Coagulation Dysfunctions in Non-Alcoholic Fatty Liver Disease—Oxidative Stress and Inflammation Relevance
Abstract
:1. Introduction
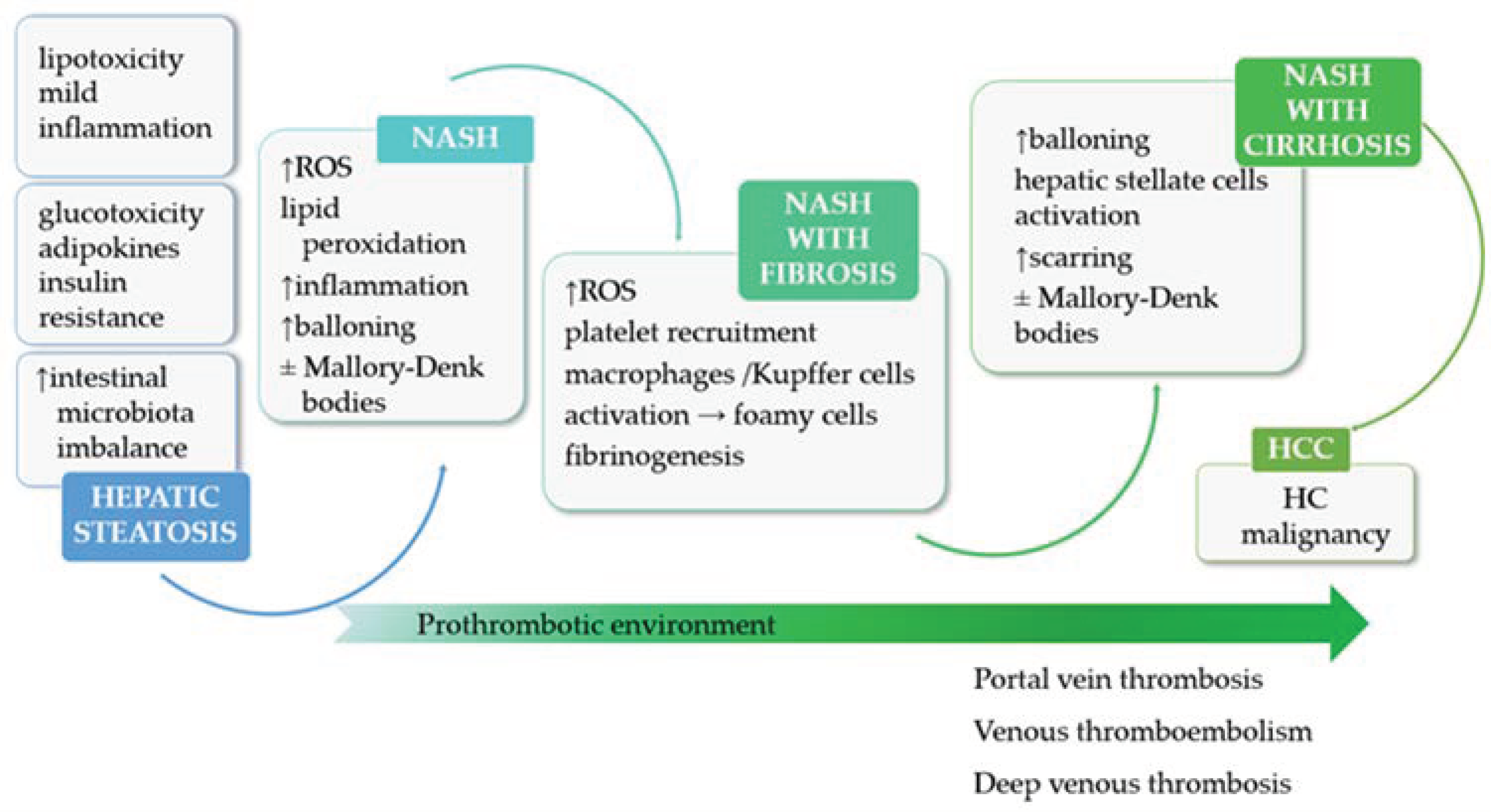
2. Non-Alcoholic Fatty Liver Disease
3. Oxidative Stress and Inflammation in Non-Alcoholic Fatty Liver Disease
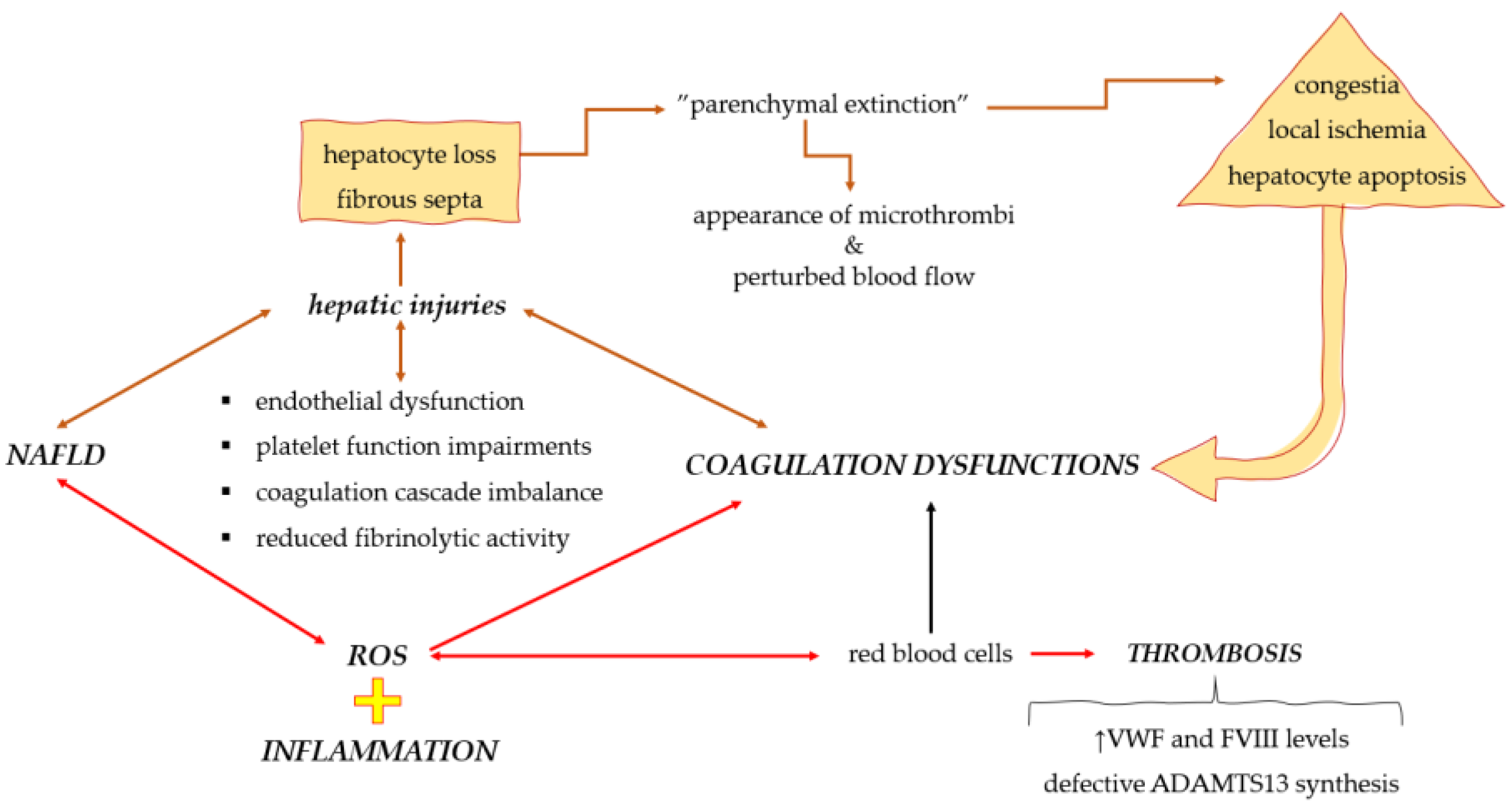
4. Coagulation Dysfunctions in Non-Alcoholic Fatty Liver Disease
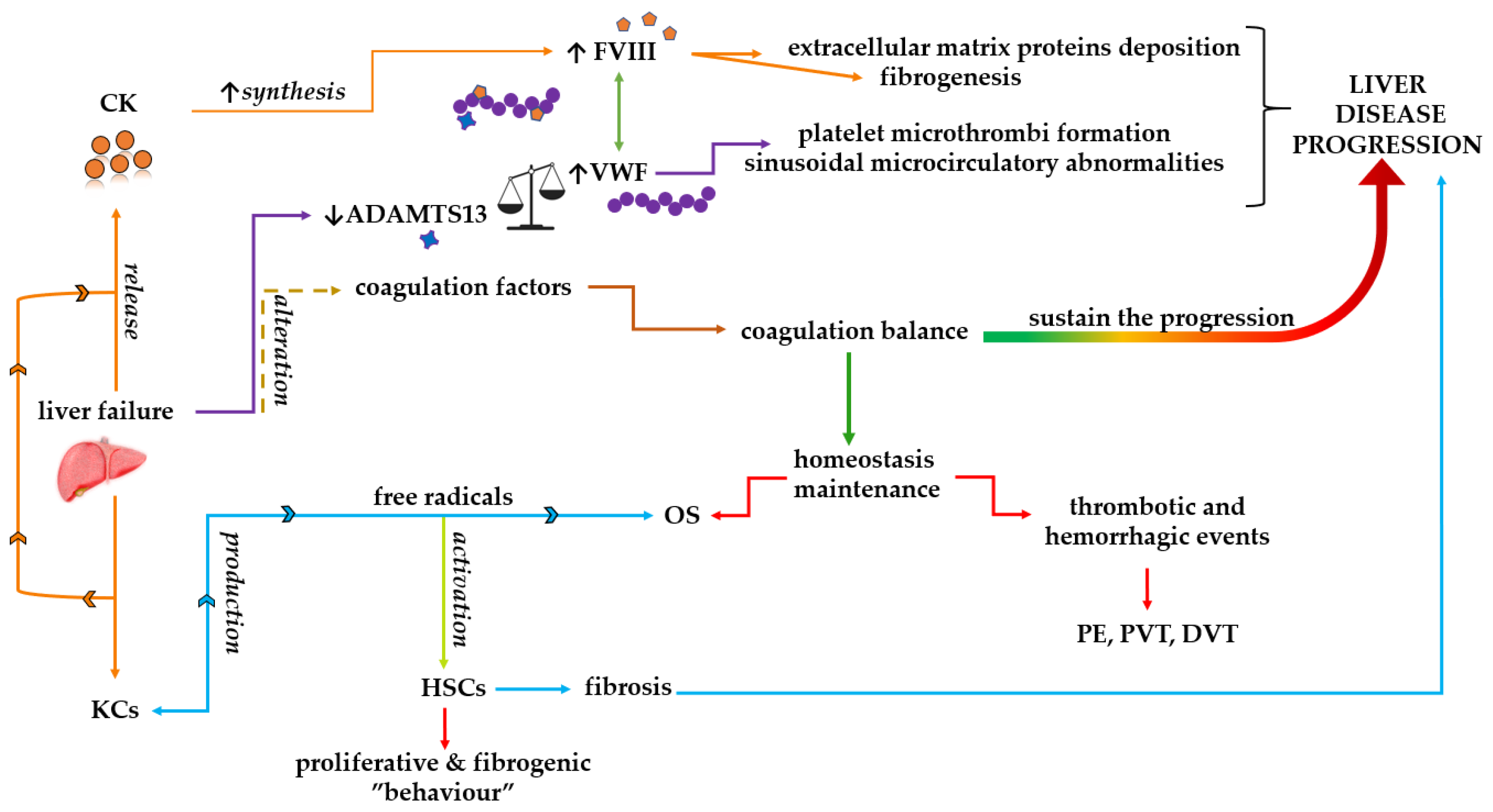
5. Possible Interaction Mechanisms among Coagulation Dyshomeostasis, Oxidative Stress, and Inflammation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Li, B.; Zhang, C.; Zhan, Y.-T. Nonalcoholic Fatty Liver Disease Cirrhosis: A Review of Its Epidemiology, Risk Factors, Clinical Presentation, Diagnosis, Management, and Prognosis. Can. J. Gastroenterol. Hepatol. 2018, 2018, 2784537. [Google Scholar] [CrossRef]
- Huang, D.Q.; El-Serag, H.B.; Loomba, R. Global Epidemiology of NAFLD-Related HCC: Trends, Predictions, Risk Factors and Prevention. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 223–238. [Google Scholar] [CrossRef]
- Lazarus, J.V.; Mark, H.E.; Anstee, Q.M.; Arab, J.P.; Batterham, R.L.; Castera, L.; Cortez-Pinto, H.; Crespo, J.; Cusi, K.; Dirac, M.A.; et al. Advancing the Global Public Health Agenda for NAFLD: A Consensus Statement. Nat. Rev. Gastroenterol. Hepatol. 2022, 19, 60–78. [Google Scholar] [CrossRef] [PubMed]
- Riazi, K.; Azhari, H.; Charette, J.H.; Underwood, F.E.; King, J.A.; Afshar, E.E.; Swain, M.G.; Congly, S.E.; Kaplan, G.G.; Shaheen, A.-A. The Prevalence and Incidence of NAFLD Worldwide: A Systematic Review and Meta-Analysis. Lancet Gastroenterol. Hepatol. 2022, 7, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A.; Nascimbeni, F.; Ballestri, S.; Fairweather, D.; Win, S.; Than, T.A.; Abdelmalek, M.F.; Suzuki, A. Sex Differences in Nonalcoholic Fatty Liver Disease: State of the Art and Identification of Research Gaps. Hepatology 2019, 70, 1457–1469. [Google Scholar] [CrossRef] [PubMed]
- Puri, P.; Sanyal, A.J. Nonalcoholic Fatty Liver Disease: Definitions, Risk Factors, and Workup. Clin. Liver Dis. 2012, 1, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; Younossi, Z.M. Nonalcoholic Fatty Liver Disease: A Manifestation of the Metabolic Syndrome. Cleve. Clin. J. Med. 2008, 75, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Gastaldelli, A. Fatty Liver Disease: The Hepatic Manifestation of Metabolic Syndrome. Hypertens. Res. 2010, 33, 546–547. [Google Scholar] [CrossRef]
- Godoy-Matos, A.F.; Silva Júnior, W.S.; Valerio, C.M. NAFLD as a Continuum: From Obesity to Metabolic Syndrome and Diabetes. Diabetol. Metab. Syndr. 2020, 12, 60. [Google Scholar] [CrossRef]
- Dyson, J.K.; Anstee, Q.M.; McPherson, S. Non-Alcoholic Fatty Liver Disease: A Practical Approach to Diagnosis and Staging. Frontline Gastroenterol. 2014, 5, 211–218. [Google Scholar] [CrossRef]
- Westfall, E.; Jeske, R.; Bader, A.R. Nonalcoholic Fatty Liver Disease: Common Questions and Answers on Diagnosis and Management. Am. Fam. Physician 2020, 102, 603–612. [Google Scholar]
- Orci, L.A.; Sanduzzi-Zamparelli, M.; Caballol, B.; Sapena, V.; Colucci, N.; Torres, F.; Bruix, J.; Reig, M.; Toso, C. Incidence of Hepatocellular Carcinoma in Patients With Nonalcoholic Fatty Liver Disease: A Systematic Review, Meta-Analysis, and Meta-Regression. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2022, 20, 283–292.e10. [Google Scholar] [CrossRef] [PubMed]
- Martinou, E.; Pericleous, M.; Stefanova, I.; Kaur, V.; Angelidi, A.M. Diagnostic Modalities of Non-Alcoholic Fatty Liver Disease: From Biochemical Biomarkers to Multi-Omics Non-Invasive Approaches. Diagnostics 2022, 12, 407. [Google Scholar] [CrossRef] [PubMed]
- Masarone, M.; Rosato, V.; Dallio, M.; Gravina, A.G.; Aglitti, A.; Loguercio, C.; Federico, A.; Persico, M. Role of Oxidative Stress in Pathophysiology of Nonalcoholic Fatty Liver Disease. Oxid. Med. Cell. Longev. 2018, 2018, 9547613. [Google Scholar] [CrossRef]
- Delli Bovi, A.P.; Marciano, F.; Mandato, C.; Siano, M.A.; Savoia, M.; Vajro, P. Oxidative Stress in Non-Alcoholic Fatty Liver Disease. An Updated Mini Review. Front. Med. 2021, 8, 595371. [Google Scholar] [CrossRef]
- Virović-Jukić, L.; Stojsavljević-Shapeski, S.; Forgač, J.; Kukla, M.; Mikolašević, I. Non-Alcoholic Fatty Liver Disease—A Procoagulant Condition? Croat. Med. J. 2021, 62, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Ogresta, D.; Mrzljak, A.; Cigrovski Berkovic, M.; Bilic-Curcic, I.; Stojsavljevic-Shapeski, S.; Virovic-Jukic, L. Coagulation and Endothelial Dysfunction Associated with NAFLD: Current Status and Therapeutic Implications. J. Clin. Transl. Hepatol. 2022, 10, 339–355. [Google Scholar] [CrossRef]
- Dolganiuc, A. Alcohol and Viral Hepatitis: Role of Lipid Rafts. Alcohol. Res. 2015, 37, 299–309. [Google Scholar]
- Kang, S.H.; Cho, Y.; Jeong, S.W.; Kim, S.U.; Lee, J.-W. From Nonalcoholic Fatty Liver Disease to Metabolic-Associated Fatty Liver Disease: Big Wave or Ripple? Clin. Mol. Hepatol. 2021, 27, 257–269. [Google Scholar] [CrossRef]
- Muzica, C.M.; Sfarti, C.; Trifan, A.; Zenovia, S.; Cuciureanu, T.; Nastasa, R.; Huiban, L.; Cojocariu, C.; Singeap, A.-M.; Girleanu, I.; et al. Nonalcoholic Fatty Liver Disease and Type 2 Diabetes Mellitus: A Bidirectional Relationship. Can. J. Gastroenterol. Hepatol. 2020, 2020, 6638306. [Google Scholar] [CrossRef]
- Chen, F.; Esmaili, S.; Rogers, G.B.; Bugianesi, E.; Petta, S.; Marchesini, G.; Bayoumi, A.; Metwally, M.; Azardaryany, M.K.; Coulter, S.; et al. Lean NAFLD: A Distinct Entity Shaped by Differential Metabolic Adaptation. Hepatology 2020, 71, 1213–1227. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Ren, J.; Zhou, W.; Huang, J.; Wu, G.; Yang, F.; Yuan, S.; Fang, J.; Liu, J.; Jin, Y.; et al. Lean Non-Alcoholic Fatty Liver Disease (Lean-NAFLD) and the Development of Metabolic Syndrome: A Retrospective Study. Sci. Rep. 2022, 12, 10977. [Google Scholar] [CrossRef]
