Depression, Metabolic Syndrome, Serum TSH, and Vitamin D Concentrations in Rural and Urban Postmenopausal Women
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Groups
2.2. Laboratory Blood Tests
2.3. Determining the Metabolic Syndrome
- (1)
- Waist circumference ≥80 cm;
- (2)
- TG ≥150 mg/dL or treatment for dyslipidemia;
- (3)
- HDL <40 mg/dL or treatment for dyslipidemia;
- (4)
- Systolic blood pressure ≥130 mm Hg and/or diastolic blood pressure ≥85 mm Hg or antihypertensive therapy;
- (5)
- Fasting glucose ≥100 mg/dL or hypoglycemic treatment.
2.4. Determining the Severity of Depression
2.5. Statistical Methods
- Pearson’s chi-square test to compare the categorical variables between the rural and the urban residents.
- Student’s t-test to compare the continuous variables between the rural and urban residents, to compare the severity of depression between the women with and without MetS, to compare the severity of depression between the women with the fulfilled criterion of metabolic syndrome and those not fulfilled.
- Pearson’s correlation coefficient to correlate the severity of depression with the serum vitamin D and TSH concentrations, as well as with the number of MetS fulfilled criteria.
- The significance level was assumed to be 0.05.
3. Results
3.1. Characteristics of Study Groups
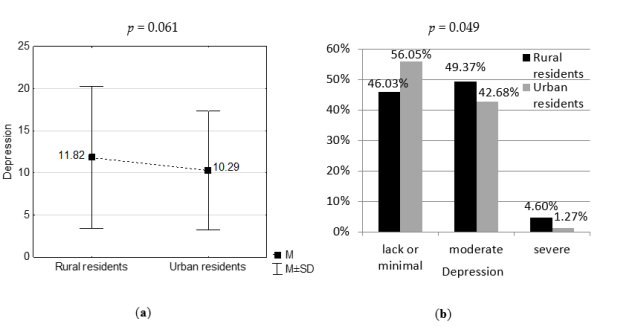
3.2. Severity of Depression in Study Groups
3.3. Serum TSH, Vitamin D Concentrations, and Metabolic Syndrome in Study Groups
3.4. Correlations between Serum TSH, Vitamin D Concentrations, Metabolic Syndrome, and Severity of Depression in Study Groups
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Depression Fact Sheet. Available online: http://www.who.int/mediacentre/factsheets/fs369/en/ (accessed on 8 August 2020).
- Mulhall, S.; Andel, R.; Anstey, K.J. Variation in symptoms of depression and anxiety in midlife women by menopausal status. Maturitas 2018, 108, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J. Vitamin D and Depression: Cellular and Regulatory Mechanisms. Pharmacol. Rev. 2017, 69, 80–92. [Google Scholar] [CrossRef] [PubMed]
- Milaneschi, Y.; Hoogendijk, W.; Lips, P.; Heijboer, A.C.; Schoevers, R.; Hemert, A.M.V.; Beekman, A.T.F.; Smit, J.H.; Penninx, B.W.J.H. The association between low vitamin D and depressive disorders. Mol. Psychiatry 2014, 19, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Anglin, R.E.; Samaan, Z.; Walter, S.D.; McDonald, S.D. Vitamin D deficiency and depression in adults: Systematic review and meta-analysis. Br. J. Psychiatry 2013, 202, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Pan, A.; Lu, L.; Franco, O.H.; Yu, Z.; Li, H.; Lin, X. Association between depressive symptoms and 25-hydroxyvitamin D in middle-aged and elderly Chinese. J. Affect Disord. 2009, 118, 240–243. [Google Scholar] [CrossRef] [PubMed]
- Eyles, D.W.; Smith, S.; Kinobe, R.; Hewison, M.; McGrath, J.J. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J. Chem. Neuroanat. 2005, 9, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Pertile, R.; Liu, P.; Eyles, D.W. Vitamin D regulates tyrosine hydroxylase expression: N-cadherin a possible mediator. Neuroscience 2015, 304, 90–100. [Google Scholar] [CrossRef]
