Advances, Applications, and Comparison of Thermal (Pasteurization, Sterilization, and Aseptic Packaging) against Non-Thermal (Ultrasounds, UV Radiation, Ozonation, High Hydrostatic Pressure) Technologies in Food Processing
Abstract
:1. Introduction
2. Thermal Technologies in Food Processing
Pasteurization, Sterilization, and Aseptic Packaging
3. Non-Thermal Technologies in Food Processing
3.1. Ultrasounds
3.1.1. Application in Fruits and Vegetables
3.1.2. Application in Meat and Fish Products
3.1.3. Application in Cereal Product
3.1.4. Application in Dairy Products
3.1.5. Application in Emulsified Products
3.2. UV Radiation
3.2.1. Application in Fruits and Vegetables
3.2.2. Application in Meat and Fish Products
3.2.3. Application in Cereal Product
3.2.4. Application in Dairy Products
3.3. Ozonation
3.3.1. Application of Ozone in Fruits and Vegetables
3.3.2. Application of Ozone in Juices and Beverages
3.3.3. Application of Ozone in Meat Products
3.4. High-Pressure Processing
3.4.1. Application of HPP in Fruits and Vegetables
3.4.2. Application of HPP in Meat and Fish Products
3.4.3. Application of HPP in Juices and Beverages
3.4.4. Application of HPP in Dairy Products
3.4.5. Application of HPP in Emulsified Product
4. Microfluidization
5. Membrane Technology
6. Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Agriopoulou, S.; Stamatelopoulou, E.; Skiada, V.; Varzakas, T. Nanobiotechnology in food preservation and molecular perspective. In Book Nanotechnology-Enhanced Food Packaging, 1st ed.; Parameswaranpillai, J., Krishnankutty, R.E., Jayakumar, A., Rangappa, S.M., Siengchin, S., Eds.; Wiley-Vch GmbH: Weinheim, Germany, 2022; pp. 327–359. [Google Scholar]
- Gizaw, Z. Public health risks related to food safety issues in the food market: A systematic literature review. Environ. Health Prev. Med. 2019, 24, 68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roobab, U.; Aadil, R.M.; Madni, G.M.; Bekhit, A.E.D. The impact of nonthermal technologies on the microbiological quality of juices: A review. Compr. Rev. Food Sci. Food Saf. 2018, 17, 437–457. [Google Scholar] [CrossRef] [PubMed]
- Giner, M.J.; Hizarci, Õ.; Martí, N.; Saura, D.; Valero, M. Novel approaches to reduce brown pigment formation and color changes in thermal pasteurized tomato juice. Eur. Food Res. Technol. 2013, 236, 507–515. [Google Scholar] [CrossRef]
- Iqbal, A.; Murtaza, A.; Hu, W.; Ahmad, I.; Ahmed, A.; Xu, X. Activation and inactivation mechanisms of polyphenol oxidase during thermal and non-thermal methods of food processing. Food Bioprod. Process. 2019, 117, 170–182. [Google Scholar] [CrossRef]
- Wang, C.Y.; Huang, H.W.; Hsu, C.P.; Yang, B.B. Recent advances in food processing using high hydrostatic pressure technology. Crit. Rev. Food Sci. Nutr. 2016, 56, 527–540. [Google Scholar] [CrossRef] [PubMed]
- Adebo, O.A.; Molelekoa, T.; Makhuvele, R.; Adebiyi, J.A.; Oyedeji, A.B.; Gbashi, S.; Adefisoye, M.A.; Ogundele, O.M.; Njobeh, P.B. A review on novel non-thermal food processing techniques for mycotoxin reduction. Int. J. Food Sci. Technol. 2021, 56, 13–27. [Google Scholar] [CrossRef]
- Hernández-Hernández, H.M.; Moreno-Vilet, L.; Villanueva-Rodríguez, S.J. Current status of emerging food processing technologies in Latin America: Novel non-thermal processing. Innov. Food Sci. Emerg. Technol. 2019, 58, 102233. [Google Scholar] [CrossRef]
- López-Gámez, G.; Elez-Martínez, P.; Martín-Belloso, O.; Soliva-Fortuny, R. Recent Advances toward the application of non-thermal technologies in food processing: An insight on the bioaccessibility of health-related constituents in plant-based products. Foods 2021, 10, 1538. [Google Scholar] [CrossRef]
- Pereira, R.N.; Vicente, A.A. Environmental impact of novel thermal and non-thermal technologies in food processing. Food Res. Int. 2010, 43, 1936–1943. [Google Scholar] [CrossRef] [Green Version]
- Putnik, P.; Bursać Kovačević, D.; Herceg, K.; Pavkov, I.; Zorić, Z.; Levaj, B. Effects of modified atmosphere, anti-browning treatments and ultrasound on the polyphenolic stability, antioxidant capacity and microbial growth in fresh-cut apples. J. Food Process Eng. 2017, 40, e12539. [Google Scholar] [CrossRef]
- Huang, D.; Men, K.; Li, D.; Wen, T.; Gong, Z.; Sunden, B.; Wu, Z. Application of ultrasound technology in the drying of food products. Ultrason. Sonochem. 2020, 63, 104950. [Google Scholar] [CrossRef]
- Musielak, G.; Mierzwa, D.; Kroehnke, J. Food drying enhancement by ultrasound—A review. Trends Food Sci. Technol. 2016, 56, 126–141. [Google Scholar] [CrossRef]
- Lozowicka, B.; Jankowska, M.; Hrynko, I.; Kaczynski, P. Removal of 16 Pesticide Residues from strawberries by washing with tap and ozone water, ultrasonic cleaning and boiling. Environ. Monit. Assess. 2016, 188, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Ramaswamy, H.S.; Awuah, G.B.; Simpson, B.K. Heat transfer and lethality considerations in aseptic processing of liquid/particle mixtures: A review. Crit. Rev. Food Sci. Nutr. 1997, 37, 253–286. [Google Scholar] [CrossRef]
- Berk, Z. Chapter 3—Heat and mass transfer, basic principles. In Food Process Engineering and Technology, 3rd ed.; Berk, Z., Ed.; Elsevier: London, UK, 2018; pp. 79–126. [Google Scholar]
- Vidyarthi, S.K.; Mishra, D.K.; Dolan, K.D.; Muramatsu, Y. Inverse estimation of fluid-to-particle heat transfer coefficient in aseptic processing of particulate foods. Biosyst. Eng. 2020, 198, 210–222. [Google Scholar] [CrossRef]
- Betta, G.; Barbanti, D.; Massini, R. Food hygiene in aseptic processing and packaging system: A survey in the Italian food industry. Trends Food Sci. Technol. 2011, 22, 327e334. [Google Scholar] [CrossRef]
- Stoforos, N.G.; Sawada, H. Aseptic processing of liquid/particulate foods. In Heat Transfer in Food Processing, WIT Transactions on State of the Art in Science and Engineering, 1st ed.; Yanniotis, S., Sundén, B., Eds.; WIT Press: Billerica, MA, USA, 2007; pp. 187–208. [Google Scholar]
- FDA. Guide to Inspections of Aseptic Processing and Packaging for the Food Industry. 2014. Available online: https://www.fda.gov/inspections-compliance-enforcement-and-criminalinvestigations/inspection-guides/aseptic-processing-andpackaging-food-industry (accessed on 31 January 2022).
- Balasubramaniam, V.M. Liquid-to-Particle Convective Heat Transfer in Aseptic Processing Systems. Ph.D. Thesis, Department of Agriculture Engineering, The Ohio State University, Columbus, OH, USA, 1993. [Google Scholar]
- Toledo, R.T.; Singh, R.K.; Kong, F. Aseptic processing. In Fundamentals of Food Process Engineering, 4th ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 245–273. [Google Scholar]
- Ibrahim, M.T.; Briesen, H.; Först, P.; Zacharias, J. Lethality calculation of particulate liquid foods during aseptic processing. Processes 2019, 7, 587. [Google Scholar] [CrossRef] [Green Version]
- Diep, B.; Moulin, J.; Bastic-Schmid, V.; Putallaz, T.; Gimonet, J.; Valles, A.D.; Klijn, A. Validation protocol for commercial sterility testing methods. Food Control 2019, 103, 1–8. [Google Scholar] [CrossRef]
- Lalpuria, M.; Anantheswaran, R.; Floros, J. Packaging technologies and their role in food safety. In Microbial Decontamination in the Food Industry; Woodhead Publishing: Sawston, UK, 2012; pp. 701–745. [Google Scholar]
- Anderson, N.M. Recent advances in low moisture food pasteurization. Curr. Opin. Food Sci. 2019, 29, 109–115. [Google Scholar] [CrossRef]
- Hosseinzadeh Samani, B.; Khoshtaghaza, M.H.; Minaei, S.; Zareifourosh, H.; Eshtiaghi, M.N.; Rostami, S. Design, development and evaluation of an automatic fruit-juice pasteurization system using microwave—Ultrasonic waves. J. Food Sci. Technol. 2016, 53, 88–103. [Google Scholar] [CrossRef] [Green Version]
- Sanchez, B.A.O.; Celestino, S.M.C.; de Abreu Gloria, M.B.; Celestino, I.C.; Lozada, M.I.O.; Júnior, S.D.A.; de Alencar, E.R.; de Oliveira, L.D.L. Pasteurization of passion fruit Passiflora setacea pulp to optimize bioactive compounds retention. Food Chem. 2020, 6, 100084. [Google Scholar] [CrossRef]
- Zhu, X.; Guo, W.; Wu, X. Frequency-and temperature-dependent dielectric properties of fruit juices associated with pasteurization by dielectric heating. J. Food Eng. 2012, 109, 258–266. [Google Scholar] [CrossRef]
- Lopez, A.A. Complete Course in Canning and Related Processes: Volume 3 Processing Procedures for Canned Food Products; The Canning Trade Inc.: Baltimore, MD, USA, 1987; p. 516. [Google Scholar]
- Potter, N.N.; Hotchkiss, J.H. Food Science, 5th ed.; Springer Science+Business Media, Inc.: Berlin/Heidelberg, Germany, 1995; p. 608. [Google Scholar]
- Tucker, G.; Featherstone, S. Essentials of Thermal Processing; John Wiley & Sons Ltd.: Chichester, UK, 2011; p. 264. [Google Scholar]
- Stoforos, N.G. Thermal processing. In Handbook of Food Processing: Food Preservation, 1st ed.; Varzakas, T., Tzia, C., Eds.; CRC Press, Taylor and Francis Group: Boca Raton, FL, USA, 2016; pp. 27–56. [Google Scholar]
- Stumbo, C.R. Thermobacteriology in Food Processing, 2nd ed.; Academic Press, Inc.: Cambridge, MA, USA, 1973; p. 329. [Google Scholar]
- Vajdi, M.; Pereira, R.R. Comparative effects of ethylene oxide, gamma irradiation and microwave treatments on selected spices. J. Food Sci. 1973, 38, 893–895. [Google Scholar] [CrossRef]
- EPA: Ethylene Oxide. Agency USEP. 2017. Available online: https://www.epa.gov/stationary-sources-air-pollution/ethylene-oxide-emissions-standards-sterilization-facilities (accessed on 31 January 2022).
- Duncan, S.E.; Moberg, K.; Amin, K.N.; Wright, M.; Newkirk, J.J.; Ponder, M.A.; Acuff, G.R.; Dickson, J.S. Processes to preserve spice and herb quality and sensory integrity during pathogen inactivation. J. Food Sci. 2017, 82, 1208–1215. [Google Scholar] [CrossRef]
- Balchem: Propylene Oxide. 2018. Available online: https://balchem.com/performance-gases/products/propylene-oxide/ (accessed on 31 January 2022).
- EPA: Propylene Oxide. Agency USEP. 2000. Available online: https://www.epa.gov/sites/default/files/2014-09/documents/propylene_oxide_final_volume9_2010.pdf (accessed on 31 January 2022).
