Cellular, Molecular and Proteomic Characteristics of Early Hepatocellular Carcinoma
Abstract
:1. Introduction
2. Materials and Methods
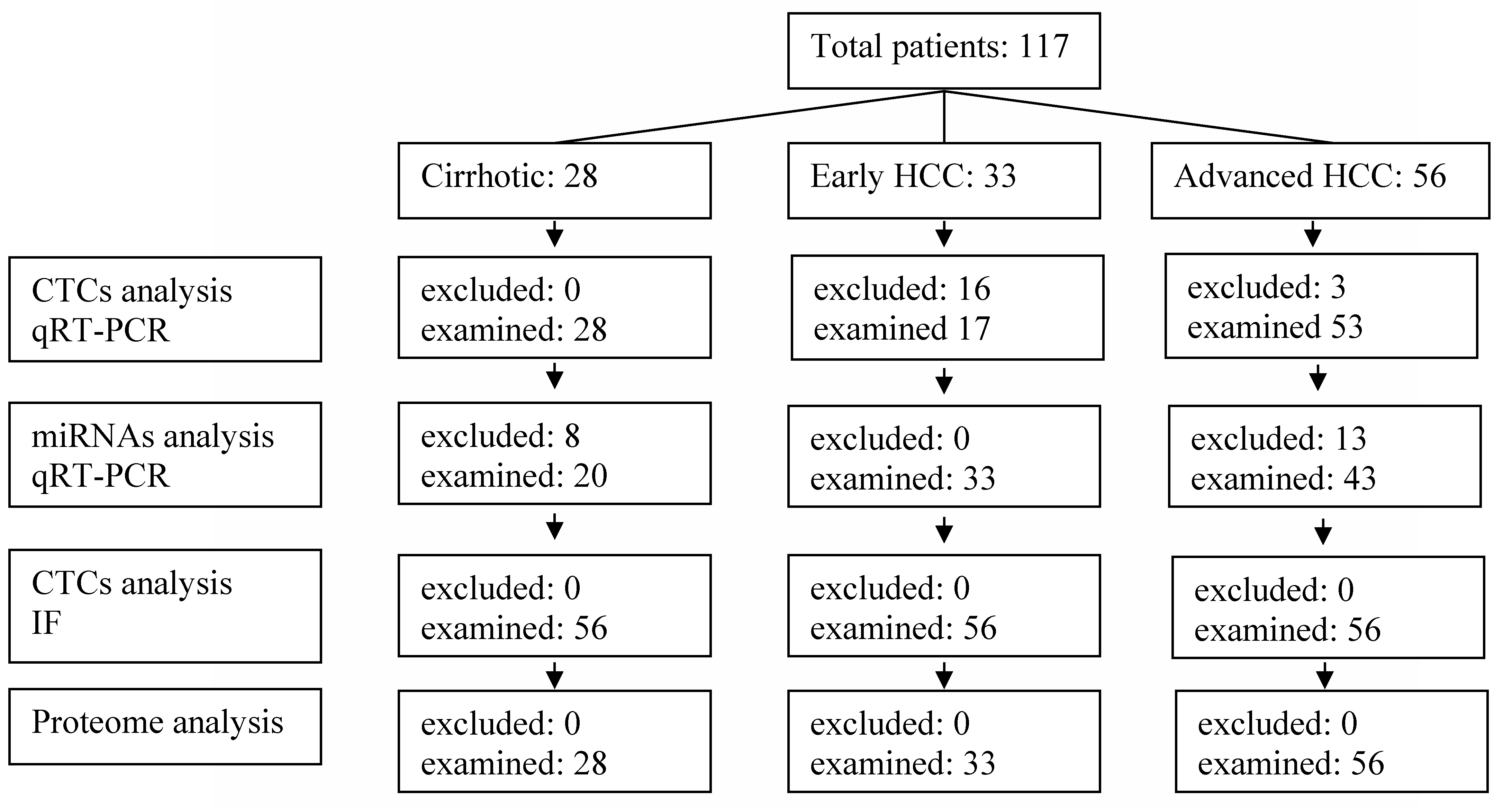
2.1. Patient Selection
2.2. CTCs
2.3. MicroRNAs
2.4. cDNA Synthesis and Real-Time PCR
2.5. Immunofluorescence Staining (IF)
2.6. Proteome Analysis
Liquid Chromatography Mass Spectrometry (LC MS)
2.7. Statistical Analysis
3. Results
3.1. CTCs
3.2. MiRNAs
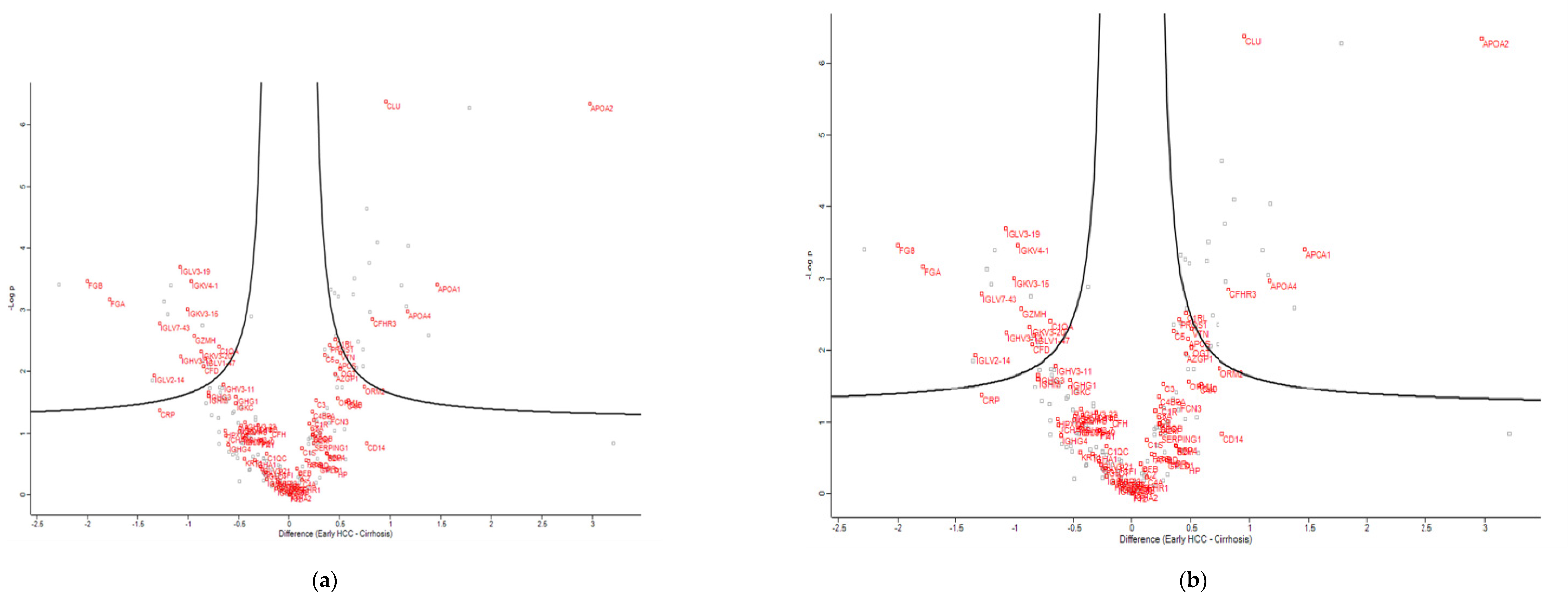
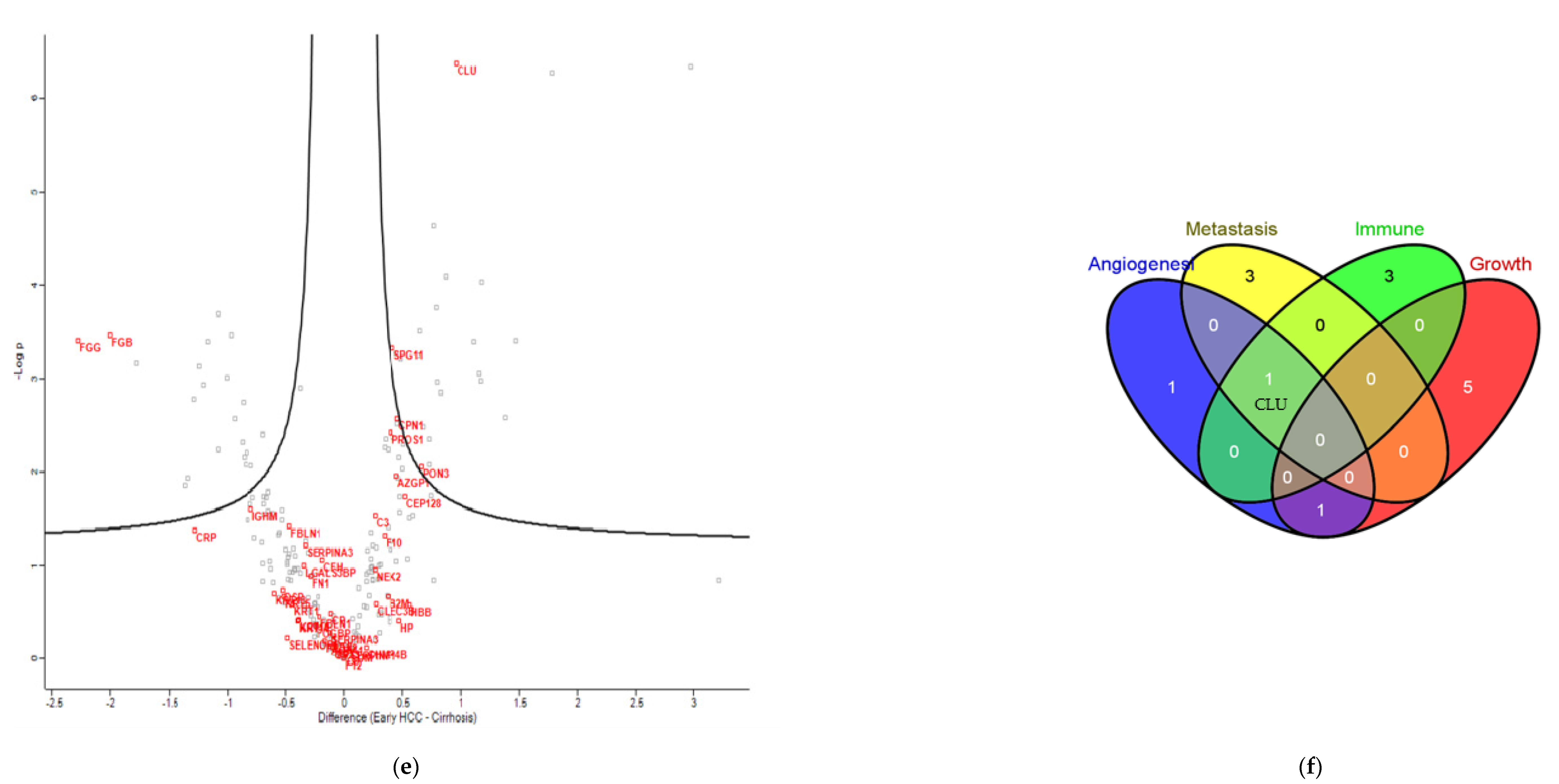
3.3. Proteomics
3.4. Clinical Significance of ApoA2, APOC3 and mir-200b
