Selected Medicinal Plants Used in the Treatment and Management of Tuberculosis and Related Symptoms in South Africa
Abstract
:1. Introduction
2. Literature Search Strategy
2.1. Botanical Description of Carpobrotus edulis (L.) Bolus
2.1.1. Ethnobotanical Uses of Carpobrotus edulis
2.1.2. Biological Effects of Carpobrotus edulis
2.1.3. Phytochemistry of Carpobrotus edulis
| Plant Part | Uses | Extraction | Biological Effect | Phytochemistry | References | ||
|---|---|---|---|---|---|---|---|
| Traditional | Method | Type of Extract | Analysis/Profile | Bioactive Components | |||
| Leaf juice Leaf pulp | Mouth and throat infections, dysentery, digestive troubles, TB, diuretic and styptic, eczema, wounds and burns, toothache, earache, oral and vaginal thrush Wounds and infections | NR | NR | Antimicrobial activity | NR | Catechin (6), malic acid (9), citric acid (8), ferulic acid (5) | [13] |
| Leaves | TB, sore throat, lung infections | NR | NR | Antimicrobial activity | TLC | Tannins and flavonoids, Hyperoside, rutin, neohesperidin | [13,17] |
| Centrifugation | Methanolic extract | Inhibits the growth of multidrug-resistant M. tuberculosis within three days of culture and methicillin-resistant S. aureus within six hours of culture below toxic levels | NR | NR | [20] | ||
| Maceration with stirring followed by centrifuging | Methanolic extract | Antimicrobial activity against M. catarrhalis with concentration of 50 mg/mL | NR | NR | [21] | ||
| Methanolic extract | Antiproliferative activity | Column chromatography | β-amyrin (1), oleanolic acid (2), uvaol (3), monogalactosyldiacylglycerol (7) (MGDG), catechin (6), epicatechin (4), and procyanidin B5 (13) | [25] | |||
2.2. Botanical Description of Drosera capensis L.
2.2.1. Ethnobotanical Uses of Drosera capensis
2.2.2. Biological Effects of Drosera capensis
2.2.3. Phytochemistry of Drosera capensis
2.3. Botanical Decription of Pelargonium reniforme Curtis
2.3.1. Ethnobotanical Uses of Pelargonium reniforme Curtis
2.3.2. Biological Effects of Pelargonium reniforme
2.3.3. Phytochemistry of Pelargonium reniforme
2.4. Botanical Description of Tulbaghia violacea Harv.
2.4.1. Ethnobotanical Uses of Tulbaghua violacea
2.4.2. Biological Effects of Tulbaghia violacea
2.4.3. Phytochemistry of Tulbaghia violacea
3. Conclusions
- From this review, it can be concluded that the four plants are not only used to treat or manage tuberculosis. They are also used to treat other lower-respiratory ailments, including cough and fever, which may be signs and symptoms of TB.
- From the four commonly used plants, the important phytochemicals were identified from different plants, including flavonoids, phenols, terpenes, naphthoquinones, and phenolics. The mentioned phytochemicals are generally abundant in nature and are also found in other plants.
- Carpobrotus edulis and Tulbaghi violacea are edible plants, and, because of this fact, it may be safe to conclude that the plant can be taken or formulated as supplements or nutraceuticals. Many more plants are taken as teas, such as Senna, Green tea, and Rooibos. The teas contain phytochemicals such as flavonoids. Therefore, the plants may most probably be taken as tea, if edible, to manage illnesses.
- The South African Health Products Regulatory Authority (SAHPRA) has a committee dedicated to evaluate complementary medicine. It will then be recommended to submit products for evaluation with the safety and efficacy profiles.
- There are reports that provide evidence that phytochemicals, including alkaloids, flavones, phenols, terpenoids, and some fatty acids, are effective against Mycobacterium strains [111]. Most of these phytochemicals were identified in the four plants, and thus there is scientific evidence that these plants and the isolated compounds from them could serve as potential drug candidates for new anti-TB drugs. However, there are no reports beyond the potential drug candidates. It is important to note that basic research has a great impact in assembling knowledge and there is, therefore, a need to report data in a systemic manner.
- Plumbagin (44) is one of the most effective isolated compounds and was identified in D. capensis as per the review. Plumbagin (44) is effective against MDR and XDR tuberculosis [112].
- From Table 10, it is clear that further research can be conducted on compounds identified in T. violacea to investigate their efficacy against TB, as well as their mode of action. In fact, Table 4, Table 6, Table 8 and Table 10 indicate that there is an opportunity to further investigate which specific compounds are responsible for the effects against M. tuberculosis. Further research into these plants may provide treatments for TB, as well as the management or treatment of the signs and symptoms of TB, including the clinical safety and efficacy aspects.
- Many plants are commercialized without any scientific evidence, which poses a danger to society. It is therefore important for SAHPRA, as well as other Medicine Regulatory Authorities (MRAs), around the globe to develop frameworks that guide the assessment of the safety, efficacy, and quality of traditional medicines, as well as to have a harmonized regulatory standard amongst the various MRAs.
- Generally, there is an assumption that the use of traditional medicine is safer than modern medicine. Therefore, there is a need to educate the public regarding the safe use of medicinal plants. Some plants are toxic and can be fatal when taken in large quantities.
- Moreover, there is a need to inform and educate healthcare professionals regarding the use of traditional medicine. Some patients take traditional medicine and do not inform their healthcare provider. This is largely due to the stigma around the use of traditional medicine, thus resulting in drug–herb interactions.
- TB is an opportunistic infection, the risk of infection increasing in HIV-positive patients. There is, therefore, a need to conduct drug interactions, especially to ensure the safe use of traditional medicine in HIV-positive patients. This thus creates a gap in pharmacovigilance studies to develop criteria for each countries’ MRA.
- For most isolated compounds, there was no progress made from the study of extracts, phytochemical profiling to isolation, and in vitro studies, as well as little progress made in few in vivo studies, to identify compounds. Most studies end there; however, there is a need for basic research that will enable further higher-level studies to be performed, such as clinical studies using animal models, pharmacodynamic and pharmacokinetic studies, and quality assurance of traditional medicines in general.
- The number of deaths from TB remains high, despite all of the interventions such as the direct observed therapy (DOT) program, which involves healthcare workers, or other designated people, making sure that patients take their medicine correctly, thereby ensuring adherence and tolerability. According to the World Health Organization, the identification of TB cases increased after the Coronavirus Disease 2019 (COVID-19) pandemic due to the renewed attention toward infectious diseases other than COVID-19.
- TB deaths, however, remain high, especially in economically burdened countries. Governments may provide treatment; however, there are challenges around food insecurity and access. Without effective nutrition, the immune system is weakened and this increases the risk of active TB. Adherence to TB treatment thus proves to be difficult due to lack of food and the multiple drugs they have to take, resulting in resistance and, consequently, death.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| COVID-19 | Coronavirus Disease 2019 |
| DOT | direct observed therapy |
| TB | Tuberculosis |
| HIV | Human Immunodeficiency Virus |
| MRAs | Medicine Regulatory Authorities |
| SAHPRA | South African Health Products Authority |
| MDR | multi-drug resistant |
| XRD | extensive-drug resistant |
References
- Wells, B.G.; Dipiro, J.T.; Dipiro, C.V.; Schwinghammer, T.L. Pharmacotherapy Handbook, 7th ed.; McGraw-Hill: New York, NY, USA, 2009; ISBN 9780071643269. [Google Scholar]
- World Health Organization. Use of Alternative Interferon-Gamma Release Assays for the Diagnosis of TB Infection: WHO Policy Statement; World Health Organization: Geneva, Switzerland, 2022. [Google Scholar]
- World Health Organization. Tuberculosis. Available online: https://www.who.int/news-room/fact-sheets/detail/tuberculosis (accessed on 25 November 2022).
- Centers for Disease Control and Prevention. Treatment for TB Disease. Available online: https://www.cdc.gov/tb/topic/treatment/tbdisease.htm (accessed on 28 June 2021).
- Seung, K.J.; Keshavjee, S.; Rich, M.L. Multidrug-Resistant Tuberculosis and Extensively Drug-Resistant Tuberculosis. Cold Spring Harb. Perspect. Med. 2015, 5, a017863. [Google Scholar] [CrossRef] [PubMed]
- TB Alliance. FDA Approves New Treatment for Highly Drug-Resistant Forms of Tuberculosis. Available online: https://www.tballiance.org/news-fda-approves-new-treatment-highly-drug-resistant-forms-tuberculosis/ (accessed on 12 March 2023).
- Centers for Disease Control and Prevention. Treatment of Tuberculosis Disease; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2013; pp. 139–187. [Google Scholar]
- Sotgiu, G.; Centis, R.; D’Ambrosio, L.; Battista Migliori, G. Tuberculosis Treatment and Drug Regimens. Cold Spring Harb. Perspect. Med. 2015, 5, a017822. [Google Scholar] [CrossRef] [PubMed]
- Leavens, J. TB Online—How TB Is Treated. Available online: https://www.tbonline.info/posts/2011/9/2/how-tb-treated/ (accessed on 29 June 2022).
- National Institute of Allergy and Infectious Diseases. Tuberculosis Drugs and Mechanisms of Action. Available online: https://www.niaid.nih.gov/diseases-conditions/tbdrugs (accessed on 15 April 2024).
