Role of Key Micronutrients from Nutrigenetic and Nutrigenomic Perspectives in Cancer Prevention
Abstract
1. Introduction
2. Cancer Risk Represents a Sum of Complex Interactions of Environmental Exposures
3. Vitamins
3.1. Vitamin C
3.2. Vitamin A
3.3. Vitamin D
3.4. Folic Acid
4. Selenium
5. Polyunsaturated Fatty Acids (PUFAs)
6. Prebiotics, Probiotics and Dietary Fibers
7. Conclusions and Further Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- You, J.S.; Jones, P.A. Cancer genetics and epigenetics: Two sides of the same coin? Cancer Cell 2012, 22, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Kussmann, M.; Krause, L.; Siffert, W. Nutrigenomics: Where are we with genetic and epigenetic markers for disposition and susceptibility? Nutr. Rev. 2010, 68 (Suppl. 1), S38–S47. [Google Scholar] [CrossRef] [PubMed]
- Cojocneanu Petric, R.; Braicu, C.; Raduly, L.; Zanoaga, O.; Dragos, N.; Monroig, P.; Dumitrascu, D.; Berindan-Neagoe, I. Phytochemicals modulate carcinogenic signaling pathways in breast and hormone-related cancers. Onco Targets Ther. 2015, 8, 2053–2066. [Google Scholar] [CrossRef] [PubMed]
- Budisan, L.; Gulei, D.; Zanoaga, O.M.; Irimie, A.I.; Sergiu, C.; Braicu, C.; Gherman, C.D.; Berindan-Neagoe, I. Dietary Intervention by Phytochemicals and Their Role in Modulating Coding and Non-Coding Genes in Cancer. Int. J. Mol. Sci. 2017, 18, 1178. [Google Scholar] [CrossRef] [PubMed]
- Gulei, D.; Irimie, A.I.; Cojocneanu-Petric, R.; Schultze, J.L.; Berindan-Neagoe, I. Exosomes-Small Players, Big Sound. Bioconjug. Chem. 2018, 29, 635–648. [Google Scholar] [CrossRef]
- Hussain, S.S.; Kumar, A.P.; Ghosh, R. Food-based natural products for cancer management: Is the whole greater than the sum of the parts? Semin. Cancer Biol. 2016, 40-41, 233–246. [Google Scholar] [CrossRef]
- Irimie, A.I.; Braicu, C.; Cojocneanu-Petric, R.; Berindan-Neagoe, I.; Campian, R.S. Novel technologies for oral squamous carcinoma biomarkers in diagnostics and prognostics. Acta Odontol. Scand. 2015, 73, 161–168. [Google Scholar] [CrossRef]
- Irimie, A.I.; Braicu, C.; Pileczki, V.; Petrushev, B.; Soritau, O.; Campian, R.S.; Berindan-Neagoe, I. Knocking down of p53 triggers apoptosis and autophagy, concomitantly with inhibition of migration on SSC-4 oral squamous carcinoma cells. Mol. Cell. Biochem. 2016, 419, 75–82. [Google Scholar] [CrossRef]
- Irimie, A.I.; Braicu, C.; Zanoaga, O.; Pileczki, V.; Gherman, C.; Berindan-Neagoe, I.; Campian, R.S. Epigallocatechin-3-gallate suppresses cell proliferation and promotes apoptosis and autophagy in oral cancer SSC-4 cells. Onco Targets Ther. 2015, 8, 461–470. [Google Scholar]
- Ilea, A.; Babtan, A.M.; Bosca, B.A.; Crisan, M.; Petrescu, N.B.; Collino, M.; Sainz, R.M.; Gerlach, J.Q.; Campian, R.S. Advanced glycation end products (AGEs) in oral pathology. Arch. Oral Biol. 2018, 93, 22–30. [Google Scholar] [CrossRef]
- Magee, R.; Telonis, A.G.; Cherlin, T.; Rigoutsos, I.; Londin, E. Assessment of isomiR Discrimination Using Commercial qPCR Methods. Non-Coding RNA 2017, 3, 18. [Google Scholar] [CrossRef] [PubMed]
- Hubé, F.; Francastel, C. “Pocket-sized RNA-Seq”: A Method to Capture New Mature microRNA Produced from a Genomic Region of Interest. Non-Coding RNA 2015, 127–138. [Google Scholar] [CrossRef]
- Cui, Z.; Chiu, N.H.L.; Wambua, D.M. MicroRNA MultiTool: A Software for Identifying Modified and Unmodified Human microRNA Using Mass Spectrometry. Non-Coding RNA 2016, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Pop, L.A.; Puscas, E.; Pileczki, V.; Cojocneanu-Petric, R.; Braicu, C.; Achimas-Cadariu, P.; Berindan-Neagoe, I. Quality control of ion torrent sequencing library. Cancer Biomark. 2014, 14, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, K. Past, present and future of nutrigenomics and its influence on drug development. Curr. Drug Discov. Technol. 2013, 10, 35–46. [Google Scholar] [PubMed]
- Ahmadi, K.R.; Andrew, T. Opportunism: A panacea for implementation of whole-genome sequencing studies in nutrigenomics research? Genes Nutr. 2014, 9, 387. [Google Scholar] [CrossRef] [PubMed]
- Lucock, M.; Beckett, E.; Martin, C.; Jones, P.; Furst, J.; Yates, Z.; Jablonski, N.G.; Chaplin, G.; Veysey, M. UV-associated decline in systemic folate: Implications for human nutrigenetics, health, and evolutionary processes. Am. J. Hum. Biol. 2017, 29, e22929. [Google Scholar] [CrossRef] [PubMed]
- Bouchard-Mercier, A.; Paradis, A.M.; Perusse, L.; Vohl, M.C. Associations between polymorphisms in genes involved in fatty acid metabolism and dietary fat intakes. Lifestyle Genomics 2012, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ooi, S.X.; Lee, P.L.; Law, H.Y.; Say, Y.H. Bitter receptor gene (TAS2R38) P49A genotypes and their associations with aversion to vegetables and sweet/fat foods in Malaysian subjects. Asia Pac. J. Clin. Nutr. 2010, 19, 491–498. [Google Scholar] [PubMed]
- Bouchard-Mercier, A.; Godin, G.; Lamarche, B.; Perusse, L.; Vohl, M.C. Effects of peroxisome proliferator-activated receptors, dietary fat intakes and gene-diet interactions on peak particle diameters of low-density lipoproteins. J. Nutr. Nutr. 2011, 4, 36–48. [Google Scholar] [CrossRef]
- Corella, D.; Coltell, O.; Sorli, J.V.; Estruch, R.; Quiles, L.; Martinez-Gonzalez, M.A.; Salas-Salvado, J.; Castaner, O.; Aros, F.; Ortega-Calvo, M.; et al. Polymorphism of the Transcription Factor 7-Like 2 Gene (TCF7L2) Interacts with Obesity on Type-2 Diabetes in the PREDIMED Study Emphasizing the Heterogeneity of Genetic Variants in Type-2 Diabetes Risk Prediction: Time for Obesity-Specific Genetic Risk Scores. Nutrients 2016, 8, 793. [Google Scholar]
- Irimie, A.I.; Sonea, L.; Jurj, A.; Mehterov, N.; Zimta, A.A.; Budisan, L.; Braicu, C.; Berindan-Neagoe, I. Future trends and emerging issues for nanodelivery systems in oral and oropharyngeal cancer. Int. J. Nanomed. 2017, 12, 4593–4606. [Google Scholar] [CrossRef] [PubMed]
- Luceri, C.; Bigagli, E.; Pitozzi, V.; Giovannelli, L. A nutrigenomics approach for the study of anti-aging interventions: Olive oil phenols and the modulation of gene and microRNA expression profiles in mouse brain. Eur. J. Nutr. 2017, 56, 865–877. [Google Scholar] [CrossRef] [PubMed]
- Seo, E.J.; Wu, C.F.; Ali, Z.; Wang, Y.H.; Khan, S.I.; Walker, L.A.; Khan, I.A.; Efferth, T. Both Phenolic and Non-phenolic Green Tea Fractions Inhibit Migration of Cancer Cells. Front. Pharmacol. 2016, 7, 398. [Google Scholar] [CrossRef] [PubMed]
- Toral, P.G.; Hervas, G.; Belenguer, A.; Carreno, D.; Frutos, P. mRNA abundance of genes involved in mammary lipogenesis during fish oil- or trans-10,cis-12 CLA-induced milk fat depression in dairy ewes. J. Dairy Sci. 2017, 100, 3182–3192. [Google Scholar] [CrossRef] [PubMed]
- Vallee Marcotte, B.; Guenard, F.; Cormier, H.; Lemieux, S.; Couture, P.; Rudkowska, I.; Vohl, M.C. Plasma Triglyceride Levels May Be Modulated by Gene Expression of IQCJ, NXPH1, PHF17 and MYB in Humans. Int. J. Mol. Sci. 2017, 18, 257. [Google Scholar] [CrossRef] [PubMed]
