.; Cardot, J.M. In vivo release of peptide-loaded PLGA microspheres assessed through deconvolution coupled with mechanistic approach. Eur. J. Pharm. Biopharm. 2018, 125, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Chereddy, K.K.; Her, C.H.; Comune, M.; Moia, C.; Lopes, A.; Porporato, P.E.; Vanacker, J.; Lam, M.C.; Steinstraesser, L.; Sonveaux, P.; et al. PLGA nanoparticles loaded with host defense peptide LL37 promote wound healing. J. Control. Release 2014, 194, 138–147. [Google Scholar] [CrossRef]
- Buyukbayraktar, H.K.; Pelit Arayici, P.; Ihlamur, M.; Gokkaya, D.; Karahan, M.; Abamor, E.S.; Topuzogullari, M. Effect of polycation coating on the long-term pulsatile release of antigenic ESAT-6(1-20) peptide from PLGA nanoparticles. Colloids Surf. B Biointerfaces 2023, 228, 113421. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Liu, L.; Lu, Y.; Sun, T.; Shen, C.; Chen, X.; Chen, Q.; An, S.; He, X.; Ruan, C.; et al. T7 Peptide-Functionalized PEG-PLGA Micelles Loaded with Carmustine for Targeting Therapy of Glioma. ACS Appl. Mater. Interfaces 2016, 8, 27465–27473. [Google Scholar] [CrossRef]
- Feiner-Gracia, N.; Dols-Perez, A.; Royo, M.; Solans, C.; Garcia-Celma, M.J.; Fornaguera, C. Cell penetrating peptide grafting of PLGA nanoparticles to enhance cell uptake. Eur. Polym. J. 2018, 108, 429–438. [Google Scholar] [CrossRef]
- Khairnar, B.D.; Padhye, A.; Madiwal, V.; Jha, A.; Jadhav, S.H.; Rajwade, J.M. Cyclic ß-hairpin peptide loaded PLGA nanoparticles: A potential anti-amyloid therapeutic. Mater. Today Commun. 2023, 35, 11. [Google Scholar] [CrossRef]
- Vijayan, A.N.; Indrakumar, J.; Gomathinayagam, S.; Gothandam, K.M.; Korrapati, P.S. Bi-Functional Aspects of Peptide Decorated PLGA Nanocarriers for Enhanced Translocation Across the Blood-Brain Barrier through Macropinocytosis. Macromol. Res. 2022, 30, 557–570. [Google Scholar] [CrossRef]
- Yan, L.; Wang, H.Y.; Jiang, Y.F.; Liu, J.H.; Wang, Z.; Yang, Y.X.; Huang, S.W.; Huang, Y.Z. Cell-Penetrating Peptide-Modified PLGA Nanoparticles for Enhanced Nose-to-Brain Macromolecular Delivery. Macromol. Res. 2013, 21, 435–441. [Google Scholar] [CrossRef]
- Du, X.; Xue, J.; Jiang, M.; Lin, S.; Huang, Y.; Deng, K.; Shu, L.; Xu, H.; Li, Z.; Yao, J.; et al. A Multiepitope Peptide, rOmp22, Encapsulated in Chitosan-PLGA Nanoparticles as a Candidate Vaccine Against Acinetobacter baumannii Infection. Int. J. Nanomed. 2021, 16, 1819–1836. [Google Scholar] [CrossRef]
- Heng, W.T.; Lim, H.X.; Tan, K.O.; Poh, C.L. Validation of Multi-epitope Peptides Encapsulated in PLGA Nanoparticles Against Influenza A Virus. Pharm. Res. 2023, 40, 1999–2025. [Google Scholar] [CrossRef]
- Roozbehani, M.; Falak, R.; Mohammadi, M.; Hemphill, A.; Razmjou, E.; Meamar, A.R.; Masoori, L.; Khoshmirsafa, M.; Moradi, M.; Gharavi, M.J. Characterization of a multi-epitope peptide with selective MHC-binding capabilities encapsulated in PLGA nanoparticles as a novel vaccine candidate against Toxoplasma gondii infection. Vaccine 2018, 36, 6124–6132. [Google Scholar] [CrossRef] [PubMed]
- Varypataki, E.M.; Silva, A.L.; Barnier-Quer, C.; Collin, N.; Ossendorp, F.; Jiskoot, W. Synthetic long peptide-based vaccine formulations for induction of cell mediated immunity: A comparative study of cationic liposomes and PLGA nanoparticles. J. Control. Release 2016, 226, 98–106. [Google Scholar] [CrossRef]
- Nie, X.; Liu, Y.; Li, M.; Yu, X.; Yuan, W.; Huang, S.; Ren, D.; Wang, Y.; Wang, Y. SP94 Peptide-Functionalized PEG-PLGA Nanoparticle Loading with Cryptotanshinone for Targeting Therapy of Hepatocellular Carcinoma. AAPS PharmSciTech 2020, 21, 124. [Google Scholar] [CrossRef]
- Paulino da Silva Filho, O.; Ali, M.; Nabbefeld, R.; Primavessy, D.; Bovee-Geurts, P.H.; Grimm, S.; Kirchner, A.; Wiesmuller, K.H.; Schneider, M.; Walboomers, X.F.; et al. A comparison of acyl-moieties for noncovalent functionalization of PLGA and PEG-PLGA nanoparticles with a cell-penetrating peptide. RSC Adv. 2021, 11, 36116–36124. [Google Scholar] [CrossRef]
