Abdominal Actinomycotic Abscess after Laparoscopic Sleeve Gastrectomy for Morbid Obesity: A Case Report
Abstract
:1. Introduction
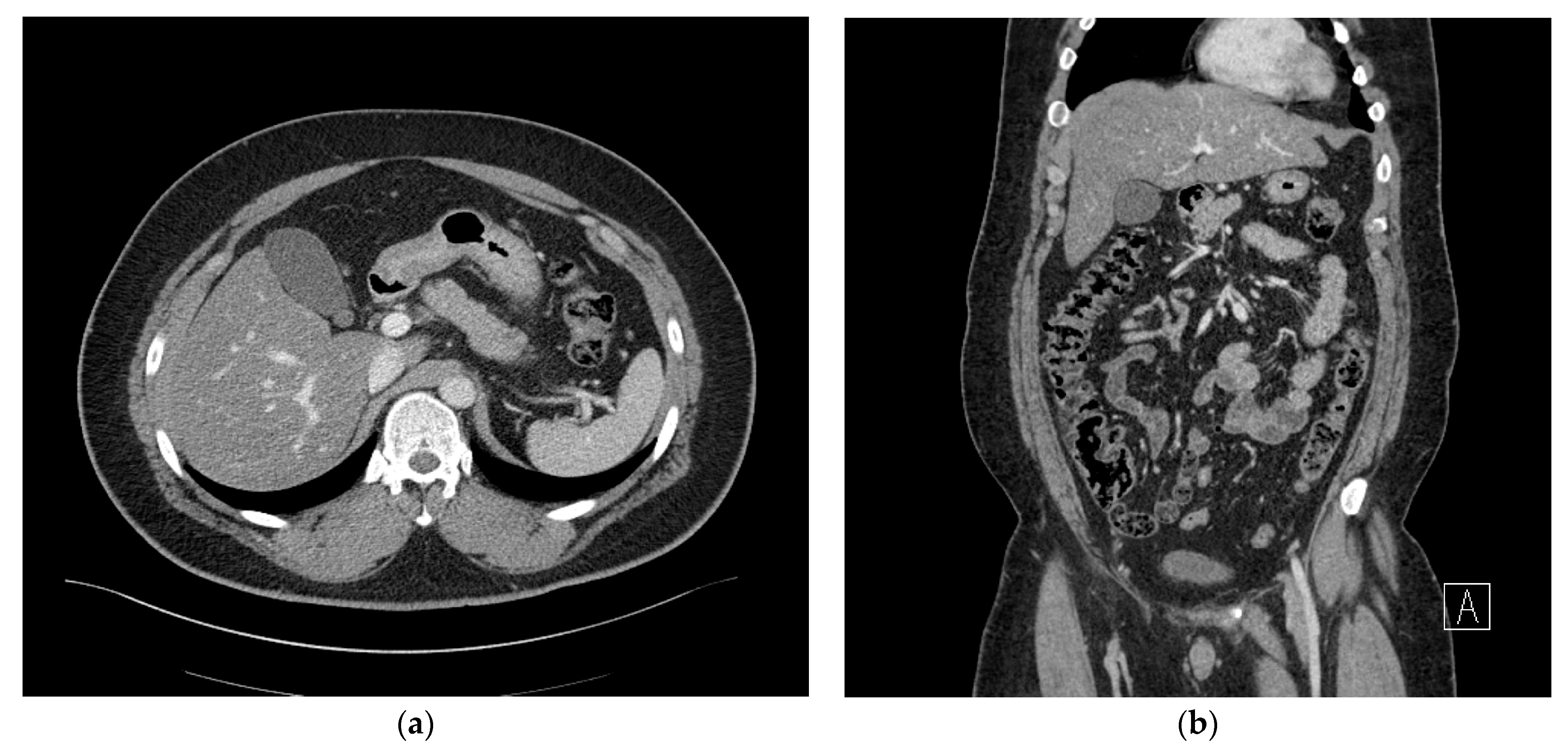
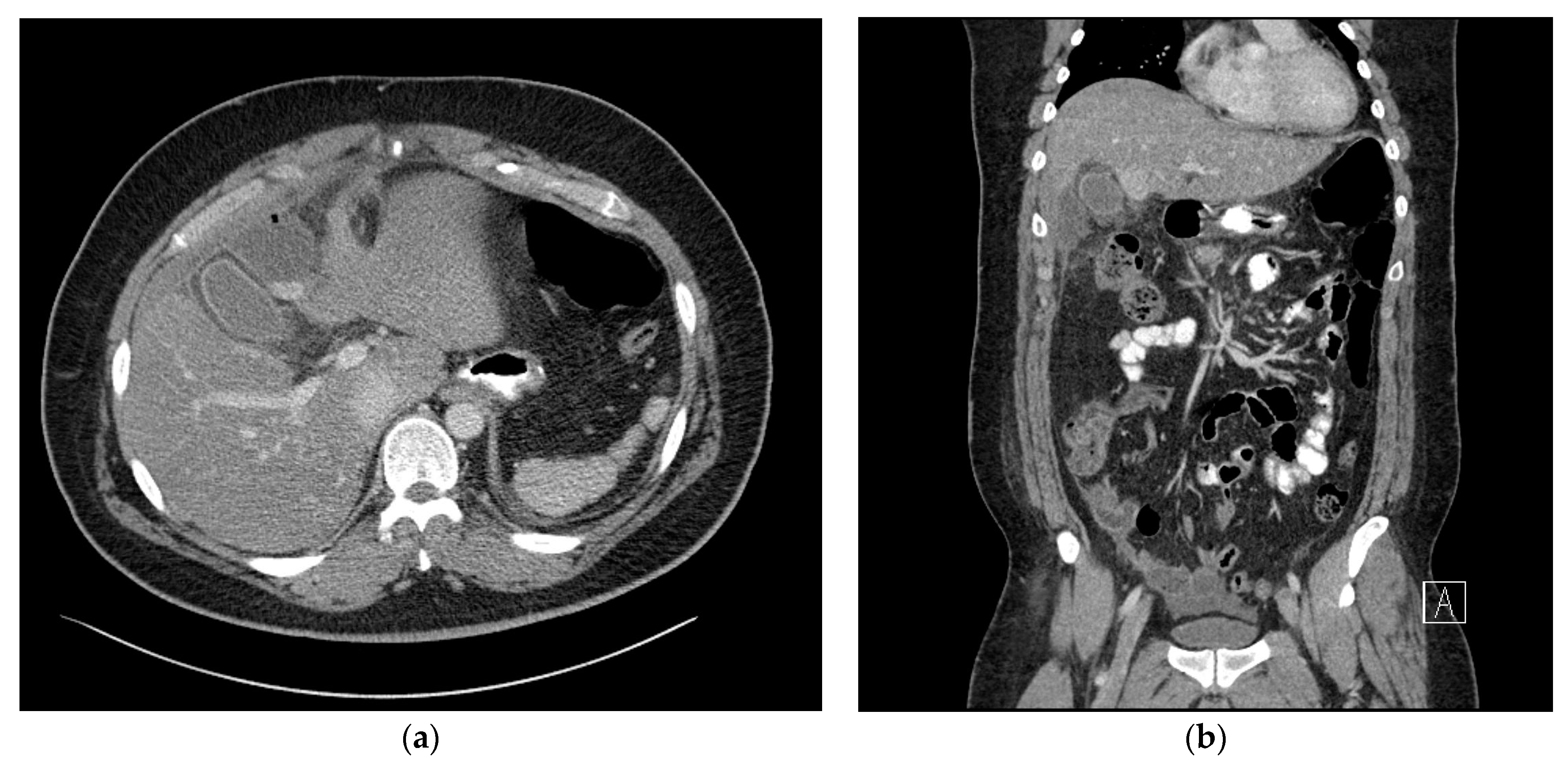
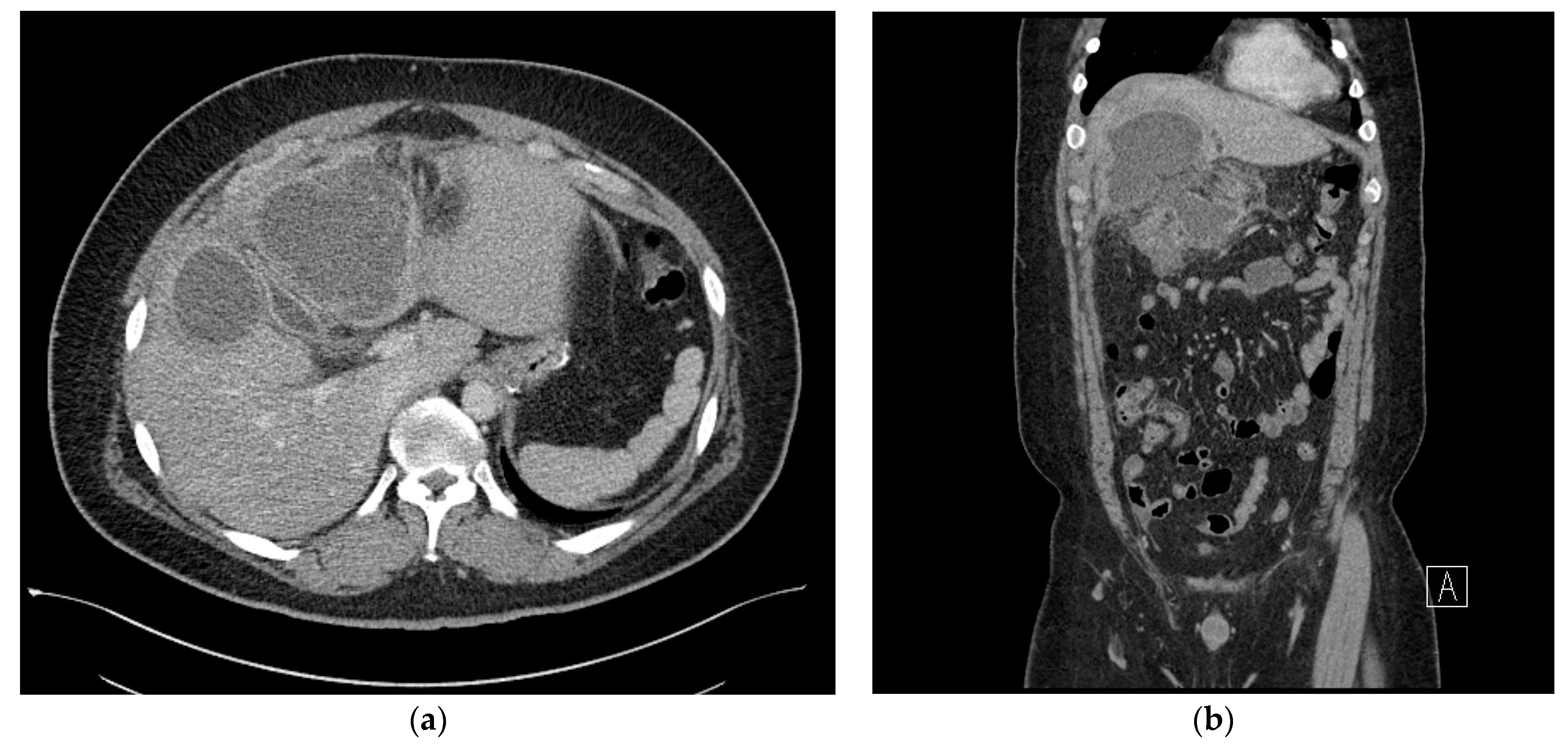
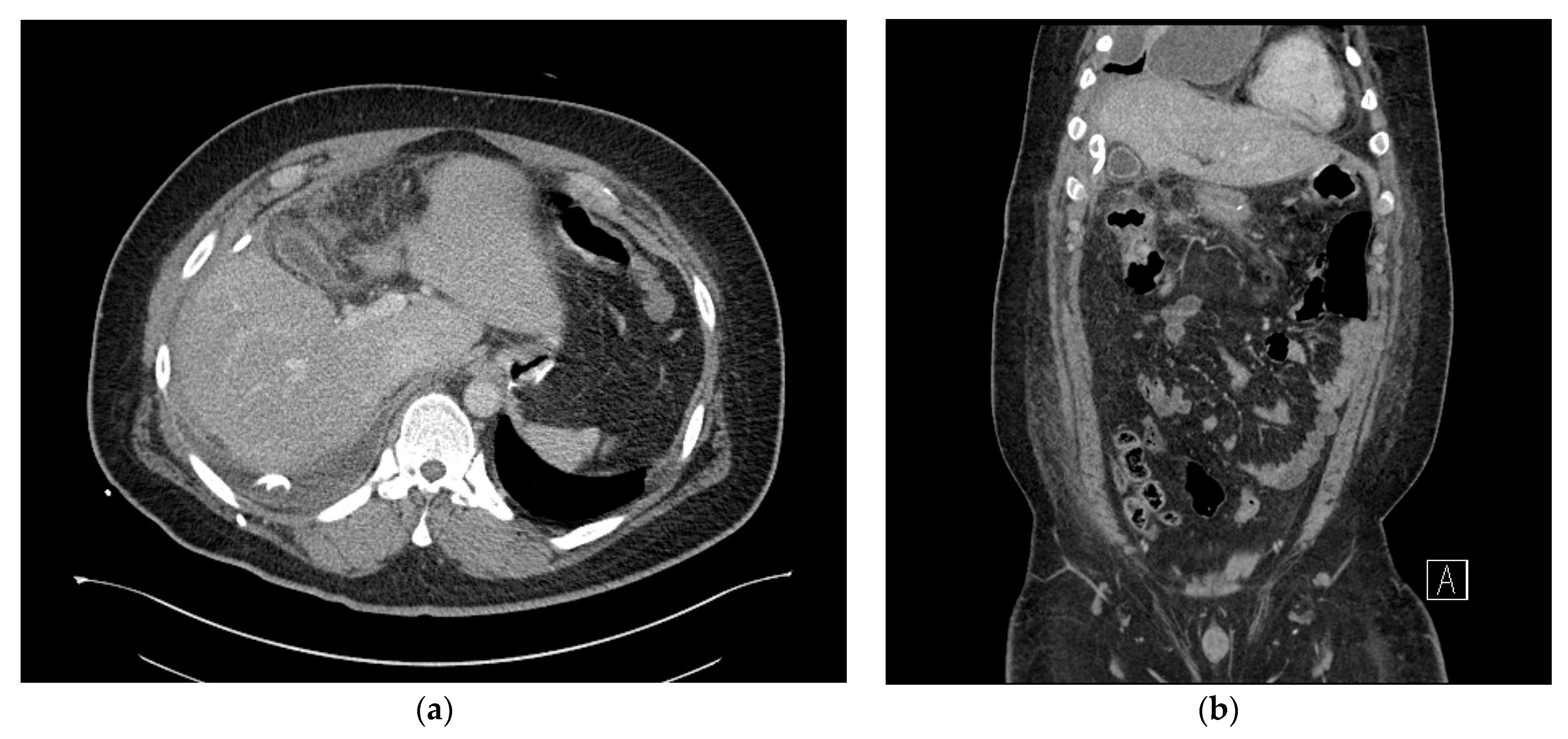
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Batty, I. Actinomyces odontolyticus, a new species of actinomycete regularly isolated from deep carious dentine. J. Pathol. Bacteriol. 1958, 75, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Stringer, M.; Br, J. Abdominal actinomycosis: A forgotten disease? Br. J. Hosp. Med. 1987, 38, 125–127. [Google Scholar] [PubMed]
- Putman, H.C.; Dockerty, M.B.; Waugh, J.M. Abdominal actinomycosis: An analysis of 122 cases. Surgery 1950, 28, 781–800. [Google Scholar]
- Fernández-Aceñero, M.; Silvestre, V.; Fernández-Roldán, R.; Cortés, L.; García-Blanch, G. Gastric actinomycosis: A rare complication after gastric bypass for morbid obesity. Obes. Surg. 2004, 14, 1012–1015. [Google Scholar] [CrossRef] [PubMed]
