Nitric Oxide Donor NOC-18-Induced Changes of Mitochondrial Phosphoproteome in Rat Cardiac Ischemia Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Animals and Induction of Heart Ischemia
2.3. Mitochondrial Preparation
2.4. Mitochondrial Incubation with PKG
2.5. Phosphoprotein Enrichment
2.6. In-Gel Protein Digestion for Mass Spectrometry Analysis
2.7. Protein Digestion and Sample Preparation for LC–MS/MS Analysis
2.8. Liquid Chromatography and Mass Spectrometry
2.9. Data Processing, Searching, and Analysis
2.10. Statistical Data Analysis
3. Results
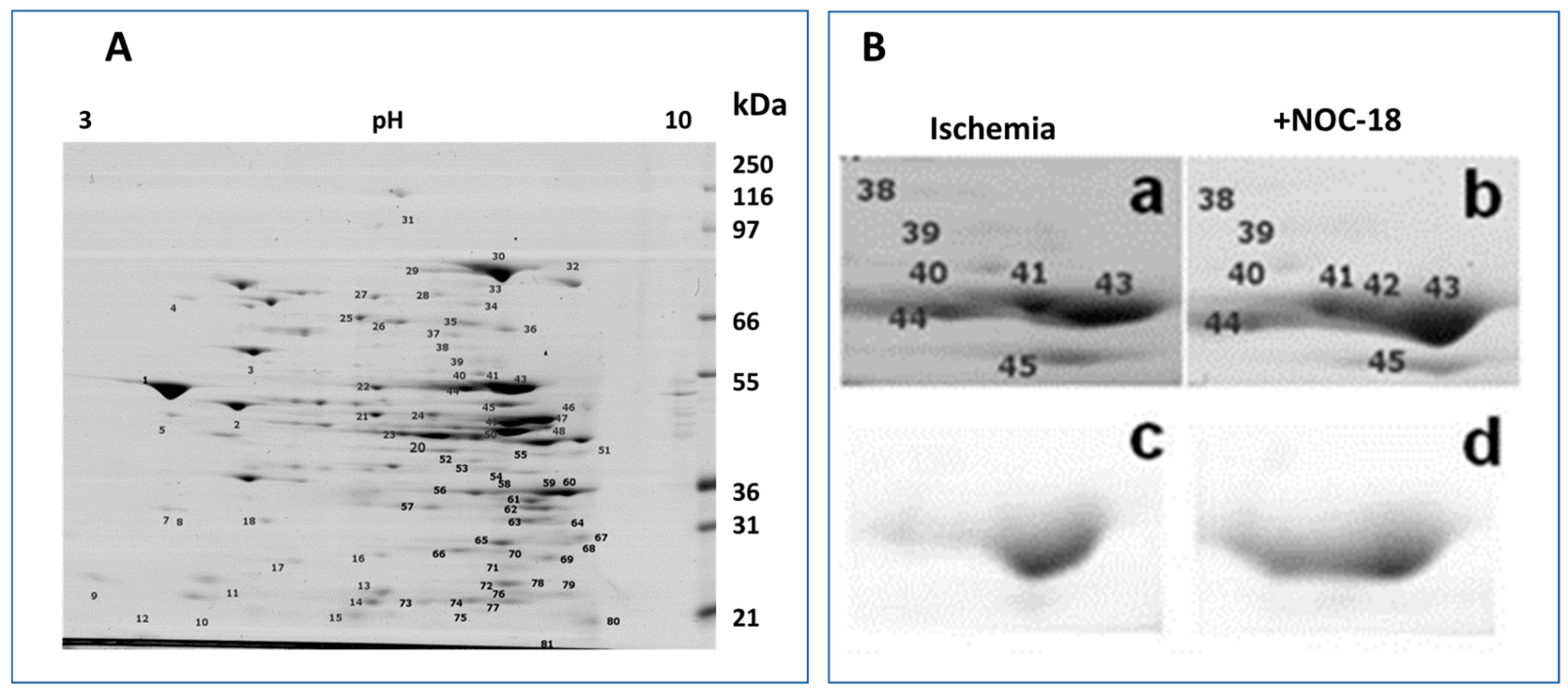
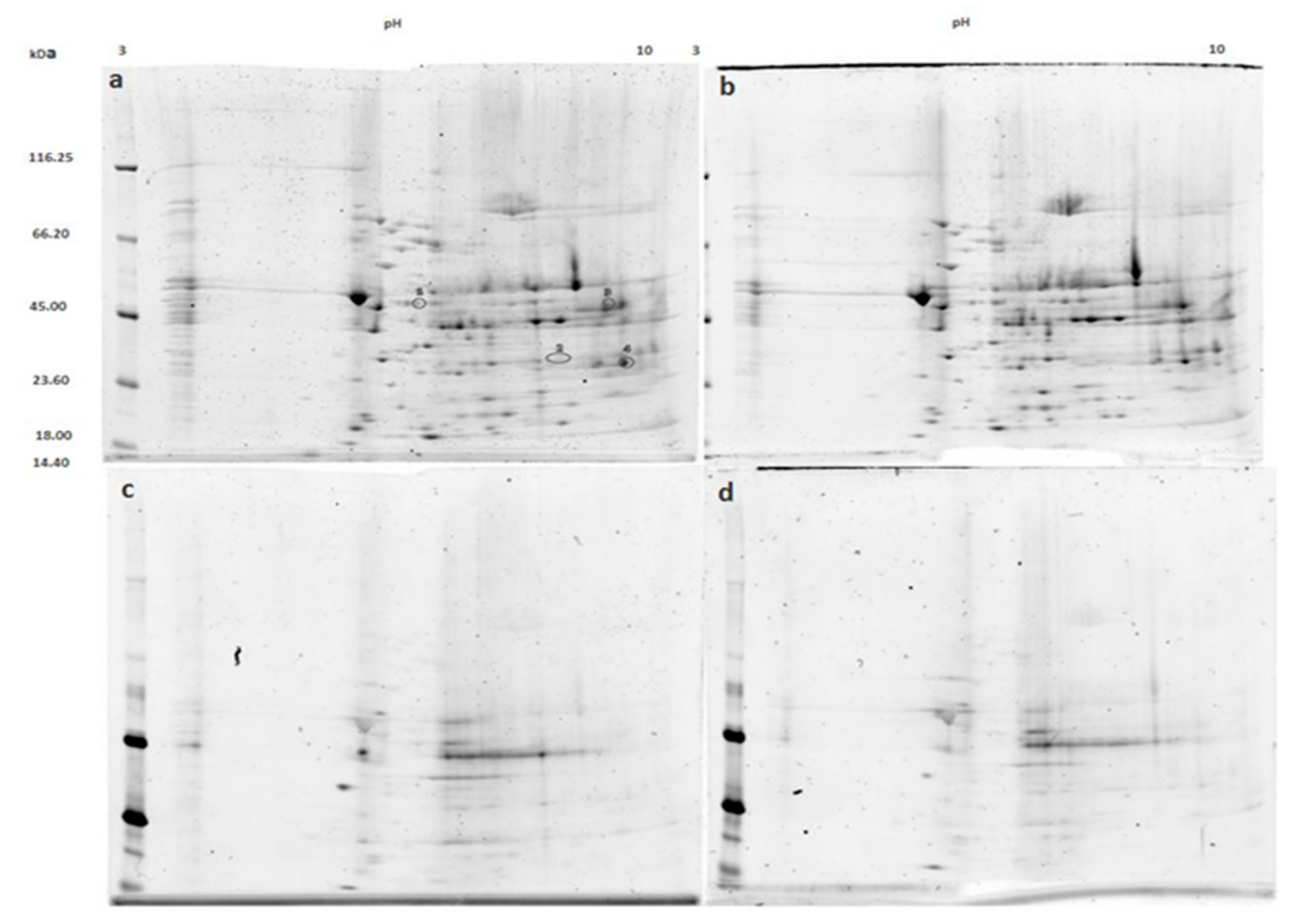
3.1. Mitochondrial Phosphoproteins in Response to NOC-18 versus Ischemia
3.2. Mitochondrial Proteins after Incubation with PKG
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Crompton, M.; Ellinger, H.; Costi, A. Inhibition by cyclosporin A of a Ca2+ dependent pore in heart mitochondria activated by inorganic phosphate and oxidative stress. Biochem. J. 1988, 255, 357–360. [Google Scholar] [PubMed]
- Crow, M.T.; Mani, K.; Nam, Y.-J.; Kitsis, R.N. The Mitochondrial Death Pathway and Cardiac Myocyte Apoptosis. Circ. Res. 2004, 95, 957–970. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boengler, K.; Lochnit, G.; Schulz, R. “THE” target of myocardial conditioning. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H1215–H1231. [Google Scholar] [CrossRef] [PubMed]
- Javadov, S.A.; Clarke, S.; Das, M.; Griffiths, E.J.; Lim, K.H.H.; Halestrap, A.P. Ischaemic preconditioning inhibits opening of mitochondrial permeability transition pores in the reperfused rat heart. J. Physiol. 2003, 549, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Argaud, L.; Gateau-Roesch, O.; Raisky, O.; Loufouat, J.; Robert, D.; Ovize, M. Postconditioning Inhibits Mitochondrial Permeability Transition. Circulation 2005, 111, 194–197. [Google Scholar] [CrossRef] [PubMed]
