Effects of Sodium–Glucose Cotransporter 2 Inhibitors in Diabetic and Non-Diabetic Patients with Advanced Chronic Kidney Disease in Peritoneal Dialysis on Residual Kidney Function: In Real-World Data
Abstract
:1. Introduction
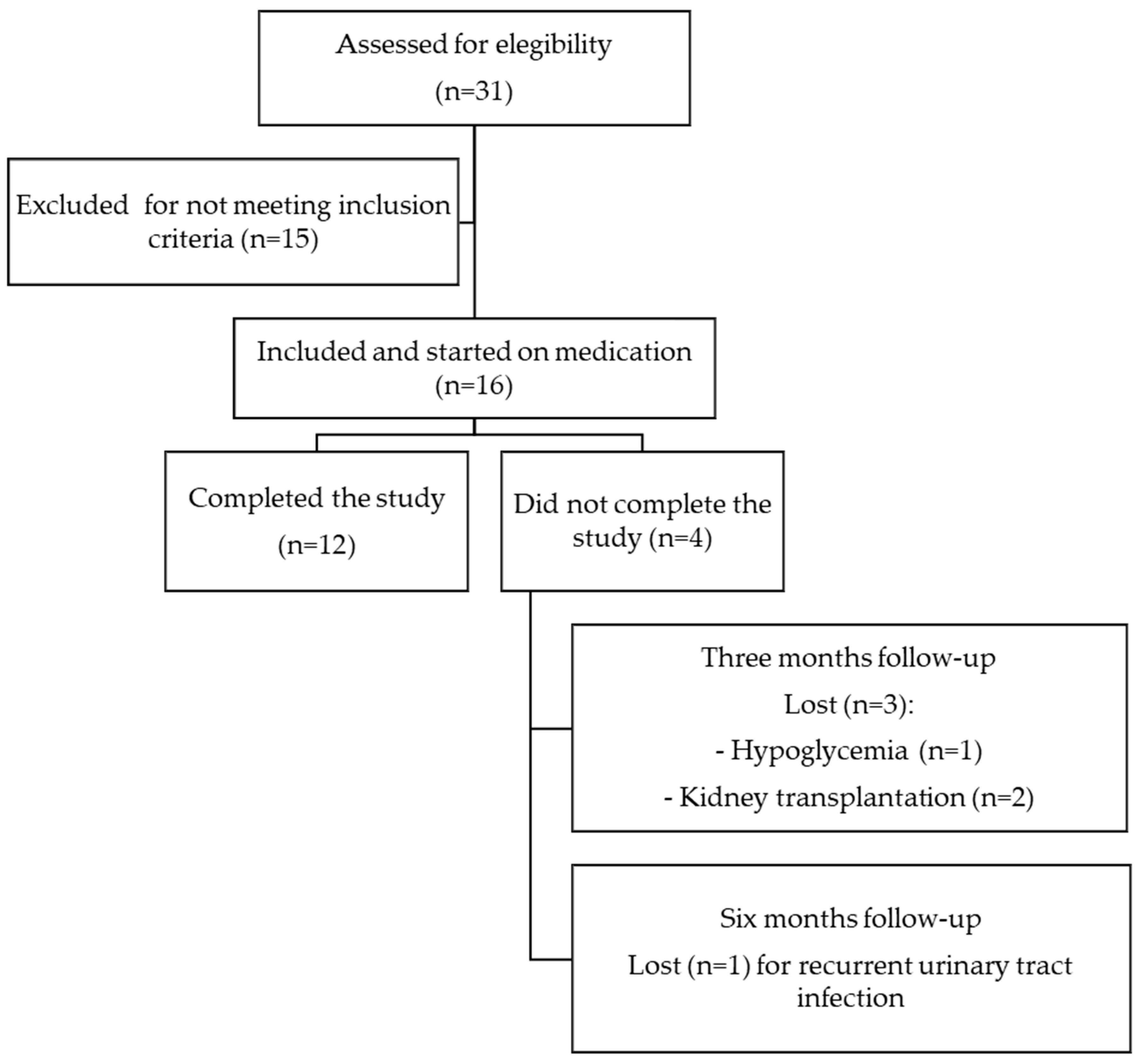
2. Materials and Methods
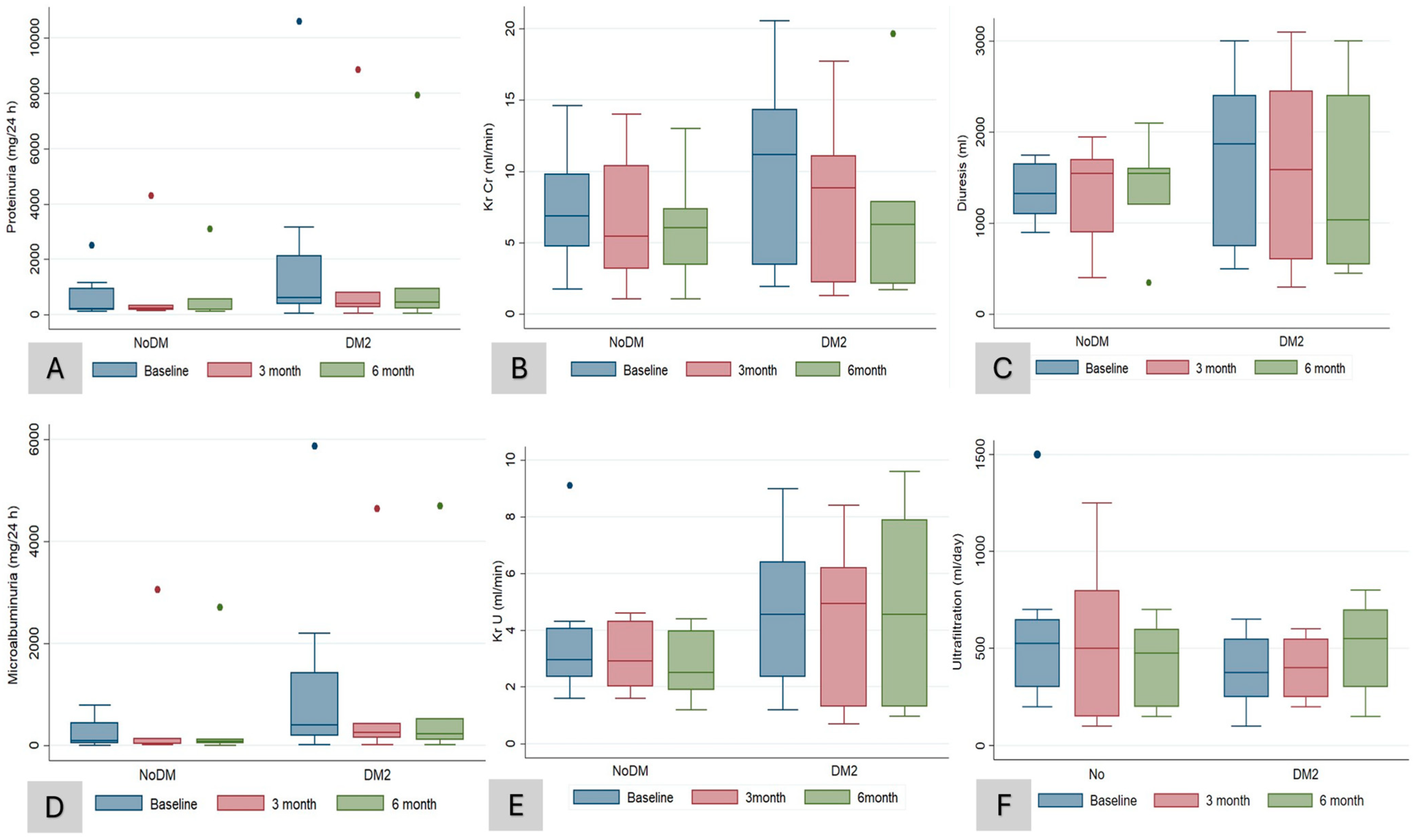
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Perkovic, V.; Jardine, M.J.; Neal, B.; Bompoint, S.; Heerspink, H.J.L.; Charytan, D.M.; Edwards, R.; Agarwal, R.; Bakris, G.; Bull, S.; et al. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. N. Engl. J. Med. 2019, 380, 2295–2306. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.L.; Stefánsson, B.V.; Correa-Rotter, R.; Chertow, G.M.; Greene, T.; Hou, F.-F.; Mann, J.F.E.; McMurray, J.J.V.; Lindberg, M.; Rossing, P.; et al. Dapagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2020, 383, 1436–1446. [Google Scholar] [CrossRef] [PubMed]
- The EMPA-KIDNEY Collaborative Group. Empagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2023, 388, 117–127. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Szarek, M.; Pitt, B.; Cannon, C.P.; Leiter, L.A.; McGuire, D.K.; Lewis, J.B.; Riddle, M.C.; Inzucchi, S.E.; Kosiborod, M.N.; et al. Sotagliflozin in Patients with Diabetes and Chronic Kidney Disease. N. Engl. J. Med. 2021, 384, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Mariani, M.V.; Manzi, G.; Pierucci, N.; Laviola, D.; Piro, A.; D’Amato, A.; Filomena, D.; Matteucci, A.; Severino, P.; Miraldi, F.; et al. SGLT2i effect on atrial fibrillation: A network meta-analysis of randomized controlled trials. J. Cardiovasc. Electrophysiol. 2024; Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Yau, K.; Dharia, A.; Alrowiyti, I.; Cherney, D.Z.I. Prescribing SGLT2 Inhibitors in Patients With CKD: Expanding Indications and Practical Considerations. Kidney Int. Rep. 2022, 7, 1463–1476. [Google Scholar] [CrossRef] [PubMed]
- Barreto, J.; Borges, C.; Rodrigues, T.B.; Jesus, D.C.; Campos-Staffico, A.M.; Nadruz, W.; da Costa, J.L.; de Oliveira, R.B.; Sposito, A.C. Pharmacokinetic Properties of Dapagliflozin in Hemodialysis and Peritoneal Dialysis Patients. Clin. J. Am. Soc. Nephrol. 2023, 18, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Bakris, G.; Oshima, M.; Mahaffey, K.W.; Agarwal, R.; Cannon, C.P.; Capuano, G.; Charytan, D.M.; De Zeeuw, D.; Edwards, R.; Greene, T.; et al. Effects of Canagliflozin in Patients with Baseline eGFR < 30 mL/min per 1.73 m2. Clin. J. Am. Soc. Nephrol. 2020, 15, 1705–1714. [Google Scholar] [CrossRef]
- Heerspink, H.L.; Wheeler, D.C.; Jong, N.; Correa-Rotter, R.; Rossing, P.; Gansevoort, R.; Mcmurray, J.; Langkilde, A.M.; Toto, R.; Chertow, G. #3382 Reasons for dialysis initiation and safety of dapagliflozin among dialysis participants: New insights from dapa-ckd. Nephrol. Dial. Transplant. 2023, 38. [Google Scholar] [CrossRef]
