ESBL and AmpC β-Lactamases in Clinical Strains of Escherichia coli from Serra da Estrela, Portugal
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains
2.2. Phenotype Identification and Antibiotics Susceptibility
2.3. Molecular Characterization of bla Genes
3. Results
3.1. Bacterial Strains
3.2. Phenotype Identification and Antibiotic Susceptibility
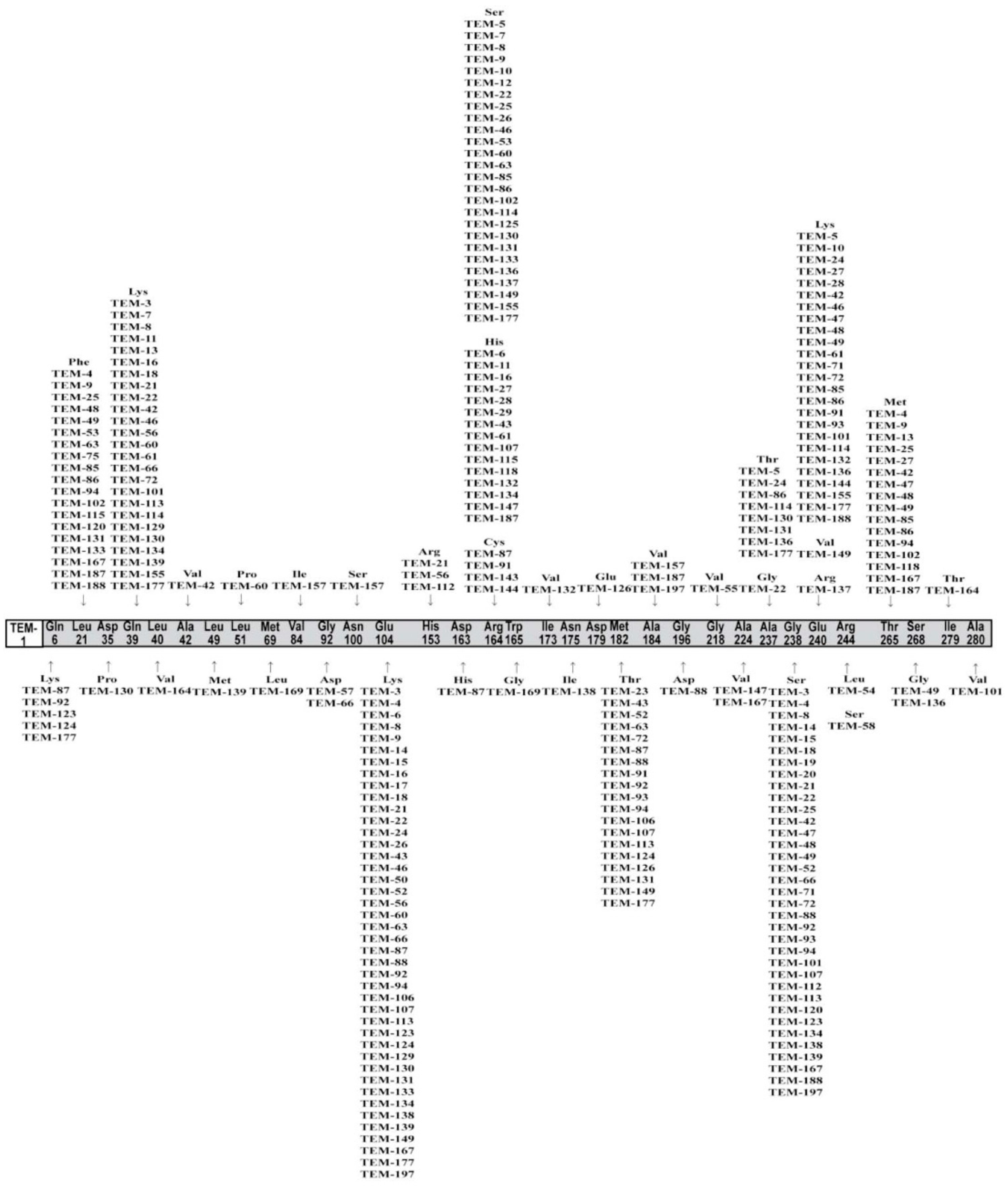
3.3. Molecular Characterization of bla Genes
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fernandes, R.; Amador, P.; Prudêncio, C. β-Lactams: chemical structure, mode of action and mechanisms of resistance. Rev. Med. Microbiol. 2013, 24, 7–17. [Google Scholar] [CrossRef]
- Hawkey, P.M.; Jones, A.M. The changing epidemiology of resistance. J. Antimicrob. Chemother. 2009, 64, i3–i10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amador, P.; Duarte, I.M.; Roberto da Costa, R.P.; Fernandes, R.; Prudêncio, C. Characterization of antibiotic resistance in Enterobacteriaceae from agricultural manure and soil in portugal. Soil Sci. 2018, 182, 292–301. [Google Scholar] [CrossRef]
- Amador, P.P.; Fernandes, R.M.; Prudêncio, M.C.; Barreto, M.P.; Duarte, I.M. Antibiotic resistance in wastewater: occurrence and fate of Enterobacteriaceae producers of class a and class c β-lactamases. J. Environ. Sci. Health Part A Toxic Hazard. Substanc. Environ. Eng. 2015, 50, 26–39. [Google Scholar] [CrossRef]
- Cunha, S.; Mendes; Rego, D.; Meireles, D.; Fernandes, R.; Carvalho, A.; Martins da Costa, P. Effect of competitive exclusion in rabbits using an autochthonous probiotic. World Rabbit Sci. 2017, 25, 123–134. [Google Scholar] [CrossRef] [Green Version]
- Coque, T.M.; Baquero, F.; Canton, R. Increasing prevalence of ESBL producing Enterobacteriaceae in Europe. Eurosurveillance 2008, 13, 9044. [Google Scholar]
- Machado, E.; Coque, T.M.; Canton, R.; Baquero, F.; Sousa, J.C.; Peixe, L. Dissemination in Portugal of CTX-M-15-, OXA-1-, and TEM-1-producing Enterobacteriaceae strains containing the aac(6’)-ib-cr gene, which encodes an aminoglycoside- and fluoroquinolone-modifying enzyme. Antimicrob. Agents Chemother. 2006, 50, 3220–3221. [Google Scholar] [CrossRef] [PubMed]
- Cantón, R.; Novais, A.; Valverde, A.; Machado, E.; Peixe, L.; Baquero, F.; Coque, T.M. Prevalence and spread of extended-spectrum β-lactamase-producing Enterobacteriaceae in Europe. Clin. Microbiol. Infect. 2008, 14, 144–153. [Google Scholar] [CrossRef]
- Machado, E.; Coque, T.M.; Cantón, R.; Novais, Â.; Sousa, J.C.; Baquero, F.; Peixe, L. High diversity of extended-spectrum β-lactamases among clinical isolates of Enterobacteriaceae from Portugal. J. Antimicrob. Chemother. 2007, 60, 1370–1374. [Google Scholar] [CrossRef]
- Naas, T.; Oueslati, S.; Bonnin, R.A.; Dabos, M.L.; Zavala, A.; Dortet, L.; Retailleau, P.; Iorga, B.I. Beta-lactamase database (BLDB)—Structure and function. J. Enzyme Inhibit. Med. Chem. 2017, 32, 917–919. [Google Scholar] [CrossRef]
