Exploring Medicinal Herbs’ Therapeutic Potential and Molecular Docking Analysis for Compounds as Potential Inhibitors of Human Acetylcholinesterase in Alzheimer’s Disease Treatment
Abstract
:1. Introduction
2. Medicinal Herbs against Alzheimer’s
2.1. Rosa damascena Herrm.
2.2. Picrasma quassioides (D.Don) Benn.
2.3. Actinidia arguta (Siebold & Zucc.) Planch. ex Miq.
2.4. Alpinia galanga (L.)
2.5. Piper nigrum L.
2.6. Rheum ribes L.
2.7. Markhamia tomentosa (Benth.) K. Schum. ex Engl.
2.8. Cassia obtusifolia L.
3. Phytochemicals against Alzheimer’s Disease
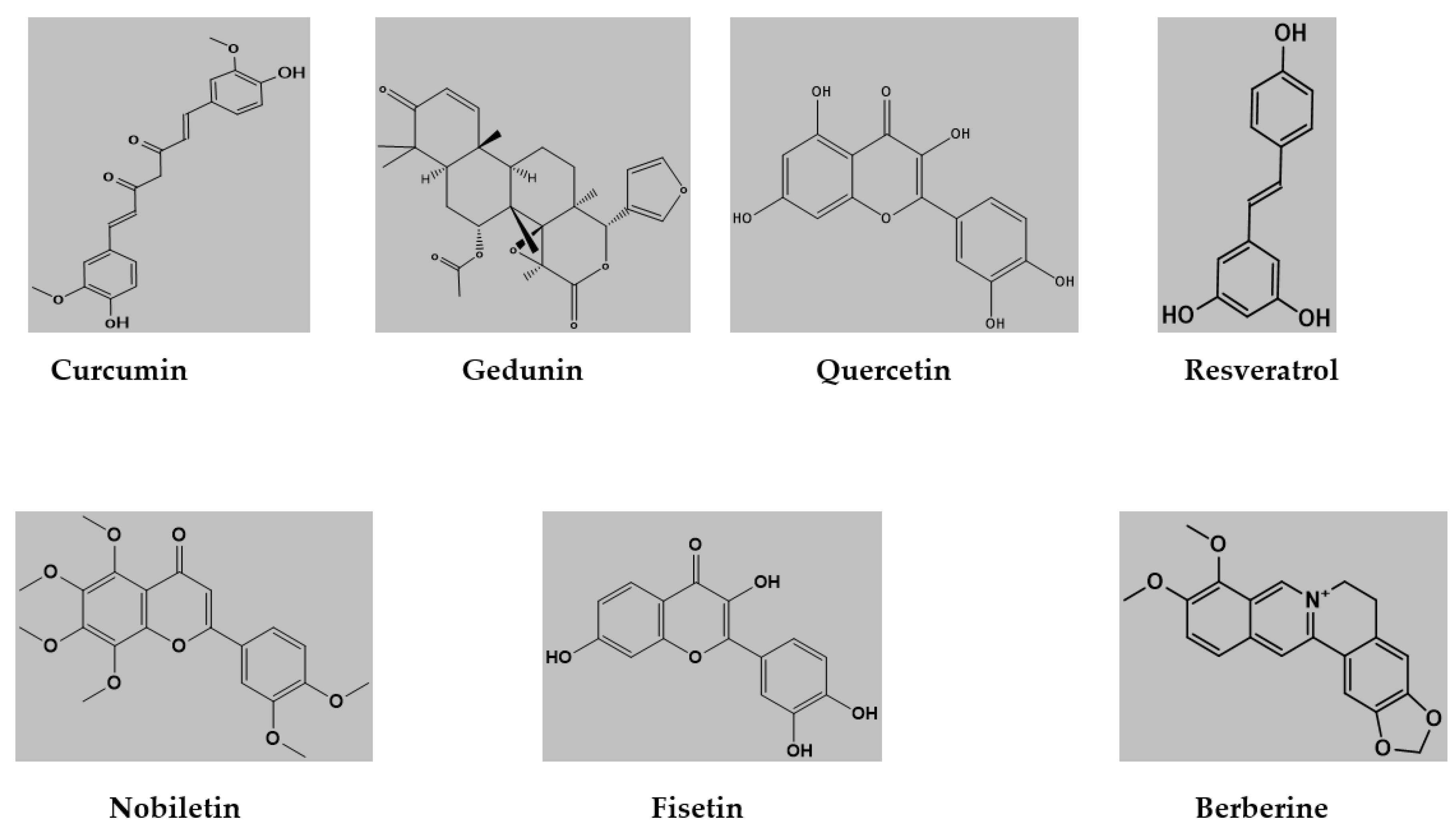
3.1. Curcumin
3.2. Gedunin
3.3. Quercetin
3.4. Resveratrol
3.5. Nobiletin
3.6. Fisetin
3.7. Berberine
4. Materials and Methods
4.1. Protein Preparation
4.2. Ligand Preparation
4.3. Molecular Docking
4.4. ADME Studies
5. Results and Discussion
5.1. Molecular Docking Results
5.2. Results of ADME Analysis
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rodini, M.; De Simone, M.S.; Caltagirone, C.; Carlesimo, G.A. Accelerated Long-Term Forgetting in Neurodegenerative Disorders: A Systematic Review of the Literature. Neurosci. Biobehav. Rev. 2022, 104815. [Google Scholar] [CrossRef] [PubMed]
- Vassar, R.; Bennett, B.D.; Babu-khan, S.; Kahn, S.; Elizabeth, A.; Denis, P.; Teplow, D.B.; Ross, S.; Amarante, P.; Loeloff, R.; et al. β-Secretase Cleavage of Alzheimer ’ s Amyloid Precursor Protein by the Transmembrane Aspartic Protease BACE. Science 1999, 286, 735–741. [Google Scholar] [CrossRef]
- Kocyigit, U.M.; Budak, Y.; Gürdere, M.B.; Ertürk, F.; Yencilek, B.; Taslimi, P.; Gülçin, İ.; Ceylan, M. Synthesis of Chalcone-Imide Derivatives and Investigation of Their Anticancer and Antimicrobial Activities, Carbonic Anhydrase and Acetylcholinesterase Enzymes Inhibition Profiles. Arch. Physiol. Biochem. 2018, 124, 61–68. [Google Scholar] [CrossRef]
- Mathew, M.; Subramanian, S. In Vitro Screening for Anti-Cholinesterase and Antioxidant Activity of Methanolic Extracts of Ayurvedic Medicinal Plants Used for Cognitive Disorders. PLoS ONE 2014, 9, e86804. [Google Scholar] [CrossRef] [PubMed]
- Ozmen Ozgun, D.; Gul, H.I.; Yamali, C.; Sakagami, H.; Gulcin, I.; Sukuroglu, M.; Supuran, C.T. Synthesis and Bioactivities of Pyrazoline Benzensulfonamides as Carbonic Anhydrase and Acetylcholinesterase Inhibitors with Low Cytotoxicity. Bioorg. Chem. 2019, 84, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Prasansuklab, A.; Tewin, T. Amyloidosis in Alzheimer’s Disease: The Toxicity of Amyloid Beta. Evid.-Based Complement. Altern. Med. 2013, 2013, 413808. [Google Scholar] [CrossRef]
- Gürbüz, P.; Martinez, A.; Pérez, C.; Martínez-González, L.; Göger, F.; Ayran, İ. Potential Anti-Alzheimer Effects of Selected Lamiaceae Plants through Polypharmacology on Glycogen Synthase Kinase-3β, β-Secretase, and Casein Kinase 1δ. Ind. Crops Prod. 2019, 138, 111431. [Google Scholar] [CrossRef]
- Griffiths, J.; Grant, S.G.N. Synapse Pathology in Alzheimer’s Disease. In Seminars in Cell & Developmental Biology; Elsevier: Amsterdam, The Netherlands, 2023; Volume 139, pp. 13–23. [Google Scholar]
- Chen, L.; Hu, L.; Zhao, J.; Hong, H.; Feng, F.; Qu, W.; Liu, W. Chotosan Improves Aβ1–42-Induced Cognitive Impairment and Neuroinflammatory and Apoptotic Responses through the Inhibition of TLR-4/NF-ΚB Signaling in Mice. J. Ethnopharmacol. 2016, 191, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Pozueta, J.; Lefort, R.; Shelanski, M.L. Synaptic Changes in Alzheimer’s Disease and Its Models. Neuroscience 2013, 251, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Schneider, L.S.; Mangialasche, F.; Andreasen, N.; Feldman, H.; Giacobini, E.; Jones, R.; Mantua, V.; Mecocci, P.; Pani, L.; Winblad, B.; et al. Clinical Trials and Late-Stage Drug Development for Alzheimer’s Disease: An Appraisal from 1984 to 2014. J. Intern. Med. 2014, 275, 251–283. [Google Scholar] [CrossRef]
- Xu, Y.; Jiang, H.; Zhu, B.; Cao, M.; Feng, T.; Sun, Z.; Du, G.; Zhao, Z. Advances and Applications of Fluids Biomarkers in Diagnosis and Therapeutic Targets of Alzheimer’s Disease. CNS Neurosci. Ther. 2023, 29, 2060–2073. [Google Scholar] [CrossRef] [PubMed]
- Scheltens, P. Brain Imaging and Alzheimer’s Disease. Eur. Neuropsychopharmacol. 1993, 3, 168–169. [Google Scholar] [CrossRef]
- Ghosh, S.; Ali, R.; Verma, S. Aβ-Oligomers: A Potential Therapeutic Target for Alzheimer’s Disease. Int. J. Biol. Macromol. 2023, 239, 124231. [Google Scholar] [CrossRef]
- Howes, M.-J.R.; Houghton, P.J. Ethnobotanical Treatment Strategies Against Alzheimers Disease. Curr. Alzheimer Res. 2012, 9, 67–85. [Google Scholar] [CrossRef]
- Zaiter, A.; Becker, L.; Petit, J.; Zimmer, D.; Karam, M.C.; Baudelaire, É.; Scher, J.; Dicko, A. Antioxidant and Antiacetylcholinesterase Activities of Different Granulometric Classes of Salix Alba (L.) Bark Powders. Powder Technol. 2016, 301, 649–656. [Google Scholar] [CrossRef]