- Delgado, P.L. Depression: The case for a monoamine deficiency. J. Clin. Psychiatry 2000, 61 (Suppl. S6), 7–11. [Google Scholar]
- Talaei, A.; Rafee, N.; Rafei, F.; Chehrei, A. TSH cut off point based on depression in hypothyroid patients. BMC Psychiatry 2017, 17, 327. [Google Scholar] [CrossRef]
- Foltyn, W.; Nowakowska-Zajdel, E.; Danikiewicz, A.; Brodziak, A. Hypothalamic-pituitary-thyroid axis in depression. Psychiatr. Pol. 2002, 36, 281–292. (In Polish) [Google Scholar]
- Du, F.M.; Kuang, H.Y.; Duan, B.H.; Liu, D.N.; Yu, X.Y. Effects of thyroid hormone and depression on common components of central obesity. J. Int. Med. Res. 2019, 47, 3040–3049. [Google Scholar] [CrossRef] [PubMed]
- Olvera, R.L.; Williamson, D.E.; Fisher-Hoch, S.P.; Vatcheva, K.P.; McCormick, J.B. Depression, obesity, and metabolic syndrome: Prevalence and risks of comorbidity in a population-based representative sample of Mexican Americans. J. Clin. Psychiatry 2015, 76, e1300–e1305. [Google Scholar] [CrossRef] [PubMed]
- Mannan, M.; Mamun, A.; Doi, S.; Clavarino, A. Prospective Associations between Depression and Obesity for Adolescent Males and Females- A Systematic Review and Meta-Analysis of Longitudinal Studies. PLoS ONE 2016, 11, e0157240. [Google Scholar] [CrossRef] [PubMed]
- García-Toro, M.; Vicens-Pons, E.; Gili, M.; Roca, M.; Serrano-Ripoll, M.J.; Vives, M.; Leiva, A.; Yáñez, A.M.; Bennasar-Veny, M.; Oliván-Blázquez, B. Obesity, metabolic syndrome and Mediterranean diet: Impact on depression outcome. J. Affect Disord. 2016, 194, 105–108. [Google Scholar] [CrossRef]
- Repousi, N.; Masana, M.F.; Sanchez-Niubo, A.; Haro, J.M.; Tyrovolas, S. Depression and metabolic syndrome in the older population: A review of evidence. J. Affect Disord. 2018, 237, 56–64. [Google Scholar] [CrossRef]
- Ferrari, A.J.; Somerville, A.J.; Baxter, A.J.; Norman, R.; Patten, S.B.; Vos, T.; Whiteford, H.A. Global variation in the prevalence and incidence of major depressive disorder: A systematic review of the epidemiological literature. Psychol. Med. 2013, 43, 71–481. [Google Scholar] [CrossRef]
- Bromberger, J.T.; Schott, L.L.; Kravitz, H.M.; Sowers, M.; Avis, N.E.; Gold, E.B.; Randolph, J.F., Jr.; Matthews, K.A. Longitudinal change in reproductive hormones and depressive symptoms across the menopausal transition: Results from the Study of Women’s Health Across the Nation (SWAN). Arch. Gen. Psychiatry 2010, 67, 598–607. [Google Scholar] [CrossRef]
- Freeman, E.W.; Sammel, M.D.; Boorman, D.W.; Zhang, R. Longitudinal pattern of depressive symptoms around natural menopause. JAMA Psychiatry 2014, 71, 36–43. [Google Scholar] [CrossRef]
- Steinberg, E.M.; Rubinow, D.R.; Bartko, J.J.; Fortinsky, P.M.; Haq, N.; Thompson, K.; Schmidt, P.J. A cross-sectional evaluation of perimenopausal depression. J. Clin. Psychiatry 2008, 69, 973–980. [Google Scholar] [CrossRef]
- Tang, R.; Luo, M.; Li, J.; Peng, Y.; Wang, Y.; Liu, B.; Liu, G.; Wang, Y.; Lin, S.; Chen, R. Symptoms of anxiety and depression among Chinese women transitioning through menopause: Findings from a prospective community-based cohort study. Fertil. Steril. 2019, 112, 1160–1171. [Google Scholar] [CrossRef]
