Ivabradine for the Prevention of Anthracycline-Induced Cardiotoxicity in Female Patients with Primarily Breast Cancer: A Prospective, Randomized, Open-Label Clinical Trial
Abstract
:1. Introduction
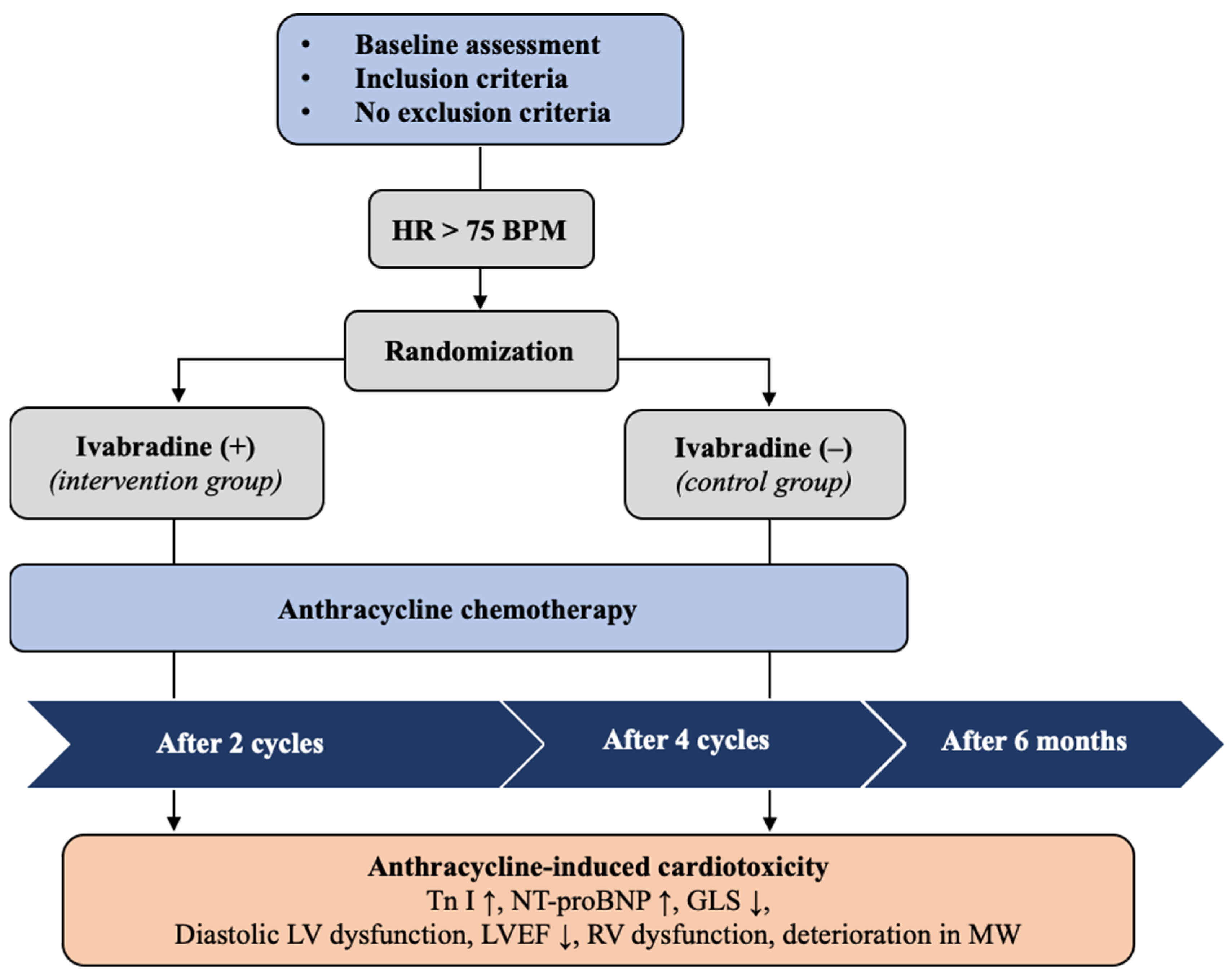
2. Materials and Methods
2.1. Study Design
2.2. Study Participants
2.3. Randomization, Allocation, and Intervention
2.4. Study Procedures
2.5. Study Endpoints
- Incidence of myocardial damage as measured by elevated high-sensitivity cardiac Tn I.
- Incidence of myocardial injury according to elevated NT-proBNP levels.
- Incidence of LV diastolic dysfunction.
- Incidence of LV systolic dysfunction measured by EF and symptomatic HF.
- Incidence of RV dysfunction.
- Changes in myocardial work parameters by the 2D strain–pressure loop.
- Incidence of adverse effects of ivabradine.
