Hypothesized Mechanisms Through Which Exercise May Attenuate Memory Interference
Abstract
:1. Introduction
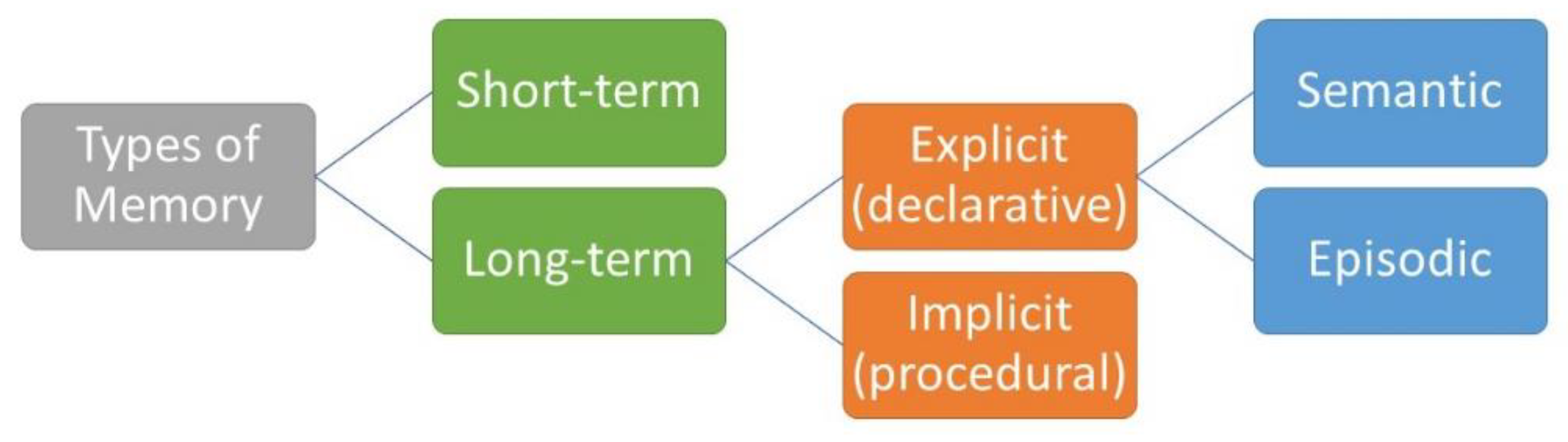
2. Types of Memory
3. Episodic Memory Stages
4. The Role of Exercise
5. Chronic Exercise
6. Acute Exercise
7. Memory Interference
8. Consequences of Memory Interference
9. The Effect of Exercise on Cognitive Memory Interference
10. Mechanisms of Attenuating Memory Interference
11. Hypothesized Mechanisms through Which Aerobic Exercise May Attenuate Memory Interference
12. Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BDNF | brain derived neurotrophic factor |
| DLPFC | dorsolateral prefrontal cortex |
| IGF-1 | insulin-like growth factor 1 |
| LC | locus coerulous |
| mPFC | medial prefrontal cortex |
| MRI | magnetic reasoning imaging |
| NTS | nucleus of the tractus solitarius |
| PI | proactive interference |
| RAVLT | Rey auditory verbal test |
| RI | retroactive interference |
| VEGF | vascular endothelial growth factor |
| VLPFC | ventrolateral prefrontal cortex |
| VO2 max | maximal oxygen uptake |
References
- Lanier, W.L.; Iaizzo, P.A.; Milde, J.H. Cerebral function and muscle afferent activity following intravenous succinylcholine in dogs anesthetized with halothane: The effects of pretreatment with a defasciculating dose of pancuronium. Anesthesiology 1989, 71, 87–95. [Google Scholar] [CrossRef]
- Lanier, W.L.; Milde, J.H.; Michenfelder, J.D. Cerebral stimulation following succinylcholine in dogs. Anesthesiology 1986, 64, 551–559. [Google Scholar] [CrossRef]
- Lanier, W.L.; Iaizzo, P.A.; Milde, J.H. The effects of intravenous succinylcholine on cerebral function and muscle afferent activity following complete ischemia in halothane-anesthetized dogs. Anesthesiology 1990, 73, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Tulving, E. Memory systems and the brain. Clin. Neuropharmacol. 1992, 15 (Suppl. 1), 327A–328A. [Google Scholar] [CrossRef]
- Tulving, E. Episodic and Semantic Memory; Academic Press: Cambridge, MA, USA, 1972. [Google Scholar]
- Tulving, E. Episodic memory and common sense: How far apart? Philos. Trans. R. Soc. Lond. B Biol. Sci. 2001, 356, 1505–1515. [Google Scholar] [CrossRef] [PubMed]
- Tulving, E. Episodic memory: From mind to brain. Rev. Neurol. 2004, 160, S9–S23. [Google Scholar] [CrossRef]
- Squire, L.R. Memory systems of the brain: A brief history and current perspective. Neurobiol. Learn. Mem. 2004, 82, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Puglisi, J.T.; Park, D.C.; Smith, A.D.; Dudley, W.N. Age differences in encoding specificity. J. Gerontol. 1988, 43, P145–P150. [Google Scholar] [CrossRef] [Green Version]
- McGaugh, J.L. Memory—A century of consolidation. Science 2000, 287, 248–251. [Google Scholar] [CrossRef] [Green Version]
- Atkinson, S.R. A key to occlusion. Am. J. Orthod. 1968, 54, 217–233. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Edwards, M.K.; Frith, E. Potential avenues for exercise to activate episodic memory-related pathways: A narrative review. Eur. J. Neurosci. 2017, 46, 2067–2077. [Google Scholar] [CrossRef] [PubMed]
- Roig, M.; Nordbrandt, S.; Geertsen, S.S.; Nielsen, J.B. The effects of cardiovascular exercise on human memory: A review with meta-analysis. Neurosci. Biobehav. Rev. 2013, 37, 1645–1666. [Google Scholar] [CrossRef] [PubMed]
- Crawford, L.K.L.; Loprinzi, P. Effects of intensity-specific acute exercise on paired-associative memory and memory interference. Psych 2019, 1, 290–305. [Google Scholar] [CrossRef] [Green Version]