- Cao, H.; Rao, X.; Jia, J.; Yan, T.; Li, D. Effects of sodium-glucose co-transporter-2 inhibitors on kidney, cardiovascular, and safety outcomes in patients with advanced chronic kidney disease: A systematic review and meta-analysis of randomized controlled trials. Acta Diabetol. 2022, 60, 325–335. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024, 105, S117–S314. [Google Scholar] [CrossRef]
- Balzer, M.S.; Rong, S.; Nordlohne, J.; Zemtsovski, J.D.; Schmidt, S.; Stapel, B.; Bartosova, M.; von Vietinghoff, S.; Haller, H.; Schmitt, C.P.; et al. SGLT2 Inhibition by Intraperitoneal Dapagliflozin Mitigates Peritoneal Fibrosis and Ultrafiltration Failure in a Mouse Model of Chronic Peritoneal Exposure to High-Glucose Dialysate. Biomolecules 2020, 10, 1573. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Fan, J.; Zheng, C.; Yin, P.; Wu, H.; Li, X.; Luo, N.; Yu, X.; Chen, C. SGLT-2 inhibitors reduce glucose absorption from peritoneal dialysis solution by suppressing the activity of SGLT-2. Biomed. Pharmacother. 2019, 109, 1327–1338. [Google Scholar] [CrossRef] [PubMed]
- Marrón, B.; Remón, C.; Pérez-Fontán, M.; Quirós, P.; Ortíz, A. Benefits of preserving residual renal function in peritoneal dialysis. Kidney Int. 2008, 73, S42–S51. [Google Scholar] [CrossRef] [PubMed]
- Borkum, M.; Jamal, A.; Singh, R.S.; Levin, A. The rationale for the need to study sodium-glucose co-transport 2 inhibitor usage in peritoneal dialysis patients. Perit. Dial. Int. J. Int. Soc. Perit. Dial. 2023, 43, 139–144. [Google Scholar] [CrossRef]
- Alhwiesh, A.K.; Sarah Al-Wa, I.; Abdul-Rahman, S.; Ahmad Nasreldin, M.; Moaz Mohammed, A.; Al-Oudah, S.; Al-Thwainy, R. The use of SGLT2 Inhibitors in Peritoneal Dialysis Patients: A Shade of Light on Dapagliflozin. Arch Nephrol. Urol. 2022, 5, 1–8. [Google Scholar]
- Lai, J.-W.; Lin, H.-J.; Chou, C.-Y. SGLT-2 inhibitors may increase ultrafiltration in incident peritoneal dialysis patients: A case report. BMC Nephrol. 2023, 24, 1–4. [Google Scholar] [CrossRef]
- Hamdan, Z.; Abdel-Hafez, Y.; Enaya, A.; Sarsour, A.; Kharraz, L.; Nazzal, Z. Dapagliflozin in peritoneal dialysis patients: A pilot study evaluating peritoneal membrane function. BMC Nephrol. 2024, 25, 1–8. [Google Scholar] [CrossRef]
- Bargman, J.M.; Thorpe, K.E.; Churchill, D.N. Relative Contribution of Residual Renal Function and Peritoneal Clearance to Adequacy of Dialysis: A Reanalysis of the CANUSA Study. J. Am. Soc. Nephrol. 2001, 12, 2158–2162. [Google Scholar] [CrossRef]
