Cerebrospinal Fluid Analysis in Multiple Sclerosis Diagnosis: An Update
Abstract
1. Introduction
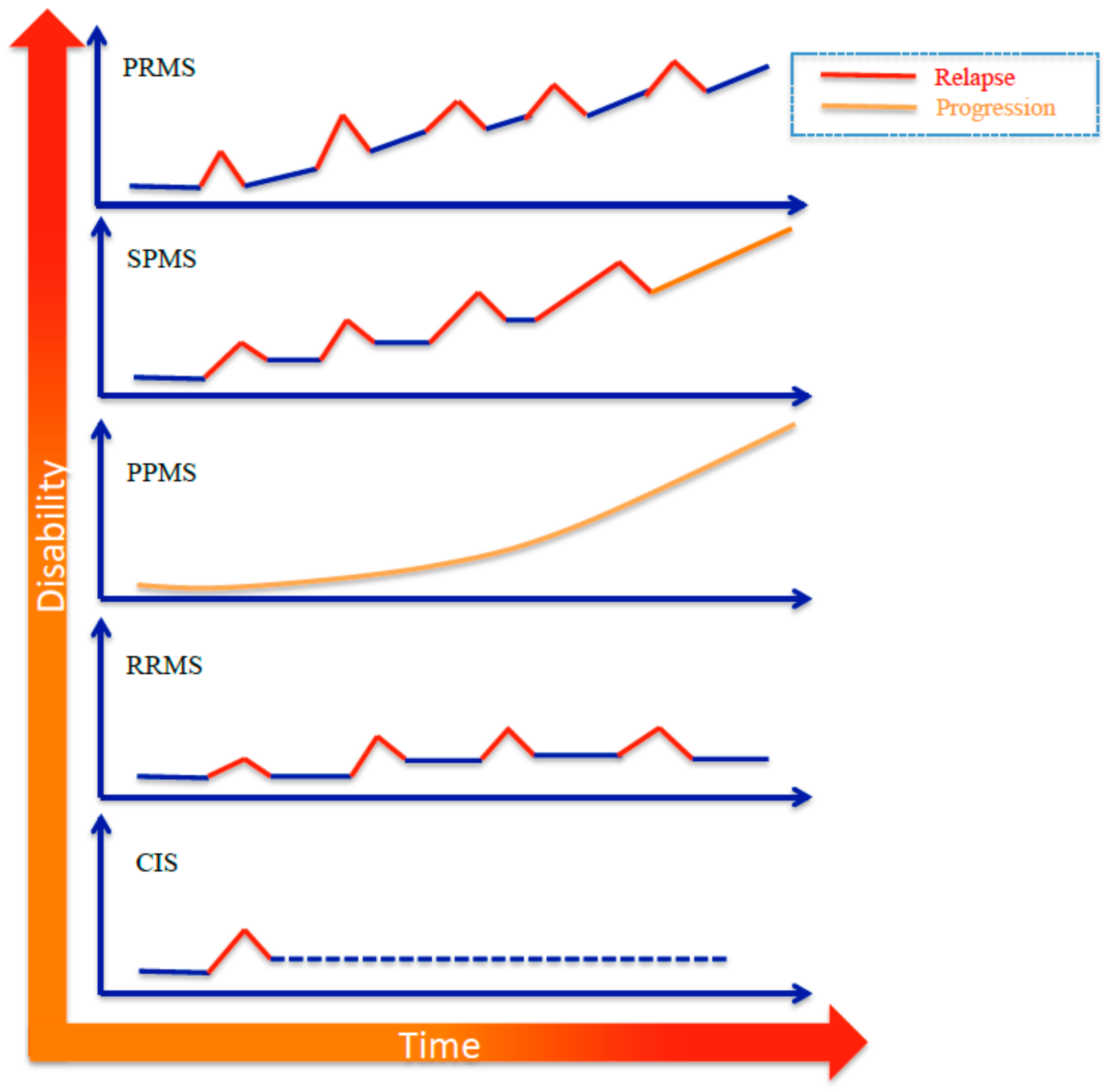
2. Etiopathogenesis
3. Diagnostic Criteria
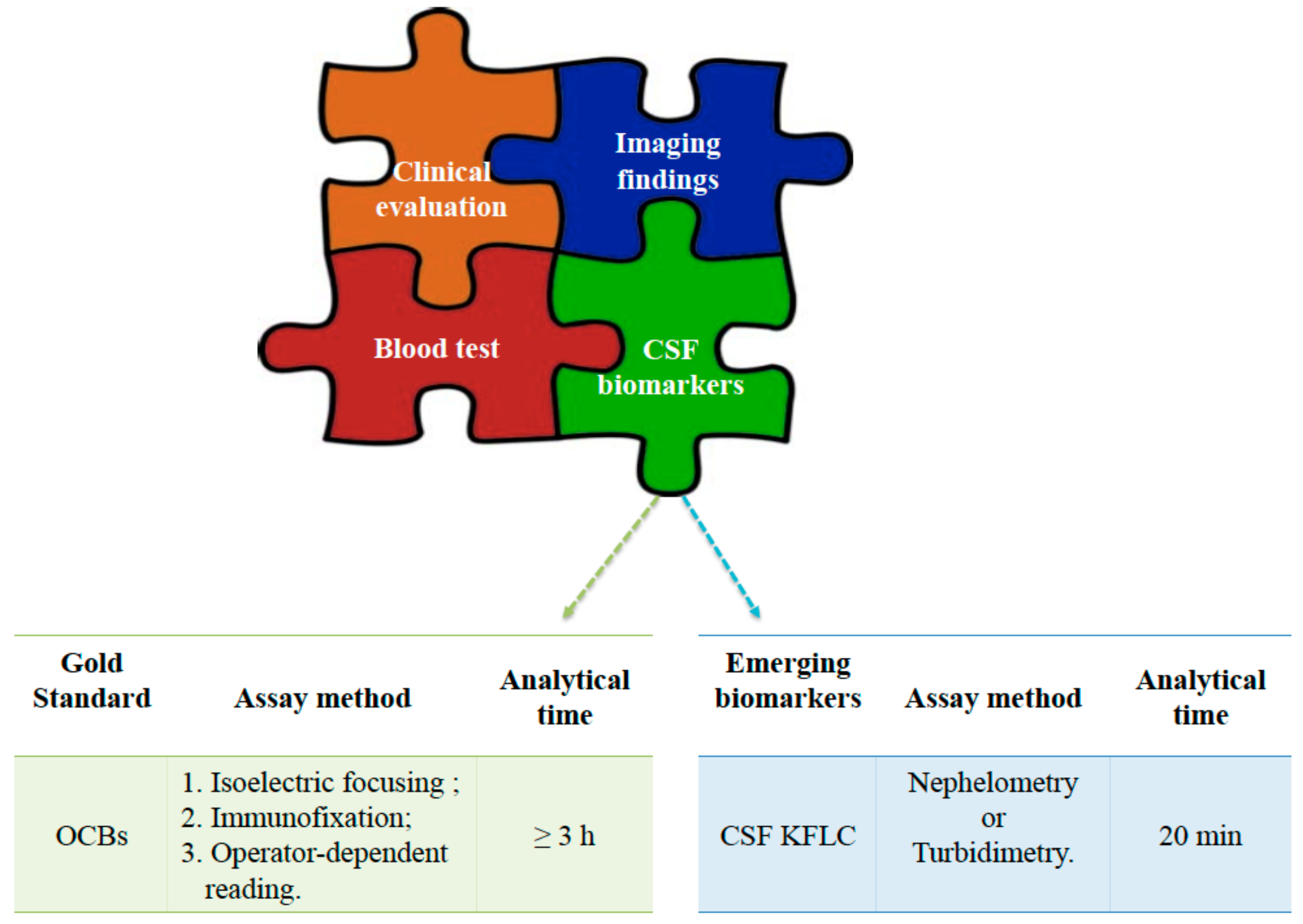
4. CSF Diagnostics
5. Conclusions
Funding
Conflicts of Interest
References
- Baranzini, S.E.; Oksenberg, J.R. The genetics of multiple sclerosis: From 0 to 200 in 50 years. Trends Genet. 2017, 33, 960–970. [Google Scholar] [CrossRef] [PubMed]
- Mallucci, G.; Peruzzotti-Jametti, L.; Bernstock, J.D.; Pluchino, S. The role of immune cells, glia and neurons in white and gray matter pathology in multiple sclerosis. Prog Neurobiol. 2015, 127, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Kingwell, E.; Marriott, J.J.; Jetté, N.; Pringsheim, T.; Makhani, N.; Morrow, SA.; Fisk, J.D.; Evans, C.; Béland, S.G.; Kulaga, S.; et al. Incidence and prevalence of multiple sclerosis in Europe: A systematic review. BMC Neurol. 2013, 13, 128. [Google Scholar] [CrossRef] [PubMed]
- Heydarpour, P.; Khoshkish, S.; Abtahi, S.; Moradi-Lakeh, M.; Sahraian, M.A. Multiple Sclerosis Epidemiology in Middle East and North Africa: A Systematic Review and Meta-Analysis. Neuroepidemiology 2015, 44, 232–244. [Google Scholar] [CrossRef]
- Ahlgren, C.; Odén, A.; Lycke, J. High nationwide incidence of multiple sclerosis in Sweden. PLoS ONE 2014, 9, e108599. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Olsson, T.; Alfredsson, L. Age at Menarche and Risk of Multiple Sclerosis: Current Progress from Epidemiological Investigations. Front. Immunol. 2018, 9, 2600. [Google Scholar] [CrossRef] [PubMed]
- Pirttisalo, A.L.; Soilu-Hänninen, M.; Sipilä, J.O.T. Multiple sclerosis epidemiology in Finland: Regional differences and high incidence. Acta Neurol. Scand. 2018, 139, 353–359. [Google Scholar] [CrossRef]
- Agnello, L.; Scazzone, C.; Lo Sasso, B.; Bellia, C.; Bivona, G.; Realmuto, S.; Brighina, F.; Schillaci, R.; Ragonese, P.; Salemi, G.; et al. VDBP, CYP27B1, and 25-Hydroxyvitamin D gene polymorphism analyses in a group of Sicilian multiple sclerosis patients. Biochem. Genet. 2017, 55, 183–192. [Google Scholar] [CrossRef]
- Bivona, G.; Agnello, L.; Ciaccio, M. Vitamin D and Immunomodulation: Is It Time to Change the Reference Values? Ann. Clin. Lab. Sci. 2017, 47, 508–510. [Google Scholar]
