Effects of Cariprazine, Aripiprazole, and Olanzapine on Mouse Fibroblast Culture: Changes in Adiponectin Contents in Supernatants, Triglyceride Accumulation, and Peroxisome Proliferator-Activated Receptor-γ Expression
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Triglyceride Content Determination of Adipocytes
2.3. Adiponectin Concentration of Cell Culture Supernatant
2.4. Peroxisome Proliferator-Activated Receptor-γ Expression
2.5. Statistical Analysis
3. Results
3.1. Triglyceride Content of Cells
3.2. Adiponectin Contents in Cell Culture Supernatants
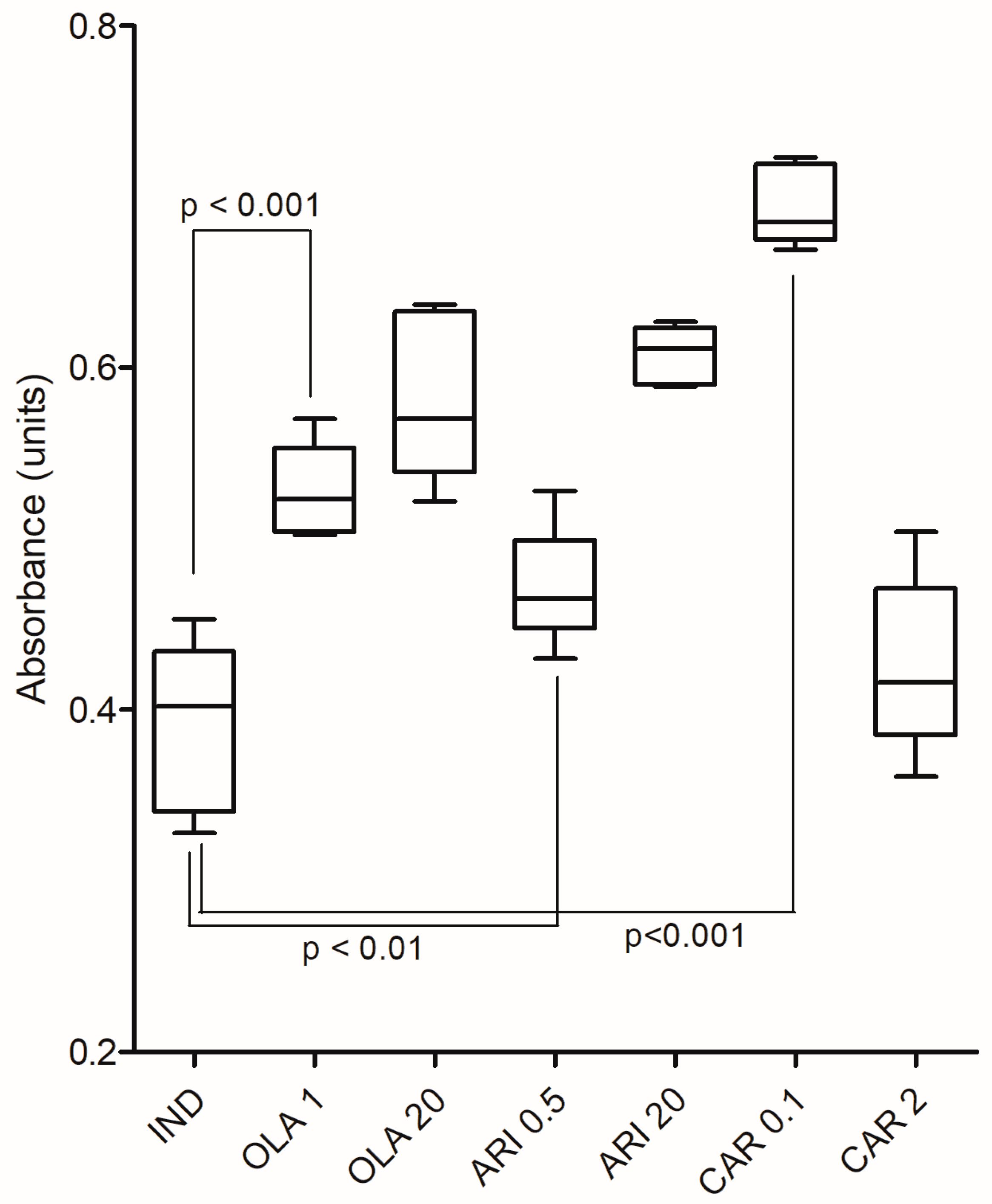
3.3. Peroxisome Proliferator-Activated Receptor-γ Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Correll, C.U.; Leucht, S.; Kane, J.M. Lower risk for tardive dyskinesia associated with second-generation antipsychotics: A systematic review of 1-year studies. Am. J. Psychiatry 2004, 161, 414–425. [Google Scholar] [CrossRef]
- Luft, B.; Taylor, D. A review of atypical antipsychotic drugs versus conventional medication in schizophrenia. Expert. Opin. Pharmacother. 2006, 7, 1739–1748. [Google Scholar] [CrossRef]
- Tandon, R.; Jibson, M.D. Efficacy of newer generation antipsychotics in the treatment of schizophrenia. Psychoneuroendocrinology 2003, 1, 9–26. [Google Scholar] [CrossRef]
- Meltzer, H.Y. What’s atypical about atypical antipsychotic drugs? Curr. Opin. Pharmacol. 2004, 4, 53–57. [Google Scholar] [CrossRef]
- Sernyak, M.J.; Leslie, D.L.; Alarcon, R.D.; Losonczy, M.F.; Rosenheck, R. Association of diabetes mellitus with use of atypical neuroleptics in the treatment of schizophrenia. Am. J. Psychiatry 2002, 159, 561–566. [Google Scholar] [CrossRef]
- Muench, J.; Hamer, A.M. Adverse effects of antipsychotic medications. Am. Fam. Phys. 2010, 81, 617–622. [Google Scholar]
- Newcomer, J.W. Second-generation (atypical) antipsychotics and metabolic effects: A comprehensive literature review. CNS Drugs 2005, 19, 1–93. [Google Scholar] [CrossRef]
- Nasrallah, H. A review of the effect of atypical antipsychotics on weight. Psychoneuroendocrinology 2003, 28, 83–96. [Google Scholar] [CrossRef]
- Nihalani, N.; Schwartz, T.L.; Siddiqui, U.A.; Megna, J.L. Weight gain, obesity, and psychotropic prescribing. J. Obes. 2011, 2011, 1–9. [Google Scholar] [CrossRef]
- Taylor, D.M.; McAskill, R. Atypical antipsychotics and weight gain: A systematic review. Acta. Psychiatr. Scand. 2000, 101, 416–432. [Google Scholar] [CrossRef]
- Ho, C.; Zhang, M.; Mak, A.; Ho, R. Metabolic syndrome in psychiatry: Advances in understanding and management. Adv. Psychiatr. Treat. 2014, 20, 101–112. [Google Scholar] [CrossRef]
- Coccurello, R.; Moles, A. Potential mechanisms of atypical antipsychotic-induced metabolic derangement: Clues for understanding obesity and novel drug design. Pharmacol. Ther. 2010, 127, 210–251. [Google Scholar] [CrossRef]
- Atkin, T.; Comai, S.; Gobbi, G. Drugs for Insomnia beyond Benzodiazepines: Pharmacology, Clinical Applications, and Discovery. Pharmacol Rev. 2018, 70, 197–245. [Google Scholar] [CrossRef]