- Almela, L.; Nieto-Sandoval, J.M.; Fernández López, J.A. Microbial inactivation of paprika by a high-temperature short-X time treatment. Influence on color properties. J. Agric. Food Chem. 2002, 50, 1435–1440. [Google Scholar] [CrossRef]
- Ivarsson, C. Nut pasteurization: Minimising impact on appearance, colour and flavor. Agro Food Ind. Hi-Tech 2011, 22, 22–24. [Google Scholar]
- Shah, M.K.; Asa, G.; Sherwood, J.; Graber, K.; Bergholz, T.M. Efficacy of vacuum steam pasteurization for inactivation of Salmonella PT 30, Escherichia coli O157:H7 and Enterococcus faecium on low moisture foods. Int. J. Food Microbiol. 2017, 244, 111–118. [Google Scholar] [CrossRef]
- Ban, G.H.; Kang, D.H. Effectiveness of superheated steam for inactivation of Escherichia coli O157:H7, Salmonella Typhimurium, Salmonella Enteritidis phage type 30, and Listeria monocytogenes on almonds and pistachios. Int. J. Food Microbiol. 2016, 220, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.S.; Han, A.R.; Reyes-De-Corcuera, J.I.; Powers, J.R.; Kang, D.H. Evaluation of steam pasteurization in controlling salmonella serotype enteritidis on raw almond surfaces. Lett. Appl. Microbiol. 2010, 50, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Johnson, J.A.; Wang, S. Radio frequency heating for postharvest control of pests in agricultural products: A review. Postharvest Biol. Technol. 2016, 113, 106–118. [Google Scholar] [CrossRef]
- Marra, F.; Zhang, L.; Lyng, J.G. Radio frequency treatment of foods: Review of recent advances. J. Food Eng. 2009, 91, 497–508. [Google Scholar] [CrossRef]
- Jeong, S.G.; Kang, D.H. Influence of moisture content on inactivation of Escherichia coli O157:H7 and Salmonella enterica serovar Typhimurium in powdered red and black pepper spices by radio-frequency heating. Int. J. Food Microbiol. 2014, 176, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Sagong, H.G.; Choi, S.H.; Ryu, S.; Kang, D.H. Radio-frequency heating to inactivate Salmonella Typhimurium and Escherichia coli O157:H7 on black and red pepper spice. Int. J. Food Microbiol. 2012, 153, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Schlisselberg, D.B.; Kler, E.; Kalily, E.; Kisluk, G.; Karniel, O.; Yaron, S. Inactivation of food borne pathogens in ground beef by cooking with highly controlled radio frequency energy. Int. J. Food Microbiol. 2013, 160, 219–226. [Google Scholar] [CrossRef]
- Li, R.; Kou, X.; Cheng, T.; Zheng, A.; Wang, S. Verification of radio frequency pasteurization process for in-shell almonds. J. Food Eng. 2017, 192, 103–110. [Google Scholar] [CrossRef]
- Awad, T.S.; Moharram, H.A.; Shaltout, O.E.; Asker, D.; Youssef, M.M. Applications of ultrasound in analysis, processing and quality control of food: A review. Food Res. Int. 2012, 48, 410–427. [Google Scholar] [CrossRef]
- Bhargava, N.; Mor, R.S.; Kumar, K.; Sharanagat, V.S. Advances in application of ultrasound in food processing: A review. Ultrason. Sonochem. 2021, 70, 105293. [Google Scholar] [CrossRef] [PubMed]
- Anaya-Esparza, L.M.; Velázquez-Estrada, R.M.; Roig, A.X.; García-Galindo, H.S.; Sayago-Ayerdi, S.G.; Montalvo-González, E. Thermosonication: An Alternative Processing for Fruit and Vegetable Juices. Trends Food Sci. Technol. 2017, 61, 26–37. [Google Scholar] [CrossRef]
- Sánchez-Rubio, M.; Taboada-Rodríguez, A.; Cava-Roda, R.; López-Gómez, A.; Marín-Iniesta, F. Combined use of thermo-ultrasound and cinnamon leaf essential oil to inactivate Saccharomyces Cerevisiae in natural orange and pomegranate juices. LWT Food Sci. Technol. 2016, 73, 140–146. [Google Scholar] [CrossRef]
- Ortega-Rivas, E. Ultrasound in food preservation. Food Eng. Ser. 2012, 5, 251–262. [Google Scholar] [CrossRef]
- Singla, M.; Sit, N. Application of ultrasound in combination with other technologies in food processing: A review. Ultrason. Sonochem. 2021, 73, 105506. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Sun, D.W. Enhancement of food processes by ultrasound: A Review. Crit. Rev. Food Sci. Nutr. 2015, 55, 570–594. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, N.K. Opportunities and challenges in application of ultrasound in food processing. Crit. Rev. Food Sci. Nutr. 2011, 51, 705–722. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Belwal, T.; Cravotto, G.; Luo, Z. Sono-physical and sono-chemical effects of ultrasound: Primary applications in extraction and freezing operations and influence on food components. Ultrason. Sonochem. 2020, 60, 104726. [Google Scholar] [CrossRef]
- Ojha, K.S.; Aznar, R.; O’Donnell, C.; Tiwari, B.K. Ultrasound technology for the extraction of biologically active molecules from plant, animal and marine sources. TrAC Trends Anal. Chem. 2020, 122, 115663. [Google Scholar] [CrossRef]
- Varzakas, T.; Tzia, C. Ultrasonic and UV disinfection of food. In Handbook of Food Processing; Contemporary Food Engineering; CRC: Boca Raton, FL, USA, 2015; ISBN 13: 978-1-4987-2176-9. [Google Scholar]
- Astráin-Redín, L.; Alejandre, M.; Raso, J.; Cebrián, G.; Álvarez, I. Direct contact ultrasound in food processing: Impact on food quality. Front. Nutr. 2021, 8, 1–11. [Google Scholar] [CrossRef]
- Cheng, X.; Zhang, M.; Xu, B.; Adhikari, B.; Sun, J. The principles of ultrasound and its application in freezing related processes of food materials: A review. Ultrason. Sonochem. 2015, 27, 576–585. [Google Scholar] [CrossRef]
- Deora, N.S.; Misra, N.N.; Deswal, A.; Mishra, H.N.; Cullen, P.J.; Tiwari, B.K. Ultrasound for improved crystallisation in food processing. Food Eng. Rev. 2013, 5, 36–44. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Ma, F.; Li, R.; Ren, G.; Yan, D.; Zhang, H.; Zhu, X.; Wu, R.; Wu, J. Degradation of tremella fuciformis polysaccharide by a combined ultrasound and hydrogen peroxide treatment: Process parameters, structural characteristics, and antioxidant activities. Int. J. Biol. Macromol. 2020, 160, 979–990. [Google Scholar] [CrossRef]
- Flórez-Fernández, N.; Domínguez, H.; Torres, M.D. A green approach for alginate extraction from sargassum muticum brown seaweed using ultrasound-assisted technique. Int. J. Biol. Macromol. 2019, 124, 451–459. [Google Scholar] [CrossRef]
- Zhang, X.; Ma, X.T.; Xu, Y.; Jiang, G.C.; Zhang, J.L.; Liu, X.J.; Yan, X.H. Extraction, purification, characterization, anticoagulant activity, and anticoagulant mechanism of polysaccharides from the heads of hypomesus olidus. Food Sci. Nutr. 2020, 8, 849–861. [Google Scholar] [CrossRef] [PubMed]
- Nuerxiati, R.; Abuduwaili, A.; Mutailifu, P.; Wubulikasimu, A.; Rustamova, N.; Jingxue, C.; Aisa, H.A.; Yili, A. Optimization of ultrasonic-assisted extraction, characterization and biological activities of polysaccharides from orchis chusua, D. Don (Salep). Int. J. Biol. Macromol. 2019, 141, 431–443. [Google Scholar] [CrossRef] [PubMed]
- Cui, R.; Zhu, F. Ultrasound modified polysaccharides: A review of structure, physicochemical properties, biological activities and food applications. Trends Food Sci. Technol. 2021, 107, 491–508. [Google Scholar] [CrossRef]
- Ni, Y.; Li, J.; Fan, L. Effects of ultrasonic conditions on the interfacial property and emulsifying property of cellulose nanoparticles from ginkgo seed shells. Ultrason. Sonochem. 2021, 70, 105335. [Google Scholar] [CrossRef] [PubMed]
- Bermúdez-Aguirre, D.; Barbosa-Cánovas, G.V. Inactivation of Saccharomyces Cerevisiae in pineapple, grape and cranberry juices under pulsed and continuous thermo-sonication treatments. J. Food Eng. 2012, 108, 383–392. [Google Scholar] [CrossRef]
- Eh, A.L.S.; Teoh, S.G. Novel modified ultrasonication technique for the extraction of lycopene from tomatoes. Ultrason. Sonochem. 2012, 19, 151–159. [Google Scholar] [CrossRef]
- Pingret, D.; Fabiano-Tixier, A.S.; Petitcolas, E.; Canselier, J.P.; Chemat, F. First investigation on ultrasound-assisted preparation of food products: Sensory and physicochemical characteristics. J. Food Sci. 2011, 76, 287–292. [Google Scholar] [CrossRef]
- Pingret, D.; Durand, G.; Fabiano-Tixier, A.S.; Rockenbauer, A.; Ginies, C.; Chemat, F. Degradation of edible oil during food processing by ultrasound: Electron paramagnetic resonance, physicochemical, and sensory appreciation. J. Agric. Food Chem. 2012, 60, 7761–7768. [Google Scholar] [CrossRef]
- Pingret, D.; Fabiano-Tixier, A.S.; Chemat, F. Degradation during application of ultrasound in food processing: A review. Food Control 2013, 31, 593–606. [Google Scholar] [CrossRef]
- Azam, S.M.R.; Ma, H.; Xu, B.; Devi, S.; Stanley, S.L.; Siddique, M.A.B.; Mujumdar, A.S.; Zhu, J. Multi-frequency multi-mode ultrasound treatment for removing pesticides from lettuce (Lactuca Sativa L.) and effects on product quality. LWT 2021, 143, 111147. [Google Scholar] [CrossRef]
- Chen, F.; Zhang, M.; Yang, C.H. Application of ultrasound technology in processing of ready-to-eat fresh food: A review. Ultrason. Sonochem. 2020, 63, 104953. [Google Scholar] [CrossRef] [PubMed]
- Soltani Firouz, M.; Farahmandi, A.; Hosseinpour, S. Recent advances in ultrasound application as a novel technique in analysis, processing and quality control of fruits, juices and dairy products industries: A review. Ultrason. Sonochem. 2019, 57, 73–88. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Liu, Y.; Huang, X.; Yang, W.; Hu, W.; Pan, S. Effect of ultrasonic processing on the changes in activity, aggregation and the secondary and tertiary structure of polyphenol oxidase in oriental sweet melon (Cucumis Melo Var. Makuwa Makino). J. Sci. Food Agric. 2017, 97, 1326–1334. [Google Scholar] [CrossRef]
- Huang, N.; Cheng, X.; Hu, W.; Pan, S. Inactivation, aggregation, secondary and tertiary structural changes of germin-like protein in satsuma mandarine with high polyphenol oxidase activity induced by ultrasonic processing. Biophys. Chem. 2015, 197, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Yeoh, W.K.; Ali, A. Ultrasound treatment on phenolic metabolism and antioxidant capacity of fresh-cut pineapple during cold storage. Food Chem. 2017, 216, 247–253. [Google Scholar] [CrossRef]
- Costa, M.G.M.; Fonteles, T.V.; de Jesus, A.L.T.; Almeida, F.D.L.; de Miranda, M.R.A.; Fernandes, F.A.N.; Rodrigues, S. High-intensity ultrasound processing of pineapple juice. Food Bioprocess Technol. 2013, 6, 997–1006. [Google Scholar] [CrossRef]
