The Short and Intensive Rehabilitation (SHAiR) Program Improves Dropped Head Syndrome Caused by Amyotrophic Lateral Sclerosis: A Case Report
Abstract
:1. Introduction
2. Case Report
2.1. Patient Information
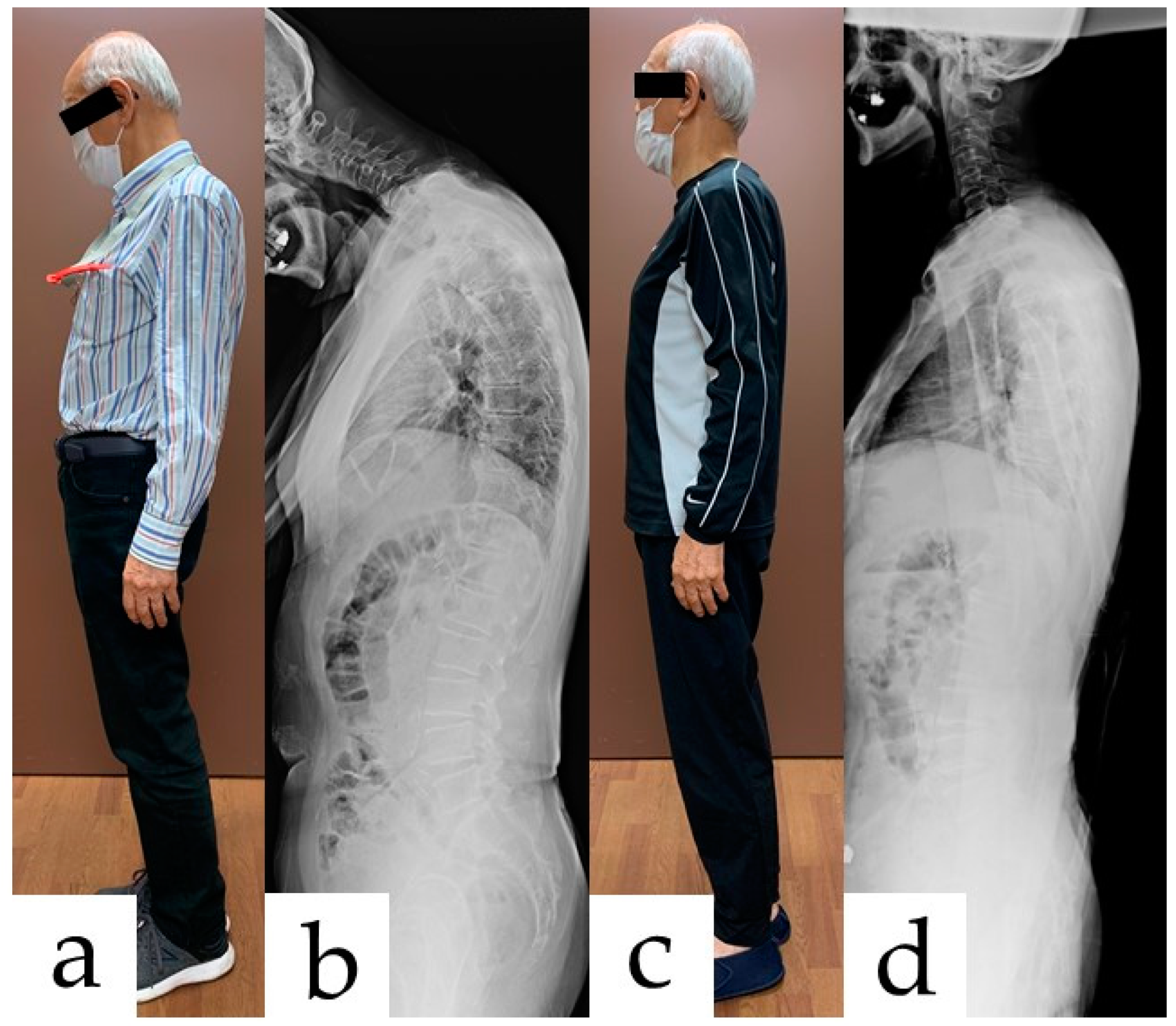
2.2. Clinical Findings and Diagnosis
2.3. Therapeutic Intervention
2.4. Follow-Up and Outcomes
3. Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sharan, A.D.; Kaye, D.; Charles Malveaux, W.M.; Riew, K.D. Dropped head syndrome: Etiology and management. J. Am. Acad. Orthop. Surg. 2012, 20, 766–774. [Google Scholar] [CrossRef] [PubMed]
- Drain, J.P.; Virk, S.S.; Jain, N.; Yu, E. Dropped Head Syndrome: A Systematic Review. Clin. Spine Surg. 2019, 32, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Gourie-Devi, M.; Nalini, A.; Sandhya, S. Early or late appearance of “dropped head syndrome” in amyotrophic lateral sclerosis. J. Neurol. Neurosurg. Psychiatry 2003, 74, 683–686. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uemura, M.; Kosaka, T.; Shimohata, T.; Ishikawa, M.; Nishihira, Y.; Toyoshima, Y.; Yanagawa, K.; Kawachi, I.; Takahashi, H.; Nishizawa, M. Dropped head syndrome in amyotrophic lateral sclerosis. Amyotroph. Lateral. Scler. Frontotemporal. Degener. 2013, 14, 232–233. [Google Scholar] [CrossRef] [PubMed]
- Igawa, T.; Isogai, N.; Suzuki, A.; Kusano, S.; Sasao, Y.; Nishiyama, M.; Funao, H.; Ishii, K. Establishment of a novel rehabilitation program for patients with dropped head syndrome: Short and intensive rehabilitation (SHAiR) program. J. Clin. Neurosci. 2020, 73, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.R.; Reddy, R.; Fehlings, M.G. Dropped head syndrome: Diagnosis and management. Evid. Based Spine Care J. 2011, 2, 41–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanikawa, S.; Tanino, M.; Wang, L.; Ishikawa, M.; Miyazaki, M.; Tsuda, M.; Orba, Y.; Sawa, H.; Matoba, K.; Nakamura, N.; et al. Autopsy findings in the early stage of amyotrophic lateral sclerosis with “dropped head” syndrome. Neuropathology 2019, 39, 374–377. [Google Scholar] [CrossRef]
- Brodell, J.D., Jr.; Sulovari, A.; Bernstein, D.N.; Mongiovi, P.C.; Ciafaloni, E.; Rubery, P.T.; Mesfin, A. Dropped Head Syndrome: An Update on Etiology and Surgical Management. JBJS Rev. 2020, 8, e0068. [Google Scholar] [CrossRef]
- Czell, D.; Weber, M. The head-up-orthosis—A good solution for ambulant patients with Dropped-head-syndrome. Praxis 2012, 101, 1499–1502. [Google Scholar] [CrossRef]
- Hansen, A.; Bedore, B.; Nickel, E.; Hanowski, K.; Tangen, S.; Goldish, G. Elastic head support for persons with amyotrophic lateral sclerosis. J. Rehabil. Res. Dev. 2014, 51, 297–303. [Google Scholar] [CrossRef] [Green Version]
- Baxter, S.; Reed, H.; Clarke, Z.; Judge, S.; Heron, N.; Mccarthy, A.; Langley, J.; Stanton, A.; Wells, O.; Squire, G.; et al. Evaluating a novel cervical orthosis, the Sheffield Support Snood, in patients with amyotrophic lateral sclerosis/motor neuron disease with neck weakness. Amyotroph. Lateral. Scler. Frontotemporal. Degener. 2016, 17, 436–442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reed, H.; Langley, J.; Stanton, A.; Heron, N.; Clarke, Z.; Judge, S.; McCarthy, A.; Squire, G.; Quinn, A.; Wells, O.; et al. Head-Up; An interdisciplinary, participatory and co-design process informing the development of a novel head and neck support for people living with progressive neck muscle weakness. J. Med. Eng. Technol. 2014, 39, 404–410. [Google Scholar] [CrossRef]