- Loizzo, M.; Tundis, R.; Menichini, F.; Menichini, F. Natural Products and Their Derivatives as Cholinesterase Inhibitors in the Treatment of Neurodegenerative Disorders: An Update. Curr. Med. Chem. 2008, 15, 1209–1228. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.L.; Sato, S.; Dai, W.; Yamanaka, N. The Protective Effect of Hepatocyte Growth-Promoting Factor (PHGF) against Hydrogen Peroxide-Induced Acute Lung Injury in Rats. Med. Electron Microsc. 2001, 34, 92–102. [Google Scholar] [CrossRef]
- Candy, J.M.; Perry, R.H.; Perry, E.K.; Irving, D.; Blessed, G.; Fairbairn, A.F.; Tomlinson, B.E. Pathological Changes in the Nucleus of Meynert in Alzheimer’s and Parkinson’s Diseases. J. Neurol. Sci. 1983, 59, 277–289. [Google Scholar] [CrossRef]
- Taslimi, P.; Köksal, E.; Gören, A.C.; Bursal, E.; Aras, A.; Kılıç, Ö.; Alwasel, S.; Gülçin, İ. Anti-Alzheimer, Antidiabetic and Antioxidant Potential of Satureja Cuneifolia and Analysis of Its Phenolic Contents by LC-MS/MS. Arab. J. Chem. 2020, 13, 4528–4537. [Google Scholar] [CrossRef]
- Rezende, F.A.G.G.; Sande, D.; Coelho, A.C.; Oliveira, G.P.; Boaventura, M.A.D.; Takahashi, J.A. Edible Flowers as Innovative Ingredients for Future Food Development: Anti-Alzheimer, Antimicrobial and Antioxidant Potential. Chem. Eng. Trans. 2019, 75, 337–342. [Google Scholar] [CrossRef]
- Falzon, C.C.; Balabanova, A. Phytotherapy: An Introduction to Herbal Medicine. Prim. Care Clin. Off. Pract. 2017, 44, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Persa, P.B.; Karimi, E. Alzheimer ’ S : Phytotherapy and the Most Important Herbs in the Treatment of Alzheimer’s. Plant Biotechnol. Persa 2020, 2, 61–62. [Google Scholar]
- Akram, M.; Riaz, M.; Munir, N.; Akhter, N.; Zafar, S.; Jabeen, F.; Ali Shariati, M.; Akhtar, N.; Riaz, Z.; Altaf, S.H.; et al. Chemical Constituents, Experimental and Clinical Pharmacology of Rosa Damascena: A Literature Review. J. Pharm. Pharmacol. 2020, 72, 161–174. [Google Scholar] [CrossRef]
- El Alami, A.; Chait, A. Enquête Ethnopharmacologique et Ethnobotanique Sur Les Plantes Médicinales Dans Le Haut Atlas Central Du Maroc. Alger. J. Nat. Prod. 2017, 5, 427–445. [Google Scholar] [CrossRef]
- Shikov, V.; Kammerer, D.R.; Mihalev, K.; Mollov, P.; Carle, R. Heat Stability of Strawberry Anthocyanins in Model Solutions Containing Natural Copigments Extracted from Rose (Rosa Damascena Mill.) Petals. J. Agric. Food Chem. 2008, 56, 8521–8526. [Google Scholar] [CrossRef] [PubMed]
- Cerezo, A.B.; Cuevas, E.; Winterhalter, P.; Garcia-Parrilla, M.C.; Troncoso, A.M. Isolation, Identification, and Antioxidant Activity of Anthocyanin Compounds in Camarosa Strawberry. Food Chem. 2010, 123, 574–582. [Google Scholar] [CrossRef]
- Zhang, L.; Ma, J.; Yang, F.; Li, S.; Ma, W.; Chang, X.; Yang, L. Neuroprotective Effects of Quercetin on Ischemic Stroke: A Literature Review. Front. Pharmacol. 2022, 13, 854249. [Google Scholar] [CrossRef]
- Yu, L.; Chen, C.; Wang, L.F.; Kuang, X.; Liu, K.; Zhang, H.; Du, J.R. Neuroprotective Effect of Kaempferol Glycosides against Brain Injury and Neuroinflammation by Inhibiting the Activation of NF-ΚB and STAT3 in Transient Focal Stroke. PLoS ONE 2013, 8, e55839. [Google Scholar] [CrossRef]
- Karimipour, M.; Mardani, M.; Ghanadian, M.; Esmaeili, A.; Mohammadnejad, D.; Alaei, H.; Esfandiary, E. Neuroprotective Effects of Rosa Damascena Extract on Learning and Memory in a Rat Model of Amyloid-β-Induced Alzheimer‘s Disease. Adv. Biomed. Res. 2015, 4, 131. [Google Scholar] [CrossRef] [PubMed]
- Esfandiary, E.; Karimipour, M.; Mardani, M.; Alaei, H.; Ghannadian, M.; Kazemi, M.; Mohammadnejad, D.; Hosseini, N.; Esmaeili, A. Novel Effects of Rosa Damascena Extract on Memory and Neurogenesis in a Rat Model of Alzheimer’s Disease. J. Neurosci. Res. 2014, 92, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Hamazaki, Y.; Kato, M.; Karasawa, K. Methylnigakinone Content Determination and Geographical Origin Discrimination for P. Quassioides via Fluorescence Fingerprint and Principal Component Analyses. J. Pharm. Biomed. Anal. 2022, 219, 114932. [Google Scholar] [CrossRef] [PubMed]
- Jiao, W.H.; Gao, H.; Zhao, F.; Lin, H.W.; Pan, Y.M.; Zhou, G.X.; Yao, X.S. Anti-Inflammatory Alkaloids from the Stems of Picrasma Quassioides BENNET. Chem. Pharm. Bull. 2011, 59, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Takasu, K.; Shimogama, T.; Saiin, C.; Kim, H.S.; Wataya, Y.; Ihara, M. π-Delocalized β-Carbolinium Cations as Potential Antimalarials. Bioorg. Med. Chem. Lett. 2004, 14, 1689–1692. [Google Scholar] [CrossRef]
- Houël, E.; Bertani, S.; Bourdy, G.; Deharo, E.; Jullian, V.; Valentin, A.; Chevalley, S.; Stien, D. Quassinoid Constituents of Quassia Amara L. Leaf Herbal Tea. Impact on Its Antimalarial Activity and Cytotoxicity. J. Ethnopharmacol. 2009, 126, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Tang, Q.; Xu, J.; Wang, S.; Li, S.; Zou, X.; Cao, Z. Dehydrocrenatidine Inhibits Voltage-Gated Sodium Channels and Ameliorates Mechanic Allodia in a Rat Model of Neuropathic Pain. Toxins 2019, 11, 229. [Google Scholar] [CrossRef] [PubMed]
- Mohd Jamil, M.D.H.; Taher, M.; Susanti, D.; Rahman, M.A.; Zakaria, Z.A. Phytochemistry, Traditional Use and Pharmacological Activity of Picrasma quassioides: A Critical Reviews. Nutrients 2020, 12, 2584. [Google Scholar] [CrossRef] [PubMed]