- Toral, P.G.; Hervas, G.; Suarez-Vega, A.; Arranz, J.J.; Frutos, P. Isolation of RNA from milk somatic cells as an alternative to biopsies of mammary tissue for nutrigenomic studies in dairy ewes. J. Dairy Sci. 2016, 99, 8461–8471. [Google Scholar] [CrossRef] [PubMed]
- Luge, T.; Fischer, C.; Sauer, S. Efficient Application of De Novo RNA Assemblers for Proteomics Informed by Transcriptomics. J. Proteome Res. 2016, 15, 3938–3943. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Qian, K.; von Toerne, C.; Hoerburger, L.; Claussnitzer, M.; Hoffmann, C.; Glunk, V.; Wahl, S.; Breier, M.; Eck, F.; et al. Allele-specific quantitative proteomics unravels molecular mechanisms modulated by cis-regulatory PPARG locus variation. Nucleic Acids Res. 2017, 45, 3266–3279. [Google Scholar] [CrossRef] [PubMed]
- Bekiares, N.; Krueger, C.G.; Meudt, J.J.; Shanmuganayagam, D.; Reed, J.D. Effect of Sweetened Dried Cranberry Consumption on Urinary Proteome and Fecal Microbiome in Healthy Human Subjects. Omics J. Integr. Biol. 2018, 22, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Meplan, C.; Johnson, I.T.; Polley, A.C.; Cockell, S.; Bradburn, D.M.; Commane, D.M.; Arasaradnam, R.P.; Mulholland, F.; Zupanic, A.; Mathers, J.C.; et al. Transcriptomics and proteomics show that selenium affects inflammation, cytoskeleton, and cancer pathways in human rectal biopsies. FASEB J. 2016, 30, 2812–2825. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Garcia, S.; Arola, L.; Pascual-Serrano, A.; Arola-Arnal, A.; Aragones, G.; Blade, C.; Suarez, M. Development and validation of a UHPLC-ESI-MS/MS method for the simultaneous quantification of mammal lysophosphatidylcholines and lysophosphatidylethanolamines in serum. J. Chromatogr. B 2017, 1055–1056, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Heianza, Y.; Qi, L. Gene-Diet Interaction and Precision Nutrition in Obesity. Int. J. Mol. Sci. 2017, 18, 787. [Google Scholar] [CrossRef] [PubMed]
- Kakkoura, M.G.; Sokratous, K.; Demetriou, C.A.; Loizidou, M.A.; Loucaides, G.; Kakouri, E.; Hadjisavvas, A.; Kyriacou, K. Mediterranean diet-gene interactions: A targeted metabolomics study in Greek-Cypriot women. Mol. Nutr. Food Res. 2017, 61, 1600558. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Garcia, S.; Del Bas, J.M.; Caimari, A.; Escorihuela, R.M.; Arola, L.; Suarez, M. Impact of a cafeteria diet and daily physical training on the rat serum metabolome. PLoS ONE 2017, 12, e0171970. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.H.; Kim, M.; Kim, M.; Kang, M.; Yoo, H.J.; Lee, N.H.; Kim, Y.H.; Song, M.; Lee, J.H. Metabolites distinguishing visceral fat obesity and atherogenic traits in individuals with overweight. Obesity 2017, 25, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Cojocneanu-Petric, R.; Chira, S.; Truta, A.; Floares, A.; Petrut, B.; Achimas-Cadariu, P.; Berindan-Neagoe, I. Clinical and pathological implications of miRNA in bladder cancer. Int. J. Nanomed. 2015, 10, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Calin, G.A.; Berindan-Neagoe, I. MicroRNAs and cancer therapy - from bystanders to major players. Curr. Med. Chem. 2013, 20, 3561–3573. [Google Scholar] [CrossRef] [PubMed]
- Verrax, J.; Calderon, P.B. The controversial place of vitamin C in cancer treatment. Biochem. Pharmacol. 2008, 76, 1644–1652. [Google Scholar] [CrossRef] [PubMed]
- Mutch, D.M.; Wahli, W.; Williamson, G. Nutrigenomics and nutrigenetics: The emerging faces of nutrition. FASEB J. 2005, 19, 1602–1616. [Google Scholar] [CrossRef] [PubMed]
- Ordovas, J.M.; Mooser, V. Nutrigenomics and nutrigenetics. Curr. Opinion Lipidol. 2004, 15, 101–108. [Google Scholar] [CrossRef]
- Braicu, C.; Selicean, S.; Cojocneanu-Petric, R.; Lajos, R.; Balacescu, O.; Taranu, I.; Marin, D.E.; Motiu, M.; Jurj, A.; Achimas-Cadariu, P.; et al. Evaluation of cellular and molecular impact of zearalenone and Escherichia coli co-exposure on IPEC-1 cells using microarray technology. BMC Genom. 2016, 17, 576. [Google Scholar] [CrossRef] [PubMed]
- Riscuta, G. Nutrigenomics at the Interface of Aging, Lifespan, and Cancer Prevention. J. Nutr. 2016, 146, 1931–1939. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Catana, C.; Calin, G.A.; Berindan-Neagoe, I. NCRNA combined therapy as future treatment option for cancer. Curr. Pharm. Des. 2014, 20, 6565–6574. [Google Scholar] [CrossRef] [PubMed]
- Mallery, S.R.; Wang, D.; Santiago, B.; Pei, P.; Schwendeman, S.P.; Nieto, K.; Spinney, R.; Tong, M.; Koutras, G.; Han, B.; et al. Benefits of Multifaceted Chemopreventives in the Suppression of the Oral Squamous Cell Carcinoma (OSCC) Tumorigenic Phenotype. Cancer Prev. Res. 2017, 10, 76–88. [Google Scholar] [CrossRef]
- Mangalath, U.; Aslam, S.A.; Abdul Khadar, A.H.K.; Francis, P.G.; Mikacha, M.S.K.; Kalathingal, J.H. Recent trends in prevention of oral cancer. J. Int. Soc. Prev. Community Dent. 2014, 4, S131–S138. [Google Scholar] [CrossRef]
- Sanchez, M.J.; Martinez, C.; Nieto, A.; Castellsague, X.; Quintana, M.J.; Bosch, F.X.; Munoz, N.; Herrero, R.; Franceschi, S. Oral and oropharyngeal cancer in Spain: Influence of dietary patterns. Eur. J. Cancer Prev. 2003, 12, 49–56. [Google Scholar] [CrossRef]
- Chen, Q.; Polireddy, K.; Chen, P.; Dong, R. The unpaved journey of vitamin C in cancer treatment. Can. J. Physiol. Pharmacol. 2015, 93, 1055–1063. [Google Scholar] [CrossRef]
- Gulei, D.; Petrut, B.; Tigu, A.B.; Onaciu, A.; Fischer-Fodor, E.; Atanasov, A.G.; Ionescu, C.; Berindan-Neagoe, I. Exosomes at a glance—Common nominators for cancer hallmarks and novel diagnosis tools. Crit. Rev. Biochem. Mol. Boil. 2018, 53, 564–577. [Google Scholar] [CrossRef]
- Fortmann, S.P.; Burda, B.U.; Senger, C.A.; Lin, J.S.; Beil, T.L.; O’Connor, E.; Whitlock, E.P. U.S. Preventive Services Task Force Evidence Syntheses, formerly Systematic Evidence Reviews. In Vitamin, Mineral, and Multivitamin Supplements for the Primary Prevention of Cardiovascular Disease and Cancer: A Systematic Evidence Review for the U.S. Preventive Services Task Force; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2013. [Google Scholar]
- Juneja, S.; Rathore, A.S.; Sharma, K.; Shetty, D.; Jain, A. Antioxidant-Oxidant Index as a Biomarker in Oral Potentially Malignant Disorders and Oral Squamous Cell Carcinoma: A Biochemical Study. J. Clin. Diagn. Res. 2017, 11, ZC05–ZC08. [Google Scholar] [CrossRef]
- Lv, W.; Zhong, X.; Xu, L.; Han, W. Association between Dietary Vitamin A Intake and the Risk of Glioma: Evidence from a Meta-analysis. Nutrients 2015, 7, 8897–8904. [Google Scholar] [CrossRef] [PubMed]
- Al Tanoury, Z.; Piskunov, A.; Rochette-Egly, C. Vitamin A and retinoid signaling: Genomic and nongenomic effects. J. Lipid Res. 2013, 54, 1761–1775. [Google Scholar] [CrossRef] [PubMed]
