1. Introduction
Endoscopic necrosectomy (EN) for treating walled-off pancreatic necrosis (WON) is less invasive and more life-saving than surgery. However, arterial bleeding during and after EN can occur in 10–20% of cases [
1,
2,
3]. Some useful hemostatic methods, including the use of endoscopic clips and vascular embolization using interventional radiology (IR), have been reported in arterial bleeding therapy during EN [
4]. However, these methods have shown some drawbacks as the clips remain within the WON cavity [
5] and the IR must temporarily interrupt the EN. Cautery hemostasis using a Coagrasper (Olympus, Tokyo, Japan) has been reported to have a high hemostatic effect that is achieved through directly grasping and coagulating the blood vessels for active gastrointestinal bleeding, as well as during endoscopic submucosal dissection procedures [
6,
7]. Herein, we report the effectiveness of using the Coagrasper for managing arterial bleeding due to arterial injury during EN for WON treatment, including the precautions regarding its use.
2. Case Presentation
A 60-year-old man without previous medical history presented to our hospital with severe abdominal pain. His heart rate was regular, at 69 beats/min, his blood pressure was 120/78 mmHg, and his body temperature was 36.7 °C. The patient was diagnosed with pancreatitis due to a common bile duct stone (CBDS) and underwent endoscopic sphincterotomy (ES) to remove it. However, although the CBDS was completely removed, the pancreatitis was worsened, and an acute kidney injury and respiratory failure were observed. The patient was treated using an artificial respirator and continuous hemodiafiltration in the intensive care unit.
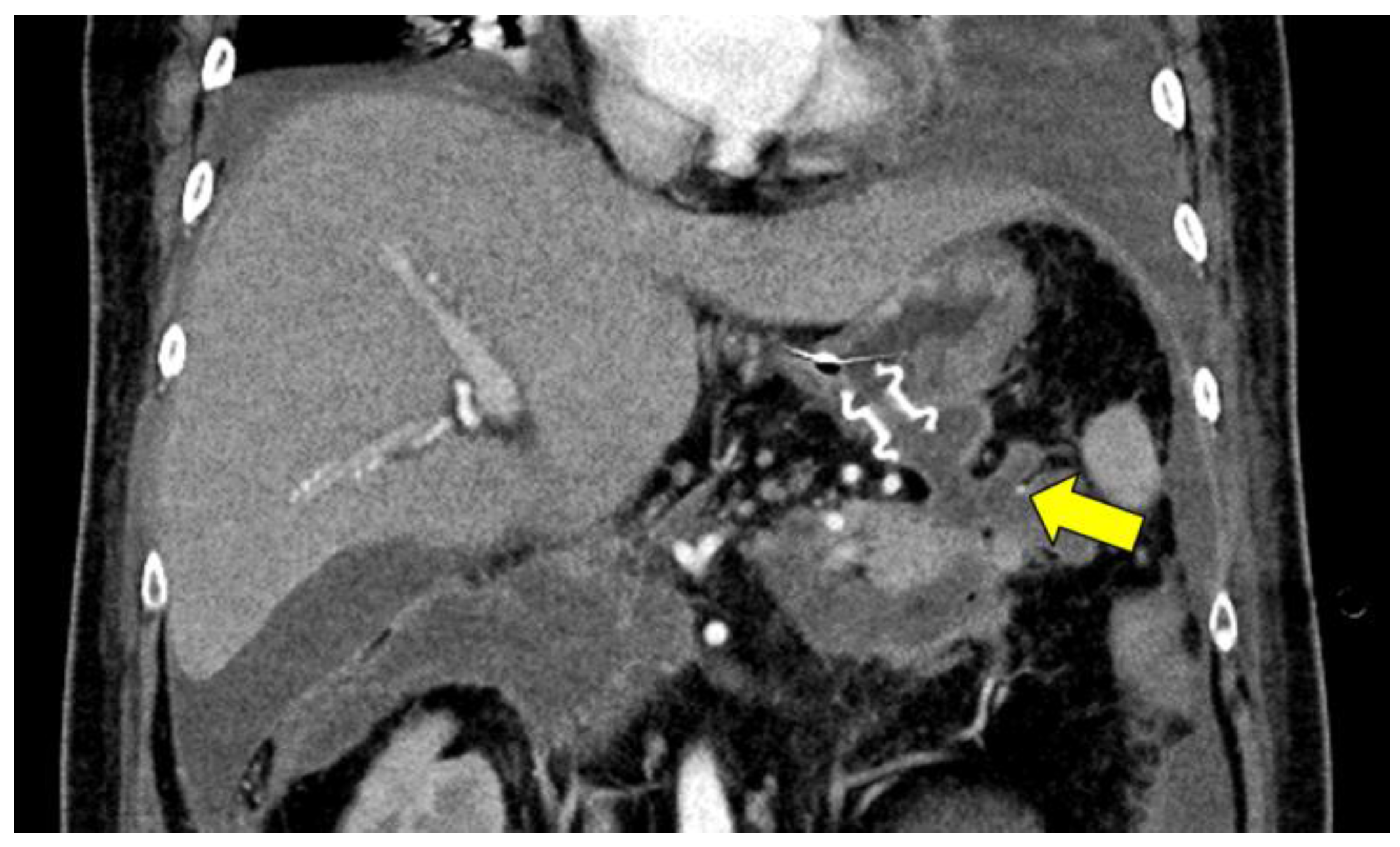
The patient’s condition improved with intensive therapy 38 days after undergoing the ES. However, computed tomography revealed a walled-off necrosis (WON) around the pancreas. An infected WON was considered because an elevated C-reactive protein level (8.8 mg/dL) and high fever were observed. Endoscopic ultrasound-guided transgastric drainage for the pancreatic tail WON cavity using a 15 mm lumen-apposing metal stent (LAMS) (Hot Axios; Boston Scientific, Marlborough, MA, USA) was performed (
Figure 1), followed by EN, which was performed using five-legged forceps.