- Guo, E.; Hu, Y.; Du, T.; Zhu, H.; Chen, L.; Qu, W.; Zhang, J.; Xie, N.; Liu, W.; Feng, F.; et al. Effects of Picrasma Quassioides and Its Active Constituents on Alzheimer’s Disease in Vitro and in Vivo. Bioorg. Chem. 2019, 92, 103258. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.G.; Seong, Y.H. Inhibitory Effect of the Leaves and Stems of Actinidia Arguta on Aβ (25-35)-Induced Neuronal Cell Death and Memory Impairment. J. Biomed. Transl. Res. 2018, 19, 26–31. [Google Scholar] [CrossRef]
- Choi, J.J.; Park, B.; Kim, D.H.; Pyo, M.Y.; Choi, S.; Son, M.; Jin, M. Blockade of Atopic Dermatitis-like Skin Lesions by DA-9102, a Natural Medicine Isolated from Actinidia Arguta, in the Mgdeficiency Induced Dermatitis Model of Hairless Rats. Exp. Biol. Med. 2008, 233, 1026–1034. [Google Scholar] [CrossRef]
- Zuo, L.L.; Wang, Z.Y.; Fan, Z.L.; Tian, S.Q.; Liu, J.R. Evaluation of Antioxidant and Antiproliferative Properties of Three Actinidia (Actinidia Kolomikta, Actinidia Arguta, Actinidia Chinensis) Extracts in Vitro. Int. J. Mol. Sci. 2012, 13, 5506–5518. [Google Scholar] [CrossRef] [PubMed]
- Montefiori, M.; Comeskey, D.J.; Wohlers, M.; McGhie, T.K. Characterization and Quantification of Anthocyanins in Red Kiwifruit (Actinidia Spp.). J. Agric. Food Chem. 2009, 57, 6856–6861. [Google Scholar] [CrossRef] [PubMed]
- Takano, F.; Tanaka, T.; Tsukamoto, E.; Yahagi, N.; Fushiya, S. Isolation of (+)-Catechin and (-)-Epicatechin from Actinidia Arguta as Bone Marrow Cell Proliferation Promoting Compounds. Planta Med. 2003, 69, 321–326. [Google Scholar] [CrossRef]
- Tan, C.; Wang, Z.; Feng, X.; Pan, B.; Irfan, M.; Liu, C. Transcriptomic and Metabolomics of Flavonoid Compounds in Actinidia Arguta Var. Arguta. J. King Saud Univ.-Sci. 2021, 33, 101605. [Google Scholar] [CrossRef]
- Ha, J.S.; Kim, J.M.; Park, S.K.; Kang, J.Y.; Lee, D.S.; Lee, U.; Kim, D.O.; Choi, S.G.; Heo, H.J. Anti-Amyloidogenic Properties of an Ethyl Acetate Fraction from: Actinidia Arguta in Aβ1-42-Induced ICR Mice. Food Funct. 2018, 9, 3264–3277. [Google Scholar] [CrossRef] [PubMed]
- Hanish Singh, J.C.; Alagarsamy, V.; Diwan, P.V.; Sathesh Kumar, S.; Nisha, J.C.; Narsimha Reddy, Y. Neuroprotective Effect of Alpinia Galanga (L.) Fractions on Aβ (25-35) Induced Amnesia in Mice. J. Ethnopharmacol. 2011, 138, 85–91. [Google Scholar] [CrossRef]
- Miyazawa, M.; Hashimoto, Y. Antimicrobial and Bactericidal Activities of Esters of 2-Endo-Hydroxy-1,8-Cineole as New Aroma Chemicals. J. Agric. Food Chem. 2002, 50, 3522–3526. [Google Scholar] [CrossRef]
- Akhtar, M.S.; Khan, M.A.; Malik, M.T. Hypoglycaemic Activity of Alpinia Galanga Rhizome and Its Extracts in Rabbits. Fitoterapia 2002, 73, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Jantan, I.; Salleh, M.; Yassin, M.; Chin, C.B.; Chen, L.L.; Sim, N.L.; Roxb, C.; Linn, K. Fungal Activity of the Essential Oils of Nine Zingiberaceae Species. Pharm. Biol. 2003, 41, 392–397. [Google Scholar] [CrossRef]
- Khattak, S.; Saeed-Ur-Rehman; Shah, H.U.; Khan, T.; Ahmad, M. In Vitro Enzyme Inhibition Activities of Crude Ethanolic Extracts Derived from Medicinal Plants of Pakistan. Nat. Prod. Res. 2005, 19, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Tungmunnithum, D.; Tanaka, N.; Uehara, A.; Iwashina, T. Flavonoids Profile, Taxonomic Data, History of Cosmetic Uses, Anti-Oxidant and Anti-Aging Potential of Alpinia Galanga (L.) Willd. Cosmetics 2020, 7, 89. [Google Scholar] [CrossRef]
- Tasleem, F.; Azhar, I.; Ali, S.N.; Perveen, S.; Mahmood, Z.A. Analgesic and Anti-Inflammatory Activities of Piper Nigrum L. Asian Pac. J. Trop. Med. 2014, 7, S461–S468. [Google Scholar] [CrossRef] [PubMed]
- Ashokkumar, K.; Murugan, M.; Dhanya, M.K.; Pandian, A.; Warkentin, T.D. Phytochemistry and Therapeutic Potential of Black Pepper [Piper Nigrum (L.)] Essential Oil and Piperine: A Review. Clin. Phytosci. 2021, 7, 52. [Google Scholar] [CrossRef]
- Shin, M.; Liu, Q.F.; Choi, B.; Shin, C.; Lee, B.; Yuan, C.; Song, Y.J.; Yun, H.S.; Lee, I.S.; Koo, B.S.; et al. Neuroprotective Effects of Limonene (+) against Aβ42-Induced Neurotoxicity in a Drosophila Model of Alzheimer’s Disease. Biol. Pharm. Bull. 2020, 43, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Hritcu, L.; Noumedem, J.A.; Cioanca, O.; Hancianu, M.; Kuete, V.; Mihasan, M. Methanolic Extract of Piper Nigrum Fruits Improves Memory Impairment by Decreasing Brain Oxidative Stress in Amyloid Beta(1-42) Rat Model of Alzheimer’s Disease. Cell. Mol. Neurobiol. 2014, 34, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Abdulla, K.K.; Taha, E.M.; Rahim, S.M. Phenolic Profile, Antioxidant, and Antibacterial Effects of Ethanol and Aqueous Extracts of Rheum Ribes L. Roots. Der Pharm. Lett. 2015, 7, 26–30. [Google Scholar]
- Noori, S.; Kiasat, A.R.; Kolahi, M.; Mirzajani, R.; Seyyed Nejad, S.M. Determination of Secondary Metabolites Including Curcumin in Rheum Ribes L. and Surveying of Its Antioxidant and Anticancer Activity. J. Saudi Chem. Soc. 2022, 26, 101479. [Google Scholar] [CrossRef]
