COVID-19 and Alzheimer’s Disease: A Literature Review
Abstract
:1. Introduction
2. Overview on Alzheimer’s Disease
2.1. Causes of Alzheimer’s Disease
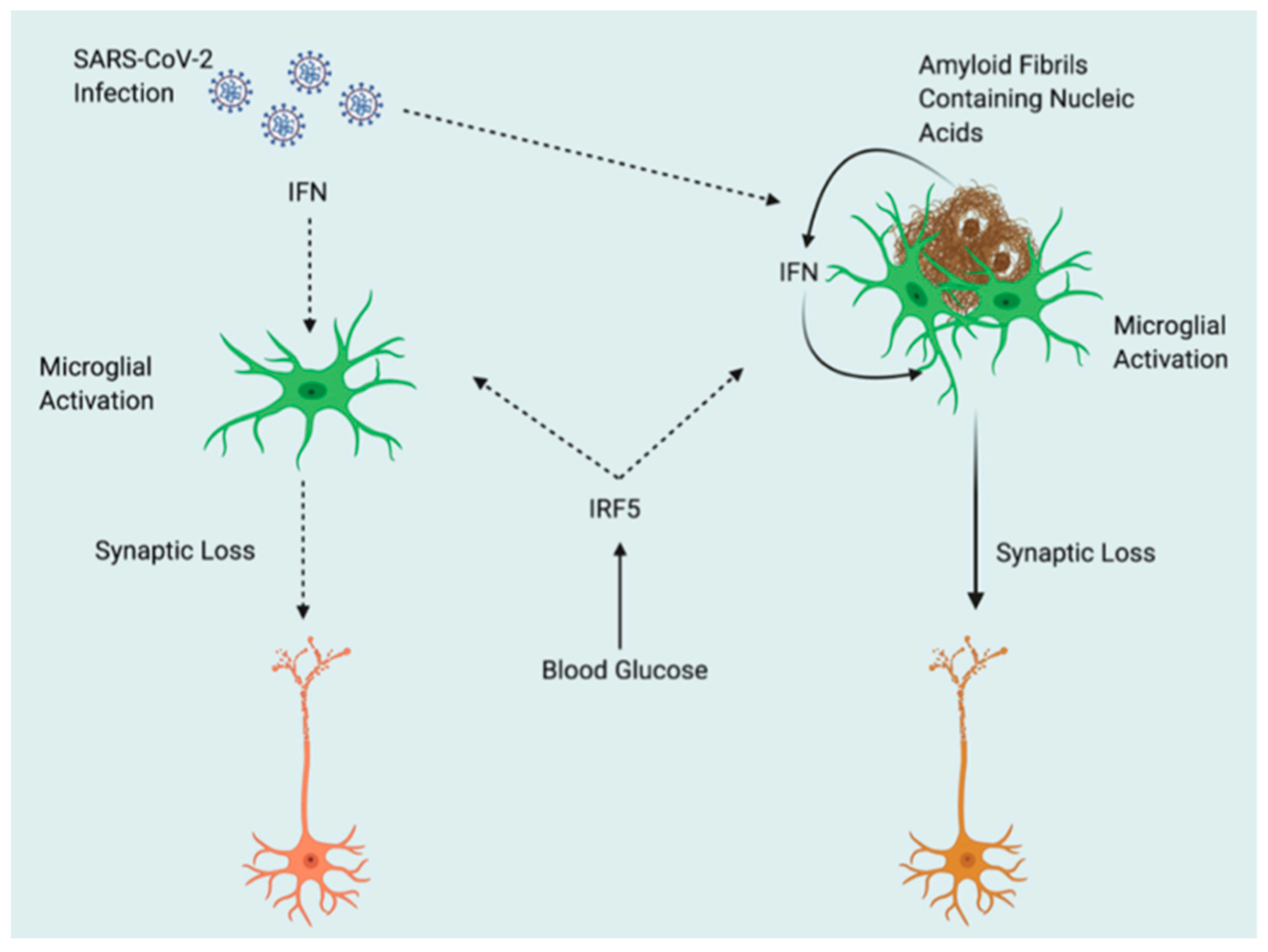
- Beta-amyloid plaque production: elevated cholesterol levels in the blood cause the creation of beta-amyloid plaques. When proteins cluster together, they form a hard, insoluble plaque. Plaque builds up between neurons in the brain, inhibiting cell communication and function. These cells eventually die as a result of this. When brain cells deteriorate and die, they lose their ability to process, store, and retrieve data. As a result, one of Alzheimer’s symptoms is memory loss [12]. AD is associated with hyperphosphorylation of tau protein and accumulation of amyloid-β peptide in the brain [13].
