1. Introduction
The Micra system (Micra TPS, Medtronic, Minneapolis, MN), which is the first leadless pacemaker, is a lead- and pocket-less implantable device having a high successful implantation rate (99.2%), a low incidence of complications (4%), and a 93.3% 6-month freedom from serious adverse events [
1,
2,
3,
4]. The post-approval registry also demonstrates similar favorable outcomes in terms of implantation and complications [
5].
Recently, we reported a patient with a greatly enlarged right atrium accompanied by atrial septal deficiency in whom a leadless pacemaker failed to be implanted [
6]. Instead, a conventional pacemaker was successfully implanted via the subclavian vein. Another team also reported a challenging case for leadless pacemaker implantation with an enlarged right heart [
7]. However, there have been few studies investigating right heart anatomical parameters associating with longer procedure time. Such data would be useful in determining the optimal therapeutic strategy, i.e., a Micra system or conventional pacemaker, for each patient. The present study aimed to investigate echocardiographic parameters associating with a longer procedure time for implanting the Micra leadless pacemaker.
2. Materials and Methods
2.1. Study Population
Consecutive patients who underwent implantation of the Micra system successfully at our institute between August 2019 and March 2021 were included retrospectively. All patients had bradycardia due to atrial fibrillation, sick sinus syndrome, or advanced atrioventricular block. The present study was approved by the institutional review board at the University of Toyama (R2020025 approved on 1 May 2020). Informed consent was obtained from all patients.
2.2. Baseline Clinical Characteristics
Baseline clinical characteristics, including demographics, comorbidity, and laboratory data, were retrieved from the electronic medical record.
2.3. Right Heart Parameters and Procedure Times
Echocardiographic parameters including right atrial diameter, right atrial area, right ventricular end-diastolic area, right ventricular basal diameter, and right ventricular mid diameter were measured according to the current guideline using the four-chamber views focusing on the right heart by an expert cardiologist blinded to the study data within 30 days before Micra implantation [
8].
The procedure time for the implantation was defined as a time from the infusion of local anesthesia to the sheath withdrawal. A procedure time > 60 min was defined as the primary endpoint.
2.4. Statistical Analysis
Data were expressed as median (interquartile range) and compared between the groups using the Mann–Whitney U test. Categorical data were expressed as numbers (percentages) and compared using Fischer’s exact test. Univariable logistic regression analyses were performed to investigate echocardiographic right heart parameters associating with the procedure time > 60 min. Receiver operating characteristics analyses were performed to calculate cutoffs to predict a procedure time > 60 min. A two-sided p value < 0.05 was considered as statistically significant. Data analysis was performed using SPSS Statistics 22 (SPSS Inc, Armonk, IL, USA).
3. Results
3.1. Patient Characteristics
Nineteen patients (median 81 years old, 10 male) were included (
Table 1). Of them, 6 (32%) were in the procedure time > 60 min group. Age, sex, body mass index, atrial fibrillation indication, hypertension, chronic obstructive pulmonary disease, and dialysis, all of which have been reported as risk factors for Micra system-associated complications, were statistically not significantly different between the two groups stratified by the procedure time [
3]. However, both atrial and ventricular parameters were significantly higher in the group with procedure time > 60 min than in the group with procedure time ≤ 60 min in the univariable analysis.