- Mustafi, S.; Camarena, V.; Volmar, C.H.; Huff, T.C.; Sant, D.W.; Brothers, S.P.; Liu, Z.J.; Wahlestedt, C.; Wang, G. Vitamin C Sensitizes Melanoma to BET Inhibitors. Cancer Res. 2018, 78, 572–583. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, A.; Takatori, A.; Hossain, M.S.; Hasan, M.K.; Tagawa, M.; Nagase, H.; Nakagawara, A. Unfavorable neuroblastoma prognostic factor NLRR2 inhibits cell differentiation by transcriptional induction through JNK pathway. Cancer Sci. 2016, 107, 1223–1232. [Google Scholar] [CrossRef] [PubMed]
- Al-Asmari, A.K.; Khan, A.Q.; Al-Asmari, S.A.; Al-Rawi, A.; Al-Omani, S. Alleviation of 5-fluorouracil-induced intestinal mucositis in rats by vitamin E via targeting oxidative stress and inflammatory markers. J. Complement. Integr. Med. 2016, 13, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Michels, A.J.; Hagen, T.M.; Frei, B. Human Genetic Variation Influences Vitamin C Homeostasis by Altering Vitamin C Transport and Antioxidant Enzyme Function. Annu. Rev. Nutr. 2013, 33, 45–70. [Google Scholar] [CrossRef]
- Carr, A.C.; McCall, C. The role of vitamin C in the treatment of pain: New insights. J. Transl. Med. 2017, 15, 77. [Google Scholar] [CrossRef]
- Yang, G.; Yan, Y.; Ma, Y.; Yang, Y. Vitamin C at high concentrations induces cytotoxicity in malignant melanoma but promotes tumor growth at low concentrations. Mol. Carcinog. 2017, 56, 1965–1976. [Google Scholar] [CrossRef]
- Chung, M.K.; Kim do, H.; Ahn, Y.C.; Choi, J.Y.; Kim, E.H.; Son, Y.I. Randomized Trial of Vitamin C/E Complex for Prevention of Radiation-Induced Xerostomia in Patients with Head and Neck Cancer. Otolaryngol. Head Neck Surg. 2016, 155, 423–430. [Google Scholar] [CrossRef]
- Ohwada, R.; Ozeki, Y.; Saitoh, Y. High-dose ascorbic acid induces carcinostatic effects through hydrogen peroxide and superoxide anion radical generation-induced cell death and growth arrest in human tongue carcinoma cells. Free. Radic. Res. 2017, 51, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Kuppusamy, P.; Yusoff, M.M.; Maniam, G.P.; Ichwan, S.J.; Soundharrajan, I.; Govindan, N. Nutraceuticals as potential therapeutic agents for colon cancer: A review. Acta Pharm. Sin. B 2014, 4, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, C.; Seuter, S. A genomic perspective on vitamin D signaling. Anticancer. Res. 2009, 29, 3485–3493. [Google Scholar] [PubMed]
- Cai, X.; Peng, D.; Wei, H.; Yang, X.; Huang, Q.; Lin, Z.; Xu, W.; Qian, M.; Yang, C.; Liu, T.; et al. Mir-215 suppresses proliferation and migration of non-small cell lung cancer cells. Oncol. Lett. 2017, 13, 2349–2353. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Layne, T.M.; Weinstein, S.J.; Graubard, B.I.; Ma, X.; Mayne, S.T.; Albanes, D. Serum 25-hydroxyvitamin D, vitamin D binding protein, and prostate cancer risk in black men. Cancer 2017, 123, 2698–2704. [Google Scholar] [CrossRef] [PubMed]
- Tsavachidou, D.; McDonnell, T.J.; Wen, S.; Wang, X.; Vakar-Lopez, F.; Pisters, L.L.; Pettaway, C.A.; Wood, C.G.; Do, K.-A.; Thall, P.F.; et al. Selenium and Vitamin E: Cell Type- and Intervention-Specific Tissue Effects in Prostate Cancer. J. Natl. Cancer Inst. 2009, 101, 306–320. [Google Scholar] [CrossRef] [PubMed]
- Dandawate, P.R.; Subramaniam, D.; Jensen, R.A.; Anant, S. Targeting cancer stem cells and signaling pathways by phytochemicals: Novel approach for breast cancer therapy. Semin. Cancer Boil. 2016, 40–41, 192–208. [Google Scholar] [CrossRef]
- Major, J.M.; Yu, K.; Wheeler, W.; Zhang, H.; Cornelis, M.C.; Wright, M.E.; Yeager, M.; Snyder, K.; Weinstein, S.J.; Mondul, A.; et al. Genome-wide association study identifies common variants associated with circulating vitamin E levels. Hum. Mol. Genet. 2011, 20, 3876–3883. [Google Scholar] [CrossRef]
- Lance, P.; Alberts, D.S.; Thompson, P.A.; Fales, L.; Wang, F.; San Jose, J.; Jacobs, E.T.; Goodman, P.J.; Darke, A.K.; Yee, M.; et al. Colorectal Adenomas in Participants of the SELECT Randomized Trial of Selenium and Vitamin E for Prostate Cancer Prevention. Cancer Prev. Res. 2017, 10, 45–54. [Google Scholar] [CrossRef]
- Ulrich, C.M. Folate and cancer prevention: A closer look at a complex picture. Am. J. Clin. Nutr. 2007, 86, 271–273. [Google Scholar] [CrossRef]
- Duthie, S.J. Folate and cancer: How DNA damage, repair and methylation impact on colon carcinogenesis. J. Inherit. Metab. Dis. 2011, 34, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Kok, D.E.G.; Dhonukshe-Rutten, R.A.M.; Lute, C.; Heil, S.G.; Uitterlinden, A.G.; van der Velde, N.; van Meurs, J.B.J.; van Schoor, N.M.; Hooiveld, G.J.E.J.; de Groot, L.C.P.G.M.; et al. The effects of long-term daily folic acid and vitamin B(12) supplementation on genome-wide DNA methylation in elderly subjects. Clin. Epigenet. 2015, 7, 121. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Wang, C.; Yu, W.; Fan, W.; Wang, S.; Shen, N.; Wu, P.; Li, X.; Wang, F. Selenium Exposure and Cancer Risk: An Updated Meta-analysis and Meta-regression. Sci. Rep. 2016, 6, 19213. [Google Scholar] [CrossRef] [PubMed]
- Xiang, N.; Zhao, R.; Song, G.; Zhong, W. Selenite reactivates silenced genes by modifying DNA methylation and histones in prostate cancer cells. Carcinogenesis 2008, 29, 2175–2181. [Google Scholar] [CrossRef]
- Kurokawa, S.; Berry, M.J. Selenium. Role of the essential metalloid in health. Metal Ions Life Sci. 2013, 13, 499–534. [Google Scholar]
- Ferguson, L.R.; Karunasinghe, N.; Zhu, S.; Wang, A.H. Selenium and its’ role in the maintenance of genomic stability. Mutat. Res. Mol. Mech. Mutagen. 2012, 733, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Azrad, M.; Turgeon, C.; Demark-Wahnefried, W. Current evidence linking polyunsaturated Fatty acids with cancer risk and progression. Front. Oncol. 2013, 3, 224. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.X. Nutrigenomics and cancer therapy. J. Nutr. Nutr. 2013, 6, I–II. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, K.; Monsen, V.T.; Hakvag Pettersen, C.H.; Overland, H.B.; Pettersen, G.; Samdal, H.; Tesfahun, A.N.; Lundemo, A.G.; Bjorkoy, G.; Schonberg, S.A. DHA-induced stress response in human colon cancer cells - Focus on oxidative stress and autophagy. Free. Radic. Boil. Med. 2016, 90, 158–172. [Google Scholar] [CrossRef]
- Chen, X.N.; Wang, K.F.; Xu, Z.Q.; Li, S.J.; Liu, Q.; Fu, D.H.; Wang, X.; Wu, B. MiR-133b regulates bladder cancer cell proliferation and apoptosis by targeting Bcl-w and Akt1. Cancer Cell Int. 2014, 14, 70. [Google Scholar] [CrossRef]
- Park, Y.; Brinton, L.A.; Subar, A.F.; Hollenbeck, A.; Schatzkin, A. Dietary fiber intake and risk of breast cancer in postmenopausal women: The National Institutes of Health–AARP Diet and Health Study. Am. J. Clin. Nutr. 2009, 90, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.Y.; He, K.; Wang, P.; Qin, L.Q. Dietary fiber intake and risk of breast cancer: A meta-analysis of prospective cohort studies. Am. J. Clin. Nutr. 2011, 94, 900–905. [Google Scholar] [CrossRef] [PubMed]
