Epoxyalcohol Synthase Branch of Lipoxygenase Cascade
Abstract
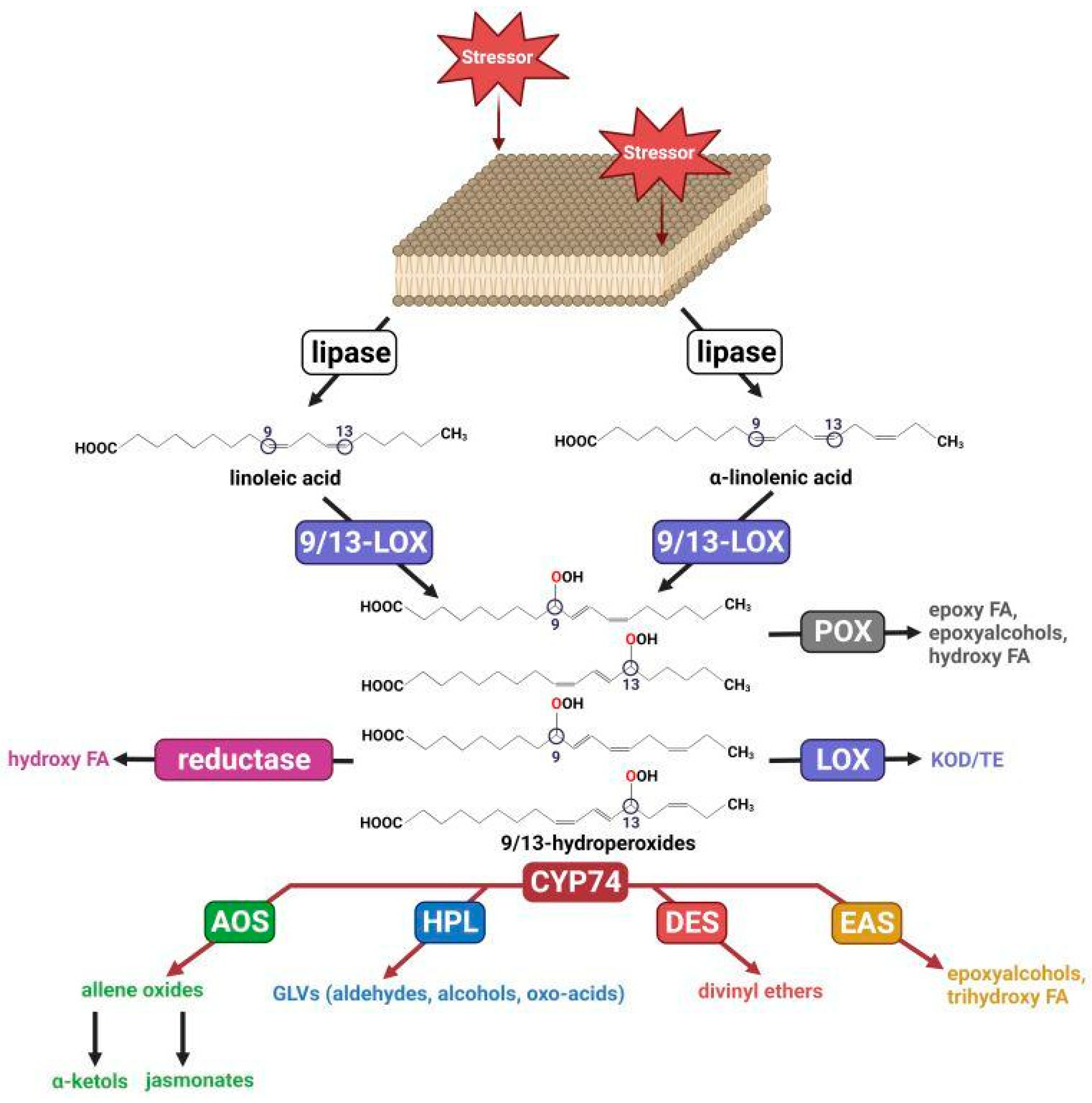
1. Introduction
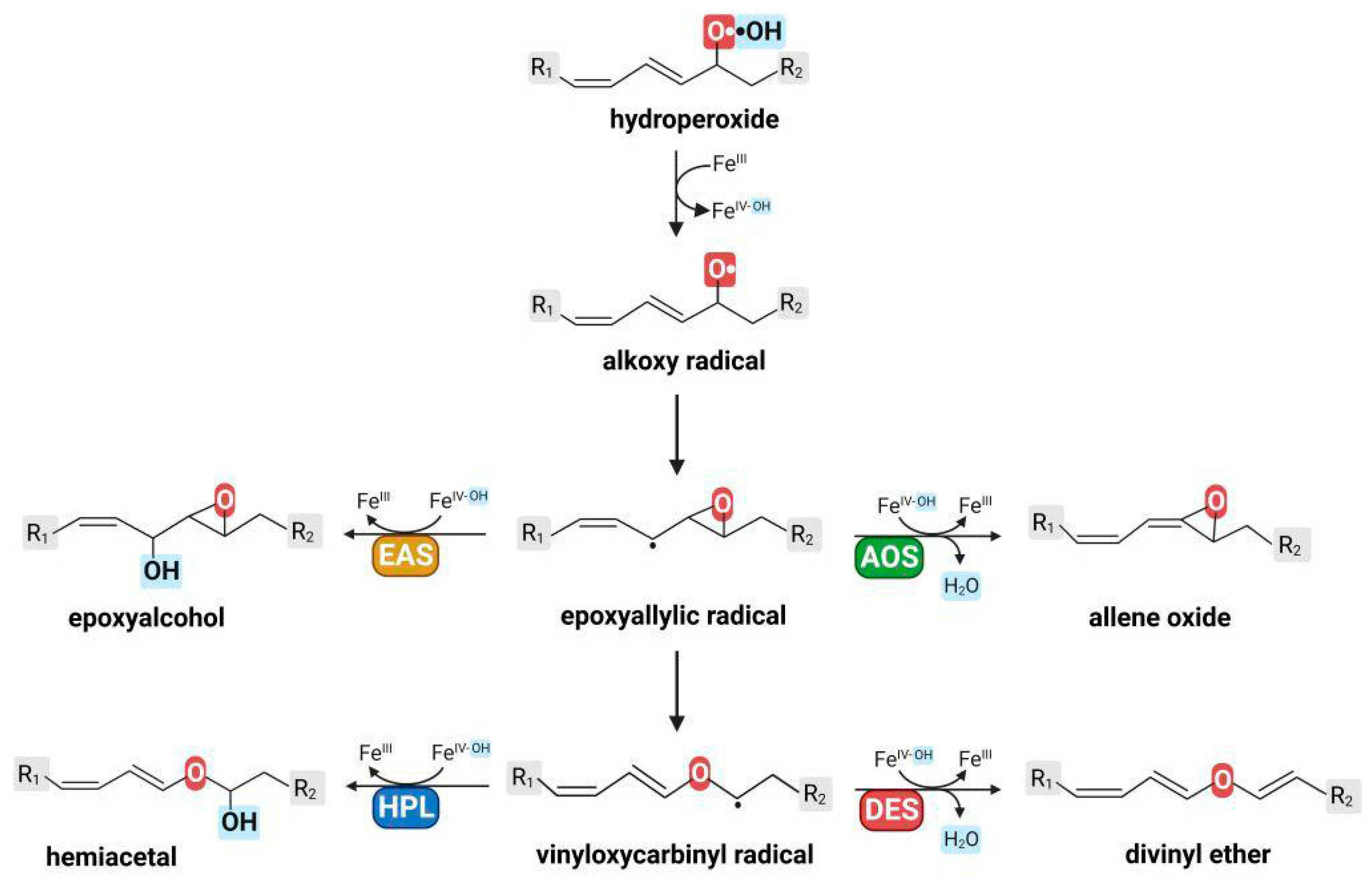
2. Epoxyalcohol Synthase Activity in Different CYP74 Enzymes
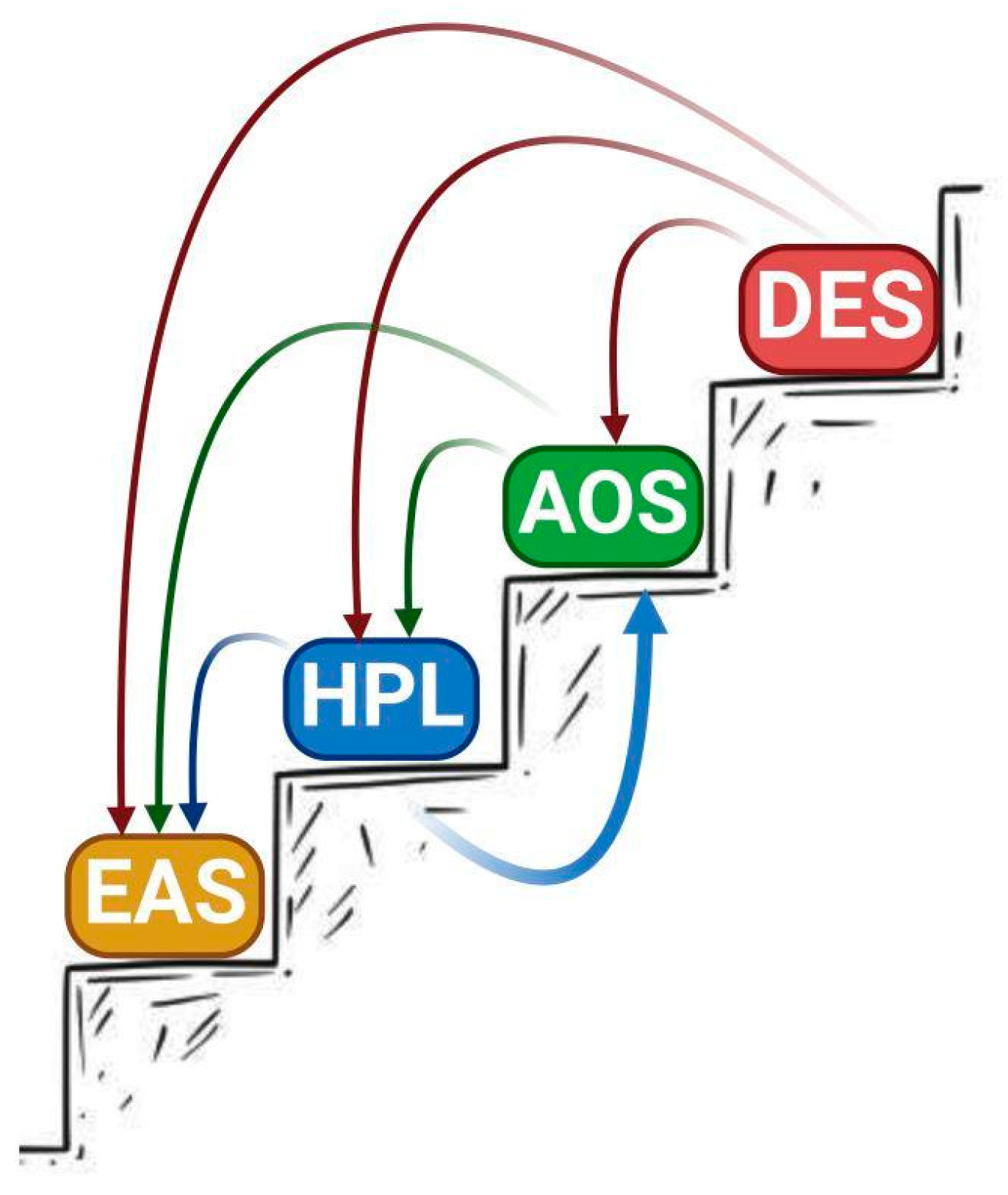
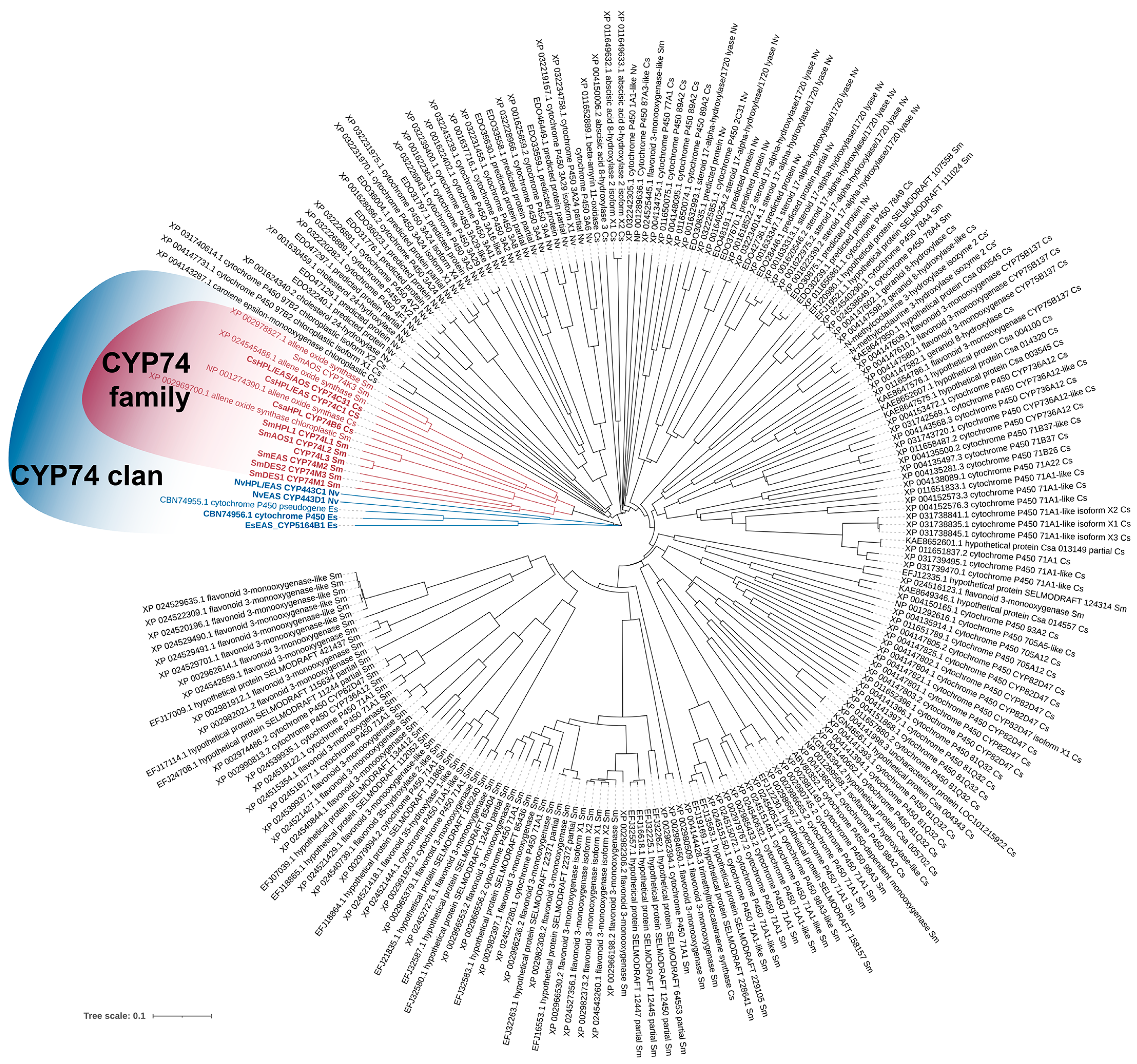
3. Phylogeny
4. Correlation between Primary Structure and Catalytic Activity of CYP74s
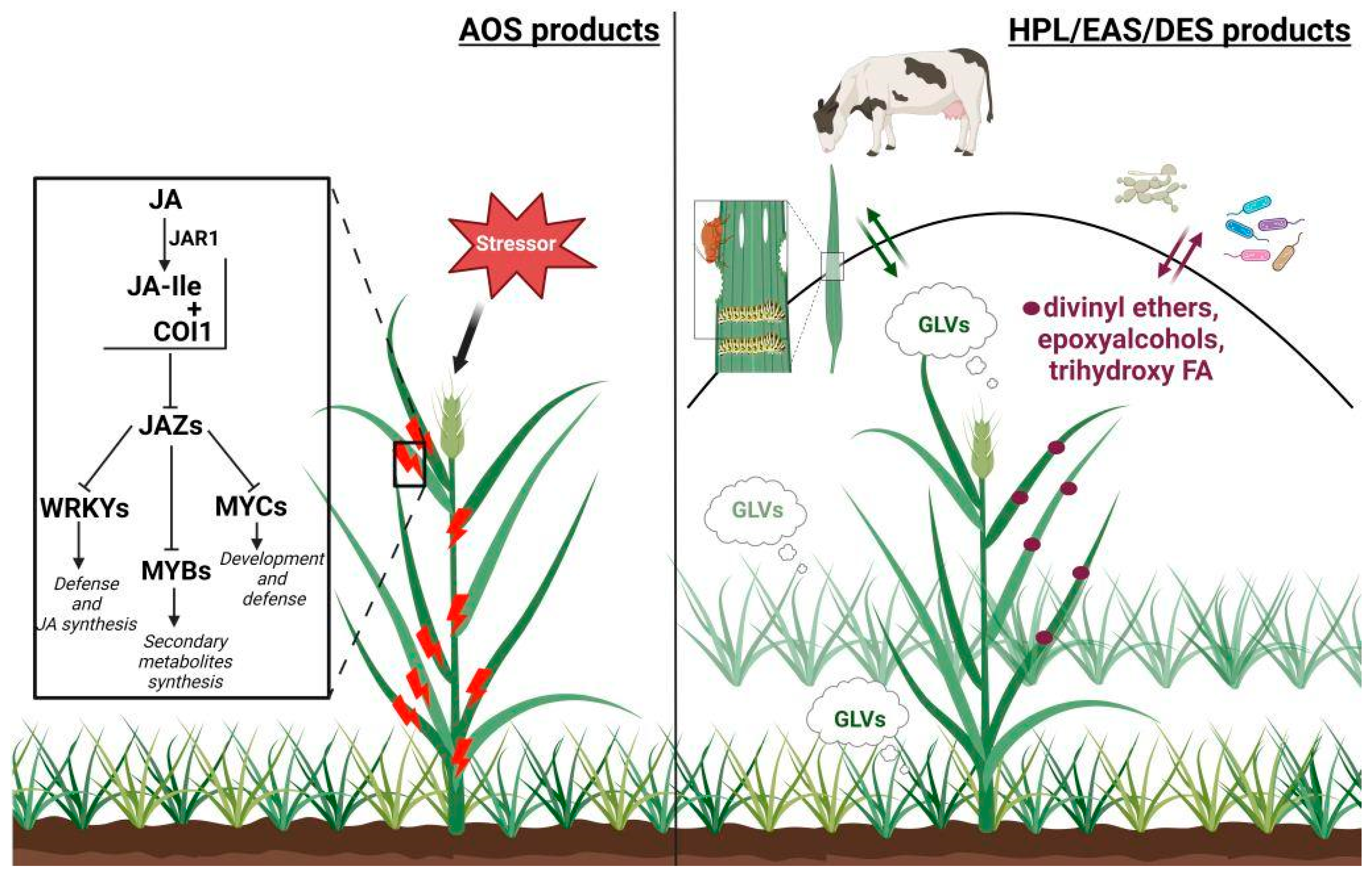
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Brodhun, F.; Feussner, I. Oxylipins in Fungi. FEBS J. 2011, 278, 1047–1063. [Google Scholar] [CrossRef] [PubMed]
- Savchenko, T.V.; Zastrijnaja, O.M.; Klimov, V.V. Oxylipins and Plant Abiotic Stress Resistance. Biochem. Mosc. 2014, 79, 362–375. [Google Scholar] [CrossRef]
- De Domenico, S.; Taurino, M.; Gallo, A.; Poltronieri, P.; Pastor, V.; Flors, V.; Santino, A. Oxylipin Dynamics in Medicago Truncatula in Response to Salt and Wounding Stresses. Physiol. Plant. 2019, 165, 198–208. [Google Scholar] [CrossRef]
- Christie, W.W.; Harwood, J.L. Oxidation of Polyunsaturated Fatty Acids to Produce Lipid Mediators. Essays Biochem. 2020, 64, 401–421. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, P.; Munné-Bosch, S. Oxylipins in Plastidial Retrograde Signaling. Redox Biol. 2020, 37, 101717. [Google Scholar] [CrossRef]
- Shipelin, V.A.; Sidorova, Y.S. Oxylipins—biologically active substances of food. Vopr. Pitan. 2020, 89, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Savchenko, T.; Degtyaryov, E.; Radzyukevich, Y.; Buryak, V. Therapeutic Potential of Plant Oxylipins. Int. J. Mol. Sci. 2022, 23, 14627. [Google Scholar] [CrossRef] [PubMed]
- Gorina, S.; Ogorodnikova, A.; Mukhtarova, L.; Toporkova, Y. Gene Expression Analysis of Potato (Solanum Tuberosum L.) Lipoxygenase Cascade and Oxylipin Signature under Abiotic Stress. Plants 2022, 11, 683. [Google Scholar] [CrossRef]
- Esser-von Bieren, J. Eicosanoids in Tissue Repair. Immunol. Cell Biol. 2019, 97, 279–288. [Google Scholar] [CrossRef]
- Wallace, J.L. Eicosanoids in the gastrointestinal tract. Br. J. Pharmacol. 2019, 176, 1000–1008. [Google Scholar] [CrossRef]
- Mitchell, J.A.; Kirkby, N.S. Eicosanoids, Prostacyclin and Cyclooxygenase in the Cardiovascular System. Br. J. Pharmacol. 2019, 176, 1038–1050. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Eicosanoids. Essays Biochem. 2020, 64, 423–441. [Google Scholar] [CrossRef]
- Hoxha, M.; Zappacosta, B. CYP-derived eicosanoids: Implications for rheumatoid arthritis. Prostaglandins Other Lipid Mediat. 2020, 146, 106405. [Google Scholar] [CrossRef] [PubMed]
- Yasukawa, K.; Okuno, T.; Yokomizo, T. Eicosanoids in Skin Wound Healing. Int. J. Mol. Sci. 2020, 21, 8435. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, M.L.; Lorenzetti, F. Role of eicosanoids in liver repair, regeneration and cancer. Biochem. Pharmacol. 2021, 192, 114732. [Google Scholar] [CrossRef]
- Radmark, O. Formation of eicosanoids and other oxylipins in human macrophages. Biochem. Pharmacol. 2022, 204, 115210. [Google Scholar] [CrossRef]
- Mukhtarova, L.S.; Brühlmann, F.; Hamberg, M.; Khairutdinov, B.I.; Grechkin, A.N. Plant Hydroperoxide-Cleaving Enzymes (CYP74 Family) Function as Hemiacetal Synthases: Structural Proof of Hemiacetals by NMR Spectroscopy. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2018, 1863, 1316–1322. [Google Scholar] [CrossRef]
- Itoh, A.; Howe, G.A. Molecular Cloning of a Divinyl Ether Synthase: Identification as a CYP74 cytochrome P-450 *. J. Biol. Chem. 2001, 276, 3620–3627. [Google Scholar] [CrossRef]
- Stumpe, M.; Kandzia, R.; Göbel, C.; Rosahl, S.; Feussner, I. A Pathogen-Inducible Divinyl Ether Synthase (CYP74D) from Elicitor-Treated Potato Suspension Cells. FEBS Lett. 2001, 507, 371–376. [Google Scholar] [CrossRef]
- Fammartino, A.; Cardinale, F.; Göbel, C.; Mène-Saffrané, L.; Fournier, J.; Feussner, I.; Esquerré-Tugayé, M.-T. Characterization of a Divinyl Ether Biosynthetic Pathway Specifically Associated with Pathogenesis in Tobacco. Plant Physiol. 2007, 143, 378–388. [Google Scholar] [CrossRef]
- Gogolev, Y.V.; Gorina, S.S.; Gogoleva, N.E.; Toporkova, Y.Y.; Chechetkin, I.R.; Grechkin, A.N. Green Leaf Divinyl Ether Synthase: Gene Detection, Molecular Cloning and Identification of a Unique CYP74B Subfamily Member. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2012, 1821, 287–294. [Google Scholar] [CrossRef]
- Werck-Reichhart, D.; Feyereisen, R. Cytochromes P450: A success story. Genome Biol. 2000, 6, 1–7. [Google Scholar] [CrossRef]
- Shengying, L.; Chaulagain, M.R.; Knauff, A.R.; Podust, L.M.; Montgomery, J.; Sherman, D.H. Selective oxidation of carbolide C–H bonds by an engineered macrolide P450 monooxygenase. Proc. Natl. Acad. Sci. USA 2009, 106, 18463–18468. [Google Scholar] [CrossRef]
- Ortiz de Montellano, P.R.; Nelson, S.D. Rearrangement reactions catalyzed by cytochrome P450s. Arch. Biochem. Biophys. 2011, 507, 95–110. [Google Scholar] [CrossRef] [PubMed]
- Poulos, T.L. Heme enzyme structure and function. Chem. Rev. 2014, 114, 3919–3962. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-S.; Nioche, P.; Hamberg, M.; Raman, C.S. Structural Insights into the Evolutionary Paths of Oxylipin Biosynthetic Enzymes. Nature 2008, 455, 363–368. [Google Scholar] [CrossRef]