- Thinwa, J. Indigenous Healing Practices and Their Effect on TB and HIV/TB Patients’ Utilization and Compliance with Anti-TB Medication. Indep. Study Proj. Collect. 2004, 498, 4–48. [Google Scholar]
- van Wyk, B.-E.; van Oudtshoorn, B.; Gericke, N. Medicinal Plants of South Africa, 2nd ed.; Briza Publications: Pretoria, South Africa, 2017. [Google Scholar]
- Akinyede, K.A.; Ekpo, O.E.; Oguntibeju, O.O. Ethnopharmacology, Therapeutic Properties and Nutritional Potentials of Carpobrotus edulis: A Comprehensive Review. Sci. Pharm. 2020, 88, 39. [Google Scholar] [CrossRef]
- Rocha, M.I.; Rodrigues, M.J.; Pereira, C.; Pereira, H.; da Silva, M.M.; da Neng, N.R.; Nogueira, J.M.F.; Varela, J.; Barreira, L.; Custódio, L. Biochemical Profile and in Vitro Neuroprotective Properties of Carpobrotus edulis L., a Medicinal and Edible Halophyte Native to the Coast of South Africa. S. Afr. J. Bot. 2017, 111, 222–231. [Google Scholar] [CrossRef]
- El-Raouf, H.S.A. Taxonomic Significance of Leaves in Family Aizoaceae. Saudi J. Biol. Sci. 2021, 28, 512–522. [Google Scholar] [CrossRef]
- Omoruyi, B.E.; Bradley, G.; Afolayan, A.J. Antioxidant and Phytochemical Properties of Carpobrotus edulis (L.) Bolus Leaf Used for the Management of Common Infections in HIV/AIDS Patients in Eastern Cape Province. BMC Complement. Altern. Med. 2012, 12, 215. [Google Scholar] [CrossRef]
- Malan, C.; Notten, A. Carpobrotus Edulis. PlantZAfrica. Available online: http://pza.sanbi.org/carpobrotus-edulis (accessed on 26 July 2021).
- Semenya, S.S.; Maroyi, A. Data on Medicinal Plants Used to Treat Respiratory Infections and Related Symptoms in South Africa. Data Br. 2018, 21, 419–423. [Google Scholar] [CrossRef]
- Awouafack, M.D.; McGaw, L.J.; Gottfried, S.; Mbouangouere, R.; Tane, P.; Spiteller, M.; Eloff, J.N. Antimicrobial Activity and Cytotoxicity of the Ethanol Extract, Fractions and Eight Compounds Isolated from Eriosema robustum (Fabaceae). BMC Complement. Altern. Med. 2013, 13, 289. [Google Scholar] [CrossRef]
- Martins, M.; Ordway, D.; Kristiansen, M.; Viveiros, M.; Leandro, C.; Molnar, J.; Amaral, L. Inhibition of the Carpobrotus edulis Methanol Extract on the Growth of Phagocytosed Multidrug-Resistant Mycobacterium tuberculosis and Methicillin-Resistant Staphylococcus aureus. Fitoterapia 2005, 76, 96–99. [Google Scholar] [CrossRef]
- Van Der Watt, E.; Pretorius, J.C. Purification and Identification of Active Antibacterial Components in Carpobrotus edulis L. J. Ethnopharmacol. 2001, 76, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Mulaudzi, R.B.; Aremu, A.O.; Rengasamy, K.R.R.; Adebayo, S.A.; McGaw, L.J.; Amoo, S.O.; Van Staden, J.; Du Plooy, C.P. Antidiabetic, Anti-Inflammatory, Anticholinesterase and Cytotoxicity Determination of Two Carpobrotus Species. S. Afr. J. Bot. 2019, 125, 142–148. [Google Scholar] [CrossRef]
- Omoruyi, B.E.; Afolayan, A.J.; Bradley, G. Chemical Composition Profiling and Antifungal Activity of the Essential Oil and Plant Extracts of Mesembryanthemum edule (L.) Bolus Leaves. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 19–30. [Google Scholar] [CrossRef]
- Martins, A.; Vasas, A.; Schelz, Z.; Viveiros, M.; Molnár, J.; Hohmann, J.; Amaral, L. Constituents of Carpobrotus edulis Inhibit P-Glycoprotein of MDR1-Transfected Mouse Lymphoma Cells. Anticancer Res. 2010, 30, 829–836. [Google Scholar] [PubMed]
- Martins, A.; Vasas, A.; Viveiros, M.; Molnár, J.; Hohmann, J.; Amaral, L. Antibacterial Properties of Compounds Isolated from Carpobrotus edulis. Int. J. Antimicrob. Agents 2011, 37, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Viet, T.D.; Xuan, T.D.; Anh, L.H. α-Amyrin and β-Amyrin Isolated from Celastrus hindsii Leaves and Their Antioxidant, Anti-Xanthine Oxidase, and Anti-Tyrosinase Potentials. Molecules 2021, 26, 7248. [Google Scholar] [CrossRef]
- Santos, F.A.; Frota, J.T.; Arruda, B.R.; De Melo, T.S.; André, A.; Almeida, D.C.; Anne, G.; Brito, D.C.; Chaves, M.H.; Rao, V.S. Antihyperglycemic and Hypolipidemic Effects of α, β-Amyrin, a Triterpenoid Mixture from Protium heptaphyllum in Mice. Lipids Health Dis. 2012, 11, 98. [Google Scholar]
- Ayeleso, T.B.; Matumba, M.G.; Mukwevho, E. Oleanolic Acid and Its Derivatives: Biological Activities and Therapeutic Potential in Chronic Diseases. Molecules 2017, 22, 1915. [Google Scholar] [CrossRef]
- Carmo, J.; Cavalcante-Araújo, P.; Silva, J.; Ferro, J.; Correia, A.C.; Lagente, V.; Barreto, E. Uvaol Improves the Functioning of Fibroblasts and Endothelial Cells and Accelerates the Healing of Cutaneous Wounds in Mice. Molecules 2020, 25, 4982. [Google Scholar] [CrossRef]
- Du, S.Y.; Huang, H.F.; Li, X.Q.; Zhai, L.X.; Zhu, Q.C.; Zheng, K.; Song, X.; Xu, C.S.; Li, C.Y.; Li, Y.; et al. Anti-Inflammatory Properties of Uvaol on DSS-Induced Colitis and LPS-Stimulated Macrophages. Chinese Med. 2020, 15, 43. [Google Scholar] [CrossRef]
- Bernatova, I. Biological Activities of (−)-Epicatechin and (−)-Epicatechin-Containing Foods: Focus on Cardiovascular and Neuropsychological Health. Biotechnol. Adv. 2018, 36, 666–681. [Google Scholar] [CrossRef] [PubMed]
- Shay, J.; Elbaz, H.A.; Lee, I.; Zielske, S.P.; Malek, M.H.; Hüttemann, M. Molecular Mechanisms and Therapeutic Effects of (−)-Epicatechin and Other Polyphenols in Cancer, Inflammation, Diabetes, and Neurodegeneration. Oxid. Med. Cell. Longev. 2015, 2015, 181260. [Google Scholar] [CrossRef]
- Kumar, N.; Pruthi, V. Potential Applications of Ferulic Acid from Natural Sources. Biotechnol. Rep. 2014, 4, 86–93. [Google Scholar] [CrossRef]
- Raj, N.D.; Singh, D. A Critical Appraisal on Ferulic Acid: Biological Profile, Biopharmaceutical Challenges and Nano Formulations. Health Sci. Rev. 2022, 5, 100063. [Google Scholar] [CrossRef]
- Bae, J.; Kim, N.; Shin, Y.; Kim, S.-Y.; Kim, Y.-J. Activity of Catechins and Their Applications. Biomed. Dermatol. 2020, 4, 8. [Google Scholar] [CrossRef]
- Fan, F.Y.; Sang, L.X.; Jiang, M.; McPhee, D.J. Catechins and Their Therapeutic Benefits to Inflammatory Bowel Disease. Molecules 2017, 22, 484. [Google Scholar] [CrossRef] [PubMed]
- Foseid, L.; Devle, H.; Naess-Andresen, C.F.; Ekeberg, D. Laminaria hyperborea as a Source of Valuable Glyceroglycolipids—A Characterization of Galactosyldiacilglycerols in Stipe and Blade by HPLC-MS/MS. AppliedChem 2022, 2, 185–198. [Google Scholar] [CrossRef]
- Basnet, R.; Zhang, J.; Hussain, N.; Shu, Q. Characterization and Mutational Analysis of a Monogalactosyldiacylglycerol Synthase Gene OsMGD2 in Rice. Front. Plant Sci. 2019, 10, 992. [Google Scholar] [CrossRef]