- Amaral, R.D.A.; Benedetti, B.C.; Pujola, M.; Achaerandio, I.; Bachelli, M.L.B. Effect of ultrasound on quality of fresh-cut potatoes during refrigerated storage. Food Eng. Rev. 2015, 7, 176–184. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound Assisted Extraction of Food and Natural Products. Mechanisms, Techniques, Combinations, Protocols and Applications. A Review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef]
- Paniwnyk, L. Applications of ultrasound in processing of liquid foods: A review. Ultrason. Sonochem. 2017, 38, 794–806. [Google Scholar] [CrossRef]
- Khandpur, P.; Gogate, P.R. Effect of novel ultrasound based processing on the nutrition quality of different fruit and vegetable juices. Ultrason. Sonochem. 2015, 27, 125–136. [Google Scholar] [CrossRef]
- Ordóñez-Santos, L.E.; Martínez-Girón, J.; Arias-Jaramillo, M.E. Effect of ultrasound treatment on visual color, vitamin C, total phenols, and carotenoids content in cape gooseberry juice. Food Chem. 2017, 233, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Oladejo, A.O.; Ma, H.; Qu, W.; Zhou, C.; Wu, B.; Yang, X.; Onwude, D.I. Effects of ultrasound pretreatments on the kinetics of moisture loss and oil uptake during deep fat frying of sweet potato (Ipomea Batatas). Innov. Food Sci. Emerg. Technol. 2017, 43, 7–17. [Google Scholar] [CrossRef]
- Dehghannya, J.; Naghavi, E.A.; Ghanbarzadeh, B. Frying of potato strips pretreated by ultrasound-assisted air-drying. J. Food Process. Preserv. 2016, 40, 583–592. [Google Scholar] [CrossRef]
- Minakawa, A.F.K.; Faria-Tischer, P.C.S.; Mali, S. Simple ultrasound method to obtain starch micro- and nanoparticles from cassava, corn and yam Starches. Food Chem. 2019, 283, 11–18. [Google Scholar] [CrossRef]
- Caraveo, O.; Alarcon-Rojo, A.D.; Renteria, A.; Santellano, E.; Paniwnyk, L. Physicochemical and microbiological characteristics of beef treated with high-intensity ultrasound and stored at 4 °C. J. Sci. Food Agric. 2015, 95, 2487–2493. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Kang, D.; Zhang, W.; Zhang, C.; Zou, Y.; Zhou, G. Changes in calpain activity, protein degradation and microstructure of beef M. Semitendinosus by the application of ultrasound. Food Chem. 2018, 245, 724–730. [Google Scholar] [CrossRef] [PubMed]
- Boateng, E.F.; Nasiru, M.M. Applications of ultrasound in meat processing technology: A review. Food Sci. Technol. USA. 2019, 7, 11–15. [Google Scholar] [CrossRef] [Green Version]
- Alarcon-Rojo, A.D.; Janacua, H.; Rodriguez, J.C.; Paniwnyk, L.; Mason, T.J. Power ultrasound in meat processing. Meat Sci. 2015, 107, 86–93. [Google Scholar] [CrossRef] [Green Version]
- Kang, D.C.; Wang, A.R.; Zhou, G.H.; Zhang, W.G.; Xu, S.M.; Guo, G.P. Power ultrasonic on mass transport of beef: Effects of ultrasound intensity and NaCl concentration. Innov. Food Sci. Emerg. Technol. 2016, 35, 36–44. [Google Scholar] [CrossRef]
- Al-Hilphy, A.R.; Al-Temimi, A.B.; Al Rubaiy, H.H.M.; Anand, U.; Delgado-Pando, G.; Lakhssassi, N. Ultrasound applications in poultry meat processing: A systematic review. J. Food Sci. 2020, 85, 1386–1396. [Google Scholar] [CrossRef] [Green Version]
- Kang, D.-C.; Gao, X.-Q.; Ge, Q.-F.; Zhou, G.-H.; Zhang, W.-G. Effects of ultrasound on the beef structure and water distribution during curing through protein degradation and modification. Ultrason. Sonochem. 2017, 38, 317–325. [Google Scholar] [CrossRef]
- Peña-Gonzalez, E.; Alarcon-Rojo, A.D.; Garcia-Galicia, I.; Carrillo-Lopez, L.; Huerta-Jimenez, M. Ultrasound as a potential process to tenderize beef: Sensory and technological parameters. Ultrason. Sonochem. 2019, 53, 134–141. [Google Scholar] [CrossRef]
- Chang, H.J.; Xu, X.L.; Zhou, G.H.; Li, C.B.; Huang, M. Effects of characteristics changes of collagen on meat physicochemical properties of beef semitendinosus muscle during ultrasonic processing. Food Bioprocess Technol. 2012, 5, 285–297. [Google Scholar] [CrossRef]
- Hu, Y.; Yu, H.; Dong, K.; Yang, S.; Ye, X.; Chen, S. Analysis of the tenderisation of jumbo squid (Dosidicus Gigas) meat by ultrasonic treatment using response surface methodology. Food Chem. 2014, 160, 219–225. [Google Scholar] [CrossRef]
- Zhu, F.; Li, H. Modification of quinoa flour functionality using ultrasound. Ultrason. Sonochem. 2019, 52, 305–310. [Google Scholar] [CrossRef]
- Fox, P.; Smith, P.P.; Sahi, S. Ultrasound measurements to monitor the specific gravity of food batters. J. Food Eng. 2004, 65, 317–324. [Google Scholar] [CrossRef]
- Ding, J.; Hou, G.G.; Dong, M.; Xiong, S.; Zhao, S.; Feng, H. Physicochemical properties of germinated dehulled rice flour and energy requirement in germination as affected by ultrasound treatment. Ultrason. Sonochem. 2018, 41, 484–491. [Google Scholar] [CrossRef]
- Harasym, J.; Satta, E.; Kaim, U. Ultrasound treatment of buckwheat grains impacts important functional properties of resulting flour. Molecules 2020, 25, 3012. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, G.; Liu, M.; Mei, X.; Yu, O.; Kan, J. Effects of multi-frequency ultrasound on physicochemical properties, structural characteristics of gluten protein and the quality of noodle. Ultrason. Sonochem. 2020, 67, 105–135. [Google Scholar] [CrossRef]
- Guimarães, J.T.; Balthazar, C.F.; Scudino, H.; Pimentel, T.C.; Esmerino, E.A.; Ashokkumar, M.; Freitas, M.Q.; Cruz, A.G. High-intensity ultrasound: A novel technology for the development of probiotic and prebiotic dairy products. Ultrason. Sonochem. 2019, 57, 12–21. [Google Scholar] [CrossRef]
- Huang, G.; Chen, S.; Tang, Y.; Dai, C.; Sun, L.; Ma, H.; He, R. Stimulation of low intensity ultrasound on fermentation of skim milk medium for yield of yoghurt peptides by Lactobacillus paracasei. Ultrason. Sonochem. 2019, 51, 315–324. [Google Scholar] [CrossRef]
- Umego, E.C.; He, R.; Huang, G.; Dai, C.; Ma, H. Ultrasound-assisted fermentation: Mechanisms, technologies, and challenges. J. Food Process. Preserv. 2021, 45, e15559. [Google Scholar] [CrossRef]
- Shanmugam, A.; Chandrapala, J.; Ashokkumar, M. The effect of ultrasound on the physical and functional properties of skim milk. Innov. Food Sci. Emerg. Technol. 2012, 16, 251–258. [Google Scholar] [CrossRef]
- Akdeniz, V.; Akalın, A.S. New approach for yoghurt and ice cream production: High-intensity ultrasound. Trends Food Sci. Technol. 2019, 86, 392–398. [Google Scholar] [CrossRef]
- Carpenter, J.; Saharan, V.K. Ultrasonic assisted formation and stability of mustard oil in water nanoemulsion: Effect of process parameters and their optimization. Ultrason. Sonochem. 2017, 35, 422–430. [Google Scholar] [CrossRef]
- Albano, K.M.; Nicoletti, V.R. Ultrasound impact on whey protein concentrate-pectin complexes and in the O/W emulsions with low oil soybean content stabilization. Ultrason. Sonochem. 2018, 41, 562–571. [Google Scholar] [CrossRef] [Green Version]
- Gayán, E.; Condón, S.; Álvarez, I. Biological aspects in food preservation by ultraviolet light: A review. Food Bioprocess Technol. 2014, 7, 1–20. [Google Scholar] [CrossRef]
- Koutchma, T.N.; Forney, L.J.; Moraru, C.I. Ultraviolet Light in Food Technology Principles and Applications, 2009th ed.; Sun, D.-W., Ed.; CRC: Boca Raton, FL, USA, 2009; ISBN 13: 978-1-4200-5950-2. [Google Scholar]
- Ramesh, T.; Nayak, B.; Amirbahman, A.; Tripp, C.P.; Mukhopadhyay, S. Application of ultraviolet light assisted titanium dioxide photocatalysis for food safety: A review. Innov. Food Sci. Emerg. Technol. 2016, 38, 105–115. [Google Scholar] [CrossRef] [Green Version]
- Yoo, S.; Ghafoor, K.; Kim, J.U.; Kim, S.; Jung, B.; Lee, D.U.; Park, J. Inactivation of Escherichia coli O157:H7 on orange fruit surfaces and in juice using photocatalysis and high hydrostatic pressure. J. Food Prot. 2015, 78, 1098–1105. [Google Scholar] [CrossRef] [Green Version]
- Shahbaz, H.M.; Yoo, S.; Seo, B.; Ghafoor, K.; Kim, J.U.; Lee, D.U.; Park, J. Combination of TiO2-UV photocatalysis and high hydrostatic pressure to inactivate bacterial pathogens and yeast in commercial apple juice. Food Bioprocess Technol. 2016, 9, 182–190. [Google Scholar] [CrossRef]
- Corrêa, T.Q.; Blanco, K.C.; Garcia, É.B.; Perez, S.M.L.; Chianfrone, D.J.; Morais, V.S.; Bagnato, V.S. Effects of ultraviolet light and curcumin-mediated photodynamic inactivation on microbiological food safety: A study in meat and fruit. Photodiagnosis Photodyn. Ther. 2020, 30, 101678. [Google Scholar] [CrossRef]
- Orlowska, M.; Koutchma, T.; Grapperhaus, M.; Gallagher, J.; Schaefer, R.; Defelice, C. Continuous and pulsed ultraviolet light for nonthermal treatment of liquid foods. Part 1: Effects on quality of fructose solution, apple juice, and milk. Food Bioprocess Technol. 2013, 6, 1580–1592. [Google Scholar] [CrossRef]
- Gayán, E.; Serrano, M.J.; Monfort, S.; Álvarez, I.; Condón, S. Combining ultraviolet light and mild temperatures for the inactivation of Escherichia coli in orange juice. J. Food Eng. 2012, 113, 598–605. [Google Scholar] [CrossRef]
- Ochoa-Velasco, C.E.; Cruz-González, M.; Guerrero-Beltrán, J.Á. Ultraviolet-C light inactivation of Escherichia coli and Salmonella typhimurium in coconut (Cocos nucifera L.) milk. Innov. Food Sci. Emerg. Technol. 2014, 26, 199–204. [Google Scholar] [CrossRef]
- Fredericks, I.N.; du Toit, M.; Krügel, M. Efficacy of ultraviolet radiation as an alternative technology to inactivate microorganisms in grape juices and wines. Food Microbiol. 2011, 28, 510–517. [Google Scholar] [CrossRef]
- Hinds, L.M.; Guclu, G.; Kelebek, H.; Selli, S.; O’Donnell, C.P.; Tiwari, B.K. Effect of ultraviolet light emitting diode treatments on microbial load, phenolic and volatile profile of black peppercorns. LWT 2021, 152, 112133. [Google Scholar] [CrossRef]
- Nyhan, L.; Przyjalgowski, M.; Lewis, L.; Begley, M.; Callanan, M. Investigating the use of ultraviolet light emitting diodes (UV-Leds) for the inactivation of bacteria in powdered food ingredients. Foods 2021, 10, 797. [Google Scholar] [CrossRef]
- Baenas, N.; Iniesta, C.; González-Barrio, R.; Nuñez-Gómez, V.; Periago, M.J.; García-Alonso, F.J. Post-harvest use of ultraviolet light (UV) and light emitting diode (LED) to enhance bioactive compounds in refrigerated tomatoes. Molecules 2021, 26, 1847. [Google Scholar] [CrossRef]
- Lázaro, C.A.; Conte-Júnior, C.A.; Monteiro, M.L.G.; Canto, A.C.V.S.; Costa-Lima, B.R.C.; Mano, S.B.; Franco, R.M. Effects of ultraviolet light on biogenic amines and other quality indicators of chicken meat during refrigerated storage. Poult. Sci. 2014, 93, 2304–2313. [Google Scholar] [CrossRef]