3.2. Right Heart Parameters and the Procedure Time
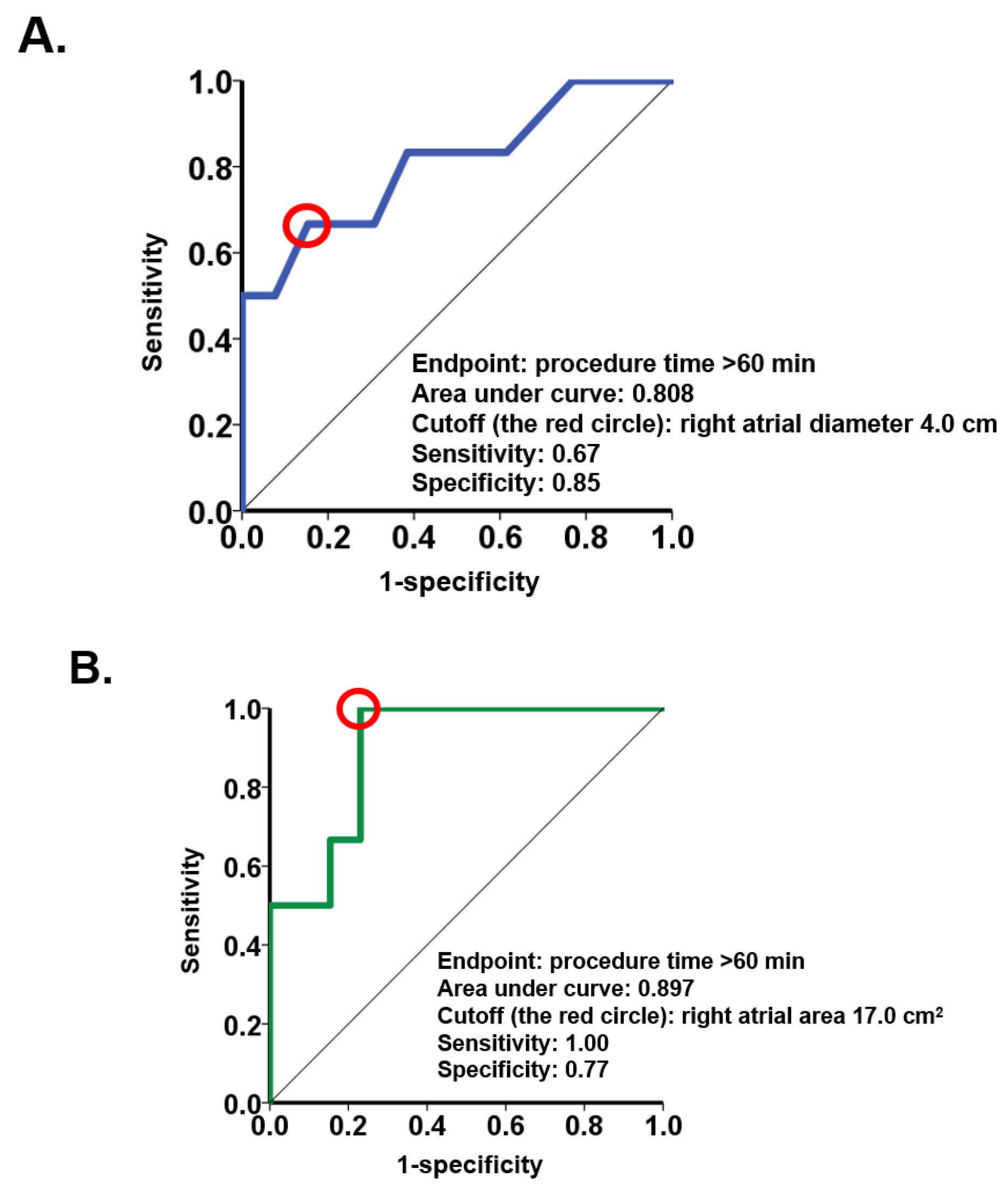
Echocardiographic right heart parameters obtained before the procedures were analyzed in the logistic regression. Baseline right atrial diameter and right atrial area were associated with procedure time > 60 min with an odds ratio of 11.3 (95% confidence interval 1.09–117;
p = 0.042) for right atrial diameter and 1.57 (95% confidence interval 1.05–2.34;
p = 0.029) for right atrial area (
Table 2). The parameters of the right ventricle failed to show significance. ROC analyses demonstrated a cutoff of 4.0 cm for right atrial diameter and a cutoff of 17.0 cm
2 for right atrial area to predict procedure time >60 min (
Figure 1).
Procedure time was significantly longer in the patients with right atrial diameter above the cutoff (63 (50, 100) minutes versus 42 (25, 50) minutes,
p = 0.022;
Figure 2A). In the same manner, patients with right atrial area above the cutoff had longer procedure time (65 (50, 70) minutes versus 33 (25, 45) minutes,
p = 0.001;
Figure 2B).
