Dose-Dependent Induction of Differential Seizure Phenotypes by Pilocarpine in Rats: Considerations for Translational Potential
Abstract
:1. Introduction
2. Materials and Methods
2.1. EEG Recordings
2.2. Statistical Analysis
3. Results
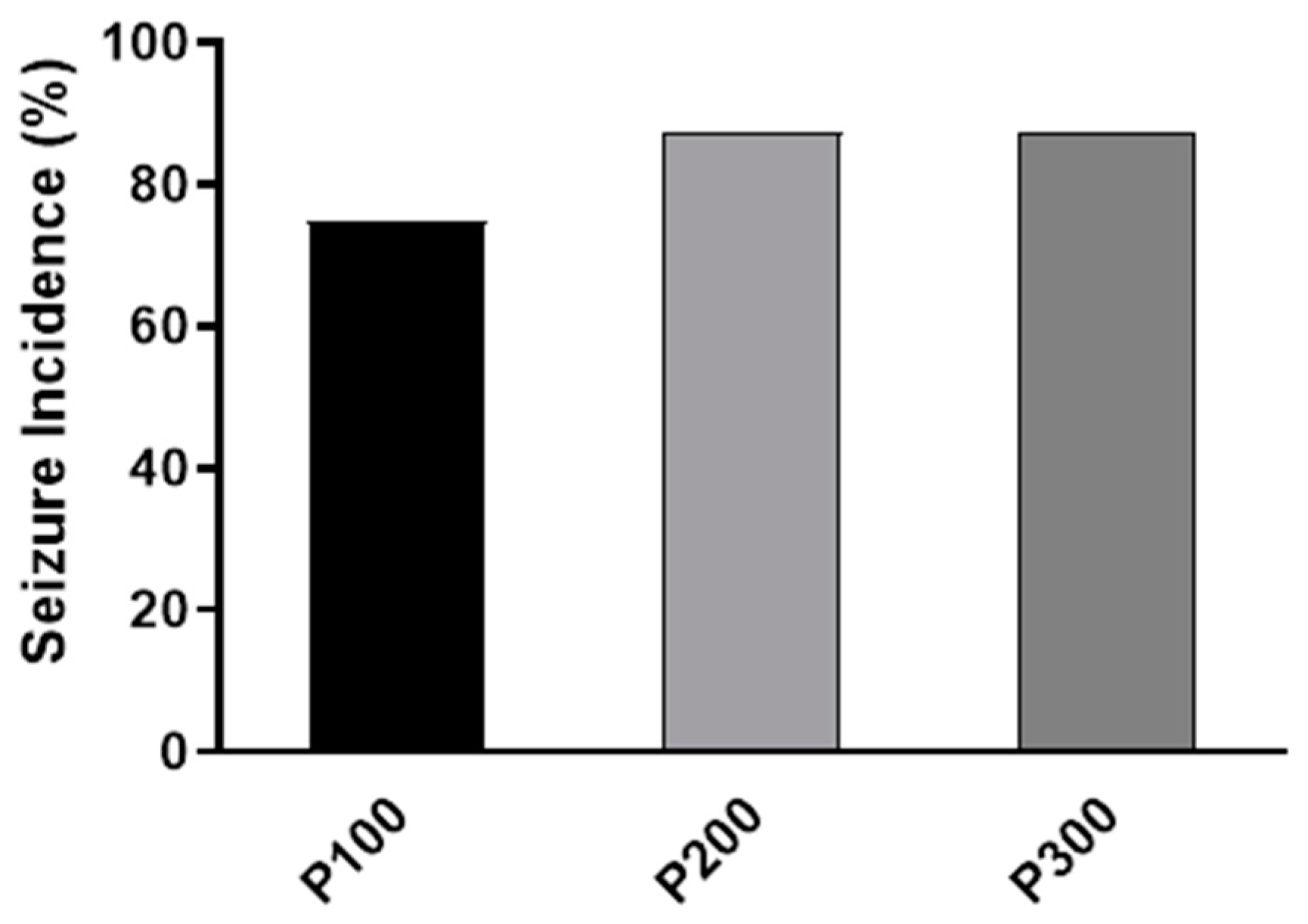
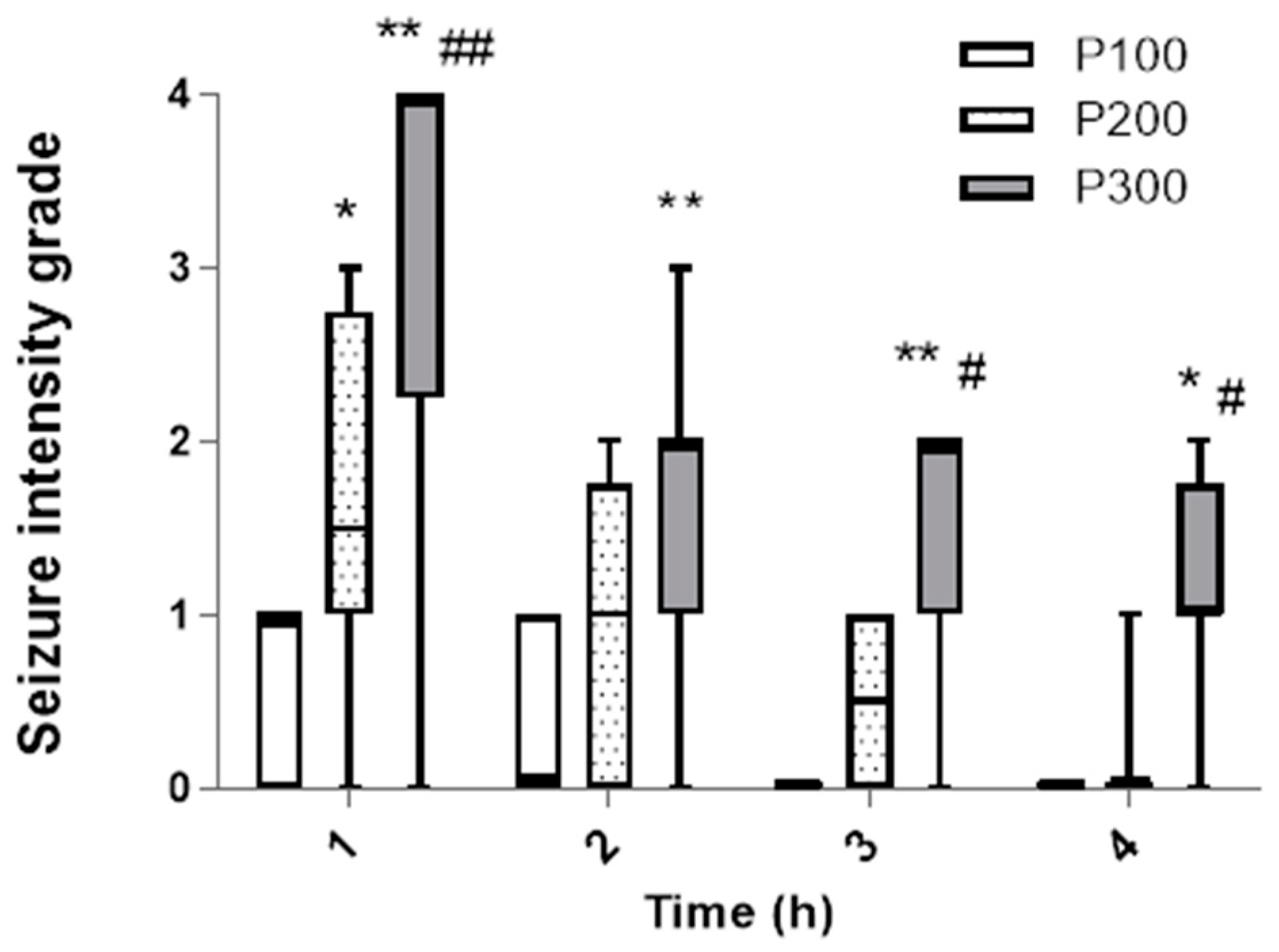
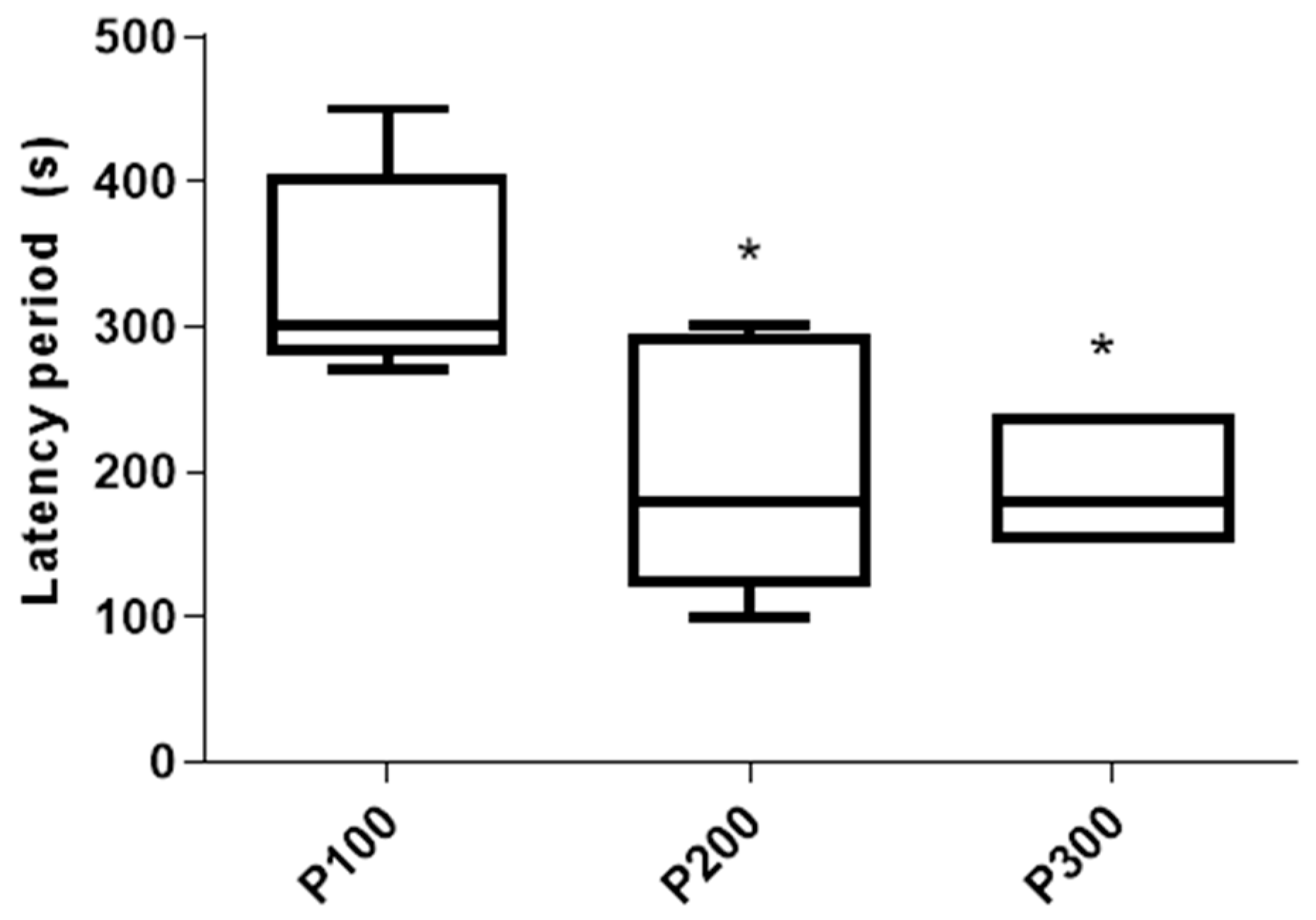
3.1. Behavioral Analysis
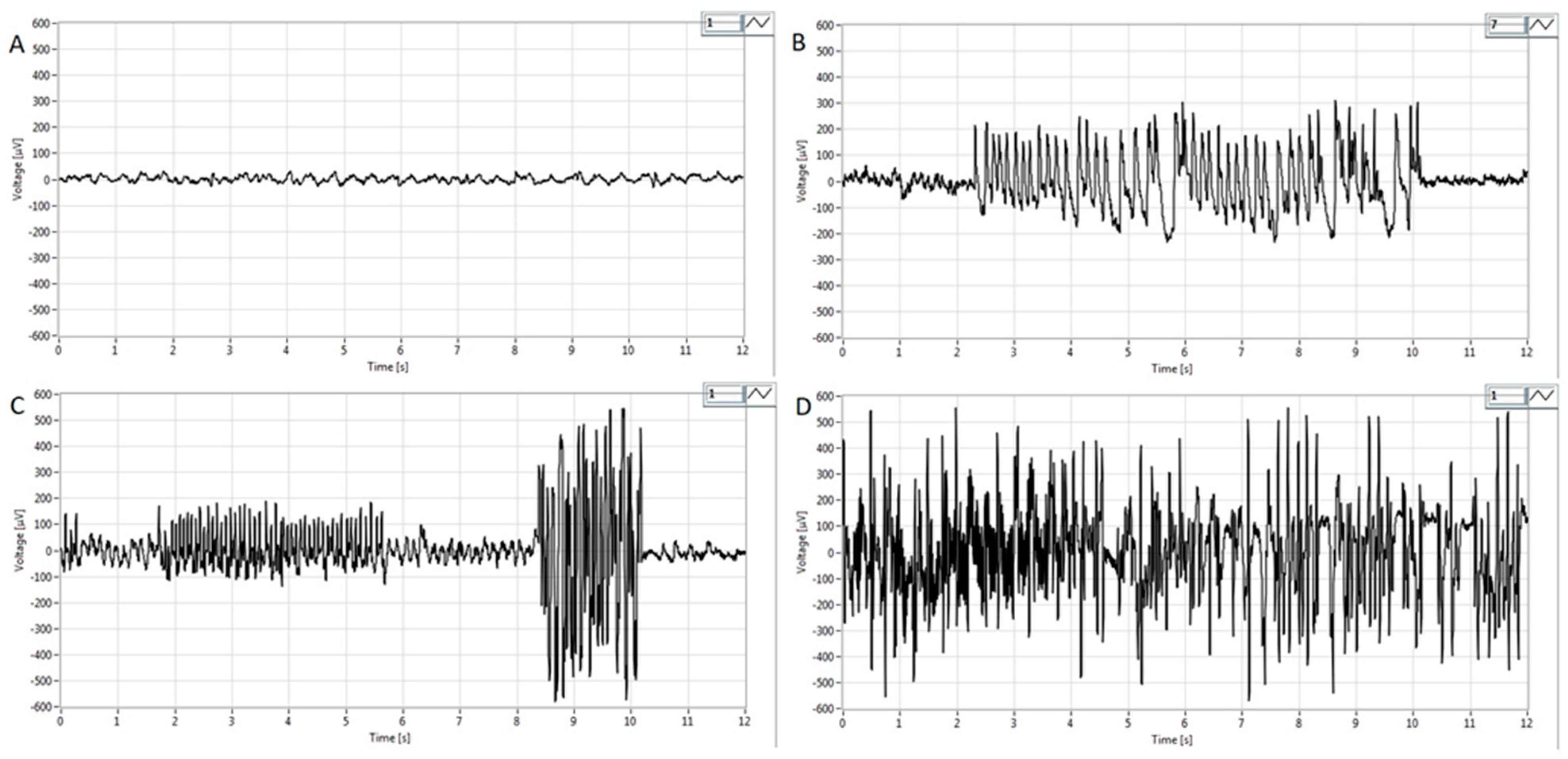
3.2. EEG Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thodeson, D.M.; Brulet, R.; Hsieh, J. Neural stem cells and epilepsy: Functional roles and disease-in-a-dish models. Cell Tissue Res. 2018, 371, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Minjarez, B.; Camarena, H.O.; Haramati, J.; Rodríguez-Yañez, Y.; Mena-Munguía, S.; Buriticá, J.; García-Leal, O. Behavioral changes in models of chemoconvulsant-induced epilepsy: A review. Neurosci. Biobehav. Rev. 2017, 83, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Puttachary, S.; Thippeswamy, T. Glial source of nitric oxide in epileptogenesis: A target for disease modification in epilepsy. J. Neurosci. Res. 2019, 97, 1363–1377. [Google Scholar] [CrossRef] [PubMed]
- Pitkänen, A.; Lukasiuk, K.; Dudek, F.E.; Staley, K.J. Epileptogenesis. Cold Spring Harb. Perspect. Med. 2015, 5, a022822. [Google Scholar] [CrossRef] [PubMed]
- Blauwblomme, T.; Jiruska, P.; Huberfeld, G. Mechanisms of ictogenesis. Int. Rev. Neurobiol. 2014, 114, 155–185. [Google Scholar]
- Aronica, E.; Bauer, S.; Bozzi, Y.; Caleo, M.; Dingledine, R.; Gorter, J.A.; Henshall, D.C.; Kaufer, D.; Koh, S.; Löscher, W.; et al. Neuroinflammatory targets and treatments for epilepsy validated in experimental models. Epilepsia 2017, 58, 27–38. [Google Scholar] [CrossRef]
