The Impact of High-Flow Nasal Cannula on the Outcome of Immunocompromised Patients with Acute Respiratory Failure: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
Study Search and Selection
3. Results
3.1. Study Selection and Characteristics
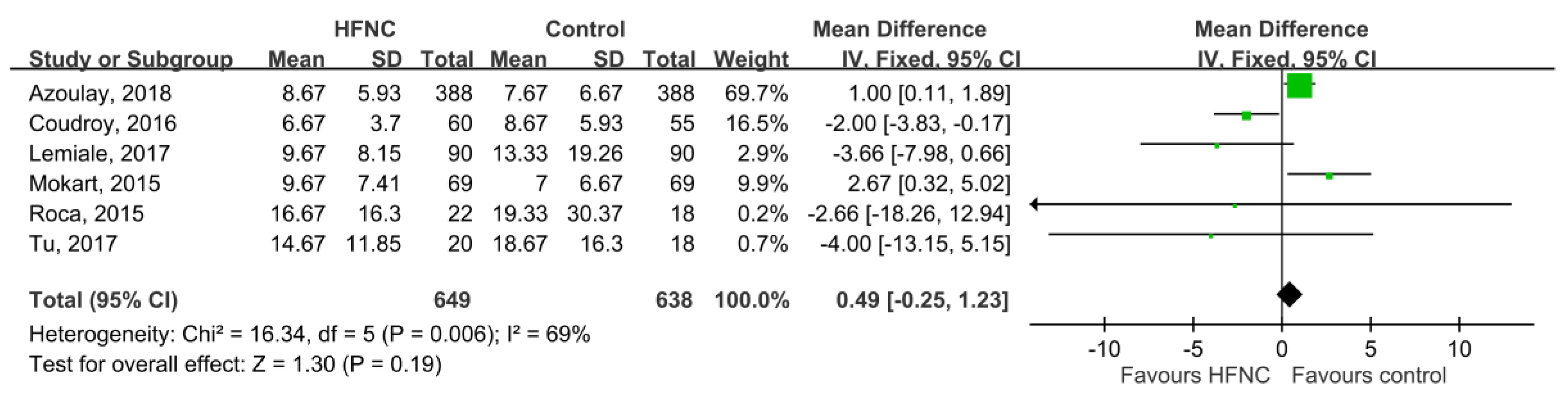
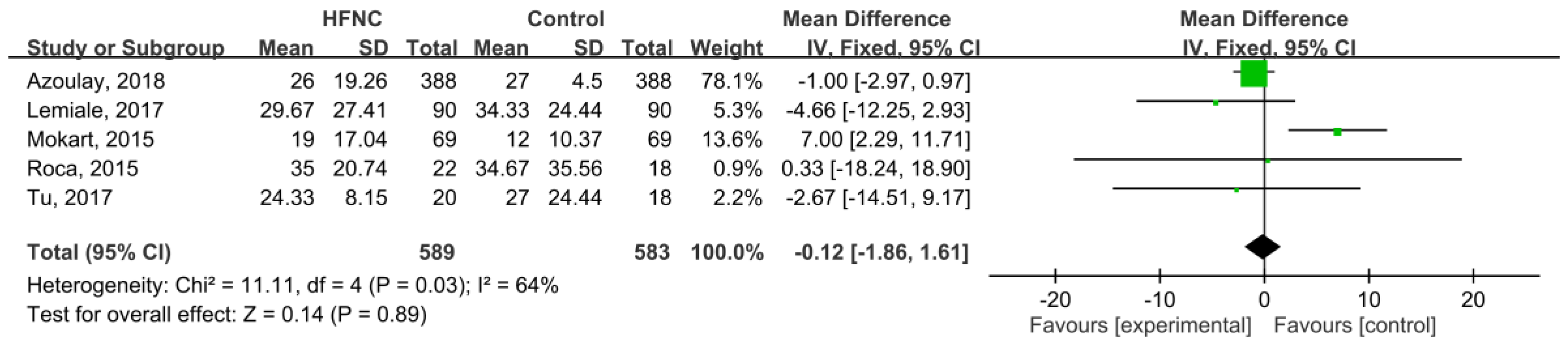
3.2. Primary Outcomes
3.3. Secondary Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Reference | Random Sequence Generation | Allocation Concealment | Blinding of Participants and Personnel | Blinding of Outcome Assessment | Incomplete Outcome Data | Selective Reporting |
|---|---|---|---|---|---|---|
| Frat, 2016 | Low | High | High | High | Low | Low |
| Lemiale, 2017 | Low | High | High | High | Low | Low |
| Lemiale, 2015 | Low | High | High | High | Low | Low |
| Azoulay, 2018 | Low | Low | High | High | Low | Low |
| Reference | Representative of Exposed Cohort | Selection of Non-Exposed Cohort | Ascertainment of Exposure | Demonstration that Outcome Was Not Present at Start of Study | Comparability of Cohorts Based on Design and Analysis | Assessment of Outcome | Timing of Follow-Up | Adequate Follow-Up | Score |
|---|---|---|---|---|---|---|---|---|---|
| Mokart, 2015 | V | V | V | V | V | V | V | V | 8 |
| Roca, 2015 | V | V | V | V | V | V | V | V | 8 |
| Coudroy, 2016 | V | V | V | V | V | V | V | V | 8 |
| Tu, 2017 | V | V | V | V | V | V | V | V | 8 |
References
- Allemani, C.; Weir, H.K.; Carreira, H.; Harewood, R.; Spika, D.; Wang, X.-S.; Bannon, F.; Ahn, J.V.; Johnson, C.J.; Bonaventure, A.; et al. Global surveillance of cancer survival 1995–2009: Analysis of individual data for 25 676 887 patients from 279 population-based registries in 67 countries (CONCORD-2). Lancet 2015, 385, 977–1010. [Google Scholar] [CrossRef]
- Allemani, C.; Matsuda, T.; Di Carlo, V.; Harewood, R.; Matz, M.; Niksic, M.; Bonaventure, A.; Valkov, M.; Johnson, C.J.; Esteve, J.; et al. Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): Analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet 2018, 391, 1023–1075. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Israni, A.K.; Zaun, D.; Rosendale, J.; Schaffhausen, C.; Snyder, J.J.; Kasiske, B.L. OPTN/SRTR 2016 Annual Data Report: Deceased Organ Donation. Arab. Archaeol. Epigr. 2018, 18, 434–463. [Google Scholar] [CrossRef]
- Weyand, C.M.; Goronzy, J.J. Clinical practice. Giant-cell arteritis and polymyalgia rheumatica. N. Engl. J. Med. 2014, 371, 50–57. [Google Scholar] [CrossRef]
- Poteat, T.; Scheim, A.; Xavier, J.; Reisner, S.; Baral, S. Global epidemiology of HIV infection and related syndemics affecting transgender people. J. Acquir. Immune Defic. Syndr. 2016, 72, S210–S219. [Google Scholar] [CrossRef]
- Azoulay, E.; Pickkers, P.; Soares, M.; Perner, A.; Rello, J.; Bauer, P.R.; van de Louw, A.; Hemelaar, P.; Lemiale, V.; Taccone, F.S.; et al. Acute hypoxemic respiratory failure in immunocompromised patients: The Efraim multinational prospective cohort study. Intensive Care Med. 2017, 43, 1808–1819. [Google Scholar] [CrossRef]