- Li, P.K.T.; Chow, K.M.; Wong, T.Y.H.; Leung, C.B.; Szeto, C.C. Effects of an Angiotensin-Converting Enzyme Inhibitor on Residual Renal Function in Patients Receiving Peritoneal Dialysis: A Randomized, Controlled Study. Ann. Intern. Med. 2003, 139, 105. [Google Scholar] [CrossRef]
- Suzuki, H.; Kanno, Y.; Sugahara, S.; Okada, H.; Nakamoto, H. Effects of an angiotensin II receptor blocker, valsartan, on residual renal function in patients on CAPD. Am. J. Kidney Dis. 2004, 43, 1056–1064. [Google Scholar] [CrossRef] [PubMed]
- Del Vecchio, L.; Beretta, A.; Jovane, C.; Peiti, S.; Genovesi, S. A Role for SGLT-2 Inhibitors in Treating Non-diabetic Chronic Kidney Disease. Drugs 2021, 81, 1491–1511. [Google Scholar] [CrossRef] [PubMed]
- Hodrea, J.; Balogh, D.B.; Hosszu, A.; Lenart, L.; Besztercei, B.; Koszegi, S.; Sparding, N.; Genovese, F.; Wagner, L.J.; Szabo, A.J.; et al. Reduced O-GlcNAcylation and tubular hypoxia contribute to the antifibrotic effect of SGLT2 inhibitor dapagliflozin in the diabetic kidney. Am. J. Physiol. Ren. Physiol. 2020, 318, F1017-29. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Nakano, D.; Guan, Y.; Hitomi, H.; Uemura, A.; Masaki, T.; Kobara, H.; Sugaya, T.; Nishiyama, A. A sodium-glucose cotransporter 2 inhibitor attenuates renal capillary injury and fibrosis by a vascular endothelial growth factor–dependent pathway after renal injury in mice. Kidney Int. 2018, 94, 524–535. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Zhao, Y.; Wang, Q.; Hillebrands, J.L.; Born, J.V.D.; Ji, L.; An, T.; Qin, G. Dapagliflozin Attenuates Renal Tubulointerstitial Fibrosis Associated With Type 1 Diabetes by Regulating STAT1/TGFβ1 Signaling. Front. Endocrinol. 2019, 3, 441. [Google Scholar] [CrossRef] [PubMed]
- Castoldi, G.; Carletti, R.; Ippolito, S.; Colzani, M.; Barzaghi, F.; Stella, A.; Zerbini, G.; Perseghin, G.; di Gioia, C.R. Renal Anti-Fibrotic Effect of Sodium Glucose Cotransporter 2 Inhibition in Angiotensin II-Dependent Hypertension. Am. J. Nephrol. 2020, 51, 119–129. [Google Scholar] [CrossRef] [PubMed]
- De La Flor, J.C.; Villa, D.; Cruzado, L.; Apaza, J.; Valga, F.; Zamora, R.; Marschall, A.; Cieza, M.; Deira, J.; Rodeles, M. Efficacy and Safety of the Use of SGLT2 Inhibitors in Patients on Incremental Hemodialysis: Maximizing Residual Renal Function, Is There a Role for SGLT2 Inhibitors? Biomedicines 2023, 11, 1908. [Google Scholar] [CrossRef] [PubMed]
- Ethier, I.; Cho, Y.; Hawley, C.; Pascoe, E.M.; Viecelli, A.K.; Campbell, S.B.; van Eps, C.; Isbel, N.M.; Cooper, B.A.; Harris, D.C.; et al. Rate of decline in residual kidney function pre and post peritoneal dialysis initiation: A post hoc analysis of the IDEAL study. PLoS ONE 2020, 15, e0242254. [Google Scholar] [CrossRef] [PubMed]
