Sorafenib in Metastatic Papillary Thyroid Carcinoma with BRAF K601E Mutation on Liquid Biopsy: A Case Report and Literature Review
Abstract
:1. Introduction
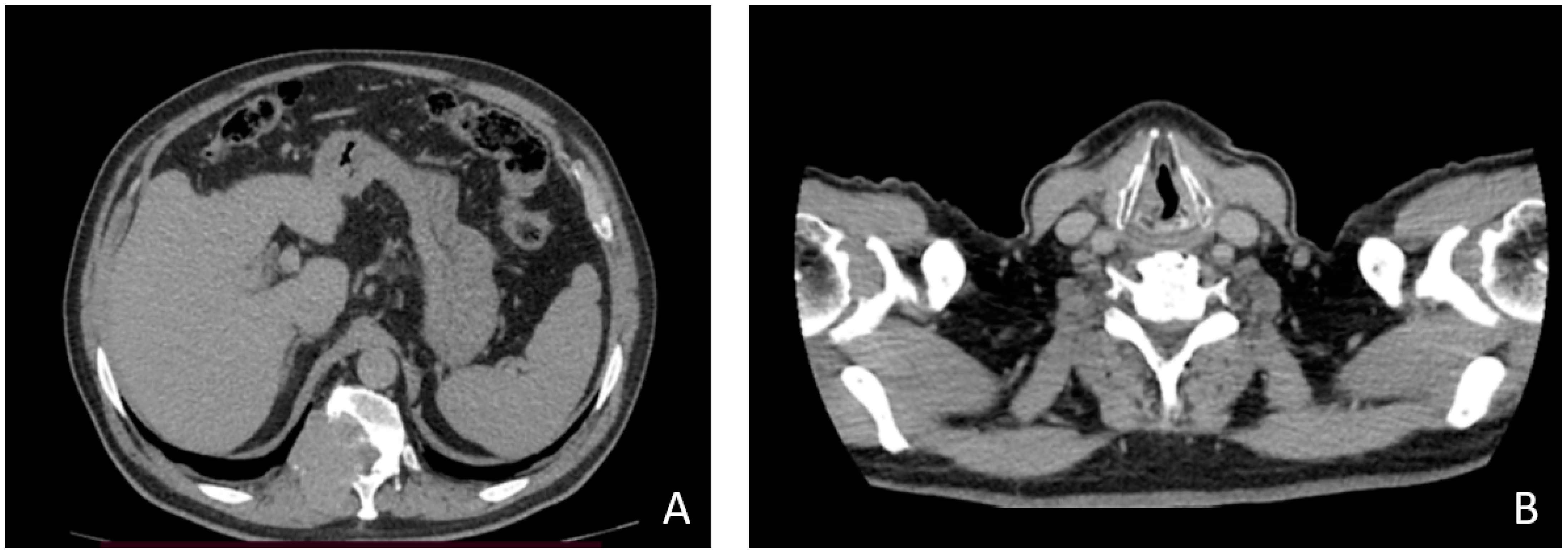
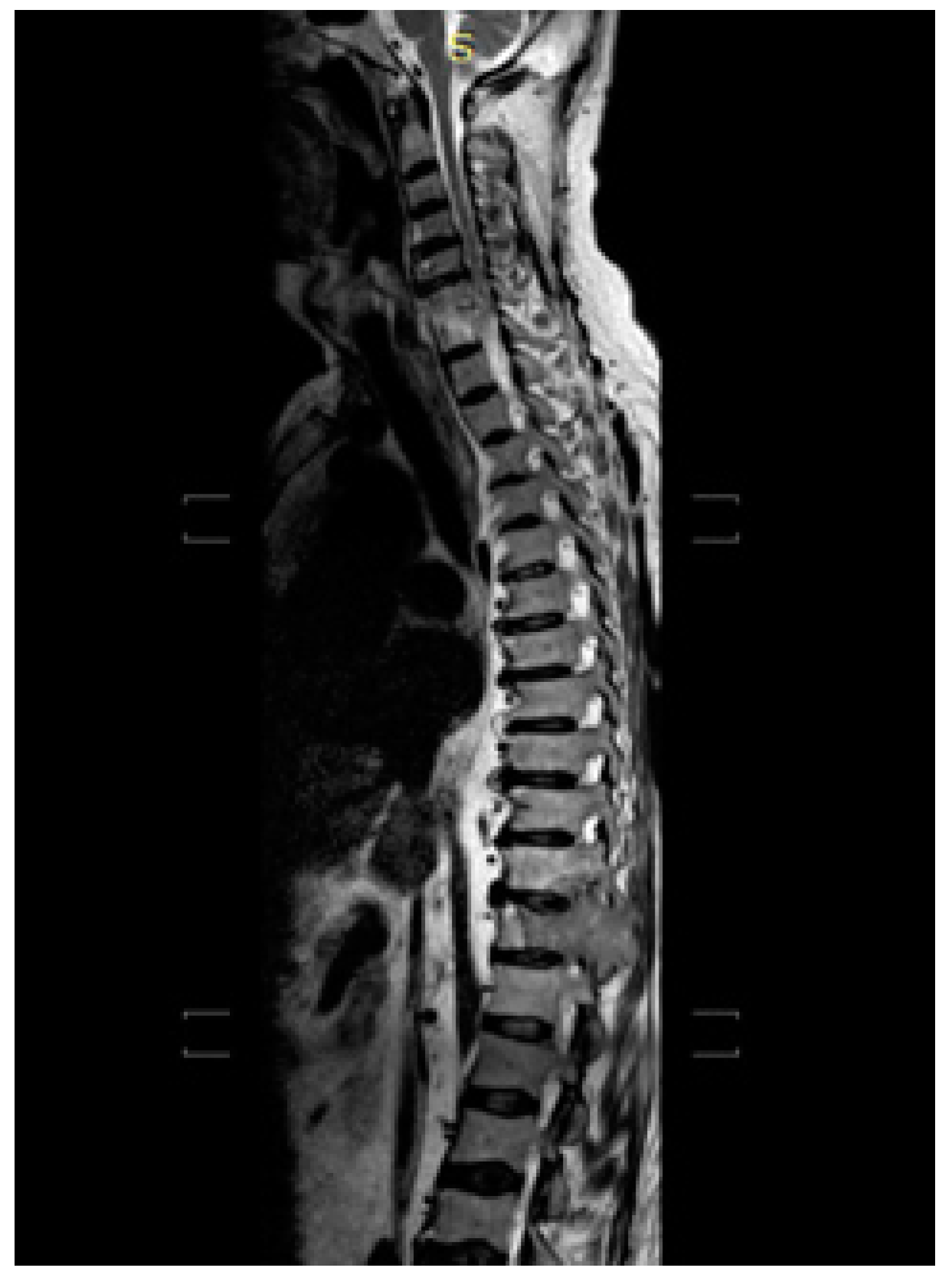
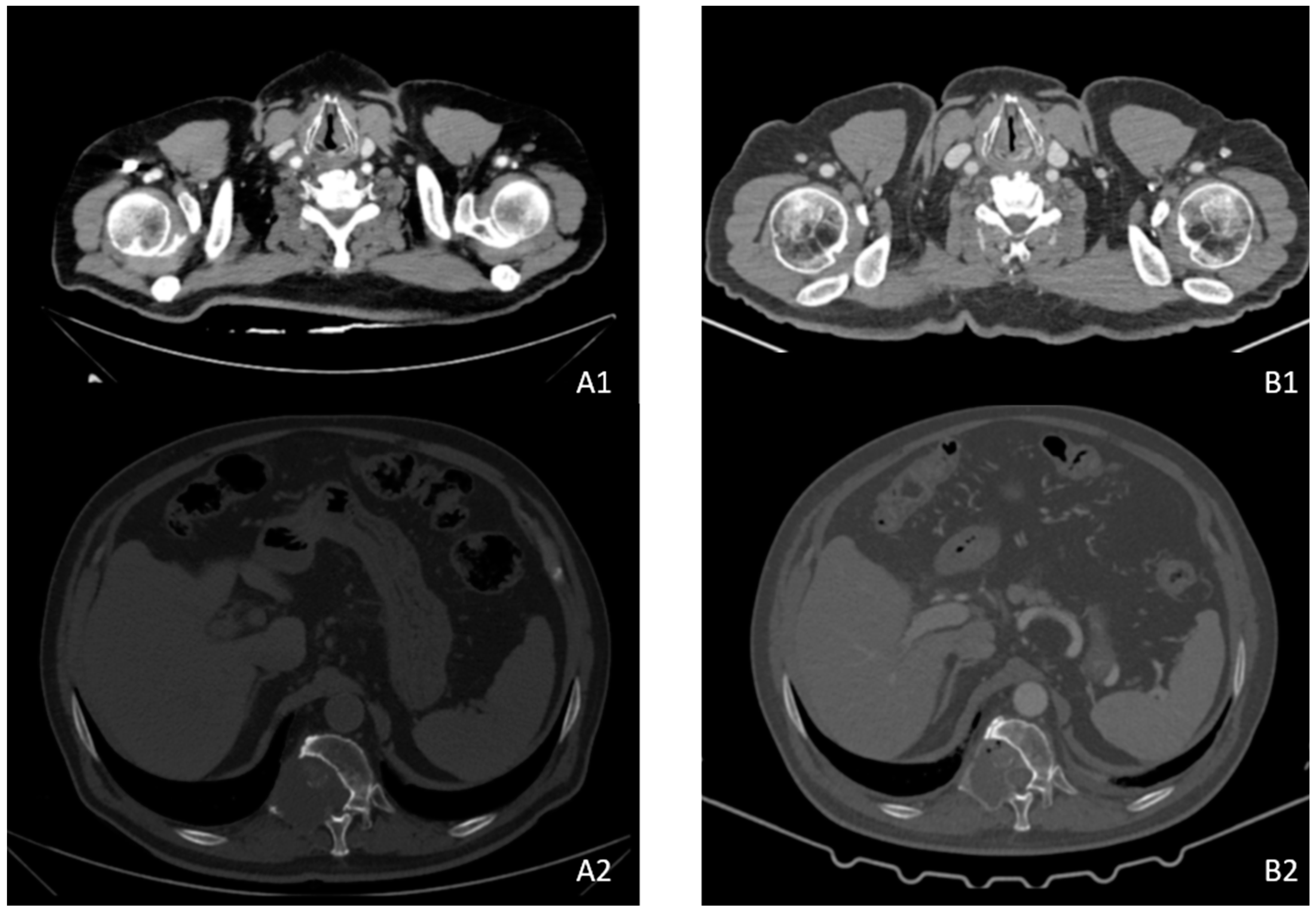
2. Case Description
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Escudier, B.; Worden, F.; Kudo, M. Sorafenib: Key Lessons from over 10 Years of Experience. Expert Rev. Anticancer Ther. 2019, 19, 177–189. [Google Scholar] [CrossRef]
- Lechner, M.G.; Hershman, J.M. Thyroid Nodules and Cancer in the Elderly; MDText. com, Inc.: Hanover, MA, USA, 