- Colejo, S.; Alvarez-Ordóñez, A.; Prieto, M.; González-Raurich, M.; López, M. Evaluation of ultraviolet light (UV), non-thermal atmospheric plasma (NTAP) and their combination for the control of foodborne pathogens in smoked salmon and their effect on quality attributes. Innov. Food Sci. Emerg. Technol. 2018, 50, 84–93. [Google Scholar] [CrossRef]
- Konak, Ü.I.; Certel, M.; Karakaş, B. Effects of ultraviolet irradiation of batter and different baking methods on cake quality. J. Microbiol. Biotechnol. Food Sci. 2017, 7, 113–117. [Google Scholar] [CrossRef] [Green Version]
- Kumar, A.; Rani, P.; Purohit, S.R.; Rao, P.S. Effect of ultraviolet irradiation on wheat (Triticum aestivum) flour: Study on protein modification and changes in quality attributes. J. Cereal Sci. 2020, 96, 103094. [Google Scholar] [CrossRef]
- Guneser, O.; Karagul Yuceer, Y. Effect of ultraviolet light on water- and fat-soluble vitamins in cow and goat milk. J. Dairy Sci. 2012, 95, 6230–6241. [Google Scholar] [CrossRef] [Green Version]
- Koca, N.; Öztürk, M.U. Application of batch system ultraviolet light on the surface of kashar cheese, a kind of pasta-filata cheese: Effects on mould inactivation, lipid oxidation, colour, hardness and sensory properties. J. Dairy Res. 2020, 87, 493–497. [Google Scholar] [CrossRef]
- Ricciardi, E.F.; Pedros-Garrido, S.; Papoutsis, K.; Lyng, J.G.; Conte, A.; Del Nobile, M.A. Novel technologies for preserving ricotta cheese: Effects of ultraviolet and near-ultraviolet–visible light. Foods 2020, 9, 580. [Google Scholar] [CrossRef]
- Ha, J.W.; Back, K.H.; Kim, Y.H.; Kang, D.H. Efficacy of UV-C Irradiation for inactivation of food-borne pathogens on sliced cheese packaged with different types and thicknesses of plastic films. Food Microbiol. 2016, 57, 172–177. [Google Scholar] [CrossRef]
- Casas, D.E.; Vargas, D.A.; Randazzo, E.; Lynn, D.; Echeverry, A.; Brashears, M.M.; Sanchez-Plata, M.X.; Miller, M.F. In-plant validation of novel on-site ozone generation technology (bio-safe) compared to lactic acid beef carcasses and trim using natural microbiota and Salmonella and E. coli O157:H7 surrogate enumeration. Foods 2021, 10, 1002. [Google Scholar] [CrossRef]
- Liao, C.; Yu, Y. Effect of vacuum cooling followed by ozone repressurization on Clostridium perfringens germination and outgrowth in cooked pork meat under temperature-sbuse conditions. Innov. Food Sci. Emerg. Technol. 2021, 68, 102599. [Google Scholar] [CrossRef]
- Megahed, A.; Aldridge, B.; Lowe, J. Antimicrobial efficacy of aqueous ozone and ozone–lactic acid blend on Salmonella-Contaminated chicken drumsticks using multiple sequential soaking and spraying approaches. Front. Microbiol. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Li, Z.; Sun, Y.; Jin, H.; Wang, Q.; Jin, Y.; Huang, X.; Sheng, L. Improvement and mechanism of emulsifying properties of liquid egg yolk by ozonation technology. LWT 2022, 156, 113038. [Google Scholar] [CrossRef]
- Bono, G.; Okpala, C.O.R.; Vitale, S.; Ferrantelli, V.; Noto, A.D.; Costa, A.; Di Bella, C.; Monaco, D.L. Effects of different ozonized slurry-ice treatments and superchilling storage (−1 °C) on microbial spoilage of two important pelagic fish species. Food Sci. Nutr. 2017, 5, 1049–1056. [Google Scholar] [CrossRef] [Green Version]
- Traore, M.B.; Sun, A.; Gan, Z.; Senou, H.; Togo, J.; Fofana, K.H. Antimicrobial capacity of ultrasound and ozone for enhancing bacterial safety on inoculated shredded green cabbage (Brassica oleracea Var. capitata). Can. J. Microbiol. 2020, 66, 125–137. [Google Scholar] [CrossRef]
- Taiye Mustapha, A.; Zhou, C.; Wahia, H.; Amanor-Atiemoh, R.; Otu, P.; Qudus, A.; Abiola Fakayode, O.; Ma, H. Sonozonation: Enhancing the antimicrobial efficiency of aqueous ozone washing techniques on cherry tomato. Ultrason. Sonochem. 2020, 64, 105059. [Google Scholar] [CrossRef]
- De Souza, L.P.; Faroni, L.R.D.A.; Heleno, F.F.; Cecon, P.R.; Gonçalves, T.D.C.; da Silva, G.J.; Prates, L.H.F. Effects of ozone treatment on postharvest carrot quality. LWT Food Sci. Technol. 2018, 90, 53–60. [Google Scholar] [CrossRef]
- Kusvuran, E.; Yildirim, D.; Mavruk, F.; Ceyhan, M. Removal of chloropyrifos ethyl, tetradifon and chlorothalonil pesticide residues from citrus by using ozone. J. Hazard. Mater. 2012, 241–242, 287–300. [Google Scholar] [CrossRef]
- García-Martínez, M.M.; Campayo, A.; Carot, J.M.; de la Hoz, K.S.; Salinas, M.R.; Alonso, G.L. Oenological characteristics of Vitis Vinifera L. cabernet sauvignon grapes from vineyards treated with ozonated water. Aust. J. Grape Wine Res. 2020, 26, 388–398. [Google Scholar] [CrossRef]
- Flores, P.; Hernández, V.; Fenoll, J.; Hellín, P. Pre-harvest application of ozonated water on broccoli crops: Effect on head quality. J. Food Compos. Anal. 2019, 83, 103260. [Google Scholar] [CrossRef]
- Aslam, R.; Alam, M.S.; Pandiselvam, R. Aqueous ozone sanitization system for fresh produce: Design, development, and optimization of process parameters for minimally processed onion. Ozone Sci. Eng. 2022, 44, 3–16. [Google Scholar] [CrossRef]
- Panigrahi, C.; Mishra, H.N.; De, S. Ozone treatment of ultrafiltered sugarcane juice: Process optimization using multi-objective genetic algorithm and correlation analysis by multivariate technique. LWT Food Sci. Technol. 2022, 154, 112861. [Google Scholar] [CrossRef]
- Ouf, S.A.; Ali, E.M. Does the treatment of dried herbs with ozone as a fungal decontaminating agent affect the active constituents? Environ. Pollut. 2021, 277, 116715. [Google Scholar] [CrossRef]
- Matłok, N.; Piechowiak, T.; Zardzewiały, M.; Gorzelany, J.; Balawejder, M. Effects of ozone treatment on microbial status and the contents of selected bioactive compounds in Origanum majorana L. plants. Plants 2020, 9, 1637. [Google Scholar] [CrossRef]
- Alexopoulos, A.; Plessas, S.; Kourkoutas, Y.; Stefanis, C.; Vavias, S.; Voidarou, C.; Mantzourani, I.; Bezirtzoglou, E. Experimental effect of ozone upon the microbial flora of commercially produced dairy fermented products. Int. J. Food Microbiol. 2017, 246, 5–11. [Google Scholar] [CrossRef]
- Sachadyn-Król, M.; Agriopoulou, S. Ozonation as a method of abiotic elicitation improving the health—Promoting properties of plant products—A review. Molecules 2020, 25, 2416. [Google Scholar] [CrossRef]
- Pandiselvam, R.; Manikantan, M.R.; Divya, V.; Ashokkumar, C.; Kaavya, R.; Kothakota, A.; Ramesh, S.V. Ozone: An advanced oxidation technology for starch modification. Ozone Sci. Eng. 2019, 41, 491–507. [Google Scholar] [CrossRef]
- García-Mateos, M.R.; Quiroz-González, B.; Corrales-García, J.; Ybarra-Moncada, C.; Leyva-Ruelas, G. Ozone-high hydrostatic pressure synergy for the stabilization of refrigerated pitaya (Stenocereus pruinosus) juice. Innov. Food Sci. Emerg. Technol. 2019, 56, 102187. [Google Scholar] [CrossRef]
- Marino, M.; Maifreni, M.; Baggio, A.; Innocente, N. Inactivation of foodborne bacteria biofilms by aqueous and gaseous ozone. Front. Microbiol. 2018, 9, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Sivaranjani, S.; Prasath, V.A.; Pandiselvam, R.; Kothakota, A.; Mousavi Khaneghah, A. Recent advances in applications of ozone in the cereal industry. LWT Food Sci. Technol. 2021, 146, 111412. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in occurrence, importance, and mycotoxin control strategies: Prevention and detoxification in foods. Foods 2020, 9, 137. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Koliadima, A.; Karaiskakis, G.; Kapolos, J. Kinetic study of aflatoxins’ degradation in the presence of ozone. Food Control 2016, 61, 221–226. [Google Scholar] [CrossRef]
- Sujayasree, O.J.; Chaitanya, A.K.; Bhoite, R.; Pandiselvam, R.; Kothakota, A.; Gavahian, M.; Mousavi Khaneghah, A. Ozone: An advanced oxidation technology to enhance sustainable food consumption through mycotoxin degradation. Ozone Sci. Eng. 2022, 44, 17–37. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in analysis and detection of major mycotoxins in foods. Foods 2020, 9, 518. [Google Scholar] [CrossRef] [Green Version]
- Bermúdez-Aguirre, D.; Barbosa-Cánovas, G.V. Ozone applications in food processing. In Handbook of Food Processing: Food Preservation, 1st ed.; Varzakas, T., Tzia, C., Eds.; CRC Press, Taylor and Francis Group: Boca Raton, FL, USA, 2016; pp. 691–704. [Google Scholar]
- Kaavya, R.; Pandiselvam, R.; Abdullah, S.; Sruthi, N.U.; Jayanath, Y.; Ashokkumar, C.; Chandra Khanashyam, A.; Kothakota, A.; Ramesh, S.V. Emerging non-thermal technologies for decontamination of salmonella in food. Trends Food Sci. Technol. 2021, 112, 400–418. [Google Scholar] [CrossRef]
- Pandiselvam, R.; Subhashini, S.; Banuu Priya, E.P.; Kothakota, A.; Ramesh, S.V.; Shahir, S. Ozone based food preservation: A promising green technology for enhanced food safety. Ozone Sci. Eng. 2019, 41, 17–34. [Google Scholar] [CrossRef]
- Niveditha, A.; Pandiselvam, R.; Prasath, V.A.; Singh, S.K.; Gul, K.; Kothakota, A. Application of cold plasma and ozone technology for decontamination of Escherichia coli in foods—A review. Food Control 2021, 130, 108338. [Google Scholar] [CrossRef]
- Premjit, Y.; Sruthi, N.U.; Pandiselvam, R.; Kothakota, A. Aqueous ozone: Chemistry, physicochemical properties, microbial inactivation, factors influencing antimicrobial effectiveness, and application in food. Compr. Rev. Food Sci. Food Saf. 2022, 20, 1–32. [Google Scholar] [CrossRef]
- Pandiselvam, R.; Sunoj, S.; Manikantan, M.R.; Kothakota, A.; Hebbar, K.B. Application and kinetics of ozone in food preservation. Ozone Sci. Eng. 2017, 39, 115–126. [Google Scholar] [CrossRef]
- Yucel Sengun, I.; Kendirci, P. Potential of ozonated water at different temperatures to improve safety and shelf-life of fresh cut lettuce. Ozone Sci. Eng. 2018, 40, 216–227. [Google Scholar] [CrossRef]
- Wani, S.; Maker, J.; Thompson, J.; Barnes, J.; Singleton, I. Effect of ozone treatment on inactivation of Escherichia coli and Listeria Sp. on spinach. Agriculture 2015, 5, 155–169. [Google Scholar] [CrossRef] [Green Version]
- Siddique, Z.; Malik, A.U.; Asi, M.R.; Anwar, R.; Inam Ur Raheem, M. Sonolytic-ozonation technology for sSanitizing microbial contaminants and pesticide residues from spinach (Spinacia Oleracea L.) leaves, at household level. Environ. Sci. Pollut. Res. 2021, 28, 52913–52924. [Google Scholar] [CrossRef]