- De Lucia Rolfe, E.; Brage, S.; Sleigh, A.; Finucane, F.; Griffin, S.J.; Wareham, N.J.; Ong, K.K.; Forouhi, N.G. Validity of ultrasonography to assess hepatic steatosis compared to magnetic resonance spectroscopy as a criterion method in older adults. PLoS ONE 2018, 13, e0207923. [Google Scholar] [CrossRef]
- Wu, M.; Sharma, P.G.; Grajo, J.R. The Echogenic Liver: Steatosis and Beyond. Ultrasound Q. 2020, 37, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Juluri, R.; Vuppalanchi, R.; Olson, J.; Unalp, A.; Van Natta, M.L.; Cummings, O.W.; Tonascia, J.; Chalasani, N. Generalizability of the Nonalcoholic Steatohepatitis Clinical Research Network Histologic Scoring System for Nonalcoholic Fatty Liver Disease. J. Clin. Gastroenterol. 2011, 45, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Grau, A.; Gabriel-Medina, P.; Rodriguez-Algarra, F.; Villena, Y.; Lopez-Martínez, R.; Augustín, S.; Pons, M.; Cruz, L.-M.; Rando-Segura, A.; Enfedaque, B.; et al. Assessing Liver Fibrosis Using the FIB4 Index in the Community Setting. Diagnostics 2021, 11, 2236. [Google Scholar] [CrossRef] [PubMed]
- Coulon, S.; Legry, V.; Heindryckx, F.; Van Steenkiste, C.; Casteleyn, C.; Olievier, K.; Libbrecht, L.; Carmeliet, P.; Jonckx, B.; Stassen, J.-M.; et al. Role of Vascular Endothelial Growth Factor in the Pathophysiology of Nonalcoholic Steatohepatitis in Two Rodent Models. Hepatology 2013, 57, 1793–1805. [Google Scholar] [CrossRef]
- Siddiqui, H.; Rawal, P.; Bihari, C.; Arora, N.; Kaur, S. Vascular Endothelial Growth Factor Promotes Proliferation of Epithelial Cell Adhesion Molecule-Positive Cells in Nonalcoholic Steatohepatitis. J. Clin. Exp. Hepatol. 2020, 10, 275–283. [Google Scholar] [CrossRef]
- Tarantino, G.; Citro, V.; Balsano, C. Liver-Spleen Axis in Nonalcoholic Fatty Liver Disease. Expert Rev. Gastroenterol. Hepatol. 2021, 15, 759–769. [Google Scholar] [CrossRef]
- Xu, X.; Jiang, L.; Wu, C.; Pan, L.; Lou, Z.; Peng, C.; Dong, Y.; Ruan, B. The Role of Fibrosis Index FIB-4 in Predicting Liver Fibrosis Stage and Clinical Prognosis: A Diagnostic or Screening Tool? J. Formos. Med. Assoc. 2022, 121, 454–466. [Google Scholar] [CrossRef]
- Powell, E.E.; Wong, V.W.-S.; Rinella, M. Non-Alcoholic Fatty Liver Disease. Lancet 2021, 397, 2212–2224. [Google Scholar] [CrossRef] [PubMed]
- Conde de la Rosa, L.; Goicoechea, L.; Torres, S.; Garcia-Ruiz, C.; Fernandez-Checa, J.C. Role of Oxidative Stress in Liver Disorders. Livers 2022, 2, 283–314. [Google Scholar] [CrossRef]
- Tandra, S.; Yeh, M.M.; Brunt, E.M.; Vuppalanchi, R.; Cummings, O.W.; Ünalp-Arida, A.; Wilson, L.A.; Chalasani, N. Presence and Significance of Microvesicular Steatosis in Nonalcoholic Fatty Liver Disease. J. Hepatol. 2011, 55, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, E.; Tokushige, K.; Ludwig, J. Diagnosis and Classification of Non-Alcoholic Fatty Liver Disease and Non-Alcoholic Steatohepatitis: Current Concepts and Remaining Challenges. Hepatol. Res. 2015, 45, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Tsutsumi, V.; Nakamura, T.; Ueno, T.; Torimura, T.; Aguirre-García, J. Structure and Ultrastructure of the Normal and Diseased Liver. In Liver Pathophysiology; Muriel, P., Ed.; Academic Press: Boston, MA, USA, 2017; Chapter 2; pp. 23–44. [Google Scholar] [CrossRef]
- Anstee, Q.M.; Day, C.P. 26—Epidemiology, Natural History, and Evaluation of Nonalcoholic Fatty Liver Disease. In Zakim and Boyer’s Hepatology, 7th ed.; Sanyal, A.J., Boyer, T.D., Lindor, K.D., Terrault, N.A.B.T.-Z., Boyer, T.D., Eds.; Elsevier: Philadelphia, PA, USA, 2018; pp. 391–405.e3. [Google Scholar] [CrossRef]
- Takahashi, Y.; Fukusato, T. Histopathology of Nonalcoholic Fatty Liver Disease/Nonalcoholic Steatohepatitis. World J. Gastroenterol. 2014, 20, 15539–15548. [Google Scholar] [CrossRef] [PubMed]
- Cataldo, I.; Sarcognato, S.; Sacchi, D.; Cacciatore, M.; Baciorri, F.; Mangia, A.; Cazzagon, N.; Guido, M. Pathology of Non-Alcoholic Fatty Liver Disease. Pathologica 2021, 113, 194–202. [Google Scholar] [CrossRef]
- Basaranoglu, M.; Turhan, N.; Sonsuz, A.; Basaranoglu, G. Mallory-Denk Bodies in Chronic Hepatitis. World J. Gastroenterol. 2011, 17, 2172–2177. [Google Scholar] [CrossRef]
- Toshikuni, N.; Tsutsumi, M.; Arisawa, T. Clinical Differences between Alcoholic Liver Disease and Nonalcoholic Fatty Liver Disease. World J. Gastroenterol. 2014, 20, 8393–8406. [Google Scholar] [CrossRef]
- Hübscher, S.G. 24—Alcohol-Induced Liver Disease. In Pattern Recognition, 2nd ed.; Theodoridis, S., Koutroumbas, K., Eds.; Elsevier: Philadelphia, PA, USA, 2018; pp. 371–390. [Google Scholar] [CrossRef]
- Zhang, P.; Wang, W.; Mao, M.; Gao, R.; Shi, W.; Li, D.; Calderone, R.; Sui, B.; Tian, X.; Meng, X. Similarities and Differences: A Comparative Review of the Molecular Mechanisms and Effectors of NAFLD and AFLD. Front. Physiol. 2021, 12, 710285. [Google Scholar] [CrossRef]
- Park, S.H.; Seo, W.; Xu, M.-J.; Mackowiak, B.; Lin, Y.; He, Y.; Fu, Y.; Hwang, S.; Kim, S.-J.; Guan, Y.; et al. Ethanol and Its Nonoxidative Metabolites Promote Acute Liver Injury by Inducing ER Stress, Adipocyte Death, and Lipolysis. Cell. Mol. Gastroenterol. Hepatol. 2023, 15, 281–306. [Google Scholar] [CrossRef]
- Hyun, J.; Han, J.; Lee, C.; Yoon, M.; Jung, Y. Pathophysiological Aspects of Alcohol Metabolism in the Liver. Int. J. Mol. Sci. 2021, 22, 5717. [Google Scholar] [CrossRef]
- Anstee, Q.M.; Reeves, H.L.; Kotsiliti, E.; Govaere, O.; Heikenwalder, M. From NASH to HCC: Current Concepts and Future Challenges. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 411–428. [Google Scholar] [CrossRef] [PubMed]