- Hrncic, D.; Rasic-Markovic, A.; Macut, D.; Mladenovic, D.; Susic, V.; Djuric, D.; Stanojlovic, O. Sulfur-Containing Amino Acids in Seizures: Current State of the Art. Curr. Med. Chem. 2018, 25, 378–390. [Google Scholar] [CrossRef]
- Löscher, W. The Search for New Screening Models of Pharmacoresistant Epilepsy: Is Induction of Acute Seizures in Epileptic Rodents a Suitable Approach? Neurochem. Res. 2017, 42, 1926–1938. [Google Scholar] [CrossRef]
- Stanojlović, O.P.; Zivanović, D.P.; Mirković, S.D.; Mikhaleva, I.I. Antiepileptic activity of delta sleep-inducing peptide and its analogue in metaphit-provoked seizures in rats. Seizure 2005, 14, 240–247. [Google Scholar] [CrossRef]
- De Deyn, P.P.; D’Hooge, R.; Marescau, B.; Pei, Y.Q. Chemical models of epilepsy with some reference to their applicability in the development of anticonvulsants. Epilepsy Res. 1992, 12, 87–110. [Google Scholar] [CrossRef]
- Samokhina, E.; Samokhin, A. Neuropathological profile of the pentylenetetrazol (PTZ) kindling model. Int. J. Neurosci. 2018, 128, 1086–1096. [Google Scholar] [CrossRef] [PubMed]
- Gorter, J.A.; van Vliet, E.A.; Lopes da Silva, F.H. Which insights have we gained from the kindling and post-status epilepticus models? J. Neurosci. Methods 2016, 260, 96–108. [Google Scholar] [CrossRef]
- Lévesque, M.; Avoli, M. The kainic acid model of temporal lobe epilepsy. Neurosci. Biobehav. Rev. 2013, 37, 2887–2899. [Google Scholar] [CrossRef] [PubMed]
- Radu, B.M.; Epureanu, F.B.; Radu, M.; Fabene, P.F.; Bertini, G. Nonsteroidal anti-inflammatory drugs in clinical and experimental epilepsy. Epilepsy Res. 2017, 131, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Scorza, F.A.; Arida, R.M.; Naffah-Mazzacoratti, M.G.; Scerni, D.A.; Calderazzo, L.; Cavalheiro, E.A. The pilocarpine model of epilepsy: What have we learned? An. Acad. Bras. Cienc. 2009, 81, 345–365. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, D.; Löscher, W. Drug Resistance in Epilepsy: Putative Neurobiologic and Clinical Mechanisms. Epilepsia 2005, 46, 858–877. [Google Scholar] [CrossRef]