- Canet, E.; Osman, D.; Lambert, J.; Guitton, C.; Heng, A.-E.; Argaud, L.; Klouche, K.; Mourad, G.; Legendre, C.; Timsit, J.F.; et al. Acute respiratory failure in kidney transplant recipients: A multicenter study. Crit. Care 2011, 15, R91. [Google Scholar] [CrossRef]
- Lai, C.C.; Ho, C.H.; Chen, C.M.; Chiang, S.R.; Chao, C.M.; Liu, W.L.; Wang, J.J.; Yang, C.C.; Cheng, K.C. Risk factors and mortality of adults with lung cancer admitted to the intensive care unit. J. Thorac. Dis. 2018, 10, 4118–4126. [Google Scholar] [CrossRef]
- Moreau, A.S.; Martin-Loeches, I.; Povoa, P.; Salluh, J.; Rodriguez, A.; Thille, A.W.; Santos, E.D.; Vedes, E.; Lobo, S.M.; Mégarbane, B.; et al. Impact of immunosuppression on incidence, aetiology and outcome of ventilator-associated lower respiratory tract infections. Eur. Respir. J. 2018, 51, 1701656. [Google Scholar] [CrossRef]
- Azoulay, E.; Lemiale, V.; Mokart, D.; Pène, F.; Kouatchet, A.; Perez, P.; Vincent, F.; Mayaux, J.; Benoit, D.; Bruneel, F.; et al. Acute respiratory distress syndrome in patients with malignancies. Intensiv. Care Med. 2014, 40, 1106–1114. [Google Scholar] [CrossRef] [PubMed]
- Lemiale, V.; Mokart, D.; Resche-Rigon, M.; Pène, F.; Mayaux, J.; Faucher, E.; Nyunga, M.; Girault, C.; Perez, P.; Guitton, C.; et al. Effect of Noninvasive Ventilation vs Oxygen Therapy on Mortality among Immunocompromised Patients with Acute Respiratory Failure: A Randomized Clinical Trial. JAMA 2015, 314, 1711–1719. [Google Scholar] [CrossRef] [PubMed]
- Hernández, G.; Roca, O.; Colinas, L. High-Flow Nasal Cannula Support Therapy: New Insights and Improving Performance. Crit. Care 2017, 315, 237–253. [Google Scholar]
- Ward, J.J. High-flow oxygen administration by nasal cannula for adult and perinatal patients. Respir. Care 2013, 58, 98–122. [Google Scholar] [CrossRef]
- Helviz, Y.; Einav, S. A Systematic Review of the High-flow Nasal Cannula for Adult Patients. Crit. Care 2018, 22, 177–191. [Google Scholar] [CrossRef]
- Ou, X.; Hua, Y.; Liu, J.; Gong, C.; Zhao, W. Effect of high-flow nasal cannula oxygen therapy in adults with acute hypoxemic respiratory failure: A meta-analysis of randomized controlled trials. Can. Med. Assoc. J. 2017, 189, E260–E267. [Google Scholar] [CrossRef]
- Ni, Y.N.; Luo, J.; Yu, H.; Liu, D.; Liang, B.M.; Liang, Z.A. The effect of high-flow nasal cannula in reducing the mortality and the rate of endotracheal intubation when used before mechanical ventilation compared with conventional oxygen therapy and noninvasive positive pressure ventilation. A systematic review and meta-analysis. Am. J. Emerg. Med. 2018, 36, 226–233. [Google Scholar]
- Leeies, M.; Flynn, E.; Turgeon, A.F.; Paunovic, B.; Loewen, H.; Rabbani, R.; Abou-Setta, A.M.; Ferguson, N.D.; Zarychanski, R. High-flow oxygen via nasal cannulae in patients with acute hypoxemic respiratory failure: A systematic review and meta-analysis. Syst. Rev. 2017, 6, 202. [Google Scholar] [CrossRef]
