The Impact of MGMT Promoter Methylation and Temozolomide Treatment in Serbian Patients with Primary Glioblastoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients and Tumor Specimens
- Group 1 (n = 10 patients): temozolomide (TMZ)—the first cycle at a dose of 150 mg/m2 for 5 days; the next 5 cycles at a dose of 200 mg/m2. Cycles were repeated every 3 weeks.
- Group 2 (n = 10 patients): procarbazine, lomustine (1-[2-chloroethyl]-3-cyclohexyl-1-chloroethylnitrosourea (CCNU)) and vincristine (PCV regimen): CCNU 110 mg/m2 p.o. day 1; procarbazine 60 mg/m2 per os (p.o.) days 8–21; vincristine 1.4 mg/m2 (maximum 2 mg), i.e., days 8 and 21. Cycles were repeated every 6–8 weeks.
- Group 3 (n = 10 patients): carmustine (BCNU) 200 mg/m2, i.e., day 1. Cycles were repeated every 8 weeks.
2.2. DNA Isolation and Bisulfite Conversion
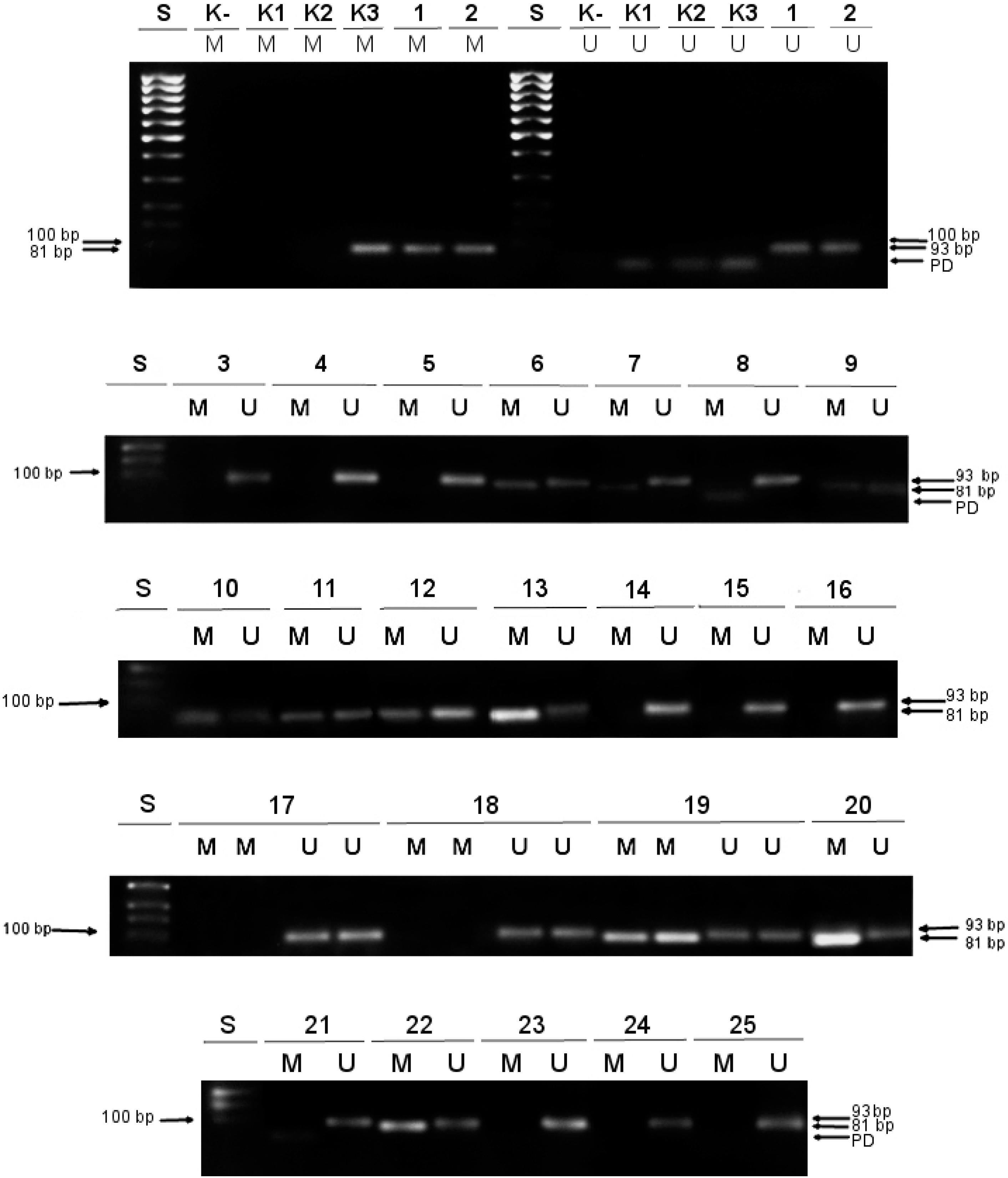
2.3. Methylation-Specific Polymerase Chain Reaction (MSP)
- -
- unmethylated and unconverted human DNA (genomic DNA purified from a human colorectal cancer cell line HCT116 DKO with double knockouts of both DNA methyltransferases (DNMT1 (-/-) and DNMT3b (-/-)) (K1 in Figure 1);
- -
- unmethylated and bisulfite-converted human DNA (genomic DNA originated from the same HCT116 DKO cell line as K1 DNA, but modified by sodium bisulfite upon isolation; as a result of bisulfite conversion non-methylated cytosines were turned to uracils) (K2 in Figure 1);
- -
- methylated and bisulfite-converted human DNA (genomic DNA derived from HCT116 DKO cell line which was in vitro methylated at all cytosine positions comprising CpG dinucleotides by M.SssI methyltransferase and then treated with sodium bisulfite; the final outcome of the bisulfite treatment was that 5-methylcytosines were left unaffected) (K3 in Figure 1).
2.4. Quantification of Methylation Data
2.5. Statistical Analysis
3. Results
3.1. Methylation Status of the MGMT Promoter and Clinical Parameters
3.2. Different Approaches in MSP Data Quantification
3.3. MGMT Status, TMZ Therapy, and Survival
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Louis, D.N.; Ohgaki, H.; Wiestler, O.D.; Cavenee, W.K.; Burger, P.C.; Jouvet, A.; Scheithauer, B.W.; Kleihues, P. The 2007 WHO Classification of Tumours of the Central Nervous System. Acta Neuropathol. 2007, 114, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed]
- Ostrom, Q.T.; Gittleman, H.; Truitt, G.; Boscia, A.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2011–2015. Neuro Oncol. 2018, 20, iv1–iv86. [Google Scholar] [CrossRef] [PubMed]