- Multi-pathogen infections: the interaction of several potential pathogens with neurodegeneration and neuroinflammation in AD has been reported [13,14]. Various pathogens, including viruses (Herpes simplex virus type 1) and oral infectious pathogens, especially periodontal infections caused by Porphyromonas gingivalis, a key pathogen in chronic periodontitis, was found in AD cases. However, other bacteria, such as Helicobacter pylori, which is associated with chronic gastric diseases, and Chlamydophila pneumoniae, which is implicated in chronic and lower-respiratory-tract diseases, also to play a role in AD [14].
- Neurofibrillary tangles: Tau proteins are disrupted by an increase in enzymes called tau kinases. They clump together and create neurofibrillary tangles as their structure changes. The tangles injure brain cells by disrupting cell communication [15].
- Blood flow deficiency: A lack of blood flow to the brain prevents the transfer all of the necessary nutrients to the cells. Blood clots, on the other hand, harm blood vessels. As a result, the memory-related areas of the brain, such as the amygdala and the hippocampus, do not receive enough blood, and this leads to the deterioration of the brain function [16].
2.2. Types of AD
- The most frequent type of AD is late-onset, which manifests symptoms in the mid-60 s. This type of AD is not produced by one gene only. One gene, however, could be a risk factor. The apolipoprotein E gene (APOE) has one allele (a variant form of a gene) that raises a person’s risk of developing this type of Alzheimer’s [19].
3. Overview on COVID-19 Disease
4. The Impact of COVID-19 on Alzheimer’s Disease
4.1. Direct Neurological Consequences of COVID-19 on People with Alzheimer’s Disease
- “SARS-COV-2” enters the brain via the olfactory system. In fact, loss of smell is common in COVID-19 infection. MRI hypersignals of the olfactory epithelial cortex indicate infection especially with the presence of ACE2 receptors and TMPRSS2 in the olfactory epithelium [36]. The virus could enter by nerve terminals and spread into the brain as described by Dubé et al. [37]. Therefore, it is not evident that the virus reaches the olfactory neurons in this manner.
- Via blood-brain barrier (BBB): the virus reaches the brain by infecting the endothelial cells [35]. The presence of ACE2 receptors, other putative “SARS-COV-2” receptors, and inflammatory cytokines like interleukin (IL)-6, IL-1b, tumor necrosis factor (TNF), and IL-17 disrupt the BBB and may facilitate the entry of the virus to the brain endothelial cells [38]. The presence of pre-existing neurological disease or comorbidities increases the permeability of BBB.
- Infiltration with infection immune cells: infected immune cells can transfer virus to the brain. It is unclear if “SARS-COV-2” can enter immune cells for infection and transport to the CNS [39]. Chen et al. demonstrated the presence of “SARS-COV-2” nucleocapsid proteins in CD68 lymphocytes and macrophage; nonetheless, it is not clear whether it is the normal process of phagocytosis or infected cells [40]. In addition, many cerebral autopsies reveal a lack of immune cells [41].