- Yen, F.-S.; Hwu, C.-M.; Liu, J.-S.; Wu, Y.-L.; Chong, K.; Hsu, C.-C. Sodium–Glucose Cotransporter-2 Inhibitors and the Risk for Dialysis and Cardiovascular Disease in Patients With Stage 5 Chronic Kidney Disease. Ann. Intern. Med. 2024, 177, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Medcalf, J.F.; Harris, K.P.G.; Walls, J. Role of diuretics in the preservation of residual renal function in patients on continuous ambulatory peritoneal dialysis. Kidney Int. 2001, 59, 1128–1133. [Google Scholar] [CrossRef]
- Thomas, M.C.; Cherney, D.Z.I. The actions of SGLT2 inhibitors on metabolism, renal function and blood pressure. Diabetologia 2018, 61, 2098–2107. [Google Scholar] [CrossRef] [PubMed]
- Zaccardi, F.; Webb, D.R.; Htike, Z.Z.; Youssef, D.; Khunti, K.; Davies, M.J. Efficacy and safety of sodium-glucose co-transporter-2 inhibitors in type 2 diabetes mellitus: Systematic review and network meta-analysis. Diabetes Obes. Metab. 2016, 18, 783–794. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Fu, Q.; Zhou, L.; Fan, Y.; Liu, F.; Fan, Y.; Zhang, X.; Lin, W.; Wu, X. Effect of SGLT-2 inhibitor, empagliflozin, on blood pressure reduction in Chinese elderly hypertension patients with type 2 diabetes and its possible mechanisms. Sci. Rep. 2022, 12, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Krämer, B.K.; Hauske, S.J.; Chilton, R.; Mann, J.F.; Gullestad, L.; Fitchett, D.; Mattheus, M.; Steubl, D.; Wanner, C. Changes in cardiac and vascular haemodynamics as potential mediators of improvements in cardiovascular and kidney outcomes with empagliflozin in type 2 diabetes. J. Diabetes Its Complicat. 2023, 37, 108588. [Google Scholar] [CrossRef] [PubMed]
- Zanchi, A.; Burnier, M.; Muller, M.; Ghajarzadeh-Wurzner, A.; Maillard, M.; Loncle, N.; Milani, B.; Dufour, N.; Bonny, O.; Pruijm, M. Acute and Chronic Effects of SGLT2 Inhibitor Empagliflozin on Renal Oxygenation and Blood Pressure Control in Nondiabetic Normotensive Subjects: A Randomized, Placebo-Controlled Trial. J. Am. Hearth Assoc. 2020, 9, e016173. [Google Scholar] [CrossRef] [PubMed]
- Zanchi, A.; Pruijm, M.; Muller, M.-E.; Ghajarzadeh-Wurzner, A.; Maillard, M.; Dufour, N.; Bonny, O.; Wuerzner, G.; Burnier, M. Twenty-Four Hour Blood Pressure Response to Empagliflozin and Its Determinants in Normotensive Non-diabetic Subjects. Front. Cardiovasc. Med. 2022, 9, 854230. [Google Scholar] [CrossRef] [PubMed]
- Stepanova, N. SGLT2 inhibitors in peritoneal dialysis: A promising frontier toward improved patient outcomes. Ren. Replace. Ther. 2024, 10, 5. [Google Scholar] [CrossRef]
- Cherney, D.Z.; Cooper, M.E.; Tikkanen, I.; Pfarr, E.; Johansen, O.E.; Woerle, H.J.; Broedl, U.C.; Lund, S.S. Pooled analysis of Phase III trials indicate contrasting influences of renal function on blood pressure, body weight, and HbA1c reductions with empagliflozin. Kidney Int. 2018, 93, 231–244. [Google Scholar] [CrossRef] [PubMed]