- El-Sayes, J.; Harasym, D.; Turco, C.V.; Locke, M.B.; Nelson, A.J. Exercise-Induced Neuroplasticity: A Mechanistic Model and Prospects for Promoting Plasticity. Neuroscientist 2019, 25, 65–85. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.I.; Leckie, R.L.; Weinstein, A.M. Physical activity, fitness, and gray matter volume. Neurobiol. Aging 2014, 35 (Suppl. 2), S20–S28. [Google Scholar] [CrossRef] [Green Version]
- Yamada, K.; Mizuno, M.; Nabeshima, T. Role for brain-derived neurotrophic factor in learning and memory. Life Sci. 2002, 70, 735–744. [Google Scholar] [CrossRef]
- Yook, J.S.; Rakwal, R.; Shibato, J.; Takahashi, K.; Koizumi, H.; Shima, T.; Ikemoto, M.J.; Oharomari, L.K.; McEwen, B.S.; Soya, H. Leptin in hippocampus mediates benefits of mild exercise by an antioxidant on neurogenesis and memory. Proc. Natl. Acad. Sci. USA 2019, 116, 10988–10993. [Google Scholar] [CrossRef] [Green Version]
- Thomas, A.G.; Dennis, A.; Bandettini, P.A.; Johansen-Berg, H. The effects of aerobic activity on brain structure. Front. Psychol. 2012, 3, 86. [Google Scholar] [CrossRef] [Green Version]
- Loprinzi, P.D. The effects of exercise on dendritic spine density: Implications for exercise-induced memory enhancement. J. Neurobehav. Sci. 2019, 6, 76–79. [Google Scholar] [CrossRef]
- Lanz, T.A.; Bove, S.E.; Pilsmaker, C.D.; Mariga, A.; Drummond, E.M.; Cadelina, G.W.; Adamowicz, W.O.; Swetter, B.J.; Carmel, S.; Dumin, J.A.; et al. Robust changes in expression of brain-derived neurotrophic factor (BDNF) mRNA and protein across the brain do not translate to detectable changes in BDNF levels in CSF or plasma. Biomarkers 2012, 17, 524–531. [Google Scholar] [CrossRef]
- Pan, W.; Banks, W.A.; Fasold, M.B.; Bluth, J.; Kastin, A.J. Transport of brain-derived neurotrophic factor across the blood-brain barrier. Neuropharmacology 1998, 37, 1553–1561. [Google Scholar] [CrossRef]
- Hotting, K.; Schickert, N.; Kaiser, J.; Roder, B.; Schmidt-Kassow, M. The Effects of Acute Physical Exercise on Memory, Peripheral BDNF, and Cortisol in Young Adults. Neural. Plast. 2016, 2016, 6860573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Etnier, J.L.; Wideman, L.; Labban, J.D.; Piepmeier, A.T.; Pendleton, D.M.; Dvorak, K.K.; Becofsky, K. The Effects of Acute Exercise on Memory and Brain-Derived Neurotrophic Factor (BDNF). J. Sport Exerc. Psychol. 2016, 38, 331–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomas, R.; Flindtgaard, M.; Skriver, K.; Geertsen, S.S.; Christiansen, L.; Korsgaard Johnsen, L.; Busk, D.V.P.; Bojsen-Moller, E.; Madsen, M.J.; Ritz, C.; et al. Acute exercise and motor memory consolidation: Does exercise type play a role? Scand. J. Med. Sci. Sports 2017, 27, 1523–1532. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D.; Blough, J.; Crawford, L.; Ryu, S.; Zou, L.; Li, H. The Temporal Effects of Acute Exercise on Episodic Memory Function: Systematic Review with Meta-Analysis. Brain Sci. 2019, 9, 87. [Google Scholar] [CrossRef] [Green Version]
- Loprinzi, P.D. An integrated model of acute exercise on memory function. Med. Hypotheses 2019, 126, 51–59. [Google Scholar] [CrossRef]
- Chatzi, C.; Zhang, Y.; Hendricks, W.D.; Chen, Y.; Schnell, E.; Goodman, R.H.; Westbrook, G.L. Exercise-induced enhancement of synaptic function triggered by the inverse BAR protein, Mtss1L. eLife 2019, 8. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Ponce, P.; Frith, E. Hypothesized mechanisms through which acute exercise influences episodic memory. Physiol. Int. 2018, 105, 285–297. [Google Scholar] [CrossRef] [Green Version]