4.2. Indirect Consequences of COVID-19 on People with Alzheimer’s Disease
5. Management Strategies
6. Future Perspective
7. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pérez-Rodríguez, P.; Ganz, F.; Torralba, R.; Oliveira, D.V.; Rodríguez-Mañas, L. Impact of Social Isolation Due to COVID-19 on Health in Older People: Mental and Physical Effects and Recommendations. J. Nutr. Health Aging 2020, 24, 938–947. [Google Scholar] [CrossRef]
- Brown, E.E.; Kumar, S.; Rajji, T.K.; Pollock, B.G.; Mulsant, B.H. Anticipating and Mitigating the Impact of the COVID-19 Pandemic on Alzheimer’s Disease and Related Dementias. Am. J. Geriatr. Psychiatry 2020, 28, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Giménez, A. COVID-19, Psychological Well-being and Physical Activity Levels in Older Adults During the Nationwide Lockdown in Spain. Am. J. Geriatr. Psychiatry 2020, 28, 1146–1155. [Google Scholar] [CrossRef]
- Mok, V.C.; Pendlebury, S.; Wong, A.; Alladi, S.; Au, L.; Bath, P.M.; Biessels, G.J.; Chen, C.; Cordonnier, C.; Dichgans, M.; et al. Tackling challenges in care of Alzheimer’s disease and other dementias amid the COVID-19 pandemic, now and in the future. Alzheimers Dement. 2020, 16, 1571–1581. [Google Scholar] [CrossRef]
- Numbers, K.; Brodaty, H. The effects of the COVID-19 pandemic on people with dementia. Nat. Rev. Neurol. 2021, 17, 69–70. [Google Scholar] [CrossRef]
- Souder, E.; Beck, C. Overview of Alzheimer’s disease. Nurs. Clin. 2004, 39, 545–559. [Google Scholar] [CrossRef]
- Wang, S.S.S.; Good, T.A. An overview of Alzheimer’s disease. J. Chin. Inst. Chem. Eng. 2005, 36, 533–559. [Google Scholar]
- Fishman, E. Risk of developing dementia at older ages in the United States. Demography 2017, 54, 1897–1919. [Google Scholar] [CrossRef] [Green Version]
- Meyer, J.C.; Harirari, P.; Schellack, N. Overview of Alzheimer’s disease and its management. SA Pharm. J. 2016, 83, 48–56. [Google Scholar]
- Ihara, Y. Overview on Alzheimer’s disease. Rinsho Shinkeigaku Clin. Neurol. 2007, 47, 902–904. [Google Scholar]
- Schneck, M.K.; Reisberg, B.; Ferris, S.H. An overview of current concepts of Alzheimer’s disease. Am. J. Psychiatry 1982, 139, 165–173. [Google Scholar] [PubMed]
- Soria Lopez, J.A.; González, H.M.; Léger, G.C. Alzheimer’s disease. Handb. Clin. Neurol. 2019, 167, 231–255. [Google Scholar] [CrossRef]
- Vigasova, D.; Nemergut, M.; Liskova, B.; Damborsky, J. Multi-pathogen infections and Alzheimer’s disease. Microb. Cell. Fact. 2021, 20, 25. [Google Scholar] [CrossRef]
- Frölich, L. Alzheimer’s disease—The ‘microbial hypothesis’ from a clinical and neuroimaging perspective. Psychiatry Res. Neuroimaging 2020, 306, 111181. [Google Scholar] [CrossRef]
- Magalingam, K.B.; Radhakrishnan, A.; Ping, N.S.; Haleagrahara, N. Current Concepts of Neurodegenerative Mechanisms in Alzheimer’s Disease. BioMed Res. Int. 2018, 2018, 3740461. [Google Scholar] [CrossRef] [Green Version]
- Varma, V.R.; Oommen, A.; Varma, S.; Casanova, R.; An, Y.; Andrews, R.M.; O’Brien, R.; Pletnikova, O.; Troncoso, J.C.; Toledo, J.; et al. Brain and blood metabolite signatures of pathology and progression in Alzheimer disease: A targeted metabolomics study. PLoS Med. 2018, 15, e1002482. [Google Scholar] [CrossRef]
- McManus, R.M.; Heneka, M.T. Role of neuroinflammation in neurodegeneration: New insights. Alzheimers Res. Ther. 2017, 9, 14. [Google Scholar] [CrossRef] [Green Version]