- Cai, Q.; Qiao, C.; Ning, J.; Ding, X.; Wang, H.; Zhou, Y. A Polysaccharide-based Hydrogel and PLGA Microspheres for Sustained P24 Peptide Delivery: An In vitro and In vivo Study Based on Osteogenic Capability. Chem. Res. Chin. Univ. 2019, 35, 908–915. [Google Scholar] [CrossRef]
- Shafiq, M.; Yuan, Z.; Rafique, M.; Aishima, S.; Jing, H.; Yuqing, L.; Ijima, H.; Jiang, S.; Mo, X. Combined effect of SDF-1 peptide and angiogenic cues in co-axial PLGA/gelatin fibers for cutaneous wound healing in diabetic rats. Colloids Surf. B Biointerfaces 2023, 223, 113140. [Google Scholar] [CrossRef]
- Wang, X.; Chen, W.; Chen, Z.; Li, Y.; Wu, K.; Song, Y. Preparation of 3D Printing PLGA Scaffold with BMP-9 and P-15 Peptide Hydrogel and Its Application in the Treatment of Bone Defects in Rabbits. Contrast Media Mol. Imaging 2022, 2022, 1081957. [Google Scholar] [CrossRef]
- Chen, J.; Li, S.; Shen, Q. Folic acid and cell-penetrating peptide conjugated PLGA-PEG bifunctional nanoparticles for vincristine sulfate delivery. Eur. J. Pharm. Sci. 2012, 47, 430–443. [Google Scholar] [CrossRef]
- Schlosser, C.S.; Morris, C.J.; Brocchini, S.; Williams, G.R. Hydrophobic ion pairing as a novel approach to co-axial electrospraying of peptide-PLGA particles. Int. J. Pharm. 2024, 667, 124885. [Google Scholar] [CrossRef]
- Streck, S.; Neumann, H.; Nielsen, H.M.; Rades, T.; McDowell, A. Comparison of bulk and microfluidics methods for the formulation of poly-lactic-co-glycolic acid (PLGA) nanoparticles modified with cell-penetrating peptides of different architectures. Int. J. Pharm. X 2019, 1, 100030. [Google Scholar] [CrossRef]
- Zhang, C.; Yang, L.; Wan, F.; Bera, H.; Cun, D.; Rantanen, J.; Yang, M. Quality by design thinking in the development of long-acting injectable PLGA/PLA-based microspheres for peptide and protein drug delivery. Int. J. Pharm. 2020, 585, 119441. [Google Scholar] [CrossRef] [PubMed]
- Balmert, S.C.; Zmolek, A.C.; Glowacki, A.J.; Knab, T.D.; Rothstein, S.N.; Wokpetah, J.M.; Fedorchak, M.V.; Little, S.R. Positive Charge of “Sticky” Peptides and Proteins Impedes Release From Negatively Charged PLGA Matrices. J. Mater. Chem. B 2015, 3, 4723–4734. [Google Scholar] [CrossRef] [PubMed]
- Xiao, P.; Qi, P.; Chen, J.; Song, Z.; Wang, Y.; He, H.; Tang, X.; Wang, P. The effect of polymer blends on initial release regulation and in vitro-in vivo relationship of peptides loaded PLGA-Hydrogel Microspheres. Int. J. Pharm. 2020, 591, 119964. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.L.; Rosalia, R.A.; Sazak, A.; Carstens, M.G.; Ossendorp, F.; Oostendorp, J.; Jiskoot, W. Optimization of encapsulation of a synthetic long peptide in PLGA nanoparticles: Low-burst release is crucial for efficient CD8(+) T cell activation. Eur. J. Pharm. Biopharm. 2013, 83, 338–345. [Google Scholar] [CrossRef]
- Zhang, N.; Li, J.; Gao, W.; Zhu, W.; Yan, J.; He, Z.; Li, L.; Wu, F.; Pu, Y.; He, B. Co-Delivery of Doxorubicin and Anti-PD-L1 Peptide in Lipid/PLGA Nanocomplexes for the Chemo-Immunotherapy of Cancer. Mol. Pharm. 2022, 19, 3439–3449. [Google Scholar] [CrossRef]
- Yaman, S.; Ramachandramoorthy, H.; Oter, G.; Zhukova, D.; Nguyen, T.; Sabnani, M.K.; Weidanz, J.A.; Nguyen, K.T. Melanoma Peptide MHC Specific TCR Expressing T-Cell Membrane Camouflaged PLGA Nanoparticles for Treatment of Melanoma Skin Cancer. Front. Bioeng. Biotechnol. 2020, 8, 943. [Google Scholar] [CrossRef] [PubMed]
- Sathya, S.; Shanmuganathan, B.; Saranya, S.; Vaidevi, S.; Ruckmani, K.; Pandima Devi, K. Phytol-loaded PLGA nanoparticle as a modulator of Alzheimer’s toxic Abeta peptide aggregation and fibrillation associated with impaired neuronal cell function. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1719–1730. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, C.Y.; He, P.; Fu, L.; Zhou, Y.M.; Chen, X.S. Preparation and Bioactivities of PLGA/Nano-hydroxyapatite Scaffold Containing Chitosan Microspheres for Controlled Delivery of Mutifuncational Peptide-adrenomedullin. Chem. J. Chin. Univ. 2011, 32, 1622–1628. [Google Scholar]