- Münzel, T.; Daiber, A.; Gori, T. Nitrate therapy: New aspects concerning molecular action and tolerance. Circulation 2011, 123, 2132–2144. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C.; Xie, Q.-W. Nitric oxide synthases: Roles, tolls, and controls. Cell 1994, 78, 915–918. [Google Scholar] [CrossRef]
- Andreadou, I.; Iliodromitis, E.K.; Rassaf, T.; Schulz, R.; Papapetropoulos, A.; Ferdinandy, P. The role of gasotransmitters NO, H2S and CO in myocardial ischaemia/reperfusion injury and cardioprotection by preconditioning, postconditioning and remote conditioning. Br. J. Pharmacol. 2015, 172, 1587–1606. [Google Scholar] [CrossRef] [PubMed]
- Borutaite, V.; Morkuniene, R.; Arandarcikaite, O.; Jekabsone, A.; Barauskaite, J.; Brown, G.C. Nitric oxide protects the heart from ischemia-induced apoptosis and mitochondrial damage via protein kinase G mediated blockage of permeability transition and cytochrome c release. J. Biomed. Sci. 2009, 16, 70. [Google Scholar] [CrossRef]
- Gornall, A.G.; Bardawill, C.J.; David, M.M. Determination of serum proteins by means of the biuret reaction. J. Boil. Chem. 1949, 177, 751–766. [Google Scholar]
- Hellman, U.; Wernstedt, C.; Gonez, J.; Heldin, C. Improvement of an “In-Gel” Digestion Procedure for the Micropreparation of Internal Protein Fragments for Amino Acid Sequencing. Anal. Biochem. 1995, 224, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Wiśniewski, J.R.; Zougman, A.; Nagaraj, N.; Mann, M. Universal sample preparation method for proteome analysis. Nat. Methods 2009, 6, 359. [Google Scholar] [CrossRef] [PubMed]
- Šimoliūnas, E.; Kaliniene, L.; Stasilo, M.; Truncaite, L.; Zajančkauskaite, A.; Staniulis, J.; Nainys, J.; Kaupinis, A.; Valius, M.; Meškys, R. Isolation and Characterization of vB_ArS-ArV2—First Arthrobacter sp. Infecting Bacteriophage with Completely Sequenced Genome. PLoS ONE 2014, 9, e111230. [Google Scholar] [CrossRef] [PubMed]
- Bonora, M.; Pinton, P. A New Current for the Mitochondrial Permeability Transition. Trends Biochem. Sci. 2019, 44, 559–561. [Google Scholar] [CrossRef] [PubMed]