- ELİUZ, E. Antimicrobial Activity of Citric Acid against Escherichia Coli, Staphylococcus Aureus and Candida Albicans as a Sanitizer Agent. Eurasian J. For. Sci. 2020, 8, 295–301. [Google Scholar] [CrossRef]
- Liu, Q.; Tang, G.Y.; Zhao, C.N.; Gan, R.Y.; Li, H. Bin Antioxidant Activities, Phenolic Profiles, and Organic Acid Contents of Fruit Vinegars. Antioxidants 2019, 8, 78. [Google Scholar] [CrossRef]
- Adamczak, A.; Ożarowski, M.; Karpiński, T.M. Antibacterial Activity of Some Flavonoids and Organic Acids Widely Distributed in Plants. J. Clin. Med. 2020, 9, 109. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Chen, S.; Xia, W.; Sui, H.; Fu, X. Hyperoside: A Review of Its Structure, Synthesis, Pharmacology, Pharmacokinetics and Toxicity. Molecules 2022, 27, 3009. [Google Scholar] [CrossRef]
- Park, J.Y.; Han, X.; Piao, M.J.; Oh, M.C.; Fernando, P.M.D.J.; Kang, K.A.; Ryu, Y.S.; Jung, U.; Kim, I.G.; Hyun, J.W. Hyperoside Induces Endogenous Antioxidant System to Alleviate Oxidative Stress. J. Cancer Prev. 2016, 21, 41–47. [Google Scholar] [CrossRef]
- Ganeshpurkar, A.; Saluja, A.K. The Pharmacological Potential of Rutin. Saudi Pharm. J. 2017, 25, 149–164. [Google Scholar] [CrossRef]
- Satari, A.; Ghasemi, S.; Habtemariam, S.; Asgharian, S.; Lorigooini, Z. Rutin: A Flavonoid as an Effective Sensitizer for Anticancer Therapy; Insights into Multifaceted Mechanisms and Applicability for Combination Therapy. Evid.-Based Complement. Altern. Med. 2021, 2021, 9913179. [Google Scholar] [CrossRef]
- Gullón, B.; Lú-Chau, T.A.; Moreira, M.T.; Lema, J.M.; Eibes, G. Rutin: A Review on Extraction, Identification and Purification Methods, Biological Activities and Approaches to Enhance Its Bioavailability. Trends Food Sci. Technol. 2017, 67, 220–235. [Google Scholar] [CrossRef]
- Sasikumar, K.; Ghosh, A.R.; Dusthackeer, A. Antimycobacterial Potentials of Quercetin and Rutin against Mycobacterium Tuberculosis H37Rv. 3 Biotech 2018, 8, 427. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, A.d.C.; Fideles, S.O.M.; Reis, C.H.B.; Bellini, M.Z.; Pereira, E.d.S.B.M.; Pilon, J.P.G.; de Marchi, M.Â.; Detregiachi, C.R.P.; Flato, U.A.P.; Trazzi, B.F.d.M.; et al. Therapeutic Effects of Citrus Flavonoids Neohesperidin, Hesperidin and Its Aglycone, Hesperetin on Bone Health. Biomolecules 2022, 12, 626. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Wang, J.; Chen, Y.; Agarwal, R. Anti-Tumor-Promoting Activity of a Polyphenolic Fraction Isolated from Grape Seeds in the Mouse Skin Two-Stage Initiation-Promotion Protocol and Identification of Procyanidin B5-3’-Gallate as the Most Effective Antioxidant Constituent. Carcinogenesis 1999, 20, 1737–1745. [Google Scholar] [CrossRef]
- MedChemExpress. Hexahydrofarnesyl Acetone. Available online: https://www.medchemexpress.com/hexahydrofarnesyl-acetone.html#:~:text=COA HandlingInstructions-,Hexahydrofarnesylacetone (6%2C10%2C14-Trimethyl-2,nociceptiveandanti-inflammationactivities (accessed on 9 April 2023).
- Nakatsuji, T.; Kao, M.C.; Fang, J.Y.; Zouboulis, C.C.; Zhang, L.; Gallo, R.L.; Huang, C.M. Antimicrobial Property of Lauric Acid against Propionibacterium Acnes: Its Therapeutic Potential for Inflammatory Acne Vulgaris. J. Investig. Dermatol. 2009, 129, 2480–2488. [Google Scholar] [CrossRef]
- Islam, M.T.; Ali, E.S.; Uddin, S.J.; Shaw, S.; Islam, M.A.; Ahmed, M.I.; Chandra Shill, M.; Karmakar, U.K.; Yarla, N.S.; Khan, I.N.; et al. Phytol: A Review of Biomedical Activities. Food Chem. Toxicol. 2018, 121, 82–94. [Google Scholar] [CrossRef]
- Siswadi, S.; Saragih, G.S. Phytochemical Analysis of Bioactive Compounds in Ethanolic Extract of Sterculia quadrifida R.Br. AIP Conf. Proc. 2021, 2353, 030098. [Google Scholar]
- Choudhary, D.; Shekhawat, J.K.; Kataria, V. GC-MS Analysis of Bioactive Phytochemicals in Methanol Extract of Aerial Part and Callus of Dipterygium Glaucum Decne. Pharmacogn. J. 2019, 11, 1055–1063. [Google Scholar] [CrossRef]
- ChEBI Octadecanoic Acid. Available online: https://www.ebi.ac.uk/chebi/searchId.do?chebiId=CHEBI:28842#:~:text=A%20C18%20straight%2Dchain%20saturated,making%20cosmetics%2C%20candles%20and%20plastics (accessed on 12 April 2023).
- Tsai, F.S.; Lin, L.W.; Wu, C.R. Lupeol and Its Role in Chronic Diseases. In Drug Discovery from Mother Nature; Spring: Berlin/Heidelberg, Germany, 2016; ISBN 9783319413426. [Google Scholar]
- Gallo, M.B.C.; Sarachine, M.J. Biological Activities of Lupeol. Int. J. Biomed. Pharm. Sci. 2009, 3, 46–66. [Google Scholar] [CrossRef]
- MacAbeo, A.P.G.; Vidar, W.S.; Chen, X.; Decker, M.; Heilmann, J.; Wan, B.; Franzblau, S.G.; Galvez, E.V.; Aguinaldo, M.A.M.; Cordell, G.A. Mycobacterium Tuberculosis and Cholinesterase Inhibitors from Voacanga Globosa. Eur. J. Med. Chem. 2011, 46, 3118–3123. [Google Scholar] [CrossRef]
- El-shahaby, O.A.; El-zayat, M.M.; El-fattah, G.A.; El-hefny, M.M. Evaluation of the Biological Activity of Capparis Spinosa Var. Aegyptiaca Essential Oils and Fatty Constituents as Anticipated Antioxidant and Antimicrobial Agents. Prog. Chem. Biochem. Res. 2019, 2, 211–221. [Google Scholar] [CrossRef]
- Kalaimagal, C. Identification of Bioactive Compounds in Flower of Tabernaemontana divaricata (L.) Using Gas Chromatography–Mass Spectrometry Analysis. Asian J. Pharm. Clin. Res. 2019, 12, 129–132. [Google Scholar] [CrossRef]
- Zahumenicka, P.; Sysova, B.; Holik, A.; Fernandez, E.C. In Vitro Induced Mitotic Polyploidy in Drosera capensis L. Agric. Trop. Subtrop. 2013, 46, 107–110. [Google Scholar] [CrossRef]
- Krausko, M.; Perutka, Z.; Šebela, M.; Šamajová, O.; Šamaj, J.; Novák, O.; Pavlovič, A. The Role of Electrical and Jasmonate Signalling in the Recognition of Captured Prey in the Carnivorous Sundew Plant Drosera Capensis. New Phytol. 2017, 213, 1818–1835. [Google Scholar] [CrossRef]
- Pavlovič, A.; Krausko, M.; Libiaková, M.; Adamec, L. Feeding on Prey Increases Photosynthetic Efficiency in the Carnivorous Sundew Drosera Capensis. Ann. Bot. 2014, 113, 69–78. [Google Scholar] [CrossRef]
- McQuillan, M. Drosera Capensis. PlantZAfrica. Available online: http://pza.sanbi.org/drosera-capensis (accessed on 3 August 2021).
- Mativandlela, S.P.N.; Meyer, J.J.M.; Hussein, A.A.; Houghton, P.J.; Hamilton, C.J.; Lall, N. Activity against Mycobacterium Smegmatis and M. Tuberculosis by Extract of South African Medicinal Plants. Phyther. Res. 2008, 22, 841–845. [Google Scholar] [CrossRef]
- Botanical Online. Sundew Toxicity. Available online: https://www.botanical-online.com/en/medicinal-plants/sundew-toxicity (accessed on 10 November 2021).