- Visser, O.; Ardanaz, E.; Botta, L.; Sant, M.; Tavilla, A.; Minicozzi, P.; Hackl, M.; Zielonke, N.; Oberaigner, W.; Van Eycken, E.; et al. Survival of adults with primary malignant brain tumours in Europe; Results of the EUROCARE-5 study. Eur. J. Cancer 2015, 51, 2231–2241. [Google Scholar] [CrossRef] [PubMed]
- Brennan, C.W.; Verhaak, R.G.W.; McKenna, A.; Campos, B.; Noushmehr, H.; Salama, S.R.; Zheng, S.; Chakravarty, D.; Sanborn, J.Z.; Berman, S.H.; et al. The Somatic Genomic Landscape of Glioblastoma. Cell 2013, 155, 462–477. [Google Scholar] [CrossRef] [PubMed]
- The Cancer Genome Atlas Research Network; McLendon, R.; Friedman, A.; Bigner, D.; Van Meir, E.G.; Brat, D.J.; Mastrogianakis, G.M.; Olson, J.J.; Mikkelsen, T.; Lehman, N.; et al. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 2008, 455, 1061. [Google Scholar] [CrossRef]
- Anvari, K.; Seilanian Toussi, M.; Ayatollahi, H.; Bahadorkhan, G.; Ghavam Nasiri, M.; Fazl Ersi, M. Prognostic Significance of MGMT Promoter Methylation in Patients with Glioblastoma Undergoing Surgical Intervention: A Retrospective Study in Northeastern Iran. Middle East J. Cancer 2018, 9, 179–185. [Google Scholar]
- Arora, I.; Gurav, M.; Rumde, R.; Dhanavade, S.; Kadam, V.; Kurani, H.; Shetty, O.; Goda, J.; Shetty, P.; Moiyadi, A.; et al. MGMT gene promoter methylation and its correlation with clinicopathological parameters in glioblastomas. Neurol. India 2018, 66, 1106–1114. [Google Scholar] [CrossRef]
- Miranda, A.; Blanco-Prieto, M.; Sousa, J.; Pais, A.; Vitorino, C. Breaching barriers in glioblastoma. Part I: Molecular pathways and novel treatment approaches. Int. J. Pharm. 2017, 531, 372–388. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, J.; Cheng, G.; Zhang, J.; Li, X. IDH mutation and MGMT promoter methylation are associated with the pseudoprogression and improved prognosis of glioblastoma multiforme patients who have undergone concurrent and adjuvant temozolomide-based chemoradiotherapy. Clin. Neurol. Neurosurg. 2016, 151, 31–36. [Google Scholar] [CrossRef]
- Pala, A.; Schmitz, A.L.; Knoll, A.; Schneider, M.; Hlavac, M.; König, R.; Wirtz, C.R.; Coburger, J. Is MGMT promoter methylation to be considered in the decision making for recurrent surgery in glioblastoma patients? Clin. Neurol. Neurosurg. 2018, 167, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Esteller, M.; Hamilton, S.R.; Burger, P.C.; Baylin, S.B.; Herman, J.G. Inactivation of the DNA Repair Gene O6-Methylguanine-DNA Methyltransferase by Promoter Hypermethylation is a Common Event in Primary Human Neoplasia. Cancer Res. 1999, 59, 793. [Google Scholar] [PubMed]