- Davis, R.L.; Zhong, Y. The Biology of Forgetting-A Perspective. Neuron 2017, 95, 490–503. [Google Scholar] [CrossRef]
- Shimamura, A.; Paul, J.; Jurica, J.A. Mangels, and Felicia B Gershberg. Susceptibility to Memory Interference Effects following Frontal Lobe Damage: Findings from Tests of Paired-Associate Learning. J. Cogn. Neurosci. 1995, 7, 144–152. [Google Scholar] [CrossRef]
- Melcher, D.; Murphy, B. The role of semantic interference in limiting memory for the details of visual scenes. Front. Psychol. 2011, 2, 262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Edwards, W.H. Motor Learning and Control: From Theory to Practice; Cengage Learning: Boston, MA, USA, 2010. [Google Scholar]
- Bergström, J.A. Experiments upon Physiological Memory by Means of the Interference of Associations. Am. J. Psychol. 1893, 5, 356–369. [Google Scholar] [CrossRef]
- Guise, K.G.; Shapiro, M.L. Medial Prefrontal Cortex Reduces Memory Interference by Modifying Hippocampal Encoding. Neuron 2017, 94, 183–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ecker, U.K.; Brown, G.D.; Lewandowsky, S. Memory Without Consolidation: Temporal Distinctiveness Explains Retroactive Interference. Cogn. Sci. 2015, 39, 1570–1593. [Google Scholar] [CrossRef]
- Irlbacher, K.; Kraft, A.; Kehrer, S.; Brandt, S.A. Mechanisms and neuronal networks involved in reactive and proactive cognitive control of interference in working memory. Neurosci. Biobehav. Rev. 2014, 46 Pt 1, 58–70. [Google Scholar] [CrossRef]
- Frith, S.; Loprinzi, P.D. Randomized Controlled Trial Considering Varied Exercises for Reducing Proactive Memory Interference. J. Clin. Med. 2018, 7, 147. [Google Scholar] [CrossRef] [Green Version]
- Haynes, J.; Loprinzi, P. Acute cardiovascular exercise on proactive memory interference. J. Cogn. Enhanc. 2018, 2, 139–143. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Frith, E.; Crawford, L. Acute exercise and retroactive memory interference. Am. J. Health Promot. 2020, 34, 25–31. [Google Scholar] [CrossRef]
- Wingate, S.; Crawford, L.; Frith, E.; Loprinzi, P.D. Experimental investigation of the effects of acute exercise on memory interference. Health Promot. Perspect. 2018, 8, 208–214. [Google Scholar] [CrossRef]
- Johnson, L.; Crawford, L.; Zou, L.; Loprinzi, P.D. Experimental Effects of Acute Exercise in Attenuating Memory Interference: Considerations by Biological Sex. Medicina 2019, 55, 331. [Google Scholar] [CrossRef] [Green Version]
- Bernstein, E.E.; McNally, R.J. Examining the Effects of Exercise on Pattern Separation and the Moderating Effects of Mood Symptoms. Behav. Ther. 2019, 50, 582–593. [Google Scholar] [CrossRef] [PubMed]
- Bolz, L.; Heigele, S.; Bischofberger, J. Running Improves Pattern Separation during Novel Object Recognition. Brain Plast. 2015, 1, 129–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suwabe, K.; Byun, K.; Hyodo, K.; Reagh, Z.M.; Roberts, J.M.; Matsushita, A.; Saotome, K.; Ochi, G.; Fukuie, T.; Suzuki, K.; et al. Rapid stimulation of human dentate gyrus function with acute mild exercise. Proc. Natl. Acad. Sci. USA 2018, 115, 10487–10492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, M.W.; Aggleton, J.P. Recognition memory: What are the roles of the perirhinal cortex and hippocampus? Nat. Rev. Neurosci. 2001, 2, 51–61. [Google Scholar] [CrossRef]