2.6. Statistical Analysis
3. Results
3.1. Cardiovascular Toxicity Risk Stratification before Anticancer Therapy
3.2. Ivabradine Dosage and Efficacy
3.3. Primary Endpoint
3.4. Secondary Endpoints
3.4.1. Changes in the Levels of Tn I
3.4.2. Changes in NT-proBNP Levels
3.4.3. LV Diastolic Dysfunction
3.4.4. LV Systolic Dysfunction and Symptomatic HF
3.4.5. RV Dysfunction
3.4.6. Myocardial Work Indices
3.4.7. Mild Asymptomatic Cancer-Therapy-Related Cardiac Dysfunction
3.4.8. Incidence of Adverse Effects of Ivabradine
4. Discussion
4.1. Reduction in the GLS during Cardiotoxic Chemotherapy
4.2. Increase in Tn I Levels
4.3. Increase in Natriuretic Peptide Levels
4.4. Development of Diastolic Dysfunction
4.5. Deterioration of LV Systolic Function
4.6. Alterations in Myocardial Performance
5. Study Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Henriksen, P. Anthracycline cardiotoxicity: An update on mechanisms, monitoring and prevention. Heart 2017, 104, 971–977. [Google Scholar] [CrossRef]
- Mele, D.; Tocchetti, C.G.; Pagliaro, P.; Madonna, R.; Novo, G.; Pepe, A.; Zito, C.; Maurea, N.; Spallarossa, P. Pathophysiology of anthracycline cardiotoxicity. J. Cardiovasc. Med. 2016, 17, e3–e11. [Google Scholar] [CrossRef]
- Kalam, K.; Marwick, T.H. Role of cardioprotective therapy for prevention of cardiotoxicity with chemotherapy: A systematic review and meta-analysis. Eur. J. Cancer 2013, 49, 2900–2909. [Google Scholar] [CrossRef]
- Cardinale, D.; Colombo, A.; Bacchiani, G.; Tedeschi, I.; Meroni, C.A.; Veglia, F.; Civelli, M.; Lamantia, G.; Colombo, N.; Curigliano, G.; et al. Early Detection of Anthracycline Cardiotoxicity and Improvement with Heart Failure Therapy. Circulation 2015, 131, 1981–1988. [Google Scholar] [CrossRef] [PubMed]
- Cardinale, D.; Ciceri, F.; Latini, R.; Franzosi, M.G.; Sandri, M.T.; Civelli, M.; Cucchi, G.; Menatti, E.; Mangiavacchi, M.; Cavina, R.; et al. Anthracycline-induced cardiotoxicity: A multicenter randomised trial comparing two strategies for guiding prevention with enalapril: The International CardioOncology Society-one trial. Eur. J. Cancer 2018, 94, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Beheshti, A.T.; Toroghi, H.M.; Hosseini, G.; Zarifian, A.; Shandiz, F.H.; Fazlinezhad, A. Carvedilol Administration Can Prevent Doxorubicin-Induced Cardiotoxicity: A Double-Blind Randomized Trial. Cardiology 2016, 134, 47–53. [Google Scholar] [CrossRef]
- Akpek, M.; Ozdogru, I.; Sahin, O.; Inanc, M.; Dogan, A.; Yazici, C.; Berk, V.; Karaca, H.; Kalay, N.; Oguzhan, A.; et al. Protective effects of spironolactone against anthracycline-induced cardiomyopathy. Eur. J. Heart Fail. 2015, 17, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Georgakopoulos, P.; Roussou, P.; Matsakas, E.; Karavidas, A.; Anagnostopoulos, N.; Marinakis, T.; Galanopoulos, A.; Georgiakodis, F.; Zimeras, S.; Kyriakidis, M.; et al. Cardioprotective effect of metoprolol and enalapril in doxorubicin-treated lymphoma patients: A prospective, parallel-group, randomized, controlled study with 36-month follow-up. Am. J. Hematol. 2010, 85, 894–896. [Google Scholar] [CrossRef]
- Anker, M.S.; Ebner, N.; Hildebrandt, B.; Springer, J.; Sinn, M.; Riess, H.; Anker, S.D.; Landmesser, U.; Haverkamp, W.; von Haehling, S. Resting heart rate is an independent predictor of death in patients with colorectal, pancreatic, and non-small cell lung cancer: Results of a prospective cardiovascular long-term study. Eur. J. Heart Fail. 2016, 18, 1524–1534. [Google Scholar] [CrossRef]
- Müller-Werdan, U.; Stöckl, G.; Werdan, K. Advances in the management of heart failure: The role of ivabradine. Vasc. Health Risk Manag. 2016, 12, 453–470. [Google Scholar] [CrossRef]
- Böhm, M. Heart rate: From heart failure to chronic diseases and cancer. Is there a role for supportive care by heart rate reduction? Eur. J. Heart Fail. 2017, 19, 250–252. [Google Scholar] [CrossRef] [PubMed]
- Dallapellegrina, L.; Sciatti, E.; Vizzardi, E. Ivabradine and endothelium: An update. Ther. Adv. Cardiovasc. Dis. 2020, 14, 1753944720934937. [Google Scholar] [CrossRef] [PubMed]
- El-Naggar, A.E.M.; El-Gowilly, S.M.; Sharabi, F.M. Possible Ameliorative Effect of Ivabradine on the Autonomic and Left Ventricular Dysfunction Induced by Doxorubicin in Male Rats. J. Cardiovasc. Pharmacol. 2018, 72, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.N.; Mousa, A.M. Effects of Ivabradine on Cardiotoxicity Induced by Doxorubicin Treatment in Rats. 2013. Available online: www.medicaljournalofcairouniversity.net (accessed on 18 September 2018).