- Binabaj, M.M.; Bahrami, A.; ShahidSales, S.; Joodi, M.; Joudi Mashhad, M.; Hassanian, S.M.; Anvari, K.; Avan, A. The prognostic value of MGMT promoter methylation in glioblastoma: A meta-analysis of clinical trials. J. Cell. Physiol. 2018, 233, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Jesien-Lewandowicz, E.; Jesionek-Kupnicka, D.; Zawlik, I.; Szybka, M.; Kulczycka-Wojdala, D.; Rieske, P.; Sieruta, M.; Jaskolski, D.; Och, W.; Skowronski, W.; et al. High incidence of MGMT promoter methylation in primary glioblastomas without correlation with TP53 gene mutations. Cancer Genet. Cytogenet. 2009, 188, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y. Temozolomide resistance in glioblastoma multiforme. Genes Dis. 2016, 3, 198–210. [Google Scholar] [CrossRef] [PubMed]
- Woo, P.; Ho, J.; Lam, S.; Ma, E.; Chan, D.; Wong, W.-K.; Mak, C.; Lee, M.; Wong, S.-T.; Chan, K.-Y.; et al. A Comparative Analysis of the Usefulness of Survival Prediction Models for Patients with Glioblastoma in the Temozolomide Era: The Importance of Methylguanine Methyltransferase Promoter Methylation, Extent of Resection, and Subventricular Zone Location. World Neurosurg. 2018, 115, e375–e385. [Google Scholar] [CrossRef] [PubMed]
- Ilic, R.; Somma, T.; Savic, D.; Frio, F.; Milicevic, M.; Solari, D.; Nikitovic, M.; Lavrnic, S.; Raicevic, S.; Milosevic, S.; et al. A Survival Analysis with Identification of Prognostic Factors in a Series of 110 Patients with Newly Diagnosed Glioblastoma Before and After Introduction of the Stupp Regimen: A Single-Center Observational Study. World Neurosurg. 2017, 104, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Nikolov, V.; Stojanović, M.; Kostić, A.; Radisavljević, M.; Simonović, N.; Jelenković, B.; Berilazić, L. Factor affecting the survival of patients with glioblastoma multiforme. J. BUON 2018, 23, 173–178. [Google Scholar]
- Meng, W.; Jiang, Y.; Ma, J. Is the prognostic significance of O6-methylguanine- DNA methyltransferase promoter methylation equally important in glioblastomas of patients from different continents? A systematic review with meta-analysis. Cancer Manag. Res. 2017, 9, 411–425. [Google Scholar] [CrossRef]
