Impact of Palliative Gastrectomy in Patients with Incurable Gastric Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Statistical Analyses
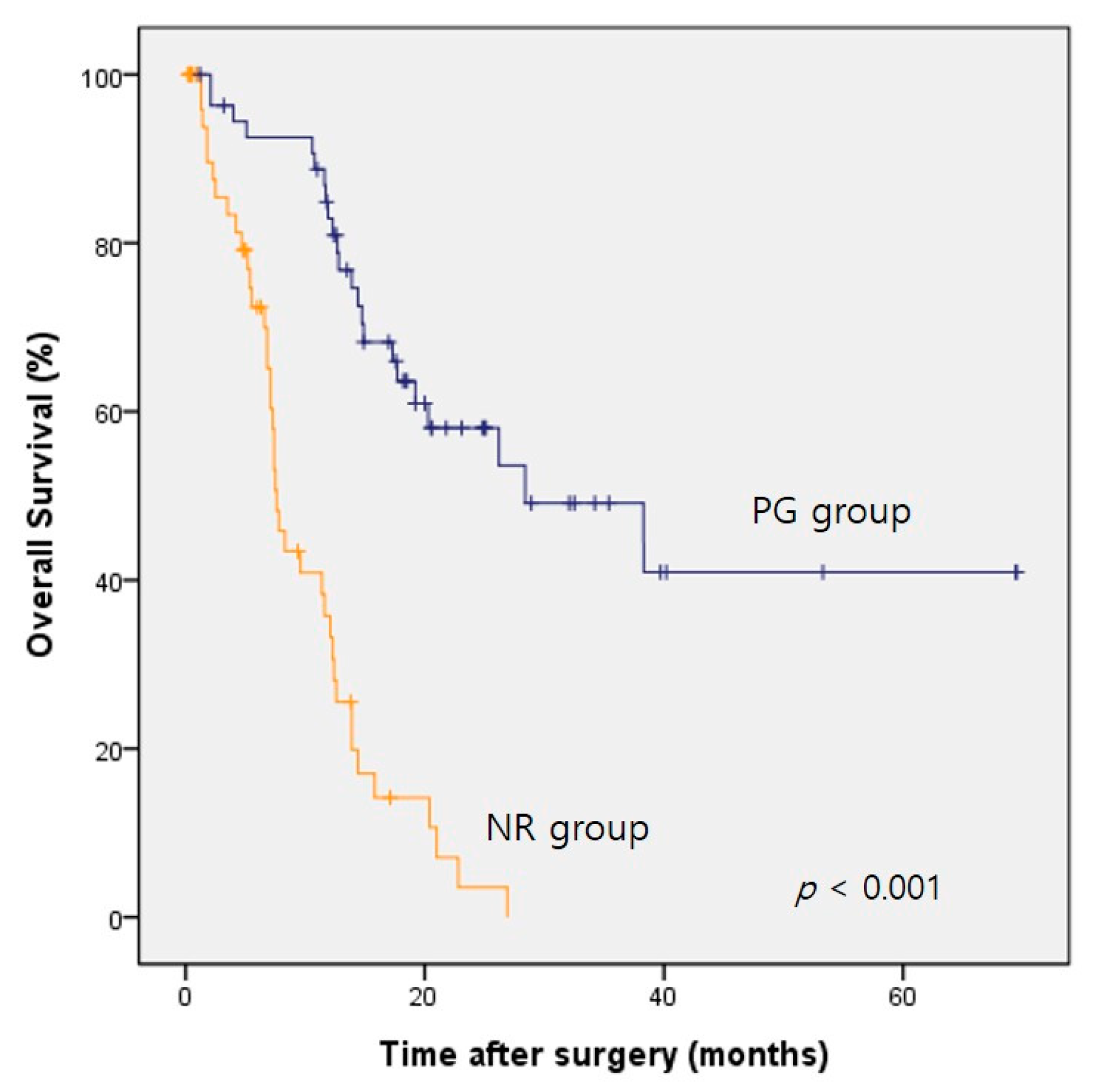
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Choi, K.S.; Jun, J.K.; Suh, M.; Park, B.; Noh, D.K.; Song, S.H.; Jung, K.W.; Lee, H.Y.; Choi, I.J.; Park, E.C. Effect of endoscopy screening on stage at gastric cancer diagnosis: Results of the National Cancer Screening Programme in Korea. Br. J. Cancer 2015, 112, 608–612. [Google Scholar] [CrossRef] [PubMed]
- Eom, B.W.; Jung, K.W.; Won, Y.J.; Yang, H.; Kim, Y.W. Trends in Gastric Cancer Incidence According to the Clinicopathological Characteristics in Korea, 1999–2014. Cancer Res. Treat. 2018, 50, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.D.; Grothe, W.; Haerting, J.; Kleber, G.; Grothey, A.; Fleig, W.E. Chemotherapy in advanced gastric cancer: A systematic review and meta-analysis based on aggregate data. J. Clin. Oncol. 2006, 24, 2903–2909. [Google Scholar] [CrossRef]
- Sun, J.; Song, Y.; Wang, Z.; Chen, X.; Gao, P.; Xu, Y.; Zhou, B.; Xu, H. Clinical significance of palliative gastrectomy on the survival of patients with incurable advanced gastric cancer: A systematic review and meta-analysis. BMC Cancer 2013, 13, 577. [Google Scholar] [CrossRef] [PubMed]
- Lasithiotakis, K.; Antoniou, S.A.; Antoniou, G.A.; Kaklamanos, I.; Zoras, O. Gastrectomy for stage IV gastric cancer: A systematic review and meta-analysis. Anticancer Res. 2014, 34, 2079–2085. [Google Scholar]
- Fujitani, K.; Yang, H.K.; Mizusawa, J.; Kim, Y.W.; Terashima, M.; Han, S.U.; Iwasaki, Y.; Hyung, W.J.; Takagane, A.; Park, D.J.; et al. Gastrectomy plus chemotherapy versus chemotherapy alone for advanced gastric cancer with a single non-curable factor (REGATTA): A phase 3, randomised controlled trial. Lancet Oncol. 2016, 17, 309–318. [Google Scholar] [CrossRef]
- Kodera, Y. Surgery with curative intent for stage IV gastric cancer: Is it a reality of illusion? Ann. Gastroenterol. Surg. 2018, 2, 339–347. [Google Scholar] [CrossRef]
- Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2014 (ver. 4). Gastric Cancer 2017, 20, 1–19. [Google Scholar] [CrossRef]