- Levi, F.; Pasche, C.; Lucchini, F.; La Vecchia, C. Dietary fibre and the risk of colorectal cancer. Eur. J. Cancer 2001, 37, 2091–2096. [Google Scholar] [CrossRef]
- Bravi, F.; Scotti, L.; Bosetti, C.; Bertuccio, P.; Negri, E.; La Vecchia, C. Dietary fiber and stomach cancer risk: A case-control study from Italy. Cancer Causes Control. 2009, 20, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Pelucchi, C.; La Vecchia, C.; Chatenoud, L.; Negri, E.; Conti, E.; Montella, M.; Calza, S.; Dal Maso, L.; Franceschi, S. Dietary fibres and ovarian cancer risk. Eur. J. Cancer 2001, 37, 2235–2239. [Google Scholar] [CrossRef]
- Lopez-Legarrea, P.; Fuller, N.R.; Zulet, M.A.; Martinez, J.A.; Caterson, I.D. The influence of Mediterranean, carbohydrate and high protein diets on gut microbiota composition in the treatment of obesity and associated inflammatory state. Asia Pac. J. Clin. Nutr. 2014, 23, 360–368. [Google Scholar]
- Narita, S.; Inoue, M.; Saito, E.; Abe, S.K.; Sawada, N.; Ishihara, J.; Iwasaki, M.; Yamaji, T.; Shimazu, T.; Sasazuki, S.; et al. Dietary fiber intake and risk of breast cancer defined by estrogen and progesterone receptor status: The Japan Public Health Center-based Prospective Study. Cancer Causes Control. 2017, 28, 569–578. [Google Scholar] [CrossRef]
- Lei, L.; Chen, C.; Zhao, J.; Wang, H.; Guo, M.; Zhou, Y.; Luo, J.; Zhang, J.; Xu, L. Targeted Expression of miR-7 Operated by TTF-1 Promoter Inhibited the Growth of Human Lung Cancer through the NDUFA4 Pathway. Mol. Ther.-Nucleic Acids 2017, 6, 183–197. [Google Scholar] [CrossRef]
- Lim, H.S.; Shin, E.J.; Yeom, J.W.; Park, Y.H.; Kim, S.K. Association between Nutrient Intake and Metabolic Syndrome in Patients with Colorectal Cancer. Clin. Nutr. Res. 2017, 6, 38–46. [Google Scholar] [CrossRef]
- Kumar, M.; Kumar, A.; Nagpal, R.; Mohania, D.; Behare, P.; Verma, V.; Kumar, P.; Poddar, D.; Aggarwal, P.K.; Henry, C.J.; et al. Cancer-preventing attributes of probiotics: An update. Int. J. Food Sci. Nutr. 2010, 61, 473–496. [Google Scholar] [CrossRef]
- Tasse, L.; Bercovici, J.; Pizzut-Serin, S.; Robe, P.; Tap, J.; Klopp, C.; Cantarel, B.L.; Coutinho, P.M.; Henrissat, B.; Leclerc, M.; et al. Functional metagenomics to mine the human gut microbiome for dietary fiber catabolic enzymes. Genome Res. 2010, 20, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Dos Reis, S.A.; da Conceicao, L.L.; Siqueira, N.P.; Rosa, D.D.; da Silva, L.L.; Peluzio, M.D. Review of the mechanisms of probiotic actions in the prevention of colorectal cancer. Nutr. Res. 2017, 37, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Park, K.I.; Kim, D.G.; Lee, B.H.; Ma, J.Y. Fermented Herbal Formulas KIOM-MA128 Ameliorate IL-6-Induced Intestinal Barrier Dysfunction in Colon Cancer Cell Line. Mediat. Inflamm. 2016, 2016, 6189590. [Google Scholar] [CrossRef] [PubMed]
- Del Carmen, S.; de Moreno de LeBlanc, A.; Levit, R.; Azevedo, V.; Langella, P.; Bermudez-Humaran, L.G.; LeBlanc, J.G. Anti-cancer effect of lactic acid bacteria expressing antioxidant enzymes or IL-10 in a colorectal cancer mouse model. Int. Immunopharmacol. 2017, 42, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, M.C.; Byrne, E.M.; Esko, T.; Nalls, M.A.; Ganna, A.; Paynter, N.; Monda, K.L.; Amin, N.; Fischer, K.; Renstrom, F.; et al. Genome-wide meta-analysis identifies six novel loci associated with habitual coffee consumption. Mol. Psychiatry 2015, 20, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Mastrangelo, D.; Pelosi, E.; Castelli, G.; Lo-Coco, F.; Testa, U. Mechanisms of anti-cancer effects of ascorbate: Cytotoxic activity and epigenetic modulation. Blood Cells Mol Dis. 2018, 69, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Nasu, K.; Kawano, Y.; Tsukamoto, Y.; Takano, M.; Takai, N.; Li, H.; Furukawa, Y.; Abe, W.; Moriyama, M.; Narahara, H. Aberrant DNA methylation status of endometriosis: Epigenetics as the pathogenesis, biomarker and therapeutic target. J. Obstet. Gynaecol. Res. 2011, 37, 683–695. [Google Scholar] [CrossRef]
- Baek, M.W.; Cho, H.S.; Kim, S.H.; Kim, W.J.; Jung, J.Y. Ascorbic Acid Induces Necrosis in Human Laryngeal Squamous Cell Carcinoma via ROS, PKC, and Calcium Signaling. J. Cell. Physiol. 2017, 232, 417–425. [Google Scholar] [CrossRef]
- Li, Y.; Schellhorn, H.E. New developments and novel therapeutic perspectives for vitamin C. J. Nutr. 2007, 137, 2171–2184. [Google Scholar] [CrossRef]
- Chen, Q.; Espey, M.G.; Sun, A.Y.; Pooput, C.; Kirk, K.L.; Krishna, M.C.; Khosh, D.B.; Drisko, J.; Levine, M. Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proc. Natl. Acad. Sci. USA 2008, 105, 11105–11109. [Google Scholar] [CrossRef]
- Azad, N.; Iyer, A.; Vallyathan, V.; Wang, L.; Castranova, V.; Stehlik, C.; Rojanasakul, Y. Role of oxidative/nitrosative stress-mediated Bcl-2 regulation in apoptosis and malignant transformation. Ann. NY Acad. Sci. 2010, 1203, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hecht, S.S. Approaches to cancer prevention based on an understanding of N-nitrosamine carcinogenesis. Exp. Boil. Med. 1997, 216, 181–191. [Google Scholar] [CrossRef]
- Carr, A.C.; Frei, B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am. J. Clin. Nutr. 1999, 69, 1086–1107. [Google Scholar] [CrossRef] [PubMed]
- Jacob, R.A.; Sotoudeh, G. Vitamin C function and status in chronic disease. Nutr. Clin. Care 2002, 5, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Harris, H.R.; Orsini, N.; Wolk, A. Vitamin C and survival among women with breast cancer: A meta-analysis. Eur. J. Cancer 2014, 50, 1223–1231. [Google Scholar] [CrossRef] [PubMed]
- Guerriero, E.; Sorice, A.; Capone, F.; Napolitano, V.; Colonna, G.; Storti, G.; Castello, G.; Costantini, S. Vitamin C effect on mitoxantrone-induced cytotoxicity in human breast cancer cell lines. PLoS ONE 2014, 9, e115287. [Google Scholar] [CrossRef] [PubMed]
- Kune, G.A.; Kune, S.; Field, B.; Watson, L.F.; Cleland, H.; Merenstein, D.; Vitetta, L. Oral and pharyngeal cancer, diet, smoking, alcohol, and serum vitamin A and beta-carotene levels: A case-control study in men. Nutr. Cancer 1993, 20, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Chi, A.C.; Day, T.A.; Neville, B.W. Oral cavity and oropharyngeal squamous cell carcinoma—An update. CA A Cancer J. Clin. 2015, 65, 401–421. [Google Scholar] [CrossRef]
- Saman, D.M. A review of the epidemiology of oral and pharyngeal carcinoma: Update. Head Neck Oncol. 2012, 4, 1. [Google Scholar] [CrossRef]
- Alizadeh, F.; Bolhassani, A.; Khavari, A.; Bathaie, S.Z.; Naji, T.; Bidgoli, S.A. Retinoids and their biological effects against cancer. Int. Immunopharmacol. 2014, 18, 43–49. [Google Scholar] [CrossRef]
- Chaitanya, N.C.; Muthukrishnan, A.; Babu, D.B.G.; Kumari, C.S.; Lakshmi, M.A.; Palat, G.; Alam, K.S. Role of Vitamin E and Vitamin A in Oral Mucositis Induced by Cancer Chemo/Radiotherapy—A Meta-analysis. J. Clin. Diagn. Res. 2017, 11, ZE06–ZE09. [Google Scholar] [CrossRef] [PubMed]