- Baierlein, S.A.; Wistop, A.; Looser, C.; Peters, T.; Riehle, H.M.; von Flüe, M.; Peterli, R. Abdominal actinomycosis: A rare complication after laparoscopic gastric bypass. Obes. Surg. 2007, 17, 1123–1126. [Google Scholar] [CrossRef] [PubMed]
- Peloux, Y.; Chardon, H.; Lagier, E.; Jauffret, P.; Latil, G.; de Cuttoli, J.P. Pathogenic role of Actinomyces apart from actinomycosis. Apropos of 2 cases of acute suppurations with Actinomyces odontolyticus. Sem. Hop. 1983, 59, 3063–3064. [Google Scholar]
- Chao, C.T.; Liao, C.H.; Lai, C.C.; Hsueh, P.R. Liver abscess due to Actinomyces odontolyticus in an immunocompetent patient. Infection. 2011, 39, 77–79. [Google Scholar] [CrossRef]
- Wu, C.M.; Noska, A. Intrauterine device infection causing concomitant streptococcal toxic shock syndrome and pelvic abscess with Actinomyces odontolyticus bacteraemia. BMJ Case Rep. 2016, 2016, bcr2015213236. [Google Scholar] [CrossRef]
- Lopes, V.N.; Dantas, M.J.; Andrade, P.; Pinto-de-Sousa, J. Secondary peritonitis by Actinomyces odontolyticus. Porto. Biomed. J. 2017, 2, 174–175. [Google Scholar] [CrossRef]