- Smith, W.L.; Urade, Y.; Jakobsson, P.-J. Enzymes of the Cyclooxygenase Pathways of Prostanoid Biosynthesis. Chem. Rev. 2011, 111, 5821–5865. [Google Scholar] [CrossRef]
- Guengerich, F.P.; Munro, A.W. Unusual cytochrome P450 enzymes and reactions. J. Biol. Chem. 2013, 288, 17065–17073. [Google Scholar] [CrossRef]
- Proteau, P.J.; Gerwick, W.H. Divinyl Ethers and Hydroxy Fatty Acids from Three Species of Laminaria (Brown Algae). Lipids 1993, 28, 783–787. [Google Scholar] [CrossRef]
- Jiang, Z.-D.; Gerwick, W.H. Novel Oxylipins from the Temperate Red Alga Polyneura latissima: Evidence for an Arachidonate 9(S)-Lipoxygenase. Lipids 1997, 32, 231–235. [Google Scholar] [CrossRef]
- Nelson, D.R.; Goldstone, J.V.; Stegeman, J.J. The Cytochrome P450 Genesis Locus: The Origin and Evolution of Animal Cytochrome P450s. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20120474. [Google Scholar] [CrossRef] [PubMed]
- Medina, S.; Gil-Izquierdo, Á.; Durand, T.; Ferreres, F.; Domínguez-Perles, R. Structural/Functional Matches and Divergences of Phytoprostanes and Phytofurans with Bioactive Human Oxylipins. Antioxidants 2018, 7, 165. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Perles, R.; Abellán, Á.; Leon, D.; Ferreres, F.; Guy, A.; Oger, C.; Galano, J.; Durand, T. Sorting out the Phytoprostane and Phytofuran Profile in Vegetable Oils. Food Res. Int. 2018, 107, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Perles, R.; Gil-Izquierdo, A.; Ferreres, F.; Medina, S. Update on Oxidative Stress and Inflammation in Pregnant Women, Unborn Children (Nasciturus), and Newborns—Nutritional and Dietary Effects. Free. Radic. Biol. Med. 2019, 142, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Pinciroli, M.; Domínguez-Perles, R.; Garbi, M.; Abellán, A.; Oger, C.; Durand, T.; Galano, J.M.; Ferreres, F.; Gil-Izquierdo, A. Impact of Salicylic Acid Content and Growing Environment on Phytoprostane and Phytofuran (Stress Biomarkers) in Oryza Sativa L. J. Agric. Food Chem. 2018, 66, 12561–12570. [Google Scholar] [CrossRef]
- Pinciroli, M.; Domínguez-Perles, R.; Abellán, Á.; Bultel-Poncé, V.; Durand, T.; Galano, J.M.; Ferreres, F.; Gil-Izquierdo, Á. Statement of Foliar Fertilization Impact on Yield, Composition, and Oxidative Biomarkers in Rice. J. Agric. Food Chem. 2019, 67, 597–605. [Google Scholar] [CrossRef]
- Lipan, L.; Collado-González, J.; Domínguez-Perles, R.; Corell, M.; Bultel-Poncé, V.; Galano, J.-M.; Durand, T.; Medina, S.; Gil-Izquierdo, Á.; Carbonell-Barrachina, Á. Phytoprostanes and Phytofurans-Oxidative Stress and Bioactive Compounds-in Almonds Are Affected by Deficit Irrigation in Almond Trees. J. Agric. Food Chem. 2020, 68, 7214–7225. [Google Scholar] [CrossRef]
- Collado-González, J.; Cano-Lamadrid, M.; Pérez-López, D.; Carbonell-Barrachina, Á.A.; Centeno, A.; Medina, S.; Griñán, I.; Guy, A.; Galano, J.-M.; Durand, T.; et al. Effects of Deficit Irrigation, Rootstock, and Roasting on the Contents of Fatty Acids, Phytoprostanes, and Phytofurans in Pistachio Kernels. J. Agric. Food Chem. 2020, 68, 8915–8924. [Google Scholar] [CrossRef]
- Ramos, L.L.; Jiménez-Aspee, F.; Theoduloz, C.; Burgos-Edwards, A.; Domínguez-Perles, R.; Oger, C.; Durand, T.; Gil-Izquierdo, Á.; Bustamante, L.; Mardones, C.; et al. Phenolic, Oxylipin and Fatty Acid Profiles of the Chilean Hazelnut (Gevuina avellana): Antioxidant Activity and Inhibition of pro-Inflammatory and Metabolic Syndrome-Associated Enzymes. Food Chem. 2019, 298, 125026. [Google Scholar] [CrossRef]
- Medina, S.; Gil-Izquierdo, Á.; Abu-Reidah, I.M.; Durand, T.; Bultel-Poncé, V.; Galano, J.-M.; Domínguez-Perles, R. Evaluation of Phoenix Dactylifera Edible Parts and Byproducts as Sources of Phytoprostanes and Phytofurans. J. Agric. Food Chem. 2020, 68, 8942–8950. [Google Scholar] [CrossRef]
- Martínez Sánchez, S.; Domínguez-Perles, R.; Montoro-García, S.; Gabaldón, J.A.; Guy, A.; Durand, T.; Oger, C.; Ferreres, F.; Gil-Izquierdo, A. Bioavailable Phytoprostanes and Phytofurans from Gracilaria longissima Have Anti-Inflammatory Effects in Endothelial Cells. Food Funct. 2020, 11, 5166–5178. [Google Scholar] [CrossRef] [PubMed]
- Grechkin, A. Recent Developments in Biochemistry of the Plant Lipoxygenase Pathway. Prog. Lipid Res. 1998, 37, 317–352. [Google Scholar] [CrossRef] [PubMed]
- Ogorodnikova, A.V.; Mukhitova, F.K.; Grechkin, A.N. Oxylipins in the Spikemoss Selaginella Martensii: Detection of Divinyl Ethers, 12-Oxophytodienoic Acid and Related Cyclopentenones. Phytochemistry 2015, 118, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Wasternack, C.; Feussner, I. The Oxylipin Pathways: Biochemistry and Function. Annu. Rev. Plant Biol. 2018, 69, 363–386. [Google Scholar] [CrossRef]
- Deboever, E.; Deleu, M.; Mongrand, S.; Lins, L.; Fauconnier, M.-L. Plant–Pathogen Interactions: Underestimated Roles of Phyto-Oxylipins. Trends Plant Sci. 2020, 25, 22–34. [Google Scholar] [CrossRef]
- Tanaka, M.; Koeduka, T.; Matsui, K. Green Leaf Volatile-Burst in Selaginella moellendorffii. Front. Plant Sci. 2021, 12, 731694. [Google Scholar] [CrossRef]