- Zhang, S.B.; Zhang, Z.; Yu, M.N.; Liu, T.B.; Wang, J.J.; Cai, Q.; Chen, L.; He, C.L.; Meng, W.Y.; Chen, X.S. Biodegradable PLGA Microspheres for Controlled Delivery of Parathyroid Hormone Related Peptide. Acta Polym. Sin. 2014, 2, 270–275. [Google Scholar] [CrossRef]
- Nune, M.; Krishnan, U.M.; Sethuraman, S. Decoration of PLGA electrospun nanofibers with designer self-assembling peptides: A “Nano-on-Nano” concept. RSC Adv. 2015, 5, 88748–88757. [Google Scholar] [CrossRef]
- Nune, M.; Krishnan, U.M.; Sethuraman, S. PLGA nanofibers blended with designer self-assembling peptides for peripheral neural regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 62, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Derman, S.; Mustafaeva, Z.A.; Abamor, E.S.; Bagirova, M.; Allahverdiyev, A. Preparation, characterization and immunological evaluation: Canine parvovirus synthetic peptide loaded PLGA nanoparticles. J. Biomed. Sci. 2015, 22, 89. [Google Scholar] [CrossRef] [PubMed]
- Ucar, B.; Acar, T.; Arayici, P.P.; Derman, S. A nanotechnological approach in the current therapy of COVID-19: Model drug oseltamivir-phosphate loaded PLGA nanoparticles targeted with spike protein binder peptide of SARS-CoV-2. Nanotechnology 2021, 32, 485601. [Google Scholar] [CrossRef]
- Ramoa, A.M.; Campos, F.; Moreira, L.; Teixeira, C.; Leiro, V.; Gomes, P.; das Neves, J.; Martins, M.C.L.; Monteiro, C. Antimicrobial peptide-grafted PLGA-PEG nanoparticles to fight bacterial wound infections. Biomater. Sci. 2023, 11, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Gomes Dos Reis, L.; Lee, W.H.; Svolos, M.; Moir, L.M.; Jaber, R.; Windhab, N.; Young, P.M.; Traini, D. Nanotoxicologic Effects of PLGA Nanoparticles Formulated with a Cell-Penetrating Peptide: Searching for a Safe pDNA Delivery System for the Lungs. Pharmaceutics 2019, 11, 12. [Google Scholar] [CrossRef]
- Gomes Dos Reis, L.; Lee, W.H.; Svolos, M.; Moir, L.M.; Jaber, R.; Engel, A.; Windhab, N.; Young, P.M.; Traini, D. Delivery of pDNA to lung epithelial cells using PLGA nanoparticles formulated with a cell-penetrating peptide: Understanding the intracellular fate. Drug Dev. Ind. Pharm. 2020, 46, 427–442. [Google Scholar] [CrossRef]
- Malik, S.; Slack, F.J.; Bahal, R. Formulation of PLGA nanoparticles containing short cationic peptide nucleic acids. Methodsx 2020, 7, 101115. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Zhou, S.C.; Zhang, Y.Z.; Wu, D.K.; Yang, X.Y. The dual delivery of growth factors and antimicrobial peptide by PLGA/GO composite biofilms to promote skin-wound healing. New J. Chem. 2020, 44, 1463–1476. [Google Scholar] [CrossRef]
- Liu, Z.H.; Tian, G.J.; Liu, L.N.; Li, Y.M.; Xu, S.D.; Du, Y.Q.; Li, M.T.; Jing, W.; Wei, P.F.; Zhao, B.; et al. A 3D-printed PLGA/HA composite scaffold modified with fusion peptides to enhance its antibacterial, osteogenic and angiogenic properties in bone defect repair. J. Mater. Res. Technol. 2024, 30, 5804–5819. [Google Scholar] [CrossRef]
- He, J.; Chen, H.; Zhou, W.; Chen, M.; Yao, Y.; Zhang, Z.; Tan, N. Kidney targeted delivery of asiatic acid using a FITC labeled renal tubular-targeting peptide modified PLGA-PEG system. Int. J. Pharm. 2020, 584, 119455. [Google Scholar] [CrossRef]
- Cui, Y.; Sun, J.; Hao, W.; Chen, M.; Wang, Y.; Xu, F.; Gao, C. Dual-Target Peptide-Modified Erythrocyte Membrane-Enveloped PLGA Nanoparticles for the Treatment of Glioma. Front. Oncol. 2020, 10, 563938. [Google Scholar] [CrossRef]
- Cheng, Y.; Qin, J.; Huang, Y.; Wang, T. The antimicrobial effects of PLGA microspheres containing the antimicrobial peptide OP-145 on clinically isolated pathogens in bone infections. Sci. Rep. 2022, 12, 14541. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Na, R.; Wang, X.; Liu, H.; Zhao, L.; Sun, X.; Ma, G.; Cui, F. Fabrication of Antimicrobial Peptide-Loaded PLGA/Chitosan Composite Microspheres for Long-Acting Bacterial Resistance. Molecules 2017, 22, 1637. [Google Scholar] [CrossRef]