- Marczak, Ł.; Kawiak, A.; Łojkowska, E.; Stobiecki, M. Secondary Metabolites in in Vitro Cultured Plants of the Genus Drosera. Phytochem. Anal. 2005, 16, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Egan, P.A.; Van Der Kooy, F. Phytochemistry of the Carnivorous Sundew Genus Drosera (Droseraceae)—Future Perspectives and Ethnopharmacological Relevance. Chem. Biodivers. 2013, 10, 1774–1790. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Zhang, J.; Chen, L.; Guo, Q.; Yang, B.; Zhang, W.; Kang, W. Anticancer Effects and Mechanisms of Action of Plumbagin: Review of Research Advances. BioMed Res. Int. 2020, 2020, 6940953. [Google Scholar] [CrossRef]
- Anand David, A.V.; Arulmoli, R.; Parasuraman, S. Overviews of Biological Importance of Quercetin: A Bioactive Flavonoid. Pharmacogn. Rev. 2016, 10, 84–89. [Google Scholar] [CrossRef]
- Sasindran, S.J.; Torrelles, J.B. Mycobacterium Tuberculosis Infection and Inflammation: What Is Beneficial for the Host and for the Bacterium? Front. Microbiol. 2011, 2, 2. [Google Scholar] [CrossRef]
- Sarkar, A.; Ghosh, S.; Shaw, R.; Patra, M.M.; Calcuttawala, F.; Mukherjee, N.; Gupta, S.K.D. Mycobacterium tuberculosis Thymidylate Synthase (ThyX) Is a Target for Plumbagin, a Natural Product with Antimycobacterial Activity. PLoS ONE 2020, 15, e0228657. [Google Scholar] [CrossRef]
- Mathew, R.; Kruthiventi, A.K.; Prasad, J.V.; Kumar, S.P.; Srinu, G.; Chatterji, D. Inhibition of Mycobacterial Growth by Plumbagin Derivatives. Chem. Biol. Drug Des. 2010, 76, 34–42. [Google Scholar] [CrossRef]
- Yadav, A.M.; Bagade, M.M.; Ghumnani, S.; Raman, S.; Saha, B.; Kubatzky, K.F.; Ashma, R. The Phytochemical Plumbagin Reciprocally Modulates Osteoblasts and Osteoclasts. Biol. Chem. 2022, 403, 211–229. [Google Scholar] [CrossRef]
- Mahapatra, A.; Mativandlela, S.P.N.; Binneman, B.; Fourie, P.B.; Hamilton, C.J.; Meyer, J.J.M.; van der Kooy, F.; Houghton, P.; Lall, N. Activity of 7-Methyljuglone Derivatives against Mycobacterium tuberculosis and as Subversive Substrates for Mycothiol Disulfide Reductase. Bioorganic Med. Chem. 2007, 15, 7638–7646. [Google Scholar] [CrossRef]
- Mbaveng, A.T.; Kuete, V. Review of the Chemistry and Pharmacology of 7-Methyljugulone. Afr. Health Sci. 2014, 14, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Maroyi, A. Review of Ethnomedicinal Uses, Phytochemistry and Pharmacological Properties of Euclea natalensis A.DC. Molecules 2017, 22, 2128. [Google Scholar] [CrossRef]
- van der Kooy, F.; Meyer, J.J.M.; Lall, N. Antimycobacterial Activity and Possible Mode of Action of Newly Isolated Neodiospyrin and Other Naphthoquinones from Euclea Natalensis. S. Afr. J. Bot. 2006, 72, 349–352. [Google Scholar] [CrossRef]
- Taheri, Y.; Suleria, H.A.R.; Martins, N.; Sytar, O.; Beyatli, A.; Yeskaliyeva, B.; Seitimova, G.; Salehi, B.; Semwal, P.; Painuli, S.; et al. Myricetin Bioactive Effects: Moving from Preclinical Evidence to Potential Clinical Applications. BMC Complement. Med. Ther. 2020, 20, 241. [Google Scholar] [CrossRef]
- Sepúlveda, L.; Ascacio, A.; Rodríguez-Herrera, R.; Aguilera-Carbó, A.; Aguilar, C.N. Ellagic Acid: Biological Properties and Biotechnological Development for Production Processes. Afr. J. Biotechnol. 2011, 10, 4518–4523. [Google Scholar] [CrossRef]
- Jones, G.; Adams, T. Pelargonium Reniforme. PlantZAfrica. Available online: http://pza.sanbi.org/pelargonium-reniforme (accessed on 4 August 2021).
- Jeiter, J.; Hilger, H.H.; Smets, E.F.; Weigend, M. The Relationship between Nectaries and Floral Architecture: A Case Study in Geraniaceae and Hypseocharitaceae. Ann. Bot. 2017, 120, 791–803. [Google Scholar] [CrossRef] [PubMed]
- Latté, K.P.; Kayser, O.; Tan, N.; Kaloga, M.; Kolodziej, H. Unusual Coumarin Patterns of Pelargonium Species Forming the Origin of the Traditional Herbal Medicine Umckaloabo. Z. Für Naturforsch. C 2000, 55, 528–533. [Google Scholar]
- Mativandlela, S.P.N.; Lall, N.; Meyer, J.J.M. Antibacterial, Antifungal and Antitubercular Activity of (the Roots of) Pelargonium Reniforme (CURT) and Pelargonium Sidoides (DC) (Geraniaceae) Root Extracts. S. Afr. J. Bot. 2006, 72, 232–237. [Google Scholar] [CrossRef]
- Kolodziej, H.; Kiderlen, A.F. In Vitro Evaluation of Antibacterial and Immunomodulatory Activities of Pelargonium Reniforme, Pelargonium Sidoides and the Related Herbal Drug Preparation EPs® 7630. Phytomedicine 2007, 14, 18–26. [Google Scholar] [CrossRef]
- Adewusi, E.A.; Afolayan, A.J. Safety Evaluation of the Extract from the Roots of Pelargonium Reniforme Curtis in Male Wistar Rats. Afr. J. Pharm. Pharmacol. 2009, 3, 368–373. [Google Scholar]
- Latté, K.P.; Kolodziej, H. Antioxidant Properties of Phenolic Compounds from Pelargonium Reniforme. J. Agric. Food Chem. 2004, 52, 4899–4902. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Zhang, L.; Liao, P.; Xiao, Z.; Zhang, F.; Sindaye, D.; Xin, Z.; Tan, C.; Deng, J.; Yin, Y.; et al. Impact of Gallic Acid on Gut Health: Focus on the Gut Microbiome, Immune Response, and Mechanisms of Action. Front. Immunol. 2020, 11, 580208. [Google Scholar] [CrossRef]
- Rehberg, N.; Omeje, E.; Ebada, S.S.; van Geelen, L.; Sureechatchayan, P.; Kassack, M.U.; Loerger, T.R.; Proksch, P.; Kalscheuer, R. 3-O-Methyl-Alkylgallates Inhibit Fatty Acid Desaturation in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2019, 63, 1–13. [Google Scholar]
- Cechinel-Zanchett, C.C.; Bolda Mariano, L.N.; Schlickmann, F.; Cechinel-Filho, V.; de Souza, P. In Vitro Effects of Two Bioactive Compounds, Gallic Acid and Methyl Gallate, on Urolithiasis. Actas Urológicas Españolas 2021, 45, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Singh, A.K.; Kumar, R.; Ganguly, R.; Rana, H.K.; Pandey, P.K.; Sethi, G.; Bishayee, A.; Pandey, A.K. Corilagin in Cancer: A Critical Evaluation of Anticancer Activities and Molecular Mechanisms. Molecules 2019, 24, 3399. [Google Scholar] [CrossRef]
- Abdulai, I.L.; Kwofie, S.K.; Gbewonyo, W.S.; Boison, D.; Puplampu, J.B.; Adinortey, M.B. Multitargeted Effects of Vitexin and Isovitexin on Diabetes Mellitus and Its Complications. Sci. World J. 2021, 2021, 6641128. [Google Scholar] [CrossRef]
- Peng, Y.; Gan, R.; Li, H.; Yang, M.; McClements, D.J.; Gao, R.; Sun, Q. Absorption, Metabolism, and Bioactivity of Vitexin: Recent Advances in Understanding the Efficacy of an Important Nutraceutical. Crit. Rev. Food Sci. Nutr. 2021, 61, 1049–1064. [Google Scholar] [CrossRef]
- Liu, S.; Lyu, Y.; Yu, S.; Cheng, J.; Zhou, J. Efficient Production of Orientin and Vitexin from Luteolin and Apigenin Using Coupled Catalysis of Glycosyltransferase and Sucrose Synthase. J. Agric. Food Chem. 2021, 69, 6578–6587. [Google Scholar] [CrossRef]
- de Jesus, C.C.M.; de Araújo, M.H.; Simão, T.L.B.V.; Lasunskaia, E.B.; Barth, T.; Muzitano, M.F.; Pinto, S.C. Natural Products from Vitex Polygama and Their Antimycobacterial and Anti-Inflammatory Activity. Nat. Prod. Res. 2022, 36, 1337–1341. [Google Scholar] [CrossRef]
- Mohammed, R.S.; Abou Zeid, A.H.; El Hawary, S.S.; Sleem, A.A.; Ashour, W.E. Flavonoid Constituents, Cytotoxic and Antioxidant Activities of Gleditsia triacanthos L. Leaves. Saudi J. Biol. Sci. 2014, 21, 547–553. [Google Scholar] [CrossRef]
- Kim, S.; Woo, E.R.; Lee, D.G. Synergistic Antifungal Activity of Isoquercitrin: Apoptosis and Membrane Permeabilization Related to Reactive Oxygen Species in Candida Albicans. IUBMB Life 2019, 71, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Mazlun, M.H.; Sabran, S.F.; Mohamed, M.; Bakar, M.F.A.; Abdullah, Z. Phenolic Compounds as Promising Drug Candidates in Tuburculosis Theraphy. Molecules 2019, 24, 2449. [Google Scholar] [PubMed]
- Çakır, D.K.; Zannou, O.; Koca, I. Scopoletin Contents and Antioxidant Properties of Some Edible Plants of Black Sea Regions. Discov. Food 2022, 2, 7. [Google Scholar] [CrossRef]
- Notten, A. Tulbaghia Simmleri. Available online: http://pza.sanbi.org/tulbaghia-simmleri (accessed on 21 April 2021).