- Bener, A.; Saleh, N.M.; Bakir, A.; Bhugra, D. Depression, Anxiety, and Stress Symptoms in Menopausal Arab Women: Shedding More Light on a Complex Relationship. Ann. Med. Health Sci. Res. 2016, 6, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Malacara, J.M.; Canto de Cetina, T.; Bassol, S.; González, N.; Cacique, L.; Vera-Ramírez, M.L.; Nava, L.E. Symptoms at pre- and postmenopause in rural and urban women from three States of Mexico. Maturitas 2002, 43, 11–19. [Google Scholar] [CrossRef]
- Devi, B.; Karki, P.; Chhetry, R.; Sharma, N.; Niroula, M.; Lepcha, P.C.; Pradhan, S.; Sherpa, Y.; Sherpa, A.; Sharma, P.; et al. Quality of life of post-menopausal women residing in rural and urban areas of Sikkim, India. Int. J. Reprod. Contracept. Obstet. Gynecol. 2018, 7, 5125–5133. [Google Scholar] [CrossRef]
- Nurbazlin, M.; Chee, W.S.; Rokiah, P.; Tan, A.T.; Chew, Y.Y.; Nusaibah, A.R.; Chan, S.P. Effects of sun exposure on 25(OH) vitamin D concentration in urban and rural women in Malaysia. Asia Pac. J. Clin. Nutr. 2013, 22, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Libuda, L.; Laabs, B.H.; Ludwig, C.; Bühlmeier, J.; Antel, J.; Hinney, A.; Naaresh, R.; Föcker, M.; Hebebrand, J.; König, I.R.; et al. Vitamin D and the Risk of Depression: A Causal Relationship? Findings from a Mendelian Randomization Study. Nutrients 2019, 11, 1085. [Google Scholar] [CrossRef] [PubMed]
- Can, M.Ş.; Baykan, H.; Baykan, Ö.; Erensoy, N.; Karlıdere, T. Vitamin D Levels and Vitamin D Receptor Gene Polymorphism in Major Depression. Psychiatr. Danub. 2017, 29, 179–185. [Google Scholar] [CrossRef]
- Gowda, U.; Mutowo, M.P.; Smith, B.J.; Wluka, A.E.; Renzaho, A.M. Vitamin D supplementation to reduce depression in adults: Meta-analysis of randomized controlled trials. Nutrition 2015, 31, 421–429. [Google Scholar] [CrossRef]
- Marsh, W.K.; Penny, J.L.; Rothschild, A.J. Vitamin D supplementation in bipolar depression: A double blind placebo controlled trial. J. Psychiatr. Res. 2017, 95, 48–53. [Google Scholar] [CrossRef]
- Mason, C.; de Dieu Tapsoba, J.; Duggan, C.; Wang, C.Y.; Korde, L.; McTiernan, A. Repletion of vitamin D associated with deterioration of sleep quality among postmenopausal women. Prev. Med. 2016, 93, 166–170. [Google Scholar] [CrossRef]
- Dadoniene, J.; Čypienė, A.; Rinkūnienė, E.; Badariene, J.; Laucevičius, A. Vitamin D, cardiovascular and bone health in postmenopausal women with metabolic syndrome. Adv. Clin. Exp. Med. 2018, 27, 1555–1560. [Google Scholar] [CrossRef]
- Yoo, T.; Choi, W.; Hong, J.H.; Lee, J.Y.; Kim, J.M.; Shin, I.S.; Yang, S.J.; Amminger, P.; Berk, M.; Yoon, J.S.; et al. Association Between Vitamin D Insufficiency and Metabolic Syndrome in Patients With Psychotic Disorders. Psychiatry Investig. 2018, 15, 396–401. [Google Scholar] [CrossRef] [PubMed]
- De Hert, M.; Detraux, J.; Vancampfort, D. The intriguing relationship between coronary heart disease and mental disorders. Dialogues Clin. Neurosci. 2018, 20, 31–40. [Google Scholar] [PubMed]
- Whooley, M.A.; Wong, J.M. Depression and cardiovascular disorders. Annu. Rev. Clin. Psychol. 2013, 9, 327–354. [Google Scholar] [CrossRef] [PubMed]
- Goldbacher, E.M.; Bromberger, J.; Matthews, K.A. Lifetime history of major depression predicts the development of the metabolic syndrome in middle-aged women. Psychosom. Med. 2009, 71, 266–272. [Google Scholar] [CrossRef]
