Preventive Impact of Long-Term Ingestion of Chestnut Honey on Glucose Disorders and Neurodegeneration in Obese Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Diets
2.2. Metabolic Parameters
2.3. Brain Tissue Preparation
2.4. TUNEL Assay
2.5. Western Blotting and PCR
2.6. RT2Profiler PCR Array
2.7. Statistical Analysis
3. Results
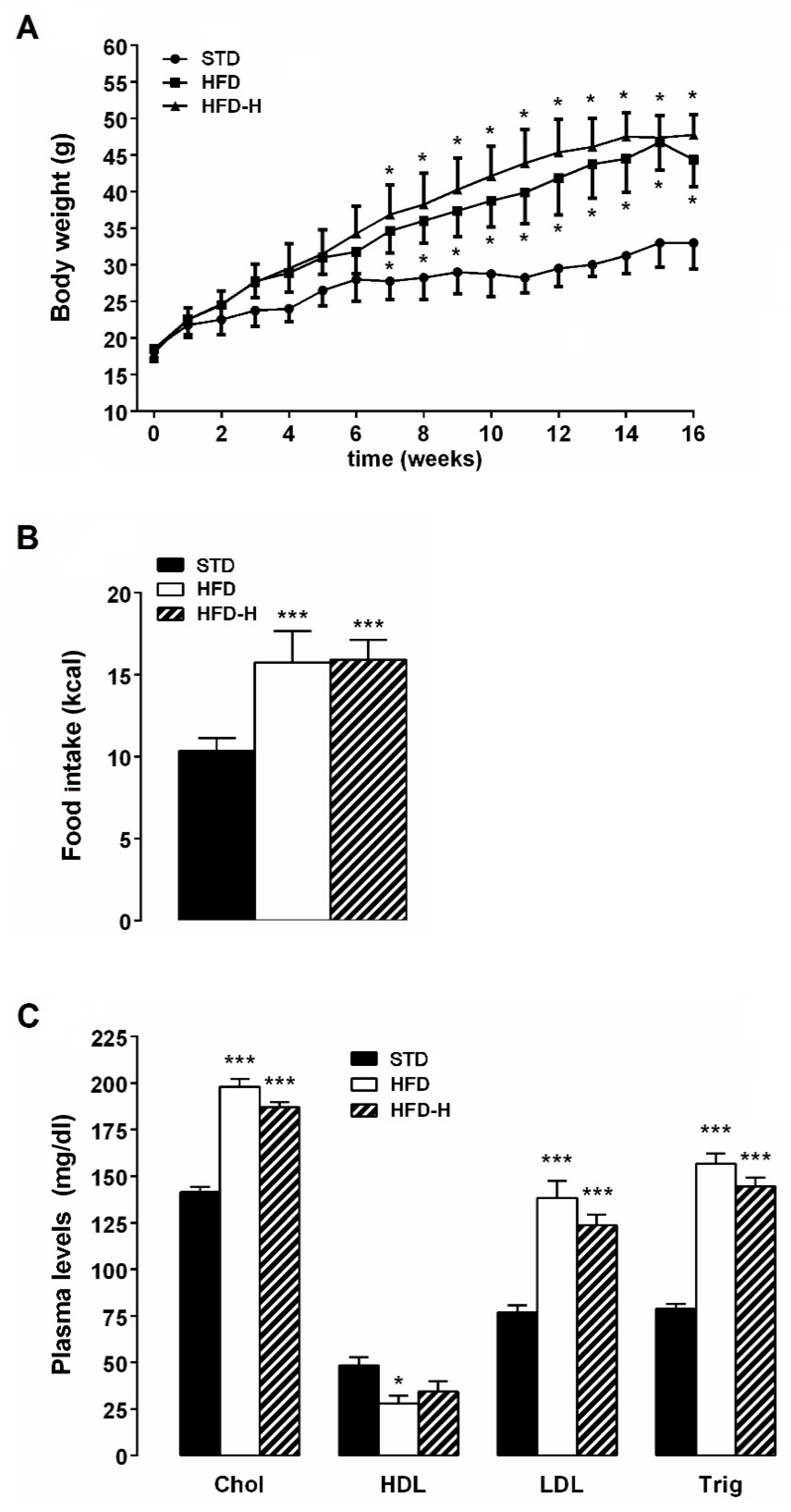
3.1. Honey Ingestion and Metabolic Parameters
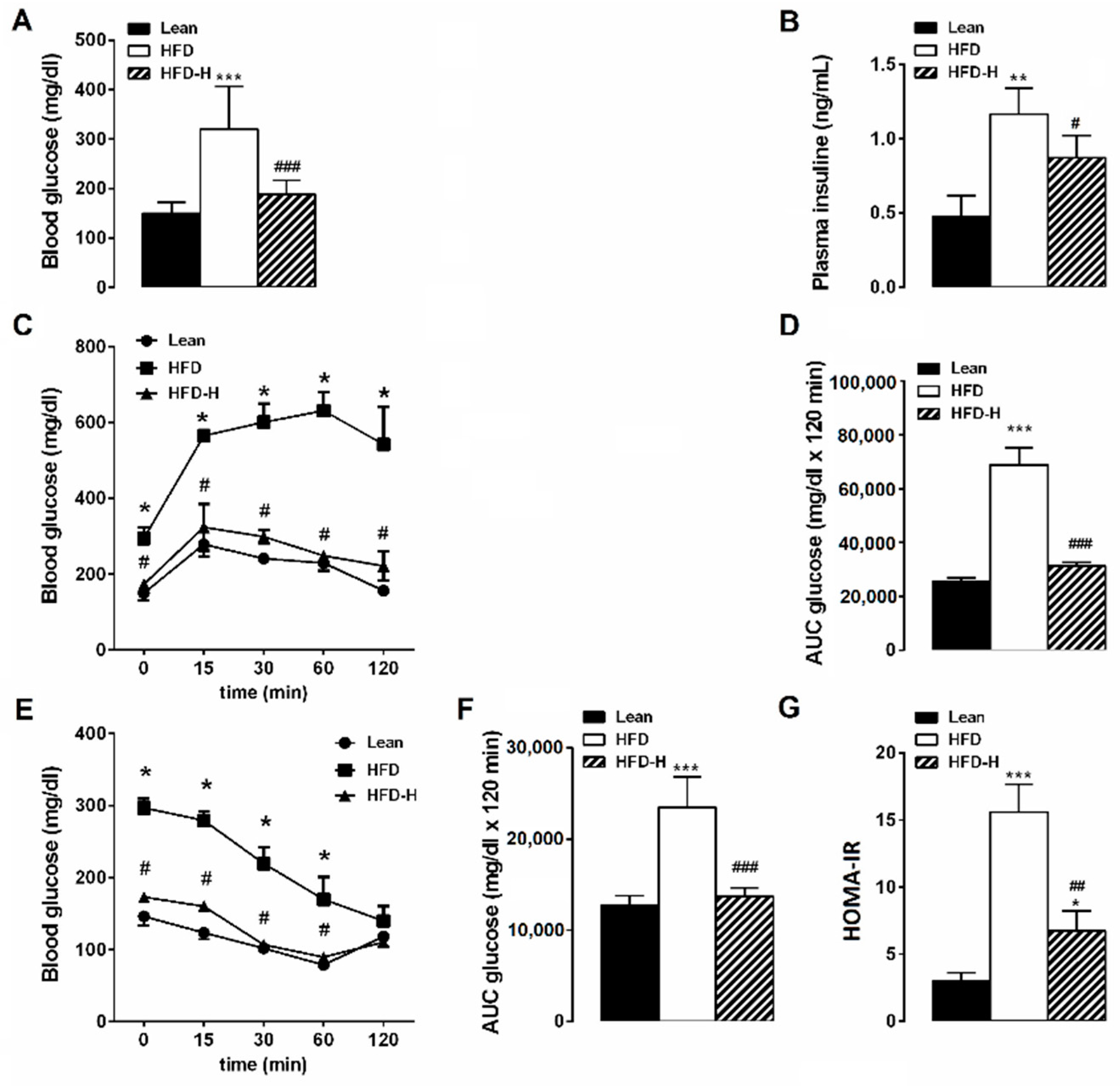
3.2. Honey Ingestion Improves Glucose Metabolism in HFD Mice
3.3. Honey Ingestion Mitigates Neurodegeneration Induced by HFD
3.4. Honey Ingestion Improves Insulin Signaling in Brains of HFD Mice
3.5. Honey Supplementation Modifies Blood Levels of Leptin and Adiponectin
4. Discussion
5. Conclusions

Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Menken, M.; Munsat, T.L.; Toole, J.F. The global burden of disease study: Implications for neurology. Arch. Neurol. 2000, 57, 418–420. [Google Scholar] [CrossRef] [PubMed]
- Amato, A.; Mulè, F. Protective potential of glucagon like peptide 2 (GLP-2) against the neurodegeneration. Neural Regen. Res. 2019, 14, 1901–1902. [Google Scholar] [PubMed]
- Iriti, M.; Vitalini, S.; Fico, G.; Faoro, F. Neuroprotective Herbs and Foods from Different Traditional Medicines and Diets. Molecules 2010, 15, 3517–3555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reitz, C.; Brayne, C.; Mayeux, R. Epidemiology of Alzheimer disease. Nat. Rev. Neurol. 2011, 7, 137. [Google Scholar] [CrossRef] [PubMed]
- Pugazhenth, S.; Qin, L.; Reddy, P.H. Common neurodegenerative pathways in obesity, diabetes, and Alzheimer’s disease. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1037–1045. [Google Scholar] [CrossRef] [PubMed]
- Kroner, Z. The relationship between Alzheimer’s disease and diabetes: Type 3 diabetes? Altern. Med. Rev. 2009, 14, 373–379. [Google Scholar]
- Singh-Manoux, A.; Dugravot, A.; Shipley, M.; Brunner, E.J.; Elbaz, A.; Sabia, S.; Kivimaki, M. Obesity trajectories and risk of dementia: 28 years of follow-up in the Whitehall II Study. Alzheimer’s Dement. 2018, 14, 178–186. [Google Scholar] [CrossRef]
- Van Dyken, P.; Lacoste, B. Impact of Metabolic Syndrome on Neuroinflammation and the Blood-Brain Barrier. Front. Neurosci. 2018, 12, 930. [Google Scholar] [CrossRef]