- Cunningham, D.; Allum, W.H.; Stenning, S.P.; Thompson, J.N.; Van de Velde, C.J.; Nicolson, M.; Scarffe, J.H.; Lofts, F.J.; Falk, S.J.; Iveson, T.J.; et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N. Engl. J. Med. 2006, 355, 11–20. [Google Scholar] [CrossRef]
- Aoyama, T.; Yoshikawa, T.; Shirai, J.; Hayashi, T.; Yamada, T.; Tsuchida, K.; Hasegawa, S.; Cho, H.; Yukawa, N.; Oshima, T.; et al. Body weight loss after surgery is an independent risk factor for continuation of S-1 adjuvant chemotherapy for gastric cancer. Ann. Surg. Oncol. 2013, 20, 2000–2006. [Google Scholar] [CrossRef]
- Yamashita, K.; Kurokawa, Y.; Yamamoto, K.; Hirota, M.; Kawabata, R.; Mikami, J.; Masuzawa, T.; Takiguchi, S.; Mori, M.; Doki, Y. Risk Factors for Poor Compliance with Adjuvant S-1 Chemotherapy for Gastric Cancer: A Multicenter Retrospective Study. Ann. Surg. Oncol. 2017, 24, 2639–2645. [Google Scholar] [CrossRef] [PubMed]
- Otowa, Y.; Suzuki, S.; Kanaji, S.; Harada, H.; Tanaka, T.; Yamamoto, M.; Matsuda, Y.; Oshikiri, T.; Nakamura, T.; Kakeji, Y. Curative Gastrectomy with Perioperative Chemotherapy Improves the Survival for Unresectable Gastric Cancer. Anticancer Res. 2018, 38, 2363–2368. [Google Scholar] [PubMed]
- Samarasam, I.; Chandran, B.S.; Sitaram, V.; Perakath, B.; Nair, A.; Mathew, G. Palliative gastrectomy in advanced gastric cancer: Is it worthwhile? ANZ J. Surg. 2006, 76, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, K.; Yoshida, K.; Tanahashi, T.; Takahashi, T.; Matsuhashi, N.; Tanaka, Y.; Tanabe, K.; Ohdan, H. The long-term survival of stage IV gastric cancer patients with conversion therapy. Gastric Cancer 2018, 21, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Fukuchi, M.; Ishiguro, T.; Ogata, K.; Suzuki, O.; Kumagai, Y.; Ishibashi, K.; Ishida, H.; Kuwano, H.; Mochiki, E. Prognostic Role of Conversion Surgery for Unresectable Gastric Cancer. Ann. Surg. Oncol. 2015, 22, 3618–3624. [Google Scholar] [CrossRef]
- Sato, Y.; Ohnuma, H.; Nobuoka, T.; Hirakawa, M.; Sagawa, T.; Fujikawa, K.; Takahashi, Y.; Shinya, M.; Katsuki, S.; Takahashi, M.; et al. Conversion therapy for inoperable advanced gastric cancer patients by docetaxel, cisplatin, and S-1 (DCS) chemotherapy: A multi-institutional retrospective study. Gastric Cancer 2017, 20, 517–526. [Google Scholar] [CrossRef]