- Webers, A.; Heneka, M.T.; Gleeson, P.A. The role of innate immune responses and neuroinflammation in amyloid accumulation and progression of Alzheimer’s disease. Immunol. Cell Biol. 2020, 98, 28–41. [Google Scholar] [CrossRef] [PubMed]
- Khachaturian, Z.S. Calcium, membranes, aging, and Alzheimer’s disease. Introduction and overview. Ann. N. Y. Acad. Sci. 1989, 568, 1–4. [Google Scholar] [CrossRef]
- Monto, A.S. Medical reviews. Coronaviruses. Yale J. Biol. Med. 1974, 47, 234–251. [Google Scholar] [PubMed]
- World Health Organization. Summary of Probable SARS Cases with Onset of Illness; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Cauchemez, S.; Fraser, C.; Van Kerkhove, M.D.; Donnelly, C.A.; Riley, S.; Rambaut, A.; Enouf, V.; van der Werf, S.; Ferguson, N.M. Middle East respiratory syndrome coronavirus: Quantification of the extent of the epidemic, surveillance biases, and transmissibility. Lancet Infect. Dis. 2014, 14, 50–56. [Google Scholar] [CrossRef] [Green Version]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Meyerowitz, E.A.; Richterman, A.; Gandhi, R.T.; Sax, P.E. Transmission of SARS-CoV-2: A Review of Viral, Host, and Environmental Factors. Ann. Intern. Med. 2021, 174, 69–79. [Google Scholar] [CrossRef]
- Rosolanka, R.; Henao-Martinez, A.F.; Pisney, L.; Franco-Paredes, C.; Krsak, M. COVID-19: A review of current knowledge regarding exposure, quarantine, isolation and other preventive measures. Ther. Adv. Infect. Dis. 2021, 8, 20499361211032039. [Google Scholar] [CrossRef]
- Chan, J.F.; Kok, K.H.; Zhu, Z.; Chu, H.; To, K.K.; Yuan, S.; Yuen, K.Y. Genomic characterization of the 2019 novel human-pathogenic coronavirus isolated from a patient with atypical pneumonia after visiting Wuhan. Emerg. Microbes Infect. 2020, 9, 221–236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xie, Y.; Wang, Z.; Liao, H.; Marley, G.; Wu, D.; Tang, W. Epidemiologic, clinical, and laboratory findings of the COVID-19 in the current pandemic: Systematic review and meta-analysis. BMC Infect. Dis. 2020, 20, 640. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 28, 1054–1062. [Google Scholar] [CrossRef]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef] [Green Version]
- Ye, Q.; Wang, B.; Mao, J. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J. Infect. 2020, 80, 607–613. [Google Scholar] [CrossRef]
- Illg, Z.; Muller, G.; Mueller, M.; Nippert, J.; Allen, B. Analysis of absolute lymphocyte count in patients with COVID-19. Am. J. Emerg. Med. 2021, 46, 16–19. [Google Scholar] [CrossRef] [PubMed]
- Ciotti, M.; Angeletti, S.; Minieri, M.; Giovannetti, M.; Benvenuto, D.; Pascarella, S.; Sagnelli, C.; Bianchi, M.; Bernardini, S.; Ciccozzi, M. COVID-19 outbreak: An overview. Chemotherapy 2019, 64, 215–223. [Google Scholar] [CrossRef]
- Sacks, D.; Baxter, B.; Campbell, B.C.V.; Carpenter, J.S.; Cognard, C.; Dippel, D.; Eesa, M.; Fischer, U.; Hausegger, K.; Hirsch, J.A.; et al. Multisociety Consensus Quality Improvement Revised Consensus Statement for Endovascular Therapy of Acute Ischemic Stroke. Int. J. Stroke 2018, 13, 612–632. [Google Scholar] [CrossRef] [Green Version]
- Hamming, I.; Timens, W.; Bulthuis, M.L.; Lely, A.T.; Navis, G.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, C.C.; Lane, T.E.; Stohlman, S.A. Coronavirus infection of the central nervous system: Host-virus stand-off. Nat. Rev. Microbiol. 2006, 4, 121–132. [Google Scholar] [CrossRef] [Green Version]