- Bivona, G.; Agnello, L.; Pivetti, A.; Milano, S.; Scazzone, C.; Lo Sasso, B.; Ciaccio, M. Association between hypovitaminosis D and systemic sclerosis: True or fake? Clin. Chim. Acta 2016, 458, 115–119. [Google Scholar] [CrossRef]
- Agnello, L.; Scazzone, C.; Ragonese, P.; Salemi, G.; Lo Sasso, B.; Schillaci, R.; Musso, G.; Bellia, C.; Ciaccio, M. Vitamin D receptor polymorphisms and 25-hydroxyvitamin D in a group of Sicilian multiple sclerosis patients. Neurol. Sci. 2016, 37, 261–267. [Google Scholar] [CrossRef]
- Agnello, L.; Scazzone, C.; Ragonese, P.; Salemi, G.; Lo Sasso, B.; Schillaci, R.; Musso, G.; Bellia, C.; Ciaccio, M. A new role of CYP2R1 in multiple sclerosis. Biochim. Clin. 2018, 42, 294–299. [Google Scholar] [CrossRef]
- Agnello, L.; Scazzone, C.; Lo Sasso, B.; Ragonese, P.; Milano, S.; Salemi, G.; Ciaccio, M. CYP27A1, CYP24A1, and RXR-α Polymorphisms, Vitamin D, and Multiple Sclerosis: A Pilot Study. J. Mol. Neurosci. 2018, 66, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Presslauer, S.; Milosavljevic, D.; Brücke, T.; Bayer, P.; Hübl, W. Elevated levels of kappa free light chains in CSF support the diagnosis of multiple sclerosis. J. Neurol. 2008, 255, 1508–1514. [Google Scholar] [CrossRef] [PubMed]
- Bernardi, G.; Cataldo, I. Quantitation of immunoglobulin free light chains in cerebrospinal fluid. Biochim. Clin. 2013, 37, 389–394. [Google Scholar]
- Napodano, C.; Pocino, K.; Rigante, D.; Stefanile, A.; Gulli, F.; Marino, M.; Basile, V.; Rapaccini, G.L.; Basile, U. Free light chains and autoimmunity. Autoimmun. Rev. 2019. [Google Scholar] [CrossRef] [PubMed]
- Basnayake, K.; Stringer, S.J.; Hutchison, C.A.; Cockwell, P. The biology of immunoglobulin free light chains and kidney injury. Kidney Int. 2011, 79, 1289–1301. [Google Scholar] [CrossRef]
- Basile, U.; Gulli, F.; Gragnani, L.; Napodano, C.; Pocino, K.; Rapaccini, G.L.; Mussap, M.; Zignego, A.L. Free light chains: Eclectic multipurpose biomarker. J. Immunol. Methods 2017, 451, 11–19. [Google Scholar] [CrossRef]
- Dutta, R.; Trapp, B.D. Pathogenesis of axonal and neuronal damage in multiple sclerosis. Neurology 2007, 68 (Suppl. 3), S22–S31. [Google Scholar] [CrossRef]
