The Effectiveness of Allergen Immunotherapy in Adult Patients with Atopic Dermatitis Allergic to House Dust Mites
Abstract
:1. Introduction
2. Material and Methods
2.1. Study Design
2.2. Patients
- Other active skin diseases;
- Systemic immunosuppressant treatment (oral corticosteroids, cyclosporine, methotrexate) during the last 12 months;
- Other chronic diseases;
- Lack of written consent provided.
2.3. Scales
2.4. Treatment
2.5. Randomisation Procedure
2.6. Diagnostic Procedures
2.7. Skin Prick Tests
2.8. Clinical Assessment
2.9. Laboratory Tests Included
2.10. Outcomes
3. Results
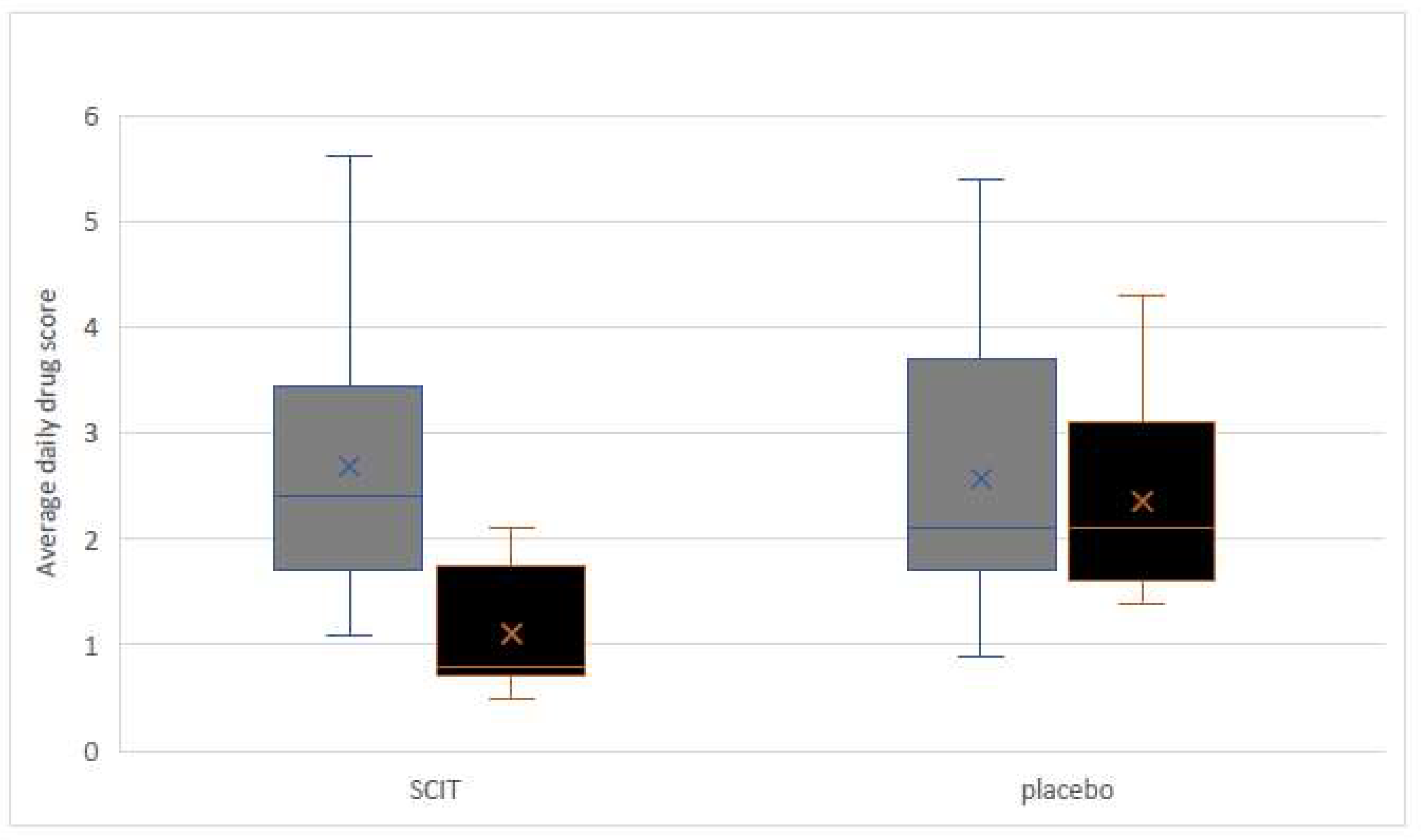
3.1. Clinical Improvement and Medication Score
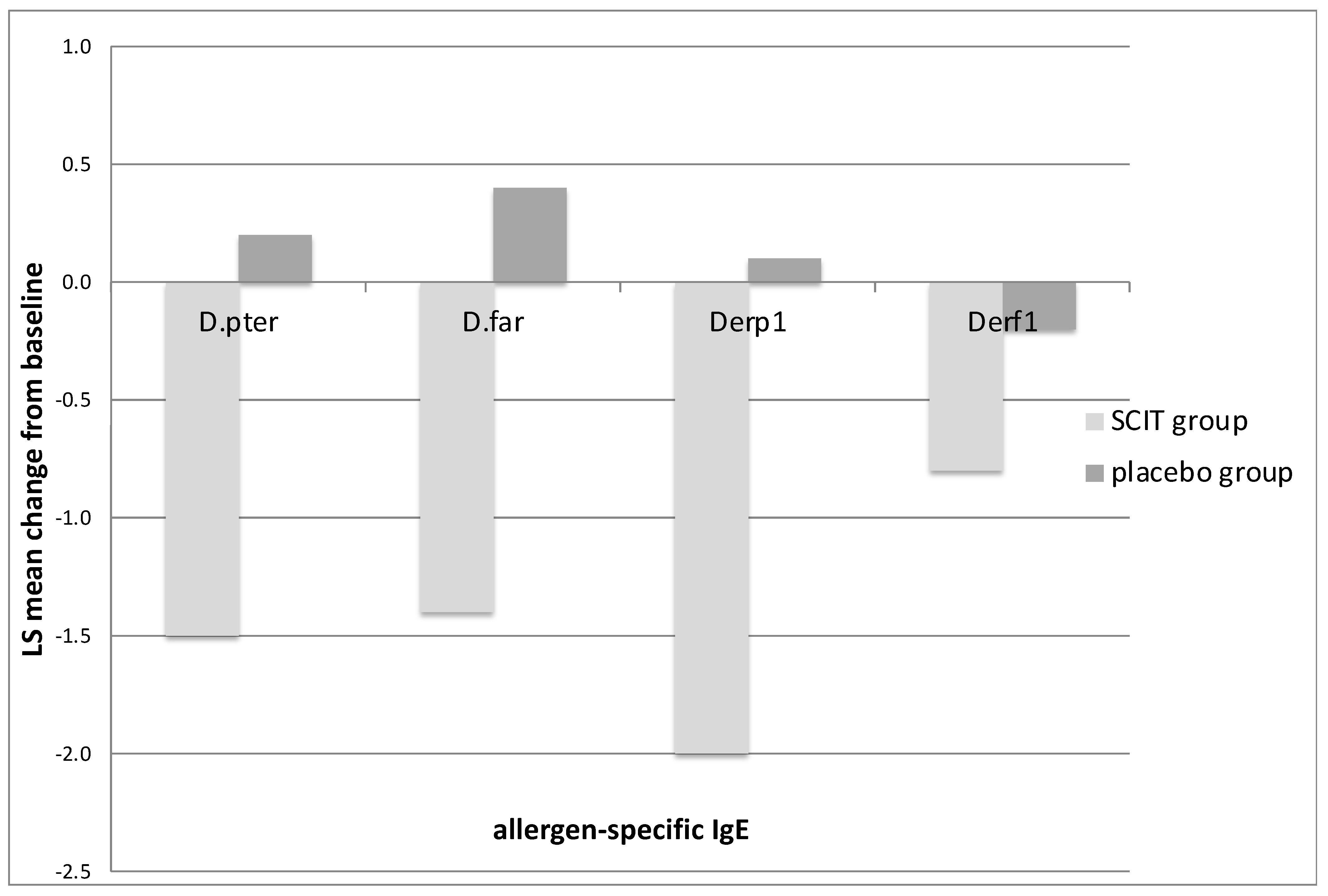
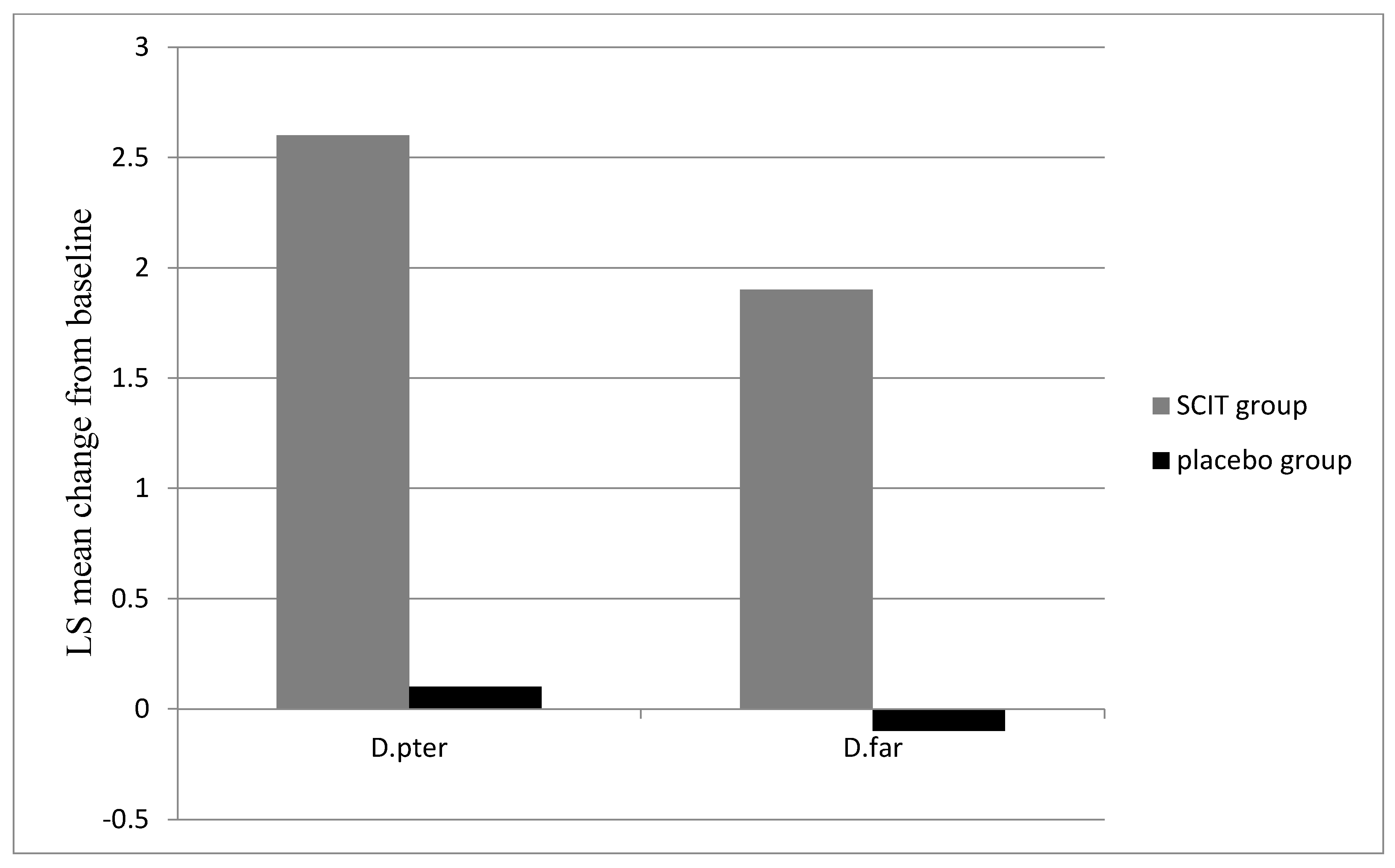
3.2. Immunological Markers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Genuneit, J.; Seibold, A.M.; Apfelbacher, C.J.; Konstantinou, G.N.; Koplin, J.J.; La Grutta, S.; Logan, K.; Perkin, M.R.; Flohr, C. Task Force ‘Overview of Systematic Reviews in Allergy Epidemiology (OSRAE)’ of the EAACI Interest Group on Epidemiology. Overview of systematic reviews in allergy epidemiology. Allergy 2017, 72, 849–856. [Google Scholar] [CrossRef] [Green Version]
- Bumbacea, R. Mite allergy and atopic dermatitis: Is there a clear link? (Review). Exp. Ther. Med. 2020, 9120, 3554–3560. [Google Scholar] [CrossRef]
- Wollenberg, A.; Barabort, S.; Bieber, T.; Christen-Zaech, S.; Deleuran, M.; Fink-Wagner, A.; Gieler, U.; Girolomoni, G.; Lau, S.; Muraro, M.; et al. Consensus-based European guidelines for treatment of atopic eczema (atopic dermatitis) in adults and children: Part I. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 657–682. [Google Scholar] [CrossRef] [Green Version]