- Tini, P.; Pastina, P.; Nardone, V.; Sebaste, L.; Toscano, M.; Miracco, C.; Cerase, A.; Pirtoli, L. The combined EGFR protein expression analysis refines the prognostic value of the MGMT promoter methylation status in glioblastoma. Clin. Neurol. Neurosurg. 2016, 149, 15–21. [Google Scholar] [CrossRef]
- De Carlo, E.; Gerratana, L.; De Maglio, G.; Buoro, V.; Cortiula, F.; Gurrieri, L.; Isola, M.; Fasola, G.; Puglisi, F.; Pizzolitto, S.; et al. Defining a prognostic score based on O6-methylguanine-DNA methyltransferase cut-off methylation level determined by pyrosequencing in patients with glioblastoma multiforme. J. Neurooncol. 2018. [Google Scholar] [CrossRef]
- Feng, E.; Sui, C.; Wang, T.; Sun, G. Temozolomide with or without Radiotherapy in Patients with Newly Diagnosed Glioblastoma Multiforme: A Meta-Analysis. Eur. Neurol. 2017, 77, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Du, X.L.; Lu, G.; Zhu, J.-J. Survival benefit of glioblastoma patients after FDA approval of temozolomide concomitant with radiation and bevacizumab: A population-based study. Oncotarget 2017, 8, 44015–44031. [Google Scholar] [CrossRef] [PubMed]
- Herman, J.G.; Graff, J.R.; Myöhänen, S.; Nelkin, B.D.; Baylin, S.B. Methylation-specific PCR: A novel PCR assay for methylation status of CpG islands. Proc. Natl. Acad. Sci. USA 1996, 93, 9821–9826. [Google Scholar] [CrossRef] [PubMed]
- Esteller, M.; Garcia-Foncillas, J.; Andion, E.; Goodman, S.N.; Hidalgo, O.F.; Vanaclocha, V.; Baylin, S.B.; Herman, J.G. Inactivation of the DNA-Repair Gene MGMT and the Clinical Response of Gliomas to Alkylating Agents. N. Engl. J. Med. 2000, 343, 1350–1354. [Google Scholar] [CrossRef] [PubMed]
- Christians, A.; Hartmann, C.; Benner, A.; Meyer, J.; von Deimling, A.; Weller, M.; Wick, W.; Weiler, M. Prognostic value of three different methods of MGMT promoter methylation analysis in a prospective trial on newly diagnosed glioblastoma. PLoS ONE 2012, 7, e33449. [Google Scholar] [CrossRef] [PubMed]
- Cankovic, M.; Nikiforova, M.N.; Snuderl, M.; Adesina, A.M.; Lindeman, N.; Wen, P.Y.; Lee, E.Q. The Role of MGMT Testing in Clinical Practice. JMD 2013, 15, 539–555. [Google Scholar] [CrossRef]
- Parrella, P.; la Torre, A.; Copetti, M.; Valori, V.M.; Barbano, R.; Notarangelo, A.; Bisceglia, M.; Gallo, A.P.; Balsamo, T.; Poeta, M.L.; et al. High specificity of quantitative methylation-specific PCR analysis for MGMT promoter hypermethylation detection in gliomas. J. Biomed. Biotechnol. 2009, 2009, 531692. [Google Scholar] [CrossRef]
- Image Processing and Analysis in Java Home page. Available online: https://imagej.nih.gov/ij/ (accessed on 30 May 2018).