- Puthenparampil, M.; Federle, L.; Miante, S.; Zito, A.; Toffanin, E.; Ruggero, S.; Ermani, M.; Pravato, S.; Poggiali, D.; Perini, P.; et al. BAFF Index and CXCL13 levels in the cerebrospinal fluid associate respectively with intrathecal IgG synthesis and cortical atrophy in multiple sclerosis at clinical onset. J. Neuroinflamm. 2017, 14, 11. [Google Scholar] [CrossRef][Green Version]
- Lisak, R.P.; Benjamins, J.A.; Nedelkoska, L.; Barger, J.L.; Ragheb, S.; Fan, B.; Ouamara, N.; Johnson, T.A.; Rajasekharan, S.; Bar-Or, A. Secretory products of multiple sclerosis B cells are cytotoxic to oligodendroglia in vitro. J. Neuroimmunol. 2012, 246, 85–95. [Google Scholar] [CrossRef]
- Myhr, K.M.; Torkildsen, Ø.; Lossius, A.; Bø, L.; Holmøy, T. B cell depletion in the treatment of multiple sclerosis. Expert Opin Biol Ther. 2019, 19, 261–271. [Google Scholar] [CrossRef]
- Polman, C.H.; Reingold, S.C.; Edan, G.; Filippi, M.; Hartung, HP.; Kappos, L.; Lublin, F.D.; Metz, L.M.; McFarland, H.F.; O’Connor, P.W.; et al. Diagnostic Criteria for Multiple Sclerosis: 2005 Revisions to the “McDonald Criteria”. Ann. Neurol. 2005, 58, 840–846. [Google Scholar] [CrossRef]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic Criteria for Multiple Sclerosis: 2010 Revisions to the McDonald Criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef]
- Thompson, A.J.; Banwell, B.L.; Barkhof, F.; Carroll, W.M.; Coetzee, T.; Comi, G.; Correale, J.; Fazekas, F.; Filippi, M.; Freedman, M.S.; et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018, 17, 162–173. [Google Scholar] [CrossRef]
- Olsson, B.; Lautner, R.; Andreasson, U.; Öhrfelt, A.; Portelius, E.; Bjerke, M.; Hölttä, M.; Rosén, C.; Olsson, C.; Strobel, G.; et al. CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: A systematic review and meta-analysis. Lancet Neurol. 2016, 15, 673–684. [Google Scholar] [CrossRef]
- Agnello, L.; Bivona, G.; Novo, G.; Scazzone, C.; Muratore, M.; Levantino, P.; Bellia, C.; Lo Sasso, B.; Ciaccio, M. Heart-type fatty acid binding protein is a sensitive biomarker for early AMI detection in troponin negative patients: A pilot study. Scand. J. Clin. Lab. Investig. 2017, 77, 428–432. [Google Scholar] [CrossRef]