- Pimenta, A.C.; Fernandes, R.; Moreira, I.S. Evolution of drug resistance: insight on TEM β-lactamases structure and activity and β-lactam antibiotics. Mini Rev. Med. Chem. 2014, 14, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Carmeli, Y. Strategies for managing today’s infections. Clin. Microbiol. Infect. 2008, 14, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Livermore, D.M.; Canton, R.; Gniadkowski, M.; Nordmann, P.; Rossolini, G.M.; Arlet, G.; Ayala, J.; Coque, T.M.; Kern-Zdanowicz, I.; Luzzaro, F.; et al. CTX-M: Changing the face of ESBLs in Europe. J. Antimicrob. Chemother. 2007, 59, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Pitout, J.D.D.; Campbell, L.; Church, D.L.; Gregson, D.B.; Laupland, K.B. Molecular characteristics of travel-related extended-spectrum—Lactamase-producing Escherichia coli isolates from the calgary health region. Antimicrob. Agents Chemother. 2009, 53, 2539–2543. [Google Scholar] [CrossRef] [PubMed]
- Kiratisin, P.; Apisarnthanarak, A.; Laesripa, C.; Saifon, P. Molecular characterization and epidemiology of extended-spectrum-β- lactamase-producing Escherichia coli and Klebsiella pneumoniae isolates causing health care-associated infection in Thailand, where the CTX-M family is endemic. Antimicrob. Agents Chemother. 2008, 52, 2818–2824. [Google Scholar] [CrossRef] [PubMed]
- Hawkey, P.M. Prevalence and clonality of extended-spectrum β-lactamases in Asia. Clin. Microbiol. Infect. 2008, 14, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.H.; Tuckman, M.; Keeney, D.; Ruzin, A.; Bradford, P.A. Characterization and sequence analysis of extended-spectrum-β- lactamase-encoding genes from Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis isolates collected during tigecycline phase 3 clinical trials. Antimicrob. Agents Chemother. 2009, 53, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, N.; Leitão, J.; Manageiro, V.; Ferreira, E.; Caniça, M. Spread of extended-spectrum β-lactamase CTX-M-producing Escherichia coli clinical isolates in community and nosocomial environments in Portugal. Antimicrob. Agents Chemother. 2007, 51, 1946–1955. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Zerr, D.M.; Weissman, S.J.; Englund, J.A.; Denno, D.M.; Klein, E.J.; Tarr, P.I.; Kwong, J.; Stapp, J.R.; Tulloch, L.G.; et al. Prevalence and mechanisms of broad-spectrum -lactam resistance in Enterobacteriaceae: A children’s hospital experience. Antimicrob. Agents Chemother. 2009, 53, 340. [Google Scholar] [CrossRef]
- Mohanty, S.; Gaind, R.; Ranjan, R.; Deb, M. Use of the cefepime-clavulanate ESBL Etest for detection of extended-spectrum beta-lactamases in AmpC co-producing bacteria. J. Infect. Dev. Countr. 2010, 4, 24–29. [Google Scholar]