- Takaidza, S. Phytochemical Analysis and Biological Activities of Crude Extracts from Selected Tulbhaghia Species. Ph.D. Thesis, Vaal University of Technology, Vanderbijlpark, South Africa, 2018. [Google Scholar]
- Bungu, L.; Van De Venter, M.; Frost, C. Evidence for an in Vitro Anticoagulant and Antithrombotic Activity in Tulbaghia Violacea. Afr. J. Biotechnol. 2008, 7, 681–688. [Google Scholar]
- Madike, L.N.; Takaidza, S.; Pillay, M. Preliminary Phytochemical Screening of Crude Extracts from the Leaves, Stems, and Roots of Tulbaghia Violacea. Int. J. Pharmacogn. Phytochem. Res. 2017, 9, 1300–1308. [Google Scholar] [CrossRef]
- Soyingbe, O.S.; Oyedeji, A.O.; Basson, A.K.; Singh, M.; Opoku, A.R. Chemical Composition, Antimicrobial and Antioxidant Properties of the Essential Oils of Tulbaghia Violacea Harv L.F. Afr. J. Microbiol. Res. 2013, 7, 1787–1793. [Google Scholar] [CrossRef]
- Motsei, M.L. Screening of Traditionally Used South African Plants against Candida albicans. Master’s Thesis, University of Natal, Pietermaritzburg, South Africa, 2003. [Google Scholar]
- Abd’quadri-Abojukoro, A.N.; Nkadimeng, S.M.; Mcgaw, L.J.; Nsahlai, I. V Phytochemical Composition and Cytotoxicity of Ethanolic Extracts of Some Selected Plants. J. Appl. Anim. Res. 2022, 50, 656–665. [Google Scholar] [CrossRef]
- Burton, S.G.; Kaye, P.T. Isolation and Characterisation of Sulphur Compounds from Tulbaghia Violacea. Planta Med. 1992, 58, 295–296. [Google Scholar] [CrossRef]
- Moodley, K.; Joseph, K.; Naidoo, Y.; Islam, S.; Mackraj, I. Antioxidant, Antidiabetic and Hypolipidemic Effects of Tulbaghia violacea Harv. (Wild Garlic) Rhizome Methanolic Extract in a Diabetic Rat Model. BMC Complement. Altern. Med. 2015, 15, 408. [Google Scholar] [CrossRef]
- Olorunnisola, O.S.; Bradley, G.; Afolayan, A.J. Chemical Composition, Antioxidant Activity and Toxicity Evaluation of Essential Oil of Tulbaghia Violacea Harv. J. Med. Plants Res. 2012, 6, 2340–2347. [Google Scholar] [CrossRef]
- Takaidza, S.; Kumar, A.M.; Ssemakalu, C.C.; Natesh, N.S.; Karanam, G.; Pillay, M. Anticancer Activity of Crude Acetone and Water Extracts of Tulbaghia Violacea on Human Oral Cancer Cells. Asian Pac. J. Trop. Biomed. 2018, 8, 456–462. [Google Scholar] [CrossRef]
- Veluthoor, S.; Badi, P.; Mukharjee, K.; Mandal, V. Phytochemicals: In Pursuit of Antitubercular Drugs. In Studies in Natural Products Chemistry; Elsevier B.V.: Amsterdam, The Netherlands, 2012; Volume 38, pp. 417–463. ISBN 9780444595300. [Google Scholar]
- Nayak, N.; Bajpai, M.; Razdan, B. Plumbagin Analogs-Synthesis, Characterization, and Antitubercular Activity. J. Adv. Pharm. Technol. Res. 2014, 5, 28–32. [Google Scholar] [CrossRef] [PubMed]
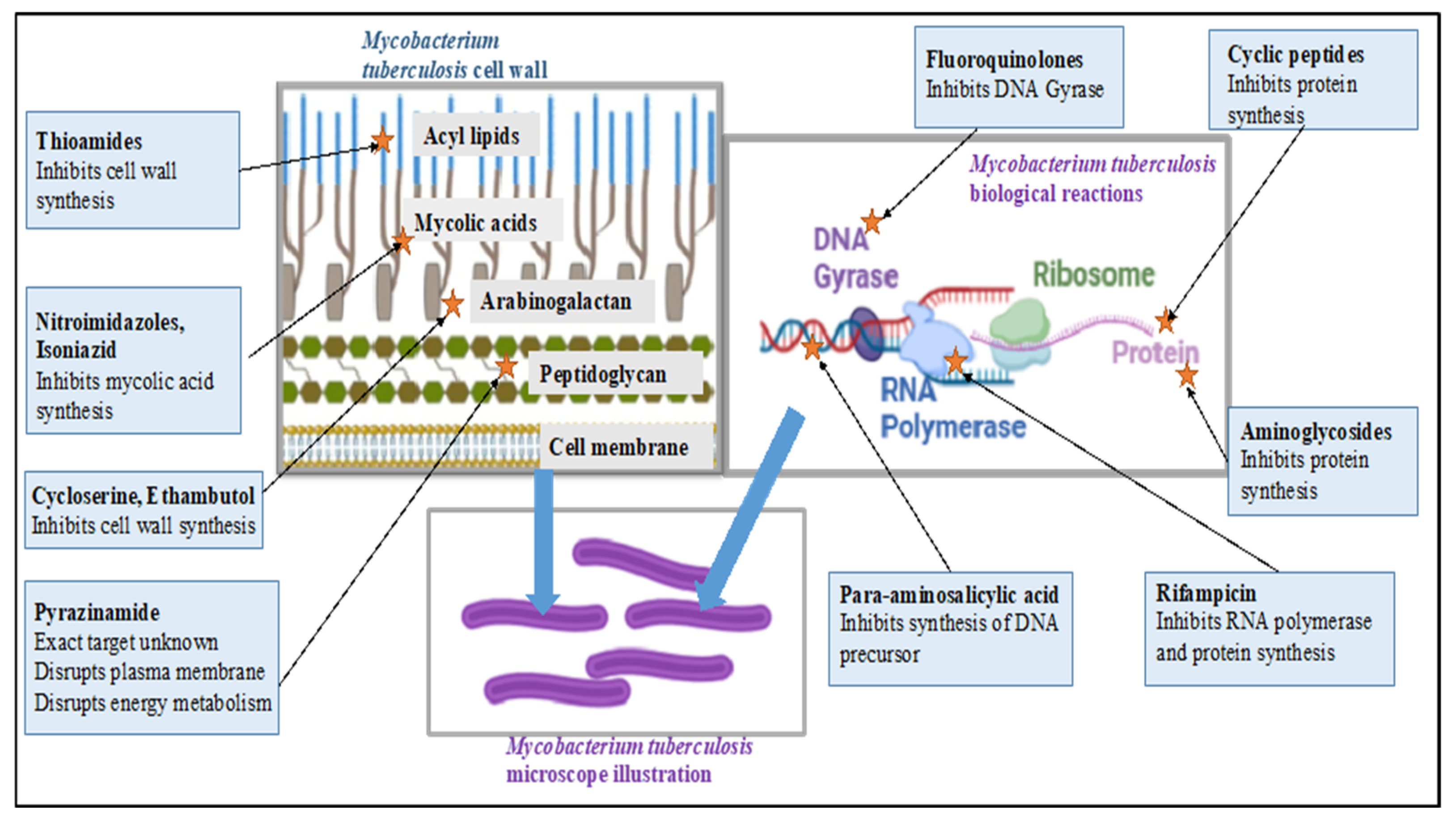




| Regimen | Initial Phase | Continuation Phase | Doses | ||
|---|---|---|---|---|---|
| Drugs | Dosing Frequency | Drugs | Dosing Frequency | ||
| 1 | Isoniazid Rifampicin Pyrazinamide Ethambutol | 7 d/w or 5 d/w (8 weeks) | Isoniazid Rifampicin | 7 d/w or 5 d/w (18 weeks) | 130–182 doses over 26 weeks |
| Isoniazid Rifampicin | 2 d/w (18 weeks) | 76–92 doses over 26 weeks | |||
| Isoniazid Rifapentine | ** 1 d/w (18 weeks) | 58–74 doses over 26 weeks | |||
| 2 | Isoniazid Rifampicin Pyrazinamide Ethambutol | 7 d/w for 2 weeks then 2 d/w for 6 weeks or 5 d/w then 2 d/w for 6 weeks | Isoniazid Rifampicin | 2 d/w (18 weeks) | 58–62 doses over 26 weeks |
| Isoniazid Rifapentine | ** 1 d/w (18 weeks) | 40–44 doses over 26 weeks | |||
| 3 | Isoniazid Rifampicin Pyrazinamide Ethambutol | 3 times/week (18 weeks) | Isoniazid Rifampicin | 54 doses 3 times/week (18 weeks) | 78 doses over 26 weeks |
| 4 | Isoniazid Rifampicin Ethambutol | 7 d/w for 8 weeks or 5 d/w for 8 weeks | Isoniazid Rifampicin | 7 d/w for (31 weeks) | 195–273 doses over 39 weeks |
| Isoniazid Rifampicin | 2 times/week (31 weeks) | 102–118 doses over 39 weeks | |||
| Dose mg/kg (Maximum Doses in mg) | |||||||
|---|---|---|---|---|---|---|---|
| Drug | Group | Daily | Once Weekly | Twice Weekly | Thrice Weekly | ||
| Isoniazid | Adults | 5 (300) | 15 (900) | 15 (900) | 15 (900) | ||
| Children | 10–15 (300) | 20–30 (900) | |||||
| Rifampicin | Adults | 10 (600) | 10 (600) | ||||
| Children | 10 (600) | 10 (600) | |||||
| Rifabutin | Adults | 5 (300) | 5 (300) | ||||
| Children | Unknown | ||||||
| Rifapentine | Adults | 10 (600) continuation phase | |||||
| Children | No approval for use in group | ||||||
| Pyrazinamide | Adults | Weight (kg) | 40–55 | 18.2–25 (1000) | 36.4–50 (2000) | 27.3–37.5 (1500) | |
| 56–75 | 20–26.8 (1500) | 40–53.6 (3000) | 33.3–44.6 (2500) | ||||
| 76–90 | 22.2–26.3 (2000) | 44.4–52.6 (4000) | 33.3–39.5 (3000) | ||||
| Children | 15–30 (2000) | 50 (2000) | |||||
| Ethambutol | Adults | Weight (kg) | 40–55 | 14.5–20 (800) | 36.4–50 (2000) | 21.8–30 (1200) | |
| 56–74 | 16–21.4 (1200) | 37.3–50 (2800) | 26.7–35.7 (2000) | ||||
| 76–90 | 17.8–21.1 (1600) | 44.4–52.6 (4000) | 26.7–31.6 (2400) | ||||
| Children | 15–20 (1000) | 50 (2500) | |||||
| Compound Number | Structure | Compound Name | Phytochemical | Bioactivity | Antimycobacterium M. tuberculosis | References |
|---|---|---|---|---|---|---|
| 1 | 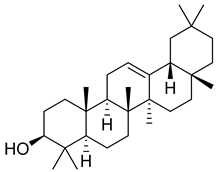 | β-amyrin | Triterpene | Anti-inflammatory, antinociceptive, antioxidant, antipruritic, gastroprotective, hepatoprotective | M. tuberculosis H37Rv MIC > 200 mg/L | [25,26,27] |