- Singeap, A.-M.; Stanciu, C.; Huiban, L.; Muzica, C.M.; Cuciureanu, T.; Girleanu, I.; Chiriac, S.; Zenovia, S.; Nastasa, R.; Sfarti, C.; et al. Association between Nonalcoholic Fatty Liver Disease and Endocrinopathies: Clinical Implications. Can. J. Gastroenterol. Hepatol. 2021, 2021, 6678142. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.P.; Stefano, J.T. Genetic Polymorphisms and Oxidative Stress in Non-Alcoholic Steatohepatitis (NASH): A Mini Review. Clin. Res. Hepatol. Gastroenterol. 2015, 39 (Suppl. S1), S35–S40. [Google Scholar] [CrossRef]
- Gabbia, D.; Cannella, L.; De Martin, S. The Role of Oxidative Stress in NAFLD-NASH-HCC Transition-Focus on NADPH Oxidases. Biomedicines 2021, 9, 687. [Google Scholar] [CrossRef]
- Smirne, C.; Croce, E.; Di Benedetto, D.; Cantaluppi, V.; Comi, C.; Sainaghi, P.P.; Minisini, R.; Grossini, E.; Pirisi, M. Oxidative Stress in Non-Alcoholic Fatty Liver Disease. Livers 2022, 2, 30–76. [Google Scholar] [CrossRef]
- Paschos, P.; Paletas, K. Non Alcoholic Fatty Liver Disease Two-Hit Process: Multifactorial Character of the Second Hit. Hippokratia 2009, 13, 128. [Google Scholar]
- Buzzetti, E.; Pinzani, M.; Tsochatzis, E.A. The Multiple-Hit Pathogenesis of Non-Alcoholic Fatty Liver Disease (NAFLD). Metabolism 2016, 65, 1038–1048. [Google Scholar] [CrossRef]
- Fang, Y.-L.; Chen, H.; Wang, C.-L.; Liang, L. Pathogenesis of Non-Alcoholic Fatty Liver Disease in Children and Adolescence: From “Two Hit Theory” to “Multiple Hit Model”. World J. Gastroenterol. 2018, 24, 2974–2983. [Google Scholar] [CrossRef]
- Riazi, K.; Raman, M.; Taylor, L.; Swain, M.G.; Shaheen, A.A. Dietary Patterns and Components in Nonalcoholic Fatty Liver Disease (NAFLD): What Key Messages Can Health Care Providers Offer? Nutrients 2019, 11, 2878. [Google Scholar] [CrossRef]
- Quek, J.; Chan, K.E.; Wong, Z.Y.; Tan, C.; Tan, B.; Lim, W.H.; Tan, D.J.H.; Tang, A.S.P.; Tay, P.; Xiao, J.; et al. Global Prevalence of Non-Alcoholic Fatty Liver Disease and Non-Alcoholic Steatohepatitis in the Overweight and Obese Population: A Systematic Review and Meta-Analysis. Lancet Gastroenterol. Hepatol. 2023, 8, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Liu, Y.; Hong, T. Epidemiological Characteristics and Management of Nonalcoholic Fatty Liver Disease/Nonalcoholic Steatohepatitis in China: A Narrative Review. Diabetes Obes. Metab. 2023, 25, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Hallsworth, K.; Thoma, C.; Moore, S.; Ploetz, T.; Anstee, Q.M.; Taylor, R.; Day, C.P.; Trenell, M.I. Non-Alcoholic Fatty Liver Disease Is Associated with Higher Levels of Objectively Measured Sedentary Behaviour and Lower Levels of Physical Activity than Matched Healthy Controls. Frontline Gastroenterol. 2015, 6, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.-G.; Kim, S.-U.; Wong, V.W.-S. New Trends on Obesity and NAFLD in Asia. J. Hepatol. 2017, 67, 862–873. [Google Scholar] [CrossRef]
- Joo, J.H.; Kim, H.J.; Park, E.-C.; Jang, S.-I. Association between Sitting Time and Non-Alcoholic Fatty Live Disease in South Korean Population: A Cross-Sectional Study. Lipids Health Dis. 2020, 19, 212. [Google Scholar] [CrossRef]
- Lim, G.-Y.; Chang, Y.; Kim, I.; Ryu, S.; Kwon, R.; Song, J. Long Working Hours and Increased Risks of Lean Non-Alcoholic Fatty Liver Disease among Korean Men and Women. Sci. Rep. 2023, 13, 12230. [Google Scholar] [CrossRef] [PubMed]
- Kuchay, M.S.; Martínez-Montoro, J.I.; Choudhary, N.S.; Fernández-García, J.C.; Ramos-Molina, B. Non-Alcoholic Fatty Liver Disease in Lean and Non-Obese Individuals: Current and Future Challenges. Biomedicines 2021, 9, 1346. [Google Scholar] [CrossRef]
- Yabe, Y.; Chihara, K.; Oshida, N.; Kamimaki, T.; Hasegawa, N.; Isobe, T.; Shoda, J. Survey of Dietary Habits and Physical Activity in Japanese Patients with Non-Obese Non-Alcoholic Fatty Liver Disease. Nutrients 2023, 15, 2764. [Google Scholar] [CrossRef]
- Albhaisi, S.; Chowdhury, A.; Sanyal, A.J. Non-Alcoholic Fatty Liver Disease in Lean Individuals. JHEP Reports 2019, 1, 329–341. [Google Scholar] [CrossRef]
- Chan, W.-K. Comparison between Obese and Non-Obese Nonalcoholic Fatty Liver Disease. Clin. Mol. Hepatol. 2023, 29, S58–S67. [Google Scholar] [CrossRef]
- Kitade, H.; Chen, G.; Ni, Y.; Ota, T. Nonalcoholic Fatty Liver Disease and Insulin Resistance: New Insights and Potential New Treatments. Nutrients 2017, 9, 387. [Google Scholar] [CrossRef] [PubMed]
- Gusdon, A.M.; Song, K.-X.; Qu, S. Nonalcoholic Fatty Liver Disease: Pathogenesis and Therapeutics from a Mitochondria-Centric Perspective. Oxid. Med. Cell. Longev. 2014, 2014, 637027. [Google Scholar] [CrossRef] [PubMed]
- Petäjä, E.M.; Yki-Järvinen, H. Definitions of Normal Liver Fat and the Association of Insulin Sensitivity with Acquired and Genetic NAFLD—A Systematic Review. Int. J. Mol. Sci. 2016, 17, 633. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.; Hatab, A.; Athwal, V.S.; Jokl, E.; Piper Hanley, K. Genetic Contribution to Non-Alcoholic Fatty Liver Disease and Prognostic Implications. Curr. Diab. Rep. 2021, 21, 8. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.-Z.; Xin, Y.-N.; Geng, N.; Jiang, M.; Zhang, D.-D.; Xuan, S.-Y. PNPLA3 I148M Variant in Nonalcoholic Fatty Liver Disease: Demographic and Ethnic Characteristics and the Role of the Variant in Nonalcoholic Fatty Liver Fibrosis. World J. Gastroenterol. 2015, 21, 794–802. [Google Scholar] [CrossRef]
- Dong, X.C. PNPLA3-A Potential Therapeutic Target for Personalized Treatment of Chronic Liver Disease. Front. Med. 2019, 6, 304. [Google Scholar] [CrossRef]
- Salari, N.; Darvishi, N.; Mansouri, K.; Ghasemi, H.; Hosseinian-Far, M.; Darvishi, F.; Mohammadi, M. Association between PNPLA3 Rs738409 Polymorphism and Nonalcoholic Fatty Liver Disease: A Systematic Review and Meta-Analysis. BMC Endocr. Disord. 2021, 21, 125. [Google Scholar] [CrossRef]
- Zain, S.M.; Mohamed, R.; Mahadeva, S.; Cheah, P.L.; Rampal, S.; Basu, R.C.; Mohamed, Z. A Multi-Ethnic Study of a PNPLA3 Gene Variant and Its Association with Disease Severity in Non-Alcoholic Fatty Liver Disease. Hum. Genet. 2012, 131, 1145–1152. [Google Scholar] [CrossRef]