- Çalman, F.; Pelit Arayıcı, P.; Büyükbayraktar, H.K.; Karahan, M.; Mustafaeva, Z.; Katsarava, R. Development of Vaccine Prototype Against Zika Virus Disease of Peptide-Loaded PLGA Nanoparticles and Evaluation of Cytotoxicity. Int. J. Pept. Res. Ther. 2018, 25, 1057–1063. [Google Scholar] [CrossRef]
- Dolen, Y.; Gileadi, U.; Chen, J.L.; Valente, M.; Creemers, J.H.A.; Van Dinther, E.A.W.; van Riessen, N.K.; Jager, E.; Hruby, M.; Cerundolo, V.; et al. PLGA Nanoparticles Co-encapsulating NY-ESO-1 Peptides and IMM60 Induce Robust CD8 and CD4 T Cell and B Cell Responses. Front. Immunol. 2021, 12, 641703. [Google Scholar] [CrossRef]
- Herrmann, V.L.; Wieland, D.E.; Legler, D.F.; Wittmann, V.; Groettrup, M. The STEAP1(262-270) peptide encapsulated into PLGA microspheres elicits strong cytotoxic T cell immunity in HLA-A*0201 transgenic mice—A new approach to immunotherapy against prostate carcinoma. Prostate 2016, 76, 456–468. [Google Scholar] [CrossRef]
- Clement, S.; Anwer, A.G.; Pires, L.; Campbell, J.; Wilson, B.C.; Goldys, E.M. Radiodynamic Therapy Using TAT Peptide-Targeted Verteporfin-Encapsulated PLGA Nanoparticles. Int. J. Mol. Sci. 2021, 22, 6425. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Shao, L.; Wang, F.; Huang, Y.; Gao, F. Enhancement in sustained release of antimicrobial peptide and BMP-2 from degradable three dimensional-printed PLGA scaffold for bone regeneration. RSC Adv. 2019, 9, 10494–10507. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Thijssen, S.; Hennink, W.E.; Garssen, J.; van Nostrum, C.F.; Willemsen, L.E.M. Oral pretreatment with beta-lactoglobulin derived peptide and CpG co-encapsulated in PLGA nanoparticles prior to sensitizations attenuates cow’s milk allergy development in mice. Front. Immunol. 2022, 13, 1053107. [Google Scholar] [CrossRef]
- Hu, F.F.; Qi, J.P.; Lu, Y.; He, H.S.; Wu, W. PLGA-based implants for sustained delivery of peptides/proteins: Current status, challenge and perspectives. Chin. Chem. Lett. 2023, 34, 11. [Google Scholar] [CrossRef]
- Liu, M.; Thijssen, S.; van Nostrum, C.F.; Hennink, W.E.; Garssen, J.; Willemsen, L.E.M. Inhibition of cow’s milk allergy development in mice by oral delivery of beta-lactoglobulin-derived peptides loaded PLGA nanoparticles is associated with systemic whey-specific immune silencing. Clin. Exp. Allergy 2022, 52, 137–148. [Google Scholar] [CrossRef]
- Choi, D.H.; Park, Y.S. Arginine-rich Peptide Coated PLGA Nanoparticles Enhance Polymeric Delivery of Antisense HIF1α-oligonucleotide to Fully Differentiated Stiff Adipocytes. Toxicol. Environ. Health Sci. 2019, 11, 1–10. [Google Scholar] [CrossRef]
- Liu, C.; Xie, Y.; Li, X.; Yao, X.; Wang, X.; Wang, M.; Li, Z.; Cao, F. Folic Acid/Peptides Modified PLGA-PEI-PEG Polymeric Vectors as Efficient Gene Delivery Vehicles: Synthesis, Characterization and Their Biological Performance. Mol. Biotechnol. 2021, 63, 63–79. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Pizarro, R.; Parrotta, G.; Vera, R.; Sanchez-Lopez, E.; Galindo, R.; Kjeldsen, F.; Badia, J.; Baldoma, L.; Espina, M.; Garcia, M.L. Ocular penetration of fluorometholone-loaded PEG-PLGA nanoparticles functionalized with cell-penetrating peptides. Nanomedicine 2019, 14, 3089–3104. [Google Scholar] [CrossRef] [PubMed]
- Galindo-Camacho, R.M.; Haro, I.; Gomara, M.J.; Espina, M.; Fonseca, J.; Martins-Gomes, C.; Camins, A.; Silva, A.M.; Garcia, M.L.; Souto, E.B. Cell penetrating peptides-functionalized Licochalcone-A-loaded PLGA nanoparticles for ocular inflammatory diseases: Evaluation of in vitro anti-proliferative effects, stabilization by freeze-drying and characterization of an in-situ forming gel. Int. J. Pharm. 2023, 639, 122982. [Google Scholar] [CrossRef]