2022. [Google Scholar]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pitoia, F.; Jerkovich, F. Selective Use of Sorafenib in the Treatment of Thyroid Cancer. Drug Des. Dev. Ther. 2016, 10, 1119–1131. [Google Scholar] [CrossRef] [Green Version]
- Tuttle, R.M.; Fleisher, M.; Francis, G.L.; Robbins, R.J. Serum Vascular Endothelial Growth Factor Levels Are Elevated in Metastatic Differentiated Thyroid Cancer but Not Increased by Short-Term TSH Stimulation. J. Clin. Endocrinol. Metab. 2002, 87, 1737–1742. [Google Scholar] [CrossRef]
- Ciampi, R.; Nikiforov, Y.E. RET/PTC Rearrangements and BRAF Mutations in Thyroid Tumorigenesis. Endocrinology 2007, 148, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Savvides, P.; Nagaiah, G.; Lavertu, P.; Fu, P.; Wright, J.J.; Chapman, R.; Wasman, J.; Dowlati, A.; Remick, S.C. Phase II Trial of Sorafenib in Patients with Advanced Anaplastic Carcinoma of the Thyroid. Thyroid 2013, 23, 600–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Food and Drug Administration. Sorafenib (Nexavar), Prescribing Information; Bayer HealthCare Pharmaceuticals Inc.: Hanover, NJ, USA, 2015.
- European Medicines Agency. Sorafenib (Nexavar). Summary of Pro-Duct Characteristics; Bayer AG: Leverkusen, Germany, 2016.