- Reiber, H.; Felgenhauer, K. Protein transfer at the blood cerebrospinal fluid barrier and the quantitation of the humoral immune response within the central nervous system. Clin. Chim. Acta 1987, 163, 319–328. [Google Scholar] [CrossRef]
- Fischer, C.; Arneth, B.; Koehler, J.; Lotz, J.; Lackner, K.J. Kappa free light chains in cerebrospinal fluid as markers of intrathecal immunoglobulin synthesis. Clin. Chem. 2004, 50, 1809–1813. [Google Scholar] [CrossRef]
- Reiber, H.; Peter, J.B. Cerebrospinal fluid analysis: Disease-related data patterns and evaluation programs. J. Neurol. Sci. 2001, 184, 101–122. [Google Scholar] [CrossRef]
- Reiber, H. Dynamics of brain-derived proteins in cerebrospinal fluid. Clin. Chim. Acta 2001, 310, 173–186. [Google Scholar] [CrossRef]
- Valencia-Vera, E.; Martinez-Escribano Garcia-Ripoll, A.; Abalos-Garcia, C.; Segovia-Cuevas, M.J. Application of κ free light chains in cerebrospinal fluid as a biomarker in multiple sclerosis diagnosis: Development of a diagnosis algorithm. Clin. Chem. Lab. Med. 2018, 56, 609–613. [Google Scholar] [CrossRef]
- Andersson, M.; Alvarez-Cermeño, J.; Bernardi, G.; Cogato, I.; Fredman, P.; Frederiksen, J.; Fredrikson, S.; Gallo, P.; Grimaldi, L.M.; Grønning, M. Cerebrospinal fluid in the diagnosis of multiple sclerosis: A consensus report. J. Neurol. Neurosurg. Psychiatry 1994, 57, 897–902. [Google Scholar] [CrossRef]
- Nylander, A.; Hafler, D.A. Multiple sclerosis. J. Clin. Investig. 2012, 122, 1180–1188. [Google Scholar] [CrossRef]
- Hassan-Smith, G.; Durant, L.; Tsentemeidou, A.; Assi, L.K.; Faint, J.M.; Kalra, S.; Douglas, M.R.; Curnow, S.J. High sensitivity and specificity of elevated cerebrospinal fluid kappa free light chains in suspected multiple sclerosis. J. Neuroimmunol. 2014, 276, 175–179. [Google Scholar] [CrossRef]
- Fortini, A.S.; Sanders, E.L.; Weinshenker, B.G.; Katzmann, J.A. Cerebrospinal fluid oligoclonal bands in the diagnosis of multiple sclerosis. Isoelectric focusing with IgG immunoblotting compared with high-resolution agarose gel electrophoresis and cerebrospinal fluid IgG index. Am. J. Clin. Pathol. 2003, 120, 672–675. [Google Scholar] [CrossRef]