WON is observed around the pancreas. The small vessels are observed in the pancreatic tail WON cavity (arrow). LAMS is placed into the pancreatic tail side WON cavity. During the fourth session, the arterial wall was damaged by forceps, and active bleeding was observed (
Figure 2A). Because the artery could be directly observed and was 2–3 mm in diameter, we considered the “Coagrasper” to grasp and coagulate the vessel to achieve hemostasis. Therefore, we grasped and coagulated the bleeding point using the electrosurgical unit ERBE VIO 300D (setting, SOFT COAG mode; effect, 5; max watts, 80; ERBE, Tubingen, Germany); however, the bleeding was aggravated after coagulation owing to the spread of vascular damage. We then grasped the entire vessel and repeatedly coagulated it. Finally, complete hemostasis was achieved (
Figure 2C,D,
Supplemental Video S1). No bleeding or adverse events were observed 3 days after the vascular cautery. The patient could avoid undergoing vascular embolization with IR and continued the EN procedure. Late adverse events related to the use of the Coagrasper were not evaluated until the completion of the EN procedure.
3. Discussion
Only two studies have reported on the use of Coagrasper for arterial bleeding during EN [
8,
9]. These reports documented successful hemostasis for small-vessel bleeding within the gastric wall or the WON cavity. When most vessels were covered by surrounding tissues, hemostasis was achieved by cauterizing the bleeding point with the surrounding tissues. In this report, we also demonstrated the usefulness of the Coagrasper for obtaining hemostasis in completely denuded vessels in the WON cavity. However, as demonstrated in
Figure 2A, inadequate grasping of the vessel can worsen the condition due to the spread of vascular damage. As the artery in our case was not covered by surrounding tissue, grasping the entire vessel was necessary for sufficient cauterization. Therefore, whether the targeted area for grasping should solely encompass the bleeding point or the entire vessel should be ascertained while considering the condition of the vessel (whether it is covered by surrounding tissues or fully exposed) before using the Coagrasper. Moreover, because the Coagrasper has a 5 mm aperture diameter, a blood vessel with a 2–3 mm diameter is considered susceptible to complete grasping and subsequent cauterization. Therefore, when cauterization of an entire vessel is required, the vessel size must also be evaluated when using the Coagrasper.
Holmes et al. reported the treatment of hemorrhages during EN and emphasized that coil embolization under IR is necessary for pseudoaneurysms [
4]. Because the Coagrasper can achieve hemostasis by cauterizing and coagulating vessels and/or tissues, attaining hemostasis for a pseudoaneurysm is challenging. It is often difficult to determine whether the bleeding vessel is a pseudoaneurysm during the EN procedure; therefore, confirming the location of the vessel and presence of a pseudoaneurysm before EN using contrast-enhanced computed tomography (CE-CT) is important for determining Coagrasper use and the safety of EN. Hemostasis using coagulation for gastrointestinal bleeding has been reported; however, a risk of delayed rebleeding or perforation persists due to the burning effect [
7]. No studies have reported rebleeding associated with coagulation for WON cavity bleeding; however, the reported number of hemostasis procedures using coagulation is small, and late adverse events may not be sufficiently evaluated. Therefore, although the space in the WON cavity is small, it is preferable to cauterize the vessel after lifting and separating the surrounding tissues to avoid coagulation-related adverse events.
In summary, the Coagrasper is useful for the coagulation of bleeding from a small artery with a diameter of <3 mm. Because attaining hemostasis for a pseudoaneurysm may be challenging and the use of a Coagrasper requires space for lifting the vessel and separating the surrounding tissues, CE-CT evaluation before EN is necessary to assess the situation around the bleeding point during EN. Under these conditions, the Coagrasper could be useful whenever an endoscope can be inserted and can grasp the whole vessel.