- Löscher, W.; White, H.S. Animal Models of Drug-Resistant Epilepsy as Tools for Deciphering the Cellular and Molecular Mechanisms of Pharmacoresistance and Discovering More Effective Treatments. Cells 2023, 12, 1233. [Google Scholar] [CrossRef]
- Turski, W.A.; Cavalheiro, E.A.; Schwarz, M.; Czuczwar, S.J.; Kleinrok, Z.; Turski, L. Limbic seizures produced by pilocarpine in rats: Behavioural, electroencephalographic and neuropathological study. Behav. Brain Res. 1983, 9, 315–335. [Google Scholar] [CrossRef]
- Nirwan, N.; Vyas, P.; Vohora, D. Animal models of status epilepticus and temporal lobe epilepsy: A narrative review. Rev. Neurosci. 2018, 29, 757–770. [Google Scholar] [CrossRef]
- Glien, M.; Brandt, C.; Potschka, H.; Löscher, W. Effects of the novel antiepileptic drug levetiracetam on sponta-neous recurrent seizures in the rat pilocarpine model of temporal lobe epilepsy. Epilepsia 2002, 43, 350–357. [Google Scholar] [CrossRef]
- Bankstahl, M.; Bankstahl, J.P.; Löscher, W. Inter-individual variation in the anticonvulsant effect of phenobarbital in the pilocarpine rat model of temporal lobe epilepsy. Exp. Neurol. 2012, 234, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Glien, M.; Brandt, C.; Potschka, H.; Voigt, H.; Ebert, U.; Löscher, W. Repeated low-dose treatment of rats with pilocarpine: Low mortality but high proportion of rats developing epilepsy. Epilepsy Res. 2001, 46, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Neumann, A.; Abele, J.; Kirschstein, T.; Engelmann, R.; Sellmann, T.; Köhling, R.; Müller-Hilke, B. Mycophenolate mofetil prevents the delayed T cell response after pilocarpine-induced status epilepticus in mice. PLoS ONE 2017, 12, e0187330. [Google Scholar] [CrossRef] [PubMed]