- Vasyuk, Y.V.; Nesvetov, V.V.; Shkolnik, E.L.; Shkolnik, L.D.; Varlan, G.V.; Gendlin, G.E.; Emelina, E.I. Possibilities of ivabradine, a selective inhibitor of ion f-channels of sinus node, in prevention of anthracycline cardiotoxicity in patients with breast cancer. Ration. Pharmacother. Cardiol. 2017, 13, 184–190. [Google Scholar] [CrossRef]
- Lyon, A.R.; Dent, S.; Stanway, S.; Earl, H.; Brezden-Masley, C.; Cohen-Solal, A.; Tocchetti, C.G.; Moslehi, J.J.; Groarke, J.D.; Bergler-Klein, J.; et al. Baseline cardiovascular risk assessment in cancer patients scheduled to receive cardiotoxic cancer therapies: A position statement and new risk assessment tools from the Cardio-Oncology Study Group of the Heart Failure Association of the European Society of Cardiology in collaboration with the International Cardio-Oncology Society. Eur. J. Heart Fail. 2020, 22, 1945–1960. [Google Scholar] [CrossRef] [PubMed]
- Čelutkienė, J.; Pudil, R.; López-Fernández, T.; Grapsa, J.; Nihoyannopoulos, P.; Bergler-Klein, J.; Cohen-Solal, A.; Farmakis, D.; Tocchetti, C.G.; von Haehling, S.; et al. Role of cardiovascular imaging in cancer patients receiving cardiotoxic therapies: A position statement on behalf of the H eart F ailure A ssociation (HFA), the E uropean A ssociation of C ardiovascular I maging (EACVI) and the Cardio-Oncology Council of the European Society of Cardiology (ESC). Eur. J. Heart Fail. 2020, 22, 1504–1524. [Google Scholar] [CrossRef] [PubMed]
- Pudil, R.; Mueller, C.; Čelutkienė, J.; Henriksen, P.A.; Lenihan, D.; Dent, S.; Barac, A.; Stanway, S.; Moslehi, J.; Suter, T.M.; et al. Role of serum biomarkers in cancer patients receiving cardiotoxic cancer therapies: A position statement from the Cardio-Oncology Study Group of the Heart Failure Association and the Cardio-Oncology Council of the European Society of Cardiology. Eur. J. Heart Fail. 2020, 22, 1966–1983. [Google Scholar] [CrossRef]
- Nagueh, S.F.; Smiseth, O.A.; Appleton, C.P.; Byrd, B.F.; Dokainish, H.; Edvardsen, T.; Flachskampf, F.A.; Gillebert, T.C.; Klein, A.L.; Lancellotti, P.; et al. Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 1321–1360. [Google Scholar] [CrossRef]
- Ferreira, V.V.; Mano, T.B.; Cardoso, I.; Cruz, M.C.; Branco, L.M.; Almeida-Morais, L.; Timóteo, A.; Galrinho, A.; Castelo, A.; Brás, P.G.; et al. Myocardial Work Brings New Insights into Left Ventricular Remodelling in Cardio-Oncology Patients. Int. J. Environ. Res. Public Health 2022, 19, 2826. [Google Scholar] [CrossRef]
- Lyon, A.R.; López-Fernández, T.; Couch, L.S.; Asteggiano, R.; Aznar, M.C.; Bergler-Klein, J.; Boriani, G.; Cardinale, D.; Cordoba, R.; Cosyns, B.; et al. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur. Heart J. Cardiovasc. Imaging 2022, 23, e333–e465. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk: The Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS). Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef]
- Thavendiranathan, P.; Negishi, T.; Somerset, E.; Negishi, K.; Penicka, M.; Lemieux, J.; Aakhus, S.; Miyazaki, S.; Shirazi, M.; Galderisi, M.; et al. Strain-Guided Management of Potentially Cardiotoxic Cancer Therapy. J. Am. Coll. Cardiol. 2020, 77, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Avila, M.S.; Ayub-Ferreira, S.M.; Wanderley, M.R.d.B.; Cruz, F.d.D.; Brandão, S.M.G.; Rigaud, V.O.C.; Higuchi-Dos-Santos, M.H.; Hajjar, L.A.; Filho, R.K.; Hoff, P.M.; et al. Carvedilol for Prevention of Chemotherapy-Related Cardiotoxicity: The CECCY Trial. J. Am. Coll. Cardiol. 2018, 71, 2281–2290. [Google Scholar] [CrossRef] [PubMed]
- Chow, S.C.; Shao, J.; Wang, H.; Lokhnygina, Y. Sample Size Calculations in Clinical Research, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the. European Society of Cardiology (ESC) With the special contributio. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Verrier, R.L. Ivabradine’s Pleiotropic Profile: Can it protect against doxorubicin-induced heart failure? J. Cardiovasc. Pharmacol. 2018, 72, 19–21. [Google Scholar] [CrossRef] [PubMed]