- El Nebrisi, E. Neuroprotective Activities of Curcumin in Parkinson’s Disease: A Review of the Literature. Int. J. Mol. Sci. 2021, 22, 11248. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, M.; Degirmenci, U.; Akkapulu, M.; Comelekoglu, U.; Balli, E.; Metin Ozcan, T.; Berköz, M.; Yalin, A.E.; Yalin, S. The Effect of Rheum Ribes L. On Oxidative Stress in Diabetic Rats. J. Basic Clin. Physiol. Pharmacol. 2021, 32, 20200058. [Google Scholar] [CrossRef] [PubMed]
- Zahedi, M.; Hojjati, M.R.; Fathpour, H.; Rabieic, Z.; Alibabaei, Z.; Basima, A. Effect of Rheum Ribes Hydro-Alcoholic Extract on Memory Impairments in Rat Model of Alzheimer’s Disease. Iran. J. Pharm. Res. 2015, 14, 1197–1206. [Google Scholar] [PubMed]
- Ibrahim, M.; Kaushik, N.; Sowemimo, A.; Chhipa, H.; Koekemoer, T.; Van De Venter, M.; Odukoya, O.A. Antifungal and Antiproliferative Activities of Endophytic Fungi Isolated from the Leaves of Markhamia Tomentosa. Pharm. Biol. 2017, 55, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.B.; Sowemimo, A.A.; Sofidiya, M.O.; Badmos, K.B.; Fageyinbo, M.S.; Abdulkareem, F.B.; Odukoya, O.A. Sub-Acute and Chronic Toxicity Profiles of Markhamia Tomentosa Ethanolic Leaf Extract in Rats. J. Ethnopharmacol. 2016, 193, 68–75. [Google Scholar] [CrossRef]
- Stanley, T. Research Paper. 10 Perform.-Based Proj. Sci. Classr. 2021, 4, 107–115. [Google Scholar] [CrossRef]
- Tantangmo, F.; Lenta, B.N.; Boyom, F.F.; Ngouela, S.; Kaiser, M.; Tsamo, E.; Weniger, B.; Rosenthal, P.J.; Vonthron-Sénécheau, C. Antiprotozoal Activities of Some Constituents of Markhamia Tomentosa (Bignoniaceae). Ann. Trop. Med. Parasitol. 2010, 104, 391–398. [Google Scholar] [CrossRef]
- Sofidiya, M.O.; Agunbiade, F.O.; Koorbanally, N.A.; Sowemimo, A.; Soesan, D.; Familusi, T. Antiulcer Activity of the Ethanolic Extract and Ethyl Acetate Fraction of the Leaves of Markhamia Tomentosa in Rats. J. Ethnopharmacol. 2014, 157, 1–6. [Google Scholar] [CrossRef]
- Sowemimo, A.; Samuel, F.; Fageyinbo, M.S. Anti-Inflammatory Activity of Markhamia Tomentosa (Benth.) K. Schum. Ex Engl. Ethanolic Leaf Extract. J. Ethnopharmacol. 2013, 149, 191–194. [Google Scholar] [CrossRef]
- Ionita, R.; Postu, P.A.; Beppe, G.J.; Mihasan, M.; Petre, B.A.; Hancianu, M.; Cioanca, O.; Hritcu, L. Cognitive-Enhancing and Antioxidant Activities of the Aqueous Extract from Markhamia Tomentosa (Benth.) K. Schum. Stem Bark in a Rat Model of Scopolamine. Behav. Brain Funct. 2017, 13, 5. [Google Scholar] [CrossRef]
- Mandel, S.; Youdim, M.B.H. Catechin Polyphenols: Neurodegeneration and Neuroprotection in Neurodegenerative Diseases. Free Radic. Biol. Med. 2004, 37, 304–317. [Google Scholar] [CrossRef]
- Guo, H.; Chang, Z.; Yang, R.; Guo, D.; Zheng, J. Anthraquinones from Hairy Root Cultures of Cassia Obtusifolia. Phytochemistry 1998, 49, 1623–1625. [Google Scholar] [CrossRef]
- Wu, D.T.; Liu, W.; Han, Q.H.; Wang, P.; Xiang, X.R.; Ding, Y.; Zhao, L.; Zhang, Q.; Li, S.Q.; Qin, W. Extraction Optimization, Structural Characterization, and Antioxidant Activities of Polysaccharides from Cassia Seed (Cassia Obtusifolia). Molecules 2019, 24, 2817. [Google Scholar] [CrossRef]
- Zhang, W.D.; Wang, Y.; Wang, Q.; Yang, W.J.; Gu, Y.; Wang, R.; Song, X.M.; Wang, X.J. Quality Evaluation of Semen Cassiae (Cassia Obtusifolia L.) by Using Ultra-High Performance Liquid Chromatography Coupled with Mass Spectrometry. J. Sep. Sci. 2012, 35, 2054–2062. [Google Scholar] [CrossRef]
- Leung, S.W.; Lai, J.H.; Wu, J.C.; Tsai, Y. Neuroprotective Effects of Emodin against Ischemia / Reperfusion Injury through Activating ERK-1/2 Signaling Pathway. Int. J. Mol. Sci. 2020, 21, 2899. [Google Scholar] [CrossRef]
- Dong, H.K.; Byung, H.Y.; Kim, Y.W.; Lee, S.; Bum, Y.S.; Ji, W.J.; Hyoung, J.K.; Yong, S.L.; Jae, S.C.; Sun, Y.K.; et al. The Seed Extract of Cassia Obtusifolia Ameliorates Learning and Memory Impairments Induced by Scopolamine or Transient Cerebral Hypoperfusion in Mice. J. Pharmacol. Sci. 2007, 105, 82–93. [Google Scholar] [CrossRef]
- Boozari, M.; Butler, A.E.; Sahebkar, A. Impact of Curcumin on Toll-like Receptors. J. Cell. Physiol. 2019, 234, 12471–12482. [Google Scholar] [CrossRef]
- Abd El-Hack, M.E.; El-Saadony, M.T.; Swelum, A.A.; Arif, M.; Abo Ghanima, M.M.; Shukry, M.; Noreldin, A.; Taha, A.E.; El-Tarabily, K.A. Curcumin, the Active Substance of Turmeric: Its Effects on Health and Ways to Improve Its Bioavailability. J. Sci. Food Agric. 2021, 101, 5747–5762. [Google Scholar] [CrossRef] [PubMed]
- Kouhpeikar, H.; Butler, A.E.; Bamian, F.; Barreto, G.E.; Majeed, M.; Sahebkar, A. Curcumin as a Therapeutic Agent in Leukemia. J. Cell. Physiol. 2019, 234, 12404–12414. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.; Monteiro-Alfredo, T.; Silva, S.; Matafome, P. Curcumin Derivatives for Type 2 Diabetes Management and Prevention of Complications. Arch. Pharm. Res. 2020, 43, 567–581. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, S.; Fathi, N.; Memar, M.Y.; Hosseiniyan Khatibi, S.M.; Khalilov, R.; Negahdari, R.; Zununi Vahed, S.; Maleki Dizaj, S. Anti-Microbial Activity of Curcumin Nanoformulations: New Trends and Future Perspectives. Phyther. Res. 2020, 34, 1926–1946. [Google Scholar] [CrossRef]