| 2 | 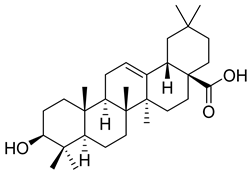 | Oleanolic acid | Triterpenoid | Antioxidant, anti-tumor, anti-inflammatory, anti-diabetic, anti-microbial, hepatoprotective | M. tuberculosis H37Rv MIC 100 mg/L | [25,28] |
| 3 | 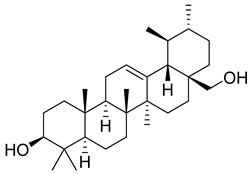 | Uvaol | Triterpene | Antioxidant, anti-inflammatory, vasodilator | M. tuberculosis H37Rv MIC > 200 mg/L | [25,29,30] |
| 4 |  | Epicatechin | Flavanol | Neuroprotective effects, blood-pressure-lowering effect | M. tuberculosis H37Rv MIC > 200 mg/L | [25,31,32] |
| 5 | 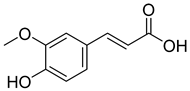 | Ferulic acid | Phenolic | Anti-inflammatory, antibacterial, anticancer, anti-arrhythmic, antithrombotic | [33,34] | |
| 6 | 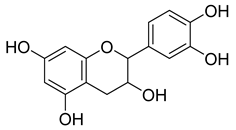 | Catechin | Polyphenolic | Antioxidant, ultraviolet (UV) protection, antimicrobial, anti-allergy, anti-inflammatory, antiviral, anticancer | M. tuberculosis H37Rv MIC 200 mg/L | [25,35,36] |
| 7 | 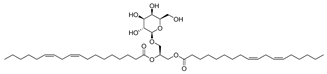 | Monogalactosyldiacylglycerol (MGDG) | Galactolipid | Anti-inflammatory | M. tuberculosis H37Rv MIC > 200 mg/L | [25,37,38] |
| 8 |  | Citric acid | Phenolic | Antimicrobial | NR | [39,40] |
| 9 |  | Malic acid | Phenolic | Antibacterial | NR | [40,41] |
| 10 | 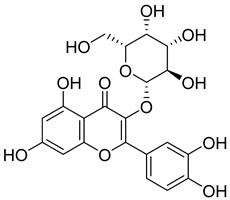 | Hyperoside | Flavonoid | Antioxidant, analgesic, anticancer, neuroprotective, kidney protective | NR | [42,43] |
| 11 | 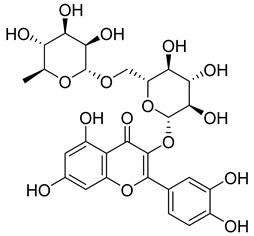 | Rutin | Flavonoid | Anticancer, anti-inflammatory, neuroprotective, antiproliferative, antimetastatic, antioxidant, antimicrobial, antiallergy, antidiabetic | M. tuberculosis H37Rv MIC 25 µg/mL | [44,45,46,47] |
| 12 | 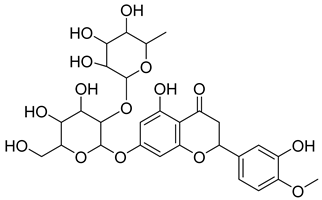 | Neohesperidin | Flavonoids | Anti-inflammatory | NR | [48] |
| 13 | 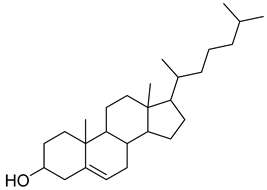 | procyanidin B5 | Polyphenol | Anti-inflammatory, anti-arthritic, anti-allergen | NR | [49] |
| 14 |  | 2-pentadecanone, 6,10,14-trimethyl | Sesquiterpene | Antibacterial, anti-nociceptive, anti-inflammatory | NR | [50] |
| 15 |  | 7-Methyl-Z-tetradecen-1-ol acetate | NR | NR | NR | |
| 16 |  | Dodecanoic acid | Fatty acid | Antibacterial | NR | [51] |
| 17 | 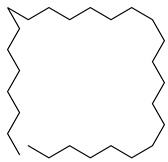 | Heptacosane | Alkane | NR | [23] | |
| 18 | 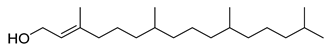 | Phytol | Diterpene | Anxiolytic, cytotoxic, antioxidant, antinociceptive, antimicrobial, anti-inflammatory, immune modulating | NR | [52,53] |
| 19 |  | n-Hexadecanoic acid | Fatty acid | Antioxidant, hypocholesterolemic, nematicide, pesticide | NR | [53] |
| 20 | 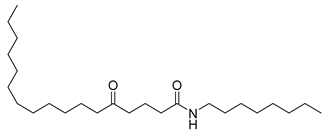 | n-Octyl-5-oxoheptadecanamide | Amide | NR | NR | [23] |
| 21 |  | Dibutyl phthalate | NR | NR | NR | [23] |
| 22 |  | 2-Tertbutyl cyclohexylpropylphosphonofluoridate | NR | NR | NR | [23] |
| 23 | 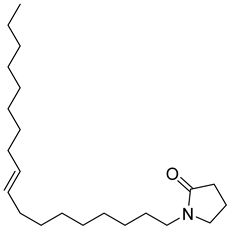 | 2-Pyrrolidinone, 1-(9-octadecenyl) | NR | NR | NR | [23] |
| 24 | 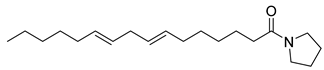 | Pyrrolidine, 1-(1-oxo-7,10-hexadecadienyl | NR | NR | NR | [23] |
| 25 |  | Nonacosane | Alkane | NR | NR | [23] |
| 26 |  | 4,8,12,16-Tetramethylheptadecan-4-olide | Terpene | NR | NR | [54] |
| 27 |  | Octadecanoic acid | Fatty acid | Used to harden soaps, soften plastics, and make cosmetics, candles, and plastics | NR | [55] |
| 28 | 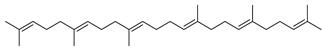 | 2,6,10,14,18,22-Tetracosahexaene, 2,6,10,15,19,23-hexamethyl | NR | NR | NR | [23] |
| 29 | 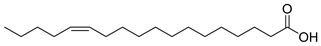 | cis-13-Octadecenoic acid | Fatty acid | NR | NR | [23] |
| 30 | 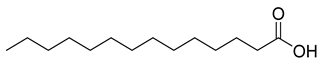 | Tetradecanoic acid | Fatty acid | NR | NR | [54] |
| 31 | 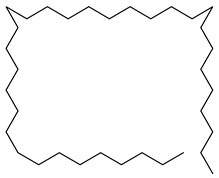 | Tetratriacontane | Alkane | NR | NR | [23] |
| 32 | 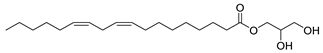 | 9,12-Octadecadienoic acid (Z,Z)-2 3-dihydroxypropyl ester | Ester | NR | NR | [23] |
| 33 | 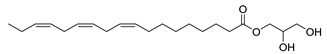 | 9,12,15-Octadecatrienoic acid, 2,3-dihydroxypropyl ester, (Z,Z,Z) | Ester | NR | NR | [23] |
| 34 |  | Eicosanoic acid | Fatty acid | NR | NR | [23] |
| 35 | 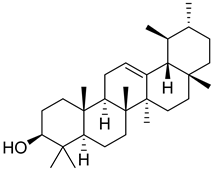 | α-Amyrin | Triterpene | NR | NR | [26] |
| 36 |  | Lupeol | Triterpene | Antiprotozoal, antimicrobial, anti-inflammatory, antioxidant, antidiabetic, antitumor, chemopreventive, wound healing | M. tuberculosis H37Rv MIC > 20 µg/mL | [56,57,58] |
| 37 |  | 9,19-Cyclolanostan-24-en-3-ol, acetate, (3β) | NR | NR | NR | [23] |
| 38 | 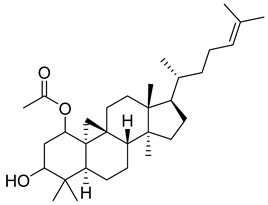 | 9,19-Cyclolanost-24-en-3-ol, acetate, (3β) | NR | NR | NR | [23] |
| 39 |  | D-α-Tocopherol (Vitamin E) | Alpha tocopherol | Antioxidant, antidiabetic, UV irradiation protections | NR | [53,59] |
| 40 | 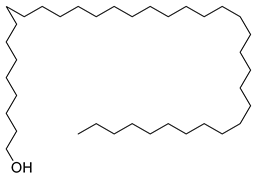 | 1-Heptatriacotanol | Alcohol [60] | Antioxidant, anti-inflammatory hypocholesterolemic, antimicrobial, anticancer [60] | NR | [23] |
| 41 | 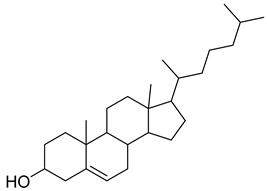 | 17-(1,5-Dimethylhexyl)-10,13-dimethyl- 2,3,4,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-3-ol | NR | NR | NR | [23] |
| 42 | 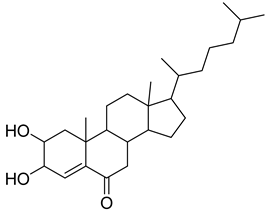 | 17-(1,5-Dimethylhexyl)-2,3-dihydroxy-10,13-dimethyl- 1,2,3,7,8,9,10,11,12,13,14,15,16,17-tetradecahydrocyclopenta[a]phenanthren-6-one | NR | NR | NR | [23] |
| 43 | 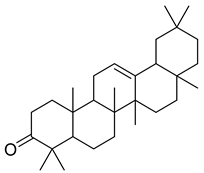 | 4,4,6a,6b,8a,11,11,14b-Octamethyl-1,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,14,14a,14b-octadecahydro-2H-picen-3-one | NR | NR | NR | [23] |