- Li, Y.; Li, Q.; Du, Y.; Jiang, X.; Tang, J.; Wang, J.; Li, G.; Jiang, Y. Prevalence of plasmid-mediated AmpC β-lactamases in a Chinese University Hospital from 2003 to 2005: First report of CMY-2-type AmpC β-lactamase resistance in China. J. Clin. Microbiol. 2008, 46, 1317–1321. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Wu, W.; Ye, X.; Xu, X.; Zhu, D. The molecular characteristics of cefepime-susceptible Escherichia coli and Klebsiella spp. isolates with a positive β-lactamase screening test result but negative confirmation. Eur. J. Clin. Microbiol. Infect. Dis. 2010, 29, 1297–1299. [Google Scholar] [CrossRef] [PubMed]
- Oteo, J.; Cercenado, E.; Fernández-Romero, S.; Saéz, D.; Padilla, B.; Zamora, E.; Cuevas, O.; Bautista, V.; Campos, J. Extended-spectrum-β-lactamase-producing Escherichia coli as a cause of pediatric infections: Report of a neonatal intensive care unit outbreak due to a CTX-M-14-producing strain. Antimicrob. Agents Chemother. 2012, 56, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, M.; Tran, J.H.; Chow, N.; Jacoby, G.A. Epidemiology of Conjugative Plasmid-Mediated AmpC β-Lactamases in the United States. Antimicrob. Agents Chemother. 2004, 48, 533–537. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, G.A. AmpC -Lactamases. Clin. Microbiol. Rev. 2009, 22, 161–182. [Google Scholar] [CrossRef]
- Oteo, J.; Cercenado, E.; Cuevas, Ó.; Bautista, V.; Delgado-Iribarren, A.; Orden, B.; Pérez-Vázquez, M.; García-Cobos, S.; Campos, J. AmpC β-lactamases in Escherichia coli: Emergence of CMY-2-producing virulent phylogroup D isolates belonging mainly to STs 57, 115, 354, 393, and 420, and phylogroup B2 isolates belonging to the international clone O25b-ST131. Diagn. Microbiol. Infect. Dis. 2010, 67, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Paterson, D.L.; Bonomo, R.A. Extended-Spectrum -Lactamases: a Clinical Update. Clin. Microbiol. Rev. 2005, 18, 657–686. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, P.; Singh, N.S.; Virdi, J.S. Escherichia coli β-Lactamases: What Really Matters. Front. Microbiol. 2016, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V. An update on newer beta-lactamases. Indian J. Med. Res. 2007, 126, 417–427. [Google Scholar]
- Amador, P.; Prudêncio, C.; Vieira, M.; Ferraz, R.; Fonte, R.; Silva, N.; Coelho, P.; Fernandes, R. β-lactamases in the biochemistry and molecular biology laboratory. Biochem. Mol. Biol. Educ. 2009, 37, 301–306. [Google Scholar]