- Winawer, M.R.; Makarenko, N.; McCloskey, D.P.; Hintz, T.M.; Nair, N.; Palmer, A.A.; Scharfman, H.E. Acute and chronic responses to the convulsant pilocarpine in DBA/2J and A/J mice. Neuroscience 2007, 149, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Larionov, S.; Pitsch, J.; Hoerold, N.; Ullmann, C.; Elger, C.E.; Schramm, J.; Becker, A.J. Expression analysis of metabotropic glutamate receptors I and III in mouse strains with different susceptibility to experimental temporal lobe epilepsy. Neurosci. Lett. 2005, 375, 192–197. [Google Scholar] [CrossRef]
- Bankstahl, M.; Brandt, C.; Löscher, W. Differences in pilocarpine-induced epileptogenesis and behavioral alterations of Sprague-Dawley rats from two different breeders. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2009, 379, 48–49. [Google Scholar]
- Schauwecker, P.E. Strain differences in seizure-induced cell death following pilocarpine-induced status epilepticus. Neurobiol. Dis. 2012, 45, 297–304. [Google Scholar] [CrossRef]
- Ahmed, M.M.; Carrel, A.J.; Cruz Del Angel, Y.; Carlsen, J.; Thomas, A.X.; González, M.I.; Gardiner, K.J.; Brooks-Kayal, A. Altered Protein Profiles During Epileptogenesis in the Pilocarpine Mouse Model of Temporal Lobe Epilepsy. Front. Neurol. 2021, 12, 654606. [Google Scholar] [CrossRef]
- Löscher, W. Animal Models of Seizures and Epilepsy: Past Present and Future Role for the Discovery of Antiseizure Drugs. Neurochem. Res. 2017, 42, 1873–1888. [Google Scholar] [CrossRef]
- Racine, R.J. Modification of seizure activity by electrical stimulation: II. Motor seizure. Electroencephalogr. Clin. Neurophysiol. 1972, 32, 281–294. [Google Scholar] [CrossRef]