The two cases with the shortest and longest procedure times are displayed in
Figure 3A,B, respectively. The patient of
Figure 3A had a smaller right atrium with 3.3 cm right atrial diameter and 14.7 cm
2 right atrial area, and a procedure time of 30 min. The patient of
Figure 3B had a larger right atrium with 4.3 cm right atrial diameter and 24.7 cm
2 right atrial area, and the procedure time was 185 min (
Figure 3B).
4. Discussion
This is the first study demonstrating the association between right atrial size and procedure time for implanting the Micra leadless pacemaker.
5. Previously Reported Risk Factors of Procedure-Related Complications
Several baseline characteristics, including body mass index < 20 kg/m
2, age ≥ 85 years, female sex, non-atrial fibrillation indication, and chronic lung disease, were reported as the risk factors for procedure-related complications, such as perforation and pericardial effusion [
9]. Detailed explanations are unknown, but these parameters may be associated with a small heart cavity that hinders safe catheter procedure and increases the risk of cardiac injury. In this study, we had no such critical complications, probably due to the very low incidence rate of these complications in general.
However, we sometimes experience Micra implantation procedures that required >60 min. A longer procedure time would be associated with minor procedure-related troubles, which might result in critical complications. A longer procedure time would also be associated with more radiation exposure. We previously experienced a case in which Micra implantation failed with a prolonged procedure time, but a conventional pacemaker was easily implanted via the subclavian vein. Factors associated with a longer procedure time would be favor choice of a conventional pacemaker over the Micra system beforehand. This is why we investigated factors associating with longer procedure time. We defined >60 min of procedure time as a challenging procedure, considering that 45 ± 15 min is the average procedure time for Micra implantation according to a previous paper [
10].
6. The Implications of Enlarged Right Heart for the Micra Implantation
A previous case report showed that a patient with an enlarged right heart due to a rheumatic fever required a unique technique to deliver the Micra system [
7]. Our case report also presented a greatly enlarged right heart due to an atrial septum defect leading to failed Micra implantation [
6]. However, there have been few studies investigating the association between cardiac anatomical features and difficulty in Micra implantation.
The Micra system should tightly contact the right ventricular septum to make a “gooseneck sign”. In patients with an enlarged right atrium, the delivery sheath may be far away from the right ventricular septum, making it difficult to contact the septum tightly compared to patients with a small right atrium (
Figure 4). This may be why right atrial size is associated with lengthened procedure times and not ventricular features.
We proposed cutoffs of right atrial diameter and area at 4.0 cm and 17 cm
2, respectively. When a patient’s right atrium reaches one of these cutoffs, the Micra implantation may be challenging, and we recommend conventional pacemaker implantation via the subclavian vein. Micra implantation via jugular vein is a feasible alternative, although specific device and implant techniques are required [
11].
7. Study Limitations
The present study has several limitations. This is a single-center retrospective observational study with a small sample size; further large-scale studies are warranted. The parameters associated with procedure time > 60 min were detected by univariable but not multivariable analysis, probably due to the small sample size. We included standard parameters to assess right heart size, but other unique parameters, for example, tricuspid valve regurgitation or pulmonary artery hypertension may exist to show the anatomical and functional features of right heart. The prognostic impacts of the prolonged procedure time should be assessed in further prospective studies. Finally, the proficiency of the operators, who were two board-certificated attending doctors in this study, or the length of vascular access procedure may affect the total procedure time.
8. Conclusions
Although larger-scale studies are needed, the diameter and area of the right atrium were associated with a Micra system implantation procedure time > 60 min in this study.
Author Contributions
Conceptualization, N.K. and T.I.; methodology, T.I.; software, T.I.; validation, N.K., T.K., and H.U.; formal analysis, T.I.; investigation, N.K. and T.K.; resources, N.K., T.K., and H.U.; data curation, N.K., T.I., and T.K.; writing—original draft preparation, N.K.; writing—review and editing, T.K.; visualization, N.K. and T.I.; supervision, K.K.; project administration, K.K.; funding acquisition, N.K. and T.I. All authors have read and agreed to the published version of the manuscript.