- Verrot, D.; Disdier, P.; Harlé, J.R.; Peloux, Y.; Garbes, L.; Arnaud, A.; Weiller, P.J. Pulmonary actinomycosis: Caused by Actinomyces odontolyticus? Rev. Med. Interne 1993, 14, 179–181. [Google Scholar] [CrossRef]
- Takiguchi, Y.; Terano, T.; Hirai, A. Lung abscess caused by Actinomyces odontolyticus. Intern. Med. 2003, 42, 723–725. [Google Scholar] [CrossRef] [PubMed]
- Crisafulli, E.; Bernardinello, N.; Alfieri, V.; Pellegrino, F.; Lazzari, C.; Gnetti, L.; Chetta, A. A pulmonary infection by Actinomyces odontolyticus and Veillonella atypica in an immunocompetent patient with dental caries. Respirol. Case Rep. 2019, 7, e00493. [Google Scholar] [CrossRef] [PubMed]
- Bennhoff, D.F. Actinomycosis: Diagnostic and therapeutic considerations and a review of 32 cases. Laryngoscope 1984, 94, 1198–1217. [Google Scholar] [CrossRef] [PubMed]
- Clarridge, J.E.; Zhang, Q. Genotypic diversity of clinical Actinomyces species: Phenotype, source, and disease correlation among genospecies. J. Clin. Microbiol. 2002, 40, 3442–3448. [Google Scholar] [CrossRef]
- Valour, F.; Sénéchal, A.; Dupieux, C.; Karsenty, J.; Lustig, S.; Breton, P.; Gleizal, A.; Boussel, L.; Laurent, F.; Braun, E.; et al. Actinomycosis: Etiology, clinical features, diagnosis, treatment, and management. Infect. Drug Resist. 2014, 7, 183. [Google Scholar] [CrossRef] [PubMed]