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
List of Abbreviations
| Hepatocellular carcinoma | (HCC) |
| microRNAs | (miRNAs) |
| Circulating tumor cells | (CTCs) |
| Epithelial cell adhesion molecule | (EPCAM) |
| Alpha fetoprotein | (aFP) |
| Surface major vault protein | (sMVP) |
| Chronic hepatitis B virus | (HBV) |
| Hepatitis C virus | (HCV) |
| Non-alcoholic steatohepatitis | (NASH) |
| Erythrocyte lysis buffer | (ELB) |
| Quantitative real-time polymerase chain reaction | (qRT-PCR) |
| Immunofluorescence staining | (IF) |
| 4′,6-diamidino-2-phenylindole | (DAPI) |
| Liquid chromatography mass spectrometry | (LC MS) |
| Gas phase fractionation | (GFP) |
| Computed tomography/magnetic resonance imaging | (CT/MRI) |
| Matrix metalloproteinase 2/9 | (MMP2/9) |
| C-X-C motif chemokine receptor 4 | (CXCR4) |
| Apolipoprotein | (APO) |
| High-density lipoprotein | (HDL) |
| Triglyceride-rich low-density lipoprotein | (VLDL) |
References
- World Health Organisation. Projections of Mortality Causes of Death, 2016 to 2060. Available online: http://www.who.int/healthinfo/global_burden_disease/projection/en/ (accessed on 2 February 2022).