- Avena-Woods, C. Overview of atopic dermatitis. Am. J. Manag. Care 2017, 23 (Suppl. 8), S115–S123. [Google Scholar]
- Hammond, M.; Gamal, A.; Mukherjee, P.K.; Damiani, G.; McCormick, T.S.; Ghannoum, M.A.; Nedorost, S. Cutaneous dysbiosis may amplify barrier dysfunction in patients with atopic dermatitis. Front. Microbiol. 2022, 14, 944365. [Google Scholar] [CrossRef]
- Ren-Bin, T. House dust mite-specific immunotherapy alters the natural course of atopic March. J. Chin. Med. Assoc. 2020, 83, 109–112. [Google Scholar]
- Werfel, T.; Breuer, K.; Rueff, F.; Przybilla, B.; Warm, M.; Grewe, M.; Ruzicka, T.; Brehler, R.; Wolf, H.; Schnitker, J.; et al. Usefulness of specific immunotherapy in patients with atopic dermatitis and allergic sensitization to house dust mites: A multi-centre, randomized, dose-response study. Allergy Eur. J. Allergy Clin. Immunol. 2006, 61, 202–205. [Google Scholar] [CrossRef]
- Hajdu, K.; Kapitany, H.; Dajnoki, Z.; Soltesz, L.; Barath, S.; Hendrik, Z.; Veres, I.; Szegedi, A.; Gaspar, K. Improvement of clinical and immunological parameters after allergen-specific immunotherapy in atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2021, 35, 1357–1361. [Google Scholar] [CrossRef]
- Muraro, A.; Roberts, G.; Halken, S.; Agache, I.; Angier, E.; Fernandez-Rivas, M.; van Wijk, R.G.; Jutel, M.; Lau, S.; Pajno, G.; et al. EAACI guidelines on allergen immunotherapy: Executive statement. Allergy 2018, 73, 739–743. [Google Scholar] [CrossRef] [Green Version]
- Tam, H.H.; Calderon, M.A.; Manikam, L.; Nankervis, H.; Núñez, I.G.; Williams, H.C.; Durham, S.R.; Boyle, R.J. Specific allergen immunotherapy for the treatment of atopic eczema: A Cochrane systematic review. Allergy 2016, 71, 1345–1356. [Google Scholar] [CrossRef]