- Kroeze, W.K.; Hufeisen, S.J.; Popadak, B.A.; Renock, S.M.; Steinberg, S.; Ernsberger, P.; Jayathilake, K.; Meltzer, H.; Roth, B. H1-Histamine Receptor Affinity Predicts Short-Term Weight Gain for Typical and Atypical Antipsychotic Drugs. Neuropsychopharmacology 2003, 28, 519–526. [Google Scholar] [CrossRef] [Green Version]
- Roering, J.; Steffen, K.; Mitchell, J. Atypical antipsychotic-induced weight gain: Insights into mechanisms of action. CNS Drugs 2011, 25, 1035–1059. [Google Scholar] [CrossRef]
- Citrome, L. The ABC’s of dopamine receptor partial agonists—Aripiprazole, brexpiprazole and cariprazine: The 15-min challenge to sort these agents out. Int. J. Clin. Pract. 2015, 69, 1211–1220. [Google Scholar] [CrossRef]
- Kershaw, E.E.; Flier, J.S. Adipose tissue as an endocrine organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556. [Google Scholar] [CrossRef]
- Lafontan, M.; Viguerie, N. Role of adipokines in the control of energy metabolism: Focus on adiponectin. Curr. Opin. Pharmacol. 2006, 6, 580–585. [Google Scholar] [CrossRef]
- Pischon, T.; Girman, C.J.; Rifai, N.; Hotamisligil, G.S.; Rimm, E.B. Association between dietary factors and plasma adiponectin concentrations in men. Am. J. Clin. Nutr. 2005, 81, 780–786. [Google Scholar] [CrossRef]
- Kadowaki, T.; Yamauchi, T.; Kubota, N.; Hara, K.; Ueki, K.; Tobe, K. Adiponectin and adiponectin receptors in insulin resistance, diabetes and the metabolic syndrome. J. Clin. Investig. 2006, 116, 1784–1792. [Google Scholar] [CrossRef]
- Fruebis, J.; Tsao, T.S.; Javorschi, S.; Ebbets-Reed, D.; Erickson, M.R.; Yen, F.T.; Bihain, B.E.; Lodish, H.F. Proteolytic cleavage product of 30-kDa adipocyte complement-related protein increases fatty acid oxidation in muscle and causes weight loss in mice. Proc. Natl. Acad. Sci. USA 2001, 98, 2005–2010. [Google Scholar] [CrossRef] [Green Version]
- Yamauchi, T.; Kamon, J.; Waki, H.; Terauchi, Y.; Kubota, N.; Hara, K.; Mori, Y.; Ide, T.; Murakami, K.; Tsuboyama-Kasaoka, N.; et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat. Med. 2001, 7, 941–946. [Google Scholar] [CrossRef]
- Berg, A.H.; Combs, T.P.; Du, X.; Brownlee, M.; Scherer, P.E. The adipocyte-secreted protein Acrp30 enhances hepatic insulin action. Nat. Med. 2001, 7, 947–953. [Google Scholar] [CrossRef]
- Mandard, S.; Muller, M.; Kersten, S. Peroxisome proliferator-activated receptor alpha target genes. Cell. Mol. Life. Sci. 2004, 61, 393–416. [Google Scholar] [CrossRef]
- Moseti, D.; Regassa, A.; Kim, W.K. Molecular regulation of adipogenesis and potential anti-adipogenic bioactive molecules. Int. J. Mol. Sci. 2016, 17, 124. [Google Scholar] [CrossRef]
- Nawrocki, A.R.; Rajala, M.W.; Tomas, E.; Pajvani, U.B.; Saha, A.K.; Trumbauer, M.E.; Pang, Z.; Chen, A.S.; Ruderman, N.B.; Chen, H.; et al. Mice lacking adiponectin show decreased hepatic insulin sensitivity and reduced responsiveness to peroxisome proliferator-activated receptor γ agonists. J. Biol. Chem. 2006, 281, 2654–2660. [Google Scholar] [CrossRef]