- Al-Kuraishy, H.M.; Issa, H.K.; Al-Gareeb, A.I.; El-Bouseary, M.M.; Youssef, A.; Abdelaziz, A.S.; Khalifa, H.A.; Batiha, G.E.-S. The role of ivabradine in doxorubicin-induced cardiotoxicity: Exploring of underlying argument. Inflammopharmacology 2022, 30, 2441–2446. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Ando, H.; Suzuki, W.; Ohashi, H.; Suzuki, Y.; Takashima, H.; Amano, T. Impact of ivabradine in decompensated heart failure due to cancer therapy-related cardiac dysfunction. Clin. Case Rep. 2021, 9, e04133. [Google Scholar] [CrossRef] [PubMed]
- Klotzka, A.; Iwańczyk, S.; Ropacka-Lesiak, M.; Misan, N.; Lesiak, M. Anthracycline-induced microcirculation disorders: AIM PILOT Study. Kardiologia Polska 2023, 81, 766–768. [Google Scholar] [CrossRef]
- Bosch, X.; Rovira, M.; Sitges, M.; Domènech, A.; Ortiz-Pérez, J.T.; de Caralt, T.M.; Morales-Ruiz, M.; Perea, R.J.; Monzó, M.; Esteve, J. Enalapril and Carvedilol for Preventing Chemotherapy-Induced Left Ventricular Systolic Dysfunction in Patients With Malignant Hemopathies: The OVERCOME trial (prevention of left ventricular dysfunction with enalapril and caRvedilol in patients submitted to intensive ChemOtherapy for the treatment of malignant hEmopathies). J. Am. Coll. Cardiol. 2013, 61, 2355–2362. [Google Scholar] [CrossRef]
- Gulati, G.; Heck, S.L.; Røsjø, H.; Ree, A.H.; Hoffmann, P.; Hagve, T.; Norseth, J.; Gravdehaug, B.; Steine, K.; Geisler, J.; et al. Neurohormonal Blockade and Circulating Cardiovascular Biomarkers During Anthracycline Therapy in Breast Cancer Patients: Results From the PRADA (Prevention of Cardiac Dysfunction During Adjuvant Breast Cancer Therapy) Study. J. Am. Heart Assoc. 2017, 6, e006513. [Google Scholar] [CrossRef]
- Evangelos K. Oikonomou, M. Assessment of Prognostic Value of Left Ventricular Global Longitudinal Strain for Early Prediction of Chemotherapy-Induced Cardiotoxicity a Systematic Review and Meta-analysis. JAMA Cardiol. 2019, 4, 1007–1018. [Google Scholar] [CrossRef] [PubMed]
- Gripp, E.D.A.; De Oliveira, G.E.; Feijó, L.A.; Garcia, M.I.; Xavier, S.S.; De Sousa, A.S. Global Longitudinal Strain Accuracy for Cardiotoxicity Prediction in a Cohort of Breast Cancer Patients During Anthracycline and/or Trastuzumab Treatment. Arq. Bras. Cardiol. 2018, 110, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Burdick, J.; Berridge, B.; Coatney, R. Strain echocardiography combined with pharmacological stress test for early detection of anthracycline induced cardiomyopathy. J. Pharmacol. Toxicol. Methods 2015, 73, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Charbonnel, C.; Convers-Domart, R.; Rigaudeau, S.; Taksin, A.L.; Baron, N.; Lambert, J.; Ghez, S.; Georges, J.-L.; Farhat, H.; Lambert, J.; et al. Assessment of global longitudinal strain at low-dose anthracycline-based chemotherapy, for the prediction of subsequent cardiotoxicity. Eur. Heart J. Cardiovasc. Imaging 2017, 18, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Santoro, C.; Arpino, G.; Esposito, R.; Lembo, M.; Paciolla, I.; Cardalesi, C.; de Simone, G.; Trimarco, B.; De Placido, S.; Galderisi, M. 2D and 3D strain for detection of subclinical anthracycline cardiotoxicity in breast cancer patients: A balance with feasibility. Eur. Heart J. Cardiovasc. Imaging 2017, 18, 930–936. [Google Scholar] [CrossRef]
- Thavendiranathan, P.; Poulin, F.; Lim, K.-D.; Plana, J.C.; Woo, A.; Marwick, T.H. Use of Myocardial Strain Imaging by Echocardiography for the Early Detection of Cardiotoxicity in Patients During and After Cancer Chemotherapy: A Systematic Review. J. Am. Coll. Cardiol. 2014, 63, 2751–2768. [Google Scholar] [CrossRef] [PubMed]
- Negishi, T.; Thavendiranathan, P.; Penicka, M.; Lemieux, J.; Aakhus, S.; Miyazaki, S.; Shirazi, M.; Galderisi, M.; Cho, G.Y.; Popescu, B.A.; et al. Cardioprotection Using Strain-Guided Management of Potentially Cardiotoxic Cancer Therapy: 1 Year Results of the SUCCOUR Trial. Available online: https://academic.oup.com/eurheartj/article/41/Supplement_2/ehaa946.3282/6004273 (accessed on 17 March 2023).