- Contreras Vidaurre, E.G.; Bagan Sebastian, J.V.; Gavalda, C.; Torres Cifuentes, E.F. Retinoids: Application in premalignant lesions and oral cancer. Med. Oral 2001, 6, 114–123. [Google Scholar] [PubMed]
- Lodi, G.; Franchini, R.; Warnakulasuriya, S.; Varoni, E.M.; Sardella, A.; Kerr, A.R.; Carrassi, A.; MacDonald, L.C.; Worthington, H.V. Interventions for treating oral leukoplakia to prevent oral cancer. Cochrane Database Syst. Rev. 2016, 7, CD001829. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.H.; Osei-Sarfo, K.; Urvalek, A.M.; Zhang, T.; Scognamiglio, T.; Gudas, L.J. Combination of bexarotene and the retinoid CD1530 reduces murine oral-cavity carcinogenesis induced by the carcinogen 4-nitroquinoline 1-oxide. Proc. Natl. Acad. Sci. USA 2014, 111, 8907–8912. [Google Scholar] [CrossRef] [PubMed]
- Li, H.X.; Zhao, W.; Shi, Y.; Li, Y.N.; Zhang, L.S.; Zhang, H.Q.; Wang, D. Retinoic acid amide inhibits JAK/STAT pathway in lung cancer which leads to apoptosis. Tumor Boil. 2015, 36, 8671–8678. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Dai, Y.; Huang, Y.; Chen, X.; Wang, H.; Hong, Y.; Xia, J.; Cheng, B. All-trans retinoic acid restores gap junctional intercellular communication between oral cancer cells with upregulation of Cx32 and Cx43 expressions in vitro. Med. Oral Patol. Oral Cir. Bucal 2013, 18, e569–e577. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Zhang, Z.; Qing, X.; Wang, X.; Liang, C.; Liu, D. Promoter methylation of APC and RAR-beta genes as prognostic markers in non-small cell lung cancer (NSCLC). Exp. Mol. Pathol. 2016, 100, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Deng, J.; Wang, S.S.; Ma, L.; Pei, J.; Zeng, X.X.; Tang, J.X. Association of methylation of the RAR-beta gene with cigarette smoking in non-small cell lung cancer with Southern-Central Chinese population. Asian Pac. J. Cancer Prev. APJCP 2014, 15, 10937–10941. [Google Scholar] [CrossRef] [PubMed]
- Jiang, A.; Wang, X.; Shan, X.; Li, Y.; Wang, P.; Jiang, P.; Feng, Q. Curcumin Reactivates Silenced Tumor Suppressor Gene RARbeta by Reducing DNA Methylation. Phytother. Res. PTR 2015, 29, 1237–1245. [Google Scholar] [CrossRef]
- Wang, L.; Chen, G.; Chen, K.; Ren, Y.; Li, H.; Jiang, X.; Jia, L.; Fu, S.; Li, Y.; Liu, X.; et al. Dual targeting of retinoid X receptor and histone deacetylase with DW22 as a novel antitumor approach. Oncotarget 2015, 6, 9740–9755. [Google Scholar] [CrossRef]
- Wang, M.; Li, G.; Yang, Z.; Wang, L.; Zhang, L.; Wang, T.; Zhang, Y.; Zhang, S.; Han, Y.; Jia, L. Uncoupling protein 2 downregulation by hypoxia through repression of peroxisome proliferator-activated receptor gamma promotes chemoresistance of non-small cell lung cancer. Oncotarget 2017, 8, 8083–8094. [Google Scholar] [PubMed]
- Lakshmi, S.P.; Reddy, A.T.; Banno, A.; Reddy, R.C. PPAR Agonists for the Prevention and Treatment of Lung Cancer. PPAR Res. 2017, 2017, 8252796. [Google Scholar] [CrossRef] [PubMed]
- Chuang, C.H.; Yeh, C.L.; Yeh, S.L.; Lin, E.S.; Wang, L.Y.; Wang, Y.H. Quercetin metabolites inhibit MMP-2 expression in A549 lung cancer cells by PPAR-gamma associated mechanisms. J. Nutr. Biochem. 2016, 33, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Gao, Y.; Zhi, X.; Ta, N.; Jiang, H.; Zheng, J. Association between vitamin A, retinol and carotenoid intake and pancreatic cancer risk: Evidence from epidemiologic studies. Sci. Rep. 2016, 6, 38936. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Boeing, H.; Stelmach-Mardas, M.; Gottschald, M.; Dietrich, S.; Hoffmann, G.; Chaimani, A. Dietary Supplements and Risk of Cause-Specific Death, Cardiovascular Disease, and Cancer: A Systematic Review and Meta-Analysis of Primary Prevention Trials. Adv. Nutr. 2017, 8, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Albanes, D.; Heinonen, O.P.; Huttunen, J.K.; Taylor, P.R.; Virtamo, J.; Edwards, B.K.; Haapakoski, J.; Rautalahti, M.; Hartman, A.M.; Palmgren, J.; et al. Effects of alpha-tocopherol and beta-carotene supplements on cancer incidence in the Alpha-Tocopherol Beta-Carotene Cancer Prevention Study. Am. J. Clin. Nutr. 1995, 62, 1427S–1430S. [Google Scholar] [CrossRef] [PubMed]
- Omenn, G.S.; Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Glass, A.; Keogh, J.P.; Meyskens, F.L., Jr.; Valanis, B.; Williams, J.H., Jr.; et al. Risk factors for lung cancer and for intervention effects in CARET, the Beta-Carotene and Retinol Efficacy Trial. J. Natl. Cancer Inst. 1996, 88, 1550–1559. [Google Scholar] [CrossRef] [PubMed]
- Peterlik, M.; Grant, W.B.; Cross, H.S. Calcium, vitamin D and cancer. Anticancer Res. 2009, 29, 3687–3698. [Google Scholar]
- Rossi, M.; McLaughlin, J.K.; Lagiou, P.; Bosetti, C.; Talamini, R.; Lipworth, L.; Giacosa, A.; Montella, M.; Franceschi, S.; Negri, E.; et al. Vitamin D intake and breast cancer risk: A case-control study in Italy. Ann. Oncol. 2009, 20, 374–378. [Google Scholar] [CrossRef]
- Ramagopalan, S.V.; Heger, A.; Berlanga, A.J.; Maugeri, N.J.; Lincoln, M.R.; Burrell, A.; Handunnetthi, L.; Handel, A.E.; Disanto, G.; Orton, S.M.; et al. A ChIP-seq defined genome-wide map of vitamin D receptor binding: Associations with disease and evolution. Genome Res. 2010, 20, 1352–1360. [Google Scholar] [CrossRef]
- Zeljic, K.; Supic, G.; Stamenkovic Radak, M.; Jovic, N.; Kozomara, R.; Magic, Z. Vitamin D receptor, CYP27B1 and CYP24A1 genes polymorphisms association with oral cancer risk and survival. J. Oral Pathol. Med. 2012, 41, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Holick, C.N.; Stanford, J.L.; Kwon, E.M.; Ostrander, E.A.; Nejentsev, S.; Peters, U. Comprehensive association analysis of the vitamin D pathway genes, VDR, CYP27B1, and CYP24A1, in prostate cancer. Cancer Epidemiol. Prev. Biomark. 2007, 16, 1990–1999. [Google Scholar] [CrossRef] [PubMed]
- Reimers, L.L.; Crew, K.D.; Bradshaw, P.T.; Santella, R.M.; Steck, S.E.; Sirosh, I.; Terry, M.B.; Hershman, D.L.; Shane, E.; Cremers, S.; et al. Vitamin D-related gene polymorphisms, plasma 25-hydroxyvitamin D, and breast cancer risk. Cancer Causes Control. 2015, 26, 187–203. [Google Scholar] [CrossRef] [PubMed]
- Trochoutsou, A.I.; Kloukina, V.; Samitas, K.; Xanthou, G. Vitamin-D in the Immune System: Genomic and Non-Genomic Actions. Mini-Reviews Med. Chem. 2015, 15, 953–963. [Google Scholar] [CrossRef]
- Vaughan-Shaw, P.G.; O’Sullivan, F.; Farrington, S.M.; Theodoratou, E.; Campbell, H.; Dunlop, M.G.; Zgaga, L. The impact of vitamin D pathway genetic variation and circulating 25-hydroxyvitamin D on cancer outcome: Systematic review and meta-analysis. Br. J. Cancer 2017, 116, 1092–1110. [Google Scholar] [CrossRef] [PubMed]