- Ghassemi, A.H.; van Steenbergen, M.J.; Barendregt, A.; Talsma, H.; Kok, R.J.; van Nostrum, C.F.; Crommelin, D.J.; Hennink, W.E. Controlled release of octreotide and assessment of peptide acylation from poly(D,L-lactide-co-hydroxymethyl glycolide) compared to PLGA microspheres. Pharm. Res. 2012, 29, 110–120. [Google Scholar] [CrossRef][Green Version]
- Zhang, C.; Wu, L.; Tao, A.; Bera, H.; Tang, X.; Cun, D.; Yang, M. Formulation and in vitro characterization of long-acting PLGA injectable microspheres encapsulating a peptide analog of LHRH. J. Mater. Sci. Technol. 2021, 63, 133–144. [Google Scholar] [CrossRef]
- Liu, M.; Feng, D.; Liang, X.; Li, M.; Yang, J.; Wang, H.; Pang, L.; Zhou, Z.; Yang, Z.; Kong, D.; et al. Old Dog New Tricks: PLGA Microparticles as an Adjuvant for Insulin Peptide Fragment-Induced Immune Tolerance against Type 1 Diabetes. Mol. Pharm. 2020, 17, 3513–3525. [Google Scholar] [CrossRef]
- Tu, S.C.; Mai, S.T.; Shu, D.; Huang, Y.X.; Nie, Z.H.; Wang, Y.; Yang, W.L. Microfluidic-based preparation of PLGA microspheres facilitating peptide sustained-release. Mater. Lett. 2024, 368, 4. [Google Scholar] [CrossRef]
- Ramezanpour, S.; Tavatoni, P.; Akrami, M.; Navaei-Nigjeh, M.; Shiri, P. Potential Wound Healing of PLGA Nanoparticles Containing a Novel L-Carnitine-GHK Peptide Conjugate. J. Nanomater. 2022, 2022, 9. [Google Scholar] [CrossRef]
- Esfandyari-Manesh, M.; Abdi, M.; Talasaz, A.H.; Ebrahimi, S.M.; Atyabi, F.; Dinarvand, R. S2P peptide-conjugated PLGA-Maleimide-PEG nanoparticles containing Imatinib for targeting drug delivery to atherosclerotic plaques. DARU J. Pharm. Sci. 2020, 28, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.H.; Heo, S.C.; Kwon, Y.W.; Kim, H.D.; Kim, S.H.; Jang, I.H.; Kim, J.H.; Hwang, N.S. Injectable PLGA microspheres encapsulating WKYMVM peptide for neovascularization. Acta Biomater. 2015, 25, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Imanparast, F.; Faramarzi, M.A.; Vatannejad, A.; Paknejad, M.; Deiham, B.; Kobarfard, F.; Amani, A.; Doosti, M. mZD7349 peptide-conjugated PLGA nanoparticles directed against VCAM-1 for targeted delivery of simvastatin to restore dysfunctional HUVECs. Microvasc. Res. 2017, 112, 14–19. [Google Scholar] [CrossRef]
- Ozcicek, I.; Aysit, N.; Balcikanli, Z.; Ayturk, N.U.; Aydeger, A.; Baydas, G.; Aydin, M.S.; Altintas, E.; Erim, U.C. Development of BDNF/NGF/IKVAV Peptide Modified and Gold Nanoparticle Conductive PCL/PLGA Nerve Guidance Conduit for Regeneration of the Rat Spinal Cord Injury. Macromol. Biosci. 2024, 24, e2300453. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.C.; Kim, J.; Kim, S.E.; Song, S.J.; Hong, S.W.; Oh, J.W.; Lee, J.; Park, J.C.; Hyon, S.H.; Han, D.W. RGD peptide and graphene oxide co-functionalized PLGA nanofiber scaffolds for vascular tissue engineering. Regen. Biomater. 2017, 4, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Singh, B.; Li, H.S.; Kim, Y.K.; Kang, S.K.; Nah, J.W.; Choi, Y.J.; Cho, C.S. Targeted oral delivery of BmpB vaccine using porous PLGA microparticles coated with M cell homing peptide-coupled chitosan. Biomaterials 2014, 35, 2365–2373. [Google Scholar] [CrossRef]
- Wang, P.Y.; Wu, T.H.; Tsai, W.B.; Kuo, W.H.; Wang, M.J. Grooved PLGA films incorporated with RGD/YIGSR peptides for potential application on skeletal muscle tissue engineering. Colloids Surf. B Biointerfaces 2013, 110, 88–95. [Google Scholar] [CrossRef]