- Stanojlović, O.; Rasić-Marković, A.; Hrncić, D.; Susić, V.; Macut, D.; Radosavljević, T.; Djuric, D. Two types of seizures in homocysteinethiolactone-treated adult rats behavioral and electroencephalographic study. Cell Mol. Neurobiol. 2009, 29, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Mladenovic, D.; Hrncic, D.; Rasic-Markovic, A.; Puskas, N.; Petrovich, S.; Stanojlovic, O. Spectral analysis of thioacetamide-induced electroencephalographic changes in rats. Hum. Exp. Toxicol. 2013, 32, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Luna-Munguia, H.; Zestos, A.G.; Gliske, S.V.; Kennedy, R.T.; Stacey, W.C. Chemical biomarkers of epileptogenesis and ictogenesis in experimental epilepsy. Neurobiol. Dis. 2019, 121, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Meral, I.; Esrefoglu, M.; Dar, K.A.; Ustunova, S.; Aydin, M.S.; Demirtas, M.; Arifoglu, Y. Effects of Nigella sativa on apoptosis and GABAA receptor density in cerebral cortical and hippocampal neurons in pentylenetetrazol induced kindling in rats. Biotech. Histochem. 2016, 91, 493–500. [Google Scholar] [CrossRef]
- Lopes, M.W.; Lopes, S.C.; Costa, A.P.; Gonçalves, F.M.; Rieger, D.K.; Peres, T.V.; Eyng, H.; Prediger, R.D.; Diaz, A.P.; Nunes, J.C.; et al. Region-specific alterations of AMPA receptor phosphorylation and signaling pathways in the pilocarpine model of epilepsy. Neurochem. Int. 2015, 87, 22–33. [Google Scholar] [CrossRef]
- Löscher, W. Fit for purpose application of currently existing animal models in the discovery of novel epilepsy therapies. Epilepsy Res. 2016, 126, 157–184. [Google Scholar] [CrossRef]
- Cavarsan, C.F.; Matsuo, A.; Blanco, M.M.; Mello, L.E. Maximal electroshock-induced seizures are able to induce Homer1a mRNA expression but not pentylenetetrazole-induced seizures. Epilepsy Behav. 2015, 44, 90–95. [Google Scholar] [CrossRef]