- Maiti, P.; Paladugu, L.; Dunbar, G.L. Solid Lipid Curcumin Particles Provide Greater Anti-Amyloid, Anti-Inflammatory and Neuroprotective Effects than Curcumin in the 5xFAD Mouse Model of Alzheimer’s Disease. BMC Neurosci. 2018, 19, 7. [Google Scholar] [CrossRef] [PubMed]
- Ray, B.; Bisht, S.; Maitra, A.; Maitra, A.; Lahiri, D.K. Neuroprotective and Neurorescue Effects of a Novel Polymeric Nanoparticle Formulation of Curcumin (NanoCurcTM) in the Neuronal Cell Culture and Animal Model: Implications for Alzheimer’s Disease. J. Alzheimer’s Dis. 2011, 23, 61–77. [Google Scholar] [CrossRef]
- Samy, D.M.; Ismail, C.A.; Nassra, R.A.; Zeitoun, T.M.; Nomair, A.M. Downstream Modulation of Extrinsic Apoptotic Pathway in Streptozotocin-Induced Alzheimer’s Dementia in Rats: Erythropoietin versus Curcumin. Eur. J. Pharmacol. 2016, 770, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.C.; Zheng, B.W.; Guo, Y.; Zhao, J.; Zhao, J.Y.; Ma, X.W.; Jiang, Z.F. Antioxidative and Neuroprotective Effects of Curcumin in an Alzheimer’s Disease Rat Model Co-Treated with Intracerebroventricular Streptozotocin and Subcutaneous D-Galactose. J. Alzheimer’s Dis. 2016, 52, 899–911. [Google Scholar] [CrossRef] [PubMed]
- Tan, Q.-G.; Luo, X.-D. Correction to Meliaceous Limonoids: Chemistry and Biological Activities. Chem. Rev. 2012, 112, 2591. [Google Scholar] [CrossRef]
- Hitotsuyanagi, Y.; Mitsui, K.; Fukaya, H.; Takeya, K. Fluorinated and Rearranged Gedunin Derivatives. Phytochem. Lett. 2012, 5, 486–489. [Google Scholar] [CrossRef]
- Nagini, S. Neem Limonoids as Anticancer Agents: Modulation of Cancer Hallmarks and Oncogenic Signaling, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2014; Volume 36, ISBN 9780128022153. [Google Scholar]
- Tom, S.; Rane, A.; Katewa, A.S.; Chamoli, M.; Matsumoto, R.R.; Andersen, J.K.; Chinta, S.J. Gedunin Inhibits Oligomeric Aβ1–42-Induced Microglia Activation Via Modulation of Nrf2-NF-ΚB Signaling. Mol. Neurobiol. 2019, 56, 7851–7862. [Google Scholar] [CrossRef] [PubMed]
- Sampson, L.; Rimm, E.; Hollman, P.C.H.; De Vries, J.H.M.; Katan, M.B. Flavonol and Flavone Intakes in US Health Professionals. J. Am. Diet. Assoc. 2002, 102, 1414–1420. [Google Scholar] [CrossRef] [PubMed]
- Wach, A.; Pyrzyńska, K.; Biesaga, M. Quercetin Content in Some Food and Herbal Samples. Food Chem. 2007, 100, 699–704. [Google Scholar] [CrossRef]
- Sandeep, M.S.; Nandini, C.D. Influence of Quercetin, Naringenin and Berberine on Glucose Transporters and Insulin Signalling Molecules in Brain of Streptozotocin-Induced Diabetic Rats. Biomed. Pharmacother. 2017, 94, 605–611. [Google Scholar] [CrossRef]
- Zheng, Y.Z.; Deng, G.; Liang, Q.; Chen, D.F.; Guo, R.; Lai, R.C. Antioxidant Activity of Quercetin and Its Glucosides from Propolis: A Theoretical Study. Sci. Rep. 2017, 7, 7543. [Google Scholar] [CrossRef]
- Van Acker, S.A.B.E.; Van Balen, G.P.; Van Den Berg, D.J.; Bast, A.; Van Der Vijgh, W.J.F. Influence of Iron Chelation on the Antioxidant Activity of Flavonoids. Biochem. Pharmacol. 1998, 56, 935–943. [Google Scholar] [CrossRef] [PubMed]
- Afanas’ev, I.B.; Dcrozhko, A.I.; Brodskii, A.V.; Kostyuk, V.A.; Potapovitch, A.I. Chelating and Free Radical Scavenging Mechanisms of Inhibitory Action of Rutin and Quercetin in Lipid Peroxidation. Biochem. Pharmacol. 1989, 38, 1763–1769. [Google Scholar] [CrossRef]
- Min, Y.D.; Choi, C.H.; Bark, H.; Son, H.Y.; Park, H.H.; Lee, S.; Park, J.W.; Park, E.K.; Shin, H.I.; Kim, S.H. Quercetin Inhibits Expression of Inflammatory Cytokines through Attenuation of NF-ΚB and P38 MAPK in HMC-1 Human Mast Cell Line. Inflamm. Res. 2007, 56, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.L.; Lyon, T.; Litwin, S.E.; Rabovsky, A.; Symons, J.D.; Jalili, T. Quercetin Reduces Blood Pressure in Hypertensive Subjects. J. Nutr. 2007, 137, 2405–2411. [Google Scholar] [CrossRef] [PubMed]
- Booyse, F.M.; Pan, W.; Grenett, H.E.; Parks, D.A.; Darley-Usmar, V.M.; Bradley, K.M.; Tabengwa, E.M. Mechanism by Which Alcohol and Wine Polyphenols Affect Coronary Heart Disease Risk. Ann. Epidemiol. 2007, 17, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.A.; Abdul, H.M.; Joshi, G.; Opii, W.O.; Butterfield, D.A. Protective Effect of Quercetin in Primary Neurons against Aβ(1-42): Relevance to Alzheimer’s Disease. J. Nutr. Biochem. 2009, 20, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Ashrafpour, M.; Parsaei, S.; Sepehri, H. Quercetin Improved Spatial Memory Dysfunctions in Rat Model of Intracerebroventricular Streptozotocin-Induced Sporadic Alzheimer’sdisease. Natl. J. Physiol. Pharm. Pharmacol. 2015, 5, 411–415. [Google Scholar] [CrossRef]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol Improves Health and Survival of Mice on a High-Calorie Diet. Nature 2006, 444, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Baur, J.A.; Sinclair, D.A. Therapeutic Potential of Resveratrol: The in Vivo Evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Gocmez, S.S.; Gacar, N.; Utkan, T.; Gacar, G.; Scarpace, P.J.; Tumer, N. Protective Effects of Resveratrol on Aging-Induced Cognitive Impairment in Rats. Neurobiol. Learn. Mem. 2016, 131, 131–136. [Google Scholar] [CrossRef]