- Hillenkamp, F.; Karas, M.; Beavis, R.C.; Chait, B.T. Matrix-assisted laser desorption/ionization mass spectrometry of biopolymers. Anal. Chem. 1991, 63, 1193A–1203A. [Google Scholar] [CrossRef]
- Meyer, P.; Nwariaku, O.; McClelland, R.N.; Gibbons, D.; Leach, F.; Sagalowsky, A.I.; Simmang, C.; Jeyarajah, D.R. Rare presentation of actinomycosis as an abdominal mass. Dis. Colon. Rectum. 2000, 43, 872–875. [Google Scholar] [CrossRef]
- Deshmukh, N.; Heaney, S.J. Actinomycosis at multiple colonic sites. Am. J. Gastroenterol. 1986, 81, 1212–1214. [Google Scholar]
- Fowler, R.; Simpkins, K. Abdominal actinomycosis: A report of three cases. Clin. Radiol. 1983, 34, 301. [Google Scholar] [CrossRef]
- Sung, H.Y.; Lee, I.S.; Kim, S.I.; Jung, S.E.; Kim, S.W.; Kim, S.Y.; Chung, M.K.; Kim, W.C.; Oh, S.T.; Kang, W.K. Clinical features of abdominal actinomycosis: A 15-year experience of a single institute. J. Korean Med. Sci. 2011, 26, 932–937. [Google Scholar] [CrossRef]