- Osonoi, T.; Shirabe, S.; Saito, M.; Hosoya, M.; Watahiki, N.; Douguchi, S.; Ofuchi, K.; Katoh, M. Dapagliflozin Improves Erythropoiesis and Iron Metabolism in Type 2 Diabetic Patients with Renal Anemia. Diabetes Metab. Syndr. Obes. Targets Ther. 2023, 16, 1799–1808. [Google Scholar] [CrossRef]
- Koshino, A.; Schechter, M.; Chertow, G.M.; Vart, P.; Jongs, N.; Toto, R.D.; Rossing, P.; Correa-Rotter, R.; McMurray, J.J.; Górriz, J.L.; et al. Dapagliflozin and Anemia in Patients with Chronic Kidney Disease. NEJM Evid. 2023, 2, EVIDoa2300049. [Google Scholar] [CrossRef]
- Fuchs Andersen, C.; Omar, M.; Glenthøj, A.; El Fassi, D.; Møller, H.J.; Lindholm Kurtzhals, J.A.; Styrishave, B.; Kistorp, C.; Tuxen, C.; Poulsen, M.K.; et al. Effects of empagliflozin on erythropoiesis in heart failure: Data from the Empire HF trial. Eur. J. Heart Fail. 2023, 25, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Cases, A.; Cigarrán, S.; Górriz, J.L.; Nuñez, J. Efecto de los inhibidores del cotransportador sodio-glucosa tipo 2 sobre la anemia: Posibles implicaciones clínicas. Nefrología 2024, 44, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Sano, M.; Goto, S. Possible Mechanism of Hematocrit Elevation by Sodium Glucose Cotransporter 2 Inhibitors and Associated Beneficial Renal and Cardiovascular Effects. Circulation 2019, 139, 1985–1987. [Google Scholar] [CrossRef] [PubMed]
- Packer, M. Critical Reanalysis of the Mechanisms Underlying the Cardiorenal Benefits of SGLT2 Inhibitors and Reaffirmation of the Nutrient Deprivation Signaling/Autophagy Hypothesis. Circulation 2022, 146, 1383–1405. [Google Scholar] [CrossRef]
- Mayne, K.J.; Staplin, N.; Keane, D.F.; Wanner, C.; Brenner, S.; Cejka, V.; Stegbauer, J.; Judge, P.K.; Preiss, D.; Emberson, J.; et al. Effects of Empagliflozin on Fluid Overload, Weight and Blood Pressure in Chronic Kidney Disease. J. Am. Soc. Nephrol. 2024, 35, 202–215. [Google Scholar] [CrossRef]
- Tang, J.; Ye, L.; Yan, Q.; Zhang, X.; Wang, L. Effects of Sodium-Glucose Cotransporter 2 Inhibitors on Water and Sodium Metabolism. Front. Pharmacol. 2022, 13, 800490. [Google Scholar] [CrossRef] [PubMed]
- Albakr, R.B.; Sridhar, V.S.; Cherney, D.Z. Novel Therapies in Diabetic Kidney Disease and Risk of Hyperkalemia: A Review of the Evidence From Clinical Trials. Am. J. Kidney Dis. 2023, 82, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Gabai, P.; Fouque, D. SGLT2 inhibitors: New kids on the block to control hyperkalemia. Nephrol. Dial. Transplant. 2023, 38, 1345–1348. [Google Scholar] [CrossRef]