- Kehne, J.H.; Klein, B.D.; Raeissi, S.; Sharma, S. The National Institute of Neurological Disorders and Stroke (NINDS) Epilepsy Therapy Screening Program (ETSP). Neurochem. Res. 2017, 42, 1894–1903. [Google Scholar] [CrossRef]
- Goddard, G.V.; McIntyre, D.C.; Leech, C.K. A permanent change in brain function resulting from daily electrical stimulation. Exp. Neurol. 1969, 25, 295–330. [Google Scholar] [CrossRef]
- Leite, J.P.; Cavalheiro, E.A. Effects of conventional antiepileptic drugs in a model of spontaneous recurrent seizures in rats. Epilepsy Res. 1995, 20, 93–104. [Google Scholar] [CrossRef]
- Chakir, A.; Fabene, P.; Ouazzani, R.; Bentivoglio, M. Drug resistance and hippocampal damage after delayed treatment of pilocarpine-induced epilepsy in the rat. Brain Res. Bull. 2006, 71, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Hellier, J.L.; Patrylo, P.R.; Buckmaster, P.S.; Dudek, F.E. Recurrent spontaneous motor seizures after repeated low-dose systemic treatment with kainate: Assessment of a rat model of temporal lobe epilepsy. Epilepsy Res. 1998, 31, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Turski, L.; Ikonomidou, C.; Turski, W.A.; Bortolotto, Z.A.; Cavalheiro, E.A. Review: Cholinergic mechanisms and epileptogenesis. The seizures induced by pilocarpine: A novel model of intractable epilepsy. Synapse 1989, 3, 154–171. [Google Scholar] [CrossRef] [PubMed]
- Cavalheiro, E.A.; Leite, J.P.; Bortolotto, Z.A.; Turski, W.A.; Ikonomidou, C.; Turski, L. Long-term effects of pilocarpine in rats: Structural damage of the brain triggers kindling and spontaneous recurrent seizures. Epilepsia 1991, 32, 778–782. [Google Scholar] [CrossRef]
- Drexel, M.; Preidt, A.P.; Sperk, G. Sequel of spontaneous seizures after kainic acid-induced status epilepticus and associated neuropathological changes in the subiculum and entorhinal cortex. Neuropharmacology 2012, 63, 806–817. [Google Scholar] [CrossRef]