- Xia, N.; Daiber, A.; Förstermann, U.; Li, H. Antioxidant Effects of Resveratrol in the Cardiovascular System. Br. J. Pharmacol. 2017, 174, 1633–1646. [Google Scholar] [CrossRef] [PubMed]
- Udenigwe, C.C.; Ramprasath, V.R.; Aluko, R.E.; Jones, P.J.H. Potential of Resveratrol in Anticancer and Anti-Inflammatory Therapy. Nutr. Rev. 2008, 66, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Yazir, Y.; Utkan, T.; Gacar, N.; Aricioglu, F. Resveratrol Exerts Anti-Inflammatory and Neuroprotective Effects to Prevent Memory Deficits in Rats Exposed to Chronic Unpredictable Mild Stress. Physiol. Behav. 2015, 138, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Gupta, Y.K. Chronic Treatment with Trans Resveratrol Prevents Intracerebroventricular Streptozotocin Induced Cognitive Impairment and Oxidative Stress in Rats. Life Sci. 2002, 71, 2489–2498. [Google Scholar] [CrossRef] [PubMed]
- Nogata, Y.; Sakamoto, K.; Shiratsuchi, H.; Ishii, T.; Yano, M.; Ohta, H. Flavonoid Composition of Fruit Tissues of Citrus Species. Biosci. Biotechnol. Biochem. 2006, 70, 178–192. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Meng, D.; Zhang, P.; Wang, X.; Du, G.; Brennan, C.; Li, S.; Ho, C.T.; Zhao, H. Antioxidant Protection of Nobiletin, 5-Demethylnobiletin, Tangeretin, and 5-Demethyltangeretin from Citrus Peel in Saccharomyces Cerevisiae. J. Agric. Food Chem. 2018, 66, 3155–3160. [Google Scholar] [CrossRef]
- Miyamoto, S.; Yasui, Y.; Tanaka, T.; Ohigashi, H.; Murakami, A. Suppressive Effects of Nobiletin on Hyperleptinemia and Colitis-Related Colon Carcinogenesis in Male ICR Mice. Carcinogenesis 2008, 29, 1057–1063. [Google Scholar] [CrossRef]
- Matsuzaki, K.; Yamakuni, T.; Hashimoto, M.; Haque, A.M.; Shido, O.; Mimaki, Y.; Sashida, Y.; Ohizumi, Y. Nobiletin Restoring β-Amyloid-Impaired CREB Phosphorylation Rescues Memory Deterioration in Alzheimer’s Disease Model Rats. Neurosci. Lett. 2006, 400, 230–234. [Google Scholar] [CrossRef]
- Nakajima, A.; Aoyama, Y.; Shin, E.J.; Nam, Y.; Kim, H.C.; Nagai, T.; Yokosuka, A.; Mimaki, Y.; Yokoi, T.; Ohizumi, Y.; et al. Nobiletin, a Citrus Flavonoid, Improves Cognitive Impairment and Reduces Soluble Aβ Levels in a Triple Transgenic Mouse Model of Alzheimer’s Disease (3XTg-AD). Behav. Brain Res. 2015, 289, 69–77. [Google Scholar] [CrossRef]
- Arai, Y.; Watanabe, S.; Kimira, M.; Shimoi, K.; Mochizuki, R.; Kinae, N. Dietary Intakes of Flavonols, Flavones and Isoflavones by Japanese Women and the Inverse Correlation between Quercetin Intake and Plasma LDL Cholesterol Concentration. J. Nutr. 2000, 130, 2243–2250. [Google Scholar] [CrossRef]
- Ishige, K.; Schubert, D.; Sagara, Y. Flavonoids Protect Neuronal Cells from Oxidative Stress by Three Distinct Mechanisms. Free Radic. Biol. Med. 2001, 30, 433–446. [Google Scholar] [CrossRef]
- Akaishi, T.; Morimoto, T.; Shibao, M.; Watanabe, S.; Sakai-Kato, K.; Utsunomiya-Tate, N.; Abe, K. Structural Requirements for the Flavonoid Fisetin in Inhibiting Fibril Formation of Amyloid β Protein. Neurosci. Lett. 2008, 444, 280–285. [Google Scholar] [CrossRef]
- Sagara, Y.; Vanhnasy, J.; Maher, P. Induction of PC12 Cell Differentiation by Flavonoids Is Dependent upon Extracellular Signal-Regulated Kinase Activation. J. Neurochem. 2004, 90, 1144–1155. [Google Scholar] [CrossRef] [PubMed]
- Das, J.; Singh, R.; Sharma, D. Antiepileptic Effect of Fisetin in Iron-Induced Experimental Model of Traumatic Epilepsy in Rats in the Light of Electrophysiological, Biochemical, and Behavioral Observations. Nutr. Neurosci. 2017, 20, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Ali, T.; Park, H.Y.; Badshah, H.; Rehman, S.U.; Kim, M.O. Neuroprotective Effect of Fisetin Against Amyloid-Beta-Induced Cognitive/Synaptic Dysfunction, Neuroinflammation, and Neurodegeneration in Adult Mice. Mol. Neurobiol. 2017, 54, 2269–2285. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Sur, B.; Shim, I.; Lee, H.; Hahm, D.H. Phellodendron Amurense and Its Major Alkaloid Compound, Berberine Ameliorates Scopolamine-Induced Neuronal Impairment and Memory Dysfunction in Rats. Korean J. Physiol. Pharmacol. 2012, 16, 79–89. [Google Scholar] [CrossRef]
- Xuan, W.-T.; Wang, H.; Zhou, P.; Ye, T.; Gao, H.-W.; Ye, S.; Wang, J.-H.; Chen, M.-L.; Song, H.; Wang, Y.; et al. Berberine Ameliorates Rats Model of Combined Alzheimer’s Disease and Type 2 Diabetes Mellitus via the Suppression of Endoplasmic Reticulum Stress. 3 Biotech 2020, 10, 359. [Google Scholar] [CrossRef] [PubMed]
- Cok, A.; Plaisier, C.; Salie, M.J.; Oram, D.S.; Chenge, J.; Louters, L.L. Berberine Acutely Activates the Glucose Transport Activity of GLUT1. Biochimie 2011, 93, 1187–1192. [Google Scholar] [CrossRef]
- Gu, Y.; Zhang, Y.; Shi, X.; Li, X.; Hong, J.; Chen, J.; Gu, W.; Lu, X.; Xu, G.; Ning, G. Effect of Traditional Chinese Medicine Berberine on Type 2 Diabetes Based on Comprehensive Metabonomics. Talanta 2010, 81, 766–772. [Google Scholar] [CrossRef]
- Peng, W.H.; Hsieh, M.T.; Wu, C.R. Effect of Long-Term Administration of Berberine on Scopolamine-Induced Amnesia in Rats. Jpn. J. Pharmacol. 1997, 74, 261–266. [Google Scholar] [CrossRef]