- Ishigami, H.; Yamaguchi, H.; Yamashita, H.; Asakage, M.; Kitayama, J. Surgery after intraperitoneal and systemic chemotherapy for gastric cancer with peritoneal metastasis or positive peritoneal cytology findings. Gastric Cancer 2017, 20, 128–134. [Google Scholar] [CrossRef]
- Morgagni, P.; Solaini, L.; Framarini, M.; Vittimberga, G.; Gardini, A.; Tringali, D.; Valgiusti, M.; Monti, M.; Ercolani, G. Conversion surgery for gastric cancer: A cohort study from a western center. Int. J. Surg. 2018, 53, 360–365. [Google Scholar] [CrossRef]
- Beom, S.H.; Choi, Y.Y.; Baek, S.E.; Li, S.X.; Lim, J.S.; Son, T.; Kim, H.I.; Cheong, J.H.; Hyung, W.J.; Choi, S.H.; et al. Multidisciplinary treatment for patients with stage IV gastric cancer: The role of conversion surgery following chemotherapy. BMC Cancer 2018, 18, 1116. [Google Scholar] [CrossRef]
- Seo, H.S.; Song, K.Y.; Jung, Y.J.; Park, S.M.; Jeon, H.M.; Kim, W.; Chin, H.M.; Kim, J.J.; Kim, S.K.; Chun, K.H.; et al. Radical Gastrectomy After Chemotherapy May Prolong Survival in Stage IV Gastric Cancer: A Korean Multi-institutional Analysis. World J. Surg. 2018, 42, 3286–3293. [Google Scholar] [CrossRef]
- Yoshida, K.; Yamaguchi, K.; Okumura, N.; Tanahashi, T.; Kodera, Y. Is conversion therapy possible in stage IV gastric cancer: The proposal of new biological categories of classification. Gastric Cancer 2016, 19, 329–338. [Google Scholar] [CrossRef] [PubMed]

| Variables | No. of Patients (%) |
|---|---|
| Age (years) | 60.6 ± 14.1 |
| Sex | |
| Male | 95 (64.2) |
| Female | 53 (35.8) |
| Body mass index (kg/m2) | 22.2 ± 3.2 |
| ASA-PS score | |
| 1 | 51 (34.5) |
| 2 | 85 (57.4) |
| 3 | 11 (7.4) |
| 4 | 1 (0.7) |
| Histology | |
| Differentiated | 39 (26.4) |
| Undifferentiated | 106 (71.6) |
| Unknown | 3 (2.0) |
| Gross type | |
| Bormann type I | 1 (0.7) |
| Bormann type II | 13 (8.8) |
| Bormann type III | 77 (52.0) |
| Bormann type IV | 38 (25.7) |
| Unspecified | 19 (12.8) |
| Metachronous cancer in the remnant stomach | 6 (4.1) |
| Outlet obstruction | 14 (9.5) |
| Bleeding from cancer | 5 (3.4) |
| Preoperative radiologic findings | |
| Adjacent organ invasion | 19 (12.8) |
| Metastatic disease | 33 (22.3) |
| Tumor markers at baseline | |
| CEA (ng/mL) | 1.8 (0.4–255.3) |
| CA 19-9 (U/mL) | 15.9 (0.2–1822.9) |
| CA 125 (U/mL) | 8.8 (0.9–200.8) |
| Type of surgery | |