- Detaille, D.; Guigas, B.; Chauvin, C.; Batandier, C.; Fontaine, E.; Wiernsperger, N.; Leverve, X. Metformin prevents high-glucose-induced endothelial cell death through a mitochondrial permeability transition-dependent process. Diabetes 2005, 54, 2179–2187. [Google Scholar] [CrossRef]
- Fontaine, E.; Detaille, D.; Vial, G. Preventing cell death with a ‘check valve’ in mitochondrial complex I? Cell Death Dis. 2016, 7, e2165. [Google Scholar] [CrossRef]
- Arandarcikaite, O.; Jokubka, R.; Borutaite, V. Neuroprotective effects of nitric oxide donor NOC-18 against brain ischemia-induced mitochondrial damages: Role of PKG and PKC. Neurosci. Lett. 2015, 586, 65–70. [Google Scholar] [CrossRef]
- Costa, A.D.T.; Garlid, K.D. Intramitochondrial signaling: Interactions among mitoK ATP, PKCε, ROS, and MPT. Am. J. Physiol. Heart Circ. Physiol. 2008, 295, H874–H882. [Google Scholar] [CrossRef]
- Oldenburg, O.; Qin, Q.; Krieg, T.; Yang, X.M.; Philipp, S.; Critz, S.D.; Cohen, M.V.; Downey, J.M. Bradykinin induces mitochondrial ROS generation via NO, cGMP, PKG, and mitoK ATP channel opening and leads to cardioprotection. Am. J. Physiol. Heart Circ. Physiol. 2004, 286, H468–H476. [Google Scholar] [CrossRef]
- Costa, A.D.; Garlid, K.D.; West, I.C.; Lincoln, T.M.; Downey, J.M.; Cohen, M.V.; Critz, S.D. Protein Kinase G Transmits the Cardioprotective Signal from Cytosol to Mitochondria. Circ. Res. 2005, 97, 329–336. [Google Scholar] [CrossRef]
- Applegate, M.A.B.; Humphries, K.M.; Szweda, L.I. Reversible Inhibition of α-Ketoglutarate Dehydrogenase by Hydrogen Peroxide: Glutathionylation and Protection of Lipoic Acid. Biochemistry 2008, 47, 473–478. [Google Scholar] [CrossRef]
- Jo, S.H.; Son, M.K.; Koh, H.J.; Lee, S.M.; Song, I.H.; Kim, Y.O.; Lee, Y.S.; Jeong, K.S.; Kim, W.B.; Park, J.W.; et al. Control of Mitochondrial Redox Balance and Cellular Defense against Oxidative Damage by Mitochondrial NADP+-dependent Isocitrate Dehydrogenase. J. Biol. Chem. 2001, 276, 16168–16176. [Google Scholar] [CrossRef]