| Plant Part | Uses | Extraction | Biological Effect | Phytochemistry | References | ||
|---|---|---|---|---|---|---|---|
| Traditional | Method | Type of Extract | Analysis/Profile | Bioactive Components | |||
| Leaves | Fever, TB | Successive extraction with Ethanol | Ethanol extract | Ethanol leaf extract showed activity against M. smegmatis with MIC value of 3.125 mg/mL with isoniazid control MIC of 2 × 104 mg/mL No activity against M. tuberculosis, control Ciprofloxacin MIC of 0.156 mg/mL and MBC of 0.312 mg/mL [65] | NR | Flavonoids | [17,65] |
| Soxhlet (dried sample) Sonication (fresh sample) [67] | Methanolic extract | NR | NR | Plumbagin (44) | [67] | ||
| Compound Number | Structure | Compound Name | Phytochemical | Bioactivity | Antimycobacterium M. tuberculosis | Mechanism of Action | References |
|---|---|---|---|---|---|---|---|
| 44 |  | Plumbagin | Naphthoquinone | Antioxidant, anti-inflammatory, anticancer, antimicrobial, neuroprotective, antidiabetic, anti-atherosclerosis, analgesic | M. tuberculosis H37Rv MIC 21.3 µM MIC 4 µg/ml | Kills mycobacterial cells primarily by targeting ThyX, an enzyme required for their survival | [69,72,73,74] |
| 45 | 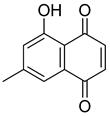 | 7-Methyljuglone | Naphthoquinone | Antifungal, antimicrobial, antitubercular, antiviral, anticancer | M. tuberculosis H37Rv MIC 0.50 µg/mL | NR | [75,76] |
| 46 |  | Mamegakinone | Naphthoquinone | NR | M. tuberculosis H37Rv MIC 100.00 µg/mL | NR | [77,78] |
| 47 |  | Neodiospyrin | Naphthoquinone | NR | M. tuberculosis H37Rv MIC 10.00 µg/mL | NR | [75,77] |
| 48 |  | Quercetin | Flavonoid | Anti-inflammatory, antibacterial, antiviral, anticancer, neurodegenerative disorders, cardiovascular disease prevention, anti-allergy | M. tuberculosis H37Rv MIC 6.25 µg/mL [47] | Inhibits subunit B of DNA gyrase and isocitratelyase | [47,70] |
| 49 | 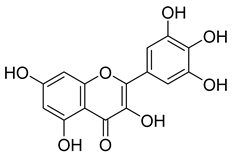 | Myricetin | Flavonol | Antimicrobial, antioxidant, neurobiological activity, antidiabetic, anticancer, immunomodulatory, antihypertensive, cardioprotective, wound healing | M. tuberculosis H37Rv MIC 50.00 µg/mL | NR | [79] |
| 50 |  | Leucocyanidin | Flavonoid | NR | NR | NR | [68] |
| 51 | 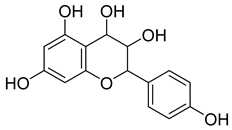 | Leucopelargonidin | NR | NR | NR | NR | [68] |
| 52 | 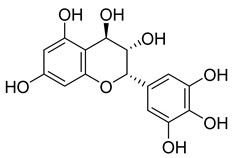 | Leucodelphinidin | NR | NR | NR | NR | [68] |
| 53 | 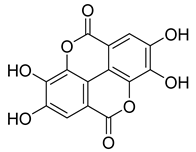 | Ellagic acid | Polyphenol | Anti-mutagenic, antimicrobial, anticancer, HIV inhibition | NR | NR | [80] |
| Plant Part | Uses | Extraction | Biological Effect | Phytochemistry | References | ||
|---|---|---|---|---|---|---|---|
| Traditional | Method | Type of Extract | Analysis/Profile | Bioactive Components | |||
| Tuber | Cough, TB | - | Ethanol, acetone, chloroform | Acetone, chloroform and ethanol root extracts showed activity against M. tuberculosis with MIC value of 10.3 mg/mL | NR | Scopoletin (67) | [17] |
| Roots | Cough, TB, diarrhea | Acetone, chloroform and ethanol Acetone and ethanol extract | Extracts inhibitory activity against drug-sensitive M. tuberculosis at 5 mg/mL Extracts active against fungal pathogens at 5 mg/mL | NR | NR | [84] | |
| NR | NR | NR | NR | Antioxidant activity with IC50 ranging from 2.6 to 32.9 µM | NT | Gallic acid (54), methyl gallate (55), glucogallin (56), corilagin (57), vitexin (58), isovitexin (59), orientin (60), isoorientin (61), vitexin 2″-gallate (62), sovitexin 2″-gallate, orientin 2″-gallate (66), isoorientin 2″-gallate, quercetin (63), isoquercitrin (64), and rutin (65) | [87] |
| Compound Number | Structure | Compound Name | Phytochemical Class | Bioactivity | Antimycobacterium M. tuberculosis | Mechanism of Action | Reference |
|---|---|---|---|---|---|---|---|
| 54 |  | Gallic acid | Phenolic acid | Antioxidant, antimicrobial, anticancer, anti-inflammatory | M. tuberculosis H37Rv MIC90 100 µM | NR | [79,88,89] |
| 55 |  | Methyl gallate | Phenolic | Antioxidant, anti-inflammatory, antimicrobial, diuretic | NR | NR | [90] |
| 56 | 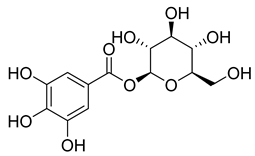 | Glucogallin | - | - | NR | NR | [87] |
| 57 |  | Corilagin | Polyphenol | Anticancer | NR | NR | [91] |
| 58 |  | Vitexin | Mono-C-glycoflavone | Antidiabetic, antioxidant, anti-inflammatory, anticancer, antimicrobial, neuroprotective, cardioprotective | NR | NR | [92,93] |
| 59 |  | Isovitexin | Mono-C-glycoflavone | Antidiabetic | NR | NR | [92] |
| 60 | 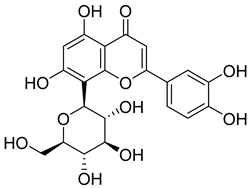 | Orientin | Flavone | Anti-inflammatory, antioxidant, anticancer, anti-diabetes, analgesic | M. tuberculosis H37Rv MIC50 23.4 ± 1.2 µg/mL (52.1 µM) | NR | [94,95] |
| 61 | 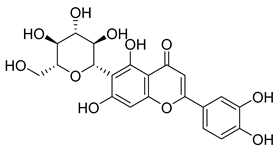 | Isoorientin | Flavonoid | Anticancer | NR | NR | [96] |
| 62 | 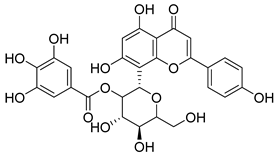 | Vitexin 2″-O-gallate | NR | NR | NR | NR | [87] |
| 63 | 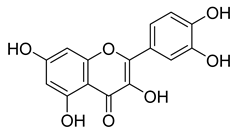 | Quercetin | Flavonoid | Anti-inflammatory, antibacterial, antiviral, anticancer, neurodegenerative disorders, cardiovascular disease prevention, anti-allergy | M. tuberculosis H37Rv MIC 6.25 µg/ml | Inhibits subunit B of DNA gyrase, inhibits β-ketoacyl ACP synthase III involved in the synthesis of mycolic acid | [47,70] |
| 64 | 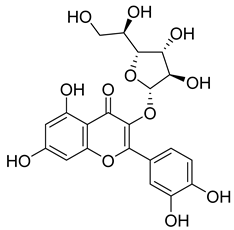 | Isoquercitrin | Flavonoid | Anti-inflammatory, anti-allergy, anti-hyperlipidemic, antioxidant, antifungal | M. tuberculosis H37Rv MIC 0 µg/mL | NR | [97,98] |