- Ramsden, D.B. Multiple sclerosis: Assay of free immunoglobulin light chains. Ann. Clin. Biochem. 2017, 54, 5–13. [Google Scholar] [CrossRef]
- Pieri, M.; Storto, M.; Pignalosa, S.; Zenobi, R.; Buttari, F.; Bernardini, S.; Centonze, D.; Dessi, M. KFLC Index utility in multiple sclerosis diagnosis: Further confirmation. J. Neuroimmunol. 2017, 309, 31–33. [Google Scholar] [CrossRef]
- Duranti, F.; Pieri, M.; Centonze, D.; Buttari, F.; Bernardini, S.; Dessi, M. Determination of kFLC and k Index in cerebrospinal fluid: A valid alternative to assess intrathecal immunoglobulin synthesis. J. Neuroimmunol. 2013, 263, 116–120. [Google Scholar] [CrossRef]
- Senel, M.; Tumani, H.; Lauda, F.; Presslauer, S.; Mojib-Yezdani, R.; Otto, M.; Brettschneider, J. Cerebrospinal fluid immunoglobulin kappa light chain in clinically isolated syndrome and multiple sclerosis. PLoS ONE 2014, 9, e88680. [Google Scholar] [CrossRef]
- Makshakov, G.; Nazarov, V.; Kochetova, O.; Surkova, E.; Lapin, S.; Evdoshenko, E. Diagnostic and prognostic value of the cerebrospinal fluid concentration of immunoglobulin free light chains in clinically isolated syndrome with conversion to multiple sclerosis. PLoS ONE 2015, 10, e0143375. [Google Scholar] [CrossRef]
- Kaplan, B.; Golderman, S.; Yahalom, G.; Yeskaraev, R.; Ziv, T.; Aizenbud, B.M.; Sela, B.A.; Livneh, A. Free light chain monomer dimer patterns in the diagnosis of multiple sclerosis. J. Immunol. Methods 2013, 390, 74–80. [Google Scholar] [CrossRef]
- Voortman, M.M.; Stojakovic, T.; Pirpamer, L.; Jehna, M.; Langkammer, C.; Scharnagl, H.; Reindl, M.; Ropele, S.; Seifert-Held, T.; Archelos, J.J. Prognostic value of free light chains lambda and kappa in early multiple sclerosis. Mult. Scler. 2017, 23, 1496–1505. [Google Scholar] [CrossRef]
- Nazarov, V.; Makshakov, G.; Kalinin, I.; Lapin, S.; Surkova, E.; Mikhailova, L.; Gilburd, B.; Skoromets, A.; Evdoshenko, E. Concentrations of immunoglobulin free light chains in cerebrospinal fluid predict increased level of brain atrophy in multiple sclerosis. Immunol. Res. 2018, 66, 761–767. [Google Scholar] [CrossRef]
- Nazarov, V.D.; Makshakov, G.S.; Mazing, A.V.; Surkova, E.A.; Krasnov, V.S.; Shumilina, M.V.; Totolyan, N.A.; Evdoshenko, E.P.; Lapin, S.V.; Emanuel, V.L.; et al. Diagnostic value of immunoglobulin free light chains at the debut of multiple sclerosis. Zhurnal Nevrol. Psikhiatrii Im. SS Korsakova 2017, 117, 60–65. [Google Scholar] [CrossRef]