- Villanueva, A. Hepatocellular Carcinoma. The New Engl. J. Med. 2019, 380, 1450–1462. [Google Scholar] [CrossRef] [Green Version]
- Llovet, J.M.; Kelley, R.K.; Villanueva, A.; Singal, A.G.; Pikarsky, E.; Roayaie, S.; Lencioni, R.; Koike, K.; Zucman-Rossi, J.; Finn, R.S. Hepatocellular carcinoma. Nat. Rev. Dis. Prim. 2021, 7, 6. [Google Scholar] [CrossRef]
- Akinyemiju, T.; Abera, S.; Ahmed, M.; Alam, N.; Alemayohu, M.A.; Allen, C.; Al-Raddadi, R.; Alvis-Guzman, N.; Amoako, Y.; Artaman, A.; et al. The Burden of Primary Liver Cancer and Underlying Etiologies From 1990 to 2015 at the Global, Regional and National Level: Results From the Global Burden of Disease Study 2015. JAMA Oncol. 2017, 3, 1683–1691. [Google Scholar] [PubMed]
- Xie, Y. Hepatitis B Virus-Associated Hepatocellular Carcinoma. Adv. Exp. Med. Biol. 2017, 1018, 11–21. [Google Scholar] [PubMed]
- Harrod, E.; Moctezuma-Velazquez, C.; Gurakar, A.; Ala, A.; Dieterich, D.; Saberi, B. Management of concomitant hepato-cellular carcinoma and chronic hepatitis C: A review. Hepatoma Res. 2019, 5, 28. [Google Scholar]
- Méndez-Sánchez, N.; Valencia-Rodríguez, A.; Coronel-Castillo, C.E.; Qi, X. Narrative review of hepatocellular carcinoma: From molecular bases to therapeutic approach. Dig. Med. Res. 2021, 4, 21037. [Google Scholar] [CrossRef]
- Wang, G.; Zhu, S.; Li, X. Comparison of values of CT and MRI imaging in the diagnosis of hepatocellular carcinoma and analysis of prognostic factors. Oncol. Lett. 2018, 17, 1184–1188. [Google Scholar] [CrossRef] [Green Version]
- Tang, Y.; Xiao, G.; Shen, Z.; Zhuang, C.; Xie, Y.; Zhang, X.; Yang, Z.; Guan, J.; Shen, Y.; Chen, Y.; et al. Noninvasive Detection of Extracellular pH in Human Benign and Malignant Liver Tumors Using CEST MRI. Front. Oncol. 2020, 10, 578985. [Google Scholar] [CrossRef]
- Lee, H.M.; Joh, J.W.; Seong, C.G.; Kim, W.-T.; Kim, M.K.; Choi, H.S.; Kim, S.Y.; Jang, Y.-J.; Sinn, D.H.; Choi, G.S.; et al. Cell-surface major vault protein promotes cancer progression through harboring mesenchymal and intermediate circulating tumor cells in hepatocellular carcinomas. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.-N.; Cui, D.-N.; Li, Y.-F.; Liu, Y.-H.; Liu, G.; Liu, L. Multiple "Omics" data-based biomarker screening for hepatocellular carcinoma diagnosis. World J. Gastroenterol. 2019, 25, 4199–4212. [Google Scholar] [CrossRef]
- Schulze, K.; Gasch, C.; Staufer, K.; Nashan, B.; Lohse, A.W.; Pantel, K.; Riethdorf, S.; Wege, H. Presence of EpCAM-positive circulating tumor cells as biomarker for systemic disease strongly correlates to survival in patients with hepatocellular carci-noma. Int. J. Cancer 2013, 133, 2165–2171. [Google Scholar] [CrossRef]