- Weghofer, M.; Thomas, W.R.; Kronqvist, M.; Mari, A.; Purohit, A.; Pauli, G.; Horak, F.; Gronlund, H.; van Hage, M.; Valenta, R.; et al. Variability of IgE reactivity profiles among European mite allergic patients. Eur. J. Clin. Investig. 2008, 38, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.W.; Zieglmayer, P.; Zieglmayer, R.; Ing, D.; Lemell, P.; Horak, F.; Bunu, C.P.; Valenta, R.; Vrtala, S. Selection of house dust mite-allergic patients by molecular diagnosis may enhance success of specific immunotherapy. J. Allergy Clin. Immunol. 2019, 143, 1248–1252. [Google Scholar] [CrossRef] [PubMed]
- Nahm, D.H.; Kim, M.E.; Kwon, B.; Cho, S.M.; Ahn, A. Clinical efficacy of subcutaneous allergen immunotherapy in patients with atopic dermatitis. Yonsei Med. J. 2006, 57, 1420–1426. [Google Scholar] [CrossRef]
- Zhou, J.; Chen, S.; Song, Z. Analysis of the long-term efficacy and safety of subcutaneous immunotherapy for atopic dermatitis. Allergy Asthma Proc. 2021, 42, 47–54. [Google Scholar] [CrossRef]
- Langer, S.S.; Cardili, R.N.; Lima Melo, J.M.; Ferriani, M.P.L.; Moreno, A.S.; Dias, M.M.; Bueno-Filho, R.; Pocente, R.H.C.; Roxo-Junior, P.; Silva, J.; et al. Efficacy of house dust mite sublingual immunotherapy in patients with atopic dermatitis: A randomized, double-blind, placebo-controlled trial. J. Allergy Clin. Immunol. 2022, 10, 539–550. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.E.; Kim, J.E.; Sung, J.M.; Lee, J.W.; Choi, G.S.; Nahm, D.H. Safety of accelerated schedules of saubcutaneous allergen immunotherapy with house dust mite wextract in patients with atopic dermatitis. J. Korean Med. Sci. 2011, 26, 1159–1164. [Google Scholar] [CrossRef] [Green Version]