- Oh, S.-W.; Lee, J.-E.; Shin, E.; Kwon, H.; Choe, E.K.; Choi, S.-Y.; Rhee, H.; Choi, S.H. Genome-Wide Association Study of Metabolic Syndrome in Korean Populations. PLoS ONE 2020, 15, e0227357. [Google Scholar] [CrossRef]
- Sookoian, S.; Pirola, C.J.; Valenti, L.; Davidson, N.O. Genetic Pathways in Nonalcoholic Fatty Liver Disease: Insights From Systems Biology. Hepatology 2020, 72, 330–346. [Google Scholar] [CrossRef]
- Sun, D.-Q.; Wang, T.-Y.; Zheng, K.I.; Zhang, H.-Y.; Wang, X.-D.; Targher, G.; Byrne, C.D.; Chen, Y.-P.; Yuan, W.-J.; Jin, Y.; et al. The HSD17B13 Rs72613567 Variant Is Associated with Lower Levels of Albuminuria in Patients with Biopsy-Proven Nonalcoholic Fatty Liver Disease. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 1822–1831. [Google Scholar] [CrossRef]
- Luo, F.; Oldoni, F.; Das, A. TM6SF2: A Novel Genetic Player in Nonalcoholic Fatty Liver and Cardiovascular Disease. Hepatol. Commun. 2022, 6, 448–460. [Google Scholar] [CrossRef] [PubMed]
- Ferro, D.; Baratta, F.; Pastori, D.; Cocomello, N.; Colantoni, A.; Angelico, F.; Del Ben, M. New Insights into the Pathogenesis of Non-Alcoholic Fatty Liver Disease: Gut-Derived Lipopolysaccharides and Oxidative Stress. Nutrients 2020, 12, 2762. [Google Scholar] [CrossRef]
- Koek, G.H.; Liedorp, P.R.; Bast, A. The Role of Oxidative Stress in Non-Alcoholic Steatohepatitis. Clin. Chim. Acta 2011, 412, 1297–1305. [Google Scholar] [CrossRef] [PubMed]
- Reiniers, M.J.; van Golen, R.F.; van Gulik, T.M.; Heger, M. Reactive Oxygen and Nitrogen Species in Steatotic Hepatocytes: A Molecular Perspective on the Pathophysiology of Ischemia-Reperfusion Injury in the Fatty Liver. Antioxid. Redox Signal. 2014, 21, 1119–1142. [Google Scholar] [CrossRef]
- Farzanegi, P.; Dana, A.; Ebrahimpoor, Z.; Asadi, M.; Azarbayjani, M.A. Mechanisms of Beneficial Effects of Exercise Training on Non-Alcoholic Fatty Liver Disease (NAFLD): Roles of Oxidative Stress and Inflammation. Eur. J. Sport Sci. 2019, 19, 994–1003. [Google Scholar] [CrossRef]
- Almazroo, O.A.; Miah, M.K.; Venkataramanan, R. Drug Metabolism in the Liver. Clin. Liver Dis. 2017, 21, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Ezhilarasan, D.; Lakshmi, T. A Molecular Insight into the Role of Antioxidants in Nonalcoholic Fatty Liver Diseases. Oxid. Med. Cell. Longev. 2022, 2022, 9233650. [Google Scholar] [CrossRef]
- Abe, R.A.M.; Masroor, A.; Khorochkov, A.; Prieto, J.; Singh, K.B.; Nnadozie, M.C.; Abdal, M.; Shrestha, N.; Mohammed, L. The Role of Vitamins in Non-Alcoholic Fatty Liver Disease: A Systematic Review. Cureus 2021, 13, e16855. [Google Scholar] [CrossRef]
- Manoppo, J.I.C.; Pateda, V.; Prayogo, C.; Langi, F.L.F.G.; Nurkolis, F.; Tsopmo, A. Relationships of 25-Hydroxyvitamin D Levels and Non-Alcoholic Fatty Liver Disease in Obese Children: A Possible Strategy to Promote Early Screening of NAFLD. Front. Nutr. 2022, 9, 1025396. [Google Scholar] [CrossRef]
- Licata, A.; Zerbo, M.; Como, S.; Cammilleri, M.; Soresi, M.; Montalto, G.; Giannitrapani, L. The Role of Vitamin Deficiency in Liver Disease: To Supplement or Not Supplement? Nutrients 2021, 13, 4014. [Google Scholar] [CrossRef] [PubMed]
- Karin, D.; Koyama, Y.; Brenner, D.; Kisseleva, T. The Characteristics of Activated Portal Fibroblasts/Myofibroblasts in Liver Fibrosis. Differentiation 2016, 92, 84–92. [Google Scholar] [CrossRef]
- Higashi, T.; Friedman, S.L.; Hoshida, Y. Hepatic Stellate Cells as Key Target in Liver Fibrosis. Adv. Drug Deliv. Rev. 2017, 121, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Longo, M.; Paolini, E.; Meroni, M.; Dongiovanni, P. Remodeling of Mitochondrial Plasticity: The Key Switch from NAFLD/NASH to HCC. Int. J. Mol. Sci. 2021, 22, 4173. [Google Scholar] [CrossRef] [PubMed]
- Shami, G.J.; Cheng, D.; Verhaegh, P.; Koek, G.; Wisse, E.; Braet, F. Three-Dimensional Ultrastructure of Giant Mitochondria in Human Non-Alcoholic Fatty Liver Disease. Sci. Rep. 2021, 11, 3319. [Google Scholar] [CrossRef]
- Tiniakos, D.G.; Anstee, Q.M.; Brunt, E.M.; Burt, A.D. Fatty Liver Disease. In MacSween’s Pathology of the Liver, 8th ed; Elsevier: Philadelphia, PA, USA, 2024; Chapter 5; pp. 330–401. [Google Scholar] [CrossRef]
- Gomes, A.L.; Teijeiro, A.; Burén, S.; Tummala, K.S.; Yilmaz, M.; Waisman, A.; Theurillat, J.-P.; Perna, C.; Djouder, N. Metabolic Inflammation-Associated IL-17A Causes Non-Alcoholic Steatohepatitis and Hepatocellular Carcinoma. Cancer Cell 2016, 30, 161–175. [Google Scholar] [CrossRef]
- Lauterbach, M.A.R.; Wunderlich, F.T. Macrophage Function in Obesity-Induced Inflammation and Insulin Resistance. Pflugers Arch. 2017, 469, 385–396. [Google Scholar] [CrossRef]
- Li, H.; Meng, Y.; He, S.; Tan, X.; Zhang, Y.; Zhang, X.; Wang, L.; Zheng, W. Macrophages, Chronic Inflammation, and Insulin Resistance. Cells 2022, 11, 3001. [Google Scholar] [CrossRef]
- Kotronen, A.; Yki-Järvinen, H. Fatty Liver: A Novel Component of the Metabolic Syndrome. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 27–38. [Google Scholar] [CrossRef]
- Dowman, J.K.; Tomlinson, J.W.; Newsome, P.N. Pathogenesis of Non-Alcoholic Fatty Liver Disease. QJM An. Int. J. Med. 2010, 103, 71–83. [Google Scholar] [CrossRef]
- Sabir, U.; Irfan, H.M.; Alamgeer; Ullah, A.; Althobaiti, Y.S.; Asim, M.H. Reduction of Hepatic Steatosis, Oxidative Stress, Inflammation, Ballooning and Insulin Resistance After Therapy with Safranal in NAFLD Animal Model: A New Approach. J. Inflamm. Res. 2022, 15, 1293–1316. [Google Scholar] [CrossRef] [PubMed]
- Min, R.W.M.; Aung, F.W.M.; Liu, B.; Arya, A.; Win, S. Mechanism and Therapeutic Targets of C-Jun-N-Terminal Kinases Activation in Nonalcoholic Fatty Liver Disease. Biomedicines 2022, 10, 2035. [Google Scholar] [CrossRef] [PubMed]