- Grechkin, A.N.; Ogorodnikova, A.V.; Egorova, A.M.; Mukhitova, F.K.; Ilyina, T.M.; Khairutdinov, B.I. Allene Oxide Synthase Pathway in Cereal Roots: Detection of Novel Oxylipin Graminoxins. ChemistryOpen 2018, 7, 336–343. [Google Scholar] [CrossRef]
- Chechetkin, I.R.; Blufard, A.S.; Yarin, A.Y.; Fedina, E.O.; Khairutdinov, B.I.; Grechkin, A.N. Detection and Identification of Complex Oxylipins in Meadow Buttercup (Ranunculus acris) Leaves. Phytochemistry 2019, 157, 92–102. [Google Scholar] [CrossRef]
- Yu, X.; Zhang, W.; Zhang, Y.; Zhang, X.; Lang, D.; Zhang, X. The Roles of Methyl Jasmonate to Stress in Plants. Funct. Plant Biol. 2019, 46, 197–212. [Google Scholar] [CrossRef]
- Ruan, J.; Zhou, Y.; Zhou, M.; Yan, J.; Khurshid, M.; Weng, W.; Cheng, J.; Zhang, K. Jasmonic Acid Signaling Pathway in Plants. Int. J. Mol. Sci. 2019, 20, 2479. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, F.; Chen, L.; Pan, Y.; Sun, L.; Bao, N.; Zhang, T.; Cui, C.-X.; Qiu, Z.; Zhang, Y.; et al. Jasmonate-Mediated Wound Signalling Promotes Plant Regeneration. Nat. Plants 2019, 5, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, D.-D.; Fang, X.; Chen, X.-Y.; Mao, Y.-B. Plant Specialized Metabolism Regulated by Jasmonate Signaling. Plant Cell Physiol. 2019, 60, 2638–2647. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Q.; Li, C. The Plant Mediator Complex and Its Role in Jasmonate Signaling. J. Exp. Bot. 2019, 70, 3415–3424. [Google Scholar] [CrossRef]
- Hellmann, E.; Helariutta, Y. Plant Genetics: Advances in Regeneration Pathways. Curr. Biol. 2019, 29, R702–R704. [Google Scholar] [CrossRef] [PubMed]
- Ueda, M.; Kaji, T.; Kozaki, W. Recent Advances in Plant Chemical Biology of Jasmonates. Int. J. Mol. Sci. 2020, 21, 1124. [Google Scholar] [CrossRef]
- Wu, X.; Ye, J. Manipulation of Jasmonate Signaling by Plant Viruses and Their Insect Vectors. Viruses 2020, 12, 148. [Google Scholar] [CrossRef]
- Gomi, K. Jasmonic Acid: An Essential Plant Hormone. Int. J. Mol. Sci. 2020, 21, 1261. [Google Scholar] [CrossRef]
- Griffiths, G. Jasmonates: Biosynthesis, Perception and Signal Transduction. Essays Biochem. 2020, 64, 501–512. [Google Scholar] [CrossRef]
- Jang, G.; Yoon, Y.; Choi, Y.D. Crosstalk with Jasmonic Acid Integrates Multiple Responses in Plant Development. Int. J. Mol. Sci. 2020, 21, 305. [Google Scholar] [CrossRef]
- Zhuo, M.; Sakuraba, Y.; Yanagisawa, S. A Jasmonate-Activated MYC2-Dof2.1-MYC2 Transcriptional Loop Promotes Leaf Senescence in Arabidopsis. Plant Cell 2020, 32, 242–262. [Google Scholar] [CrossRef]
- Ali, M.S.; Baek, K.-H. Jasmonic Acid Signaling Pathway in Response to Abiotic Stresses in Plants. Int. J. Mol. Sci. 2020, 21, 621. [Google Scholar] [CrossRef]
- Yang, Z.; Huang, Y.; Yang, J.; Yao, S.; Zhao, K.; Wang, D.; Qin, Q.; Bian, Z.; Li, Y.; Lan, Y.; et al. Jasmonate Signaling Enhances RNA Silencing and Antiviral Defense in Rice. Cell Host Microbe 2020, 28, 89–103.e8. [Google Scholar] [CrossRef]
- Zhao, L.; Li, X.; Chen, W.; Xu, Z.; Chen, M.; Wang, H.; Yu, D. The Emerging Role of Jasmonate in the Control of Flowering Time. J. Exp. Bot. 2022, 73, 11–21. [Google Scholar] [CrossRef]
- Bonner, J.; English, J., Jr. Purification of traumatin, a plant wound hormone. Science 1937, 86, 352–353. [Google Scholar] [CrossRef] [PubMed]
- Noordermeer, M.A.; Veldink, G.A.; Vliegenthart, J.F.G. Fatty Acid Hydroperoxide Lyase: A Plant Cytochrome P450 Enzyme Involved in Wound Healing and Pest Resistance. ChemBioChem 2001, 2, 494–504. [Google Scholar] [CrossRef] [PubMed]
- Stumpe, M.; Bode, J.; Göbel, C.; Wichard, T.; Schaaf, A.; Frank, W.; Frank, M.; Reski, R.; Pohnert, G.; Feussner, I. Biosynthesis of C9-Aldehydes in the Moss Physcomitrella patens. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2006, 1761, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Bouwmeester, H.; Schuurink, R.C.; Bleeker, P.M.; Schiestl, F. The Role of Volatiles in Plant Communication. Plant J. 2019, 100, 892–907. [Google Scholar] [CrossRef]
- Stolterfoht, H.; Rinnofner, C.; Winkler, M.; Pichler, H. Recombinant Lipoxygenases and Hydroperoxide Lyases for the Synthesis of Green Leaf Volatiles. J. Agric. Food Chem. 2019, 67, 13367–13392. [Google Scholar] [CrossRef]
- Hashem, C.; Hochrinner, J.; Bürgler, M.B.; Rinnofner, C.; Pichler, H.; Winkler, M. From linoleic acid to hexanal and hexanol by whole cell catalysis with a lipoxygenase, hydroperoxide lyase and reductase cascade in Komagataella phaffii. Front. Mol. Biosci. 2022, 9, 965315. [Google Scholar] [CrossRef]
- Matsui, K.; Engelberth, J. Green leaf volatiles—The forefront of plant responses against biotic attack. Plant Cell Physiol. 2022, 63, 1378–1390. [Google Scholar] [CrossRef]
- Yactayo-Chang, J.P.; Hunter, C.T.; Alborn, H.T.; Christensen, S.A.; Block, A.K. Production of the Green Leaf Volatile (Z)-3-Hexenal by a Zea mays Hydroperoxide Lyase. Plants 2022, 11, 2201. [Google Scholar] [CrossRef]
- Yan, B.; Zheng, H.; Sang, Y.; Wang, Y.; Sun, J.; Li, F.; Wang, J.; Wang, X. A Single Amino Acid Substitution in MIL1 Leads to Activation of Programmed Cell Death and Defense Responses in Rice. Int. J. Mol. Sci. 2022, 23, 8853. [Google Scholar] [CrossRef]
- Zhou, Z.W.; Wu, Q.Y.; Yang, Y.; Hu, Q.C.; Wu, Z.J.; Huang, H.Q.; Lin, H.Z.; Lai, Z.X.; Sun, Y. The Dynamic Change in Fatty Acids during the Postharvest Process of Oolong Tea Production. Molecules 2022, 27, 4298. [Google Scholar] [CrossRef] [PubMed]