- Keshk, W.A.; Ibrahim, M.A.; Shalaby, S.M.; Zalat, Z.A.; Elseady, W.S. Redox status, inflammation, necroptosis and inflammasome as indispensable contributors to high fat diet (HFD)-induced neurodegeneration; Effect of N-acetylcysteine (NAC). Arch. Biochem. Biophys. 2020, 680, 108227. [Google Scholar] [CrossRef]
- Nuzzo, D.; Picone, P.; Baldassano, S.; Caruana, L.; Messina, E.; Gammazza, A.; Cappello, F.; Mulè, F.; Carlo, M. Insulin resistance as common molecular denominator linking obesity to Alzheimer’s disease. Curr. Alzheimer Res. 2015, 12, 723–735. [Google Scholar] [CrossRef] [Green Version]
- Stranahan, A.M.; Norman, E.D.; Lee, K.; Cutler, R.G.; Telljohann, R.S.; Egan, J.M.; Mattson, M.P. Diet-induced insulin resistance impairs hippocampal synaptic plasticity and cognition in middle-aged rats. Hippocampus 2008, 18, 1085–1088. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stacchiotti, A.; Corsetti, G. Natural Compounds and Autophagy: Allies against Neurodegeneration. Front. Cell Dev. Biol. 2020, 8, 555409. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.W.; Nash, P.; Buttar, H.S.; Griffiths, K.; Singh, R.; De Meester, F.; Horiuchi, R.; Takahashi, T. The Role of Food Antioxidants, Benefits of Functional Foods, and Influence of Feeding Habits on the Health of the Older Person: An Overview. Antioxidants 2017, 6, 81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasnieza Mohd Rosli, N.; Mastura Yahya, H.; Shahar, S.; Wahida Ibrahim, F.; Fadilah Rajab, N. Alzheimer’s Disease and Functional Foods: An Insight on Neuroprotective Effect of its Combination. Pak. J. Biol. Sci. 2020, 23, 575–589. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, D.; Galizzi, G.; Amato, A.; Terzo, S.; Picone, P.; Cristaldi, L.; Mule, F.; Di Carlo, M. Regular Intake of Pistachio Mitigates the Deleterious Effects of a High Fat-Diet in the Brain of Obese Mice. Antioxidants 2020, 9, 317. [Google Scholar] [CrossRef] [Green Version]
- Nuzzo, D.; Amato, A.; Picone, P.; Terzo, S.; Galizzi, G.; Bonina, F.P.; Mulè, F.; Di Carlo, M. A Natural Dietary Supplement with a Combination of Nutrients Prevents Neurodegeneration Induced by a High Fat Diet in Mice. Nutrients 2018, 10, 1130. [Google Scholar] [CrossRef] [Green Version]
- Terzo, S.; Mulè, F.; Amato, A. Honey and obesity-related dysfunctions: A summary on health benefits. J. Nutr. Biochem. 2020, 82, 108401. [Google Scholar] [CrossRef]