- Win, S.; Than, T.A.; Zhang, J.; Oo, C.; Min, R.W.M.; Kaplowitz, N. New Insights into the Role and Mechanism of C-Jun-N-Terminal Kinase Signaling in the Pathobiology of Liver Diseases. Hepatology 2018, 67, 2013–2024. [Google Scholar] [CrossRef]
- Jiang, Y.; Xu, J.; Huang, P.; Yang, L.; Liu, Y.; Li, Y.; Wang, J.; Song, H.; Zheng, P. Scoparone Improves Nonalcoholic Steatohepatitis Through Alleviating JNK/Sab Signaling Pathway-Mediated Mitochondrial Dysfunction. Front. Pharmacol. 2022, 13, 863756. [Google Scholar] [CrossRef] [PubMed]
- Pierantonelli, I.; Svegliati-Baroni, G. Nonalcoholic Fatty Liver Disease: Basic Pathogenetic Mechanisms in the Progression From NAFLD to NASH. Transplantation 2019, 103, e1–e13. [Google Scholar] [CrossRef] [PubMed]
- Legaki, A.-I.; Moustakas, I.I.; Sikorska, M.; Papadopoulos, G.; Velliou, R.-I.; Chatzigeorgiou, A. Hepatocyte Mitochondrial Dynamics and Bioenergetics in Obesity-Related Non-Alcoholic Fatty Liver Disease. Curr. Obes. Rep. 2022, 11, 126–143. [Google Scholar] [CrossRef]
- Jornayvaz, F.R.; Shulman, G.I. Regulation of Mitochondrial Biogenesis. Essays Biochem. 2010, 47, 69–84. [Google Scholar] [CrossRef]
- Popov, L.-D. Mitochondrial Biogenesis: An Update. J. Cell. Mol. Med. 2020, 24, 4892–4899. [Google Scholar] [CrossRef]
- Sarwar, R.; Pierce, N.; Koppe, S. Obesity and Nonalcoholic Fatty Liver Disease: Current Perspectives. Diabetes. Metab. Syndr. Obes. 2018, 11, 533–542. [Google Scholar] [CrossRef]
- Polyzos, S.A.; Kountouras, J.; Mantzoros, C.S. Obesity and Nonalcoholic Fatty Liver Disease: From Pathophysiology to Therapeutics. Metabolism 2019, 92, 82–97. [Google Scholar] [CrossRef]
- Karjoo, S.; Auriemma, A.; Fraker, T.; Bays, H.E. Nonalcoholic Fatty Liver Disease and Obesity: An Obesity Medicine Association (OMA) Clinical Practice Statement (CPS) 2022. Obes. Pillars 2022, 3, 100027. [Google Scholar] [CrossRef]
- Miyao, M.; Kawai, C.; Kotani, H.; Minami, H.; Abiru, H.; Hamayasu, H.; Eguchi, S.; Tamaki, K. Mitochondrial Fission in Hepatocytes as a Potential Therapeutic Target for Nonalcoholic Steatohepatitis. Hepatol. Res. 2022, 52, 1020–1033. [Google Scholar] [CrossRef] [PubMed]
- Aharoni-Simon, M.; Hann-Obercyger, M.; Pen, S.; Madar, Z.; Tirosh, O. Fatty Liver Is Associated with Impaired Activity of PPARγ-Coactivator 1α (PGC1α) and Mitochondrial Biogenesis in Mice. Lab. Investig. 2011, 91, 1018–1028. [Google Scholar] [CrossRef] [PubMed]
- Morris, E.M.; Meers, G.M.E.; Booth, F.W.; Fritsche, K.L.; Hardin, C.D.; Thyfault, J.P.; Ibdah, J.A. PGC-1α Overexpression Results in Increased Hepatic Fatty Acid Oxidation with Reduced Triacylglycerol Accumulation and Secretion. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G979–G992. [Google Scholar] [CrossRef] [PubMed]
- Katoch, S.; Sharma, V.; Patial, V. Peroxisome Proliferator-Activated Receptor Gamma as a Therapeutic Target for Hepatocellular Carcinoma: Experimental and Clinical Scenarios. World J. Gastroenterol. 2022, 28, 3535–3554. [Google Scholar] [CrossRef]
- Mastropasqua, F.; Girolimetti, G.; Shoshan, M. PGC1α: Friend or Foe in Cancer? Genes 2018, 9, 48. [Google Scholar] [CrossRef]
- Li, R.; Toan, S.; Zhou, H. Role of Mitochondrial Quality Control in the Pathogenesis of Nonalcoholic Fatty Liver Disease. Aging 2020, 12, 6467–6485. [Google Scholar] [CrossRef]
- Wang, L.; Li, X.; Hanada, Y.; Hasuzawa, N.; Moriyama, Y.; Nomura, M.; Yamamoto, K. Dynamin-Related Protein 1 Deficiency Accelerates Lipopolysaccharide-Induced Acute Liver Injury and Inflammation in Mice. Commun. Biol. 2021, 4, 894. [Google Scholar] [CrossRef]
- Shan, S.; Liu, Z.; Wang, S.; Liu, Z.; Huang, Z.; Yang, Y.; Zhang, C.; Song, F. Drp1-Mediated Mitochondrial Fission Promotes Carbon Tetrachloride-Induced Hepatic Fibrogenesis in Mice. Toxicol. Res. 2022, 11, 486–497. [Google Scholar] [CrossRef]
- Babbar, M.; Sheikh, M.S. Metabolic Stress and Disorders Related to Alterations in Mitochondrial Fission or Fusion. Mol. Cell. Pharmacol. 2013, 5, 109–133. [Google Scholar]
- Zacharioudakis, E.; Agianian, B.; Kumar Mv, V.; Biris, N.; Garner, T.P.; Rabinovich-Nikitin, I.; Ouchida, A.T.; Margulets, V.; Nordstrøm, L.U.; Riley, J.S.; et al. Modulating Mitofusins to Control Mitochondrial Function and Signaling. Nat. Commun. 2022, 13, 3775. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, J.S.; Bhatti, G.K.; Reddy, P.H. Mitochondrial Dysfunction and Oxidative Stress in Metabolic Disorders—A Step towards Mitochondria Based Therapeutic Strategies. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1066–1077. [Google Scholar] [CrossRef] [PubMed]
- Sebastián, D.; Hernández-Alvarez, M.I.; Segalés, J.; Sorianello, E.; Muñoz, J.P.; Sala, D.; Waget, A.; Liesa, M.; Paz, J.C.; Gopalacharyulu, P.; et al. Mitofusin 2 (Mfn2) Links Mitochondrial and Endoplasmic Reticulum Function with Insulin Signaling and Is Essential for Normal Glucose Homeostasis. Proc. Natl. Acad. Sci. USA 2012, 109, 5523–5528. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Alvarez, M.I.; Sebastián, D.; Vives, S.; Ivanova, S.; Bartoccioni, P.; Kakimoto, P.; Plana, N.; Veiga, S.R.; Hernández, V.; Vasconcelos, N.; et al. Deficient Endoplasmic Reticulum-Mitochondrial Phosphatidylserine Transfer Causes Liver Disease. Cell 2019, 177, 881–895.e17. [Google Scholar] [CrossRef]
- Shunkina, D.; Dakhnevich, A.; Shunkin, E.; Khaziakhmatova, O.; Shupletsova, V.; Vulf, M.; Komar, A.; Kirienkova, E.; Litvinova, L. Gp130 Activates Mitochondrial Dynamics for Hepatocyte Survival in a Model of Steatohepatitis. Biomedicines 2023, 11, 396. [Google Scholar] [CrossRef]
- Sangwung, P.; Petersen, K.F.; Shulman, G.I.; Knowles, J.W. Mitochondrial Dysfunction, Insulin Resistance, and Potential Genetic Implications. Endocrinology 2020, 161, bqaa017. [Google Scholar] [CrossRef]