- Jo, H.E.; Song, K.; Kim, J.G.; Lee, C.H. Non-targeted metabolomic analysis for the comparative evaluation of volatile organic compounds in 20 globally representative cucumber lines. Front. Plant Sci. 2022, 13, 1028735. [Google Scholar] [CrossRef] [PubMed]
- Yue, R.; Zhang, Z.; Shi, Q.; Duan, X.; Wen, C.; Shen, B.; Li, X. Identification of the key genes contributing to the LOX-HPL volatile aldehyde biosynthesis pathway in jujube fruit. Int. J. Biol. Macromol. 2022, 222, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Kaur, I.; Korrapati, N.; Bonello, J.; Mukherjee, A.; Rishi, V.; Bendigiri, C. Biosynthesis of natural aroma compounds using recombinant whole-cell tomato hydroperoxide lyase biocatalyst. J. Biosci. 2022, 47, 37. [Google Scholar] [CrossRef]
- Aratani, Y.; Uemura, T.; Hagihara, T.; Matsui, K.; Toyota, M. Green leaf volatile sensory calcium transduction in Arabidopsis. Nat. Commun. 2023, 14, 6236. [Google Scholar] [CrossRef]
- Coenen, A.; Ferrer, M.; Jaeger, K.E.; Schörken, U. Synthesis of 12-aminododecenoic acid by coupling transaminase to oxylipin pathway enzymes. Appl. Microbiol. Biotechnol. 2023, 107, 2209–2221. [Google Scholar] [CrossRef]
- Kato, T.; Yamaguchi, Y.; Abe, N.; Uyehara, T.; Namai, T.; Kodama, M.; Shiobara, Y. Structure and Synthesis of Unsaturaded Trihydroxy C18 Fatty: Acids in Rice Plants Suffering from Rice Blast Disease. Tetrahedron Lett. 1985, 26, 2357–2360. [Google Scholar] [CrossRef]
- Kato, T.; Yamaguchi, Y.; Hirukawa, T.; Hoshino, N. Structural Elucidation of Naturally Occurring 9, 12, 13-Trihydroxy Fatty Acids by a Synthetic Study. Agric. Biol. Chem. 1991, 55, 1349–1357. [Google Scholar] [CrossRef]
- Hamberg, M. An Epoxy Alcohol Synthase Pathway in Higher Plants: Biosynthesis of Antifungal Trihydroxy Oxylipins in Leaves of Potato. Lipids 1999, 34, 1131–1142. [Google Scholar] [CrossRef]
- Hamberg, M. Biosynthesis of New Divinyl Ether Oxylipins in Ranunculus Plants. Lipids 2002, 37, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Hamberg, M.; Olsson, U. Efficient and Specific Conversion of 9-Lipoxygenase Hydroperoxides in the Beetroot. Formation of Pinellic Acid. Lipids 2011, 46, 873–878. [Google Scholar] [CrossRef]
- Jin, J.; Boeglin, W.E.; Cha, J.K.; Brash, A.R. 8R-Lipoxygenase-catalyzed synthesis of a prominent cis-epoxyalcohol from dihomo-γ-linolenic acid: A distinctive transformation compared with S-lipoxygenases. J. Lipid Res. 2012, 53, 292–299. [Google Scholar] [CrossRef]
- Wennman, A.; Oliw, E.H. Secretion of Two Novel Enzymes, Manganese 9S-Lipoxygenase and Epoxy Alcohol Synthase, by the Rice Pathogen Magnaporthe salvinii. J. Lipid Res. 2013, 54, 762–775. [Google Scholar] [CrossRef] [PubMed]
- Aghofack-Nguemezi, J.; Schwab, W. Spatiotemporal Changes in the Content and Metabolism of 9, 12, 13—Trihydorxy-10(E)-Octadecenoic Acid in Tomato (Solanum lycopersicum L. CV Balkonsar) Fruits. J. Sci. Technol. 2013, 33, 12–22. [Google Scholar] [CrossRef]
- d’Ippolito, G.; Nuzzo, G.; Sardo, A.; Manzo, E.; Gallo, C.; Fontana, A. Lipoxygenases and Lipoxygenase Products in Marine Diatoms. Methods Enzymol. 2018, 605, 69–100. [Google Scholar] [CrossRef] [PubMed]
- An, J.-U.; Hong, S.-H.; Oh, D.-K. Regiospecificity of a Novel Bacterial Lipoxygenase from Myxococcus Xanthus for Polyunsaturated Fatty Acids. Biochim. Et Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2018, 1863, 823–833. [Google Scholar] [CrossRef]
- An, J.-U.; Lee, I.-G.; Ko, Y.-J.; Oh, D.-K. Microbial Synthesis of Linoleate 9 S -Lipoxygenase Derived Plant C18 Oxylipins from C18 Polyunsaturated Fatty Acids. J. Agric. Food Chem. 2019, 67, 3209–3219. [Google Scholar] [CrossRef]
- Oliw, E.H. Linoleate Diol Synthase Related Enzymes of the Human Pathogens Histoplasma capsulatum and Blastomyces dermatitidis. Arch. Biochem. Biophys. 2020, 696, 108669. [Google Scholar] [CrossRef]
- Oliw, E.H. Fatty Acid Dioxygenase-Cytochrome P450 Fusion Enzymes of Filamentous Fungal Pathogens. Fungal Genet. Biol. 2021, 157, 103623. [Google Scholar] [CrossRef]
- Edin, M.L.; Yamanashi, H.; Boeglin, W.E.; Graves, J.P.; DeGraff, L.M.; Lih, F.B.; Zeldin, D.C.; Brash, A.R. Epoxide Hydrolase 3 (Ephx3) Gene Disruption Reduces Ceramide Linoleate Epoxide Hydrolysis and Impairs Skin Barrier Function. J. Biol. Chem. 2021, 296, 100198. [Google Scholar] [CrossRef]
- Blée, E.; Schuber, F. Efficient Epoxidation of Unsaturated Fatty Acids by a Hydroperoxide-Dependent Oxygenase. J. Biol. Chem. 1990, 265, 12887–12894. [Google Scholar] [CrossRef]
- Hamberg, M.; Hamberg, G. Peroxygenase-catalyzed fatty acid epoxidation in cereal seeds (sequential oxidation of linoleic acid into 9 (S), 12 (S), 13 (S)-trihydroxy-10 (E)-octadecenoic acid). Plant Physiol. 1996, 110, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Blée, E.; Flenet, M.; Boachon, B.; Fauconnier, M.-L. A Non-Canonical Caleosin from Arabidopsis Efficiently Epoxidizes Physiological Unsaturated Fatty Acids with Complete Stereoselectivity. FEBS J. 2012, 279, 3981–3995. [Google Scholar] [CrossRef]