- Vancampfort, D.; Correll, C.U.; Wampers, M.; Sienaert, P.; Mitchell, A.J.; De Herdt, A.; Probst, M.; Scheewe, T.W.; De Hert, M. Metabolic syndrome and metabolic abnormalities in patients with major depressive disorder: A meta-analysis of prevalences and moderating variables. Psychol. Med. 2014, 44, 2017–2028. [Google Scholar] [CrossRef]
- Lichtman, J.H.; Froelicher, E.S.; Blumenthal, J.A.; Carney, R.M.; Doering, L.V.; Frasure-Smith, N.; Freedland, K.E.; Jaffe, A.S.; Leifheit-Limson, E.C.; Sheps, D.S.; et al. American Heart Association Statistics Committee of the Council on Epidemiology and Prevention and the Council on Cardiovascular and Stroke Nursing. Depression as a risk factor for poor prognosis among patients with acute coronary syndrome: Systematic review and recommendations: A scientific statement from the American Heart Association. Circulation 2014, 129, 1350–1369. [Google Scholar] [CrossRef]
- Mohammad, M.Y.H.; Bushulaybi, N.A.; AlHumam, A.S.; AlGhamdi, A.Y.; Aldakhil, H.A.; Alumair, N.A.; Shafey, M.M. Prevalence of depression among hypothyroid patients attending the primary healthcare and endocrine clinics of King Fahad Hospital of the University (KFHU). J. Family Med. Prim. Care 2019, 8, 2708–2713. [Google Scholar] [CrossRef]
- Fischer, S.; Ehlert, U. Hypothalamic-pituitary-thyroid (HPT) axis functioning in anxiety disorders. A systematic review. Depress Anxiety 2018, 35, 98–110. [Google Scholar] [CrossRef]
- Guimarães, J.M.; de Souza Lopes, C.; Baima, J.; Sichieri, R. Depression symptoms and hypothyroidism in a population-based study of middle-aged Brazilian women. J. Affect Disord. 2009, 117, 120–123. [Google Scholar] [CrossRef]
- Loh, H.H.; Lim, L.L.; Yee, A.; Loh, H.S. Association between subclinical hypothyroidism and depression: An updated systematic review and meta-analysis. BMC Psychiatry 2019, 19, 12. [Google Scholar] [CrossRef]
- Prättälä, R.; Paalanen, L.; Grinberga, D.; Helasoja, V.; Kasmel, A.; Petkeviciene, J. Gender differences in the consumption of meat, fruit and vegetables are similar in Finland and the Baltic countries. Eur. J. Public Health 2007, 17, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.C.; Moran, L.J.; Teede, H.J.; Ranasinha, S.; Lombard, C.B.; Harrison, C.L. Exploring Diet Quality between Urban and Rural Dwelling Women of Reproductive Age. Nutrients 2017, 9, 586. [Google Scholar] [CrossRef] [PubMed]
- Nemati, A.; Naghizadeh Baghi, A. Assessment of Nutritional Status in Post Menopausal Women of Ardebil, Iran. J. Biol. Sci. 2008, 8, 196–200. [Google Scholar] [CrossRef][Green Version]

| Parameter | Rural Residents (n = 239) | Urban Residents (n = 157) | Comparison between Rural and Urban | |
|---|---|---|---|---|
| Test 1 | p | |||
| Age (years), M ± SD | 56.82 ± 4.40 | 56.38 ± 3.34 | 1.072 | 0.285 |
| Age at last menstruation (years), M ± SD | 48.95 ± 4.22 | 50.27 ± 4.08 | −3.016 | 0.003 |
| BMI (kg/m2), M ± SD | 28.83 ± 5.01 | 26.36 ± 4.36 | 5.045 | <0.001 |
| normal weight | 56 (23.43) | 65 (41.40) | 21.271 | <0.001 |
| overweight | 96 (40.17) | 64 (40.76) | ||
| obesity | 87 (36.40) | 28 (17.83) | ||
| Level of education, n (%) | ||||