- López-Sendón, J.; Álvarez-Ortega, C.; Zamora Auñon, P.; Buño Soto, A.; Lyon, A.R.; Farmakis, D.; Cardinale, D.; Canales Albendea, M.; Feliu Batlle, J.; Rodríguez Rodríguez, I.; et al. Classification, prevalence, and outcomes of anticancer therapy-induced cardiotoxicity: The CARDIOTOX registry. Eur. Heart J. 2020, 41, 1720–1729. [Google Scholar] [CrossRef]
- Gulati, G.; Heck, S.L.; Ree, A.H.; Hoffmann, P.; Schulz-Menger, J.; Fagerland, M.W.; Gravdehaug, B.; von Knobelsdorff-Brenkenhoff, F.; Bratland, Å.; Storås, T.H.; et al. Prevention of cardiac dysfunction during adjuvant breast cancer therapy (PRADA): A 2 3 2 factorial, randomized, placebo-controlled, double-blind clinical trial of candesartan and metoprolol In patients treated for early breast cancer with adjuvant anthracycline-containing regimens with or without trastuzumab and radiation, concomitant treatment with candesartan provides protection against early decline in global left ventricu-lar function. Eur. Heart J. 2016, 37, 1671–1680. [Google Scholar] [CrossRef]
- Tzolos, E.; Adamson, P.; Hall, P.; Macpherson, I.; Oikonomidou, O.; MacLean, M.; Lewis, S.; McVicars, H.; Newby, D.; Mills, N.; et al. Dynamic Changes in High-Sensitivity Cardiac Troponin I in Response to Anthracycline-Based Chemotherapy. Clin. Oncol. 2020, 32, 292–297. [Google Scholar] [CrossRef]
- Kitayama, H.; Kondo, T.; Sugiyama, J.; Kurimoto, K.; Nishino, Y.; Kawada, M.; Hirayama, M.; Tsuji, Y. High-sensitive troponin T assay can predict anthracycline- and trastuzumab-induced cardiotoxicity in breast cancer patients. Breast Cancer 2017, 24, 774–782. [Google Scholar] [CrossRef]
- Upshaw, J.N.; Finkelman, B.; Hubbard, R.A.; Smith, A.M.; Narayan, H.K.; Arndt, L.; Domchek, S.; DeMichele, A.; Fox, K.; Shah, P.; et al. Comprehensive Assessment of Changes in Left Ventricular Diastolic Function with Contemporary Breast Cancer Therapy. JACC Cardiovasc. Imaging 2020, 13, 198–210. [Google Scholar] [CrossRef]
- Zhang, K.W.; Finkelman, B.S.; Gulati, G.; Narayan, H.K.; Upshaw, J.; Narayan, V.; Plappert, T.; Englefield, V.; Smith, A.M.; Zhang, C.; et al. Abnormalities in 3-Dimensional Left Ventricular Mechanics with Anthracycline Chemotherapy Are Associated with Systolic and Diastolic Dysfunction. JACC Cardiovasc. Imaging 2018, 11, 1059–1068. [Google Scholar] [CrossRef]
- Čiburienė, E.; Aidietienė, S.; Ščerbickaitė, G.; Brasiūnienė, B.; Drobnienė, M.; Baltruškevičienė, E.; Žvirblis, T.; Čelutkienė, J. Development of a Cardio-Oncology Service in Lithuania: Prediction, Prevention, Monitoring and Treatment of Cancer Treatment-Induced Cardiotoxicity. J. Cardiovasc. Dev. Dis. 2022, 9, 134. [Google Scholar] [CrossRef] [PubMed]
- Caspani, F.; Tralongo, A.C.; Campiotti, L.; Asteggiano, R.; Guasti, L.; Squizzato, A. Prevention of anthracycline-induced cardiotoxicity: A systematic review and meta-analysis. Intern. Emerg. Med. 2020, 16, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Cardinale, D.; Colombo, A.; Sandri, M.T.; Lamantia, G.; Colombo, N.; Civelli, M.; Martinelli, G.; Veglia, F.; Fiorentini, C.; Cipolla, C.M.; et al. Prevention of High-Dose Chemotherapy–Induced Cardiotoxicity in High-Risk Patients by Angiotensin-Converting Enzyme Inhibition. Circulation 2006, 114, 2474–2481. [Google Scholar] [CrossRef] [PubMed]