- Uysal, S.; Armutcu, F.; Aydogan, T.; Akin, K.; Ikizek, M.; Yigitoglu, M.R. Some Inflammatory Cytokine Levels, Iron Metabolism and Oxidan Stress Markers in Subjects with Nonalcoholic Steatohepatitis. Clin. Biochem. 2011, 44, 1375–1379. [Google Scholar] [CrossRef]
- Rautou, P.-E.; Cazals-Hatem, D.; Feldmann, G.; Mansouri, A.; Grodet, A.; Barge, S.; Martinot-Peignoux, M.; Duces, A.; Bièche, I.; Lebrec, D.; et al. Changes in Autophagic Response in Patients with Chronic Hepatitis C Virus Infection. Am. J. Pathol. 2011, 178, 2708–2715. [Google Scholar] [CrossRef]
- Léveillé, M.; Estall, J.L. Mitochondrial Dysfunction in the Transition from NASH to HCC. Metabolites 2019, 9, 233. [Google Scholar] [CrossRef]
- Lebeaupin, C.; Vallée, D.; Hazari, Y.; Hetz, C.; Chevet, E.; Bailly-Maitre, B. Endoplasmic Reticulum Stress Signalling and the Pathogenesis of Non-Alcoholic Fatty Liver Disease. J. Hepatol. 2018, 69, 927–947. [Google Scholar] [CrossRef]
- Wang, J.; He, W.; Tsai, P.-J.; Chen, P.-H.; Ye, M.; Guo, J.; Su, Z. Mutual Interaction between Endoplasmic Reticulum and Mitochondria in Nonalcoholic Fatty Liver Disease. Lipids Health Dis. 2020, 19, 72. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, S.; Hwang, S.; Cherrington, N.J.; Ryu, D.-Y. Dysregulated Expression of Proteins Associated with ER Stress, Autophagy and Apoptosis in Tissues from Nonalcoholic Fatty Liver Disease. Oncotarget 2017, 8, 63370–63381. [Google Scholar] [CrossRef]
- Ma, Y.; Lee, G.; Heo, S.-Y.; Roh, Y.-S. Oxidative Stress Is a Key Modulator in the Development of Nonalcoholic Fatty Liver Disease. Antioxidants 2022, 11, 91. [Google Scholar] [CrossRef] [PubMed]
- Flessa, C.-M.; Kyrou, I.; Nasiri-Ansari, N.; Kaltsas, G.; Papavassiliou, A.G.; Kassi, E.; Randeva, H.S. Endoplasmic Reticulum Stress and Autophagy in the Pathogenesis of Non-Alcoholic Fatty Liver Disease (NAFLD): Current Evidence and Perspectives. Curr. Obes. Rep. 2021, 10, 134–161. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Lee, A.S. Role of the Unfolded Protein Response, GRP78 and GRP94 in Organ Homeostasis. J. Cell. Physiol. 2015, 230, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.-L.; Li, C.; Huang, J.-J.; Xiong, Q.; Li, Q.-Z.; Peng, B.-K.; Tang, Z. The Relationship between Endoplasmic Reticulum Stress and Liver Function, Insulin Resistance and Vascular Endothelial Function in Patients with Non-Alcoholic Fatty Liver Disease. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 11707–11715. [Google Scholar] [CrossRef]
- Wang, Q.; Zhou, H.; Bu, Q.; Wei, S.; Li, L.; Zhou, J.; Zhou, S.; Su, W.; Liu, M.; Liu, Z.; et al. Role of XBP1 in Regulating the Progression of Non-Alcoholic Steatohepatitis. J. Hepatol. 2022, 77, 312–325. [Google Scholar] [CrossRef]
- Rieusset, J.; Fauconnier, J.; Paillard, M.; Belaidi, E.; Tubbs, E.; Chauvin, M.-A.; Durand, A.; Bravard, A.; Teixeira, G.; Bartosch, B.; et al. Disruption of Calcium Transfer from ER to Mitochondria Links Alterations of Mitochondria-Associated ER Membrane Integrity to Hepatic Insulin Resistance. Diabetologia 2016, 59, 614–623. [Google Scholar] [CrossRef]
- Beaulant, A.; Dia, M.; Pillot, B.; Chauvin, M.-A.; Ji-Cao, J.; Durand, C.; Bendridi, N.; Chanon, S.; Vieille-Marchiset, A.; Da Silva, C.C.; et al. Endoplasmic Reticulum-Mitochondria Miscommunication Is an Early and Causal Trigger of Hepatic Insulin Resistance and Steatosis. J. Hepatol. 2022, 77, 710–722. [Google Scholar] [CrossRef]
- Jin, C.; Felli, E.; Lange, N.F.; Berzigotti, A.; Gracia-Sancho, J.; Dufour, J.-F. Endoplasmic Reticulum and Mitochondria Contacts Correlate with the Presence and Severity of NASH in Humans. Int. J. Mol. Sci. 2022, 23, 8348. [Google Scholar] [CrossRef]
- Palta, S.; Saroa, R.; Palta, A. Overview of the Coagulation System. Indian J. Anaesth. 2014, 58, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Ding, F.; O’Donnell, J.; Xu, Q.; Kang, N.; Goldman, N.; Nedergaard, M. Changes in the Composition of Brain Interstitial Ions Control the Sleep-Wake Cycle. Science 2016, 352, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Valenti, L.; Tripodi, A.; La Mura, V.; Pelusi, S.; Bianco, C.; Scalambrino, E.; Margarita, S.; Malvestiti, F.; Ronzoni, L.; Clerici, M.; et al. Clinical and Genetic Determinants of the Fatty Liver-Coagulation Balance Interplay in Individuals with Metabolic Dysfunction. JHEP Rep. Innov. Hepatol. 2022, 4, 100598. [Google Scholar] [CrossRef] [PubMed]
- Northup, P.G.; Caldwell, S.H. Coagulation in Liver Disease: A Guide for the Clinician. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2013, 11, 1064–1074. [Google Scholar] [CrossRef]
- Amitrano, L.; Guardascione, M.A.; Brancaccio, V.; Balzano, A. Coagulation Disorders in Liver Disease. Semin. Liver Dis. 2002, 22, 83–96. [Google Scholar] [CrossRef]
- Pant, A.; Kopec, A.K.; Luyendyk, J.P. Role of the Blood Coagulation Cascade in Hepatic Fibrosis. Am. J. Physiol. Gastrointest. Liver Physiol. 2018, 315, G171–G176. [Google Scholar] [CrossRef]
- Premkumar, M.; Sarin, S.K. Current Concepts in Coagulation Profile in Cirrhosis and Acute-on-Chronic Liver Failure. Clin. liver Dis. 2020, 16, 158–167. [Google Scholar] [CrossRef]
- Aller, R.; de Luis, D.A.; Fernandez, L.; Calle, F.; Velayos, B.; Olcoz, J.L.; Izaola, O.; Sagrado, M.G.; Conde, R.; Gonzalez, J.M. Influence of Insulin Resistance and Adipokines in the Grade of Steatosis of Nonalcoholic Fatty Liver Disease. Dig. Dis. Sci. 2008, 53, 1088–1092. [Google Scholar] [CrossRef]
- Boutari, C.; Tziomalos, K.; Athyros, V.G. The Adipokines in the Pathogenesis and Treatment of Nonalcoholic Fatty Liver Disease. Hippokratia 2016, 20, 259–263. [Google Scholar]
- Divella, R.; Mazzocca, A.; Daniele, A.; Sabbà, C.; Paradiso, A. Obesity, Nonalcoholic Fatty Liver Disease and Adipocytokines Network in Promotion of Cancer. Int. J. Biol. Sci. 2019, 15, 610–616. [Google Scholar] [CrossRef]