- Yu, W.; Dittenhafer-Reed, K.E.; Denu, J.M. SIRT3 Protein Deacetylates Isocitrate Dehydrogenase 2 (IDH2) and Regulates Mitochondrial Redox Status. J. Boil. Chem. 2012, 287, 14078–14086. [Google Scholar] [CrossRef] [Green Version]
- Kim, E.Y.; Kim, W.K.; Kang, H.J.; Kim, J.-H.; Chung, S.J.; Seo, Y.S.; Park, S.G.; Lee, S.C.; Bae, K.-H. Acetylation of malate dehydrogenase 1 promotes adipogenic differentiation via activating its enzymatic activity. J. Lipid Res. 2012, 53, 1864–1876. [Google Scholar] [CrossRef] [Green Version]
- Shi, Q.; Gibson, G.E. Up-regulation of the mitochondrial malate dehydrogenase by oxidative stress is mediated by miR-743a. J. Neurochem. 2011, 118, 440–448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, S.; Fu, Y.; Wang, X.; Shi, H.; Huang, Y.; Song, X.; Li, L.; Song, N.; Luo, Y. Voltage-dependent anion channel 1 is involved in endostatin-induced endothelial cell apoptosis. FASEB J. 2008, 22, 2809–2820. [Google Scholar] [CrossRef]
- Ottani, F.; Latini, R.; Staszewsky, L.; La Vecchia, L.; Locuratolo, N.; Sicuro, M.; Masson, S.; Barlera, S.; Milani, V.; Lombardi, M.; et al. CYCLE Investigators. Cyclosporine A in Reperfused Myocardial Infarction: The Multicenter, Controlled, Open-Label CYCLE Trial. J. Am. Coll. Cardiol. 2016, 67, 365–374. [Google Scholar] [CrossRef]

| Mitochondrial Membrane-Bound Proteins | UniProt/SwissProt Number | Fold Change NOC-18/Ischemia | P-Value |
|---|---|---|---|
| Mitochondrial pyruvate carrier 1 (MPC1) | P63031 | 1.64 | 0.003 |
| NADH dehydrogenase ubiquinone flavoprotein 2 (NDUFV2) | P19234 | −1.53 | 0.009 |
| Very long chain specific acyl CoA dehydrogenase mitochondrial (ACADVL) | P45953 | 1.58 | 0.013 |
| Mitochondrial 2 oxoglutarate malatecarrier protein (SLC25A11) | P97700 | 1.66 | 0.013 |
| ADP/ATP translocase 1 (SLC25A4) | Q05962 | 1.57 | 0.022 |
| ATP synthase subunit alpha mitochondrial (ATP5A1) | P15999 | 1.55 | 0.028 |
| Trifunctional enzyme subunit beta mitochondrial (HADHB) | Q60587 | 1.61 | 0.032 |