- Wilhelm, S.M.; Carter, C.; Tang, L.Y.; Wilkie, D.; McNabola, A.; Rong, H.; Chen, C.; Zhang, X.; Vincent, P.; McHugh, M.; et al. BAY 43-9006 Exhibits Broad Spectrum Oral Antitumor Activity and Targets the RAF/MEK/ERK Pathway and Receptor Tyrosine Kinases Involved in Tumor Progression and Angiogenesis. Cancer Res. 2004, 64, 7099–7109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salvatore, G.; de Falco, V.; Salerno, P.; Nappi, T.C.; Pepe, S.; Troncone, G.; Carlomagno, F.; Melillo, R.M.; Wilhelm, S.M.; Santoro, M. BRAF Is a Therapeutic Target in Aggressive Thyroid Carcinoma. Clin. Cancer Res. 2006, 12, 1623–1629. [Google Scholar] [CrossRef] [Green Version]
- Carlomagno, F.; Anaganti, S.; Guida, T.; Salvatore, G.; Troncone, G.; Wilhelm, S.M.; Santoro, M. BAY 43-9006 Inhibition of Oncogenic RET Mutants. J. Natl. Cancer Inst. 2006, 98, 326–334. [Google Scholar] [CrossRef] [Green Version]
- Yu, C.; Bruzek, L.M.; Xue, W.M.; Gores, G.J.; Carter, C.A.; Kaufmann, S.H.; Adjei, A.A. The Role of Mcl-1 Downregulation in the Proapoptotic Activity of the Multikinase Inhibitor BAY 43-9006. Oncogene 2005, 24, 6861–6869. [Google Scholar] [CrossRef] [Green Version]
- Palmirotta, R.; Lovero, D.; Cafforio, P.; Felici, C.; Mannavola, F.; Pellè, E.; Quaresmini, D.; Tucci, M.; Silvestris, F. Liquid Biopsy of Cancer: A Multimodal Diagnostic Tool in Clinical Oncology. Ther. Adv. Med. Oncol. 2018, 10, 1758835918794630. [Google Scholar] [CrossRef] [PubMed]
- Parameswaran, R.; Shulin Hu, J.; Min En, N.; Tan, W.B.; Yuan, N.K. Patterns of Metastasis in Follicular Thyroid Carcinoma and the Difference between Early and Delayed Presentation. Ann. R Coll. Surg. Engl. 2017, 99, 151–154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brose, M.S.; Nutting, C.M.; Jarzab, B.; Elisei, R.; Siena, S.; Bastholt, L.; de La Fouchardiere, C.; Pacini, F.; Paschke, R.; Shong, Y.K.; et al. Sorafenib in Radioactive Iodine-Refractory, Locally Advanced or Metastatic Differentiated Thyroid Cancer: A Randomised, Double-Blind, Phase 3 Trial. Lancet 2014, 384, 319–328. [Google Scholar] [CrossRef] [Green Version]
- Tirrò, E.; Martorana, F.; Romano, C.; Vitale, S.R.; Motta, G.; di Gregorio, S.; Massimino, M.; Pennisi, M.S.; Stella, S.; Puma, A.; et al. Molecular Alterations in Thyroid Cancer: From Bench to Clinical Practice. Genes 2019, 10, 709. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khatami, F.; Tavangar, S.M. A Review of Driver Genetic Alterations in Thyroid Cancers. Iran. J. Pathol. 2018, 13, 125. [Google Scholar] [CrossRef]
- Khatami, F.; Tavangar, S.M. Liquid Biopsy in Thyroid Cancer: New Insight. Int. J. Hematol.-Oncol. Stem Cell Res. 2018, 12, 235. [Google Scholar]
- Lin, L.H.; Allison, D.H.R.; Feng, Y.; Jour, G.; Park, K.; Zhou, F.; Moreira, A.L.; Shen, G.; Feng, X.; Sabari, J.; et al. Comparison of Solid Tissue Sequencing and Liquid Biopsy Accuracy in Identification of Clinically Relevant Gene Mutations and Rearrangements in Lung Adenocarcinomas. Mod. Pathol. 2021, 34, 2168–2174. [Google Scholar] [CrossRef]
- Liebs, S.; Eder, T.; Klauschen, F.; Schütte, M.; Yaspo, M.L.; Keilholz, U.; Tinhofer, I.; Kidess-Sigal, E.; Braunholz, D. Applicability of Liquid Biopsies to Represent the Mutational Profile of Tumor Tissue from Different Cancer Entities. Oncogene 2021, 40, 5204–5212. [Google Scholar] [CrossRef]
- Pupilli, C.; Pinzani, P.; Salvianti, F.; Fibbi, B.; Rossi, M.; Petrone, L.; Perigli, G.; De Feo, M.L.; Vezzosi, V.; Pazzagli, M.; et al. Circulating BRAFV600E in the Diagnosis and Follow-up of Differentiated Papillary Thyroid Carcinoma. J. Clin. Endocrinol. Metab. 2013, 98, 3359–3365. [Google Scholar] [CrossRef] [Green Version]