- Loprinzi, P.D. The effects of physical exercise on parahippocampal function. Physiol. Int. 2019, 106, 114–127. [Google Scholar] [CrossRef]
- Vivar, C.; Peterson, B.D.; van Praag, H. Running rewires the neuronal network of adult-born dentate granule cells. Neuroimage 2016, 131, 29–41. [Google Scholar] [CrossRef]
- Hopkins, M.E.; Bucci, D.J. BDNF expression in perirhinal cortex is associated with exercise-induced improvement in object recognition memory. Neurobiol. Learn. Mem. 2010, 94, 278–284. [Google Scholar] [CrossRef] [Green Version]
- Hopkins, M.E.; Nitecki, R.; Bucci, D.J. Physical exercise during adolescence versus adulthood: Differential effects on object recognition memory and brain-derived neurotrophic factor levels. Neuroscience 2011, 194, 84–94. [Google Scholar] [CrossRef] [Green Version]
- Griffin, E.W.; Bechara, R.G.; Birch, A.M.; Kelly, A.M. Exercise enhances hippocampal-dependent learning in the rat: Evidence for a BDNF-related mechanism. Hippocampus 2009, 19, 973–980. [Google Scholar] [CrossRef]
- Cayco-Gajic, N.A.; Silver, R.A. Re-evaluating Circuit Mechanisms Underlying Pattern Separation. Neuron 2019, 101, 584–602. [Google Scholar] [CrossRef] [Green Version]
- Zheng, J.; Stevenson, R.F.; Mander, B.A.; Mnatsakanyan, L.; Hsu, F.P.K.; Vadera, S.; Knight, R.T.; Yassa, M.A.; Lin, J.J. Multiplexing of Theta and Alpha Rhythms in the Amygdala-Hippocampal Circuit Supports Pattern Separation of Emotional Information. Neuron 2019. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.E.; Ross, R.S.; Stern, C.E. Predictability matters: Role of the hippocampus and prefrontal cortex in disambiguation of overlapping sequences. Learn. Mem. 2018, 25, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, N.S.; Allen, T.A.; Nguyen, N.; Fortin, N.J. Critical role of the hippocampus in memory for elapsed time. J. Neurosci. 2013, 33, 13888–13893. [Google Scholar] [CrossRef] [PubMed]
- Klimesch, W. EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Res. Brain Res.Rev. 1999, 29, 169–195. [Google Scholar] [CrossRef]
- Basso, J.C.; Suzuki, W.A. The Effects of Acute Exercise on Mood, Cognition, Neurophysiology, and Neurochemical Pathways: A Review. Brain Plast. 2017, 2, 127–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zang, J.; Liu, Y.; Li, W.; Xiao, D.; Zhang, Y.; Luo, Y.; Liang, W.; Liu, F.; Wei, W. Voluntary exercise increases adult hippocampal neurogenesis by increasing GSK-3beta activity in mice. Neuroscience 2017, 354, 122–135. [Google Scholar] [CrossRef]
- Lin, T.W.; Shih, Y.H.; Chen, S.J.; Lien, C.H.; Chang, C.Y.; Huang, T.Y.; Chen, S.H.; Jen, C.J.; Kuo, Y.M. Running exercise delays neurodegeneration in amygdala and hippocampus of Alzheimer’s disease (APP/PS1) transgenic mice. Neurobiol. Learn. Mem. 2015, 118, 189–197. [Google Scholar] [CrossRef]
- Voss, M.W.; Soto, C.; Yoo, S.; Sodoma, M.; Vivar, C.; van Praag, H. Exercise and Hippocampal Memory Systems. Trends Cogn. Sci. 2019, 23, 318–333. [Google Scholar] [CrossRef]
- Azab, M.; Stark, S.M.; Stark, C.E. Contributions of human hippocampal subfields to spatial and temporal pattern separation. Hippocampus 2014, 24, 293–302. [Google Scholar] [CrossRef] [Green Version]
- Cameron, H.A.; McKay, R.D. Adult neurogenesis produces a large pool of new granule cells in the dentate gyrus. J. Comp. Neurol. 2001, 435, 406–417. [Google Scholar] [CrossRef]