- Aziz, M.S.; Giribabu, N.; Rao, P.V.; Salleh, N. Pancreatoprotective effects of Geniotrigona thoracica stingless bee honey in streptozotocin-nicotinamide-induced male diabetic rats. Biomed. Pharmacother. 2017, 89, 135–145. [Google Scholar] [CrossRef]
- Galal, R.M.; Zaki, H.F.; Seif El-Nasr, M.M.; Agha, A.M. Potential protective effect of honey against paracetamol-induced hepatotoxicity. Arch. Iran Med. 2012, 15, 674–680. [Google Scholar]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S.; Sirajudeen, K.N.; Salleh, S.; Gurtu, S. Honey supplementation in spontaneously hypertensive rats elicits antihypertensive effect via amelioration of renal oxidative stress. Oxid. Med. Cell. Longev. 2012, 2012, 374037. [Google Scholar] [CrossRef] [Green Version]
- Nemoseck, T.M.; Carmody, E.G.; Furchner-Evanson, A.; Gleason, M.; Li, A.; Potter, H.; Rezende, L.M.; Lane, K.J.; Kern, M. Honey promotes lower weight gain, adiposity, and triglycerides than sucrose in rats. Nutr. Res. 2011, 31, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Hashim, K.N.; Chin, K.Y.; Ahmad, F. The Mechanism of Honey in Reversing Metabolic Syndrome. Molecules 2021, 26, 808. [Google Scholar] [CrossRef] [PubMed]
- Terzo, S.; Mulè, F.; Caldara, G.F.; Baldassano, S.; Puleio, R.; Vitale, M.; Cassata, G.; Ferrantelli, V.; Amato, A. Pistachio Consumption Alleviates Inflammation and Improves Gut Microbiota Composition in Mice Fed a High-Fat Diet. Int. J. Mol. Sci. 2020, 21, 365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nasuti, C.; Gabbianelli, R.; Falcioni, G.; Cantalamessa, F. Antioxidative and gastroprotective activities of anti-inflammatory formulations derived from chestnut honey in rats. Nutr. Res. 2006, 26, 130–137. [Google Scholar] [CrossRef]
- Moroz, N.; Tong, M.; Longato, L.; Xu, H.; de la Monte, S.M. Limited alzheimer-type neurodegeneration in experimental obesity and type 2 diabetes mellitus. J. Alzheimer’s Dis. 2008, 15, 29–44. [Google Scholar] [CrossRef]
- Frisardi, V.; Solfrizzi, V.; Seripa, D.; Capurso, C.; Santamato, A.; Sancarlo, D.; Vendemiale, G.; Pilotto, A.; Panza, F. Metabolic-cognitive syndrome: A cross-talk between metabolic syndrome and Alzheimer’s disease. Ageing Res. Rev. 2010, 9, 399–417. [Google Scholar] [CrossRef]