- Qiu, Z.L.; Wei, W.J.; Sun, Z.K.; Shen, C.T.; Song, H.J.; Zhang, X.Y.; Zhang, G.Q.; Chen, X.Y.; Luo, Q.Y. Circulating Tumor Cells Correlate with Clinicopathological Features and Outcomes in Differentiated Thyroid Cancer. Cell. Physiol. Biochem. 2018, 48, 718–730. [Google Scholar] [CrossRef]
- Kebebew, E.; Weng, J.; Bauer, J.; Ranvier, G.; Clark, O.H.; Duh, Q.Y.; Shibru, D.; Bastian, B.; Griffin, A. The Prevalence and Prognostic Value of BRAF Mutation in Thyroid Cancer. Ann. Surg. 2007, 246, 466–470. [Google Scholar] [CrossRef] [PubMed]
- Ganly, I.; Makarov, V.; Deraje, S.; Dong, Y.Y.; Reznik, E.; Seshan, V.; Nanjangud, G.; Eng, S.; Bose, P.; Kuo, F.; et al. Integrated Genomic Analysis of Hürthle Cell Cancer Reveals Oncogenic Drivers, Recurrent Mitochondrial Mutations, and Unique Chromosomal Landscapes. Cancer Cell 2018, 34, 256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agrawal, N.; Akbani, R.; Aksoy, B.A.; Ally, A.; Arachchi, H.; Asa, S.L.; Auman, J.T.; Balasundaram, M.; Balu, S.; Baylin, S.B.; et al. Integrated Genomic Characterization of Papillary Thyroid Carcinoma. Cell 2014, 159, 676–690. [Google Scholar] [CrossRef] [Green Version]
- Schulten, H.-J.; Salama, S.; Al-Ahmadi, A.; Al-Mansouri, Z.; Mirza, Z.; Al-Ghamdi, K.; Al-Hamour, O.A.; Huwait, E.; Gari, M.; Al-Qahtani, M.H.; et al. Comprehensive Survey of HRAS, KRAS, and NRAS Mutations in Proliferative Thyroid Lesions from an Ethnically Diverse Population—PubMed. Anticancer Res. 2013, 11, 4779–4784. [Google Scholar]
- Xing, M. BRAF Mutation in Thyroid Cancer. Endocr. Relat. Cancer 2005, 12, 245–262. [Google Scholar] [CrossRef] [Green Version]
- Schulten, H.J.; Salama, S.; Al-Mansouri, Z.; Alotibi, R.; Al-Ghamdi, K.; Al-Hamour, O.A.; Sayadi, H.; Al-Aradati, H.; Al-Johari, A.; Huwait, E.; et al. BRAF Mutations in Thyroid Tumors from an Ethnically Diverse Group. Hered. Cancer Clin. Pract. 2012, 10, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pennelli, G.; Vianello, F.; Barollo, S.; Pezzani, R.; Boschin, I.M.; Pelizzo, M.R.; Mantero, F.; Rugge, M.; Mian, C. BRAF(K601E) Mutation in a Patient with a Follicular Thyroid Carcinoma. Thyroid 2011, 21, 1393–1396. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caterino, M.; Pirozzi, M.; Facchini, S.; Zotta, A.; Sica, A.; Lo Giudice, G.; Rauso, R.; Varriale, E.; Ciardiello, F.; Fasano, M. Sorafenib in Metastatic Papillary Thyroid Carcinoma with BRAF K601E Mutation on Liquid Biopsy: A Case Report and Literature Review. Medicina 2022, 58, 666. https://doi.org/10.3390/medicina58050666
Caterino M, Pirozzi M, Facchini S, Zotta A, Sica A, Lo Giudice G, Rauso R, Varriale E, Ciardiello F, Fasano M. Sorafenib in Metastatic Papillary Thyroid Carcinoma with BRAF K601E Mutation on Liquid Biopsy: A Case Report and Literature Review. Medicina. 2022; 58(5):666. https://doi.org/10.3390/medicina58050666
Chicago/Turabian StyleCaterino, Marianna, Mario Pirozzi, Sergio Facchini, Alessia Zotta, Antonello Sica, Giorgio Lo Giudice, Raffaele Rauso, Elisa Varriale, Fortunato Ciardiello, and Morena Fasano. 2022. "Sorafenib in Metastatic Papillary Thyroid Carcinoma with BRAF K601E Mutation on Liquid Biopsy: A Case Report and Literature Review" Medicina 58, no. 5: 666. https://doi.org/10.3390/medicina58050666
APA StyleCaterino, M., Pirozzi, M., Facchini, S., Zotta, A., Sica, A., Lo Giudice, G., Rauso, R., Varriale, E., Ciardiello, F., & Fasano, M. (2022). Sorafenib in Metastatic Papillary Thyroid Carcinoma with BRAF K601E Mutation on Liquid Biopsy: A Case Report and Literature Review. Medicina, 58(5), 666. https://doi.org/10.3390/medicina58050666