| primary | 27 (11.29) | 1 (0.64) | 168.621 | <0.001 |
| basic vocational | 104 (43.51) | 5 (3.18) | ||
| secondary | 97 (40.59) | 65 (41.40) | ||
| tertiary | 11 (4.60) | 86 (54.78) | ||
| Marital status, n (%) | ||||
| married | 204 (85.36) | 128 (81.53) | 24.504 | <0.001 |
| never married | 3 (1.26) | 7 (4.46) | ||
| divorced | 4 (1.67) | 16 (10.19) | ||
| widowed | 28 (11.72) | 6 (3.82) | ||
| Having children, n (%) | 228 (95.40) | 144 (91.72) | 2.251 | 0.134 |
| Parameter | Rural Residents (n = 239) | Urban Residents (n = 157) | Comparison between Rural and Urban | |
|---|---|---|---|---|
| Test 1 | p | |||
| Vitamin D (ng/mL), M ± SD | 22.79 ± 8.01 | 17.31 ± 8.79 | 6.402 | <0.001 |
| severe deficiency, n (%) | 7 (2.93) | 32 (20.38) | 48.650 | <0.001 |
| moderate deficiency, n (%) | 90 (37.66) | 77 (49.04) | ||
| mild deficiency, n (%) | 95 (39.75) | 32 (20.38) | ||
| optimal, n (%) | 47 (19.67) | 16 (10.19) | ||
| TSH (mIU/L), M ± SD | 1.46 ± 1.28 | 1.55 ± 0.98 | −0.759 | 0.448 |
| below normal, n (%) | 40 (16.74) | 13 (8.28) | 6.832 | 0.033 |
| normal, n (%) | 193 (80.75) | 142 (90.45) | ||
| above normal, n (%) | 6 (2.51) | 2 (1.27) | ||
| Metabolic syndrome, n (%) | 168 (70.29) | 34 (21.66) | 89.695 | <0.001 |
| Number of fulfilled criteria of metabolic syndrome, n (%) | ||||
| 0 | 2 (0.84) | 18 (11.46) | 106.489 | <0.001 |
| 1 | 15 (6.28) | 47 (29.94) | ||
| 2 | 54 (22.59) | 58 (36.94) | ||
| 3 | 110 (46.03) | 25 (15.92) | ||
| 4 | 41 (17.15) | 7 (4.46) | ||
| 5 | 17 (7.11) | 2 (1.27) | ||
| Parameter | Test 1 | Rural Residents (n = 239) | Urban Residents (n = 157) | ||
|---|---|---|---|---|---|
| Result | p | Result | p | ||
| Vitamin D (ng/mL) | r | 0.062 | 0.343 | 0.010 | 0.900 |
| TSH (mIU/L) | r | −0.211 | 0.049 | 0.060 | 0.487 |
| Metabolic syndrome | t | −0.083 | 0.934 | 0.759 | 0.449 |
| Number of fulfilled criteria of metabolic syndrome | r | 0.020 | 0.765 | 0.051 | 0.524 |
| abdominal obesity | t | 0.258 | 0.796 | 0.788 | 0.432 |
| hypertriglyceridaemia | t | −0.423 | 0.671 | 0.273 | 0.785 |
| low HDL-cholesterol | t | −0.459 | 0.647 | 1.350 | 0.179 |
| arterial hypertension | t | 0.623 | 0.534 | −2.264 | 0.025 |
| hyperglycaemia | t | −0.224 | 0.823 | 0.729 | 0.467 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bojar, I.; Raczkiewicz, D.; Sarecka-Hujar, B. Depression, Metabolic Syndrome, Serum TSH, and Vitamin D Concentrations in Rural and Urban Postmenopausal Women. Medicina 2020, 56, 511. https://doi.org/10.3390/medicina56100511
Bojar I, Raczkiewicz D, Sarecka-Hujar B. Depression, Metabolic Syndrome, Serum TSH, and Vitamin D Concentrations in Rural and Urban Postmenopausal Women. Medicina. 2020; 56(10):511. https://doi.org/10.3390/medicina56100511
Chicago/Turabian StyleBojar, Iwona, Dorota Raczkiewicz, and Beata Sarecka-Hujar. 2020. "Depression, Metabolic Syndrome, Serum TSH, and Vitamin D Concentrations in Rural and Urban Postmenopausal Women" Medicina 56, no. 10: 511. https://doi.org/10.3390/medicina56100511
APA StyleBojar, I., Raczkiewicz, D., & Sarecka-Hujar, B. (2020). Depression, Metabolic Syndrome, Serum TSH, and Vitamin D Concentrations in Rural and Urban Postmenopausal Women. Medicina, 56(10), 511. https://doi.org/10.3390/medicina56100511