- Meireles, D.; Leite-Martins, L.; Bessa, L.J.; Cunha, S.; Fernandes, R.; de Matos, A.; Manaia, C.M.; Martins da Costa, P. Molecular characterization of quinolone resistance mechanisms and extended-spectrum β-lactamase production in Escherichia coli isolated from dogs. Comp. Immunol. Microbiol. Infect. Dis. 2015, 41, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.; Vieira, M.; Ferraz, R.; Prudêncio, C. Bloodstream infections caused by multidrug-resistant Enterobacteriaceae: report from two Portuguese hospitals. J. Hosp. Infect. 2008, 70, 93–95. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.; Prudêncio, C. Post-surgical wound infections involving Enterobacteriaceae with reduced susceptibility to β-lactams in two Portuguese hospitals. Int. Wound J. 2010, 7, 508–514. [Google Scholar] [CrossRef] [PubMed]
- CLSI Performance Standards for Antimicrobial Susceptibility Testing, 26th ed.; CLSI supplement M100S; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2016.
- Polsfuss, S.; Bloemberg, G.V.; Giger, J.; Meyer, V.; Bottger, E.C.; Hombach, M. Practical Approach for reliable detection of ampc beta-lactamase-producing Enterobacteriaceae. J. Clin. Microbiol. 2011, 49, 2798–2803. [Google Scholar] [CrossRef] [PubMed]
- Dallenne, C.; da Costa, A.; Decré, D.; Favier, C.; Arlet, G. Development of a set of multiplex PCR assays for the detection of genes encoding important β-lactamases in Enterobacteriaceae. J. Antimicrob. Chemother. 2010, 65, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.; Amador, P.; Oliveira, C.; Prudêncio, C. Molecular characterization of ESBL-producing Enterobacteriaceae in northern Portugal. Sci. World J. 2014, 2014, 782897. [Google Scholar] [CrossRef] [PubMed]
- Baudry, P.J.; Mataseje, L.; Zhanel, G.G.; Hoban, D.J.; Mulvey, M.R. Characterization of plasmids encoding CMY-2 AmpC β-lactamases from Escherichia coli in Canadian intensive care units. Diag. Microbiol. Infect. Dis. 2009, 65, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Shannon, E.; Abu-Ghannam, N. Antibacterial derivatives of marine algae: An Overview of pharmacological mechanisms and applications. Mar. Drugs 2016, 14, 81. [Google Scholar] [CrossRef] [PubMed]
- Bradford, P.A. Extended-spectrum -lactamases in the 21st century: Characterization, epidemiology, and detection of this important resistance threat. Clin. Microbiol. Rev. 2001, 14, 933–951. [Google Scholar] [CrossRef]
- Kaye, K.S.; Pogue, J.M. Infections Caused by Resistant Gram-Negative Bacteria: Epidemiology and Management. Pharmacother. J. Hum. Pharmacol. Drug Therap. 2015, 35, 949–962. [Google Scholar] [CrossRef]
- Manchanda, V. Occurrence and detection of AmpC beta-lactamases among gram-negative clinical isolates using a modified three-dimensional test at Guru Tegh Bahadur Hospital, Delhi, India. J. Antimicrob. Chemother. 2003, 51, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Rawat, A.; Handa, S.; Thakuria, B.; Handa, D.; Asthana, A. Evaluation of phenotypic tests for the detection of AmpC beta-lactamase in clinical isolates of Escherichia coli. Indian J. Pathol. Microbiol. 2013, 56, 135. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.; Gestoso, Á.; Freitas, J.M.; Santos, P.; Prudêncio, C. High resistance to fourth-generation cephalosporins among clinical isolates of Enterobacteriaceae producing extended-spectrum β-lactamases isolated in Portugal. Int. J. Antimicrob. Agents 2009, 33, 184–185. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.Y.; Ng, L.S.Y.; He, J.; Koh, T.H.; Hsu, L.Y. Evaluation of screening methods to detect plasmid-mediated AmpC in Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis. Antimicrob. Agents Chemother. 2009, 53, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Pimenta, A.C.; Martins, J.M.; Fernandes, R.; Moreira, I.S. Ligand-induced structural changes in TEM-1 probed by molecular dynamics and relative binding free energy calculations. J. Chem. Inf. Model. 2013, 53. [Google Scholar] [CrossRef] [PubMed]
- Woodford, N.; Reddy, S.; Fagan, E.J.; Hill, R.L.R.; Hopkins, K.L.; Kaufmann, M.E.; Kistler, J.; Palepou, M.F.I.; Pike, R.; Ward, M.E.; et al. Wide geographic spread of diverse acquired AmpC β-lactamases among Escherichia coli and Klebsiella spp. in the UK and Ireland. J. Antimicrob. Chemother. 2007, 59, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Xu, J.; He, F. Draft genome sequence of an NDM-5, CTX-M-15 and OXA-1 co-producing Escherichia coli ST167 clinical strain isolated from a urine sample. J. Glob. Antimicrob. Resist. 2018, 14, 284–286. [Google Scholar] [CrossRef]


| PCR Multiplex | Targeted β-lactamases | Primer’s Name | Sequence (5′-3′) | Product’s Size (bp) |
|---|---|---|---|---|
| PCR multiplex TSO (TEM, SHV, and OXA-1-like) | TEM variants (including TEM-1 and TEM-2) | MultiTSO-T_for | CATTTCCGTGTCGCCCTTATTC | 800 |
| MultiTSO-T_rev | CGTTCATCCATAGTTGCCTGAC | |||
| SHV variants (including SHV-1) | MultiTSO-S_for | AGCCGCTTGAGCAAATTAAAC | 713 | |
| MultiTSO-S_rev | ATCCCGCAGATAAATCACCAC | |||
| OXA-1-like variants OXA-1, OXA-4 and OXA-30 | MultiTSO-O_for | GGCACCAGATTCAACTTTCAAG | 564 | |
| MultiTSO-O_rev | GACCCCAAGTTTCCTGTAAGTG | |||
| PCR multiplex CTX-M (CTX-M Group 1, CTX-M Group 2, and CTX-M Group 9) | CTX-M Group 1 variants (including CTX-M-1, CTX-M-3 and CTX-M-15) | MultiCTX-MGrp1_for | TTAGGAARTGTGCCGCTGYAa | 688 |
| MultiCTX-MGrp1-2_rev | CGATATCGTTGGTGGTRCCATa | |||
| CTX-M Group 2 variants | MultiCTX-MGrp2_for | CGTTAACGGCACGATGAC | 404 | |
| MultiCTX-MGrp1-2_rev | CGATATCGTTGGTGGTRCCATa | |||
| CTX-M Group 9 variants (including CTX-M-9 and CTX-M-14) | MultiCTX-MGrp9_for | TCAAGCCTGCCGATCTGGT | 561 | |
| MultiCTX-MGrp9_rev | TGATTCTCGCCGCTGAAG | |||
| PCR CTX-M Group 8/25 | CTX-M Group 8/25 variants CTX-M-8, CTX-M-25, CTX-M-26, and CTX-M-39 to CTX-M-41 | CTX-MGrp8/25_for | AACRCRCAGACGCTCTACa | 326 |
| CTX-MGrp8/25_rev | TCGAGCCGGAASGTGYATa | |||
| PCR multiplex AmpC (ACC, FOX, MOX, DHA, CIT, and EBC) | ACC variants ACC-1 and ACC-2 | MultiACC_for | CACCTCCAGCGACTTGTTAC | 346 |