- Argulian, E.; Narula, J. Myocardial Work in Cardio-Oncology. JACC Cardiovasc. Imaging 2022, 15, 1377–1379. [Google Scholar] [CrossRef]
- Di Lisi, D.; Manno, G.; Novo, G. Subclinical Cardiotoxicity: The Emerging Role of Myocardial Work and Other Imaging Techniques. Curr. Probl. Cardiol. 2021, 46, 100818. [Google Scholar] [CrossRef]








| Baseline Characteristics | Ivabradine n = 21 (%) | Controls n = 27 (%) | p-Value |
|---|---|---|---|
| Age, years (mean ± SD, range) | 47.8 ± 9.9, 34–66.5 | 48 ± 10.2, 34–66.5 | 0.935 |
| Cancer type, n (%) | 0.449 | ||
| Right breast | 10 (47.6) | 10 (37) | |
| Left breast | 11 (52.4) | 14 (51.9) | |
| Both breasts | 0 | 1 (3.7) | |
| Sarcoma | 0 | 2 (7.4) | |
| Cancer stage, n (%) | 0.509 | ||
| I | 2 (9.5) | 6 (22.2) | |
| II | 14 (66.7) | 15 (55.6) | |
| III | 5 (23.8) | 5 (18.5) | |
| IV | 0 | 1 (3.7) | |
| Cancer grade, n (%) | 0.309 | ||
| 1 | 0 | 0 | |
| 2 | 13 (65) | 13 (50) | |
| 3 | 7 (35) | 13 (50) | |
| HER2, n (%) | 0.333 | ||
| Positive | 7 (35) | 6 (22.2) | |
| Negative | 13 (65) | 21 (77.8) | |
| BRCA, n (%) | 0.928 | ||
| Positive | 3 (15.8) | 4 (14.8) | |
| Negative | 16 (84.2) | 23 (85.2) | |
| Chemotherapy, n (%) | 0.060 | ||
| Adjuvant | 10 (47.6) | 20 (74.1) | |
| Neoadjuvant | 11 (52.4) | 7 (25.9) | |
| Anthracyclines, n (%) | 0.545 | ||
| Doxorubicin | 15 (71.4) | 21 (77.8) | |
| Epirubicin | 6 (28.6) | 6 (22.2) | |
| Cumulative anthracycline dose | 0.425 | ||
| Doxorubicin, mg/m2 (mean ± SD) | 236 ± 70 | 246 ± 69 | |
| Epirubicin, mg/m2 (mean ± SD) | 360 ± 70 | 360 ± 70 | |
| CV risk factors, n (%) | |||
| Hypertension | 3 (15) | 2 (7.4) | 0.404 |
| Diabetes | 0 | 0 | |
| Dyslipidaemia | 13 (65) | 21 (77.8) | 0.333 |
| Smoking | 1 (5) | 6 (22.2) | 0.101 |
| Obesity (BMI > 30 kg/m2) | 4 (19) | 2 (7.4) | 0.226 |
| Kidney dysfunction (GFR < 60 mL/min./1.73 m2) | 0 | 0 | |
| Cardiotoxicity risk group, n (%) | 0.519 | ||
| Low | 17 (81) | 23 (85.2) | |
| Medium | 4 (19) | 3 (11.1) | |
| High | 0 | 1 (3.7) | |
| Very high | 0 | 0 | |
| HF stage, n (%) | 0.356 | ||
| A | 4 (19) | 4 (14.8) | |
| B | 4 (19) | 8 (29.6) | |
| C | 0 | 1 (3.7) | |
| D | 0 | 0 | |
| Anaemia (Hb < 117 g/L), n (%) | 12 (60) | 15 (55.6) | 0.761 |
| CRP > 5 mg/L, n (%) | 2 (9.5) | 5 (18.5) | 0.381 |
| Vitamin D < 75 nmol/L, n (%) | 12 (70.6) | 19 (86.4) | 0.226 |
| Myocardial damage markers at baseline, n (%) | |||
| Tn I > 16 ng/L | 0 | 0 | |
| NT-proBNP > 125 ng/L | 3 (14.3) | 9 (33.3) | 0.131 |
| GLS > −18% | 0 | 1 (3.7) | 0.373 |
| LVEF < 55% | 0 | 0 | |
| Diastolic LV dysfunction | 1 (4.8) | 2 (7.4) | 0.707 |
| Echocardiographic parameters at baseline, n (%) | |||
| LAVI > 34 mL/m2 | 5 (23.8) | 5 (18.5) | 0.654 |
| Transmitral E velocity > 50 cm/s | 7 (33.3) | 10 (37) | 0.790 |
| Transmitral A velocity < 60 to < 120 cm/s | 12 (57.1) | 11 (40.7) | 0.259 |
| Transmitral E/A ratio < 0.8 to > 2.0 | 7 (33.3) | 6 (22.2) | 0.390 |