- Yang, L.H.; Chen, T.M.; Yu, S.T.; Chen, Y.H. Olanzapine induces SREBP-1-related adipogenesis in 3T3-L1 cells. Pharmacol. Res. 2007, 56, 202–208. [Google Scholar] [CrossRef]
- Cai, H.L.; Tan, Q.Y.; Jiang, P.; Dang, R.L.; Xue, Y.; Tang, M.M.; Xu, P.P.; Deng, R.L.; Li, H.D.; Yao, J.K. A potential mechanism underlying atypical antipsychotics-induced lipid disturbances. Transl. Psychiatry 2015, 5, e661. [Google Scholar] [CrossRef]
- Liu, X.; Wu, Z.; Lian, J.; Hu, C.H.; Huang, X.F.; Deng, C. Time-dependent changes and potential mechanisms of glucose-lipid metabolic disorders associated with chronic clozapine or olanzapine treatment in rats. Sci. Rep. 2017, 7, 2762. [Google Scholar] [CrossRef] [Green Version]
- Fajas, L.; Schoonjans, K.; Gelman, L.; Kim, J.B.; Najib, J.; Martin, G.; Fruchart, J.C.; Briggs, M.; Spiegelman, B.M.; Auwerx, J. Regulation of peroxisome proliferator-activated receptor gamma expression by adipocyte differentiation and determination factor 1/sterol regulatory element binding protein 1: Implications for adipocyte differentiation and metabolism. Mol. Cell. Biol. 1999, 19, 5495–5503. [Google Scholar] [CrossRef]
- Sárvári, A.K.; Veréb, Z.; Uray, I.P.; Fesüs, L.; Balajthy, Z. Atypical antipsychotics induce both proinflammatory and adipogenic gene expression in human adipocytes in vitro. Biochem. Biophys. Res. Commun. 2014, 450, 1383–1389. [Google Scholar] [CrossRef]
- Nagy, A.; Gertsenstein, M.; Vintersten, K.; Behringer, R. Preparing Feeder Cell Layers from STO or Mouse Embryo Fibroblast (MEF) Cells: Treatment with γ-Irradiation. CSH Protoc. 2006, 1. [Google Scholar] [CrossRef]
- Kirschbaum, K.M.; Müller, M.J.; Malevani, J.; Mobascher, A.; Burchardt, C.; Piel, M.; Hiemke, C. Serum levels of aripiprazole and dehydroaripiprazole, clinical response and side effects. World J. Biol. Psychiatry 2008, 9, 212–218. [Google Scholar] [CrossRef]
- Lu, M.L.; Wu, Y.X.; Chen, C.H.; Kuo, P.T.; Chen, Y.H.; Lin, C.H.; Wu, T.H. Application of Plasma Levels of Olanzapine and n-Desmethyl-Olanzapine to Monitor Clinical Efficacy in Patients with Schizophrenia. PLoS ONE 2016, 11, e0148539. [Google Scholar] [CrossRef]
- Nakamura, T.; Kubota, T.; Iwakaji, A.; Imada, M.; Kapás, M.; Morio, Y. Clinical pharmacology study of cariprazine (MP-214) in patients with schizophrenia (12-week treatment). Drug. Des. Devel. Ther. 2016, 10, 327–338. [Google Scholar] [CrossRef] [Green Version]
- Ramirez-Zacarias, J.L.; Castro-Munozledo, F.; Kuri-Harcuch, W. Quantitation of adipose conversion and triglycerides by staining intracytoplasmic lipids with Oil red O. Histochemistry 1992, 97, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Minet-Ringuet, J.; Even, P.C.; Valet, P.; Carpéné, C.; Visentin, V.; Prévot, D.; Daviaud, D.; Quignard-Boulange, A.; Tomé, D.; de Beaurepaire, R. Alterations of lipid metabolism and gene expression in rat adipocytes during chronic olanzapine treatment. Mol. Psychiatry 2007, 12, 562–571. [Google Scholar] [CrossRef] [Green Version]