- Ummat, V.; Singh, A.K.; Sidhu, G.K. Effect of aqueous ozone on quality and shelf life of shredded green bell pepper (Capsicum annuum). J. Food Process. Preserv. 2018, 42, 1–14. [Google Scholar] [CrossRef]
- Nayak, S.L.; Sethi, S.; Sharma, R.R.; Sharma, R.M.; Singh, S.; Singh, D. Aqueous ozone controls decay and maintains quality attributes of strawberry (Fragaria × Ananassa Duch.). J. Food Sci. Technol. 2020, 57, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Contigiani, E.V.; Kronberg, M.F.; Jaramillo Sánchez, G.; Gómez, P.L.; García-Loredo, A.B.; Munarriz, E.; Alzamora, S.M. Ozone washing decreases strawberry susceptibility to Botrytis cinerea while maintaining antioxidant, optical and sensory quality. Heliyon 2020, 6, e05416. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Chen, C.; Jiang, A.; Zhang, Y.; Zhao, Q.; Hu, W. Effects of aqueous ozone treatment on microbial growth, quality, and pesticide residue of fresh-cut cabbage. Food Sci. Nutr. 2021, 9, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Liu, C.; Jiang, A.; Zhao, Q.; Liu, S.; Hu, W. Effects of ozonated water on microbial growth, quality retention and pesticide residue removal of fresh-cut onions. Ozone Sci. Eng. 2020, 42, 399–407. [Google Scholar] [CrossRef]
- Gonçalves, A.A.; Lira Santos, T.C. Improving quality and shelf-life of whole chilled pacific white shrimp (Litopenaeus vannamei) by ozone technology combined with modified atmosphere packaging. LWT 2019, 99, 568–575. [Google Scholar] [CrossRef]
- Giannoglou, M.; Dimitrakellis, P.; Efthimiadou, A.; Gogolides, Ε.; Katsaros, G. Comparative study on the effect of cold atmospheric plasma, ozonation, pulsed electromagnetic fields and high-pressure technologies on sea bream fillet quality indices and shelf life. Food Eng. Rev. 2021, 13, 175–184. [Google Scholar] [CrossRef]
- López Hernández, K.; Pardío Sedas, V.; Rodríguez Dehaibes, S.; Suárez Valencia, V.; Rivas Mozo, I.; Martínez Herrera, D.; Flores Primo, A.; Uscanga Serrano, R. Improved microbial safety of direct ozone-depurated shellstock Eastern oysters (Crassostrea virginica) by superchilled storage. Front Microbiol. 2018, 9, 2802. [Google Scholar] [CrossRef]
- Zalewska, M.; Górska-Horczyczak, E.; Marcinkowska-Lesiak, M. Effect of Applied Ozone Dose, Time of Ozonization and Storage Time on Selected Physicochemical Characteristics of Mushrooms (Agaricus bisporus). Agriculture 2021, 11, 748. [Google Scholar] [CrossRef]
- Ayranci, U.G.; Ozunlu, O.; Ergezer, H.; Karaca, H. Effects of ozone treatment on microbiological quality and physicochemical properties of turkey breast meat. Ozone Sci. Eng. 2020, 42, 95–103, 541. [Google Scholar] [CrossRef]
- Giménez, B.; Graiver, N.; Giannuzzi, L.; Zaritzky, N. Treatment of beef with gaseous ozone: Physicochemical aspects and antimicrobial effects on heterotrophic microflora and Listeria monocytogenes. Food Control 2021, 121, 1–9. [Google Scholar] [CrossRef]
- Degala, H.L.; Scott, J.R.; Rico Espinoza, F.I.; Mahapatra, A.K.; Kannan, G. Synergistic effect of ozonated and electrolyzed water on the inactivation kinetics of Escherichia coli on goat meat. J. Food Saf. 2020, 40, e12740. [Google Scholar] [CrossRef]
- Jaramillo Sánchez, G.M.; Garcia Loredo, A.B.; Contigiani, E.V.; Gómez, P.L.; Alzamora, S.M. Inactivation kinetics of peroxidase and polyphenol oxidase in peach juice treated with gaseous ozone. Int. J. Food Sci. Technol. 2018, 53, 347–355. [Google Scholar] [CrossRef]
- Patil, S.; Valdramidis, V.P.; Tiwari, B.K.; Cullen, P.J.; Bourke, P. Quantitative assessment of the shelf life of ozonated apple juice. Eur. Food Res. Technol. 2011, 232, 469–477. [Google Scholar] [CrossRef] [Green Version]
- Timpanaro, N.; Strano, M.C.; Allegra, M.; Foti, P.; Granuzzo, G.; Carboni, C.; Romeo, F.V. Assessing the effect of ozonated water on microbial load and quality of Nocellara Etnea table olives. Ozone Sci. Eng. 2021, 43, 1–8. [Google Scholar] [CrossRef]
- Werlang, G.O.; Kich, J.D.; Lopes, G.V.; Coldebella, A.; Feddern, V.; Cardoso, M. Effect of gaseous ozone application during chilling on microbial and quality attributes of pig carcasses. Food Sci. Technol. Int. 2021, 27, 10820132211014985. [Google Scholar] [CrossRef] [PubMed]
- Khanashyam, A.C.; Shanker, M.A.; Kothakota, A.; Mahanti, N.K.; Pandiselvam, R. Ozone applications in milk and meat industry. Ozone Sci. Eng. 2021, 44, 1–16. [Google Scholar] [CrossRef]
- Muhlisin, M.; Utama, D.T.; Lee, J.H.; Choi, J.H.; Lee, S.K. Effects of gaseous ozone exposure on bacterial counts and oxidative properties in chicken and duck breast meat. Korean J. Food Sci. Anim. Resour. 2016, 36, 405–411. [Google Scholar] [CrossRef] [Green Version]
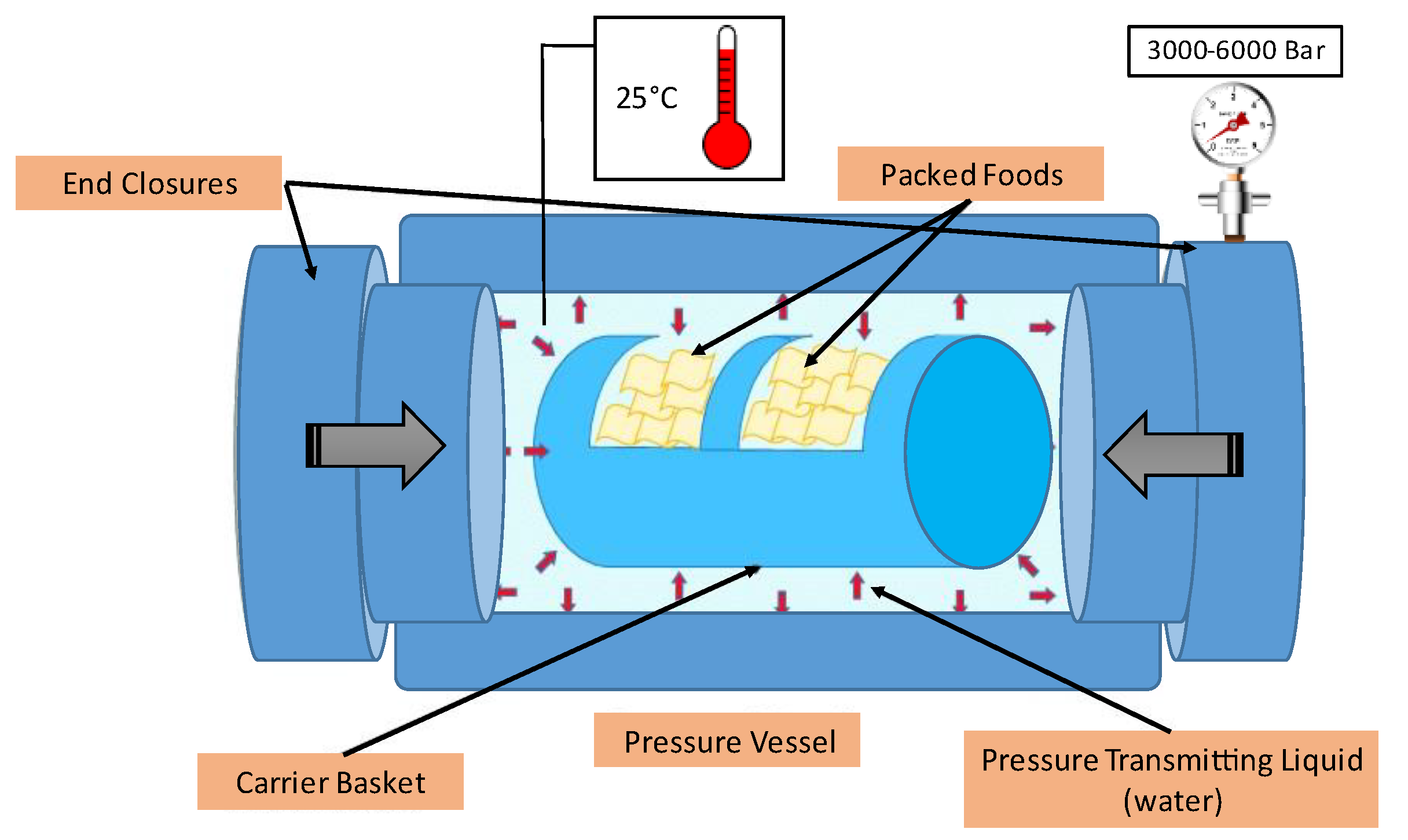
- Aganovic, K.; Hertel, C.; Vogel, R.F.; Johne, R.; Schlüter, O.; Schwarzenbolz, U.; Jäger, H.; Holzhauser, T.; Bergmair, J.; Roth, A.; et al. Aspects of high hydrostatic pressure food processing: Perspectives on technology and food safety. Compr. Rev. Food Sci. Food Saf. 2021, 20, 3225–3266. [Google Scholar] [CrossRef]
- Tsikrika, K.; Walsh, D.; Joseph, A.; Burgess, C.M.; Rai, D.K. High-pressure processing and ultrasonication of minimally processed potatoes: Effect on the colour, microbial counts, and bioactive compounds. Molecules 2021, 26, 2614. [Google Scholar] [CrossRef]
- Cano-Lamadrid, M.; Artés-Hernández, F. By-products revalorization with non-thermal treatments to enhance phytochemical compounds of fruit and vegetables derived products: A review. Foods 2022, 11, 59. [Google Scholar] [CrossRef]
- Morata, A.; Escott, C.; Loira, I.; López, C.; Palomero, F.; González, C. Emerging non-thermal technologies for the extraction of grape anthocyanins. Antioxidants 2021, 10, 1863. [Google Scholar] [CrossRef]
- Ozkan, G.; Guldiken, B.; Capanoglu, E. Effect of novel food processing technologies on beverage antioxidants. In Processing and Sustainability of Beverages; Grumezescu, A.M., Holban, A.M., Eds.; Woodhead Publishing: Cambridge, UK, 2019; pp. 413–449. [Google Scholar]
- Roobab, U.; Shabbir, M.A.; Khan, A.W.; Arshad, R.N.; Bekhit, A.E.; Zeng, X.A.; Inam-Ur-Raheem, M.; Aadil, R.M. High-pressure treatments for better quality clean-label juices and beverages: Overview and advances. LWT Food Sci. Technol. 2021, 149, 111828. [Google Scholar] [CrossRef]
- Balakrishna, A.K.; Wazed, M.A.; Farid, M. A Review on the effect of high pressure processing (HPP) on gelatinization and infusion of nutrients. Molecules 2020, 25, 2369. [Google Scholar] [CrossRef] [PubMed]
- Katsaros, G.; Alexandrakis, Z.; Taoukis, P. High-pressure processing of foods: Technology and applications. In Handbook of Food Processing: Food Preservation, 1st ed.; Varzakas, T., Tzia, C., Eds.; CRC Press, Taylor and Francis Group: Boca Raton, FL, USA, 2016; pp. 443–468. [Google Scholar]
- Barba, F.J.; Terefe, N.S.; Buckow, R.; Knorr, D.; Orlien, V. New opportunities and perspectives of high pressure treatment to improve health and safety attributes of foods. A review. Food Res. Int. 2015, 77, 725–742. [Google Scholar] [CrossRef]
- Woldemariam, H.W.; Emire, S.A. High pressure processing of foods for microbial and mycotoxins control: Current trends and future prospects. Cogent Food Agric. 2019, 5, 1622184. [Google Scholar] [CrossRef]
- Balasubramaniam, V.M.; Martinez-Monteagudo, S.I.; Gupta, R. Principles and application of high pressure–based technologies in the food industry. Ann. Rev. Food Sci. Technol. 2015, 6, 435–462. [Google Scholar] [CrossRef]
- Huang, H.W.; Hsu, C.P.; Wang, C.Y. Healthy expectations of high hydrostatic pressure treatment in food processing industry. J. Food Drug Anal. 2020, 28, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Škegro, M.; Putnik, P.; Bursać Kovačević, D.; Kovač, A.P.; Salkić, L.; Čanak, I.; Frece, J.; Zavadlav, S.; Ježek, D. Chemometric comparison of high-pressure processing and thermal pasteurization: The nutritive, sensory, and microbial quality of smoothies. Foods 2021, 10, 1167. [Google Scholar] [CrossRef] [PubMed]
- Gavahian, M.; Mathad, G.N.; Pandiselvam, R.; Lin, J.; Sun, D.-W. Emerging technologies to obtain pectin from food processing by-products: A strategy for enhancing resource efficiency. Trends Food Sci. Technol. 2021, 115, 42–54. [Google Scholar] [CrossRef]
- Huang, H.W.; Wu, S.J.; Lu, J.K.; Shyu, Y.T.; Wang, C.Y. Current status and future trends of high-pressure processing in food industry. Food Control 2017, 72, 1–8. [Google Scholar] [CrossRef]
- Amsasekar, A.; Mor, R.S.; Kishore, A.; Singh, A.; Sid, S. Impact of high pressure processing on microbiological, nutritional and sensory properties of food: A review. Nutr. Food Sci. 2022, 52, 1–22. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E.; Sachadyn-Król, M.; Varzakas, T. Lactic acid bacteria as antibacterial agents to extend the shelf life of fresh and minimally processed fruits and vegetables: Quality and safety aspects. Microorganisms 2020, 8, 952. [Google Scholar] [CrossRef] [PubMed]
- Kyriacou, M.C.; Rouphael, Y. Towards a new definition of quality for fresh fruits and vegetables. Sci. Hortic. 2018, 234, 463–469. [Google Scholar] [CrossRef]
- Corato, U.D. Improving the shelf- life and quality of fresh and minimally- processed fruits and vegetables for a modern food industry: A comprehensive critical review from the traditional technologies into the most promising advancements. Crit. Rev. Food Sci. Nutr. 2020, 60, 940–975. [Google Scholar] [CrossRef]
- Baranda, A.B.; Montes, P. HPP for improving preservation of vitamin and antioxidant contents in vegetable matrices. In Present and Future of High Pressure Processing: A Tool for Developing Innovative, Sustainable, Safe and Healthy Foods; Barba, F.J., Tonello-Samson, C., Puértolas, E., Lavilla, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 15–70. [Google Scholar]
- Viacava, F.; Ortega, E.; Welti-Chanes, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. Using high hydrostatic pressure processing come-up time as an innovative tool to induce the biosynthesis of free and bound phenolics in whole carrots. Food Bioprocess Technol. 2020, 13, 1717–1727. [Google Scholar] [CrossRef]
- Jacobo-Velázquez, D.A.; Benavides, J. Non-thermal technologies as tools to increase the content of health-promoting compounds in whole fruits and vegetables while retaining quality attributes. Foods 2021, 10, 2904. [Google Scholar] [CrossRef]