| Mitral E/e’ ratio > 14 | 0 | 0 | |
| e’ med. < 7 cm/s | 1 (4.8) | 4 (14.8) | 0.258 |
| e’ lat. < 10 cm/s | 2 (9.5) | 2 (7.4) | 0.792 |
| IVRT < 70 to > 100 msec | 12 (57.1) | 8 (29.6) | 0.037 |
| Transmitral E velocity DT < 160 to > 220 msec | 11 (52.4) | 9 (33.3) | 0.184 |
| RV S’ < 9.5 cm/s | 0 | 0 | |
| TAPSE < 17 cm | 0 | 0 | |
| 2D LVEF < 55% | 0 | 0 | |
| GWI < 1310 mmHg% | 0 | 0 | |
| GCW < 1543 mmHg% | 0 | 0 | |
| GWW > 287 mmHg% | 0 | 0 | |
| GWE < 90% | 1 (5.3) | 0 | 0.376 |
| 3D LVEF< 55% * | 0 | 0 |
| Parameter | All (n = 48) | Ivabradine (n = 21) | Controls (n = 27) | p-Value |
|---|---|---|---|---|
| Baseline | ||||
| 2D LVEF (mean ± SD) | 64 ± 4.0 | 63.5 ± 3.4 | 64.4 ± 4.5 | 0.478 |
| 3D LVEF (mean ± SD) | 64.3 ± 4.1 | 62.7 ± 4.0 | 65.5 ± 3.8 | 0.055 |
| RV function | ||||
| TAPSE (mean ± SD) | 22 ± 2.7 | 22.2 ± 3.4 | 21.8 ± 2.2 | 0.661 |
| RV S’ (mean ± SD) | 14.8 ± 1.6 | 14.4 ± 1.3 | 15.07 ± 1.8 | 0.152 |
| Myocardial work indices | ||||
| GWI (median; Q1–Q3; range) | 2055.5 (1895–2288; 1552–2904) | 2090 (1911–2341; 1552–2697) | 2021 (1846–2288; 1571–2904) | 0.820 |
| GCW (mean ± SD) | 2471.3 ± 359.2 | 2445.2 ± 404.4 | 2492.8 ± 325 | 0.674 |
| GWW (median; Q1–Q3; range) | 98 (75–131; 35–263) | 82 (63–144; 35–263) | 105 (77–131; 38–156) | 0.622 |
| GWE (median; Q1–Q3; range) | 95 (95–97; 86–98) | 96 (94–97; 86–98) | 95 (95–97; 93–98) | 0.826 |
| After 2 anthracyclines cycles | ||||
| 2D LVEF (mean ± SD) | 63.3 ± 4.0 | 62.1 ± 3.4 | 64.0 ± 4.3 | 0.129 |
| 3D LVEF (mean ± SD) | 63.6 ± 2.6 | 63.5 ± 2.3 | 63.8 ± 3.0 | 0.751 |
| RV function | ||||
| TAPSE (mean ± SD) | 21.5 ± 2.9 | 22 ± 3.1 | 21.1 ± 2.7 | 0.327 |
| RV S’ (mean ± SD) | 14.1 ± 1.7 | 14.3 ± 1.4 | 14.0 ± 1.8 * | 0.534 |
| Myocardial work indices | ||||
| GWI (median; Q1–Q3; range) | 1911.5 (1671–2229.5; 1043–2587) | 1959 (1766–2253; 1043–2587) | 1826 (1632–2206; 1416–2470) * | 0.412 |
| GCW (mean ± SD) | 2235.3 (308.1) | 2278.5 (300.8) | 2203.4 (316.2) * | 0.453 |
| GWW (median; Q1–Q3; range) | 84 (68.5–108; 27–438) | 81 (48–125; 35–438) | 86 (70–106; 27–226) | 1.000 |
| GWE (median; Q1–Q3; range) | 96 (95–96.5; 84–98) | 96 (95–97; 84–98) | 95 (95–96; 90–98) | 0.348 |
| After 4 anthracyclines cycles | ||||
| 2D LVEF (mean ± SD) | 61.4 ± 5.492 | 62 ± 3.1 * | 61.1 ± 6.7 * | 0.534 |
| 3D LVEF (mean ± SD) | 63.0 ± 3.8 | 63.1 ± 4.3 | 63 ± 3.3 * | 0.964 |
| RV function | ||||
| TAPSE (mean ± SD) | 21.3 ± 2.9 | 22.2 ± 2.5 | 20.7 ± 3.0 | 0.087 |
| RV S’ (mean ± SD) | 14.4 ± 1.3 | 14.4 ± 1.7 | 14.4 ± 1.1 | 0.902 |
| Myocardial work indices | ||||
| GWI (median; Q1–Q3; range) | 1906.5 (1747.5–2111.5; 684–2518) | 1946 (1616–2240; 1468–2518) * | 1890 (1769–2073; 684–2453) * | 0.591 |
| GCW (mean ± SD) | 2205.5 ± 356.1 | 2257.9 ± 388.7 * | 2169.3 ± 334.7 * | 0.423 |
| GWW (median; Q1–Q3; range) | 89 (59–114; 23–150) | 76.5 (61–107; 32–149) | 96 (58–121; 23–150) | 0.424 |
| GWE (median; Q1–Q3; range) | 96 (95–97; 84–98) | 96 (95–97; 94–98) | 95 (94–97; 84–98) | 0.113 |
| At 6 moths follow-up | ||||
| 2D LVEF (mean ± SD) | 61.8 ± 4.4 | 61.2 ± 3.5 * | 62.1 ± 4.9 * | 0.493 |