- Riban, V.; Bouilleret, V.; Pham-Le, B.T.; Fritschy, J.M.; Marescaux, C.; Depaulis, A. Evolution of hippocampal epileptic activity during the development of hippocampal sclerosis in a mouse model of temporal lobe epilepsy. Neuroscience 2002, 112, 101–111. [Google Scholar] [CrossRef]
- Klein, S.; Bankstahl, M.; Löscher, W. Inter-individual variation in the effect of antiepileptic drugs in the intrahippocampalkainate model of mesial temporal lobe epilepsy in mice. Neuropharmacology 2015, 90, 53–62. [Google Scholar] [CrossRef]
- Duveau, V.; Pouyatos, B.; Bressand, K.; Bouyssières, C.; Chabrol, T.; Roche, Y.; Depaulis, A.; Roucard, C. Differential effects of antiepileptic drugs on focal seizures in the intrahippocampalkainate mouse model of mesial temporal lobe epilepsy. CNS Neurosci. Ther. 2016, 22, 497–506. [Google Scholar] [CrossRef]
- Stead, M.; Bower, M.; Brinkmann, B.H.; Lee, K.; Marsh, W.R.; Meyer, F.B.; Litt, B.; Van Gompel, J.; Worrell, G.A. Microseizures and the spatiotemporal scales of human partial epilepsy. Brain 2010, 133, 2789–2797. [Google Scholar] [CrossRef]
- Pitkänen, A.; Bolkvadze, T.; Immonen, R. Anti-epileptogenesis in rodent post-traumatic epilepsy models. Neurosci. Lett. 2011, 497, 163–171. [Google Scholar] [CrossRef]
- Eastman, C.L.; Verley, D.R.; Fender, J.S.; Temkin, N.R.; D’Ambrosio, R. ECoG studies of valproate, carbamazepine and halothane in frontal-lobe epilepsy induced by head injury in the rat. Exp. Neurol. 2010, 224, 369–388. [Google Scholar] [CrossRef] [PubMed]
- Eastman, C.L.; Verley, D.R.; Fender, J.S.; Stewart, T.H.; Nov, E.; Curia, G.; D’Ambrosio, R. Antiepileptic and antiepileptogenic performance of carisbamate after head injury in the rat: Blind and randomized studies. J. Pharmacol. Exp. Ther. 2011, 336, 779–790. [Google Scholar] [CrossRef] [PubMed]