- Aithal, M.G.S.; Rajeswari, N. Validation of housekeeping genes for gene expression analysis in glioblastoma using quantitative real-time polymerase chain reaction. Brain Tumor Res. Treat. 2015, 3, 24–29. [Google Scholar] [CrossRef]
- Rezvani, N.; Alibakhshi, R.; Vaisi-Raygani, A.; Bashiri, H.; Saidijam, M. Detection of SPG20 gene promoter-methylated DNA, as a novel epigenetic biomarker, in plasma for colorectal cancer diagnosis using the MethyLight method. Oncol. Lett. 2017, 13, 3277–3284. [Google Scholar] [CrossRef] [PubMed]
- Håvik, A.B.; Brandal, P.; Honne, H.; Dahlback, H.-S.S.; Scheie, D.; Hektoen, M.; Meling, T.R.; Helseth, E.; Heim, S.; Lothe, R.A.; et al. MGMT promoter methylation in gliomas-assessment by pyrosequencing and quantitative methylation-specific PCR. J. Transl. Med. 2012, 10, 36. [Google Scholar] [CrossRef] [PubMed]
- Dullea, A.; Marignol, L. MGMT testing allows for personalised therapy in the temozolomide era. Tumor Biol. 2016, 37, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Hegi, M.E.; Mason, W.P.; van den Bent, M.J.; Taphoorn, M.J.B.; Janzer, R.C.; Ludwin, S.K.; Allgeier, A.; Fisher, B.; Belanger, K.; et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009, 10, 459–466. [Google Scholar] [CrossRef]
- Kalkan, R.; Atli, E.İ.; Özdemir, M.; Çiftçi, E.; Aydin, H.E.; Artan, S.; Arslantaş, A. IDH1 mutations is prognostic marker for primary glioblastoma multiforme but MGMT hypermethylation is not prognostic for primary glioblastoma multiforme. Gene 2015, 554, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Parker, N.R.; Khong, P.; Parkinson, J.F.; Howell, V.M.; Wheeler, H.R. Molecular heterogeneity in glioblastoma: Potential clinical implications. Front. Oncol. 2015, 5, 55. [Google Scholar] [CrossRef] [PubMed]
- Wiencke, J.K; Aldape, K.; McMillan, A.; Wiemels, J.; Moghadassi, M.; Miike, R.; Kelsey, K.T.; Patoka, J.; Long, J.; Wrensch, M. Molecular Features of Adult Glioma Associated with Patient Race/Ethnicity, Age, and a Polymorphism in O6-Methylguanine-DNA-Methyltransferase. Cancer Epidemiol. Biomark. Prev. 2005, 14, 1774–1783. [Google Scholar] [CrossRef]
- Zou, P.; Xu, H.; Chen, P.; Yan, Q.; Zhao, L.; Zhao, P.; Gu, A. IDH1/IDH2 mutations define the prognosis and molecular profiles of patients with gliomas: A meta-analysis. PLoS ONE 2013, 8, e68782. [Google Scholar] [CrossRef]
- Ohgaki, H.; Kleihues, P. Genetic pathways to primary and secondary glioblastoma. Am. J. Pathol. 2007, 170, 1445–1453. [Google Scholar] [CrossRef]
- Shamsara, J.; Sharif, S.; Afsharnezhad, S.; Lotfi, M.; Raziee, H.R.; Ghaffarzadegan, K.; Moradi, A.; Rahighi, S.; Behravan, J. Association Between MGMT Promoter Hypermethylation and p53 Mutation in Glioblastoma. Cancer Invest. 2009, 27, 825–829. [Google Scholar] [CrossRef]
- Wang, K.; Wang, Y.; Ma, J.; Wang, J.; Li, S.; Jiang, T.; Dai, J. Prognostic value of MGMT promoter methylation and TP53 mutation in glioblastomas depends on IDH1 mutation. Asian Pac. J. Cancer Prev. 2014, 15, 10893–10898. [Google Scholar] [CrossRef] [PubMed]


| Type of Therapy | Patient Mark |
|---|---|
| RT + TMZ (Group 1) | 1, 2, 5, 8, 9, 15, 16, 17, 19 and 25 |