- Garscha, U.; Oliw, E.H. Leucine/valine residues direct oxygenation of linoleic acid by (10R)-and (8R)-dioxygenases: Expression and site-directed mutagenesis of (10R)-dioxygenase with epoxyalcohol synthase activity. J. Biol. Chem. 2009, 284, 13755–13765. [Google Scholar] [CrossRef] [PubMed]
- Gardner, H.W.; Weisleder, D.; Nelson, E.C. Acid catalysis of a linoleic acid hydroperoxide: Formation of epoxides by an intramolecular cyclization of the hydroperoxide group. J. Org. Chem. 1984, 49, 508–515. [Google Scholar]
- Gardner, H.; Nelson, E.C.; Tjarks, L.W.; England, R.E. Acid-Catalyzed Transformation of 13(S)-Hydroperoxylinoleic Acid into Epoxyhydroxyoctadecenoic and Trihydroxyoctadecenoic Acids. Chem. Phys. Lipids 1984, 35, 87–101. [Google Scholar] [CrossRef]
- Gardner, H.W. Decomposition of linoleic acid hydroperoxides. Enzymic reactions compared with nonenzymic. J. Agric. Food Chem. 1975, 23, 129–136. [Google Scholar] [CrossRef]
- Gardner, H.W.; Kleiman, R. Degradation of Linoleic Acid Hydroperoxides by a Cysteine FeCl3 Catalyst as a Model for Similar Biochemical Reactions: II. Specificity in Formation of Fatty Acid Epoxides. Biochim. Et Biophys. Acta (BBA)-Lipids Lipid Metab. 1981, 665, 113–125. [Google Scholar] [CrossRef]
- Dix, T.A.; Marnett, L.J. Conversion of Linoleic Acid Hydroperoxide to Hydroxy, Keto, Epoxyhydroxy, and Trihydroxy Fatty Acids by Hematin. J. Biol. Chem. 1985, 260, 5351–5357. [Google Scholar] [CrossRef] [PubMed]
- Gardner, H.W. Oxygen Radical Chemistry of Polyunsaturated Fatty Acids. Free. Radic. Biol. Med. 1989, 7, 65–86. [Google Scholar] [CrossRef]
- Hamberg, M.; Gotthammar, B. A New Reaction of Unsaturated Fatty Acid Hydroperoxides: Formation of 11-Hydroxy-12,13-Epoxy-9-Octadecenoic Acid from 13-Hydroperoxy-9,11-Octadecadienoic Acid. Lipids 1973, 8, 737–744. [Google Scholar] [CrossRef]
- Chang, M.S.; Boeglin, W.E.; Guengerich, F.P.; Brash, A.R. Cytochrome P450-Dependent Transformations of 15R- and 15S-Hydroperoxyeicosatetraenoic Acids: Stereoselective Formation of Epoxy Alcohol Products. Biochemistry 1996, 35, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Song, W.C.; Baertschi, S.W.; Boeglin, W.E.; Harris, T.M.; Brash, A.R. Formation of Epoxyalcohols by a Purified Allene Oxide Synthase. Implications for the Mechanism of Allene Oxide Synthesis. J. Biol. Chem. 1993, 268, 6293–6298. [Google Scholar] [CrossRef] [PubMed]
- Hughes, R.K.; Yousafzai, F.K.; Ashton, R.; Chechetkin, I.R.; Fairhurst, S.A.; Hamberg, M.; Casey, R. Evidence for Communality in the Primary Determinants of CYP74 Catalysis and of Structural Similarities between CYP74 and Classical Mammalian P450 Enzymes. Proteins 2008, 72, 1199–1211. [Google Scholar] [CrossRef]
- Hoffmann, I.; Oliw, E.H. Discovery of a Linoleate 9S-Dioxygenase and an Allene Oxide Synthase in a Fusion Protein of Fusarium Oxysporum. J. Lipid Res. 2013, 54, 3471–3480. [Google Scholar] [CrossRef]
- Hoffmann, I.; Jernerén, F.; Oliw, E.H. Expression of Fusion Proteins of Aspergillus Terreus Reveals a Novel Allene Oxide Synthase. J. Biol. Chem. 2013, 288, 11459–11469. [Google Scholar] [CrossRef]
- Graveland, A. Enzymatic Oxidations of Linoleic Acid and Glycerol-1-Monolinoleate in Doughs and Flour-Water Suspensions. J. Am. Oil Chem. Soc. 1970, 47, 352–361. [Google Scholar] [CrossRef]
- Panossian, A.G.; Avetissian, G.M.; Mnatsakanian, V.A.; Batrakov, S.G.; Vartanian, S.A.; Gabrielian, E.S.; Amroyan, E.A. Unsaturated Polyhydroxy Acids Having Prostaglandin-Like Activity from Bryonia Alba II. Major Components. Planta Med. 1983, 47, 17–25. [Google Scholar] [CrossRef]
- Gorshkov, V.Y.; Toporkova, Y.Y.; Tsers, I.D.; Smirnova, E.O.; Ogorodnikova, A.V.; Gogoleva, N.E.; Parfirova, O.I.; Petrova, O.E.; Gogolev, Y.V. Differential Modulation of the Lipoxygenase Cascade during Typical and Latent Pectobacterium Atrosepticum Infections. Ann. Bot. 2021, 129, 271–286. [Google Scholar] [CrossRef] [PubMed]
- Toporkova, Y.Y.; Gorina, S.S.; Bessolitsyna, E.K.; Smirnova, E.O.; Fatykhova, V.S.; Brühlmann, F.; Ilyina, T.M.; Mukhtarova, L.S.; Grechkin, A.N. Double Function Hydroperoxide Lyases/Epoxyalcohol Synthases (CYP74C) of Higher Plants: Identification and Conversion into Allene Oxide Synthases by Site-Directed Mutagenesis. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2018, 1863, 369–378. [Google Scholar] [CrossRef]
- Gorina, S.S.; Iljina, T.M.; Mukhtarova, L.S.; Toporkova, Y.Y.; Grechkin, A.N. Detection of Unprecedented CYP74 Enzyme in Mammal: Hydroperoxide Lyase CYP74C44 of the Bat Sturnira Hondurensis. Int. J. Mol. Sci. 2022, 23, 8009. [Google Scholar] [CrossRef] [PubMed]
- Grechkin, A.N.; Mukhtarova, L.S.; Latypova, L.R.; Gogolev, Y.; Toporkova, Y.Y.; Hamberg, M. Tomato CYP74C3 Is a Multifunctional Enzyme Not Only Synthesizing Allene Oxide but Also Catalyzing Its Hydrolysis and Cyclization. ChemBioChem 2008, 9, 2498–2505. [Google Scholar] [CrossRef]
- Grechkin, A.N.; Lantsova, N.V.; Mukhtarova, L.S.; Khairutdinov, B.I.; Gorina, S.S.; Iljina, T.M.; Toporkova, Y.Y. Distinct Mechanistic Behaviour of Tomato CYP74C3 and Maize CYP74A19 Allene Oxide Synthases: Insights from Trapping Experiments and Allene Oxide Isolation. Int. J. Mol. Sci. 2023, 24, 2230. [Google Scholar] [CrossRef] [PubMed]
- Toporkova, Y.Y.; Smirnova, E.O.; Mukhtarova, L.S.; Gorina, S.S.; Grechkin, A.N. Catalysis by Allene Oxide Synthases (CYP74A and CYP74C): Alterations by the Phe/Leu Mutation at the SRS-1 Region. Phytochemistry 2020, 169, 112152. [Google Scholar] [CrossRef] [PubMed]