- Pedditzi, E.; Peters, R.; Beckett, N. The risk of overweight/obesity in mid-life and late life for the development of dementia: A systematic review and meta-analysis of longitudinal studies. Age Ageing 2016, 45, 14–21. [Google Scholar] [CrossRef] [Green Version]
- De la Monte, S.M. Insulin resistance and neurodegeneration: Progress towards the development of new therapeutics for Alzheimer’s disease. Drugs 2017, 77, 47–65. [Google Scholar] [CrossRef] [Green Version]
- Terzo, S.; Amato, A.; Mulè, F. From obesity to Alzheimer’s disease through insulin resistance. J. Diabetes Its Complicat. 2021, 18, 108026. [Google Scholar] [CrossRef]
- Mercer, J.G.; Archer, Z.A. Diet-induced obesity in the Sprague-Dawley rat: Dietary manipulations and their effect on hypothalamic neuropeptide energy balance systems. Biochem. Soc. Trans. 2005, 33, 1068–1072. [Google Scholar] [CrossRef]
- Sa-Nguanmoo, P.; Tanajak, P.; Kerdphoo, S.; Jaiwongkam, T.; Pratchayasakul, W.; Chattipakorn, N.; Chattipakorn, S.C. SGLT2-inhibitor and DPP-4 inhibitor improve brain function via attenuating mitochondrial dysfunction, insulin resistance, inflammation, and apoptosis in HFD-induced obese rats. Toxicol. Appl. Pharmacol. 2017, 333, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Pratchayasakul, W.; Sa-Nguanmoo, P.; Sivasinprasasn, S.; Pintana, H.; Tawinvisan, R.; Sripetchwandee, J.; Kumfu, S.; Chattipakorn, N.; Chattipakorn, S.C. Obesity accelerates cognitive decline by aggravating mitochondrial dysfunction, insulin resistance and synaptic dysfunction under estrogen-deprived conditions. Horm. Behav. 2015, 72, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Pintana, H.; Apaijai, N.; Pratchayasakul, W.; Chattipakorn, N.; Chattipakorn, S.C. Effects of metformin on learning and memory behaviors and brain mitochondrial functions in high fat diet induced insulin resistant rats. Life Sci. 2012, 91, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, D.; Baldassano, S.; Amato, A.; Picone, P.; Galizzi, G.; Caldara, G.F.; Di Carlo, M.; Mule, F. Glucagon-like peptide-2 reduces the obesity-associated inflammation in the brain. Neurobiol. Dis. 2019, 121, 296–304. [Google Scholar] [CrossRef]