- Zhu, Y.; Yin, H.; Zhang, R.; Wei, J. High-flow nasal cannula oxygen therapy versus conventional oxygen therapy in patients with acute respiratory failure: A systematic review and meta-analysis of randomized controlled trials. BMC Pulm. Med. 2017, 17, 201. [Google Scholar] [CrossRef]
- Huang, H.B.; Peng, J.M.; Weng, L.; Liu, G.Y.; Du, B. High-flow oxygen therapy in immunocompromised patients with acute respiratory failure: A review and meta-analysis. J. Crit. Care 2018, 43, 300–305. [Google Scholar] [CrossRef]
- Azoulay, E.; Lemiale, V.; Mokart, D.; Nseir, S.; Argaud, L.; Pène, F.; Kontar, L.; Bruneel, F.; Klouche, K.; Barbier, F.; et al. Effect of High-Flow Nasal Oxygen vs Standard Oxygen on 28-Day Mortality in Immunocompromised Patients With Acute Respiratory Failure: The HIGH Randomized Clinical Trial. JAMA 2018, 320, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Wang, W.; Liu, J.; Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014, 14, 135. [Google Scholar] [CrossRef] [PubMed]
- Mokart, D.; Geay, C.; Chow-Chine, L.; Brun, J.P.; Faucher, M.; Blache, J.L.; Bisbal, M.; Sannini, A. High-flow oxygen therapy in cancer patients with acute respiratory failure. Intensive Care Med. 2015, 41, 2008–2010. [Google Scholar] [CrossRef] [PubMed]
- Roca, O.; De Acilu, M.G.; Caralt, B.; Sacanell, J.; Masclans, J.R. Humidified High Flow Nasal Cannula Supportive Therapy Improves Outcomes in Lung Transplant Recipients Readmitted to the Intensive Care Unit Because of Acute Respiratory Failure. Transplantion 2015, 99, 1092–1098. [Google Scholar] [CrossRef]
- Lemiale, V.; Resche-Rigon, M.; Mokart, D.; Pène, F.; Argaud, L.; Mayaux, J.; Guitton, C.; Rabbat, A.; Girault, C.; Kouatchet, A.; et al. High-flow nasal cannula oxygenation in immunocompromised patients with acute hypoxemic respiratory failure: A Groupe de Recherche Respiratoire en Reanimation Onco-Hematologique Study. Crit. Care Med. 2017, 45, e274–e280. [Google Scholar] [CrossRef]
- Lemiale, V.; Mokart, D.; Mayaux, J.; Lambert, J.; Rabbat, A.; Demoule, A.; Azoulay, E. The effects of a 2-h trial of high-flow oxygen by nasal cannula versus Venturi mask in immunocompromised patients with hypoxemic acute respiratory failure: A multicenter randomized trial. Crit. Care 2015, 19, 380. [Google Scholar] [CrossRef]
- Tu, G.; He, H.; Yin, K.; Ju, M.; Zheng, Y.; Zhu, D.; Luo, Z. High-flow Nasal Cannula Versus Noninvasive Ventilation for Treatment of Acute Hypoxemic Respiratory Failure in Renal Transplant Recipients. Transplant. Proc. 2017, 49, 1325–1330. [Google Scholar] [CrossRef]
- Frat, J.P.; Ragot, S.; Girault, C.; Perbet, S.; Prat, G.; Boulain, T.; Demoule, A.; Ricard, J.D.; Coudroy, R.; Robert, R.; et al. Effect of non-invasive oxygenation strategies in immunocompromised patients with severe acute respiratory failure: A post-hoc analysis of a randomised trial. Lancet Respir. Med. 2016, 4, 646–652. [Google Scholar] [CrossRef]