- Guo, W.; Yang, X.R.; Sun, Y.F.; Shen, M.N.; Ma, X.L.; Wu, J.; Zhang, C.Y.; Zhou, Y.; Xu, Y.; Hu, B.; et al. Clinical significance of EpCAM mRNA-positive circulating tumor cells in hepatocellular carcinoma by an optimized negative enrichment and qRT-PCR-based platform. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2014, 20, 4794–4805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bandiera, S.; Pfeffer, S.; BaumertT, F.; ZeiselM, B. miR-122—A key factor and therapeutic target in liver disease. J. Hepatol. 2015, 62, 448–457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhayat, S.A.; Mardin, W.A.; Kφhler, G.; Bahde, R.; Vowinkel, T.; Wolters, H.; Senninger, N.; Haier, J.; Mees, S.T. The microRNA-200 family—A potential diagnostic marker in hepatocellular carcinoma? J. Surg. Oncol. 2014, 110, 430–438. [Google Scholar] [CrossRef]
- Mizuguchi, Y.; Takizawa, T.; Yoshida, H.; Uchida, E. Dysregulated miRNA in progression of hepatocellular carcinoma: A systematic review. Hepatol. Res. 2015, 46, 391–406. [Google Scholar] [CrossRef]
- Kaur, H.; Lathwal, A.; Raghava, G.P.S. Integrative multi-omics approach for stratification of tumor recurrence risk groups of Hepatocellular Carcinoma patients. bioRxiv 2021. [Google Scholar] [CrossRef]
- Cui, D.; Li, W.; Jiang, D.; Wu, J.; Xie, J.; Wu, Y. Advances in Multi-Omics Applications in HBV-Associated Hepatocellular Carcinoma. Front. Med. 2021, 8, 8. [Google Scholar] [CrossRef]
- Wang, P.-X.; Cheng, J.-W.; Yang, X.-R. Detection of circulating tumor cells in hepatocellular carcinoma: Applications in di-agnosis prognosis prediction and personalized treatment. Hepatoma Res. 2020, 6, 61. [Google Scholar] [CrossRef]
- Di Bisceglie, A.M.; Hoofnagle, J.H. Elevations in serum alpha-fetoprotein levels in patients with chronic hepatitis B. Cancer 1989, 64, 2117–2120. [Google Scholar] [CrossRef]
- Li, Y.; Tang, Z.-Y.; Ye, S.-L.; Liu, Y.-K.; Chen, J.; Xue, Q.; Chen, J.; Gao, D.-M.; Bao, W.-H. Establishment of cell clones with different metastatic potential from the metastatic hepatocellular carcinoma cell line MHCC97. World J. Gastroenterol. 2001, 7, 630–636. [Google Scholar] [CrossRef]
- Genna, A.; Vanwynsberghe, A.M.; Villard, A.V.; Pottier, C.; Ancel, J.; Polette, M.; Gilles, C. EMT-Associated Heterogeneity in Circulating Tumor Cells: Sticky Friends on the Road to Metastasis. Cancers 2020, 12, 1632. [Google Scholar] [CrossRef] [PubMed]
- Espejo-Cruz, M.L.; Gonzαlez-Rubio, S.; Zamora-Olaya, J.; Amado-Torres, V.; Alejandre, R.; Sαnchez-Frνas, M.; Ciria, R.; De la Mata, M.; Rodrνguez-Perαlvarez, M.; Ferrνn, G. Circulating Tumor Cells in Hepatocellular Carcinoma: A Comprehensive Re-view and Critical Appraisal. Int. J. Mol. Sci. 2021, 22, 13073. [Google Scholar] [CrossRef]
- Fan, J.-L.; Yang, Y.-F.; Yuan, C.-H.; Chen, H.; Wang, F.-B. Circulating tumor cells for predicting the prognostic of patients with hepatocellular carcinoma: A meta analysis. Cell. Physiol. Biochem. 2015, 37, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; He, X.; Xu, Y.; Zhang, W. FuF MiR-200b is upregulated in plasma-derived exosomes and functions as an oncogene by promoting macrophage M2 polarization in ovarian cancer. J. Ovarian Res. 2021, 14, 1–10. [Google Scholar] [CrossRef]
- Wang, X.; Liao, X.; Huang, K.; Zeng, X.; Liu, Z.; Zhou, X.; Yu, T.; Yang, C.; Yu, L.; Wang, Q.; et al. Clustered microRNAs hsa-miR-221-3p/hsa-miR-222-3p and their targeted genes might be prognostic predictors for hepato-cellular carcinoma. J. Cancer 2019, 10, 2520–2533. [Google Scholar] [CrossRef] [Green Version]