4. Conclusions
Hemostasis using the Coagrasper is useful for arterial bleeding during EN. However, because the Coagrasper worsens bleeding due to its use in incompatible cases (vessels), it should only be employed after its characteristics and the cases in which its use is compatible have been understood. To evaluate the utility and limitations of the Coagrasper in detail, more studies are needed in the future.
Author Contributions
Conceptualization, Y.I. (Yuki Ito), M.K., and M.O.; methodology, Y.I. (Yuki Ito), M.K., K.I., and M.O.; software, Y.I. (Yuki Ito), and M.O.; validation, Y.I. (Yuki Ito), and M.O.; formal analysis, Y.I. (Yuki Ito), M.K., and M.O.; investigation, Y.I. (Yuki Ito), M.K., and M.O.; resources, Y.I. (Yuki Ito), M.K., and M.O.; data curation, Y.I. (Yuki Ito), M.K., and M.O.; writing—original draft preparation, Y.I. (Yuki Ito), and M.O.; writing—review and editing, K.I., and Y.I. (Yuhei Iwasa); visualization, Y.I. (Yuki Ito), M.K., and M.O.; supervision, A.S., Y.N., and E.T.; project administration, E.T.; funding acquisition, A.S., Y.N., and E.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of Gifu Municipal Hospital (protocol code 523).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. Written informed consent has been obtained from the patient to publish this paper.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Siddiqui, A.A.; Kowalski, T.E.; Loren, D.E.; Khalid, A.; Soomro, A.; Mazhar, S.M.; Isby, L.; Kahaleh, M.; Karia, K.; Yoo, J.; et al. Fully covered self-expanding metal stents versus lumen-apposing fully covered self-expanding metal stent versus plastic stents for endoscopic drainage of pancreatic walled-off necrosis: Clinical outcomes and success. Gastrointest. Endosc. 2017, 85, 758–765. [Google Scholar] [CrossRef] [PubMed]
- van Brunschot, S.; Fockens, P.; Bakker, O.J.; Besselink, M.G.; Voermans, R.P.; Poley, J.W.; Gooszen, H.G.; Bruno, M.; van Santvoort, H.C. Endoscopic transluminal necrosectomy in necrotising pancreatitis: A systematic review. Surg. Endosc. 2014, 28, 1425–1438. [Google Scholar] [CrossRef] [PubMed]
- DeSimone, M.L.; Asombang, A.W.; Berzin, T.M. Lumen apposing metal stents for pancreatic fluid collections: Recognition and management of complications. World J. Gastrointest. Endosc. 2017, 9, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Holmes, I.; Shinn, B.; Mitsuhashi, S.; Boortalary, T.; Bashir, M.; Kowalski, T.; Loren, D.; Kumar, A.; Schlachterman, A.; Chiang, A. Prediction and management of bleeding during endoscopic necrosectomy for pancreatic walled-off necrosis: Results of a large retrospective cohort at a tertiary referral center. Gastrointest. Endosc. 2022, 95, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Luigiano, C.; Pellicano, R.; Fusaroli, P.; Iabichino, G.; Arena, M.; Lisotti, A.; Consolo, P.; Morace, C.; Opocher, E.; Caletti, G.; et al. Pancreatic necrosectomy: An evidence-based systematic review of the levels of evidence and a comparison of endoscopic versus non-endoscopic techniques. Minerva Chir. 2016, 71, 262–269. [Google Scholar] [PubMed]
- Tanaka, S.; Toyonaga, T.; Morita, Y.; Ishida, T.; Hoshi, N.; Grimes, K.L.; Ohara, Y.; Yoshizaki, T.; Kawara, F.; Umegaki, E.; et al. Efficacy of a new hemostatic forceps during gastric endoscopic submucosal dissection: A prospective randomized controlled trial. J. Gastroenterol. Hepatol. 2017, 32, 846–851. [Google Scholar] [CrossRef] [PubMed]
- Coumaros, D.; Tsesmeli, N. Active gastrointestinal bleeding: Use of hemostatic forceps beyond endoscopic submucosal dissection. World J. Gastroenterol. 2010, 16, 2061–2064. [Google Scholar] [CrossRef] [PubMed]
- Andueep, K.V.; Katukuri, G.R.; Lakhtakia, S.; Ramchandani, M.; Reddy, D.N. Hemostasis During Endoscopic Necrosectomy: Spray Coagulation Can be a Savior! Am. J. Gastroenterol. 2022, 117, 375–376. [Google Scholar]
- Matsumi, A.; Matsumoto, K.; Okada, H. Hemostasis with hemostatic forceps for bleeding during endoscopic necrosectomy. Dig. Endosc. 2018, 30, 810–811. [Google Scholar] [CrossRef] [PubMed]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).