- Park, J.S.; Yang, H.N.; Yi, S.W.; Kim, J.H.; Park, K.H. Neoangiogenesis of human mesenchymal stem cells transfected with peptide-loaded and gene-coated PLGA nanoparticles. Biomaterials 2016, 76, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Egusquiaguirre, S.P.; Manguan-Garcia, C.; Perona, R.; Pedraz, J.L.; Hernandez, R.M.; Igartua, M. Development and validation of a rapid HPLC method for the quantification of GSE4 peptide in biodegradable PEI-PLGA nanoparticles. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2014, 972, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Zheng, Q.; Yang, S.; Guo, X. Effects of functionalization of PLGA-[Asp-PEG]n copolymer surfaces with Arg-Gly-Asp peptides, hydroxyapatite nanoparticles, and BMP-2-derived peptides on cell behavior in vitro. J. Biomed. Mater. Res. A 2014, 102, 4526–4535. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, L.; Wang, Y.; Chen, X.; Zhang, P. Improved Cell Adhesion and Osteogenesis of op-HA/PLGA Composite by Poly(dopamine)-Assisted Immobilization of Collagen Mimetic Peptide and Osteogenic Growth Peptide. ACS Appl. Mater. Interfaces 2016, 8, 26559–26569. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xu, Y.; Wang, Y.; Ren, H.; Meng, Z.; Liu, K.; Liu, Z.; Huang, H.; Li, X. Proton Oriented-”Smart Depot” for Responsive Release of Ca(2+) to Inhibit Peptide Acylation in PLGA Microspheres. Pharm. Res. 2019, 36, 119. [Google Scholar] [CrossRef]
- Zhang, Y.; Schwendeman, S.P. Minimizing acylation of peptides in PLGA microspheres. J. Control. Release 2012, 162, 119–126. [Google Scholar] [CrossRef][Green Version]
- Wang, M.; Guo, X.; Tan, R.; She, Z.; Feng, Q. Effect of stabilizers on bioactivity of peptide-24 in PLGA microspheres. Med. Chem. 2013, 9, 1123–1128. [Google Scholar] [CrossRef] [PubMed]
- Senturk, F.; Cakmak, S. Fabrication of curcumin-loaded magnetic PEGylated-PLGA nanocarriers tagged with GRGDS peptide for improving anticancer activity. Methodsx 2023, 10, 102229. [Google Scholar] [CrossRef]
- Wang, J.H.; Li, Y.; Jing, J.; Yue, H.L.; Zhang, L.L.; Hua, W.; Li, N.; Liu, X.; Han, J.A. Practical evaluations of bioactive peptide-modified Fluorapatite/PLGA multifunctional nano-clustery composite against for root caries restorations to inhibit periodontitis-related pathogens in periodontitis care. Mater. Res. Express 2021, 8, 055013. [Google Scholar] [CrossRef]
- Shirangi, M.; Najafi, M.; Rijkers, D.T.; Kok, R.J.; Hennink, W.E.; van Nostrum, C.F. Inhibition of Octreotide Acylation Inside PLGA Microspheres by Derivatization of the Amines of the Peptide with a Self-Immolative Protecting Group. Bioconjug. Chem. 2016, 27, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Lau, C.Y.J.; Cabello, I.T.; Garssen, J.; Willemsen, L.E.M.; Hennink, W.E.; van Nostrum, C.F. Live Cell Imaging by Forster Resonance Energy Transfer Fluorescence to Study Trafficking of PLGA Nanoparticles and the Release of a Loaded Peptide in Dendritic Cells. Pharmaceuticals 2023, 16, 818. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Tian, G.; Zhi, M.; Liu, Z.; Du, Y.; Lu, X.; Li, M.; Bai, J.; Li, X.; Deng, J.; et al. Functionalized PLGA Microsphere Loaded with Fusion Peptide for Therapy of Bone Defects. ACS Biomater. Sci. Eng. 2024, 10, 2463–2476. [Google Scholar] [CrossRef]
- Cai, H.; Liang, Z.; Huang, W.; Wen, L.; Chen, G. Engineering PLGA nano-based systems through understanding the influence of nanoparticle properties and cell-penetrating peptides for cochlear drug delivery. Int. J. Pharm. 2017, 532, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Chen, S.; Gao, Y.; Guo, F.; Li, F.; Xie, B.; Zhou, J.; Zhong, H. Enhanced oral bioavailability of insulin using PLGA nanoparticles co-modified with cell-penetrating peptides and Engrailed secretion peptide (Sec). Drug Deliv. 2016, 23, 1980–1991. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.C.; Lee, J.H.; Kim, M.J.; Hong, S.W.; Kim, B.; Hyun, J.K.; Choi, Y.S.; Park, J.C.; Han, D.W. Stimulating effect of graphene oxide on myogenesis of C2C12 myoblasts on RGD peptide-decorated PLGA nanofiber matrices. J. Biol. Eng. 2015, 9, 22. [Google Scholar] [CrossRef] [PubMed]