- Nelson, H.S. Immunotherapy for house dust mite allergy. Allergy Asthma Proc. 2018, 39, 264–272. [Google Scholar] [CrossRef]
- Panjo, G.D.; Caminiti, L.; Vita, D.; Barberio, G.; Salzano, G.; Lombardo, F.; Canonica, G.W.; Passalacqua, G. Sublingual immunotherapy in mite-sensitized children with atopic dermatitis: A randomized, double-blind, placebo-controlled study. J. Allergy Clin. Immunol. 2007, 120, 164–170. [Google Scholar]
- Lee, J.; Park, C.O.; Lee, K.H. Specific immunotherapy in atopic dermatitis. Allergy Asthma Immunol. Res. 2015, 7, 221–229. [Google Scholar] [CrossRef] [Green Version]
- Koskeridis, F.; Evangelou, E.; Ntzani, E.E.; Kostikas, K.; Tsabouri, S. Treatment with Dupilumab in Patients with Atopic Dermatitis: Systematic Review and Meta-Analysis. J. Cutan Med. Surg. 2022, 26, 613–621. [Google Scholar] [CrossRef]
- Halling, A.S.; Loft, N.; Silverberg, J.I.; Guttman-Yassky, E.; Thyssen, J.P. Real-world evidence of dupilumab efficacy and risk of adverse events: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2021, 84, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Bridgewood, C.; Wittmann, M.; Macleod, T.; Watad, A.; Newton, D.; Bhan, K.; Amital, H.; Damiani, G.; Giryes, S.; Bragazzi, N.L.; et al. T Helper 2 IL-4/IL-13 Dual Blockade with Dupilumab Is Linked to Some Emergent T Helper 17–Type Diseases, Including Seronegative Arthritis and Enthesitis/Enthesopathy, but Not to Humoral Autoimmune Diseases. J. Investig. Dermatol. 2022, 142, 2660–2667. [Google Scholar] [CrossRef] [PubMed]

| SCIT Group n = 21 | Placebo Group n = 16 | p | |
|---|---|---|---|
| Women | 10 | 8 | NS |
| Mean age (years) SD | 18.5 ± 11.3 | 20.1 ± 8.3 | NS |
| Mean EASI SD | 43 ± 8.2 | 39 ± 10.3 | NS |
| Range | 20–58 | 12–62 | |