- Thumbikat, P.; Bailey, C.; Datta, D. Orthoses for neck control. ACNR 2006, 6, 18–19. [Google Scholar]
- Lin, H.N.; Nagaoka, M.; Hayashi, Y.; Yonezawa, I. Pathophysiological analysis of dropped head syndrome caused by various diagnoses—Based on surface EMG findings and responses to physiotherapy. Rinsho Shinkeigaku 2013, 53, 430–438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahnof, R. The dropped head syndrome: Rehabilitation of cervical focal myositis. Disabil. Rehabil. 1999, 21, 563–565. [Google Scholar] [CrossRef] [PubMed]
- Stefano, V.D.; Attanasi, C.; Ferrante, C.; Di Muzio, A. Late onset of dropped head syndrome following mantle radiation therapy for Hodgkin lymphoma. BMJ Case Rep. 2018, 11, e226822. [Google Scholar] [CrossRef] [PubMed]
- Kusakabe, T.; Endo, K.; Sawaji, Y.; Suzuki, H.; Nishimura, H.; Matsuoka, Y.; Murata, K.; Takamatsu, T.; Maekawa, A.; Aihara, T.; et al. Mode of onset of dropped head syndrome and efficacy of conservative treatment. J. Orthop. Surg. 2020, 28, 2309499020938882. [Google Scholar] [CrossRef] [PubMed]
- Macé, Y.; Yahia, M.; Rannou, F.; Lefevre-Colau, M.M.; Poiraudeau, S.; Revel, M. Value of intensive rehabilitation in fixed dropped head syndrome. Ann. Readapt. Med. Phys. 2005, 48, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Petheram, T.G.; Hourigan, P.G.; Emran, I.M.; Weatherley, C.R. Dropped head syndrome: A case series and literature review. Spine 2008, 33, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Kadone, H.; Miura, K.; Kubota, S.; Abe, T.; Shimizu, Y.; Hada, Y.; Suzuki, K.; Sankai, Y.; Koda, M.; Yamazaki, M. Dropped Head Syndrome Attenuation by Hybrid Assistive Limb: A Preliminary Study of Three Cases on Cervical Alignment during Walking. Medicina 2020, 56, 291. [Google Scholar] [CrossRef] [PubMed]
- Miura, K.; Koda, M.; Kadone, H.; Kubota, S.; Shimizu, Y.; Kumagai, H.; Nagashima, K.; Mataki, K.; Fujii, K.; Noguchi, H.; et al. Gait training using a hybrid assistive limb (HAL) attenuates head drop: A case report. J. Clin. Neurosci. 2018, 52, 141–144. [Google Scholar] [CrossRef]
- Chu, E.C.; Wong, A.Y.; Lin, A.F. Isolated Neck Extensor Myopathy Associated with Cervical Spondylosis: A Case Report and Brief Review. Clin. Med. Insights Arthritis Musculoskelet. Disord. 2020, 13, 1179544120977844. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Mataki, K.; Shimizu, Y.; Matsuba, K.; Miura, K.; Takahashi, H.; Koda, M.; Kamada, H.; Yamazaki, M. Dropped Head Syndrome Treated with Physical Therapy Based on the Concept of Athletic Rehabilitation. Case Rep. Orthop. 2020, 2020, 8811148. [Google Scholar] [CrossRef] [PubMed]
- Bello-Haas, V.D.; Florence, J.M. Therapeutic exercise for people with amyotrophic lateral sclerosis or motor neuron disease. Cochrane Database Syst. Rev. 2013, 2013, CD005229. [Google Scholar] [CrossRef] [PubMed]