- Sartelli, M.; Boermeester, M.A.; Cainzos, M.; Coccolini, F.; de Jonge, S.W.; Rasa, K.; Dellinger, E.P.; McNamara, D.A.; Fry, D.E.; Cui, Y.; et al. Six Long-Standing Questions about Antibiotic Prophylaxis in Surgery. Antibiotics 2023, 12, 908. [Google Scholar] [CrossRef] [PubMed]
- Wagenlehner, F.M.; Mohren, B.; Naber, K.G.; Männl, H.F. Abdominal actinomycosis. Clin. Microbiol. Infect. 2003, 9, 881–885. [Google Scholar] [CrossRef]
- Koneman, E.W.; Allen, S.D.; Janda, W.M.; Schreckenberger, P.C.; Winn, W.C. Diagnostic microbiology. In The Nonfermentative Gram-Negative Bacilli Philadelphia; Lippincott-Raven Publishers: Philadelphia, PA, USA, 1997; pp. 253–320. [Google Scholar]
- Vyas, J.M.; Kasmar, A.; Chang, H.R.; Holden, J.; Hohmann, E. Abdominal abscesses due to actinomycosis after laparoscopic cholecystectomy: Case reports and review. Clin. Infect. Dis. 2007, 44, e1–e4. [Google Scholar] [CrossRef] [PubMed]
- Saleem, A.; Clement, L.K.C.; Sameer, P.J.; Winston, W.; Vishal, G.S. Percutaneous drainage for giant pyogenic liver abscess--is it safe and sufficient? Am. J. Surg. 2016, 211, 95–101. [Google Scholar] [CrossRef]