| 65 | 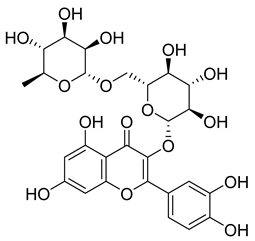 | Rutin | Flavonoid | Anticancer, anti-inflammatory, neuroprotective, antiproliferative, antimetastatic, antioxidant, antimicrobial, antiallergy, antidiabetic | M. tuberculosis H37Rv MIC 25 µg/mL | NR | [44,45,46,47] |
| 66 | 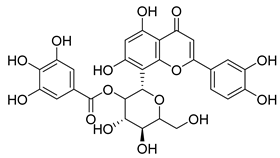 | Orientin 2-O-gallate | Flavone | NR | NR | NR | [94] |
| 67 | 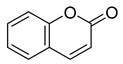 | Scopoletin | Coumarin | NR | M. tuberculosis H37Rv MIC ≥ 100 µg/mL | NR | [98,99] |
| Plant Part | Uses | Extraction | Biological Effect | Phytochemistry | References | ||
|---|---|---|---|---|---|---|---|
| Traditional | Method | Type of Extract | Analysis/Profile | Bioactive Components | |||
| Rhizome | Asthma, cold, fever, chest complaints, cough, influenza, sinusitis, TB, lung ulceration [17] | Maceration | Methanolic extract | NR | GC-MS | Beta-1,5-O-Dibenzoyl-ribofuranose (81), 4-Methoxybenzaldehyde (82), Oleyl alcoho, trifluoroacetate (83), Disulfide, bis(2-sulfhydrylethyl)- (84), Benzene, 1-methyl-4-(methylthio)- (85), 2,4-Dithiapentane (73), n-propyl 9,12-octadecadienoate (87), Methyl 5,13-Docosadienoate (89) | [17,108,109] |
| Soxhelt extraction | Hexane extract | NR | IR, MS, NMR | Asymmetrically substituted novel dialkyl sulphone, deoxygenated analog of dialkyl sulphone, putative alkyl thiosulphinates | [107] | ||
| Hydro-distillation | Essential oil | Antioxidant activity, cytotoxic effect, alternative source of anticancer, antibiotic, and antimicrobial agents [109] | GC-MS | Dimethy trisulfide (86) Dimethy disulfide, methyl (methylthio) meth 2,4-dithiapentane (Methylthio) acetic acid (88) (Methylthio) acetic acid, 2-(methylthiol) ethanol, propanitrile, 3-(methylthio)- 2,4dithiapntane,bis-(methlythio), disulfide | [109] | ||
| Bulbs | Enema for stomach problems | - | Aqueous extract | Antifungal activity against Candida albicans with an MIC value of 3.25 mg/mL | NR | NR | [13,105] |
| Leaves | Esophagus cancer | Maceration/homogenized Boiling | Acetone, Water | Ability to scavenge free radicals, IC50 for acetone extract at 207.33 µg/mL (68%) and IC50 for water extract at 168.88 µg/mL (73%) Anticancer effect, acetone extract indicated a dose-dependent inhibitory effect on human oral cancer cells, IC50 for acetone at 0.2 mg/mL and IC50 for aqueous extract at 1 mg/mL | NR | NR | [13,110] |
| Compound Number | Structure | Compound Name | Phytochemical/Class | References |
|---|---|---|---|---|
| 68 |  | Chlorodifluoro acetamide | Acetic amide | [104] |
| 69 | 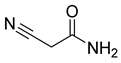 | Acetamide, 2-cyano | Acetic amide | [104] |
| 70 | 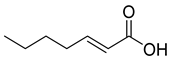 | (E)-2-heptenoic acid | Fatty acid | [104] |
| 71 | 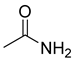 | Acetamide | Acetic amide | [104] |
| 72 | 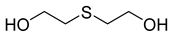 | Thiodiglycol | Organosulfur compound | [104] |
| 73 |  | 2,4-Dithiapentane | Organosulfur compound | [104] |
| 74 | 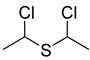 | Chloromethyl sulfide | Sulfur compound | [104] |
| 75 |  | Phthalic acid | Dicarboxylic acid | [104] |
| 76 | 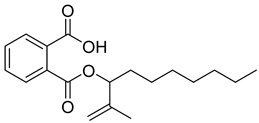 | Phthalic acid, heptyl2-methylallyl ester | Dicarboxylic acid ester | [104] |
| 77 | 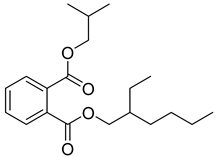 | Phthalic acid 2-ethylhexyl isobutyl ester | Dicarboxylic acid ester | [104] |
| 78 |  | Nonadecane | Alkane | [104] |
| 79 |  | Heptacosane | Alkane | [104] |
| 80 |  | Tetracosane | Alkane | [104] |
| 81 | 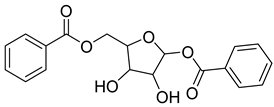 | Beta-1,5-O-Dibenzoyl-ribofuranose | Pentose | [108] |
| 82 |  | 4-Methoxybenzaldehyde | Aldehyde | [108] |
| 83 | 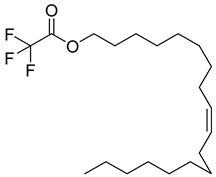 | Oleyl alcohol, trifluoroacetate | Ester | [108] |
| 84 | 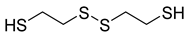 | Disulfide, bis(2-sulfhydrylethyl)- | Organosulfur compound | [108] |
| 85 | 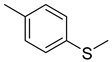 | Benzene, 1-methyl-4-(methylthio)- | Sulfur compound | [108] |
| 86 |  | Dimethyl trisulfide | Organosulfur compound | [108] |
| 87 | 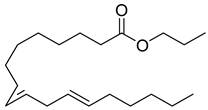 | n-propyl 9,12-octadecadienoate | Linoleic acid | [108] |
| 88 | 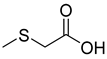 | (Methylthio) acetic acid | Acetic acid | [108] |
| 89 | 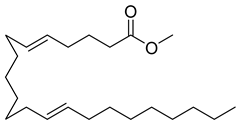 | Methyl 5,13-Docosadienoate | Ester | [108] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mamabolo, M.P.; Tembeni, B.; Siwe Noundou, X.; Mncwangi, N.P. Selected Medicinal Plants Used in the Treatment and Management of Tuberculosis and Related Symptoms in South Africa. Pharmaceuticals 2025, 18, 513. https://doi.org/10.3390/ph18040513
Mamabolo MP, Tembeni B, Siwe Noundou X, Mncwangi NP. Selected Medicinal Plants Used in the Treatment and Management of Tuberculosis and Related Symptoms in South Africa. Pharmaceuticals. 2025; 18(4):513. https://doi.org/10.3390/ph18040513
Chicago/Turabian StyleMamabolo, Makosha P., Babalwa Tembeni, Xavier Siwe Noundou, and Nontobeko P. Mncwangi. 2025. "Selected Medicinal Plants Used in the Treatment and Management of Tuberculosis and Related Symptoms in South Africa" Pharmaceuticals 18, no. 4: 513. https://doi.org/10.3390/ph18040513
APA StyleMamabolo, M. P., Tembeni, B., Siwe Noundou, X., & Mncwangi, N. P. (2025). Selected Medicinal Plants Used in the Treatment and Management of Tuberculosis and Related Symptoms in South Africa. Pharmaceuticals, 18(4), 513. https://doi.org/10.3390/ph18040513