- Butowt, R.; von Bartheld, C.S. Anosmia in COVID-19: Underlying Mechanisms and Assessment of an Olfactory Route to Brain Infection. Neuroscientist 2020, 1073858420956905. [Google Scholar] [CrossRef]
- Dubé, M.; Le Coupanec, A.; Wong, A.H.M.; Rini, J.M.; Desforges, M.; Talbot, P.J. Axonal Transport Enables Neuron-to-Neuron Propagation of Human Coronavirus OC43. J. Virol. 2018, 92, e00404-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erickson, M.A.; Banks, W.A. Neuroimmune Axes of the Blood-Brain Barriers and Blood-Brain Interfaces: Bases for Physiological Regulation, Disease States, and Pharmacological Interventions. Pharmacol. Rev. 2018, 70, 278–314. [Google Scholar] [CrossRef]
- Merad, M.; Martin, J.C. Pathological inflammation in patients with COVID-19: A key role for monocytes and macrophages. Nat. Rev. Immunol. 2020, 20, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Wang, K.; Yu, J.; Howard, D.; French, L.; Chen, Z.; Wen, C.; Xu, Z. The Spatial and Cell-Type Distribution of SARS-CoV-2 Receptor ACE2 in the Human and Mouse Brains. Front. Neurol. 2021, 11, 573095. [Google Scholar] [CrossRef]
- Kantonen, J.; Mahzabin, S.; Mäyränpää, M.I.; Tynninen, O.; Paetau, A.; Andersson, N.; Sajantila, A.; Vapalahti, O.; Carpén, O.; Kekäläinen, E.; et al. Neuropathologic features of four autopsied COVID-19 patients. Brain. Pathol. 2020, 30, 1012–1016. [Google Scholar] [CrossRef]
- Xia, X.; Wang, Y.; Zheng, J. COVID-19 and Alzheimer’s disease: How one crisis worsens the other. Transl. Neurodegener. 2021, 10, 15. [Google Scholar] [CrossRef]
- Nguyen, S.; Major, K.; Cochet, C.; Bizzozzero, T.; Barbarossa, L.; Bosshard, W.; Humbert, M.; Rubli, E.; Jox, R.; D’Amelio, P.; et al. COVID-19 infection in the elderly in French-speaking Switzerland: An inventory of beliefs, convictions and certainties. Rev. Med. Suisse 2020, 16, 835–838. [Google Scholar]
- Cesari, M.; Proietti, M. COVID-19 in Italy: Ageism and decision making in a pandemic. J. Am. Med. Dir. Assoc. 2020, 21, 576–577. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Islam, K.; Rahman, S.; Alamin, M. Neurobiochemical Cross-talk Between COVID-19 and Alzheimer’s Disease. Mol. Neurobiol. 2021, 58, 1017–1023. [Google Scholar] [CrossRef] [PubMed]
- Iodice, F.; Cassano, V.; Rossini, P.M. Direct and indirect neurological, cognitive, and behavioral effects of COVID-19 on the healthy elderly, mild-cognitive-impairment, and Alzheimer’s disease populations. Neurol. Sci. 2021, 42, 455–465. [Google Scholar] [CrossRef]
- Jacus, J.P.; Dupont, M.P.; Herades, Y.; Pelix, C.; Large, H.; Baud, M. Conscience des troubles dans la maladie d’Alzheimer et le mild cognitive impairment. Encephale 2014, 40, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Xiong, M.; Liang, X.; Wei, Y.-D. Changes in blood coagulation in patients with severe coronavirus disease 2019 (COVID-19): A meta-analysis. Br. J. Haematol. 2020, 189, 1050–1052. [Google Scholar] [CrossRef] [Green Version]
- Helms, J.; Kremer, S.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Kummerlen, C.; Collange, O.; Boulay, C.; Fafi-Kremer, S.; Ohana, M.; et al. Neurologic features in severe SARS-CoV-2 infection. N. Engl. J. Med. 2020, 382, 2268–2270. [Google Scholar] [CrossRef] [PubMed]