- Neuen, B.L.; Oshima, M.; Agarwal, R.; Arnott, C.; Cherney, D.Z.; Edwards, R.; Langkilde, A.M.; Mahaffey, K.W.; McGuire, D.K.; Neal, B.; et al. Sodium-Glucose Cotransporter 2 Inhibitors and Risk of Hyperkalemia in People With Type 2 Diabetes: A Meta-Analysis of Individual Participant Data From Randomized, Controlled Trials. Circulation 2022, 145, 1460–1470. [Google Scholar] [CrossRef]
- Rau, M.; Thiele, K.; Hartmann, N.-U.K.; Möllmann, J.; Wied, S.; Hohl, M.; Marx, N.; Lehrke, M. Effects of empagliflozin on markers of calcium and phosphate homeostasis in patients with type 2 diabetes—Data from a randomized, placebo-controlled study. Bone Rep. 2022, 16, 101175. [Google Scholar] [CrossRef]
- Goicoechea, M.; de Vinuesa, S.G.; Verdalles, U.; Ruiz-Caro, C.; Ampuero, J.; Rincón, A.; Arroyo, D.; Luño, J. Effect of Allopurinol in Chronic Kidney Disease Progression and Cardiovascular Risk. Clin. J. Am. Soc. Nephrol. 2010, 5, 1388–1393. [Google Scholar] [CrossRef] [PubMed]
- Shiau, C.-H.; Tsau, L.-Y.; Kao, C.-C.; Peng, Y.-C.; Bai, C.-H.; Wu, J.; Hou, W.-H. Efficacy and safety of sodium-glucose cotransporter-2 inhibitors in patients with chronic kidney disease: A systematic review and meta-analysis. Int. Urol. Nephrol. 2023, 56, 1359–1381. [Google Scholar] [CrossRef] [PubMed]
- Karimzadeh, I.; Barreto, E.F.; Kellum, J.A.; Awdishu, L.; Murray, P.T.; Ostermann, M.; Bihorac, A.; Mehta, R.L.; Goldstein, S.L.; Kashani, K.B.; et al. Moving toward a contemporary classification of drug-induced kidney disease. Crit. Care 2023, 27, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Sampani, E.; Sarafidis, P.; Dimitriadis, C.; Kasimatis, E.; Daikidou, D.; Bantis, K.; Papanikolaou, A.; Papagianni, A. Severe euglycemic diabetic ketoacidosis of multifactorial etiology in a type 2 diabetic patient treated with empagliflozin: Case report and literature review. BMC Nephrol. 2020, 21, 276. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Luo, D.; Luo, N.; Li, B.; Fu, D.; Fan, L.; Li, Z.; Chen, W.; Mao, H. Serum Hepcidin-25 and Risk of Mortality in Patients on Peritoneal Dialysis. Front. Med. 2021, 8, 684548. [Google Scholar] [CrossRef] [PubMed]
- Schröppel, B.; Fischereder, M.; Wiese, P.; Segerer, S.; Huber, S.; Kretzler, M.; Heiss, P.; Sitter, T.; Schlöndorff, D. Expression of glucose transporters in human peritoneal mesothelial cells. Kidney Int. 1998, 53, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Schricker, S.; Oberacker, T.; Fritz, P.; Ketteler, M.; Alscher, M.D.; Schanz, M. Peritoneal Expression of SGLT-2, GLUT1, and GLUT3 in Peritoneal Dialysis Patients. Kidney Blood Press. Res. 2022, 47, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Martus, G.; Bergling, K.; de Arteaga, J.; Öberg, C.M. SGLT2 inhibition does not reduce glucose absorption during experimental peritoneal dialysis. Perit. Dial. Int. J. Int. Soc. Perit. Dial. 2021, 41, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Shentu, Y.; Li, Y.; Xie, S.; Jiang, H.; Sun, S.; Lin, R.; Chen, C.; Bai, Y.; Zhang, Y.; Zheng, C.; et al. Empagliflozin, a sodium glucose cotransporter-2 inhibitor, ameliorates peritoneal fibrosis via suppressing TGF-β/Smad signaling. Int. Immunopharmacol. 2021, 93, 107374. [Google Scholar] [CrossRef]