- Goffette, S.; Schluep, M.; Henry, H.; Duprez, T.; Sindic, C.J. Detection of oligoclonal free kappa chains in the absence of oligoclonal IgG in the CSF of patients with suspected multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2004, 75, 308–310. [Google Scholar] [CrossRef]
- Sáez, M.S.; Rojas, J.I.; Lorenzón, M.V.; Sánchez, F.; Patrucco, L.; Míguez, J.; Azcona, C.; Sorroche, P.; Cristiano, E. Validation of CSF free light chain in diagnosis and prognosis of multiple sclerosis and clinically isolated syndrome: Prospective cohort study in Buenos Aires. J. Neurol. 2019, 266, 112–118. [Google Scholar] [CrossRef]
- Christiansen, M.; Gjelstrup, M.C.; Stilund, M.; Christensen, T.; Petersen, T.; Jon Møller, H. Cerebrospinal fluid free kappa light chains and kappa index perform equal to oligoclonal bands in the diagnosis of multiple sclerosis. Clin. Chem. Lab. Med. 2018, 57, 210–220. [Google Scholar] [CrossRef]
- Gurtner, K.M.; Shosha, E.; Bryant, S.C.; Andreguetto, B.D.; Murray, D.L.; Pittock, S.J.; Willrich, M.A.V. CSF free light chain identification of demyelinating disease: Comparison with oligoclonal banding and other CSF indexes. Clin. Chem. Lab. Med. 2018, 56, 1071–1080. [Google Scholar] [CrossRef]
- Menéndez-Valladares, P.; García-Sánchez, M.I.; Adorna Martínez, M.; García De Veas Silva, J.L.; Bermudo Guitarte, C.; Izquierdo Ayuso, G. Validation and meta-analysis of kappa index biomarker in multiple sclerosis diagnosis. Autoimmun. Rev. 2019, 18, 43–49. [Google Scholar] [CrossRef]
- Mader, S.; Brimberg, L. Aquaporin-4 Water Channel in the Brain and Its Implication for Health and Disease. Cells 2019, 8, E90. [Google Scholar] [CrossRef] [PubMed]
- Sato, D.K.; Callegaro, D.; Lana-Peixoto, M.A.; Waters, P.J.; de Haidar Jorge, F.M.; Takahashi, T.; Nakashima, I.; Apostolos-Pereira, S.L.; Talim, N.; Simm, R.F.; et al. Distinction between MOG antibody-positive and AQP4 antibody-positive NMO spectrum disorders. Neurology 2014, 82, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Waters, P.; Woodhall, M.; O’Connor, K.C.; Reindl, M.; Lang, B.; Sato, D.K.; Juryńczyk, M.; Tackley, G.; Rocha, J.; Takahashi, T.; et al. MOG cell-based assay detects non-MS patients with inflammatory neurologic disease. Neurol. Neuroimmunol. Neuroinflamm. 2015, 2, e89. [Google Scholar] [CrossRef] [PubMed]