- Aydeger, A.; Aysit, N.; Baydas, G.; Cakici, C.; Erim, U.C.; Arpa, M.D.; Ozcicek, I. Design of IKVAV peptide/gold nanoparticle decorated, micro/nano-channeled PCL/PLGA film scaffolds for neuronal differentiation and neurite outgrowth. Biomater. Adv. 2023, 152, 213472. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Hao, S.; Zheng, Q.; Li, J.; Zheng, J.; Hu, Z.; Yang, S.; Guo, X.; Yang, Q. Bone induction by biomimetic PLGA copolymer loaded with a novel synthetic RADA16-P24 peptide in vivo. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 3336–3345. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Wei, T.; Hua, Y.; Wang, Z.; Zhang, L. Effective Antitumor of Orally Intestinal Targeting Penetrating Peptide-Loaded Tyroserleutide/PLGA Nanoparticles in Hepatocellular Carcinoma. Int. J. Nanomed. 2021, 16, 4495–4513. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Feng, L.; Fan, L.; Zha, Y.; Guo, L.; Zhang, Q.; Chen, J.; Pang, Z.; Wang, Y.; Jiang, X.; et al. Targeting the brain with PEG-PLGA nanoparticles modified with phage-displayed peptides. Biomaterials 2011, 32, 4943–4950. [Google Scholar] [CrossRef]
- Galindo, R.; Sanchez-Lopez, E.; Gomara, M.J.; Espina, M.; Ettcheto, M.; Cano, A.; Haro, I.; Camins, A.; Garcia, M.L. Development of Peptide Targeted PLGA-PEGylated Nanoparticles Loading Licochalcone-A for Ocular Inflammation. Pharmaceutics 2022, 14, 285. [Google Scholar] [CrossRef]
- Zhao, X.; Ni, S.; Song, Y.; Hu, K. Intranasal delivery of Borneol/R8dGR peptide modified PLGA nanoparticles co-loaded with curcumin and cisplatin alleviate hypoxia in pediatric brainstem glioma which improves the synergistic therapy. J. Control. Release 2023, 362, 121–137. [Google Scholar] [CrossRef]
- Bisht, R.; Rupenthal, I.D. PLGA nanoparticles for intravitreal peptide delivery: Statistical optimization, characterization and toxicity evaluation. Pharm. Dev. Technol. 2018, 23, 324–333. [Google Scholar] [CrossRef]
- Song, X.; Li, X.; Wang, F.; Wang, L.; Lv, L.; Xie, Q.; Zhang, X.; Shao, X. Bioinspired Protein/Peptide Loaded 3D Printed PLGA Scaffold Promotes Bone Regeneration. Front. Bioeng. Biotechnol. 2022, 10, 832727. [Google Scholar] [CrossRef]
- Liu, J.; Xu, Y.; Wang, Y.; Ren, H.; Meng, Z.; Liu, K.; Liu, Z.; Huang, H.; Li, X. Effect of inner pH on peptide acylation within PLGA microspheres. Eur. J. Pharm. Sci. 2019, 134, 69–80. [Google Scholar] [CrossRef]
- Chintapula, U.; Yang, S.; Nguyen, T.; Li, Y.; Jaworski, J.; Dong, H.; Nguyen, K.T. Supramolecular Peptide Nanofiber/PLGA Nanocomposites for Enhancing Pulmonary Drug Delivery. ACS Appl. Mater. Interfaces 2022, 14, 56498–56509. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.C.; Lee, J.H.; Jin, O.S.; Lee, E.J.; Jin, L.H.; Kim, C.S.; Hong, S.W.; Han, D.W.; Kim, C.; Oh, J.W. RGD peptide-displaying M13 bacteriophage/PLGA nanofibers as cell-adhesive matrices for smooth muscle cells. J. Korean Phys. Soc. 2015, 66, 12–16. [Google Scholar] [CrossRef]
- Wu, C.; Wang, C.; Sun, L.; Xu, K.; Zhong, W. PLGA nanoparticle-reinforced supramolecular peptide hydrogels for local delivery of multiple drugs with enhanced synergism. Soft Matter 2020, 16, 10528–10536. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.; Smyth, J.W.; Will, J.; Roberts, P.; Grek, C.L.; Ghatnekar, G.S.; Sheng, Z.; Gourdie, R.G.; Lamouille, S.; Foster, E.J. Development of PLGA nanoparticles for sustained release of a connexin43 mimetic peptide to target glioblastoma cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 108, 110191. [Google Scholar] [CrossRef]
- Jin, L.; Pan, Y.; Pham, A.C.; Boyd, B.J.; Norton, R.S.; Nicolazzo, J.A. Prolonged Plasma Exposure of the Kv1.3-Inhibitory Peptide HsTX1[R14A] by Subcutaneous Administration of a Poly(Lactic-co-Glycolic Acid) (PLGA) Microsphere Formulation. J. Pharm. Sci. 2021, 110, 1182–1188. [Google Scholar] [CrossRef]
- Sneh-Edri, H.; Likhtenshtein, D.; Stepensky, D. Intracellular targeting of PLGA nanoparticles encapsulating antigenic peptide to the endoplasmic reticulum of dendritic cells and its effect on antigen cross-presentation in vitro. Mol. Pharm. 2011, 8, 1266–1275. [Google Scholar] [CrossRef]
- Mohan, A.K.; Minsa, M.; Kumar, T.R.S.; Kumar, G.S.V. Multi-Layered PLGA-PEI Nanoparticles Functionalized with TKD Peptide for Targeted Delivery of Pep5 to Breast Tumor Cells and Spheroids. Int. J. Nanomed. 2022, 17, 5581–5600. [Google Scholar] [CrossRef] [PubMed]