| MultiACC_rev | GTTAGCCAGCATCACGATCC | |||
| FOX variants FOX-1 to FOX-5 | MultiFOX_for | CTACAGTGCGGGTGGTTT | 162 | |
| MultiFOX_rev | CTATTTGCGGCCAGGTGA | |||
| MOX variants MOX-1, MOX-2, CMY-1, CMY-8 to CMY-11, and CMY-19 | MultiMOX_for | GCAACAACGACAATCCATCCT | 895 | |
| MultiMOX_rev | GGGATAGGCGTAACTCTCCCAA | |||
| DHA variants DHA-1 and DHA-2 | MultiDHA_for | TGATGGCACAGCAGGATATTC | 997 | |
| MultiDHA_rev | GCTTTGACTCTTTCGGTATTCG | |||
| CIT variants LAT-1 to LAT-3, BIL-1, CMY-2 to CMY-7, CMY-12 to CMY-18, and CMY-21 to CMY-23 | MultiCIT_for | CGAAGAGGCAATGACCAGAC | 538 | |
| MultiCIT_rev | ACGGACAGGGTTAGGATAGYa | |||
| EBC variants ACT-1 and MIR-1 | MultiEBC_for | CGGTAAAGCCGATGTTGCG | 683 | |
| MultiEBC_rev | AGCCTAACCCCTGATACA |
| Strain | Source | Antibiogram | Presumptive Tests | Confirmatory | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ESBL | AmpC | Molecular Tests | ||||||||||||||||||||||||||||||||||||
| Etest | FOX | Etest | PCR | |||||||||||||||||||||||||||||||||||
| AMP | AMC | TZP | CXM | CT | TZ | ERT | MEM | GEN | CIP | LVX | TIG | SXT | CT/CTL | TZ/TZL | Diameter | CN/CNI | ESBL | AmpC | ||||||||||||||||||||
| MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | MIC (mg/L) | ratio | ratio | (mm) | ratio | ||||||||||||||||||||||
| A1 | Urine | HAI | ≥32 | R | 4 | ≤4 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | >8 | + | >8 | + | 27 | <8 | N.D. | N.D. | |||||||||
| A2 | Urine | HAI | ≥32 | R | ≥32 | R | ≥128 | R | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | 40 | >8 | + | >8 | + | 23 | <8 | OXA-1-like | N.D. | ||||||
| A3 | Respiratory | HAI | ≥32 | R | 16 | ≤4 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | 1 | ≤20 | >8 | + | P.Z. | + | 24 | <8 | OXA-1-like | N.D. | |||||||||
| A4 | Urine | HAI | ≥32 | R | 4 | ≤4 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | >8 | + | >8 | + | 25 | <8 | N.D. | N.D. | |||||||||
| A5 | Urine | HAI | ≥32 | R | 16 | ≥128 | R | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | P.Z. | + | P.Z. | + | 24 | <8 | N.D. | N.D. | |||||||
| A6 | Urine | HAI | ≥32 | R | 4 | ≤4 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | P.Z. | + | >8 | + | 24 | <8 | CTX-M Grp1 + OXA-1-like | N.D. | |||||||||
| A7 | Respiratory | HAI | ≥32 | R | 16 | 8 | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | 40 | >8 | + | >8 | + | 16 | + | <8 | CTX-M Grp1 | N.D. | ||||||||
| A8 | Urine | HAI | ≥32 | R | ≥32 | R | ≥128 | R | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | 40 | >8 | + | >8 | + | 24 | <8 | OXA-1-like | N.D. | ||||||
| B1 | Urine | HAI | ≥32 | R | 4 | ≥128 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | P.Z. | + | >8 | + | 26 | <8 | OXA-1-like | N.D. | ||||||||
| B2 | Urine | HAI | ≥32 | R | ≥32 | R | 8 | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | P.Z. | + | >8 | + | 22 | <8 | N.D. | N.D. | ||||||||