- Hussien, H.M.; Abd-Elmegied, A.; Ghareeb, D.A.; Hafez, H.S.; Ahmed, H.E.A.; El-moneam, N.A. Neuroprotective Effect of Berberine against Environmental Heavy Metals-Induced Neurotoxicity and Alzheimer’s-like Disease in Rats. Food Chem. Toxicol. 2018, 111, 432–444. [Google Scholar] [CrossRef]
- Güzel, E.; Koçyiğit, Ü.M.; Taslimi, P.; Erkan, S.; Taskin, O.S. Biologically Active Phthalocyanine Metal Complexes: Preparation, Evaluation of α-Glycosidase and Anticholinesterase Enzyme Inhibition Activities, and Molecular Docking Studies. J. Biochem. Mol. Toxicol. 2021, 35, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Elbouzidi, A.; Taibi, M.; Ouassou, H.; Ouahhoud, S.; Ou-Yahia, D.; Loukili, E.H.; Aherkou, M.; Mansouri, F.; Bencheikh, N.; Laaraj, S. Exploring the Multi-Faceted Potential of Carob (Ceratonia siliqua var. Rahma) Leaves from Morocco: A Comprehensive Analysis of Polyphenols Profile, Antimicrobial Activity, Cytotoxicity against Breast Cancer Cell Lines, and Genotoxicity. Pharmaceuticals 2023, 16, 840. [Google Scholar] [CrossRef]
- Taibi, M.; Elbouzidi, A.; Ou-Yahia, D.; Dalli, M.; Bellaouchi, R.; Tikent, A.; Roubi, M.; Gseyra, N.; Asehraou, A.; Hano, C.; et al. Assessment of the Antioxidant and Antimicrobial Potential of Ptychotis Verticillata Duby Essential Oil from Eastern Morocco: An In Vitro and In Silico Analysis. Antibiotics 2023, 12, 655. [Google Scholar] [CrossRef] [PubMed]
- Mendie, L.E.; Hemalatha, S. Molecular Docking of Phytochemicals Targeting GFRs as Therapeutic Sites for Cancer: An In Silico Study. Appl. Biochem. Biotechnol. 2022, 194, 215–231. [Google Scholar] [CrossRef] [PubMed]
- Mrabti, H.N.; El Hachlafi, N.; Al-Mijalli, S.H.; Jeddi, M.; Elbouzidi, A.; Abdallah, E.M.; Flouchi, R.; Assaggaf, H.; Qasem, A.; Zengin, G.; et al. Phytochemical Profile, Assessment of Antimicrobial and Antioxidant Properties of Essential Oils of Artemisia Herba-Alba Asso., and Artemisia Dracunculus L.: Experimental and Computational Approaches. J. Mol. Struct. 2023, 1294, 136479. [Google Scholar] [CrossRef]
- Al-Mijalli, S.H.; Mrabti, H.N.; El Hachlafi, N.; El Kamili, T.; Elbouzidi, A.; Abdallah, E.M.; Flouchi, R.; Assaggaf, H.; Qasem, A.; Zengin, G. Integrated Analysis of Antimicrobial, Antioxidant, and Phytochemical Properties of Cinnamomum Verum: A Comprehensive In Vitro and In Silico Study. Biochem. Syst. Ecol. 2023, 110, 104700. [Google Scholar] [CrossRef]
- Ishola, A.A.; Oyinloye, B.E.; Ajiboye, B.O.; Kappo, A.P. Molecular Docking Studies of Flavonoids from Andrographis Paniculata as Potential Acetylcholinesterase, Butyrylcholinesterase and Monoamine Oxidase Inhibitors towards the Treatment of Neurodegenerative Diseases. Biointerface Res. Appl. Chem. 2021, 11, 9871–9879. [Google Scholar] [CrossRef]
- Kandsi, F.; Elbouzidi, A.; Lafdil, F.Z.; Meskali, N.; Azghar, A.; Addi, M.; Hano, C.; Maleb, A.; Gseyra, N. Antibacterial and Antioxidant Activity of Dysphania Ambrosioides (L.) Mosyakin and Clemants Essential Oils: Experimental and Computational Approaches. Antibiotics 2022, 11, 482. [Google Scholar] [CrossRef] [PubMed]
- Elbouzidi, A.; Ouassou, H.; Aherkou, M.; Kharchoufa, L.; Meskali, N.; Baraich, A.; Mechchate, H.; Bouhrim, M.; Idir, A.; Hano, C.; et al. LC–MS/MS Phytochemical Profiling, Antioxidant Activity, and Cytotoxicity of the Ethanolic Extract of Atriplex Halimus L. against Breast Cancer Cell Lines: Computational Studies and Experimental Validation. Pharmaceuticals 2022, 15, 1156. [Google Scholar] [CrossRef] [PubMed]
- Gligorić, E.; Igić, R.; Suvajdžić, L.; Grujić-Letić, N. Species of the Genus Salix L.: Biochemical Screening and Molecular Docking Approach to Potential Acetylcholinesterase Inhibitors. Appl. Sci. 2019, 9, 1842. [Google Scholar] [CrossRef]
- Fan, J.; Fu, A.; Zhang, L. Progress in Molecular Docking. Quant. Biol. 2019, 7, 83–89. [Google Scholar] [CrossRef]
- Rizvi, S.M.D.; Shakil, S.; Haneef, M. A Simple Click by Click Protocol to Perform Docking: Autodock 4.2 Made Easy for Non-Bioinformaticians. EXCLI J. 2013, 12, 830–857. [Google Scholar]
- Paul Gleeson, M.; Hersey, A.; Hannongbua, S. In-Silico ADME Models: A General Assessment of Their Utility in Drug Discovery Applications. Curr. Top. Med. Chem. 2011, 11, 358–381. [Google Scholar] [CrossRef] [PubMed]
- Daina, A.; Zoete, V. A BOILED-Egg To Predict Gastrointestinal Absorption and Brain Penetration of Small Molecules. ChemMedChem 2016, 11, 1117–1121. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A Free Web Tool to Evaluate Pharmacokinetics, Drug-Likeness and Medicinal Chemistry Friendliness of Small Molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [PubMed]
- Ishola, A.A.; Adewole, K.E. In Silico Screening of Anticholinesterase Alkaloids for Cyclooxygenase-2 (COX-2) and Matrix Metalloproteinase 8 (MMP-8) Inhibitory Potentials as Multi-Target Inhibitors of Alzheimer’s Disease. Med. Chem. Res. 2019, 28, 1704–1717. [Google Scholar] [CrossRef]