- Tsai, S.-C.; Lin, C.-C.; Shih, T.-C.; Tseng, R.-J.; Yu, M.-C.; Lin, Y.-J.; Hsieh, S.-Y. The miR-200b-ZEB1 circuit regulates diverse stemness of human hepatocellular carcinoma. Mol. Carcinog. 2017, 56, 2035–2047. [Google Scholar] [CrossRef]
- Sokolowski, K.M.; Balamurugan, M.; Kunnimalaiyaan, S.; Gamblin, T.C.; Kunnimalaiyaan, M. Notch signaling in hepato-cellular carcinoma: Molecular targeting in an advanced disease. Hepatoma Res. 2015, 1, 11–18. [Google Scholar]
- Wong, Q.W.-L.; Ching, A.K.-K.; Chan, A.W.-H.; Choy, K.-W.; To, K.-F.; Lai, P.B.-S.; Wong, N. MiR-222 overexpression confers cell migratory advantages in hepatocellular carcinoma through enhancing AKT signaling. Clin. Cancer Res. 2010, 16, 867–875. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malik, G.; Ward, M.D.; Gupta, S.K.; Trosset, M.W.; EGrizzle, W.; Adam, B.-L.; IDiaz, J.; Semmes, O.J. Serum levels of an isoform of apolipoprotein A-II as a potential marker for prostate cancer. Clin. Cancer Res. 2005, 11, 1073–1085. [Google Scholar] [CrossRef]
- Mendivil, C.O.; Zheng, C.; Furtado, J.; Lel, J.; Sacks, F.M. Metabolism of very-low-density lipoprotein and low-density lipo-protein containing apolipoprotein C-III and not other small apolipoproteins. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 239–245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Sogawa, K.; Sunaga, M.; Umemura, H.; Satoh, M.; Kazami, T.; Yoshikawa, M.; Tomonaga, T.; Yokosuka, O.; Nomura, F. Increased Concentrations of Apo A-I and Apo A-II Fragments in the Serum of Patients With Hepatocellular Carcinoma by Magnetic Beads–Assisted MALDI-TOF Mass Spectrometry. Am. J. Clin. Pathol. 2014, 141, 52–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zewinger, S.; Reiser, J.; Jankowski, V.; Alansary, D.; Hahm, E.; Triem, S.; Klug, M.; Schunk, S.J.; Schmit, D.; Kramann, R.; et al. Apolipoprotein C3 induces inflammation and organ damage by al-ternative inflammasome activation. Nat. Immunol. 2020, 21, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Mόller, I.; Strozyk, E.; Schindler, S.; Beissert, S.; Oo, H.Z.; Sauter, T.; Lucarelli, P.; Raeth, S.; Hausser, A.; Al Nakouzi, N.; et al. Cancer Cells Employ Nuclear Caspase-8 to Overcome the p53-Dependent G2/M Checkpoint through Cleavage of USP28. Mol. Cell 2020, 77, 970–984.e7. [Google Scholar] [CrossRef]
- Luo, P.; Wu, S.; Yu, Y.; Ming, X.; Li, S.; Zuo, X.; Tu, J. Current Status and Perspective Biomarkers in AFP Negative HCC: Towards Screening for and Diagnosing Hepatocellular Carcinoma at an Earlier Stage. Pathol. Oncol. Res. 2020, 26, 599–603. [Google Scholar] [CrossRef]






| HCC Patients (n = 89) | Cirrhotic Patients (n = 28) | |
|---|---|---|
| Median age, years (range) | 65 ± 11 years | 60 ± 7 years |
| Male sex, n (%) | 67, (75%) | 20, (71%) |
| Liver Disease, n (%) | ||
| Viral hepatitis | 32, (36%) | 9, (32%) |
| Non-viral hepatitis | 57, (64%) | 19, (68%) |
| HCC stage, n (%) | ||
| BCLC A | 33, (37%) | n/a |
| BCLC C | 56, (63%) | |