| B3 | Urine | HAI | ≥32 | R | ≥32 | R | ≥128 | R | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | P.Z. | + | P.Z. | + | 24 | <8 | CTX-M Grp1 + OXA-1-like | N.D. | ||||||
| B4 | Respiratory | HAI | ≥32 | R | 16 | 64 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | P.Z. | + | >8 | + | 24 | <8 | OXA-1-like | N.D. | ||||||||
| B5 | Respiratory | HAI | ≥32 | R | 16 | 64 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | P.Z. | + | P.Z. | + | 24 | <8 | N.D. | N.D. | ||||||||
| B6 | Urine | HAI | ≥32 | R | 16 | 16 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | P.Z. | + | P.Z. | + | 24 | <8 | OXA-1-like | N.D. | |||||||
| B7 | Respiratory | HAI | ≥32 | R | 16 | 8 | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | P.Z. | + | P.Z. | + | 24 | <8 | CTX-M Grp1 + TEM | N.D. | |||||||||
| B8 | Urine | HAI | ≥32 | R | ≥32 | R | 16 | R | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | P.Z. | + | 17 | + | <8 | OXA-1-like | N.D. | ||||
| B9 | Urine | HAI | ≥32 | R | ≥32 | R | 8 | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | >8 | + | 26 | <8 | OXA-1-like | N.D. | ||||||
| C1 | Urine | HAI | ≥32 | R | ≥32 | R | ≥128 | R | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | >8 | + | 24 | <8 | CTX-M Grp1 + OXA-1-like | N.D. | |||||
| C2 | Urine | HAI | ≥32 | R | 8 | ≤4 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | P.Z. | + | 24 | <8 | CTX-M Grp1 + TEM + OXA-1-like | N.D. | |||||||
| C3 | Urine | HAI | ≥32 | R | ≥32 | R | 32 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | P.Z. | + | 17 | + | <8 | OXA-1-like | N.D. | |||||
| C4 | Urine | HAI | ≥32 | R | ≥32 | R | 16 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | >8 | + | 16 | + | <8 | OXA-1-like | N.D. | |||||
| C5 | Urine | HAI | ≥32 | R | ≥32 | R | ≥128 | R | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | 40 | >8 | + | P.Z. | + | 22 | <8 | OXA-1-like | N.D. | ||||||
| C6 | Urine | HAI | ≥32 | R | ≥32 | R | 64 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | P.Z. | + | >8 | + | 17 | + | <8 | OXA-1-like | N.D. | |||||
| C7 | Urine | HAI | ≥32 | R | 16 | 16 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | P.Z. | + | >8 | + | 24 | <8 | OXA-1-like | N.D. | |||||||
| C8 | Skin and soft tissue | HAI | ≥32 | R | ≥32 | R | 64 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | >8 | + | 15 | + | <8 | OXA-1-like | N.D. | |||||
| C9 | Bloodstream | HAI | ≥32 | R | 16 | ≤4 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | P.Z. | + | >8 | + | 24 | <8 | CTX-M Grp1 | N.D. | ||||||||
| D1 | Urine | HAI | ≥32 | R | 4 | ≤4 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | P.Z. | + | >8 | + | 20 | <8 | CTX-M Grp1 | N.D. | |||||||||
| D2 | Skin and soft tissue | HAI | ≥32 | R | 16 | 32 | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | 1 | ≥320 | R | P.Z. | + | P.Z. | + | 15 | + | <8 | CTX-M Grp1 + OXA-1-like | N.D. | ||||||