- Lopergolo, D.; Scribano, D.; Dell’Abate, M.T.; Zuppi, C.; Urbani, A.; De Michele, T. La misura delle catene leggere libere nel liquor nella diagnosi della sclerosi. Bioch. Clin. 2018, 42, e22–e25. [Google Scholar] [CrossRef]
| Clinical Presentation | Additional Data Needed for MS Diagnosis |
| In patient with a typical attack/CIS at onset | |
| ≥2 clinical attacks and evidence of ≥2 lesions | None * |
| ≥2 clinical attacks and evidence of 1 lesion with history of previous attack involving lesions in different location | None * |
| ≥2 clinical attacks and evidence of 1 lesion | Evidence for DIS established by an additional clinical attack implicating different CNS site OR by MRI |
| 1 clinical attack and evidence of ≥2 lesions | Evidence for DIT established by an additional clinical attack implicating different CNS site or by MRI OR positive CSF-specific (i.e., not in serum) oligoclonal bands ** |
| 1 clinical attack and evidence of 1 lesion | Evidence for DIS established by an additional clinical attack implicating different CNS site or by MRI ORevidence for DIT established by an additional clinical attack implicating different CNS site OR by MRI OR positive CSF-specific (i.e., not in serum) oligoclonal bands ** |
| Clinical presentation | Additional data needed for MS Diagnosis |
| In patient with a progression of disability from onset (PPMS) | |
| Progression from onset | 1 year of disability progression (retrospective or prospective) AND two of these criteria:
|
| Authors [Ref.] | Number of Subjects | Assay Method | Metrics | Cut-Off | Sensitivity, % | Specificity, % |
|---|---|---|---|---|---|---|
| Menendez-Valladares, P. et al. [50] | 334 patients | Nephelometry | kFLC Index | 10.62 | 91% | 89% |
| Saez, M.S. et al. [47] | 77 patients | Turbidimetry | OCB | Positive | 93% | 90.4% |
| CSF kFLC | 7.1 mg/L | 95% | 97% | |||
| CSF λFLC | 0.7 mg/L | 71% | 81% | |||
| kFLC + λFLC | / | 95% | ||||