- Toporkova, Y.Y.; Askarova, E.K.; Gorina, S.S.; Ogorodnikova, A.V.; Mukhtarova, L.S.; Grechkin, A.N. Epoxyalcohol Synthase Activity of the CYP74B Enzymes of Higher Plants. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2020, 1865, 158743. [Google Scholar] [CrossRef]
- Gorina, S.S.; Mukhitova, F.K.; Ilyina, T.M.; Toporkova, Y.Y.; Grechkin, A.N. Detection of Unprecedented Allene Oxide Synthase Member of CYP74B Subfamily: CYP74B33 of Carrot (Daucus carota). Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2019, 1864, 1580–1590. [Google Scholar] [CrossRef]
- Toporkova, Y.Y.; Smirnova, E.O.; Iljina, T.M.; Mukhtarova, L.S.; Gorina, S.S.; Grechkin, A.N. The CYP74B and CYP74D Divinyl Ether Synthases Possess a Side Hydroperoxide Lyase and Epoxyalcohol Synthase Activities That Are Enhanced by the Site-Directed Mutagenesis. Phytochemistry 2020, 179, 112512. [Google Scholar] [CrossRef]
- Gorina, S.S.; Toporkova, Y.Y.; Mukhtarova, L.S.; Chechetkin, I.R.; Khairutdinov, B.I.; Gogolev, Y.V.; Grechkin, A.N. Detection and Molecular Cloning of CYP74Q1 Gene: Identification of Ranunculus acris Leaf Divinyl Ether Synthase. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2014, 1841, 1227–1233. [Google Scholar] [CrossRef]
- Gorina, S.S.; Toporkova, Y.Y.; Mukhtarova, L.S.; Smirnova, E.O.; Chechetkin, I.R.; Khairutdinov, B.I.; Gogolev, Y.V.; Grechkin, A.N. Oxylipin Biosynthesis in Spikemoss Selaginella moellendorffii: Molecular Cloning and Identification of Divinyl Ether Synthases CYP74M1 and CYP74M3. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2016, 1861, 301–309. [Google Scholar] [CrossRef]
- Gorina, S.S.; Mukhtarova, L.S.; Iljina, T.M.; Toporkova, Y.Y.; Grechkin, A.N. Detection of divinyl ether synthase CYP74H2 biosynthesizing (11Z)-etheroleic and (1′Z)-colnelenic acids in asparagus (Asparagus officinalis L.). Phytochemistry 2022, 200, 113212. [Google Scholar] [CrossRef]
- Grechkin, A.N.; Ilyasov, A.V.; Hamberg, M. On the mechanism of biosynthesis of divinyl ether oxylipins by enzyme from garlic bulbs. Eur. J. Biochem. 1997, 245, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Toporkova, Y.Y.; Smirnova, E.O.; Gorina, S.S.; Mukhtarova, L.S.; Grechkin, A.N. Detection of the First Higher Plant Epoxyalcohol Synthase: Molecular Cloning and Characterisation of the CYP74M2 Enzyme of Spikemoss Selaginella moellendorffii. Phytochemistry 2018, 156, 73–82. [Google Scholar] [CrossRef]
- Toporkova, Y.Y.; Fatykhova, V.S.; Gorina, S.S.; Mukhtarova, L.S.; Grechkin, A.N. Epoxyalcohol Synthase RjEAS (CYP74A88) from the Japanese Buttercup (Ranunculus japonicus): Cloning and Characterization of Catalytic Properties. Biochemistry 2019, 84, 171–180. [Google Scholar] [CrossRef]
- Froehlich, J.E.; Itoh, A.; Howe, G.A. Tomato allene oxide synthase and fatty acid hydroperoxide lyase, two cytochrome P450s involved in oxylipin metabolism, are targeted to different membranes of chloroplast envelope. Plant Physiol. 2001, 125, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Toporkova, Y.Y.; Fatykhova, V.S.; Gogolev, Y.V.; Khairutdinov, B.I.; Mukhtarova, L.S.; Grechkin, A.N. Epoxyalcohol Synthase of Ectocarpus Siliculosus. First CYP74-Related Enzyme of Oxylipin Biosynthesis in Brown Algae. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2017, 1862, 167–175. [Google Scholar] [CrossRef]
- Toporkova, Y.Y.; Gorina, S.S.; Mukhitova, F.K.; Hamberg, M.; Ilyina, T.M.; Mukhtarova, L.S.; Grechkin, A.N. Identification of CYP443D1 (CYP74 Clan) of Nematostella vectensis as a First Cnidarian Epoxyalcohol Synthase and Insights into Its Catalytic Mechanism. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2017, 1862, 1099–1109. [Google Scholar] [CrossRef]
- Gorina, S.S.; Toporkova, Y.Y.; Mukhtarova, L.S.; Grechkin, A.N. The CYP443C1 (CYP74 Clan) Cytochrome of Sea Anemone Nematostella Vectensis—The First Metazoan Enzyme Possessing Hydroperoxide Lyase/Epoxyalcohol Synthase Activity. Dokl. Biochem. Biophys. 2019, 486, 192–196. [Google Scholar] [CrossRef]
- Toporkova, Y.Y.; Smirnova, E.O.; Lantsova, N.V.; Mukhtarova, L.S.; Grechkin, A.N. Detection of the First Epoxyalcohol Synthase/Allene Oxide Synthase (CYP74 Clan) in the Lancelet (Branchiostoma belcheri, Chordata). Int. J. Mol. Sci. 2021, 22, 4737. [Google Scholar] [CrossRef]
- Toporkova, Y.Y.; Smirnova, E.O.; Mukhtarova, L.S.; Grechkin, A.N. Lipoxygenase Pathway in Brown Algae: The Biosynthesis of Novel Oxylipins “ectocarpins” by Hydroperoxide Bicyclase CYP5164A3 of Ectocarpus Siliculosus. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2022, 1867, 159205. [Google Scholar] [CrossRef]
- Grechkin, A.N.; Lantsova, N.V.; Khairutdinov, B.I.; Toporkova, Y.Y. Hydroperoxide Bicyclase CYP50918A1 of Plasmodiophora Brassicae (Rhizaria, SAR): Detection of Novel Enzyme of Oxylipin Biosynthesis. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2021, 1866, 159042. [Google Scholar] [CrossRef] [PubMed]
- Gotoh, O. Substrate Recognition Sites in Cytochrome P450 Family 2 (CYP2) Proteins Inferred from Comparative Analyses of Amino Acid and Coding Nucleotide Sequences. J. Biol. Chem. 1992, 267, 83–90. [Google Scholar] [PubMed]