| Electron transfer flavoprotein ubiquinone oxidoreductase mitochondrial (ETFD) | Q6UPE1 | 1.63 | 0.041 |
| Cytochrome c oxidase subunit 4isoform 1 mitochondrial (COX4I1) | P10888 | 1.54 | 0.042 |
| Cytochrome b c1 complex subunit Rieske (UQCRFS1) | P20788 | 1.54 | 0.043 |
| Mitochondrial Matrix Proteins | |||
| Pyruvate dehydrogenase E1 component subunit alpha somatic form mitochondrial (PDHA1) | P26284 | 1.55 | 0.024 |
| Short chain specific acyl CoAdehydrogenase mitochondrial (ACADS) | P15651 | 1.58 | 0.027 |
| Long chain specific acyl CoAdehydrogenase mitochondrial (ACADVL) | P15650 | 1.59 | 0.027 |
| Isovaleryl CoA dehydrogenase mitochondrial (IVD) | P12007 | 1.59 | 0.030 |
| Succinyl CoA ligase ADP GDP forming subunit alpha mitochondrial (SUCLG1) | P13086 | 1.62 | 0.030 |
| Lon protease homolog mitochondrial (LONP1) | Q924S5 | 1.75 | 0.032 |
| Hexaprenyldihydroxybenzoatemethyltransferase mitochondrial (COQ3) | Q63159 | 1.67 | 0.032 |
| Citrate synthase mitochondrial (CS) | Q8VHF5 | 1.57 | 0.034 |
| 3-ketoacyl CoA thiolase mitochondrial (ACAA2) | P13437 | 1.57 | 0.044 |
| Malate dehydrogenase mitochondrial (MDH2) | P04636 | 1.54 | 0.049 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umbrasas, D.; Jokubka, R.; Kaupinis, A.; Valius, M.; Arandarčikaitė, O.; Borutaitė, V. Nitric Oxide Donor NOC-18-Induced Changes of Mitochondrial Phosphoproteome in Rat Cardiac Ischemia Model. Medicina 2019, 55, 631. https://doi.org/10.3390/medicina55100631
Umbrasas D, Jokubka R, Kaupinis A, Valius M, Arandarčikaitė O, Borutaitė V. Nitric Oxide Donor NOC-18-Induced Changes of Mitochondrial Phosphoproteome in Rat Cardiac Ischemia Model. Medicina. 2019; 55(10):631. https://doi.org/10.3390/medicina55100631
Chicago/Turabian StyleUmbrasas, Danielius, Ramūnas Jokubka, Algirdas Kaupinis, Mindaugas Valius, Odeta Arandarčikaitė, and Vilmantė Borutaitė. 2019. "Nitric Oxide Donor NOC-18-Induced Changes of Mitochondrial Phosphoproteome in Rat Cardiac Ischemia Model" Medicina 55, no. 10: 631. https://doi.org/10.3390/medicina55100631