- Gewirtz, D.A.; Hilliker, M.L.; Wilson, E.N. Promotion of autophagy as a mechanism for radiation sensitization of breast tumor cells. Radiother. Oncol. 2009, 92, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Bertone-Johnson, E.R. Vitamin D and breast cancer. Ann. Epidemiol. 2009, 19, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Chiang, K.C.; Chen, T.C. Vitamin D for the prevention and treatment of pancreatic cancer. World J. Gastroenterol. 2009, 15, 3349–3354. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.; Cetindis, M.; Biegner, T.; Lehman, M.; Munz, A.; Teriete, P.; Reinert, S. Serum vitamin D levels of patients with oral squamous cell carcinoma (OSCC) and expression of vitamin D receptor in oral precancerous lesions and OSCC. Med. Oral Patol. Oral Cir. Bucal 2015, 20, e188–e195. [Google Scholar] [CrossRef] [PubMed]
- Berger, S.H.; Pittman, D.L.; Wyatt, M.D. Uracil in DNA: Consequences for carcinogenesis and chemotherapy. Biochem. Pharmacol. 2008, 76, 697–706. [Google Scholar] [CrossRef]
- Crider, K.S.; Yang, T.P.; Berry, R.J.; Bailey, L.B. Folate and DNA methylation: A review of molecular mechanisms and the evidence for folate’s role. Adv. Nutr. 2012, 3, 21–38. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.T.; Hsu, S.W.; Tsai, S.J.; Chang, Y.T.; Huang, C.W.; Liu, M.E.; Chen, N.C.; Chang, W.N.; Hsu, J.L.; Lee, C.C.; et al. Genetic effect of MTHFR C677T polymorphism on the structural covariance network and white-matter integrity in Alzheimer’s disease. Hum. Brain Mapp. 2017, 38, 3039–3051. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, M.C.; Yu, P.; Shiao, S.P. MTHFR Gene Polymorphism-Mutations and Air Pollution as Risk Factors for Breast Cancer: A Metaprediction Study. Nurs. Res. 2017, 66, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.C.; Lai, C.Y.; Chang, S.N.; Hsieh, L.L.; Tang, R.; Sung, F.C.; Lin, Y.K. Polymorphisms of MTHFR C677T and A1298C associated with survival in patients with colorectal cancer treated with 5-fluorouracil-based chemotherapy. Int. J. Clin. Oncol. 2017, 22, 484–493. [Google Scholar] [CrossRef] [PubMed]
- Lien, S.A.; Young, L.; Gau, B.S.; SP, K.S. Meta-prediction of MTHFR gene polymorphism-mutations, air pollution, and risks of leukemia among world populations. Oncotarget 2017, 8, 4387–4398. [Google Scholar] [CrossRef] [PubMed]
- Fryar-Williams, S. Fundamental Role of Methylenetetrahydrofolate Reductase 677 C → T Genotype and Flavin Compounds in Biochemical Phenotypes for Schizophrenia and Schizoaffective Psychosis. Front. Psychiatry 2016, 7, 172. [Google Scholar] [CrossRef] [PubMed]
- Kaur, A.; Kaur, A. Maternal MTHFR polymorphism (677 C-T) and risk of Down’s syndrome child: Meta-analysis. J. Genet. 2016, 95, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Carlus, S.J.; Sarkar, S.; Bansal, S.K.; Singh, V.; Singh, K.; Jha, R.K.; Sadasivam, N.; Sadasivam, S.R.; Gireesha, P.S.; Thangaraj, K.; et al. Is MTHFR 677 C>T Polymorphism Clinically Important in Polycystic Ovarian Syndrome (PCOS)? A Case-Control Study, Meta-Analysis and Trial Sequential Analysis. PLoS ONE 2016, 11, e0151510. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, L.J.; Deng, M.Z.; Luo, Y.Y.; Wu, S.; Xiong, L.; Wang, D.; Liu, Y.; Liu, H. MTHFR C677T and A1298C polymorphisms and risk of lung cancer: A comprehensive evaluation. Genet. Mol. Res. 2016, 15, gmr.15027615. [Google Scholar] [CrossRef]
- Wang, X.; Yue, K.; Hao, L. Meta-analysis of methylenetetrahydrofolate reductase polymorphism and lung cancer risk in Chinese. Int. J. Clin. Exp. Med. 2015, 8, 1521–1525. [Google Scholar]
- Liu, N.B.; Li, J.; Qi, J.F.; Zhang, Z.Z.; Wu, X.; Zhang, J.H. Methylenetetrahydrofolate reductase 677TT genotype may be associated with an increased lung cancer risk in North China: An updated meta-analysis. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2014, 20, 2817–2823. [Google Scholar]
- Rai, V. Folate pathway gene MTHFR C677T polymorphism and risk of lung cancer in Asian populations. Asian Pac. J. Cancer Prev. 2014, 15, 9259–9264. [Google Scholar] [CrossRef] [PubMed]
- Fenech, M. The role of folic acid and Vitamin B12 in genomic stability of human cells. Mutat. Res. Mol. Mech. Mutagen. 2001, 475, 57–67. [Google Scholar] [CrossRef]
- Succi, M.; de Castro, T.B.; Galbiatti, A.L.; Arantes, L.M.; da Silva, J.N.; Maniglia, J.V.; Raposo, L.S.; Pavarino, E.C.; Goloni-Bertollo, E.M. DNMT3B C46359T and SHMT1 C1420T polymorphisms in the folate pathway in carcinogenesis of head and neck. Mol. Biol. Rep. 2014, 41, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Tower, R.L.; Spector, L.G. The epidemiology of childhood leukemia with a focus on birth weight and diet. Crit. Rev. Clin. Lab. Sci. 2007, 44, 203–242. [Google Scholar] [CrossRef]
- Xu, X.; Chen, J. One-carbon metabolism and breast cancer: An epidemiological perspective. J. Genet. Genom. 2009, 36, 203–214. [Google Scholar] [CrossRef]
- Kennedy, D.A.; Stern, S.J.; Moretti, M.; Matok, I.; Sarkar, M.; Nickel, C.; Koren, G. Folate intake and the risk of colorectal cancer: A systematic review and meta-analysis. Cancer Epidemiol. 2011, 35, 2–10. [Google Scholar] [CrossRef]
- Collin, S.M.; Metcalfe, C.; Refsum, H.; Lewis, S.J.; Zuccolo, L.; Smith, G.D.; Chen, L.; Harris, R.; Davis, M.; Marsden, G.; et al. Circulating folate, vitamin B12, homocysteine, vitamin B12 transport proteins, and risk of prostate cancer: A case-control study, systematic review, and meta-analysis. Cancer Epidemiol. Biomark. Prev. 2010, 19, 1632–1642. [Google Scholar] [CrossRef]
- Tomuleasa, C.; Braicu, C.; Irimie, A.; Craciun, L.; Berindan-Neagoe, I. Nanopharmacology in translational hematology and oncology. Int. J. Nanomed. 2014, 9, 3465–3479. [Google Scholar]
- Zhao, Q.S.; Hu, L.L.; Wang, Z.D.; Li, Z.P.; Wang, A.W.; Liu, J. Resveratrol-loaded folic acid-grafted dextran stearate submicron particles exhibits enhanced antitumor efficacy in non-small cell lung cancers. Mater. Sci. Eng. C 2017, 72, 185–191. [Google Scholar] [CrossRef]
- Yuan, D.; Xu, J.; Wang, J.; Pan, Y.; Fu, J.; Bai, Y.; Zhang, J.; Shao, C. Extracellular miR-1246 promotes lung cancer cell proliferation and enhances radioresistance by directly targeting DR5. Oncotarget 2016, 7, 32707–32722. [Google Scholar] [CrossRef] [PubMed]
- Dang, W.; Qin, Z.; Fan, S.; Wen, Q.; Lu, Y.; Wang, J.; Zhang, X.; Wei, L.; He, W.; Ye, Q.; et al. miR-1207-5p suppresses lung cancer growth and metastasis by targeting CSF1. Oncotarget 2016, 7, 32421–32432. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.F.; Liu, Y.T.; Lin, X.L.; Fan, Y.Y.; Zhang, X.L.; Xu, C.H.; Chen, Y.M. Folate, vitamin B6, vitamin B12 and methionine intakes and risk for nasopharyngeal carcinoma in Chinese adults: A matched case-control study. Br. J. Nutr. 2016, 115, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Galeone, C.; Edefonti, V.; Parpinel, M.; Leoncini, E.; Matsuo, K.; Talamini, R.; Olshan, A.F.; Zevallos, J.P.; Winn, D.M.; Jayaprakash, V.; et al. Folate intake and the risk of oral cavity and pharyngeal cancer: A pooled analysis within the International Head and Neck Cancer Epidemiology Consortium. Int. J. Cancer 2015, 136, 904–914. [Google Scholar] [CrossRef] [PubMed]