- Coccolini, F.; Sartelli, M.; Sawyer, R.; Rasa, K.; Viaggi, B.; Abu-Zidan, F.; Soreide, K.; Hardcastle, T.; Gupta, D.; Bendinelli, C.; et al. Source control in emergency general surgery: WSES, GAIS, SIS-E, SIS-A guidelines. World J. Emerg. Surg. 2023, 18, 41. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.-G.; Lee, H.-K.; Park, E. Abdominal Actinomycotic Abscess after Laparoscopic Sleeve Gastrectomy for Morbid Obesity: A Case Report. Medicina 2023, 59, 1516. https://doi.org/10.3390/medicina59091516
Kim H-G, Lee H-K, Park E. Abdominal Actinomycotic Abscess after Laparoscopic Sleeve Gastrectomy for Morbid Obesity: A Case Report. Medicina. 2023; 59(9):1516. https://doi.org/10.3390/medicina59091516
Chicago/Turabian StyleKim, Ho-Goon, Ho-Kyun Lee, and Eunkyu Park. 2023. "Abdominal Actinomycotic Abscess after Laparoscopic Sleeve Gastrectomy for Morbid Obesity: A Case Report" Medicina 59, no. 9: 1516. https://doi.org/10.3390/medicina59091516
APA StyleKim, H.-G., Lee, H.-K., & Park, E. (2023). Abdominal Actinomycotic Abscess after Laparoscopic Sleeve Gastrectomy for Morbid Obesity: A Case Report. Medicina, 59(9), 1516. https://doi.org/10.3390/medicina59091516