- Nabi, B.G.; Mukhtar, K.; Arshad, R.N.; Radicetti, E.; Tedeschi, P.; Shahbaz, M.U.; Walayat, N.; Nawaz, A.; Inam-Ur-Raheem, M.; Aadil, R.M. High-pressure processing for sustainable food supply. Sustainability 2021, 13, 13908. [Google Scholar] [CrossRef]
- Hu, K.; Peng, D.; Wang, L.; Liu, H.; Xie, B.; Sun, Z. Effect of mild high hydrostatic pressure treatments on physiological and physicochemical characteristics and carotenoid biosynthesis in postharvest mango. Postharvest Biol. Technol. 2021, 172, 111381. [Google Scholar] [CrossRef]
- Kim, T.; Gil, B.; Kim, C.; Cho, Y. Enrichment of phenolics in harvested strawberries by high-pressure treatment. Food Bioprocess Technol. 2017, 10, 222–227. [Google Scholar] [CrossRef]
- Morata, A.; Loira, I.; Vejarano, R.; Bañuelos, M.A.; Sanz, P.D.; Otero, L.; Suárez-Lepe, J.A. Grape processing by high hydrostaticPressure: Effect on microbial populations, phenol extraction and wine quality. Food Bioprocess Technol. 2014, 8, 277–286. [Google Scholar] [CrossRef] [Green Version]
- Ninčević Grassino, A.; Ostojić, J.; Miletić, V.; Djaković, S.; Bosiljkov, T.; Zorić, Z.; Ježek, D.; Rimac Brnčić, S.; Brnčić, M. Application of high hydrostatic pressure and ultrasound-assisted extractions as a novel approach for pectin and polyphenols recovery from tomato peel waste. Innov. Food Sci. Emerg. Technol. 2020, 64, 102424. [Google Scholar] [CrossRef]
- Strati, I.F.; Gogou, E.; Oreopoulou, V. Enzyme and high pressure assisted extraction of carotenoids from tomato waste. Food Bioprod. Process. 2015, 94, 668–674. [Google Scholar] [CrossRef]
- Xie, F.; Zhang, W.; Lan, X.; Gong, S.; Wu, J.; Wang, Z. Effects of high hydrostatic pressure and high pressure homogenization processing on characteristics of potato peel waste pectin. Carbohydr. Polym. 2018, 196, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Boziaris, I.S.; Parlapani, F.F.; DeWitt, C.A.M. High pressure processing at ultra-low temperatures: Inactivation of foodborne bacterial pathogens and quality changes in frozen fish fillets. Innov. Food Sci. Emerg. Technol. 2021, 74, 102811. [Google Scholar] [CrossRef]
- Bechstein, D.V.; Popp, J.; Sudhaus-Joern, N.; Krischek, C. Effect of ethyl-lauroyl-arginate hypochloride in combination with high hydrostatic pressure processing on the microbial load and physico-chemical characteristics of minced and portioned chicken breast meat. Poult Sci. 2019, 98, 966–976. [Google Scholar] [CrossRef]
- Sheen, S.; Huang, C.-Y.; Ramos, R.; Chien, S.-Y.; Scullen, O.J.; Sommers, C. Lethality prediction for Escherichia coli O157:H7 and uropathogenic E. coli in ground chicken treated with high pressure processing and trans-cinnamaldehyde. J. Food Sci. 2018, 83, 740–749. [Google Scholar] [CrossRef]
- Possas, A.; Pérez-Rodríguez, F.; Valero, A.; García-Gimeno, R.M. Modelling the inactivation of Listeria monocytogenes by high hydrostatic pressure processing in foods: A review. Trends Food Sci. Technol. 2017, 70, 45–55. [Google Scholar] [CrossRef]
- Cava, R.; Higuero, N.; Ladero, L. High-pressure processing and storage temperature on Listeria monocytogenes, microbial counts and oxidative changes of two traditional dry-cured meat products. Meat Sci. 2021, 171, 108273. [Google Scholar] [CrossRef] [PubMed]
- Argyri, A.A.; Papadopoulou, O.S.; Nisiotou, A.; Tassou, C.C.; Chorianopoulos, N. Effect of high pressure processing on the survival of Salmonella Enteritidis and shelf-life of chicken fillets. Food Microbiol. 2018, 70, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.; Sheen, S.; Sommers, C.H.; Zhou, S.; Sheen, L.Y. Survival evaluation of Salmonella and Listeria monocytogenes on selective and nonselective media in ground chicken meat subjected to high hydrostatic pressure and carvacrol. J. Food Prot. 2020, 83, 37–44. [Google Scholar] [CrossRef]
- Chuang, S.; Sheen, S. High pressure processing of raw meat with essential oils-microbial survival, meat quality, and models: A review. Food Control 2022, 132, 108529. [Google Scholar] [CrossRef]
- Yu, H.H.; Chin, Y.-W.; Paik, H.-D. Application of natural preservatives for meat and meat products against food-borne pathogens and spoilage bacteria: A review. Foods 2021, 10, 2418. [Google Scholar] [CrossRef] [PubMed]
- Koutchma, T.; Popović, V.; Ros-Polski, V.; Popielarz, A. Effects of ultraviolet light and high-pressure processing on quality and health-related constituents of fresh juice products. Compr. Rev. Food Sci. Food Saf. 2016, 15, 844–867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pérez-Lamela, C.; Franco, I.; Falqué, E. Impact of high-pressure processing on antioxidant activity during storage of fruits and fruit products: A review. Molecules 2021, 26, 5265. [Google Scholar] [CrossRef]
- Panigrahi, C.; Vishwakarma, S.; Mishra, H.N.; De, S. Kinetic modeling for inactivation of polyphenoloxidase and peroxidase enzymes during ozonation of sugarcane juice. J. Food Process. Preserv. 2021, 45, 1–13. [Google Scholar] [CrossRef]
- Zhang, L.; Dai, S.; Brannan, R.G. Effect of high pressure processing, browning treatments, and refrigerated storage on sensory analysis, color, and polyphenol oxidase activity in pawpaw (Asimina triloba L.) pulp. LWT-Food Sci. Technol. 2017, 86, 49–54. [Google Scholar] [CrossRef]
- Zhao, G.; Zhang, R.; Zhang, M. Effects of high hydrostatic pressure processing and subsequent storage on phenolic contents and antioxidant activity in fruit and vegetable products. Int. J. Food Sci. Technol. 2017, 52, 3–12. [Google Scholar] [CrossRef]
- Sousa, S.G.; Delgadillo, I.; Saraiva, J.A. Effect of thermal pasteurisation and high-pressure processing on immunoglobulin content and lysozyme and lactoperoxidase activity in human colostrum. Food Chem. 2014, 151, 79–85. [Google Scholar] [CrossRef]
- Pallarés, N.; Sebastià, A.; Martínez-Lucas, V.; González-Angulo, M.; Barba, F.J.; Berrada, H.; Ferrer, E. High pressure processing impact on alternariol and aflatoxins of grape juice and fruit juice-milk based beverages. Molecules 2021, 26, 3769. [Google Scholar] [CrossRef]
- Sethi, S.; Chauhan, O.P.; Anurag, R.K. Effect of high-pressure processing on quality and stability of green mango blended mayonnaise. J. Food Sci. Technol. 2017, 54, 2341–2350. [Google Scholar] [CrossRef]
- Nuñez-Mancilla, Y.; Pérez-Won, M.; Uribe, E.; Vega-Gálvez, A.; Di Scala, K. Osmotic dehydration under high hydrostatic pressure: Effects on antioxidant activity, total phenolics compounds, vitamin C and colour of strawberry (Fragaria vesca). LWT Food Sci. Technol. 2013, 52, 151–156. [Google Scholar] [CrossRef]
- Cap, M.; Paredes, P.F.; Fernández, D.; Mozgovoj, M.; Vaudagna, S.R.; Rodriguez, A. Effect of high hydrostatic pressure on Salmonella spp inactivation and meat- quality of frozen chicken breast. LWT Food Sci. Technol. 2020, 118, 108873. [Google Scholar] [CrossRef]
- Borges, A.F.; Cózar, A.; Patarata, L.; Gama, L.T.; Alfaia, C.M.; Fernandes, M.J.; Fernandes, M.H.; Pérez, H.V.; Fraqueza, M.J. Effect of high hydrostatic pressure challenge on biogenic amines, microbiota, and sensory profile in traditional poultry- and pork-based semidried fermented sausage. J. Food Sci. 2020, 85, 1256–1264. [Google Scholar] [CrossRef] [PubMed]
- Suprabha Raj, A.; Chakraborty, S.; Rao, P.S. Optimizing the thermal assisted high-pressure process parameters for a sugarcane based mixed beverage using response surface methodology. J. Food Process Eng. 2020, 43, e13274. [Google Scholar] [CrossRef]
- Stinco, C.M.; Szczepańska, J.; Marszałek, K.; Pinto, C.A.; Inácio, R.S.; Mapelli-Brahm, P.; Barba, F.J.; Lorenzo, J.M.; Saraiva, J.A.; Meléndez-Martínez, A.J. Effect of high-pressure processing on carotenoids profile, colour, microbial and enzymatic stability of cloudy carrot juice. Food Chem. 2019, 299, 125112. [Google Scholar] [CrossRef]
- Stratakos, A.C.; Inguglia, E.S.; Linton, M.; Tollerton, J.; Murphy, L.; Corcionivoschi, N.; Koidis, A.; Tiwari, B.K. Effect of high pressure processing on the safety, shelf life and quality of raw milk. Innov. Food Sci. Emerg. Technol. 2019, 52, 325–333. [Google Scholar] [CrossRef] [Green Version]
- Tan, S.F.; Chin, N.L.; Tee, T.P.; Chooi, S.K. Physico-chemical changes, microbiological properties, and storage shelf life of cow and goat milk from industrial high-pressure processing. Processes 2020, 8, 697. [Google Scholar] [CrossRef]
- Guo, X.; Chen, M.; Li, Y.; Dai, T.; Shuai, X.; Chen, J.; Liu, C. Modification of food macromolecules using dynamic high pressure microfluidization: A review. Trends Food Sci. Technol. 2020, 100, 223–234. [Google Scholar] [CrossRef]
- Ozturk, O.K.; Turasan, H. Latest developments in the applications of microfluidization to modify the structure of macromolecules leading to improved physicochemical and functional properties. Crit. Rev. Food Sci. Nutr. 2021, 1–23. [Google Scholar] [CrossRef]
- Kumar, A.; Dhiman, A.; Suhag, R.; Sehrawat, R.; Upadhyay, A.; McClements, D.J. Comprehensive review on potential applications of microfluidization in food processing. Food Sci. Biotechnol. 2022, 31, 17–36. [Google Scholar] [CrossRef]
- Mert, I.D. The applications of microfluidization in cereals and cereal-based products: An overview. Crit. Rev. Food Sci. Nutr. 2018, 60, 1007–1024. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Deng, L.; Dai, T.; Li, Y.; Chen, J.; Liu, W.; Liu, W. Microfluidization: A promising food processing technology and its challenges in industrial application. Food Control 2021, 108794. [Google Scholar] [CrossRef]
- Charcosset, C. Classical and recent applications of membrane processes in the food industry. Food Eng Rev. 2021, 13, 322–343. [Google Scholar] [CrossRef]
- Cassano, A.; Drioli, E. Integrated Membrane Operations: In the Food Production; De Gruyter: Berlin, Germany, 2013. [Google Scholar] [CrossRef]
- Guzel-Seydim, Z.B.; Greene, A.K.; Seydim, A.C. Use of ozone in the food industry. LWT Food Sci. Technol. 2004, 37, 453–460. [Google Scholar] [CrossRef]
- Huang, H.W.; Lung, H.M.; Yang, B.B.; Wang, C.Y. Responses of microorganisms to high hydrostatic pressure processing. Food Control 2014, 40, 250–259. [Google Scholar] [CrossRef]
- HPP: Equipment, Trends and the Science Behind the Technology. Available online: https://www.foodengineeringmag.com/articles/98497-hpp-equipment-trends-and-the-science-behind-the-technology (accessed on 14 February 2022).