- Hickman, I.J.; Sullivan, C.M.; Flight, S.; Campbell, C.; Crawford, D.H.; Masci, P.P.; O’Moore-Sullivan, T.M.; Prins, J.B.; Macdonald, G.A. Altered Clot Kinetics in Patients with Non-Alcoholic Fatty Liver Disease. Ann. Hepatol. 2009, 8, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Mirza, M.S. Obesity, Visceral Fat, and NAFLD: Querying the Role of Adipokines in the Progression of Nonalcoholic Fatty Liver Disease. ISRN Gastroenterol. 2011, 2011, 592404. [Google Scholar] [CrossRef] [PubMed]
- Sookoian, S.; Pirola, C.J. Non-Alcoholic Fatty Liver Disease Is Strongly Associated with Carotid Atherosclerosis: A Systematic Review. J. Hepatol. 2008, 49, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Oni, E.T.; Agatston, A.S.; Blaha, M.J.; Fialkow, J.; Cury, R.; Sposito, A.; Erbel, R.; Blankstein, R.; Feldman, T.; Al-Mallah, M.H.; et al. A Systematic Review: Burden and Severity of Subclinical Cardiovascular Disease among Those with Nonalcoholic Fatty Liver; Should We Care? Atherosclerosis 2013, 230, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Zeina, A.-R.; Kopelman, Y.; Mari, A.; Ahmad, H.S.; Artul, S.; Khalaila, A.S.; Taher, R.; Villannueva, F.Z.; Safadi, R.; Abu Mouch, S.; et al. Pulmonary Embolism Risk in Hospitalized Patients with Nonalcoholic Fatty Liver Disease: A Case-Control Study. Medicine 2022, 101, e31710. [Google Scholar] [CrossRef]
- Yin, H.; Shi, A.; Wu, J. Platelet-Activating Factor Promotes the Development of Non-Alcoholic Fatty Liver Disease. Diabetes Metab. Syndr. Obes. 2022, 15, 2003–2030. [Google Scholar] [CrossRef]
- Nasiri-Ansari, N.; Androutsakos, T.; Flessa, C.-M.; Kyrou, I.; Siasos, G.; Randeva, H.S.; Kassi, E.; Papavassiliou, A.G. Endothelial Cell Dysfunction and Nonalcoholic Fatty Liver Disease (NAFLD): A Concise Review. Cells 2022, 11, 2511. [Google Scholar] [CrossRef]
- Guerrini, V.; Gennaro, M.L. Foam Cells: One Size Doesn’t Fit All. Trends Immunol. 2019, 40, 1163–1179. [Google Scholar] [CrossRef]
- Gui, Y.; Zheng, H.; Cao, R.Y. Foam Cells in Atherosclerosis: Novel Insights Into Its Origins, Consequences, and Molecular Mechanisms. Front. Cardiovasc. Med. 2022, 9, 845942. [Google Scholar] [CrossRef]
- Baffy, G. Kupffer Cells in Non-Alcoholic Fatty Liver Disease: The Emerging View. J. Hepatol. 2009, 51, 212–223. [Google Scholar] [CrossRef]
- Anstee, Q.M.; Darlay, R.; Cockell, S.; Meroni, M.; Govaere, O.; Tiniakos, D.; Burt, A.D.; Bedossa, P.; Palmer, J.; Liu, Y.-L.; et al. Genome-Wide Association Study of Non-Alcoholic Fatty Liver and Steatohepatitis in a Histologically Characterised Cohort (☆). J. Hepatol. 2020, 73, 505–515. [Google Scholar] [CrossRef]
- Carpino, G.; Pastori, D.; Baratta, F.; Overi, D.; Labbadia, G.; Polimeni, L.; Di Costanzo, A.; Pannitteri, G.; Carnevale, R.; Del Ben, M.; et al. PNPLA3 Variant and Portal/Periportal Histological Pattern in Patients with Biopsy-Proven Non-Alcoholic Fatty Liver Disease: A Possible Role for Oxidative Stress. Sci. Rep. 2017, 7, 15756. [Google Scholar] [CrossRef]
- Longo, M.; Meroni, M.; Paolini, E.; Erconi, V.; Carli, F.; Fortunato, F.; Ronchi, D.; Piciotti, R.; Sabatini, S.; Macchi, C.; et al. TM6SF2/PNPLA3/MBOAT7 Loss-of-Function Genetic Variants Impact on NAFLD Development and Progression Both in Patients and in In Vitro Models. Cell. Mol. Gastroenterol. Hepatol. 2022, 13, 759–788. [Google Scholar] [CrossRef] [PubMed]
- Kasper, P.; Martin, A.; Lang, S.; Kütting, F.; Goeser, T.; Demir, M.; Steffen, H.-M. NAFLD and Cardiovascular Diseases: A Clinical Review. Clin. Res. Cardiol. 2021, 110, 921–937. [Google Scholar] [CrossRef] [PubMed]
- Hassen, G.; Singh, A.; Belete, G.; Jain, N.; De la Hoz, I.; Camacho-Leon, G.P.; Dargie, N.K.; Carrera, K.G.; Alemu, T.; Jhaveri, S.; et al. Nonalcoholic Fatty Liver Disease: An Emerging Modern-Day Risk Factor for Cardiovascular Disease. Cureus 2022, 14, e25495. [Google Scholar] [CrossRef] [PubMed]
- Tripodi, A.; Anstee, Q.M.; Sogaard, K.K.; Primignani, M.; Valla, D.C. Hypercoagulability in Cirrhosis: Causes and Consequences. J. Thromb. Haemost. 2011, 9, 1713–1723. [Google Scholar] [CrossRef] [PubMed]
- McMurry, H.S.; Jou, J.; Shatzel, J. The Hemostatic and Thrombotic Complications of Liver Disease. Eur. J. Haematol. 2021, 107, 383–392. [Google Scholar] [CrossRef]
- Wanless, I.R. The Role of Vascular Injury and Congestion in the Pathogenesis of Cirrhosis: The Congestive Escalator and the Parenchymal Extinction Sequence. Curr. Hepatol. Reports 2020, 19, 40–53. [Google Scholar] [CrossRef]
- Kang, M.K.; Choi, J.H. Parenchymal Extinction Mimicking Hepatocellular Carcinoma in a Patient with Chronic Hepatitis B-Related Liver Cirrhosis. Diagnostics 2021, 11, 1171. [Google Scholar] [CrossRef]
- Wang, Q.; Zennadi, R. Oxidative Stress and Thrombosis during Aging: The Roles of Oxidative Stress in RBCs in Venous Thrombosis. Int. J. Mol. Sci. 2020, 21, 4259. [Google Scholar] [CrossRef] [PubMed]
- Bettiol, A.; Galora, S.; Argento, F.R.; Fini, E.; Emmi, G.; Mattioli, I.; Bagni, G.; Fiorillo, C.; Becatti, M. Erythrocyte Oxidative Stress and Thrombosis. Expert Rev. Mol. Med. 2022, 24, e31. [Google Scholar] [CrossRef] [PubMed]
- Violi, F.; Loffredo, L.; Carnevale, R.; Pignatelli, P.; Pastori, D. Atherothrombosis and Oxidative Stress: Mechanisms and Management in Elderly. Antioxid. Redox Signal. 2017, 27, 1083–1124. [Google Scholar] [CrossRef] [PubMed]
- Pradhan-Sundd, T.; Gudapati, S.; Kaminski, T.W.; Ragni, M.V. Exploring the Complex Role of Coagulation Factor VIII in Chronic Liver Disease. Cell. Mol. Gastroenterol. Hepatol. 2021, 12, 1061–1072. [Google Scholar] [CrossRef] [PubMed]
- Cortes, G.A.; Moore, M.J.; El-Nakeep, S. Physiology, Von Willebrand Factor; StatPearls: Treasure Island, FL, USA, 2023. [Google Scholar]
- Wiley, C.D.; Liu, S.; Limbad, C.; Zawadzka, A.M.; Beck, J.; Demaria, M.; Artwood, R.; Alimirah, F.; Lopez-Dominguez, J.-A.; Kuehnemann, C.; et al. SILAC Analysis Reveals Increased Secretion of Hemostasis-Related Factors by Senescent Cells. Cell Rep. 2019, 28, 3329–3337.e5. [Google Scholar] [CrossRef]