- Galizzi, G.; Palumbo, L.; Amato, A.; Conigliaro, A.; Nuzzo, D.; Terzo, S.; Caruana, L.; Picone, P.; Alessandro, R.; Mule, F.; et al. Altered insulin pathway compromises mitochondrial function and quality control both in in vitro and in vivo model systems. Mitochondrion 2021, 60, 178–188. [Google Scholar] [CrossRef]
- Siino, V.; Amato, A.; Di Salvo, F.; Caldara, G.; Filogamo, M.; James, P.; Vasto, S. Impact of diet-induced obesity on the mouse brain phosphoproteome. J. Nutr. Biochem. 2018, 58, 102–109. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Nwobodo, N.N.; Akpan, J.L.; Okorie, U.A.; Ezeonu, C.T.; Ezeokpo, B.C.; Nwadike, K.I.; Erhiano, E.; Wahab, M.S.A.; Sulaiman, S.A. Nigerian Honey Ameliorates Hyperglycemia and Dyslipidemia in Alloxan-Induced Diabetic Rats. Nutrients 2016, 8, 95. [Google Scholar] [CrossRef] [Green Version]
- Farakla, I.; Koui, E.; Arditi, J.; Papageorgiou, I.; Bartzeliotou, A.; Papadopoulos, G.E.; Mantzou, A.; Papathanasiou, C.; Dracopoulou, M.; Papastamataki, M.; et al. Effect of honey on glucose and insulin concentrations in obese girls. Eur. J. Clin. Invest. 2019, 49, e13042. [Google Scholar] [CrossRef]
- De la Monte, S.M. Brain insulin resistance and deficiency as therapeutic targets in Alzheimer’s disease. Curr. Alzheimer Res. 2012, 9, 35–66. [Google Scholar] [CrossRef]
- Chepulis, L.M.; Starkey, N.J.; Waas, J.R.; Molan, P.C. The effects of long-term honey, sucrose or sugar-free diets on memory and anxiety in rats. Physiol. Behav. 2009, 97, 359–368. [Google Scholar] [CrossRef]
- Akanmu, M.A.; Olowookere, T.A.; Atunwa, S.A.; Ibrahim, B.O.; Lamidi, O.F.; Adams, P.A.; Ajimuda, B.O.; Adeyemo, L.E. Neuropharmacological effects of Nigerian honey in mice. Afr. J. Tradit. Complementary Altern. Med. 2011, 8, 230–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Rahbi, B.; Zakaria, R.; Othman, Z.; Hassan, A.; Mohd Ismail, Z.I.; Muthuraju, S. Tualang honey supplement improves memory performance and hippocampal morphology in stressed ovariectomized rats. Acta Histochem. 2014, 116, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Rosli, N.H.M.; Yahya, H.M.; Ibrahim, F.W.; Shahar, S.; Ismail, I.S.; Azam, A.A.; Rajab, N.F. Serum Metabolomics Profiling of Commercially Mixed Functional Foods-Effects in Beta-Amyloid Induced Rats Measured Using 1H NMR Spectroscopy. Nutrients 2020, 12, 3812. [Google Scholar] [CrossRef] [PubMed]