| Mean % BSA SD | 72 ± 18 | 79 ± 21 | NS |
| Range | 23–84 | 19–88 | |
| Mean IsGA SD | 4.5 ± 0.5 | 4 ± 0.75 | NS |
| Range | 3–5 | 3–5 | |
| Duration of AD (yrs) | 18.6 ± 5.7 | 21.9 ± 9.4 | 0.05 |
| Number of subjects with asthma | 4 | 3 | NS |
| Number of patients with allergic rhinitis | 8 | 7 | NS |
| Number of smokers | 4 | 2 | NS |
| Total IgE (kU/L) SD | 7287.43 ± 1664 | 6450 ± 2018 | 0.04 |
| Total IgE (kU/L) SD in patients with AD and IA | 6933.12 ± 2070 | 6201.1 ± 2652 | NS |
| Total IgE (kU/L) SD in patients with only AD | 7356.4 ± 1980 | 6953.5 ± 2542 | NS |
| sIgE to D. pter (kU/L) SD | 25.1 ± 6.34 | 21.5 ± 9.11 | NS |
| sIgE to D. pter (kU/L) SD in patients with AD and IA | 23.4 ± 9.11 | 19.4 ± 7.21 | NS |
| sIgE to D. pter (kU/L) SD in patients with only AD | 27.4 ± 6.2 | 23.8 ± 5.19 | NS |
| sIgE to D. far (kU/L) SD | 13.04 ± 4.71 | 11.9 ± 5.2 | NS |
| sIgE to D. far (kU/L) SD in patients with AD and IA | 16.4 ± 5.3 | 9.31 ± 4.2 | 0.03 |
| sIgE to D. far (kU/L) SD in patients with only AD | 11.2 ± 4.5 | 15.4 ± 8.3 ** | NS |
| Treatment before study * | |||
| Antihistamines | 21 (100%) | 16 (100%) | NS |
| Topical glucocorticosteroids | 17 (80%) | 14 (88%) | NS |
| Calcineurin inhibitors topically | 11 (52%) | 9 (56%) | |
| NS | |||
| Systemic glucocorticosteroid | 4 (19%) | 3 (18%) | |
| NS | |||
| Cyclosporine | 13 (62%) | 9 (56%) | NS |
| Methotrexate | 1 (5%) | 0 | NS |
| Dupilimab | 0 | 0 | NS |
| PUVA | 7 (33%) | 3 (19%) | 0.03 |
| SCIT Group n = 20 | Placebo Group n = 14 | |||||
|---|---|---|---|---|---|---|
| Baseline | After 12 m | p | Baseline | After 12 m | p | |
| Mean EASI ± SD Range | 43 ± 8.2 20–58 | 21 ± 5.9 0–54 | <0.05 | 39 ± 10.3 12–62 | 32 ± 12.8 10–69 | NS |