| RT + PCV (Group 2) | 4, 7, 10, 12, 18, 22, 23 and 24 |
| RT + BCNU (Group 3) | 3, 6, 11, 13, 14, 20 and 21 |
| Gene | Primer Sequence (5’–3’) | Amplicon Size (bp) | References |
|---|---|---|---|
| MGMT unmethylated (U) | F: TTTGTGTTTTGATGTTTGTAGGTTTTTGT | 93 | [26] |
| R: AACTCCACACTCTTCCAAAAACAAAACA | |||
| MGMT methylated (M) | F: TTTCGACGTTCGTAGGTTTTCGC | 81 | [26] |
| R: GCACTCTTCCGAAAACGAAACG | |||
| ALU–C4 | F: GGTTAGGTATAGTGGTTTATATTTGTAATTTTAGTA | 98 | [31] |
| R: ATTAACTAAACTAATCTTAAACTCCTAACCTCA |
| Unmethylated (n = 13) | Methylated (n = 12) | ||
|---|---|---|---|
| Sex, n (%) | male | 9 (69.2) | 10 (83.3) |
| female | 4 (30.8) | 2 (16.7) | |
| Age, mean ± SD, years | 58.00 ± 12.85 | 61.33 ± 13.65 | |
| Age, n (%) | <50 yr | 4 (30.8) | 2 (16.7) |
| >50 yr | 9 (69.2) | 10 (83.3) | |
| Preoperative KPS, (%) | 81.64 ± 12.01 | 81.64 ± 12.01 | |
| Postoperative KPS, (%) | 80.00 ± 12.06 | 80.00 ± 12.06 | |
| PMR | (I) | (II) | (III) |
|---|---|---|---|
| <1, n (%) | 5 (20.0) | 6 (24.0) | 5 (20.0) |
| >1, n (%) | 7 (28.0) | 6 (24.0) | 7 (28.0) |
| 0, n (%) | 13 (52.0) | 13 (52.0) | 13 (52.0) |
| ICC | 95% CI | p | |
|---|---|---|---|
| (I) vs. (II) | 0.763 | 0.532–0.888 | <0.001 |
| (I) vs. (III) | 0.493 | 0.139–0.739 | 0.005 |
| (II) vs. (III) | 0.844 | 0.678–0.928 | <0.001 |
| SE | 95% CI | Log-Rank | p | |||
|---|---|---|---|---|---|---|
| Lower Limit | Upper Limit | |||||
| No TMZ | 5.000 | 0.781 | 3.469 | 6.531 | 15.721 | <0.001 |
| TMZ | 15.000 | 1.799 | 11.473 | 18.527 | ||
| Male | 8.167 | 1.462 | 5.300 | 11.033 | 0.221 | 0.638 |
| Female | 7.167 | 2.151 | 2.950 | 11.383 | ||
| Unmethylated MGMT | 7.083 | 1.574 | 3.999 | 10.168 | 0.727 | 0.394 |
| Methylated MGMT | 8.750 | 1.855 | 5.114 | 12.386 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jovanović, N.; Mitrović, T.; Cvetković, V.J.; Tošić, S.; Vitorović, J.; Stamenković, S.; Nikolov, V.; Kostić, A.; Vidović, N.; Krstić, M.; et al. The Impact of MGMT Promoter Methylation and Temozolomide Treatment in Serbian Patients with Primary Glioblastoma. Medicina 2019, 55, 34. https://doi.org/10.3390/medicina55020034
Jovanović N, Mitrović T, Cvetković VJ, Tošić S, Vitorović J, Stamenković S, Nikolov V, Kostić A, Vidović N, Krstić M, et al. The Impact of MGMT Promoter Methylation and Temozolomide Treatment in Serbian Patients with Primary Glioblastoma. Medicina. 2019; 55(2):34. https://doi.org/10.3390/medicina55020034
Chicago/Turabian StyleJovanović, Nikola, Tatjana Mitrović, Vladimir J. Cvetković, Svetlana Tošić, Jelena Vitorović, Slaviša Stamenković, Vesna Nikolov, Aleksandar Kostić, Nataša Vidović, Miljan Krstić, and et al. 2019. "The Impact of MGMT Promoter Methylation and Temozolomide Treatment in Serbian Patients with Primary Glioblastoma" Medicina 55, no. 2: 34. https://doi.org/10.3390/medicina55020034
APA StyleJovanović, N., Mitrović, T., Cvetković, V. J., Tošić, S., Vitorović, J., Stamenković, S., Nikolov, V., Kostić, A., Vidović, N., Krstić, M., Jevtović-Stoimenov, T., & Pavlović, D. (2019). The Impact of MGMT Promoter Methylation and Temozolomide Treatment in Serbian Patients with Primary Glioblastoma. Medicina, 55(2), 34. https://doi.org/10.3390/medicina55020034