- Al-Himyari, F.A. The use of honey as a natural preventive therapy of cognitive decline and dementia in the Middle East. Alzheimer’s Dement. 2009, 5, 24. [Google Scholar] [CrossRef]
- Mijanur Rahman, M.; Gan, S.H.; Khalil, M.I. Neurological effects of honey: Current and future prospects. Evid. -Based Complementary Altern. Med. 2014, 2014, 958721. [Google Scholar] [CrossRef]
- Cui, Y.; Zhang, Y.; Zhao, X.; Shao, L.; Liu, G.; Sun, C.; Xu, R.; Zhang, Z. ACSL4 exacerbates ischemic stroke by promoting ferroptosis-induced brain injury and neuroinflammation. Brain Behav. Immun. 2021, 93, 312–321. [Google Scholar] [CrossRef]
- Nono Nankam, P.; Blüher, M. Retinol-binding protein 4 in obesity and metabolic dysfunctions. Mol. Cell. Endocrinol. 2021, 531, 111312. [Google Scholar] [CrossRef]
- Höpfinger, A.; Berghoff, M.; Karrasch, T.; Schmid, A.; Schäffler, A. Systematic Quantification of Neurotrophic Adipokines RBP4, PEDF, and Clusterin in Human Cerebrospinal Fluid and Serum. J. Clin. Endocrinol. Metab. 2021, 106, e2239–e2250. [Google Scholar] [CrossRef]
- Qin, H.; Yeh, W.I.; De Sarno, P.; Holdbrooks, A.T.; Liu, Y.; Muldowney, M.T. Signal transducer and activator of transcription-3/suppressor of cytokine signaling-3 (STAT3/SOCS3) axis in myeloid cells regulates neuroinflammation. Proc. Natl. Acad. Sci. USA 2012, 109, 5004–5009. [Google Scholar] [CrossRef] [Green Version]
- Oliva, A.A., Jr.; Kang, Y.; Sanchez-Molano, J.; Furones, C.; Atkins, C.M. STAT3 signaling after traumatic brain injury. J. Neurochem. 2012, 120, 710–720. [Google Scholar] [CrossRef]
- Zheng, Z.V.; Chen, J.; Lyu, H.; Lam, S.Y.E.; Lu, G.; Chan, W.Y.; Wong, G.K.C. Novel role of STAT3 in microglia-dependent neuroinflammation after experimental subarachnoid haemorrhage. Stroke Vasc. Neurol. 2021, svn-2021-001028. [Google Scholar] [CrossRef] [PubMed]
- Picone, P.; Di Carlo, M.; Nuzzo, D. Obesity and Alzheimer’s disease: Molecular bases. Eur. J. Neurosci. 2020, 52, 3944–3950. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zhao, F.; Shen, K.; Wang, W.; Siedlak, S.L.; Lee, H.-G.; Phelix, C.F.; Perry, G.; Shen, L.; Tang, B.; et al. The sterol regulatory element-binding protein 2 is dysregulated by tau alterations in Alzheimer disease. Brain Pathol. 2019, 29, 530–543. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; Cheng, K.K.; Hoo, R.L.; Siu, P.M.; Yau, S.Y. The Novel Perspectives of Adipokines on Brain Health. Int. J. Mol. Sci. 2019, 20, 5638. [Google Scholar] [CrossRef] [Green Version]