- Jung, M.W.; McNaughton, B.L. Spatial selectivity of unit activity in the hippocampal granular layer. Hippocampus 1993, 3, 165–182. [Google Scholar] [CrossRef] [PubMed]
- Poo, M.M.; Pignatelli, M.; Ryan, T.J.; Tonegawa, S.; Bonhoeffer, T.; Martin, K.C.; Rudenko, A.; Tsai, L.H.; Tsien, R.W.; Fishell, G.; et al. What is memory? The present state of the engram. BMC Biol. 2016, 14, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dery, N.; Pilgrim, M.; Gibala, M.; Gillen, J.; Wojtowicz, J.M.; Macqueen, G.; Becker, S. Adult hippocampal neurogenesis reduces memory interference in humans: Opposing effects of aerobic exercise and depression. Front. Neurosci. 2013, 7, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Voss, M.W.; Heo, S.; Prakash, R.S.; Erickson, K.I.; Alves, H.; Chaddock, L.; Szabo, A.N.; Mailey, E.L.; Wojcicki, T.R.; White, S.M.; et al. The influence of aerobic fitness on cerebral white matter integrity and cognitive function in older adults: Results of a one-year exercise intervention. Hum. Brain Mapp. 2013, 34, 2972–2985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tseng, B.Y.; Gundapuneedi, T.; Khan, M.A.; Diaz-Arrastia, R.; Levine, B.D.; Lu, H.; Huang, H.; Zhang, R. White matter integrity in physically fit older adults. Neuroimage 2013, 82, 510–516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bennett, I.J.; Huffman, D.J.; Stark, C.E. Limbic Tract Integrity Contributes to Pattern Separation Performance Across the Lifespan. Cereb Cortex 2015, 25, 2988–2999. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frankland, P.W.; Kohler, S.; Josselyn, S.A. Hippocampal neurogenesis and forgetting. Trends Neurosci. 2013, 36, 497–503. [Google Scholar] [CrossRef]
- Dulas, M.R.; Duarte, A. Age-related changes in overcoming proactive interference in associative memory: The role of PFC-mediated executive control processes at retrieval. Neuroimage 2016, 132, 116–128. [Google Scholar] [CrossRef] [Green Version]
- Thompson-Schill, S.L.; Jonides, J.; Marshuetz, C.; Smith, E.E.; D’Esposito, M.; Kan, I.P.; Knight, R.T.; Swick, D. Effects of frontal lobe damage on interference effects in working memory. Cogn. Affect. Behav. Neurosci. 2002, 2, 109–120. [Google Scholar] [CrossRef] [Green Version]
- Feredoes, E.; Heinen, K.; Weiskopf, N.; Ruff, C.; Driver, J. Causal evidence for frontal involvement in memory target maintenance by posterior brain areas during distracter interference of visual working memory. Proc. Natl. Acad. Sci. USA 2011, 108, 17510–17515. [Google Scholar] [CrossRef] [Green Version]
- Addis, D.R.; Giovanello, K.S.; Vu, M.A.; Schacter, D.L. Age-related changes in prefrontal and hippocampal contributions to relational encoding. Neuroimage 2014, 84, 19–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]







© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crawford, L.K.; Li, H.; Zou, L.; Wei, G.-X.; Loprinzi, P.D. Hypothesized Mechanisms Through Which Exercise May Attenuate Memory Interference. Medicina 2020, 56, 129. https://doi.org/10.3390/medicina56030129
Crawford LK, Li H, Zou L, Wei G-X, Loprinzi PD. Hypothesized Mechanisms Through Which Exercise May Attenuate Memory Interference. Medicina. 2020; 56(3):129. https://doi.org/10.3390/medicina56030129
Chicago/Turabian StyleCrawford, Lindsay K., Hong Li, Liye Zou, Gao-Xia Wei, and Paul D. Loprinzi. 2020. "Hypothesized Mechanisms Through Which Exercise May Attenuate Memory Interference" Medicina 56, no. 3: 129. https://doi.org/10.3390/medicina56030129