| Palliative resection | 55 (37.2) |
| Distal gastrectomy | 28 (18.9) |
| Total gastrectomy | 25 (16.9) |
| Whipple’s operation | 2 (1.4) |
| Nonresection | 93 (62.8) |
| Bypass surgery (including gastrojejunostomy) | 39 (26.4) |
| Opening and closure | 54 (36.5) |
| Combined resection | 24 (16.2) |
| Extent of lymph-node dissection | |
| D0 | 93 (62.8) |
| Less than D2 | 4 (2.7) |
| D2 or more | 51 (34.5) |
| Reason for incurability | |
| Advanced primary disease | 65 (43.9) |
| Extended nodal disease | 21 (14.2) |
| Metastatic disease | 120 (81.1) |
| Peritoneal seeding | 100 (67.6) |
| Hepatic metastasis | 9 (6.1) |
| Metastasis to other organs (except for liver) | 9 (6.1) |
| Two or more noncurative factors | 48 (32.4) |
| Intraperitoneal chemotherapy † | 14 (9.5) |
| Postoperative chemotherapy | 102 (68.9) |
| Overall survival (months) | 10.9 (0.2–69.5) |
| Variables | Before Matching | After Matching | ||||
|---|---|---|---|---|---|---|
| PG Group (n = 55) | NR Group (n = 93) | p-Value | PG Group (n = 55) | NR Group (n = 55) | p-Value | |
| Age at operation (years) | 54.0 ± 14.1 | 64.5 ± 12.6 | <0.001 | 54.0 ± 14.1 | 59.9 ± 12.1 | 0.020 |
| Sex | ||||||
| Male | 28 (50.9%) | 67 (72.0%) | 0.013 | 28 (50.9%) | 36 (65.5%) | 0.122 |
| Female | 27 (49.1%) | 26 (28.0%) | 27 (49.1%) | 19 (34.5%) | ||
| ASA-PS classification | 0.054 | 0.076 | ||||
| 1 | 21 (38.2%) | 30 (32.3%) | 21 (38.2%) | 16 (29.1%) | ||
| 2 | 26 (47.3%) | 59 (63.4%) | 26 (47.3%) | 37 (67.3%) | ||
| 3 | 7 (12.7%) | 4 (4.3%) | 8 (14.5%) | 2 (3.6%) | ||
| 4 | 1 (1.8%) | 0 (0%) | 1 (1.8%) | 0 (0%) | ||
| Clinical stage * | ||||||
| T category | 0.109 | 0.495 | ||||
| T2 | 3 (5.5%) | 2 (2.2%) | 3 (5.5%) | 1 (1.8%) | ||
| T3 | 9 (16.4%) | 18 (19.4%) | 9 (16.4%) | 12 (21.8%) | ||
| T4a | 24 (43.6%) | 26 (28.0%) | 24 (43.6%) | 19 (34.5%) | ||
| T4b | 19 (34.5%) | 47 (50.5%) | 19 (34.5%) | 23 (41.8%) | ||
| N category | 0.311 | |||||
| N0 | 5 (9.1%) | 8 (8.6%) | 0.086 | 5 (9.1%) | 4 (7.3%) | |
| N1 | 7 (12.7%) | 24 (25.8%) | 7 (12.7%) | 13 (23.6%) | ||
| N2 | 24 (43.6%) | 45 (48.4%) | 24 (43.6%) | 27 (49.1%) | ||
| N3a | 18 (32.7%) | 15 (16.1%) | 18 (32.7%) | 11 (20.0%) | ||
| N3b | 1 (1.8%) | 1 (1.1%) | 1 (1.8%) | 0 (0%) | ||
| Reason for incurability | ||||||