- Leuner, B.; Gould, E. Structural plasticity and hippocampal function. Annu. Rev. Psychol. 2010, 61, 111–140. [Google Scholar] [CrossRef] [Green Version]
- Lin, S.; Thomas, T.C.; Storlien, L.H.; Huang, X.F. Development of high fat diet-induced obesity and leptin resistance in C57Bl/6J mice. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 639–646. [Google Scholar] [CrossRef] [Green Version]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; McKee, L.J.; Bauer, T.L.; et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef]
- Forny-Germano, L.; De Felice, F.G.; Vieira, M.N.D.N. The Role of Leptin and Adiponectin in Obesity-Associated Cognitive Decline and Alzheimer’s Disease. Front. Neurosci. 2019, 12, 1027. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.-S.; Lee, W.-J.; Funahashi, T.; Tanaka, S.; Matsuzawa, Y.; Chao, C.-L.; Chen, C.-L.; Tai, T.-Y.; Chuang, L.-M. Weight reduction increases plasma levels of an adipose-derived anti-inflammatory protein, adiponectin. J. Clin. Endocrinol. Metab. 2001, 86, 3815–3819. [Google Scholar] [CrossRef]
- Qiu, G.; Wan, R.; Hu, J.; Mattson, M.P.; Spangler, E.; Liu, S.; Yau, S.-Y.; Lee, T.M.C.; Gleichmann, M.; Ingram, D.K.; et al. Adiponectin protects rat hippocampal neurons against excitotoxicity. Age 2011, 33, 155–165. [Google Scholar] [CrossRef]
- Guo, F.; Jiang, T.; Song, W.; Wei, H.; Wang, F.; Liu, L.; Ma, L.; Yin, H.; Wang, Q.; Xiong, L. Electroacupuncture attenuates cerebral ischemia-reperfusion injury in diabetic mice through adiponectin receptor 1-mediated phosphorylation of GSK-3β. Mol. Neurobiol. 2015, 51, 685–695. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. March 2016, 7, 27–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]




| Gene | Forward Primer | Reverse Primer | T° Annealing |
|---|---|---|---|
| Fas-L | 5′-CAAGTCCAACTCAAGGTCCATGCC-3′ | 5′-AGAGAGAGCTCAGATACGTTTGAC-3′ | 58 °C |
| Bim | 5′-AACCTTCTGATGTAAGTTCT-3′ | 5′-GTGATTGCCTTCAGGATTAC-3′ | 58 °C |
| p27 | 5′-TGCGAGTGTCTAACGGGAG-3′ | 5′-GTTTGACGTCTTCTGAGGCC-3′ | 59 °C |
| Bcl-2 | 5′-ATGTGTGTGGAGAGCGTCAA-3′ | 5′-AGAGACAGCCAGGAGAAATCA-3′ | 47 °C |
| BDNF | 5′-GGCTGACACTTTTGAGCACGTC-3′ | 5′-CTCCAAAGGCACTTGACTGCTG-3′ | 52 °C |