| Mean EASI ± SD in patients with AD and IA Range | 45 ± 6.8 22–61 | 24 ± 4.1 0–49 | <0.05 | 38 ± 9.4 11–58 | 30 ± 9.12 11–71 | NS |
| Mean EASI ± SD in patients with only AD Range | 39 ± 7.2 18–55 | 18 ± 5 0–61 | <0.05 | 41.2 ± 3 14–65 | 35 ± 11.8 8–65 | NS |
| Mean % BSA ± SD Range | 72 ± 18 23–84 | 28 ± 11 0–78 | <0.05 | 79 ± 21 19–88 | 69 ± 21 19–88 | 0.05 |
| Mean % BSA ± SD in patients with AD and IA Range | 70 ± 9.4 20–81 | 26 ± 9.5 0–67 | <0.05 | 77 ± 6.8 15–91 | 67 ± 14.8 14–90 | NS |
| Mean % BSA ± SD in patients with only AD Range | 75 ± 11.2 25–83 | 30 ± 11.5 0–78 | <0.05 | 80 ± 12.3 10–87 | 70.1 ± 4.1 17–91 | 0.05 |
| Mean IsGA ± SD Range | 4.5 ± 0.5 3–5 | 1.5 ± 0.5 0–5 | <0.05 | 4 ± 0.75 3–5 | 3.5 ± 0.5 1–5 | NS |
| Mean IsGA ± SD in patients with AD and IA Range | 4.5 ± 0.25 3–5 | 1.75 ± 0.5 0–5 | <0.05 | 4 ± 0.25 3–5 | 3.5 ± 0.75 1–5 | NS |
| Mean IsGA ± SD in patients with only AD Range | 4.5 ± 0.75 3–5 | 1.5 ± 2.5 0–5 | <0.05 | 3.75 ± 0.75 3–4.75 | 3.5 ± 0.25 1–5 | NS |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bogacz-Piaseczyńska, A.; Bożek, A. The Effectiveness of Allergen Immunotherapy in Adult Patients with Atopic Dermatitis Allergic to House Dust Mites. Medicina 2023, 59, 15. https://doi.org/10.3390/medicina59010015
Bogacz-Piaseczyńska A, Bożek A. The Effectiveness of Allergen Immunotherapy in Adult Patients with Atopic Dermatitis Allergic to House Dust Mites. Medicina. 2023; 59(1):15. https://doi.org/10.3390/medicina59010015
Chicago/Turabian StyleBogacz-Piaseczyńska, Agnieszka, and Andrzej Bożek. 2023. "The Effectiveness of Allergen Immunotherapy in Adult Patients with Atopic Dermatitis Allergic to House Dust Mites" Medicina 59, no. 1: 15. https://doi.org/10.3390/medicina59010015