- Naughton, S.X.; Raval, U.; Pasinetti, G.M. Potential Novel Role of COVID-19 in Alzheimer’s Disease and Preventative Mitigation Strategies. J. Alzheimers Dis. 2020, 76, 21–25. [Google Scholar] [CrossRef]
- Carod-Artal, F.J. Neurological complications of coronavirus and COVID-19. Rev. Neurol. 2020, 70, 311–322. [Google Scholar] [CrossRef]
- Wang, H.; Li, T.; Barbarino, P.; Gauthier, S.; Brodaty, H.; Molinuevo, J.L.; Xie, H.; Sun, Y.; Yu, E.; Tang, Y.; et al. Dementia care during COVID-19. Lancet 2020, 395, 1190–1191. [Google Scholar] [CrossRef]
- Palmer, K.; Monaco, A.; Kivipelto, M.; Onder, G.; Maggi, S.; Michel, J.-P.; Prieto, R.; Sykara, G.; Donde, S. The potential long-term impact of the COVID-19 outbreak on patients with non-communicable diseases in Europe: Consequences for healthy ageing. Aging Clin. Exp. Res. 2020, 32, 1189–1194. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, L.; Vanacore, N.; Donfrancesco, C.; Noce, C.L.; Canevelli, M.; Punzo, O.; Raparelli, V.; Pezzotti, P.; Riccardo, F.; Bella, A.; et al. Clinical characteristics of hospitalized individuals dying with COVID-19 by age Group in Italy. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 1796–1800. [Google Scholar] [CrossRef]
- Boutoleau-Bretonnière, C.; Pouclet-Courtemanche, H.; Gillet, A.; Bernard, A.; Deruet, A.L.; Gouraud, I.; Mazoue, A.; Lamy, E.; Rocher, L.; Kapogiannis, D.; et al. The Effects of Confinement on Neuropsychiatric Symptoms in Alzheimer’s Disease During the COVID-19 Crisis. J. Alzheimers Dis. 2020, 76, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Chiao, C.-Y.; Wu, H.-S.; Hsiao, C.-Y. Caregiver burden for informal caregivers of patients with dementia: A systematic review. Int. Nurs. Rev. 2015, 62, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Abbatecola, A.M.; Antonelli-Incalzi, R. Editorial: COVID-19 spiraling of frailty in older Italian patients. J. Nutr. Health Aging 2020, 24, 453–455. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.Y.Y.; Gobat, N.; Kim, J.H.; Newnham, E.A.; Huang, Z.; Hung, H.; Dubois, C.; Hung, K.K.; Wong, E.L.Y.; Wong, S.Y.S. Informal home care providers: The forgotten health-care workers during the COVID-19 pandemic. Lancet 2020, 395, 1957–1959. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hardan, L.; Filtchev, D.; Kassem, R.; Bourgi, R.; Lukomska-Szymanska, M.; Tarhini, H.; Salloum-Yared, F.; Mancino, D.; Kharouf, N.; Haikel, Y. COVID-19 and Alzheimer’s Disease: A Literature Review. Medicina 2021, 57, 1159. https://doi.org/10.3390/medicina57111159
Hardan L, Filtchev D, Kassem R, Bourgi R, Lukomska-Szymanska M, Tarhini H, Salloum-Yared F, Mancino D, Kharouf N, Haikel Y. COVID-19 and Alzheimer’s Disease: A Literature Review. Medicina. 2021; 57(11):1159. https://doi.org/10.3390/medicina57111159
Chicago/Turabian StyleHardan, Louis, Dimitar Filtchev, Ratiba Kassem, Rim Bourgi, Monika Lukomska-Szymanska, Hassan Tarhini, Fouad Salloum-Yared, Davide Mancino, Naji Kharouf, and Youssef Haikel. 2021. "COVID-19 and Alzheimer’s Disease: A Literature Review" Medicina 57, no. 11: 1159. https://doi.org/10.3390/medicina57111159
APA StyleHardan, L., Filtchev, D., Kassem, R., Bourgi, R., Lukomska-Szymanska, M., Tarhini, H., Salloum-Yared, F., Mancino, D., Kharouf, N., & Haikel, Y. (2021). COVID-19 and Alzheimer’s Disease: A Literature Review. Medicina, 57(11), 1159. https://doi.org/10.3390/medicina57111159