| D3 | Skin and soft tissue | HAI | ≥32 | R | 16 | 64 | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≤20 | P.Z. | + | P.Z. | + | 23 | <8 | TEM + OXA-1-like | N.D. | ||||||||
| D4 | Skin and soft tissue | HAI | ≥32 | R | ≥32 | R | 16 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | P.Z. | + | 17 | + | <8 | CTX-M Grp1 + OXA-1-like | N.D. | |||||
| D5 | Urine | HAI | ≥32 | R | ≥32 | R | ≥128 | R | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | >8 | + | 17 | + | <8 | CTX-M Grp1 + OXA-1-like | N.D. | ||||
| D6 | Urine | HAI | ≥32 | R | ≥32 | R | ≥128 | R | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | P.Z. | + | >8 | + | 24 | <8 | OXA-1-like | N.D. | |||||
| D7 | Skin and soft tissue | HAI | ≥32 | R | ≥32 | R | ≥128 | R | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | P.Z. | + | >8 | + | 17 | + | <8 | CTX-M Grp1 + OXA-1-like | N.D. | ||||
| D8 | Urine | HAI | ≥32 | R | 8 | ≤4 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≤1 | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | P.Z. | + | P.Z. | + | 25 | <8 | CTX-M Grp1 + TEM | N.D. | ||||||||
| D9 | Urine | CAI | ≥32 | R | ≥32 | R | ≥128 | R | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | >8 | + | 17 | + | <8 | CTX-M Grp1 + OXA-1-like | N.D. | ||||
| E2 | Skin and soft tissue | HAI | ≥32 | R | ≥32 | R | 64 | >64 | R | >64 | R | ≥64 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | >8 | + | >8 | + | 17 | + | <8 | CTX-M Grp1 + OXA-1-like | N.D. | |||||
| E3 | Urine | HAI | ≥32 | R | 16 | 16 | >64 | R | >64 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | ≥4 | R | ≥8 | R | ≤0,5 | ≥320 | R | P.Z. | + | >8 | + | 15 | + | <8 | CTX-M Grp1 + OXA-1-like | N.D. | ||||||
| E4 | Skin and soft tissue | HAI | ≥32 | R | ≥32 | R | 32 | >64 | R | 8 | R | 16 | R | ≤0,5 | ≤0,25 | ≥16 | R | 0,5 | 1 | ≤0,5 | ≤20 | Inconclusive | Inconclusive | 9 | + | >8 | + | TEM | CIT | |||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, C.; Amador, P.; Prudêncio, C.; Tomaz, C.T.; Tavares-Ratado, P.; Fernandes, R. ESBL and AmpC β-Lactamases in Clinical Strains of Escherichia coli from Serra da Estrela, Portugal. Medicina 2019, 55, 272. https://doi.org/10.3390/medicina55060272
Oliveira C, Amador P, Prudêncio C, Tomaz CT, Tavares-Ratado P, Fernandes R. ESBL and AmpC β-Lactamases in Clinical Strains of Escherichia coli from Serra da Estrela, Portugal. Medicina. 2019; 55(6):272. https://doi.org/10.3390/medicina55060272
Chicago/Turabian StyleOliveira, Cátia, Paula Amador, Cristina Prudêncio, Cândida T Tomaz, Paulo Tavares-Ratado, and Rúben Fernandes. 2019. "ESBL and AmpC β-Lactamases in Clinical Strains of Escherichia coli from Serra da Estrela, Portugal" Medicina 55, no. 6: 272. https://doi.org/10.3390/medicina55060272