- Koutchma, T. Advances in ultraviolet light technology for non-thermal processing of liquid foods. Food Bioprocess Technol. 2009, 2, 138–155. [Google Scholar] [CrossRef]




| Process | Food | Experimental Parameters | Target Organism | Microbial Inactivation or Reduction | Reference |
|---|---|---|---|---|---|
| Steam | Paprika | 130–170 °C, 4–6 s | Indicators (e.g., Enterobacteriaceae, coliforms, yeast, mold) | 3–4 log reduction | [40] |
| Steam | Pistachios | 88 °C, 4 min | Enterococcus faecium | 4 log reduction | [41] |
| Steam | Seeds, black peppercorns | 85 °C, 1 min | Salmonella enterica, Escherichia coli O157:H7 | >5 log reduction | [42] |
| Steam | Almonds, pistachios | 200 °C, 15–30 s | S. enterica, E. coli O157:H7, Listeria monocytogenes | >5 log reduction in 15 s (almonds) and 30 s (pistachios) | [43] |
| Steam | Black peppercorn, cumin seeds | 85 °C, 1–2 min | S. enterica | 5 log reduction | [37] |
| Steam | Almonds | 95 °C, 25 s | Salmonella Enteritidis PT 30 | 5 log reduction | [44] |
| Category | Type | Process Parameters (Type/Power/Frequency/Intensity/Time) | Use | Effects | Reference |
|---|---|---|---|---|---|
| Fruits and Vegetables | Melon | Water bath/25 kHz/4870 W/m2/10, 20, and 30 min | Pre-drying treatment | Samples immersed in distilled water present negative water loss values, in sucrose solution, values are positive and are higher when the sonication time increased. | [4] |
| Papaya | Water bath/25 kHz/4870 W/m2/10, 20, and 30 min | Pre-drying treatment | Water loss increased with sonication time. | [4] | |
| Pineapple | Water bath/25 kHz/55.5 W/L/20 and 40 min | Pre-drying treatment | The moisture content increased with increasing sonication time. | [4] | |
| Mushrooms | Water bath/25 kHz/154 W/20 and 25 min | Pre-drying treatment | Water gain increased with increasing sonication time. | [4] | |
| Strawberry | Water bath/480 W/ 40 kHz/5 min | Pesticides removal | Reduction of 91.2% of 16 pesticide residues. | [6] | |
| Lettuce | Water bath/300 W/20, 40, 60 kHz/8 min | Pesticides removal | Removal of 92.31% of abamectin b1, 89.36% of alphamethrin and 95.25% of emamectin benzoate. No changes in nutritional properties. | [76] | |
| Oriental sweet melon juice | Ice-water bath/100–500 W/20 kHz/20 min | Inhibitory effect on enzymes | 65% of inactivation of PPO. | [79] | |
| Satsuma mandarin | Ice-water bath/400 W/20 kHz/30 min | Inhibitory effect on enzymes | 63,7% of inactivation of PPO. | [80] | |
| Pineapple | Water bath/25–29 W/20, 40, 37 kHz/10–15 min | Inhibitory effect on enzymes | Decrease in PPO and POD activity. Enhancement of phenylalanine ammonia lyase activity. | [81] | |
| Pineapple juice | Titanium probe/500 W/19 kHz/376 W/cm2/10 min | Inhibitory effect on enzymes | PPO activity reduced; viscosity drop of 75%. | [82] | |
| Fresh cut potatoes | Water bath/200 W/40 kHz/5 min | Inhibitory effect on enzymes | PPO activity reduced. pH goes down with longer treatments. No change in color. After 10 min, the potatoes’ cells are damaged. | [83] | |
| Garlic cloves | Solvent bath/35 kHz/30 min | Aroma extraction | - | [84] | |
| Grape seeds | 33–67% ethanol–water bath/250 W/40 kHz/16–34 min | Phenol, antioxidants, anthocyanins extraction | - | [84] | |
| Grapefruit juice | Probe/28 kHz/30, 60 and 90 min | Microorganism reduction | Enhancement of total antioxidant capacity, ascorbic acid, flavonoids, flavonols, and total phenolics. | [85] | |
| Apple juice | Probe/25 kHz/2 Wcm^−2 /30 and 60 min | Microorganism reduction | Rise of total carotenoids, viscosity, minerals such as Na, K, and Ca, and of the concentration of sugar and polyphenolic compounds. | [85] | |
| Orange, sweet lime, carrot, and spinach juices | Probe/100 W/20 kHz/15 min | Microorganism reduction | Sterilization without loss of nutrients. | [86] | |
| Cape gooseberry juice | Water bath/240 W/42 kHz/10, 20, 40 min | Post-US process modifications | Decrease in chromaticity, juice yellowing, increased total phenols, RAE value, and carotenoids. | [87] | |
| Sweet potatoes | Probe/300 W/28 kHz/30 min | Prior to frying treatment | Reduction of 71.47% the oil uptake during the frying step. | [88] | |
| Potatoes strips | Water bath/160 W/28–40 kHz/240 s | Treatment before drying | Useful to avoid excessive oil uptake during the frying step. | [89] | |
| Cassava, corn, and yam starch nanoparticles | Probe/20 kHz/30 min | Starch nanoparticles production | - | [90] | |
| Meat and Fish Products | Bovine semitendinosus muscle | Water bath/40 kHz/11 Wcm−2/60–90 s | Post-US process modifications | Improved water-holding capacity, controlled growth of mesophilic and psychrophilic bacteria and total coliforms. Increased meat luminosity and lowers pH without affecting the redness or yellowness. | [91] |
| Beef | Probe/150 W/20 kHz/2,39 Wcm−2/60–90 s | Meat brining | Rate of mass transfer is accelerated, and the value of D for NaCl is higher than D of water. | [95] | |
| Chicken broilers | US bath/25–130 kHz/28 W/L/5 to 30 min | Tenderization and decontamination | Reduction of about 40% prechiller process time. | [96] | |
| Beef | Probe/300 W/20 kHz/20–30 min | Curing | Water-holding capacity and tenderness are enhanced. | [97] | |
| Beef m. Longissimus dorsi muscle | US Bath/40 kHz/11 Wcm−2/60 min | Tenderization | Reduction in red color and increased pH, luminosity, size of fascicle, and greater interfibrillar space. | [98] | |
| Beef semitendinosus muscle | Water bath/1500 W/40 kHz/10–60 min | Post-US process modifications | US treatment deteriorates the stability of collagen and meat textural properties. | [99] | |
| Squid | Water bath/186.9 W/25.6 kHz/30.8 min | Tenderization | Broken fiber and proteins degradation create a softer meat. | [100] | |
| Cereal Products | Quinoa (Chenopodium quinoa) | Water bath/250 W/20 kHz/Up to 19 h | Post-US process modifications | Increased water solubility and in vitro starch digestibility, decreased gelatinization temperatures, enthalpy changes in viscosity, gelling capacity, antioxidant activity, and total phenolic content. | [101] |
| Flour batters and similar thick liquids | Probe/2.25 MHz | Monitoring the specific gravity | - | [102] | |
| Dehulled rice flour | Water bath 2000 W/25 kHz/16 W/L/5 min | Post-US process modifications | Transformed surface microstructure to facilitate moisture transfer during steam-cooking, enhanced starch hydrolysis, and glucose content. | [103] | |
| Buckwheat grains | Water bath/100 W/45 kHz/15 min | Post-US process modifications | Rise in the water absorption index, the swelling power of the flour, the water solubility index, and insoluble polyphenols content. Redness and yellowish enhancement. | [104] | |
| Noodles | US reaction tank/67 W/L/28–40-80 kHz/10 min | Post-US process modifications | Solubility, water-holding capacity and oil-holding capacity of gluten increased. Particle size reduced. UV absorption and fluorescence intensity of the treated gluten increased. The surface hydrophobicity of gluten increased. | [105] | |
| Dairy Products | Skimmed milk | Probe/28 kHz/100 W/L/30 min | Fermentation | Peptide content and viable cells increased by 49.5% and 43.5%, respectively. | [107] |
| Skimmed milk and goat milk | Probe/20–41 W/20 kHz /Up to 60 min | Stabilization and droplet size reduction | Droplets size reduced both for skimmed milk and goat milk. Increased homogenization for cream. | [107] | |
| Yogurt | Probe/150–750 W/20 kHz /10 min | Homogenization | Reduced milk fat globule size. | [108] | |
| Yogurt | Probe/100 W/30 kHz/2–15 min | Emulsification | Reduced milk fat globule size. | [109] | |
| Yogurt | Probe/250 W/20 kHz/20 min | Fermentation | Fermentation time reduced, enhanced gel strength and firmness. | [110] | |
| Ice cream | Probe/20 kHz/0.21 W/cm2/5 s | Freezing process support | Reduced ice crystal size, freezing time, and block crust on the freezing surface. | [110] | |
| Milk | Probe/0.4–1.6 MHz/5 min | Fractionation | Arrangement in layers. | [86] | |
| Emulsified Products | Mustard | Probe/750 W/20 kHz/30 min | Post-US process modifications | Increased stability index. | [111] |
| Emulsions with low oil soybean content | Probe/120 W/20 kHz/1 min | Post-US process modifications | Reduced suspension viscosity and size of the biopolymer complexes. | [112] |
| Category | Type | Process Parameters (Power/Wavelength/Intensity/Time/Distance from the Sample) | Use | Effects on Microorganism and Properties | Reference |
|---|---|---|---|---|---|
| Fruits and Vegetables | Orange surface | TUV 254 nm/35 W/17.2 mW/cm2/up to 20 min | Disinfection | E. coli reduced by 4.3 log10 CFU/mL. | [116] |
| Orange juice | TUV 254 nm/35 W/17.2 mW/cm2/20 min + HHP 400 MPa 1 min | Disinfection | E. coli reduced by 2.4 log10 CFU/mL. | [116] | |
| Apple juice | TUV 254 nm/16 W/8.45 J/cm2/+ HHP 400–500–600 MPa | Disinfection | L. monocytogenes, S. aureus totally inactivated. E. coli and S. typhimurium reduced by 7.1 and 7.2 log10 CFU/mL, respectively. S. cerevisiae reduced by 6.2 log10 CFU/mL. | [117] | |
| Apple juice | UV-C 254 nm/4 W/13 mW/cm2/5–10 min/1 cm | Disinfection | E. coli reduced by (3.2 ± 0.4) and (3.8 ± 0.2) log10 CFU/mL. | [118] | |
| Apple juice | LMP UV 254 nm/20 W/10 mJ/cm2/140 min/30.48 cm MPM UV 245 nm/2660 W/10 mJ/cm2/5 min/45.72 cm | Post-UV irradiation alterations | Vitamin C reduced by −1.30 ± 0.07% with LPM lamp after about 140 min and of −5.45 ± 0. 27% with MPM. pH not affected. | [119] | |
| Orange juice | LP UV 245 nm/8 W/23.72 J/mL/3.6 min at 55 °C | Disinfection | Reduction of 16.45% ascorbic acid content and 63.96% pectinmethylesterase activity. Acidity, pH, and color not affected; 6 log10 cycles of inactivation of E. coli. | [120] | |