- Kato, N.; Hashida, G.; Kobayashi, M.; Konaka, K. Physical therapy improves lower limb muscle strength but not function in individuals with amyotrophic lateral sclerosis: A case series study. Ann. Phys. Rehabil. Med. 2018, 61, 108–110. [Google Scholar] [CrossRef] [PubMed]
- Körner, S.; Kollewe, K.; Abdulla, S.; Zapf, A.; Dengler, R.; Petri, S. Interaction of physical function, quality of life and depression in Amyotrophic lateral sclerosis: Characterization of a large patient cohort. BMC Neurol. 2015, 16, 84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caga, J.; Hsieh, S.; Lillo, P.; Dudley, K.; Mioshi, E. The impact of cognitive and behavioral symptoms on ALS patients and their caregivers. Front. Neurol. 2019, 11, 192. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, K.; Hosono, N.; Kawaguchi, Y. Validity, reliability and responsiveness of the Japanese version of the Neck Disability Index. J. Orthop. Sci. 2013, 18, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Rahmati, M.; Malakoutinia, F. Aerobic, resistance and combined exercise training for patients with amyotrophic lateral sclerosis: A systematic review and meta-analysis. Physiotherapy 2021, 113, 12–28. [Google Scholar] [CrossRef]
- Meng, L.; Li, X.; Li, C.; Tsang, R.C.C.; Chen, Y.; Ge, Y.; Gao, Q. Effects of Exercise in Patients with Amyotrophic Lateral Sclerosis: A Systematic Review and Meta-Analysis. Am. J. Phys. Med. Rehabil. 2020, 99, 801–810. [Google Scholar] [CrossRef] [PubMed]


| Physical Examination |
|---|
Upper motor neuron signs
|
Lower motor neuron signs
|
| Electrophysiological Examination |
|
|
|
| Blood and Cerebrospinal Fluid Examination |
|
|
| Before Rehabilitation | 2-Weeks after Rehabilitation | |
|---|---|---|
| NRS | ||
| Neck pain | 9 | 6 |
| NDI (points) | 23 | 10 |
| SF-36 | ||
| Physical functioning | 30.0 | 75.0 |
| Role physical | 6.3 | 12.5 |
| Bodily pain | 12.0 | 12.0 |
| General health | 35.0 | 57.0 |
| Vitality | 25.0 | 68.8 |
| Social functioning | 0.0 | 25.0 |
| Role emotional | 0.0 | 8.3 |
| Mental health | 25.0 | 75.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urata, R.; Igawa, T.; Suzuki, A.; Sasao, Y.; Isogai, N.; Funao, H.; Ishii, K. The Short and Intensive Rehabilitation (SHAiR) Program Improves Dropped Head Syndrome Caused by Amyotrophic Lateral Sclerosis: A Case Report. Medicina 2022, 58, 452. https://doi.org/10.3390/medicina58030452
Urata R, Igawa T, Suzuki A, Sasao Y, Isogai N, Funao H, Ishii K. The Short and Intensive Rehabilitation (SHAiR) Program Improves Dropped Head Syndrome Caused by Amyotrophic Lateral Sclerosis: A Case Report. Medicina. 2022; 58(3):452. https://doi.org/10.3390/medicina58030452
Chicago/Turabian StyleUrata, Ryunosuke, Tatsuya Igawa, Akifumi Suzuki, Yutaka Sasao, Norihiro Isogai, Haruki Funao, and Ken Ishii. 2022. "The Short and Intensive Rehabilitation (SHAiR) Program Improves Dropped Head Syndrome Caused by Amyotrophic Lateral Sclerosis: A Case Report" Medicina 58, no. 3: 452. https://doi.org/10.3390/medicina58030452