- Koyuncu, R.; Duruksu, G.; Ozcelik, B.; Mert, S.; Yazir, Y. Effect of Peptide-Lipid Conjugates Loaded PLGA Nanoparticles Against Cancer Cells In-Vitro. J. Biomed. Nanotechnol. 2023, 19, 1321–1336. [Google Scholar] [CrossRef]
- Margaroni, M.; Agallou, M.; Kontonikola, K.; Karidi, K.; Kammona, O.; Kiparissides, C.; Gaitanaki, C.; Karagouni, E. PLGA nanoparticles modified with a TNFalpha mimicking peptide, soluble Leishmania antigens and MPLA induce T cell priming in vitro via dendritic cell functional differentiation. Eur. J. Pharm. Biopharm. 2016, 105, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Derman, S.; Akdeste, Z.M.; Ates, S.C.; Mansuroglu, B.; Kizilbey, K.; Bagirova, M.; Allahverdiyev, A. The study of syntetic peptide loaded plga nanoparticles cytotoxicity in vitro. Fresen. Environ. Bull. 2017, 26, 1646–1653. [Google Scholar]
- Santos, A.P.d.; Oliveira, R.C.R.d.; Louchard, B.O.; Uchoa, A.F.J.; Ricardo, N.M.P.S.; Leal, L.K.A.M.; Rádis-Baptista, G.; Araújo, T.G.d. Preparation of PLGA Nanoparticles Loaded with the Anti-Infective Ctn[15-34] Peptide for Antifungal Application. Braz. Arch. Biol. Technol. 2023, 66, e20220775. [Google Scholar] [CrossRef]
- Marinelli, L.; Ciulla, M.; Ritsema, J.A.S.; van Nostrum, C.F.; Cacciatore, I.; Dimmito, M.P.; Palmerio, F.; Orlando, G.; Robuffo, I.; Grande, R.; et al. Preparation, Characterization, and Biological Evaluation of a Hydrophilic Peptide Loaded on PEG-PLGA Nanoparticles. Pharmaceutics 2022, 14, 1821. [Google Scholar] [CrossRef]
- Situ, J.Q.; Wang, X.J.; Zhu, X.L.; Xu, X.L.; Kang, X.Q.; Hu, J.B.; Lu, C.Y.; Ying, X.Y.; Yu, R.S.; You, J.; et al. Multifunctional SPIO/DOX-loaded A54 Homing Peptide Functionalized Dextran-g-PLGA Micelles for Tumor Therapy and MR Imaging. Sci. Rep. 2016, 6, 35910. [Google Scholar] [CrossRef] [PubMed]
- Tondeur, E.G.M.; Voerman, J.S.A.; Geleijnse, M.A.A.; van Hofwegen, L.S.; van Krimpen, A.; Koerner, J.; Mishra, G.; Song, Z.; Schliehe, C. Sec22b and Stx4 Depletion Has No Major Effect on Cross-Presentation of PLGA Microsphere-Encapsulated Antigen and a Synthetic Long Peptide In Vitro. J. Immunol. 2023, 211, 1203–1215. [Google Scholar] [CrossRef]
- Ma, W.; Chen, M.; Kaushal, S.; McElroy, M.; Zhang, Y.; Ozkan, C.; Bouvet, M.; Kruse, C.; Grotjahn, D.; Ichim, T.; et al. PLGA nanoparticle-mediated delivery of tumor antigenic peptides elicits effective immune responses. Int. J. Nanomed. 2012, 7, 1475–1487. [Google Scholar] [CrossRef]
- Mahmoud, M.Y.; Steinbach-Rankins, J.M.; Demuth, D.R. Functional assessment of peptide-modified PLGA nanoparticles against oral biofilms in a murine model of periodontitis. J. Control. Release 2019, 297, 3–13. [Google Scholar] [CrossRef]
- Jiang, T.; Yu, X.; Carbone, E.J.; Nelson, C.; Kan, H.M.; Lo, K.W. Poly aspartic acid peptide-linked PLGA based nanoscale particles: Potential for bone-targeting drug delivery applications. Int. J. Pharm. 2014, 475, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Kostadinova, A.I.; Middelburg, J.; Ciulla, M.; Garssen, J.; Hennink, W.E.; Knippels, L.M.J.; van Nostrum, C.F.; Willemsen, L.E.M. PLGA nanoparticles loaded with beta-lactoglobulin-derived peptides modulate mucosal immunity and may facilitate cow’s milk allergy prevention. Eur. J. Pharmacol. 2018, 818, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Li, W.; Yuan, X.; Rakhmanov, Y.; Wang, P.; Lu, R.; Mao, Z.; Shang, X.; You, H. Chondrogenic effect of cell-based scaffold of self-assembling peptides/PLGA-PLL loading the hTGFbeta3 plasmid DNA. J. Mater. Sci. Mater. Med. 2016, 27, 19. [Google Scholar] [CrossRef]
- Chen, H.A.; Ma, Y.H.; Hsu, T.Y.; Chen, J.P. Preparation of Peptide and Recombinant Tissue Plasminogen Activator Conjugated Poly(Lactic-Co-Glycolic Acid) (PLGA) Magnetic Nanoparticles for Dual Targeted Thrombolytic Therapy. Int. J. Mol. Sci. 2020, 21, 2690. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).