| Advanced primary disease | 19 (34.5%) | 46 (54.1%) | 0.025 | 19 (34.5%) | 23 (41.8%) | 0.432 |
| Extended nodal disease | 7 (12.7%) | 10 (10.8%) | 0.792 | 7 (12.7%) | 7 (12.7%) | >0.999 |
| Metastatic disease | ||||||
| Peritoneal seeding | 41 (74.5%) | 59 (63.4%) | 0.204 | 41 (74.5%) | 37 (67.3%) | 0.401 |
| Hepatic metastasis | 1 (1.8%) | 8 (8.6%) | 0.154 | 1 (1.8%) | 1 (1.8%) | >0.999 |
| Metastasis to other organs (except for liver) | 5 (9.1%) | 4 (4.3%) | 0.293 | 5 (9.1%) | 3 (5.5%) | 0.716 |
| Two or more noncurative factors | 20 (36.4%) | 28 (30.1%) | 0.470 | 20 (36.4%) | 13 (23.6%) | 0.146 |
| Postoperative complications † | <0.001 | 0.003 | ||||
| None | 44 (80.0%) | 90 (96.8%) | 44 (80.0%) | 54 (98.2%) | ||
| Grade I | 7 (12.7%) | 1 (1.1%) | 7 (12.7%) | 0 (0%) | ||
| Grade II | 3 (5.5%) | 2 (2.2%) | 3 (5.5%) | 1 (1.8%) | ||
| Grade IV | 1 (1.8%) | 0 | 1 (1.8%) | 0 (0%) | ||
| Length of hospital stay (days) | 13.0 ± 11.1 | 9.3 ± 7.3 | 0.017 | 13.0 ± 11.1 | 9.5 ± 8.0 | 0.063 |
| Preoperative chemotherapy | 4 (7.3%) | 6 (6.5%) | >0.999 | 4 (7.3%) | 5 (9.1%) | >0.999 |
| Intraperitoneal chemotherapy | 14 (25.5%) | 0 | <0.001 | 14 (25.5%) | 0 (0%) | <0.001 |
| Postoperative chemotherapy | 47 (85.5%) | 55 (59.1%) | 0.001 | 47 (85.5%) | 31 (56.4%) | 0.001 |
| No. of chemotherapy cycles ‡ | 13 (1–104) | 8 (1–44) | 0.004 | 13 (1–104) | 8 (1–39) | 0.027 |
| Variables | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Palliative resection (vs. nonresection) | 0.185 (0.106–0.321) | <0.001 | 0.169 (0.089–0.321) | <0.001 |
| Age < 70 years (vs. age ≥ 70) | 0.545 (0.307–0.969) | 0.039 | 0.617 (0.322–1.181) | 0.145 |
| Male (vs. female) | 1.382 (0.832–2.295) | 0.211 | ||
| ASA-PS ≤ 1 | 0.476 (0.270–0.842) | 0.011 | 0.425 (0.215–0.838) | 0.014 |
| Outlet obstruction | 1.466 (0.631–3.409) | 0.374 | ||
| Differentiated (vs. undifferentiated) | 0.943 (0.527–1.685) | 0.842 | ||
| Tumor markers | ||||
| CEA > 7 | 1.390 (0.502–3.849) | 0.527 | ||
| CA 19-9 > 37 | 0.858 (0.476–1.545) | 0.609 | ||
| CA 125 > 30 | 2.020 (0.908–4.494) | 0.085 | 1.373 (0.532–3.543) | 0.512 |
| Surgical findings | ||||
| Advanced primary disease | 1.127 (0.676–1.880) | 0.646 | ||
| Extended nodal disease | 1.435 (0.705–2.921) | 0.319 | ||
| Peritoneal seeding | 0.662 (0.389–1.127) | 0.129 | ||
| Hepatic metastasis | 0.627 (0.087–4.549) | 0.645 | ||