| Coconut milk | UV-C 254 nm/17 W/0.342–1.026 kJ/m2 /30 min at 4 °C | Disinfection | E. coli and S. typhimurium reduced by (4.1 ± 0.1) log10 CFU/mL. pH, acidity and soluble solids not affected. Phenolic compounds reduced by 26.6%. | [121] | |
| Grape juice and wine | UV-C 245 nm/30 W/3672 J L−1 | Disinfection | Bacterial drop of 4.97 and 4.89 log10 CFU/mL in Chardonnay and Pinotage, respectively. Bacterial drop of 4.48 and 4.25 log10 CFU/mL in Chenin blanc and Shiraz juice, respectively. | [122] | |
| Black peppercorns | UV-LED 280 nm/20 min/1 cm | Disinfection | B. subtilis concentration decreased to (6.20 ± 0.44) log10 CFU/g. | [123] | |
| Onion, garlic, cheese and onion powders and chilli powder | UV-C-LEDs 270 nm/128 mJ/cm2/40 s/20 mm | Disinfection | L. monocytogenes, E. coli, B. subtilis, and S. typhimurium reduced by 0.75 up to 3 log10 CFU/g. | [124] | |
| Tomatoes |
UV-C 254 nm/8 W/1 kJ/m2/5 h UV-A 366 nm/8 W/1 kJ/m2/5 h | Compounds enhancement | Carotenoid content, phenols, or polyphenols concentration not considerably influenced. | [125] | |
| Meat and Fish Products | Beef | UV-C 254 nm/4 W/13 mW/cm2 /5 min/1 cm | Disinfection | E. coli reduced by (1.0 ± 0.2) log10 CFU/mL. | [118] |
| Chicken | UV-C 254 nm/4 W/13 mW/cm2 /5 min/1 cm | Disinfection | E. coli reduced by (1.6 ± 0.7) log10 CFU/mL. | [118] | |
| Pork | UV-C 254 nm/4 W/13 mW/cm2 /5 min/1 cm | Disinfection | E. coli reduced by (1.6 ± 0.4) log10 CFU/mL. | [118] | |
| Chicken breast | UV-C 254 nm/30–55 W/1.13–1.95 mW/cm2/up to 120 s/14 cm | Disinfection | Extended shelf life, decrease in bacterial load of (0.6 ± 0.03) log10 CFU/g. Rise in biogenic amines content, tyramine, cadaverine, and putrescine. | [126] | |
| Smoked salmon | UV-C 254 nm/30–55 W/900 mJ/cm2 | Disinfection | Drop of −0.5 /–1.3 log10 CFU unit/tot unit of bacterial population. No variation in sensory quality after 28 days of storage. | [127] | |
| Cereal Products | Cake batters | UV-C 254 nm/3.636 mJ/m2/up to 4 h | Post-UV irradiation alterations | Enhancement in browning reactions on the cake. Rise in specific volume. Unappealing taste and fragrance as the irradiation time increases. | [128] |
| Wheat flour | UV-C 254 nm/30 W/0.568 ± 0.026 mW/cm2/from 50 up to 250 s | Post-UV irradiation alterations | Decrease in gluten content. Rise in total volatile basic nitrogen content and photo-induced thiol-disulfide bridge exchange. Reduction in pH. | [129] | |
| Dairy Products | Milk | UV-C LPM 254 nm/20 W/10 mJ/cm2 /234 min UV-C MPM 254 nm/2.660 W/10 mJ/cm2/11 min | Post-UV irradiation alterations | Vitamin C reduced by −35.13 ± 1.56% with LPM UV lamp and of −61.67 ± 3.08% with MPM UV lamp. pH not affected. | [119] |
| Milk | UV-C 254 nm/28 W/88.2 J/mL | Post-UV irradiation alterations | Vitamin A, B2, and E decrease by 8 to 13%, 3 to 10%, and 16 to 33%, respectively. | [130] | |
| Goat milk | UV-C 254 nm/28 W/82.04 J/mL | Post-UV irradiation alterations | Vitamin A, B2, and E decrease by 1 to 9%, 1 to 2%, and 1 to 48%, respectively. | [130] | |
| Kashar cheese | UV-C 254 nm/32.1 W/m2/up to 300 s/4 cm | Disinfection | Bacterial reduction of up to (2.49) log10. CFU/g. Lipid oxidation causes a perception of off-flavor. No differences in color and hardness value. | [131] | |
| Ricotta | UV-C 254 nm/95 W/6.54 J/cm2/30 s/3.5 cm | Disinfection | P. fluorescens reduced by (−1.03 ± 0.02) log10 CFU/g. Ricotta lasts for 6 days without any alteration in sensory properties. | [132] | |
| Sliced cheese | UV-C 254 nm/3.04 mW/cm2/1 min/10 cm | Disinfection | PP and PE films reduced the most the levels of the pathogens. A thickness of 0,07 mm allows equal reduction in the three bacteria compared to non-packaged UV-treated samples. | [133] |
| Food Category | Product | Ozone Form and Concentration | Exposure Time | Effect | Reference |
|---|---|---|---|---|---|
| Fruits and Vegetables | Fresh-cut lettuce | Ozonated water, 2 mg L−1 | 5 min | 2.57 log reductions against S. Typhimurium color properties and sensory quality without any effect | [165] |
| 15 min | 3.47 log reductions against E. coli color properties and sensory quality without any effect | ||||
| Spinach | Gaseous ozone, 1 ppm | 10 min | 1 log reduction in E. coli and Listeria spp. | [166] | |
| Spinach | Combination of ozone 3.33 g min− 1 and ultrasound 40 kHz | 10 min | 1.46 log reduction in E. coli, Salmonella, and Listeria | [167] | |
| Fresh-cut green bell peppers | Aqueous ozone, 2.4 mg L−1 | 5 min | 3.71 log reduction in total plate count | [168] | |
| Strawberry | Aqueous ozone, 0.1 ppm | 2 min | Retention the fruit quality and extension the storage life | [169] | |
| Strawberry | Aqueous ozone, 3.5 mg L−1 | 5 min | Reduced decay caused by B. cinerea almost 17% | [170] | |
| Fresh-cut cabbage | Aqueous ozone, 1.4 mg L−1 | 5 min | Significantly inhibited aerobic bacteria, coliforms, and yeasts, reduced ethylene production | [171] | |
| Fresh-cut onions | Aqueous ozone, 1.4 mg L−1 | 5 min | Significantly inhibited aerobic bacteria, coliforms, and yeasts, reduced respiration rate, reduced residual levels of five tested pesticides | [172] | |
| Fish Products | White shrimp | Ozonated water, 1 ppm | 10 min | Increased shelf life (up to 24 days), maintained acceptable sensorial attributes | [173] |
| Sea bream | Aqueous ozone, 640 ppm | 15 min | 0.29 log reductions in Enterobacteriaceae | [174] | |
| Oyster | 0.6 mg/L/ | 6 h | 1.3 log reductions in E. coli | [175] | |
| Mushrooms | Agaricus bisporus | Gaseous ozone, 2.0 mg/L | 30 and 60 min | Increased firmness | [176] |
| Meat Products | Turkey breast meat | Gaseous ozone, 1 × 10−2 kg m−3 | up to 8 h | 2.9 log reductions in the counts of total aerobic mesophilic bacteria, 2.3 log reductions in enterobacteriaceae, 1.9 log reductions in yeast–mold, significant changes in color and pH, acceptable sensory properties | [177] |
| Beef | Gaseous ozone, 280 mg m−3 | 5 and 10 min every 30 min for 5 h | 1 log reductions in lactic acid bacteria, mesophilic and enterobacteriaceae | [178] | |
| Goat meat | Ozonated water, 0.68 mg/L | 6 min | 0.50 log reductions in E. coli | [179] | |
| Juices and Beverages | Peach juice | Gaseous ozone, 0.11 mg O3 min1 mL | 12 min | 99.5% decreased POD activities 93.9% decreased PPO activities | [180] |
| Gaseous ozone, 0.20 mg O3 min1 mL | 99.8% decreased POD activities 97.3% decreased PPO activities | ||||
| Apple juice | Gaseous ozone, 33–40 μg/mL | 8 min | Increased shelf life in 34 days at 8 °C storage | [181] | |
| Table Olives | Nocellara Etnea | Ozonated water, 6.5 ppm | 10 min | 1 log reductions for mesophilic aerobic bacteria and 1.47 log reductions for yeasts and mold | [182] |
| Food Category | Product | Pressure | Exposure Time | Temperature | Effect | Reference | |
|---|---|---|---|---|---|---|---|
| Fruits and Vegetables | Dried strawberry | 400 MPa | 10 min | 5 °C | Increased total phenolic content, same content in vitamin C | [232] | |
| Grapes | 200–550 MPa | 10 min | <30 °C | +80% extraction of anthocyanins | [211] | ||
| Fish Products | Frozen pink salmon fillets | 250 MPa | 3 min | −22 °C | 3 log reduction against L. monocytogenes and S. enterica | [215] | |
| Meat Products | Frozen chicken breast | 500 MPa | 1 min | 5 °C | Salmonella spp inactivation and preserve color parameters | [233] | |
| Chicken fille | 500 MPa | 10 min | 20 °C | S. Enteritidis inactivation and increased shelf life | [220] | ||
| Poultry- and pork-based semidried fermented sausage | 600 MPa | 960 s | 5 °C | L. monocytogenes and C. perfringens were below LOQ3 | [234] | ||
| Juices and Beverages | Sugarcane based mixed beverage | 300–500 MPa | 10–20 min | 40–60 °C | Inactivation in PPO1 (79%) and POD2 (72%) activity | [235] | |
| Pawpaw pulp | 600 MPa | 76 s | 4 °C | Significantly decreased PPO | [236] | ||
| Cloudy carrot juice | 300 MPa | 5 min | 22 °C | Inactivation in POD (31%) | [236] | ||
| 600 MPa | Inactivation in PPO (57%) | ||||||
| Dairy Products | Raw milk | 600 MPa | 5 min | 18 °C | 5 log reductions for E. coli, Salmonella and L. monocytogenes | [237] | |
| Cow and goat milk | 450 MPa | 7 min | 15 °C | Increased shelf life (up to 22 days to 8 °C) | [238] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiozzi, V.; Agriopoulou, S.; Varzakas, T. Advances, Applications, and Comparison of Thermal (Pasteurization, Sterilization, and Aseptic Packaging) against Non-Thermal (Ultrasounds, UV Radiation, Ozonation, High Hydrostatic Pressure) Technologies in Food Processing. Appl. Sci. 2022, 12, 2202. https://doi.org/10.3390/app12042202
Chiozzi V, Agriopoulou S, Varzakas T. Advances, Applications, and Comparison of Thermal (Pasteurization, Sterilization, and Aseptic Packaging) against Non-Thermal (Ultrasounds, UV Radiation, Ozonation, High Hydrostatic Pressure) Technologies in Food Processing. Applied Sciences. 2022; 12(4):2202. https://doi.org/10.3390/app12042202
Chicago/Turabian StyleChiozzi, Viola, Sofia Agriopoulou, and Theodoros Varzakas. 2022. "Advances, Applications, and Comparison of Thermal (Pasteurization, Sterilization, and Aseptic Packaging) against Non-Thermal (Ultrasounds, UV Radiation, Ozonation, High Hydrostatic Pressure) Technologies in Food Processing" Applied Sciences 12, no. 4: 2202. https://doi.org/10.3390/app12042202
APA StyleChiozzi, V., Agriopoulou, S., & Varzakas, T. (2022). Advances, Applications, and Comparison of Thermal (Pasteurization, Sterilization, and Aseptic Packaging) against Non-Thermal (Ultrasounds, UV Radiation, Ozonation, High Hydrostatic Pressure) Technologies in Food Processing. Applied Sciences, 12(4), 2202. https://doi.org/10.3390/app12042202