- Dulai, P.S.; Singh, S.; Marquez, E.; Khera, R.; Prokop, L.J.; Limburg, P.J.; Gupta, S.; Murad, M.H. Chemoprevention of colorectal cancer in individuals with previous colorectal neoplasia: Systematic review and network meta-analysis. BMJ 2016, 355, i6188. [Google Scholar] [CrossRef] [PubMed]
- Takagi, Y.; Hosomi, Y.; Nagamata, M.; Watanabe, K.; Takahashi, S.; Nakahara, Y.; Yomota, M.; Sunami, K.; Okuma, Y.; Shimokawa, T.; et al. Phase II study of oral vitamin B12 supplementation as an alternative to intramuscular injection for patients with non-small cell lung cancer undergoing pemetrexed therapy. Cancer Chemother. Pharmacol. 2016, 77, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Cole, B.F.; Baron, J.A.; Sandler, R.S.; Haile, R.W.; Ahnen, D.J.; Bresalier, R.S.; McKeown-Eyssen, G.; Summers, R.W.; Rothstein, R.I.; Burke, C.A.; et al. Folic acid for the prevention of colorectal adenomas: A randomized clinical trial. Jama 2007, 297, 2351–2359. [Google Scholar] [CrossRef]
- Crider, K.S.; Quinlivan, E.P.; Berry, R.J.; Hao, L.; Li, Z.; Maneval, D.; Yang, T.P.; Rasmussen, S.A.; Yang, Q.; Zhu, J.H.; et al. Genomic DNA methylation changes in response to folic acid supplementation in a population-based intervention study among women of reproductive age. PLoS ONE 2011, 6, e28144. [Google Scholar] [CrossRef]
- Castellano, S.; Gladyshev, V.N.; Guigo, R.; Berry, M.J. SelenoDB 1.0: A database of selenoprotein genes, proteins and SECIS elements. Nucleic Acids Res. 2008, 36, D332–D338. [Google Scholar] [CrossRef]
- Mariotti, M.; Guigo, R. Selenoprofiles: Profile-based scanning of eukaryotic genome sequences for selenoprotein genes. Bioinformatics 2010, 26, 2656–2663. [Google Scholar] [CrossRef]
- Meplan, C. Association of Single Nucleotide Polymorphisms in Selenoprotein Genes with Cancer Risk. Methods Mol. Biol. 2018, 1661, 313–324. [Google Scholar] [PubMed]
- Pelosof, L.; Yerram, S.; Armstrong, T.; Chu, N.; Danilova, L.; Yanagisawa, B.; Hidalgo, M.; Azad, N.; Herman, J.G. GPX3 Promoter Methylation Predicts Platinum Sensitivity in Colorectal Cancer. Epigenetics 2016, 12, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.J.; Ping, C.; Tang, J.; Zhang, W. MicroRNA-455 suppresses non-small cell lung cancer through targeting ZEB1. Cell Boil. Int. 2016, 40, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Chen, C.; Qu, Y.; Xiang, H.; Ai, Q.; Yang, F.; Tan, X.; Zhou, Y.; Jiang, G.; Zhang, Z. Selenium-binding protein 1 in head and neck cancer is low-expression and associates with the prognosis of nasopharyngeal carcinoma. Medicine 2016, 95, e4592. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Li, F.; Younes, M.; Liu, H.; Chen, C.; Yao, Q. Reduced selenium-binding protein 1 in breast cancer correlates with poor survival and resistance to the anti-proliferative effects of selenium. PLoS ONE 2013, 8, e63702. [Google Scholar] [CrossRef] [PubMed]
- Ha, Y.S.; Lee, G.T.; Kim, Y.H.; Kwon, S.Y.; Choi, S.H.; Kim, T.H.; Kwon, T.G.; Yun, S.J.; Kim, I.Y.; Kim, W.J. Decreased selenium-binding protein 1 mRNA expression is associated with poor prognosis in renal cell carcinoma. World J. Surg. Oncol. 2014, 12, 288. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, M.; Filippini, T.; Wise, L.A. Environmental Selenium and Human Health: An Update. Curr Environ Health Rep. 2018, 5, 464–485. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, P.A.; Carlson, B.A.; Anderson, C.B.; Seifried, H.E.; Hatfield, D.L.; Howard, M.T. Dietary Selenium Levels Affect Selenoprotein Expression and Support the Interferon-gamma and IL-6 Immune Response Pathways in Mice. Nutrients 2015, 7, 6529–6549. [Google Scholar] [CrossRef] [PubMed]
- Tobe, R.; Carlson, B.A.; Tsuji, P.A.; Lee, B.J.; Gladyshev, V.N.; Hatfield, D.L. Differences in Redox Regulatory Systems in Human Lung and Liver Tumors Suggest Different Avenues for Therapy. Cancers 2015, 7, 2262–2276. [Google Scholar] [CrossRef] [PubMed]
- Dolinoy, D.C. The agouti mouse model: An epigenetic biosensor for nutritional and environmental alterations on the fetal epigenome. Nutr. Rev. 2008, 66, S7–S11. [Google Scholar] [CrossRef] [PubMed]
- Farhud, D.D.; Zarif Yeganeh, M.; Zarif Yeganeh, M. Nutrigenomics and Nutrigenetics. Iran. J. Public Health 2010, 39, 1–14. [Google Scholar] [PubMed]
- Ordovas, J.M. Genetic interactions with diet influence the risk of cardiovascular disease. Am. J. Clin. Nutr. 2006, 83, 443S–446S. [Google Scholar] [CrossRef] [PubMed]
- Marques-Rocha, J.L.; Garcia-Lacarte, M.; Samblas, M.; Bressan, J.; Martinez, J.A.; Milagro, F.I. Regulatory roles of miR-155 and let-7b on the expression of inflammation-related genes in THP-1 cells: Effects of fatty acids. J. Physiol. Biochem. 2018, 74, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Nie, D.; Wang, Z.; Zhang, Y.; Pang, D.; Ouyang, H.; Li, L.I. Fat-1 gene inhibits human oral squamous carcinoma cell proliferation through downregulation of β-catenin signaling pathways. Exp. Ther. Med. 2016, 11, 191–196. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nikolakopoulou, Z.; Nteliopoulos, G.; Michael-Titus, A.T.; Parkinson, E.K. Omega-3 polyunsaturated fatty acids selectively inhibit growth in neoplastic oral keratinocytes by differentially activating ERK1/2. Carcinogenesis 2013, 34, 2716–2725. [Google Scholar] [CrossRef] [PubMed]
- Zarco, M.F.; Vess, T.J.; Ginsburg, G.S. The oral microbiome in health and disease and the potential impact on personalized dental medicine. Oral Dis. 2012, 18, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Kuper, H.; Adami, H.O.; Trichopoulos, D. Infections as a major preventable cause of human cancer. J. Intern. Med. 2000, 248, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Ng, Q.X.; Loke, W.; Venkatanarayanan, N.; Lim, D.Y.; Soh, A.Y.S.; Yeo, W.S. A Systematic Review of the Role of Prebiotics and Probiotics in Autism Spectrum Disorders. Medicina 2019, 55, 129. [Google Scholar] [CrossRef]
- Le Bars, P.; Matamoros, S.; Montassier, E.; Le Vacon, F.; Potel, G.; Soueidan, A.; Jordana, F.; de La Cochetière, M.-F. The oral cavity microbiota: Between health, oral disease, and cancers of the aerodigestive tract. Can. J. Microbiol. 2017, 63, 475–492. [Google Scholar] [CrossRef]
- Lazar, M.A.; Vodnar, D.; Prodan, D.; Rotaru, H.; Roman, C.R.; Sorcoi, L.A.; Baciut, G.; Campian, R.S. Antibacterial coating on biocomposites for cranio-facial reconstruction. Clujul Med. 2016, 89, 430–434. [Google Scholar] [CrossRef]
- Meurman, J.H. Oral microbiota and cancer. J. Oral Microbiol. 2010, 2, 5195. [Google Scholar] [CrossRef] [PubMed]
- Catana, C.S.; Cristea, V.; Miron, N.; Neagoe, I.B. Is interleukin-17 a proatherogenic biomarker? Roum. Arch. Microbiol. Immunol. 2011, 70, 124–128. [Google Scholar] [PubMed]
- Gata, V.A.; Lisencu, C.I.; Vlad, C.I.; Piciu, D.; Irimie, A.; Achimas-Cadariu, P. Tumor infiltrating lymphocytes as a prognostic factor in malignant melanoma. Review of the literature. J. BUON 2017, 22, 592–598. [Google Scholar] [PubMed]