- Uemura, M.; Fujimura, Y.; Ko, S.; Matsumoto, M.; Nakajima, Y.; Fukui, H. Determination of ADAMTS13 and Its Clinical Significance for ADAMTS13 Supplementation Therapy to Improve the Survival of Patients with Decompensated Liver Cirrhosis. Int. J. Hepatol. 2011, 2011, 759047. [Google Scholar] [CrossRef]
- Lombardi, A.M.; Fabris, R.; Berti de Marinis, G.; Marson, P.; Navaglia, F.; Plebani, M.; Vettor, R.; Fabris, F. Defective ADAMTS13 Synthesis as a Possible Consequence of NASH in an Obese Patient with Recurrent Thrombotic Thrombocytopenic Purpura. Eur. J. Haematol. 2014, 92, 497–501. [Google Scholar] [CrossRef]
- Tripodi, A.; Fracanzani, A.L.; Primignani, M.; Chantarangkul, V.; Clerici, M.; Mannucci, P.M.; Peyvandi, F.; Bertelli, C.; Valenti, L.; Fargion, S. Procoagulant Imbalance in Patients with Non-Alcoholic Fatty Liver Disease. J. Hepatol. 2014, 61, 148–154. [Google Scholar] [CrossRef]
- Patil, A.G.; Bihari, C.; Shewade, H.D.; Nigam, N.; Sarin, S.K. Decreased Protein C Function Predicts Mortality in Patients with Cirrhosis. Int. J. Lab. Hematol. 2018, 40, 466–472. [Google Scholar] [CrossRef]
- Castellino, F.J.; Ploplis, V.A. The Protein C Pathway and Pathologic Processes. J. Thromb. Haemost. 2009, 7 (Suppl. S1), 140–145. [Google Scholar] [CrossRef]
- Sinegre, T.; Duron, C.; Lecompte, T.; Pereira, B.; Massoulier, S.; Lamblin, G.; Abergel, A.; Lebreton, A. Increased Factor VIII Plays a Significant Role in Plasma Hypercoagulability Phenotype of Patients with Cirrhosis. J. Thromb. Haemost. 2018, 16, 1132–1140. [Google Scholar] [CrossRef]
- Kumar, S.; Chandran Nair, S.; Zachariah, U.; Geevar, T.; Eapen, C.E.; Goel, A. Analysis of Coagulation Factor VIII in Liver Failure Syndromes. J. Clin. Exp. Hepatol. 2022, 12, S64. [Google Scholar] [CrossRef]
- Shen, M.-C.; Chiou, S.-S.; Chou, S.-C.; Weng, T.-F.; Lin, C.-Y.; Wang, J.-D.; Lee, S.-W.; Peng, C.-T. Prevalence of Non-Alcoholic Fatty Liver Disease and Associated Factors in Patients with Moderate or Severe Hemophilia: A Multicenter-Based Study. Clin. Appl. Thromb. Off. J. Int. Acad. Clin. Appl. Thromb. 2022, 28, 10760296221128294. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, C.; Shaw, S.; Webster, C.R.L. Coagulation in Hepatobiliary Disease. J. Vet. Emerg. Crit. Care 2011, 21, 589–604. [Google Scholar] [CrossRef] [PubMed]
- Lisman, T.; Jenne, C.N. Fibrin Fuels Fatty Liver Disease. J. Thromb. Haemost. 2018, 16, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.-N.; Yang, X.; Wu, J.-F.; Ai, W.-B.; Ni, Y.-R. Interaction of Non-parenchymal Hepatocytes in the Process of Hepatic Fibrosis (Review). Mol. Med. Rep. 2021, 23, 364. [Google Scholar] [CrossRef]
- de Andrade, K.Q.; Moura, F.A.; dos Santos, J.M.; de Araújo, O.R.P.; de Farias Santos, J.C.; Goulart, M.O.F. Oxidative Stress and Inflammation in Hepatic Diseases: Therapeutic Possibilities of N-Acetylcysteine. Int. J. Mol. Sci. 2015, 16, 30269–30308. [Google Scholar] [CrossRef] [PubMed]
- Poothong, J.; Pottekat, A.; Siirin, M.; Campos, A.R.; Paton, A.W.; Paton, J.C.; Lagunas-Acosta, J.; Chen, Z.; Swift, M.; Volkmann, N.; et al. Factor VIII Exhibits Chaperone-Dependent and Glucose-Regulated Reversible Amyloid Formation in the Endoplasmic Reticulum. Blood 2020, 135, 1899–1911. [Google Scholar] [CrossRef]
- Dalbeni, A.; Castelli, M.; Zoncapè, M.; Minuz, P.; Sacerdoti, D. Platelets in Non-Alcoholic Fatty Liver Disease. Front. Pharmacol. 2022, 13, 842636. [Google Scholar] [CrossRef]
- Henkel, A.S.; Khan, S.S.; Olivares, S.; Miyata, T.; Vaughan, D.E. Inhibition of Plasminogen Activator Inhibitor 1 Attenuates Hepatic Steatosis but Does Not Prevent Progressive Nonalcoholic Steatohepatitis in Mice. Hepatol. Commun. 2018, 2, 1479–1492. [Google Scholar] [CrossRef]
- Olivares, S.; Henkel, A.S. Endoplasmic Reticulum Stress Induces Hepatic Plasminogen Activator Inhibitor 1 in Murine Nonalcoholic Steatohepatitis. FASEB BioAdvances 2020, 2, 695–704. [Google Scholar] [CrossRef]
- Meijnikman, A.S.; Herrema, H.; Scheithauer, T.P.M.; Kroon, J.; Nieuwdorp, M.; Groen, A.K. Evaluating Causality of Cellular Senescence in Non-Alcoholic Fatty Liver Disease. JHEP Rep. Innov. Hepatol. 2021, 3, 100301. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, N. The Roles and Mechanisms of Senescence-Associated Secretory Phenotype (SASP): Can It Be Controlled by Senolysis? Inflamm. Regen. 2022, 42, 11. [Google Scholar] [CrossRef] [PubMed]
| Chronic Hepatic Diseases | Pathophysiological Traits | References |
|---|---|---|
| Hepatic steatosis |
| [22,24] |
| NAFLD |
| [18,19,22,25,26] |
| NASH |
| [25,27,28,29,30] |
| Cirrhosis |
| [26,30,31,32] |
| Chronic Hepatic Diseases | Histological Changes | Oxidative Stress | Inflammation | References |
|---|---|---|---|---|
| NAFLD |
|
|
| [35,36,51,85,86,87,88] |
| NASH |
|
|
| [37,38,87,89] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robea, M.A.; Balmus, I.-M.; Girleanu, I.; Huiban, L.; Muzica, C.; Ciobica, A.; Stanciu, C.; Cimpoesu, C.D.; Trifan, A. Coagulation Dysfunctions in Non-Alcoholic Fatty Liver Disease—Oxidative Stress and Inflammation Relevance. Medicina 2023, 59, 1614. https://doi.org/10.3390/medicina59091614
Robea MA, Balmus I-M, Girleanu I, Huiban L, Muzica C, Ciobica A, Stanciu C, Cimpoesu CD, Trifan A. Coagulation Dysfunctions in Non-Alcoholic Fatty Liver Disease—Oxidative Stress and Inflammation Relevance. Medicina. 2023; 59(9):1614. https://doi.org/10.3390/medicina59091614
Chicago/Turabian StyleRobea, Madalina Andreea, Ioana-Miruna Balmus, Irina Girleanu, Laura Huiban, Cristina Muzica, Alin Ciobica, Carol Stanciu, Carmen Diana Cimpoesu, and Anca Trifan. 2023. "Coagulation Dysfunctions in Non-Alcoholic Fatty Liver Disease—Oxidative Stress and Inflammation Relevance" Medicina 59, no. 9: 1614. https://doi.org/10.3390/medicina59091614