| Microvascular invasion | ||
| (Available data in 55 patients) | 23, (26%) | n/a |
| Serum aFP values >20 ng/ml | ||
| Early HCC | 7/33 (21%) | - |
| Advanced HCC | 42/56 (75%) | - |
| Cirrhotic | - | 0/28 (0%) |
| EPCAM | Vimentin | AFP | sMVP | |
|---|---|---|---|---|
| Advanced HCC (53 pts) | 27/53 | 10/53 | 12/53 | 6/53 |
| Early HCC (17 pts) | 5/17 | 3/17 | 1/17 | 1/17 |
| Cirrhosis (28 pts) | 2/28 | 1/28 | 1/28 | 0/28 |
| (i) | ||||
| EPCAM | Vimentin | AFP | sMVP | |
| Cirrhotic | ||||
| miR-200b | * 2/2 | * 1/1 | * 1/1 | 0/2 |
| miR-122 | * 2/2 | * 1/1 | 0/1 | 0/2 |
| miR-221 | 0/2 | 0/1 | 0/1 | 0/2 |
| miR-222 | 0/2 | 0/1 | 0/1 | 0/2 |
| miR-200a | 0/2 | 0/1 | 0/1 | 0/2 |
| Early HCC | ||||
| miR-200b | * 4/5 | * 3/3 | * 1/1 | 0/1 |
| miR-122 | * 3/5 | * 1/3 | 0/1 | 0/1 |
| miR-221 | * 2/5 | 0/3 | 0/1 | 0/1 |
| miR-222 | * 3/5 | * 1/3 | 0/1 | 0/1 |
| miR-200a | 0/5 | 0/3 | 0/1 | 0/1 |
| Advanced HCC | ||||
| miR-200b | * 7/27 | * 7/10 | * 3/12 | * 2/6 |
| miR-122 | * 11/27 | * 9/10 | * 7/12 | * 4/6 |
| miR-221 | * 5/27 | * 5/10 | * 3/12 | * 2/6 |
| miR-222 | * 12/27 | * 8/10 | * 10/12 | * 4/6 |
| miR-200a | * 3/27 | * 3/10 | * 3/12 | 0/6 |
| (ii) | ||||
| miR-200b | Cirrhotic | Early HCC | Advanced HCC | |
| CTCs | 2 | 4 | 7 | |
| No CTCs | 1 | 2 | 3 | |
| miR-122 | Cirrhotic | Early HCC | Advanced HCC | |
| CTCs | 2 | 3 | 11 | |
| No CTCs | 7 | 7 | 6 | |
| miR-221 | Cirrhotic | Early HCC | Advanced HCC | |
| CTCs | 0 | 2 | 5 | |
| No CTCs | 13 | 8 | 9 | |
| miR-222 | Cirrhotic | Early HCC | Advanced HCC | |
| CTCs | 0 | 3 | 12 | |
| No CTCs | 13 | 7 | 8 | |
| miR-200a | Cirrhotic | Early HCC | Advanced HCC | |
| CTCs | 0 | 0 | 3 | |
| No CTCs | 7 | 4 | 6 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Armakolas, A.; Dimopoulou, V.; Nezos, A.; Stamatakis, G.; Samiotaki, M.; Panayotou, G.; Tampaki, M.; Stathaki, M.; Dourakis, S.; Koskinas, J. Cellular, Molecular and Proteomic Characteristics of Early Hepatocellular Carcinoma. Curr. Issues Mol. Biol. 2022, 44, 4714-4734. https://doi.org/10.3390/cimb44100322
Armakolas A, Dimopoulou V, Nezos A, Stamatakis G, Samiotaki M, Panayotou G, Tampaki M, Stathaki M, Dourakis S, Koskinas J. Cellular, Molecular and Proteomic Characteristics of Early Hepatocellular Carcinoma. Current Issues in Molecular Biology. 2022; 44(10):4714-4734. https://doi.org/10.3390/cimb44100322
Chicago/Turabian StyleArmakolas, Athanasios, Vasiliki Dimopoulou, Adrianos Nezos, George Stamatakis, Martina Samiotaki, George Panayotou, Maria Tampaki, Martha Stathaki, Spyridon Dourakis, and John Koskinas. 2022. "Cellular, Molecular and Proteomic Characteristics of Early Hepatocellular Carcinoma" Current Issues in Molecular Biology 44, no. 10: 4714-4734. https://doi.org/10.3390/cimb44100322
APA StyleArmakolas, A., Dimopoulou, V., Nezos, A., Stamatakis, G., Samiotaki, M., Panayotou, G., Tampaki, M., Stathaki, M., Dourakis, S., & Koskinas, J. (2022). Cellular, Molecular and Proteomic Characteristics of Early Hepatocellular Carcinoma. Current Issues in Molecular Biology, 44(10), 4714-4734. https://doi.org/10.3390/cimb44100322