- Coudroy, R.; Jamet, A.; Petua, P.; Robert, R.; Frat, J.P.; Thille, A.W. High-flow nasal cannula oxygen therapy versus noninvasive ventilation in immunocompromised patients with acute respiratory failure: An observational cohort study. Ann. Intensive Care 2016, 6, 45. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Li, Y.; Zhou, J.; Li, X.; Huang, Y.; Liu, X.; Burns, K.E.A.; Zhong, N.; Zhang, H. High-flow nasal cannula in adults with acute respiratory failure and after extubation: A systematic review and meta-analysis. Respir. Res. 2018, 19, 202. [Google Scholar] [CrossRef] [PubMed]




| Study, Year | Study Design | Study Period | Study Site | Immunocompromised Condition | Inclusion Criteria |
|---|---|---|---|---|---|
| Tu, 2017 | Retrospective | 2011–2015 | Single tertiary mixed ICU in China | Renal transplant | RR > 25/min, PaO2/FiO2 ≤ 200 mm Hg, PaCO2 ≤ 45 mm Hg |
| Coudroy, 2016 | Retrospective | 2007–2014 | Single tertiary medical ICU in France | Hematologic or solid cancer, stem or solid organ transplantation, steroid, cytotoxic drug, AIDS | RR > 25/min, sign of respiratory distress, PaO2/FiO2 ≤ 300 mm Hg |
| Frat, 2016 | Post-hoc analysis of RCT | 2011–2013 | 23 ICUs in France and Belgium | Solid or hematological cancer, AIDS, immunosuppressive drug or steroid | RR > 25/min, PaO2/FiO2 ≤ 300 mm Hg, PaCO2 ≤ 45 mm Hg |
| Lemiale, 2017 | Post-hoc analysis of RCT | 2013–2015 | 28 ICUs in France and Belgium | Hematologic malignancy or solid tumor, solid organ transplant, long-term or high-dose steroid, immunosuppressive drug | PaO2 < 60 mm Hg on room air or tachypnea> 30/min or symptoms of respiratory distress |
| Lemiale, 2015 | Open, parallel-group RCT | 2012–2014 | 4 ICUs in France | Solid or hematological cancer, solid organ transplant, long-term or high-dose steroid, immunosuppressive drug, HIV infection | O2 delivery >6 L/min to maintain SpO2 > 95% or symptoms of respiratory distress |
| Mokart, 2015 | Retrospective | 2009–2014 | Single center in France | Cancer | Severe acute respiratory failure (O2 delivery >9 L/min) |
| Roca, 2015 | Retrospective | 2007–2011 | Single center in Spain | Lung transplant | Inability to maintain SpO2 > 95% and an RR ≥ 25/min |
| Azoulay, 2018 | RCT | 2016–2017 | 32 ICUs in France | Hematologic malignancy or solid tumor, solid organ transplant, long-term or high-dose steroid, immunosuppressive drug, primary immune deficiency | PaO2 < 60 mm Hg or SpO2 < 90% on room air or tachypnea >30/min or symptoms of respiratory distress, need for oxygen ≥6 L/min |
| Study, Year | No. of Patients | Mean Age | Percentage of Common ARF Etiology | Severity | ||||
|---|---|---|---|---|---|---|---|---|
| HFNC | Control Group | HFNC | Control Group | HFNC | Control Group | HFNC | Control Group | |
| Tu, 2017 | 20 | 18 (NIV) | 47 | 47 | NA | NA | SAPS II 37 (4) | SAPS II 35 (6) |