- Wang, J.; Lv, X.; A-Ni-Wan, A.S.J.; Tian, S.S.; Wang, J.M.; Liu, H.Y.; Fan, X.G.; Zhou, S.J.; Yu, P. Canagliflozin alleviates high glucose-induced peritoneal fibrosis via HIF-1α inhibition. Front Pharmacol. 2023, 14, 1152611. [Google Scholar] [CrossRef]
| All (N = 16) | NoDM | DM | p-Value | |
|---|---|---|---|---|
| (N = 8) | (N = 8) | |||
| Age—years, mean (SD) | 67.3 (10.3) | 64.5 (9.9) | 70 (10.6) | 0.2 |
| Sex—female, n (%) | 6 (37.5) | 4 (50) | 2 (25) | 0.3 |
| Dry weight (Kg), mean (SD) | 75.7 (18.3) | 70.4 (21.3) | 81.1 (14.1) | 0.3 |
| BMI (kg/m2), mean (SD) | 28.4 (6.1) | 25.73 (6.31) | 31.1 (4.9) | 0.07 |
| Causes of CKD, n (%) | 0.1 | |||
| Unknown origin | 7 (43.8) | 2 (25) | 5 (62.5) | |
| Glomerular disease | 5 (31.3) | 2 (25) | 3 (37.5) | |
| CTIN | 1 (6.3) | 1 (12.5) | 0 | |
| ADPKD | 3 (18.8) | 3 (37.5) | 0 | |
| HbA1c level, n (%) | 6.0 (0.9) | 5.4 (0.6) | 6.6 (0.8) | <0.001 |
| Hypertension, n (%) | 16 (100) | 16 (100) | 16 (100) | |
| ACE inhibitors/ARB | 16 (100) | 8 (100) | 8 (100) | |
| ACC | 6 (37.5) | 3 (37.5) | 3 (37.5) | |
| Loop diuretics | 13 (81.3) | 6 (75) | 7 (87.5) | |
| MRA | 9 (56.3) | 1 (12.5) | 8 (100) | |
| Thiazides | 6 (37.5) | 4 (50) | 2 (25) | |
| Alpha blockers | 5 (31.3) | 2 (25) | 3 (37.5) | |
| Beta blockers | 7 (43.8) | 4 (50) | 3 (37.55) | |
| SGLT-2 inhibitors, n (%) | 0.5 | |||
| Empagliflozin | 13 (81.2) | 7 (87.5) | 6 (75) | |
| Dapagliflozin | 3 (18.8) | 1 (12.5) | 2 (25) | |
| PD vintage (months), mean (SD) | 21.1 (15.0) | 13.1 (8.1) | 29.1 (16.5) | 0.03 |
| Technical peritoneal dialysis, n (%) | ||||
| APD | 4 (25) | 3 (37.5) | 1 (12.5) | 0.2 |
| CAPD | 12 (75) | 5 (62.5) | 7 (87.5) | |
| Technical prescription, n (%) | ||||
| Icodextrin | 10 (62.5) | 5 (62.5) | 5 (62.5) | 1 |
| Peritoneal equilibrium test: | ||||
| D/P, mean (SD) | 0.76 (0.12) | 0.78 (0.11) | 0.74 (0.13) | 0.9 |
| D/D0, mean (SD) | 0.29 (0.1) | 0.29 (0.09) | 0.29 (0.09) | |
| Peritoneal membrane function classification: | 0.4 | |||
| Low transporter, number (%) | 1 (6.3) | 0 | 1 (12.5) | |
| Average transporter, number (%) | 11 (68.8) | 5 (62.5) | 6 (75) | |
| High transporter, number (%) | 4 (25.0) | 3 (37.5) | 1 (12.5) | |
| Ultrafiltration volume (mL/day), median [IQR] | 425 [250–600] | 525 [300–650] | 375 [250–550] | 0.3 |
| nPCR, mean (SD) | 0.94 (0.16) | 0.94 (0.16) | 0.94 (0.18) | 0.9 |
| Residual diuresis (L), mean (SD) | 1.5 (0.7) | 1.35 Lt (0.315) | 1.69 (0.9) | 0.3 |