| IL-1β | 5′-TCATGGGATGATGATAACCTGCT-3′ | 5′-CCCATACTTTAGGAAGACACGATT-3′ | 50 °C |
| IL-6 | 5′-CTGGTGACAACCACGGCCTTCCCT-3′ | 5′-ATGCTTAGGCATAACGCACTAGGT-3′ | 54 °C |
| TNF-α | 5′-AGCCCACGTCGTAGCAAACCA-3′ | 5′-GCAGGGGCTCTTGACGGCAG-3′ | 53 °C |
| Β-actin | 5′-CGGGATCCCCGCCCTAGGCACCAGGGT-3′ | 5′-GGAATTCGGCTGGGGTGTTGAAGGTCTCAAA-3′ | 60 °C |
| Gene Name | Protein | HFD-H/HFD |
|---|---|---|
| Acsl4 | Acyl-CoA synthetase long-chain family member 4 | −2.30 |
| Adgre1 | Adhesion G Protein-Coupled Receptor E1 | −2.12 |
| Adipor1 | Adiponectin receptor 1 | 2.47 |
| Adipor2 | Adiponectin receptor 2 | 2.32 |
| Cd36 | CD36 antigen | −3.33 |
| Cebpa | CCAAT/enhancer binding protein (C/EBP), alpha | −2.83 |
| Insr | Insulin receptor | 3.77 |
| Irs1 | Insulin receptor substrate 1 | 4.17 |
| Lepr | Leptin receptor | 5.44 |
| Rbp4 | Retinol binding protein 4, plasma | −4.46 |
| Srebf2 | Sterol regulatory element binding factor 2 | −2.12 |
| Stat3 | Signal transducer and activator of transcription 3 | −2.39 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terzo, S.; Calvi, P.; Nuzzo, D.; Picone, P.; Galizzi, G.; Caruana, L.; Di Carlo, M.; Lentini, L.; Puleio, R.; Mulè, F.; et al. Preventive Impact of Long-Term Ingestion of Chestnut Honey on Glucose Disorders and Neurodegeneration in Obese Mice. Nutrients 2022, 14, 756. https://doi.org/10.3390/nu14040756
Terzo S, Calvi P, Nuzzo D, Picone P, Galizzi G, Caruana L, Di Carlo M, Lentini L, Puleio R, Mulè F, et al. Preventive Impact of Long-Term Ingestion of Chestnut Honey on Glucose Disorders and Neurodegeneration in Obese Mice. Nutrients. 2022; 14(4):756. https://doi.org/10.3390/nu14040756
Chicago/Turabian StyleTerzo, Simona, Pasquale Calvi, Domenico Nuzzo, Pasquale Picone, Giacoma Galizzi, Luca Caruana, Marta Di Carlo, Laura Lentini, Roberto Puleio, Flavia Mulè, and et al. 2022. "Preventive Impact of Long-Term Ingestion of Chestnut Honey on Glucose Disorders and Neurodegeneration in Obese Mice" Nutrients 14, no. 4: 756. https://doi.org/10.3390/nu14040756
APA StyleTerzo, S., Calvi, P., Nuzzo, D., Picone, P., Galizzi, G., Caruana, L., Di Carlo, M., Lentini, L., Puleio, R., Mulè, F., & Amato, A. (2022). Preventive Impact of Long-Term Ingestion of Chestnut Honey on Glucose Disorders and Neurodegeneration in Obese Mice. Nutrients, 14(4), 756. https://doi.org/10.3390/nu14040756