| Coudroy, 2016 | 60 | 55 (NIV) | 58 | 62 | Infection (44%), cardiogenic edema (9%) | Infection (52%), cardiogenic edema (8%) | SAPS II 42 (11) SOFA 3 (1–6) | SAPS II 46 (13) SOFA 4 (1–6) |
| Frat, 2016 | 26 | 30 (COT) | 62 | 63 | Pneumonia (66%), cancer (12%), others (23%) | Pneumonia (94%), cancer (3%), others (3%) | SAPS II 29 (11) | SAPS II 30 (17) |
| Lemiale, 2017 | 90 | 90 (COT) | 64 | 63 | Infection (73.3%), cardiogenic edema (2.2%) other (24.4%) | Infection (68.8%), cardiogenic edema (2.2) other (28.8%) | SOFA 4 (2–6) | SOFA 3 (2–6) |
| Lemiale, 2015 | 52 | 48 (COT) | 50 | 49 | Sepsis (48.1%), PJP (9.6%), cardiogenic edema (9.6%) | Sepsis (52%), PJP (4.1%), cardiogenic edema (4.1%) | SOFA 3.5 (2–6) SAPS II 42 (29.5–52) | SOFA 3 (2–5) SAPS II 37.5 (31–47) |
| Mokart, 2015 | 69 (HFNC + NIV) | 59 (NIV + COT) | 56 | 59 | Pulmonary sepsis (65%), cancer (19%), others (26%) | Pulmonary sepsis (65%), cancer (9%), others (43%) | SOFA 6 (4–8) SPAS II 47 (37–55) | SOFA 6 (4–9) SAPS 48 (3859) |
| Roca, 2015 | 22 | 18 (COT) | 56 | 53.5 | Infection (91.0%), rejection (4.5%) | Infection (72.2%), rejection (5.6%) | SOFA 4 (4–6) | SOFA 4 (4–6) |
| Azoulay, 2018 | 388 | 388 (COT) | 64 | 63 | NA | NA | SAPS II 36 (28–46); SOFA 6 (4–8) | SAPS II 37 (28–48); SOFA 6 (4–8) |
| Subgroup | No of Study | No of Patients | Random-Effect Model | I2 (%) | Test of Heterogeneity P | ||
|---|---|---|---|---|---|---|---|
| HFNC | Control | Risk Ratio | 95% CI | ||||
| Comparator | |||||||
| HFNC vs. COT | 5 | 578 | 574 | 0.86 | 0.75–0.95 | 0 | 0.57 |
| HFNC vs. NIV | 2 | 80 | 73 | 0.59 | 0.40–0.86 | 0 | 0.39 |
| HFNC + NIV vs. COT + NIV | 1 | 69 | 69 | 0.92 | 0.66–1.28 | NA | NA |
| Study design | |||||||
| RCT | 2 | 440 | 436 | 0.89 | 0.76–1.06 | 0 | 0.38 |
| Retrospective study | 4 | 171 | 160 | 0.73 | 0.58–0.90 | 21 | 0.28 |
| Post-hoc analysis | 2 | 116 | 120 | 0.81 | 0.61–1.07 | 0 | 0.68 |
| Study site | |||||||
| Single center | 4 | 171 | 160 | 0.73 | 0.58–0.90 | 21 | 0.28 |
| Multicenter | 4 | 556 | 556 | 0.87 | 0.76–1.01 | 0 | 0.88 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, L.-C.; Chang, S.-P.; Wang, J.-J.; Hsiao, S.-Y.; Lai, C.-C.; Chao, C.-M. The Impact of High-Flow Nasal Cannula on the Outcome of Immunocompromised Patients with Acute Respiratory Failure: A Systematic Review and Meta-Analysis. Medicina 2019, 55, 693. https://doi.org/10.3390/medicina55100693
Cheng L-C, Chang S-P, Wang J-J, Hsiao S-Y, Lai C-C, Chao C-M. The Impact of High-Flow Nasal Cannula on the Outcome of Immunocompromised Patients with Acute Respiratory Failure: A Systematic Review and Meta-Analysis. Medicina. 2019; 55(10):693. https://doi.org/10.3390/medicina55100693
Chicago/Turabian StyleCheng, Li-Chin, Shen-Peng Chang, Jian-Jhong Wang, Sheng-Yen Hsiao, Chih-Cheng Lai, and Chien-Ming Chao. 2019. "The Impact of High-Flow Nasal Cannula on the Outcome of Immunocompromised Patients with Acute Respiratory Failure: A Systematic Review and Meta-Analysis" Medicina 55, no. 10: 693. https://doi.org/10.3390/medicina55100693