| 3D LVEF (mean ± SD) | 62.3 ± 4.7 | 61.7 ± 5.1 | 62.8 ± 4.4 * | 0.512 |
| RV function | ||||
| TAPSE (mean ± SD) | 21.3 ± 2.7 | 21.3 (2.3) | 21.3 ± 3 | 0.972 |
| RV S’ (mean ± SD) | 14.5 ± 2.04 | 14.2 (1.7) | 14.6 ± 2.3 | 0.497 |
| Myocardial work indices | ||||
| GWI (median; Q1–Q3; range) | 1839.5 (1637–2084.5; 1055–2677) | 1979.5 (1665–2339; 1396–2677) * | 1795 (1632–2009; 1055–2171) * | 0.044 |
| GCW (mean ± SD) | 2134.1 ± 357.0 | 2290.6 ± 373.1 | 2025.8 ± 307.8 * | 0.014 |
| GWW (median; Q1–Q3; range) | 100.5 (70–142; 29–266) | 104.5 (75–150; 42–260) | 96.5 (65–132; 29–266) | 0.676 |
| GWE (median; Q1–Q3; range) | 95 (92.5–96; 87–98) | 95 (94–96; 90–98) | 95 (92–96; 87–98) | 0.709 |
| Parameters | All (n = 48) | Ivabradine (n = 21) | Controls (n = 27) | p-Value |
|---|---|---|---|---|
| Baseline | ||||
| Tn I (median; Q1–Q3; range) | 1; 0–1.5; 0–5 | 1; 0–1; 0–3 | 1; 0–2; 0–5 | 0.465 |
| NT-proBNP (median; Q1–Q3; range) | 70.8 (59.2–128.3; 21–846.6) | 68.5 (60.3–98; 21–190.5) | 74.8 (58.3–152; 47–846.6) | 0.418 |
| After 2 anthracyclines cycles | ||||
| Tn I (median; Q1–Q3; range) | 3 (2–5; 0–172) | 3 (2–4; 1–9) * | 3 (2–6; 0–172) * | 0.579 |
| NT-proBNP (median; Q1–Q3; range) | 82.1 (55.3–130.6; 26.6–945) | 84 (52.4–138; 32–281.6) | 80.2 (65.7–116.4; 26.6–945) | 0.918 |
| After 4 anthracyclines cycles | ||||
| Tn I (median; Q1–Q3; range) | 12 (7–24; 2–73) | 10 (6–21; 2–73) * | 14 (9–31; 2–70) * | 0.444 |
| NT-proBNP (median; Q1–Q3; range) | 84 (49.3–148; 26.9–16,048.7) | 80 (53.4–153.1; 26.9–233.4) | 84 (44–148; 29.5–16,048.7) | 0.908 |
| At 6 months follow-up | ||||
| Tn I (median; Q1–Q3; range) | 5.5 (3–13; 1–109) | 4 (3–7; 1–42) * | 7.5 (3–20; 2–109) * | 0.124 |
| NT-proBNP (median; Q1–Q3; range) | 84.4 (51.2–150.4; 20–10,032.7) | 71.8 (49.7–131.1; 29.8–504.9) | 86.4 (57.2–163.8; 20–10,032.7) | 0.334 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Čiburienė, E.; Aidietienė, S.; Ščerbickaitė, G.; Sadauskienė, E.; Sudavičienė, D.; Baltruškevičienė, E.; Brasiūnienė, B.; Drobnienė, M.; Čelutkienė, J. Ivabradine for the Prevention of Anthracycline-Induced Cardiotoxicity in Female Patients with Primarily Breast Cancer: A Prospective, Randomized, Open-Label Clinical Trial. Medicina 2023, 59, 2140. https://doi.org/10.3390/medicina59122140
Čiburienė E, Aidietienė S, Ščerbickaitė G, Sadauskienė E, Sudavičienė D, Baltruškevičienė E, Brasiūnienė B, Drobnienė M, Čelutkienė J. Ivabradine for the Prevention of Anthracycline-Induced Cardiotoxicity in Female Patients with Primarily Breast Cancer: A Prospective, Randomized, Open-Label Clinical Trial. Medicina. 2023; 59(12):2140. https://doi.org/10.3390/medicina59122140
Chicago/Turabian StyleČiburienė, Eglė, Sigita Aidietienė, Greta Ščerbickaitė, Eglė Sadauskienė, Diana Sudavičienė, Edita Baltruškevičienė, Birutė Brasiūnienė, Monika Drobnienė, and Jelena Čelutkienė. 2023. "Ivabradine for the Prevention of Anthracycline-Induced Cardiotoxicity in Female Patients with Primarily Breast Cancer: A Prospective, Randomized, Open-Label Clinical Trial" Medicina 59, no. 12: 2140. https://doi.org/10.3390/medicina59122140