- Hamaker, B.R.; Tuncil, Y.E. A perspective on the complexity of dietary fiber structures and their potential effect on the gut microbiota. J. Mol. Boil. 2014, 426, 3838–3850. [Google Scholar] [CrossRef] [PubMed]
- Lepage, P.; Leclerc, M.C.; Joossens, M.; Mondot, S.; Blottiere, H.M.; Raes, J.; Ehrlich, D.; Dore, J. A metagenomic insight into our gut’s microbiome. Gut 2013, 62, 146–158. [Google Scholar] [CrossRef] [PubMed]

| Nutrigenetics | Nutrigenomics | Practical Application | Ref. | |
|---|---|---|---|---|
| DNA | Next generation sequencing (NGS), pyrosequencing, nanostring, polymerase chain reaction (PCR)-based methods | Microarray, NGS, nanostring | Methods assessing DNA are more prone to be applied in nutrigenetics, with emphasis on particular mutations or single nucleotide polymorphisms (SNPs) that affect the response to a particular diet. This entails prediction of genotype/mutation patterns caused by the indirect interaction of genes with certain nutrients. | [14,15,16,17,18,19,20,21,22] |
| Coding and non-coding RNA | Next generation sequencing, pyrosequencing, PCR-based methods | Microarray, NGS, nanostring | Methods assessing RNA are more prone to be applied in nutrigenomics, to evaluate the effect on the alteration of coding and non-coding genes of a particular nutrient. This means determining RNA levels from different tissues to observe the effects of nutrients on transcriptomic profile in terms of impact on physiological or pathological status. | [23,24,25,26,27,28] |
| Proteins | Mass spectrometry (MS), high performance liquid chromatography (HPLC), high performance liquid chromatography–tandem mass spectrometry (HPLC/MS), ultra-high performance liquid chromatography–tandem mass spectrometry (UHPLC/MS) | HPLC/MS, UHPLC/MS | Proteomics is also more prone to be found in nutrigenomic studies. Being an extension of transcriptomics, it allows for validating mRNA expression protein levels. | [28,29,30,31] |
| Metabolites | Nuclear magnetic resonance, HPLC/MS, UHPLC/MS | Nuclear magnetic resonance, HPLC/MS, ultra-high performance liquid chromatography (UHPLC) | Giving a complete picture, metabolites are able to be more accurate in predicting the effect of nutrients. Furthermore, they could be used for validation of the other “omics.” | [22,32,33,34,35,36] |
| Nutrient | Cancer type | Expected Outcomes | Genes effected | Comment | Ref. |
|---|---|---|---|---|---|
| Vitamin A | Glioma, lung, colorectal cancer | Pro/anti-oxidant action, cell differentiation and immune response | Expression level and polymorphism of RARs, RXRs, and PPARβ/δ, Akt, Erk, JNK, p38 | Epidemiological data are not consistent | [51,53,54,55,56] |
| Vitamin C | Solid tumors and hematological malignancies | Selective activation of apoptosis and autophagy. Interferes with redox-sensitive transcription factors and associated target molecules. Selective metabolic and genotoxic stress on tumor cells. | Expression level and polymorphism of GLUT, GST, MnSOD, SVCT, Hp | Low toxicity to normal tissues, but with controversial data due to its dual effect as a pro/antioxidant. The molecular mechanism(s) of selective toxicity on tumor cells remains to be deciphered | [40,49,57,58,59,60,61,62] |
| Vitamin D | Colorectal, breast, prostate or pancreatic cancer | Correlated with lower risks of specific cancers. | Expression level and polymorphism of VDR target genes like p21WAF1/CIP TP53, p27, Cyclin C, CYP24 gene | The results of these studies have been inconsistent, possibly because of the challenges in carrying out such studies. | [63,64,65,66] |
| Vitamin E | Prostate, breast colorectal cancer | Reduces unwanted side effect of cytotoxicity by targeting oxidative stress and inflammatory markers | Polymorphism of APOA5, CYP4F2 | This might also have a pro-oxidant effect. | [51,57,67,68,69,70] |
| Folic acid | Gastric colorectal, breast, pancreatic cancer | Carcinogenesis and embryonic development. At low doses, it decreases cancer risk but overdoses might increase cancer risk | Methylation of DIRAS3, ARMC8, NODAL, MTHFR and HOX genes | Dual role: protection early in carcinogenesis and at high doses in late stages of cancer | [71,72,73] |
| Selenium | Prostate, breast, lung, oropharyngeal, colorectal, bladder, skin, leukemias, uterine, ovarian cancers | Antioxidant, reduces cancer risk; restores epigenetic altered events; genomic stability | Expression and polymorphism of GPxsang, TrxRs | Still highly controversial, being tumor specific and dose specific (pro/antioxidant effect) | [63,67,74,75,76,77] |
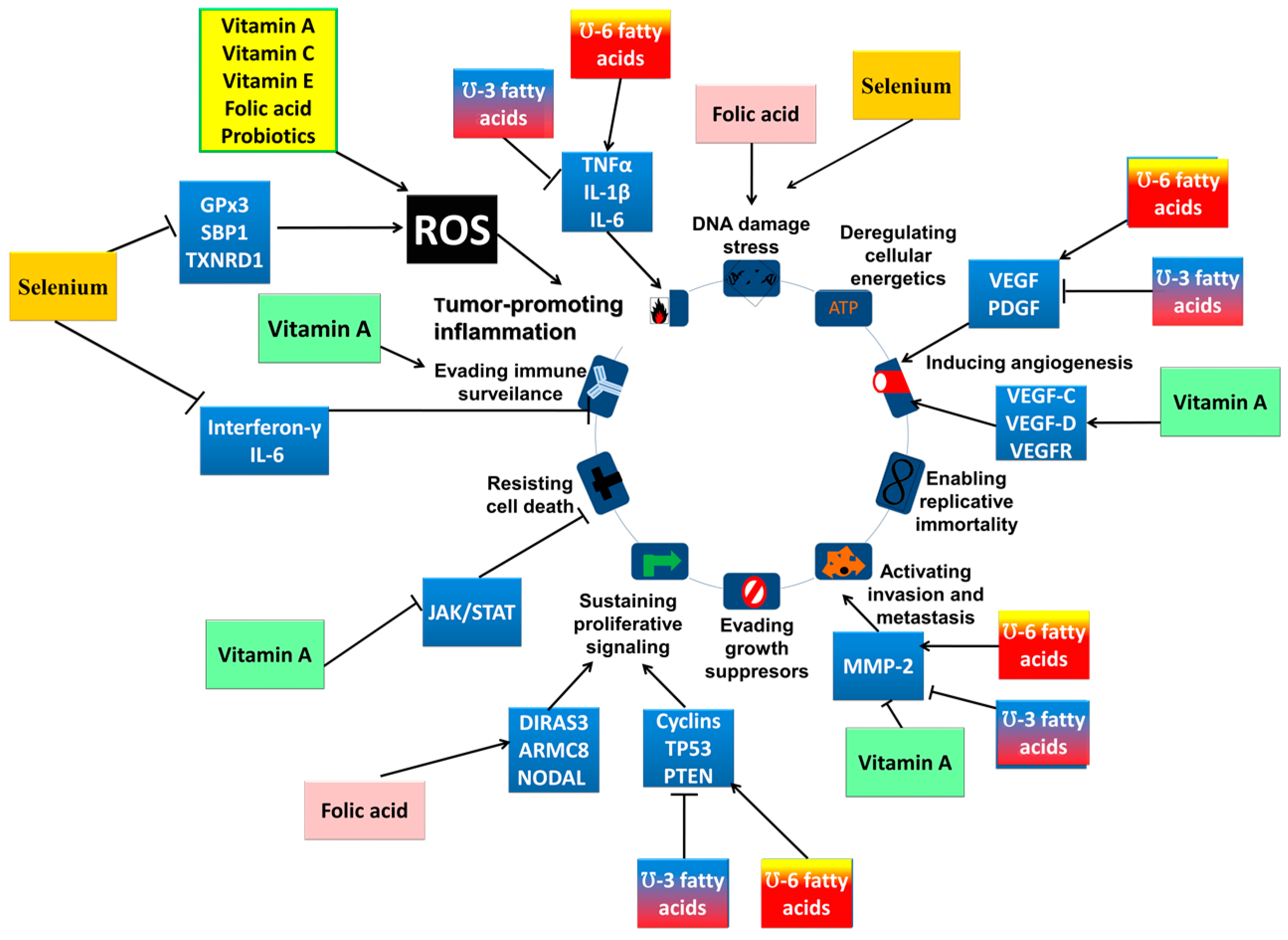
| Polyunsaturated fatty acids (PUFAs) | Breast, colorectal cancer | Regulate cytokine production; stimulate the immune response and enhances apoptosis in cancer cells; regulate cell proliferation and angiogenesis | Transcription factors: PPARs or NFκβ; immune response: TNFα, IL-1β, IL-6; angiogenesis mechanisms: VEGF, PDGF, MMP-2; cell proliferation: cyclins, p53, PTEN | Involved in tumor biology and cancer patients’ prognosis; epidemiologic data furnish inconsistent picture | [63,78,79,80] |
| Dietary fibers | Colorectal, breast, pancreatic, ovarian or stomach cancer | Increased intestinal transit blocking the absorption of external or internal toxic factors | Expression level and polymorphism of CAZymes family | Highly controversial epidemiological data, due to the different types of soluble or insoluble fibers used in studies | [81,82,83,84,85,86,87,88,89,90] |
| Probiotics | Colorectal cancer | Cell-mediated immune responses; increase the activity of antioxidant enzymes | Expression level and polymorphism of CAZymes family | Presently there is no direct evidence in epidemiological data | [87,91,92,93,94,95] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Irimie, A.I.; Braicu, C.; Pasca, S.; Magdo, L.; Gulei, D.; Cojocneanu, R.; Ciocan, C.; Olariu, A.; Coza, O.; Berindan-Neagoe, I. Role of Key Micronutrients from Nutrigenetic and Nutrigenomic Perspectives in Cancer Prevention. Medicina 2019, 55, 283. https://doi.org/10.3390/medicina55060283
Irimie AI, Braicu C, Pasca S, Magdo L, Gulei D, Cojocneanu R, Ciocan C, Olariu A, Coza O, Berindan-Neagoe I. Role of Key Micronutrients from Nutrigenetic and Nutrigenomic Perspectives in Cancer Prevention. Medicina. 2019; 55(6):283. https://doi.org/10.3390/medicina55060283
Chicago/Turabian StyleIrimie, Alexandra Iulia, Cornelia Braicu, Sergiu Pasca, Lorand Magdo, Diana Gulei, Roxana Cojocneanu, Cristina Ciocan, Andrei Olariu, Ovidiu Coza, and Ioana Berindan-Neagoe. 2019. "Role of Key Micronutrients from Nutrigenetic and Nutrigenomic Perspectives in Cancer Prevention" Medicina 55, no. 6: 283. https://doi.org/10.3390/medicina55060283
APA StyleIrimie, A. I., Braicu, C., Pasca, S., Magdo, L., Gulei, D., Cojocneanu, R., Ciocan, C., Olariu, A., Coza, O., & Berindan-Neagoe, I. (2019). Role of Key Micronutrients from Nutrigenetic and Nutrigenomic Perspectives in Cancer Prevention. Medicina, 55(6), 283. https://doi.org/10.3390/medicina55060283