- Guo, H.; Chen, Y.H.; Wang, T.M.; Kang, T.G.; Sun, H.Y.; Pei, W.H.; Song, H.P.; Zhang, H. A Strategy to Discover Selective α-Glucosidase/Acetylcholinesterase Inhibitors from Five Function-Similar Citrus Herbs through LC-Q-TOF-MS, Bioassay and Virtual Screening. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2021, 1174, 122722. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, A.; Yamakuni, T.; Haraguchi, M.; Omae, N.; Song, S.Y.; Kato, C.; Nakagawasai, O.; Tadano, T.; Yokosuka, A.; Mimaki, Y.; et al. Nobiletin, a Citrus Flavonoid That Improves Memory Impairment, Rescues Bulbectomy-Induced Cholinergic Neurodegeneration in Mice. J. Pharmacol. Sci. 2007, 105, 122–126. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Tundis, R.; Bonesi, M.; Menichini, F.; De Luca, D.; Colica, C.; Menichini, F. Evaluation of Citrus Aurantifolia Peel and Leaves Extracts for Their Chemical Composition, Antioxidant and Anti-Cholinesterase Activities. J. Sci. Food Agric. 2012, 92, 2960–2967. [Google Scholar] [CrossRef]
- Mishra, A. Molecular Docking Analysis of Modified Gedunin from Neem with Snake Venom Enzymes. Bioinformation 2021, 17, 776–783. [Google Scholar] [CrossRef]
- Bajda, M.; Wiȩckowska, A.; Hebda, M.; Guzior, N.; Sotriffer, C.A.; Malawska, B. Structure-Based Search for New Inhibitors of Cholinesterases. Int. J. Mol. Sci. 2013, 14, 5608–5632. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.D.; Nguyen, T.C.V.; Nguyen, N.S.; Nguyen, D.M.; Nguyen, T.T.H.; Le, M.T.; Thai, K.M. Synthesis of Novel Chalcones as Acetylcholinesterase Inhibitors. Appl. Sci. 2016, 6, 198. [Google Scholar] [CrossRef]
- Lipinski, C.A. Drug-like Properties and the Causes of Poor Solubility and Poor Permeability. J. Pharmacol. Toxicol. Methods 2000, 44, 235–249. [Google Scholar] [CrossRef]
- Veber, D.F.; Johnson, S.R.; Cheng, H.-Y.; Smith, B.R.; Ward, K.W.; Kopple, K.D. Molecular Properties That Influence the Oral Bioavailability of Drug Candidates. J. Med. Chem. 2002, 45, 2615–2623. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, J.T.; Clark, D.E. In Silico Predictions of Blood-Brain Barrier Penetration: Considerations to “Keep in Mind”. J. Pharmacol. Exp. Ther. 2005, 315, 477–483. [Google Scholar] [CrossRef]
- Pardridge, W.M. Alzheimer’s Disease Drug Development and the Problem of the Blood-Brain Barrier. Alzheimer’s Dement. 2009, 5, 427–432. [Google Scholar] [CrossRef] [PubMed]



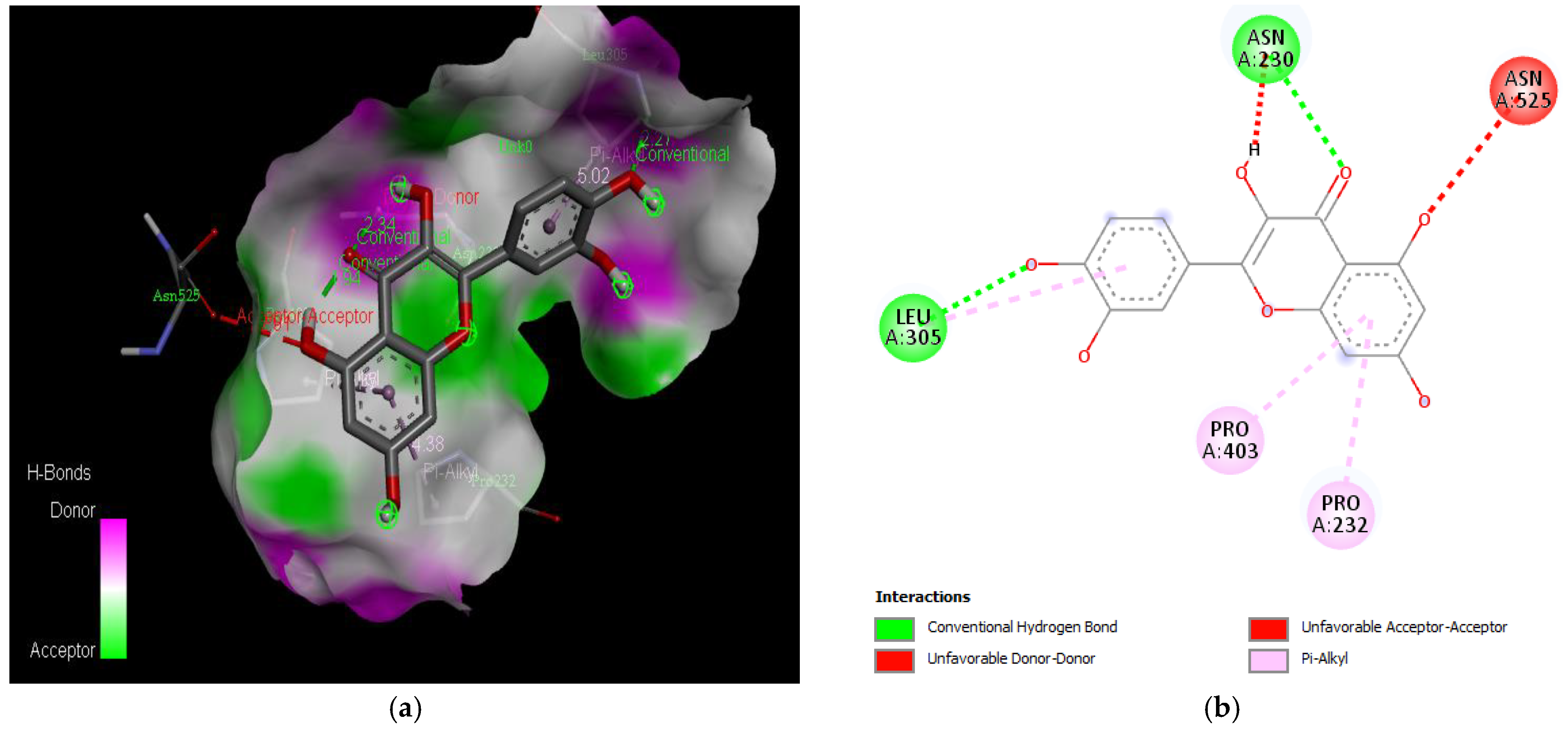
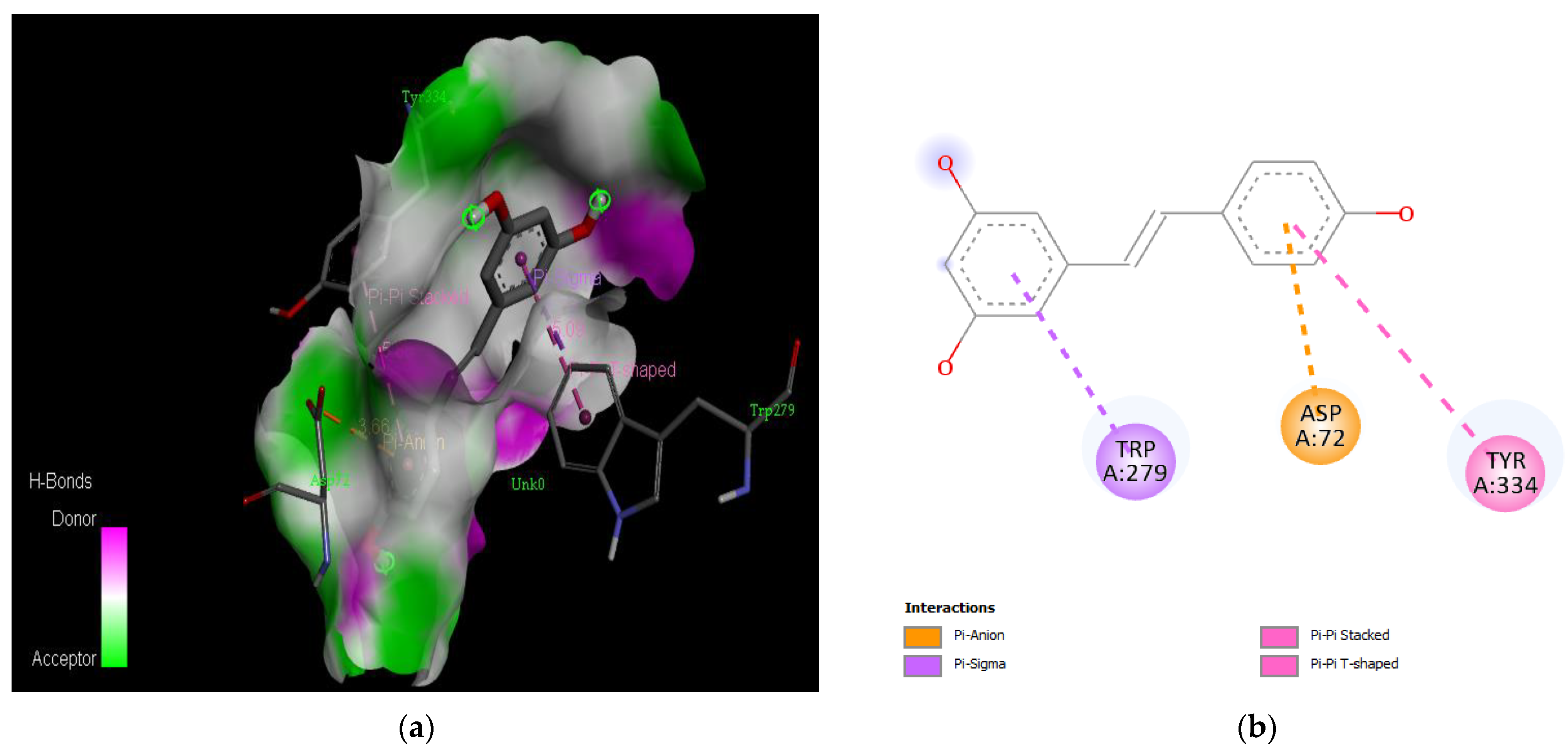
| Compound | Binding Affinity (Kcal/mol) | Interaction Site |
|---|---|---|
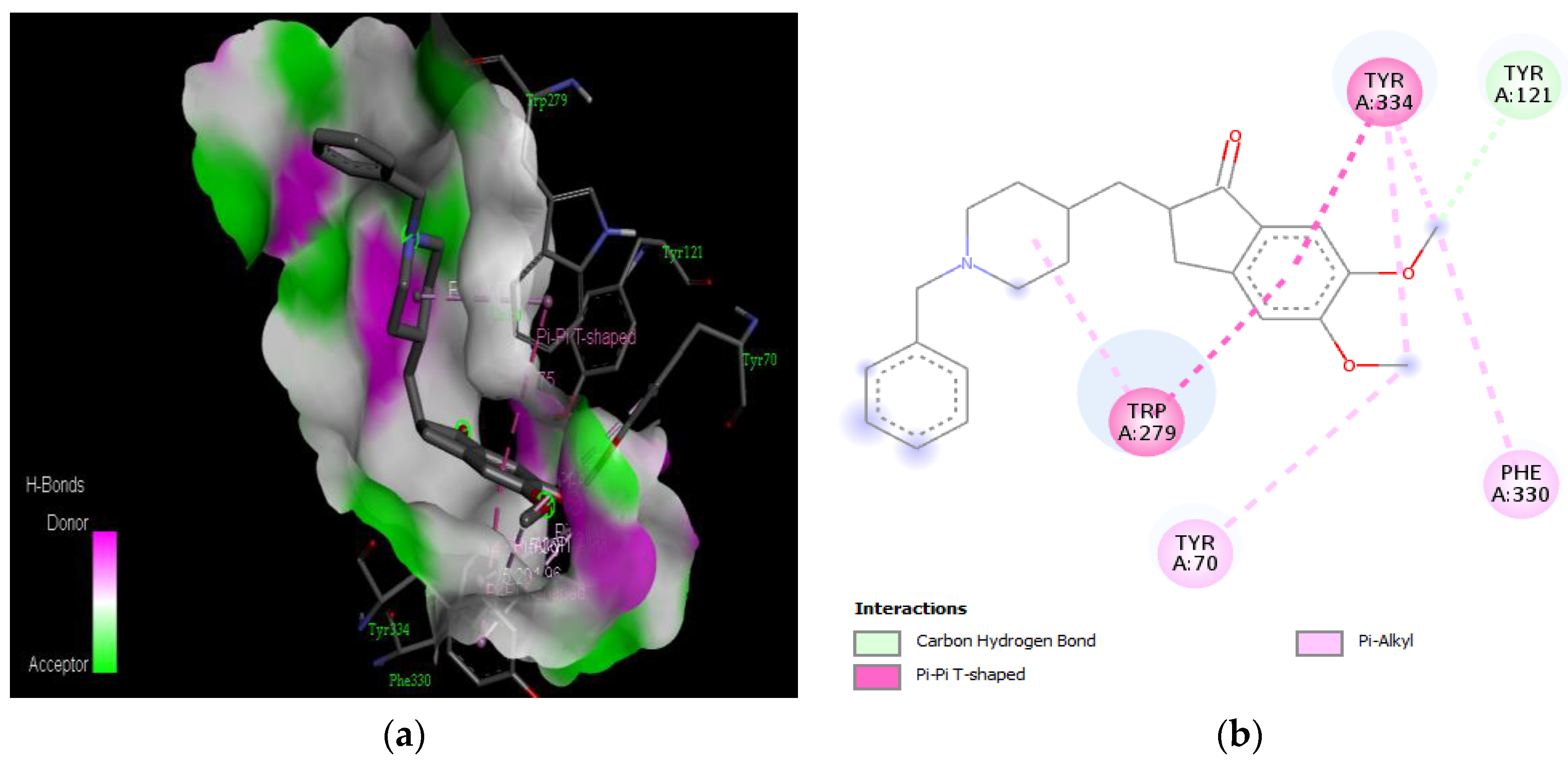
| Donepezil | −8.6 | Tyr 70, Tyr 121, Trp 279, Phe 330, Tyr 334 |
| Curcumin | −7.5 | Tyr 121, Trp 279, Leu 282, Phe 288, Arg 289, Tyr 334 |
| Gedunin | −8.7 | Asn 230, Pro 232, His 398, Pro 403, His 406 |
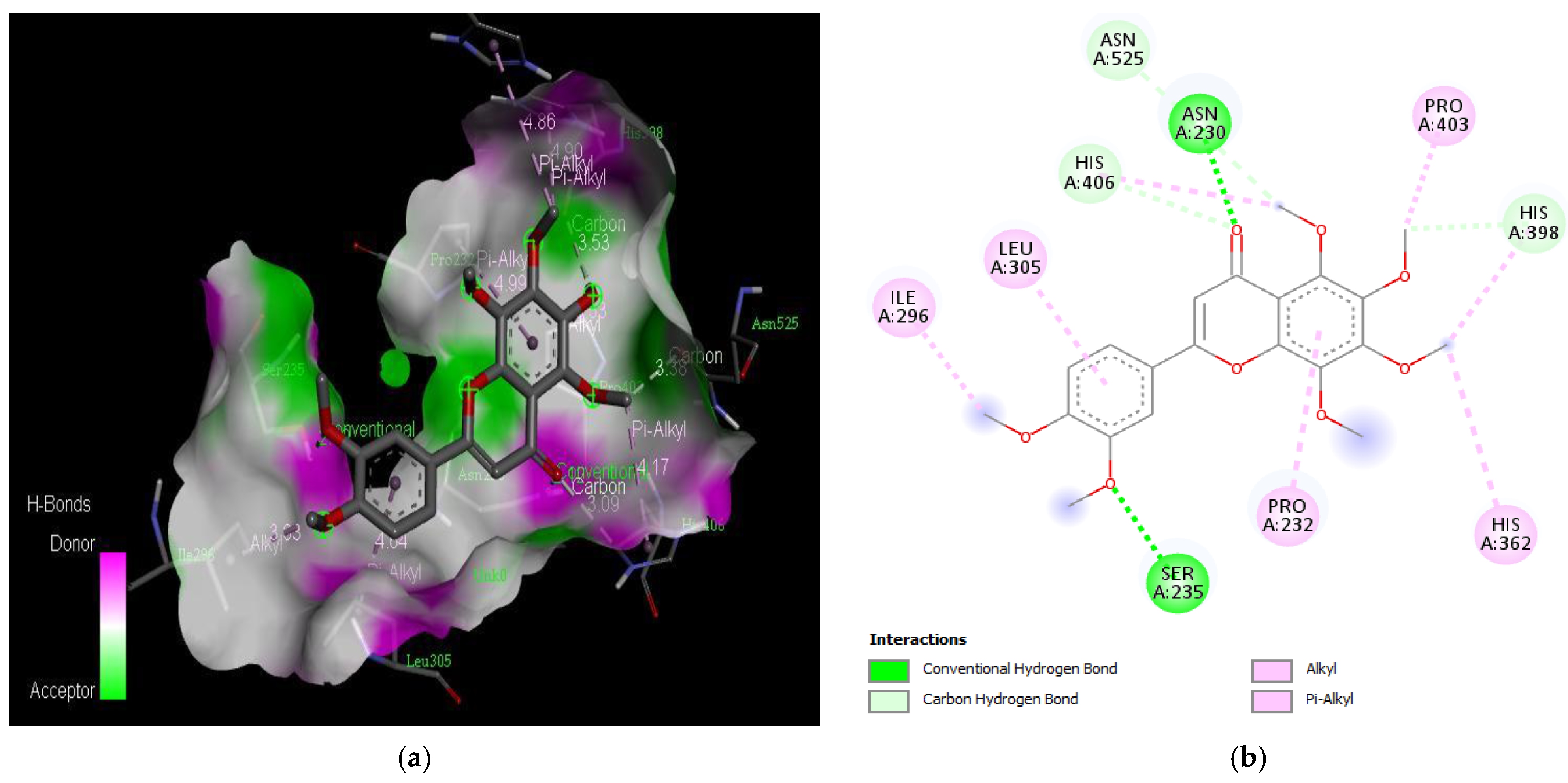
| Quercetin | −7.5 | Asn 230, Pro 232, Leu 305, Pro 403, Asn 525 |
| Resveratrol | −7.0 | Asp 72, Trp 279, Tyr 334 |
| Nobiletin | −6.9 | Asn 230, Pro 232, Ser 235, Ile 296, Leu 305, His 362, His 398, Pro 403, His 406, Asn 525 |
| Fisetin | −8.2 | Tyr 121, Trp 279, Leu 282, Ser 286, Ser 291, Phe 330 |
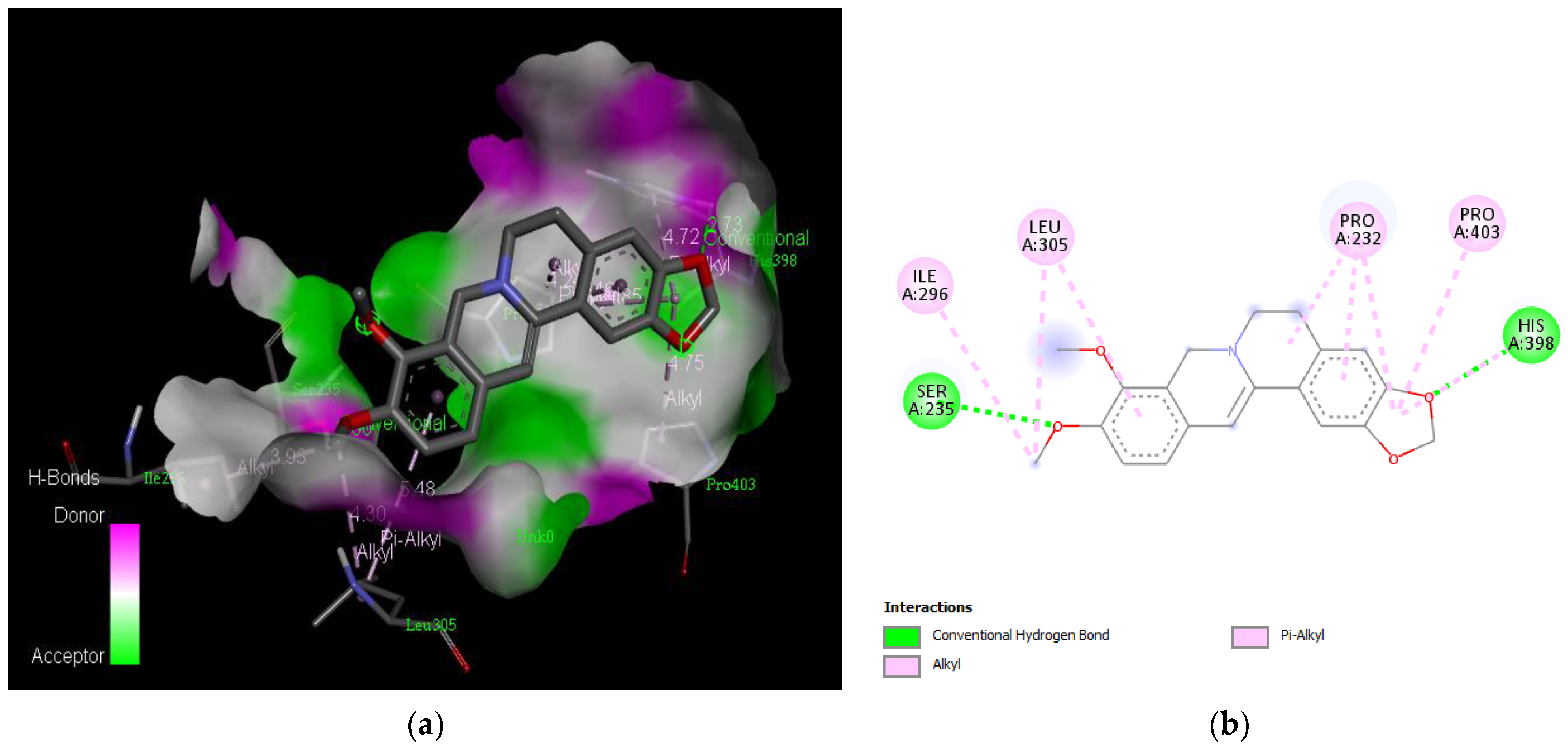
| Berberine | −7.7 | Pro 232, Ser 235, Ile 296, Leu 305, His 398, Pro 403 |
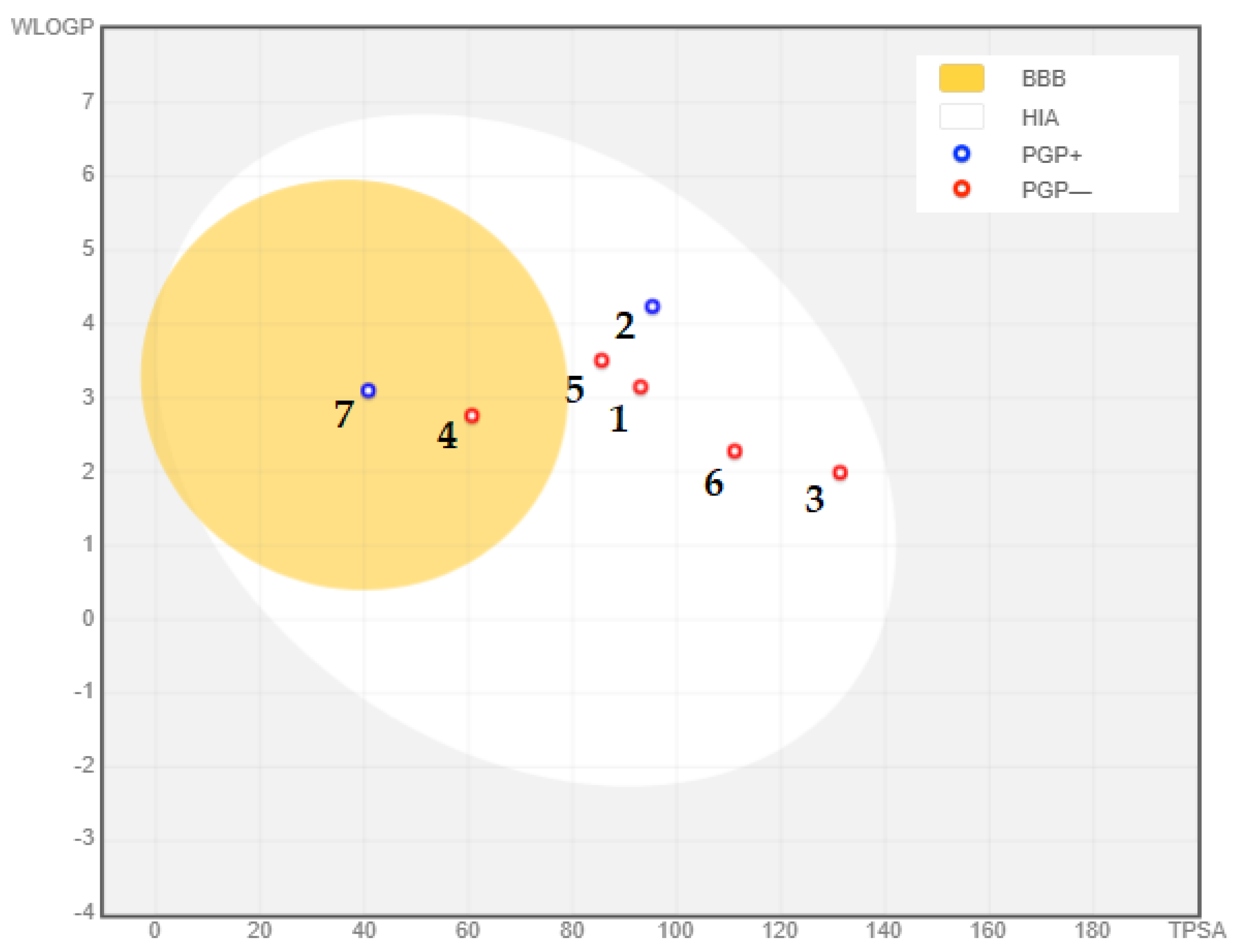
| S/N | Compounds | Molecular Weight | HBA | HBD | MLogP | Lipinski’s Violations | Rotatable Bonds | TPSA | Veber’s Violation | WLogP | BBB Permeation |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Curcumin | 368.38 | 6 | 2 | 1.47 | 0 | 8 | 93.06 | 0 | 3.15 | No |
| 2 | Gedunin | 482.57 | 7 | 0 | 2.56 | 0 | 3 | 95.34 | 0 | 4.24 | No |
| 3 | Quercetin | 302.24 | 7 | 5 | −0.56 | 0 | 1 | 131.36 | 0 | 1.99 | No |
| 4 | Resveratrol | 228.24 | 3 | 3 | 2.26 | 0 | 2 | 60.69 | 0 | 2.76 | Yes |
| 5 | Nobiletin | 402.39 | 8 | 0 | 0.34 | 0 | 7 | 85.59 | 0 | 3.51 | No |
| 6 | Fisetin | 286.24 | 6 | 4 | −0.03 | 0 | 1 | 111.13 | 0 | 2.28 | No |
| 7 | Berberine | 336.36 | 4 | 0 | 2.19 | 0 | 2 | 40.80 | 0 | 3.10 | Yes |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farihi, A.; Bouhrim, M.; Chigr, F.; Elbouzidi, A.; Bencheikh, N.; Zrouri, H.; Nasr, F.A.; Parvez, M.K.; Alahdab, A.; Ahami, A.O.T. Exploring Medicinal Herbs’ Therapeutic Potential and Molecular Docking Analysis for Compounds as Potential Inhibitors of Human Acetylcholinesterase in Alzheimer’s Disease Treatment. Medicina 2023, 59, 1812. https://doi.org/10.3390/medicina59101812
Farihi A, Bouhrim M, Chigr F, Elbouzidi A, Bencheikh N, Zrouri H, Nasr FA, Parvez MK, Alahdab A, Ahami AOT. Exploring Medicinal Herbs’ Therapeutic Potential and Molecular Docking Analysis for Compounds as Potential Inhibitors of Human Acetylcholinesterase in Alzheimer’s Disease Treatment. Medicina. 2023; 59(10):1812. https://doi.org/10.3390/medicina59101812
Chicago/Turabian StyleFarihi, Ayoub, Mohamed Bouhrim, Fatiha Chigr, Amine Elbouzidi, Noureddine Bencheikh, Hassan Zrouri, Fahd A. Nasr, Mohammad Khalid Parvez, Ahmad Alahdab, and Ahmed Omar Touhami Ahami. 2023. "Exploring Medicinal Herbs’ Therapeutic Potential and Molecular Docking Analysis for Compounds as Potential Inhibitors of Human Acetylcholinesterase in Alzheimer’s Disease Treatment" Medicina 59, no. 10: 1812. https://doi.org/10.3390/medicina59101812