- Gorina, S.S.; Smirnova, E.O.; Mukhtarova, L.S.; Toporkova, Y.Y.; Grechkin, A.N. Conversion of Tomato Allene Oxide Synthase LeAOS3 (CYP74C3) into Epoxyalcohol Synthase by Site-Directed Mutagenesis. Dokl. Biochem. Biophys. 2018, 483, 329–332. [Google Scholar] [CrossRef]
- Toporkova, Y.Y.; Gogolev, Y.V.; Mukhtarova, L.S.; Grechkin, A.N. Determinants Governing the CYP74 Catalysis: Conversion of Allene Oxide Synthase into Hydroperoxide Lyase by Site-Directed Mutagenesis. FEBS Lett. 2008, 582, 3423–3428. [Google Scholar] [CrossRef]
- Toporkova, Y.Y.; Ermilova, V.S.; Gorina, S.S.; Mukhtarova, L.S.; Osipova, E.V.; Gogolev, Y.V.; Grechkin, A.N. Structure-Function Relationship in the CYP74 Family: Conversion of Divinyl Ether Synthases into Allene Oxide Synthases by Site-Directed Mutagenesis. FEBS Lett. 2013, 587, 2552–2558. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wei, K. Comparative Functional Genomics Analysis of Cytochrome P450 Gene Superfamily in Wheat and Maize. BMC Plant Biol. 2020, 20, 93. [Google Scholar] [CrossRef]
- Pandian, B.A.; Sathishraj, R.; Djanaguiraman, M.; Prasad, P.V.V.; Jugulam, M. Role of Cytochrome P450 Enzymes in Plant Stress Response. Antioxidants 2020, 9, 454. [Google Scholar] [CrossRef]
- Hansen, C.C.; Nelson, D.R.; Møller, B.L.; Werck-Reichhart, D. Plant Cytochrome P450 Plasticity and Evolution. Mol. Plant 2021, 14, 1244–1265. [Google Scholar] [CrossRef]
- Xu, L.-H.; Du, Y.-L. Rational and Semi-Rational Engineering of Cytochrome P450s for Biotechnological Applications. Synth. Syst. Biotechnol. 2018, 3, 283–290. [Google Scholar] [CrossRef]
- Nelson, D.R. Cytochrome P450 Diversity in the Tree of Life. Biochim. Biophys. Acta Proteins Proteom. 2018, 1866, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Omura, T.; Gotoh, O. Evolutionary Origin of Mitochondrial Cytochrome P450. J. Biochem. 2017, 161, 399–407. [Google Scholar] [CrossRef][Green Version]
- Lacchini, E.; Venegas-Molina, J.; Goossens, A. Structural and Functional Diversity in Plant Specialized Metabolism Signals and Products: The Case of Oxylipins and Triterpenes. Curr. Opin. Plant Biol. 2023, 74, 102371. [Google Scholar] [CrossRef]
- Rustgi, S.; Springer, A.; Kang, C.; von Wettstein, D.; Reinbothe, C.; Reinbothe, S.; Pollmann, S. Allene oxide synthase and hydroperoxide lyase, two non-canonical cytochrome P450s in Arabidopsis thaliana and their different roles in plant defense. Int. J. Mol. Sci. 2019, 20, 3064. [Google Scholar] [CrossRef] [PubMed]
- Carella, P. Xylem-Mobile Oxylipins Are Critical Regulators of Induced Systemic Resistance in Maize. Plant Cell 2020, 32, 13–14. [Google Scholar] [CrossRef]
- Wang, K.-D.; Borrego, E.J.; Kenerley, C.M.; Kolomiets, M.V. Oxylipins Other Than Jasmonic Acid Are Xylem-Resident Signals Regulating Systemic Resistance Induced by Trichoderma Virens in Maize. Plant Cell 2020, 32, 166–185. [Google Scholar] [CrossRef] [PubMed]
- Berg-Falloure, K.M.; Kolomiets, M.V. Ketols Emerge as Potent Oxylipin Signals Regulating Diverse Physiological Processes in Plants. Plants 2023, 12, 2088. [Google Scholar] [CrossRef]
- Ruocco, N.; Albarano, L.; Esposito, R.; Zupo, V.; Costantini, M.; Ianora, A. Multiple Roles of Diatom-Derived Oxylipins within Marine Environments and Their Potential Biotechnological Applications. Mar. Drugs 2020, 18, 342. [Google Scholar] [CrossRef]
- Masui, H.; Kondo, T.; Kojima, M. An Antifungal Compound, 9,12,13-Trihydroxy-(E)-10-Octadecenoic Acid, from Colocasia antiquorum Inoculated with Ceratocystis fimbriata. Phytochemistry 1989, 28, 2613–2615. [Google Scholar] [CrossRef]
- Weber, H.; Chételat, A.; Caldelari, D.; Farmer, E.E. Divinyl ether fatty acid synthesis in late blight–diseased potato leaves. Plant Cell 1999, 11, 485–493. [Google Scholar] [CrossRef]
- Toporkova, Y.Y.; Bessolitsyna, E.K.; Smirnova, E.O.; Gorina, S.S.; Petrova, O.E.; Mukhtarova, L.S.; Grechkin, A.N. Antimicrobial Activity of Geometric Isomers of Etherolenic Acid—The Products of Plant Lipoxygenase Cascade. Dokl. Biochem. Biophys. 2018, 480, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Oldham, M.L.; Brash, A.R.; Newcomer, M.E. The Structure of Coral Allene Oxide Synthase Reveals a Catalase Adapted for Metabolism of a Fatty Acid Hydroperoxide. Proc. Natl. Acad. Sci. USA 2005, 102, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Koljak, R.; Boutaud, O.; Shieh, B.H.; Samel, N.; Brash, A.R. Identification of a naturally occurring peroxidase-lipoxygenase fusion protein. Science 1997, 277, 1994–1996. [Google Scholar] [CrossRef]
- Varvas, K.; Järving, I.; Koljak, R.; Valmsen, K.; Brash, A.R.; Samel, N. Evidence of a Cyclooxygenase-Related Prostaglandin Synthesis in Coral: The allene oxide pathway is not involved in prostaglandin biosynthesis. J. Biol. Chem. 1999, 274, 9923–9929. [Google Scholar] [CrossRef]
- Lõhelaid, H.; Teder, T.; Tõldsepp, K.; Ekins, M.; Samel, N. Up-regulated expression of AOS-LOXa and increased eicosanoid synthesis in response to coral wounding. PLoS ONE 2014, 9, e89215. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Boeglin, W.E.; Zheng, Y.; Schneider, C.; Brash, A.R. Evidence for an Ionic Intermediate in the Transformation of Fatty Acid Hydroperoxide by a Catalase-Related Allene Oxide Synthase from the Cyanobacterium acaryochloris Marina. J. Biol. Chem. 2009, 284, 22087–22098. [Google Scholar] [CrossRef]