| Neoadjuvant chemotherapy | 1.983 (0.899–4.371) | 0.090 | 0.797 (0.270–2.350) | 0.681 |
| Intraperitoneal chemotherapy | 0.561 (0.255–1.234) | 0.151 | ||
| Postoperative chemotherapy | 0.326 (0.189–0.563) | <0.001 | 0.665 (0.360–1.231) | 0.194 |
| Variables | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Age < 70 years (vs. age ≥ 70) | 0.432 (0.174–1.072) | 0.070 | 1.170 (0.371–3.689) | 0.788 |
| Male (vs. female) | 1.246 (0.545–2.848) | 0.601 | ||
| ASA-PS ≤ 1 | 0.409 (0.152–1.104) | 0.078 | 0.528 (0.181–1.537) | 0.241 |
| Differentiated (vs. undifferentiated) | 0.850 (0.252–2.867) | 0.793 | ||
| Extent of surgery | ||||
| Distal gastrectomy | Ref. | 0.624 | ||
| Total gastrectomy | 0.845 (0.365–1.956) | 0.695 | ||
| Whipples’ operation | 2.308 (0.292–18.252) | 0.428 | ||
| LND ≥ D2 (vs. LND < D2) | 1.527 (0.204–11.419) | 0.680 | ||
| Pathologic findings | ||||
| No. of positive nodes < 15 | 0.351 (0.148–0.832) | 0.014 | 0.327 (0.129–0.831) | 0.019 |
| Peritoneal seeding | 0.862 (0.339–2.190) | 0.755 | ||
| Extended nodal disease | 3.472 (1.237–9.745) | 0.018 | 3.078 (0.931–10.176) | 0.065 |
| Advanced primary disease | 1.017 (0.415–2.495) | 0.971 | ||
| Intraperitoneal chemotherapy | 1.380 (0.559–3.407) | 0.484 | ||
| Postoperative chemotherapy | 0.339 (0.112–1.024) | 0.055 | 0.258 (0.068–0.984) | 0.047 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.Y.; Yu, B.; Park, K.B.; Kwon, O.K.; Lee, S.S.; Chung, H.Y. Impact of Palliative Gastrectomy in Patients with Incurable Gastric Cancer. Medicina 2021, 57, 198. https://doi.org/10.3390/medicina57030198
Park JY, Yu B, Park KB, Kwon OK, Lee SS, Chung HY. Impact of Palliative Gastrectomy in Patients with Incurable Gastric Cancer. Medicina. 2021; 57(3):198. https://doi.org/10.3390/medicina57030198
Chicago/Turabian StylePark, Ji Yeon, Byunghyuk Yu, Ki Bum Park, Oh Kyoung Kwon, Seung Soo Lee, and Ho Young Chung. 2021. "Impact of Palliative Gastrectomy in Patients with Incurable Gastric Cancer" Medicina 57, no. 3: 198. https://doi.org/10.3390/medicina57030198
APA StylePark, J. Y., Yu, B., Park, K. B., Kwon, O. K., Lee, S. S., & Chung, H. Y. (2021). Impact of Palliative Gastrectomy in Patients with Incurable Gastric Cancer. Medicina, 57(3), 198. https://doi.org/10.3390/medicina57030198