| Puthenparampil, M. et al. [20] | 70 patients | Nephelometry | IgG Index | / | / | / |
| CSF and serum kFLC | / | / | / | |||
| 37 controls | CSF and serum λFLC | / | / | / | ||
| kFLC Index | 4.25 | 93.8% | 100.0% | |||
| λFLC Index | / | / | / | |||
| Christiansen, M. et al. [48] | 230 patients | Turbidimetry | OCB | Positive | 82.3% (MS) 56.8% (CIS) | 93.8% |
| IgG Index | 0.64 | 72.9% (MS) 51.3% (CIS) | 95.9% | |||
| CSF kFLC | 0.42 mg/L | 93.8% (MS) 70.3% (CIS) | 85.6% | |||
| CSF λFLC | 0.14 mg/L | 93.8% (MS) 86.5% (CIS) | 35.1% | |||
| kFLC Index | 5.9 | 92.7% (MS) 70.3% (CIS) | 86.6% | |||
| λFLC Index | 2.8 | 93.8% (MS) 81,1% (CIS) | 46.4% | |||
| Gurtner, K.M. et al. [49] | 325 residual paired CSF and serum samples | Nephelometry | OCB | Positive (≥4 bands) | 86.6% | 89.6% |
| CSF kFLC | 0.0611 mg/dL | 92.5% | 86.1% | |||
| CSF λFLC | 0.0244 mg/dL | 75.8% | 84.4% | |||
| kFLC Index | ≥8.87 | 88.1% | 88.7% | |||
| Valencia-Vera, E. et al. [32] | 123 unselected consecutive patients with CSF OCB request | Nephelometry | OCB | Positive | 89.2% | 81.2% |
| ≥0.92 | 97.3% | 45.88% | ||||
| kFLC index | ≥2.91 | 83.8% | 85.8% | |||
| ≥8.33 | 70.2% | 95.6% | ||||
| Goffette et al. [46] | 33 patients with clinical suspicion of MS | Immunoaffinity-mediated capillary blot | Free K Bands | Presence of free kappa bands | / | / |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lo Sasso, B.; Agnello, L.; Bivona, G.; Bellia, C.; Ciaccio, M. Cerebrospinal Fluid Analysis in Multiple Sclerosis Diagnosis: An Update. Medicina 2019, 55, 245. https://doi.org/10.3390/medicina55060245
Lo Sasso B, Agnello L, Bivona G, Bellia C, Ciaccio M. Cerebrospinal Fluid Analysis in Multiple Sclerosis Diagnosis: An Update. Medicina. 2019; 55(6):245. https://doi.org/10.3390/medicina55060245
Chicago/Turabian StyleLo Sasso, Bruna, Luisa Agnello, Giulia Bivona, Chiara Bellia, and Marcello Ciaccio. 2019. "Cerebrospinal Fluid Analysis in Multiple Sclerosis Diagnosis: An Update" Medicina 55, no. 6: 245. https://doi.org/10.3390/medicina55060245
APA StyleLo Sasso, B., Agnello, L., Bivona, G., Bellia, C., & Ciaccio, M. (2019). Cerebrospinal Fluid Analysis in Multiple Sclerosis Diagnosis: An Update. Medicina, 55(6), 245. https://doi.org/10.3390/medicina55060245