- Teder, T.; Lõhelaid, H.; Boeglin, W.E.; Calcutt, W.M.; Brash, A.R.; Samel, N. A Catalase-Related Hemoprotein in Coral Is Specialized for Synthesis of Short-Chain Aldehydes: Discovery of p450-type hydroperoxide lyase activity in a catalase. J. Biol. Chem. 2015, 290, 19823–19832. [Google Scholar] [CrossRef]
- Mashhadi, Z.; Newcomer, M.E.; Brash, A.R. The Thr–His connection on the distal heme of catalase-related hemoproteins: A hallmark of reaction with fatty acid hydroperoxides. ChemBioChem 2016, 17, 2000–2006. [Google Scholar] [CrossRef]
- Brash, A.R. Mechanistic Aspects of CYP74 Allene Oxide Synthases and Related Cytochrome P450 Enzymes. Phytochemistry 2009, 70, 1522–1531. [Google Scholar] [CrossRef]
- Feussner, I.; Wasternack, C. The Lipoxygenase Pathway. Annu. Rev. Plant Biol. 2002, 53, 275–297. [Google Scholar] [CrossRef]
- Hughes, R.K.; De Domenico, S.; Santino, A. Plant Cytochrome CYP74 Family: Biochemical Features, Endocellular Localisation, Activation Mechanism in Plant Defence and Improvements for Industrial Applications. ChemBioChem 2009, 10, 1122–1133. [Google Scholar] [CrossRef]
- Vick, B.A.; Zimmerman, D.C. Pathways of fatty acid hydroperoxide metabolism in spinach leaf chloroplasts. Plant Physiol. 1987, 85, 1073–1078. [Google Scholar] [CrossRef]
- Droillard, M.J.; Rouet-Mayer, M.A.; Bureau, J.M.; Lauriere, C. Membrane-Associated and Soluble Lipoxygenase Isoforms in Tomato pericarp (Characterization and Involvement in Membrane Alterations). Plant Physiol. 1993, 103, 1211–1219. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Royo, J.; Vancanneyt, G.; Pérez, A.G.; Sanz, C.; Störmann, K.; Rosahl, S.; Sánchez-Serrano, J.J. Characterization of three potato lipoxygenases with distinct enzymatic activities and different organ-specific and wound-regulated expression patterns. J. Biol. Chem. 1996, 271, 21012–21019. [Google Scholar] [CrossRef]
- Ishiguro, S.; Kawai-Oda, A.; Ueda, J.; Nishida, I.; Okada, K. The DEFECTIVE IN ANTHER DEHISCIENCE gene encodes a novel phospholipase A1 catalyzing the initial step of jasmonic acid biosynthesis, which synchronizes pollen maturation, anther dehiscence, and flower opening in Arabidopsis. Plant Cell 2001, 13, 2191–2209. [Google Scholar] [CrossRef]
- Stelmach, B.A.; Müller, A.; Hennig, P.; Gebhardt, S.; Schubert-Zsilavecz, M.; Weiler, E.W. A novel class of oxylipins, sn1-O-′12-oxophytodienoyl)-sn2-O-(hexadecatrienoyl)-monogalactosyl diglycerides, from Arabidopsis thaliana. J. Biol. Chem. 2001, 276, 12832–12838. [Google Scholar] [CrossRef] [PubMed]
- Porta, H.; Rocha-Sosa, M. Plant lipoxygenases. Physiological and molecular features. Plant Physiol. 2002, 130, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H.; Yokota, K. Characterization of metabolic pathway of linoleic acid 9-hydroperoxide in cytosolic fraction of potato tubers and identification of reaction products. Appl. Biochem. Biotechnol. 2004, 118, 115–132. [Google Scholar] [CrossRef]
- Xu, Y.; Ishida, H.; Reisen, D.; Hanson, M.R. Upregulation of a tonoplast-localized cytochrome P450 during petal senescence in Petunia inflate. BMC Plant Biol. 2006, 6, 8. [Google Scholar] [CrossRef]
- Wasternack, C. Jasmonates: An Update on Biosynthesis, Signal Transduction and Action in Plant Stress Response, Growth and Development. Ann. Bot. 2007, 100, 681–697. [Google Scholar] [CrossRef]
- De Domenico, S.; Tsesmetzis, N.; Di Sansebastiano, G.P.; Hughes, R.K.; Casey, R.; Santino, A. Subcellular localisation of Medicago truncatula 9/13-hydroperoxide lyase reveals a new localisation pattern and activation mechanism for CYP74C enzymes. BMC Plant Biol. 2007, 7, 58. [Google Scholar] [CrossRef] [PubMed]
- Acosta, I.F.; Farmer, E.E. Jasmonates. Arab. Book 2010, 8, e0129. [Google Scholar] [CrossRef]
- Wasternack, C.; Song, S. Jasmonates: Biosynthesis, metabolism, and signaling by proteins activating and repressing transcription. J. Exp. Bot. 2017, 68, 1303–1321. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yu, G.; Cao, C.; Liu, P. Metabolism, signaling, and transport of jasmonates. Plant Commun. 2021, 2, 100231. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.; Werck-Reichhart, D. A P450-Centric View of Plant Evolution. Plant J. 2011, 66, 194–211. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toporkova, Y.Y.; Smirnova, E.O.; Gorina, S.S. Epoxyalcohol Synthase Branch of Lipoxygenase Cascade. Curr. Issues Mol. Biol. 2024, 46, 821-841. https://doi.org/10.3390/cimb46010053
Toporkova YY, Smirnova EO, Gorina SS. Epoxyalcohol Synthase Branch of Lipoxygenase Cascade. Current Issues in Molecular Biology. 2024; 46(1):821-841. https://doi.org/10.3390/cimb46010053
Chicago/Turabian StyleToporkova, Yana Y., Elena O. Smirnova, and Svetlana S. Gorina. 2024. "Epoxyalcohol Synthase Branch of Lipoxygenase Cascade" Current Issues in Molecular Biology 46, no. 1: 821-841. https://doi.org/10.3390/cimb46010053
APA StyleToporkova, Y. Y., Smirnova, E. O., & Gorina, S. S. (2024). Epoxyalcohol Synthase Branch of Lipoxygenase Cascade. Current Issues in Molecular Biology, 46(1), 821-841. https://doi.org/10.3390/cimb46010053