- Libbey, J.E.; Fujinami, R.S. Neurotropic viral infections leading to epilepsy: Focus on Theiler’s murine encephalomyelitis virus. Future Virol. 2011, 6, 1339–1350. [Google Scholar] [CrossRef] [PubMed]
| Time (h) | Seizure Duration (s) | ||
|---|---|---|---|
| P100 | P200 | P300 | |
| 1 | 2.5 (2.1, 3.4) | 4.9 (2.3, 10.5) | 30.4 (20.1, 70.25) **# |
| 2 | 2.6 (1.9, 3.5) | 10.05 (7.6, 11.75) | 9.35 (5.975, 11.65) |
| 3 | 0 (0, 0) | 2.2 (1.6, 4.2) | 8 (5, 9.95) ** |
| 4 | 0 (0, 0) | 1.6 (1.2, 2.55) | 4.2 (2.725, 6.175) * |
| Time (h) | The Number of Seizures (n) | ||
|---|---|---|---|
| P100 | P200 | P300 | |
| 1 | 9 (1.75, 12.5) | 22.5 (17.75, 28) | 26.5 (18, 30.25) |
| 2 | 0 (0, 3.75) | 8.5 (0, 10.75) | 20 (17.25, 22.75) |
| 3 | 0 (0, 0) | 2.5 (0, 5.75) | 9.5 (4.75, 13.25) |
| 4 | 0 (0, 0) | 0 (0, 1.5) | 8.5 (6.5, 10) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasović, D.; Stanojlović, O.; Hrnčić, D.; Šutulović, N.; Vesković, M.; Ristić, A.J.; Radunović, N.; Mladenović, D. Dose-Dependent Induction of Differential Seizure Phenotypes by Pilocarpine in Rats: Considerations for Translational Potential. Medicina 2024, 60, 1579. https://doi.org/10.3390/medicina60101579
Vasović D, Stanojlović O, Hrnčić D, Šutulović N, Vesković M, Ristić AJ, Radunović N, Mladenović D. Dose-Dependent Induction of Differential Seizure Phenotypes by Pilocarpine in Rats: Considerations for Translational Potential. Medicina. 2024; 60(10):1579. https://doi.org/10.3390/medicina60101579
Chicago/Turabian StyleVasović, Dolika, Olivera Stanojlović, Dragan Hrnčić, Nikola Šutulović, Milena Vesković, Aleksandar J. Ristić, Nebojša Radunović, and Dušan Mladenović. 2024. "Dose-Dependent Induction of Differential Seizure Phenotypes by Pilocarpine in Rats: Considerations for Translational Potential" Medicina 60, no. 10: 1579. https://doi.org/10.3390/medicina60101579