- Citrome, L. Cariprazine: Chemistry, pharmacodynamics, pharmacokinetics, and metabolism, clinical efficacy, safety, and tolerability. Expert. Opin. Drug. Metab. Toxicol. 2013, 9, 193–206. [Google Scholar] [CrossRef]
- Gáll, Z.; Vancea, S.; Mezei, T.; Kolcsár, M. Adipocyte triglyceride content and adipogenesis in aripiprazole treated rats. Int. J. Pharmacol. 2013, 9, 251–257. [Google Scholar]
- Vestri, H.S.; Maianu, L.; Moellering, D.R.; Garvey, W.T. Atypical antipsychotic drugs directly impair insulin action in adipocytes: Effects on glucose transport, lipogenesis, and antilipolysis. Neuropsychopharmacology 2007, 32, 765–772. [Google Scholar] [CrossRef]
- Martella, E.; Bellotti, C.; Dozza, B.; Perrone, S.; Donati, D.; Lucarelli, E. Secreted adiponectin as a marker to evaluate in vitro the adipogenic differentiation of human mesenchymal stromal cells. Cytotherapy 2014, 16, 1476–1485. [Google Scholar] [CrossRef] [PubMed]
- Asterholm, I.W.; Scherer, P.E. Enhanced metabolic flexibility associated with elevated adiponectin levels. Am. J. Pathol. 2010, 176, 1364–1376. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, M.; Kacirova, I.; Urinovska, R. Therapeutic drug monitoring of atypical antipsychotic drugs. Acta. Pharm. 2014, 64, 387–401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, D.; Wang, Z.; Dou, X.; Zhang, X.; Li, S.; Vu, L.; Yao, T.; Song, Z. Inhibition of ERK1/2 pathway suppresses adiponectin secretion via accelerating protein degradation by Ubiquitin-proteasome system: Relevance to obesity-related adiponectin decline. Metabolism 2013, 62, 1137–1148. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Ojeda, J.F.; Rupérez, A.I.; Gomez-Llorente, C.; Gil, A.; Aguilera, C.M. Cell models and their application for studying adipogenic differentiation in relation to obesity: A review. Int. J. Mol. Sci. 2016, 17, 1040. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bába, L.-I.; Kolcsár, M.; Kun, I.Z.; Ulakcsai, Z.; Bagaméry, F.; Szökő, É.; Tábi, T.; Gáll, Z. Effects of Cariprazine, Aripiprazole, and Olanzapine on Mouse Fibroblast Culture: Changes in Adiponectin Contents in Supernatants, Triglyceride Accumulation, and Peroxisome Proliferator-Activated Receptor-γ Expression. Medicina 2019, 55, 160. https://doi.org/10.3390/medicina55050160
Bába L-I, Kolcsár M, Kun IZ, Ulakcsai Z, Bagaméry F, Szökő É, Tábi T, Gáll Z. Effects of Cariprazine, Aripiprazole, and Olanzapine on Mouse Fibroblast Culture: Changes in Adiponectin Contents in Supernatants, Triglyceride Accumulation, and Peroxisome Proliferator-Activated Receptor-γ Expression. Medicina. 2019; 55(5):160. https://doi.org/10.3390/medicina55050160
Chicago/Turabian StyleBába, László-István, Melinda Kolcsár, Imre Zoltán Kun, Zsófia Ulakcsai, Fruzsina Bagaméry, Éva Szökő, Tamás Tábi, and Zsolt Gáll. 2019. "Effects of Cariprazine, Aripiprazole, and Olanzapine on Mouse Fibroblast Culture: Changes in Adiponectin Contents in Supernatants, Triglyceride Accumulation, and Peroxisome Proliferator-Activated Receptor-γ Expression" Medicina 55, no. 5: 160. https://doi.org/10.3390/medicina55050160