Funding
This work was partly supported by JSPS KAKENHI Grant Number JP19K20724 to N.K. and JSPS KAKENHI Grant Number JP20K17143 to T.I.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Ethics Committee of University of Toyama (protocol code R2020025 and approved at 1 May 2020).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Conflicts of Interest
The authors declare no conflict of interest associated with this manuscript.
References
- Okabe, T.; Afzal, M.R.; Houmsse, M.; Makary, M.S.; Elliot, E.D.; Daoud, E.G.; Augostini, R.S.; Hummel, J.D. Tine-based leadless pacemaker: Strategies for safe implantation in unconventional clinical scenarios. Clin. Electrophysiol. 2020, 6, 1318–1331. [Google Scholar] [CrossRef] [PubMed]
- Duray, G.Z.; Ritter, P.; El-Chami, M.; Narasimhan, C.; Omar, R.; Tolosana, J.M.; Zhang, S.; Soejima, K.; Steinwender, C.; Rapallini, L.; et al. Long-term performance of a transcatheter pacing system: 12-Month results from the Micra Transcatheter Pacing Study. Heart Rhythm 2017, 14, 702–709. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reynolds, D.; Duray, G.Z.; Omar, R.; Soejima, K.; Neuzil, P.; Zhang, S.; Narasimhan, C.; Steinwender, C.; Brugada, J.; Lloyd, M.; et al. A leadless intracardiac transcatheter pacing system. N. Engl. J. Med. 2016, 374, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.Y.; Exner, D.V.; Cantillon, D.J.; Doshi, R.; Bunch, T.J.; Tomassoni, G.F.; Friedman, P.A.; Mark Estes, N.A., III; Ip, J.; Niazi, I.; et al. Percutaneous implantation of an entirely intracardiac leadless pacemaker. N. Engl. J. Med. 2015, 373, 1125–1135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roberts, P.R.; Clementy, N.; Al Samadi, F.; Garweg, C.; Martinez-Sande, J.L.; Iacopino, S.; Johansen, J.B.; Prat, X.V.; Kowal, R.C.; Klug, D.; et al. A leadless pacemaker in the real-world setting: The Micra Transcatheter Pacing System Post-Approval Registry. Heart Rhythm 2017, 14, 1375–1379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imamura, T.; Koi, T.; Kataoka, N.; Tanaka, S.; Ueno, H.; Kinugawa, K. Micra trans-catheter leadless pacemaker implantation in a patient with large right heart. J. Cardiol. Cases 2020, in press. [Google Scholar]
- Alyesh, D.; Cunnane, R. Successful leadless pacemaker deployment in a patient with challenging right heart anatomy using a double snare technique. HeartRhythm Case Rep. 2019, 5, 399–401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. Heart J.-Cardiovasc. Imaging 2015, 16, 233–271. [Google Scholar] [CrossRef] [PubMed]
- El-Chami, M.F.; Al-Samadi, F.; Clementy, N.; Garweg, C.; Martinez-Sande, J.L.; Piccini, J.P.; Iacopino, S.; Lloys, M.; Prat, X.V.; Jacobsen, M.D.; et al. Updated performance of the Micra transcatheter pacemaker in the real-world setting: A comparison to the investigational study and a transvenous historical control. Heart Rhythm 2018, 15, 1800–1807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garweg, C.; Vandenberk, B.; Foulon, S.; Hermans, P.; Poels, P.; Haemers, P.; Ector, J.; Willems, R. Determinants of the difficulty of leadless pacemaker implantation. Pacing Clin. Electrophysiol. 2020, 43, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Kolek, M.J.; Crossley, G.H.; Ellis, C.R. Implantation of a MICRA leadless pacemaker via right internal jugular vein. JACC Clin. Electrophysiol. 2018, 4, 420–421. [Google Scholar] [CrossRef] [PubMed]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).