| KrU (SD), mL/min, mean (SD) | 4.1 (2.5) | 3.67 (2.36) | 4.6 (2.8) | 0.5 |
| ClCr (SD), mL/min, mean (SD) | 7.45 (4.2) | 10.05 (6.6) | ||
| Kt/V week, mean (SD) | 2.1 (0.4) | 2.1 (0.2) | 2.1(0.6) | 0.9 |
| 24 h proteinuria (g/day), median [IQR] | 489.7 [192.6–1128] | 229 [172.8–955.3] | 622.9 [398.8–2130] | 0.3 |
| MAU 24 h urine (mg/day), median [IQR] | 283 [65.2–553] | 105.6 [47.6–450.0] | 408 [188.7–1414.8] | 0.2 |
| SBP (mmHg), mean (SD) | 139.8 (10.2) | 141 (9.9) | 138.6 (11.0) | 0.7 |
| DBP (mmHg), mean (SD) | 71.4 (8.1) | 73.6 (7.4) | 69.3 (8.6) | 0.3 |
| Bicarbonate, mean (SD) | 22.1 (2.1) | 22.0 (1.5) | 22.1 (2.7) | 0.9 |
| Baseline | 3 Months | 6 Months | p-Value ANOVA | |
|---|---|---|---|---|
| N | 16 | 13 | 12 | |
| Weight (kg), mean (SD) | 75.7 (18.3) | 73.6 (16.9) | 72.8 (16.7) | 0.2 |
| Hb (g/dL), mean (SD) | 12.2 (1.5) | 11.6 (1.6) | 11.8 (0.8) | 0.5 |
| Cr (g/dL), mean (SD) | 6.6 (2.3) | 7.2 (2) | 7.1 (1.9) | 0.9 |
| CKD-EPI 2021 (mL/min/1.73m2), mean (SD) | 8.3 (3.6) | 7.2 (2.9) | 7.5 (3.6) | 0.8 |
| Urea (mmol/mL), mean (SD) | 139.4 (32.9) | 135.8 (28) | 131.8 (21.2) | 0.2 |
| Sodium (mmol/L), mean (SD) | 137.1 (3) | 136.4 (3.2) | 137.1 (1.9) | 0.7 |
| Potassium (mmol/L), mean (SD) | 4.8 (0.7) | 4.4 (0.5) | 4.6 (0.6) | 0.1 |
| Chlorine (mmol/L), mean (SD) | 100.2 (5) | 99 (4.2) | 99.6 (3.3) | 0.8 |
| Calcium (mg/dL), mean (SD) | 9.3 (0.8) | 9 (0.4) | 9.2 (0.5) | 0.3 |
| Phosphorus (mg/dL), mean (SD) | 4.8 (1.2) | 4.5 (1.1) | 4.7 (1.1) | 0.6 |
| Magnesium (mg/dL), mean (SD) | 2 (0.5) | 2.2 (0.5) | 2.2 (0.5) | 0.7 |
| HbA1c (%), mean (SD) | 6 (0.9) | 6 (0.7) | 5.9 (0.6) | 0.8 |
| Uric acid (mg/dL), mean (SD) | 5.2 (1.4) | 5.1 (1.2) | 5.4 (1.3) | 0.5 |
| Cholesterol (mg/dL), mean (SD) | 143.9 (29.5) | 148.8 (47.1) | 150.2 (47.8) | 0.9 |
| Triglycerides (mg/dL), mean (SD) | 152.3 (81.5) | 163.5 (80) | 193.6 (213.9) | 0.7 |
| HDL (mg/dL), mean (SD) | 50.1 (15.8) | 46.9 (14.5) | 43.5 (17.8) | 0.5 |
| LDL (mg/dL), mean (SD) | 71.8 (17.2) | 70.2 (40.3) | 64.3 (27.2) | 0.6 |
| Bilirubin (mg/dL), mean (SD) | 0.4 (0.2) | 0.4 (0.1) | 0.4 (0.2) | 0.6 |
| AST (IU/L), mean (SD) | 20.9 (11.7) | 23.4 (22.1) | 20.5 (15.7) | 0.5 |
| ALT (IU/L), mean (SD) | 24 (15.8) | 27.1 (27.3) | 20.5 (12.4) | 0.3 |
| GGT (IU/L), mean (SD) | 28.2 (21.7) | 43.7 (55.3) | 36.7 (35.5) | 0.2 |
| KT/V weekly (Lt), mean (SD) | 2.1 (0.4) | 2.1 (0.4) | 2 (0.4) | 0.5 |
| nPCR (g Urea/Kg/d), mean (SD) | 0.9 (0.2) | 0.8 (0.1) | 0.8 (0.1) | 0.08 |
| Ultrafiltration PD (mL/day), median [IQR] | 425 [250–600] | 500 [250–550] | 475 [250–700] | 0.8 |
| KrU (mL/min), mean (SD) | 4.1 (2.5) | 3.7 (2.2) | 3.8 (2.7) | 0.5 |
| Diuresis (mL/day), mean (SD) | 1521.9 (698.8) | 1448.5 (837.8) | 1402.5 (810) | 0.8 |
| ClCr (mL/min), mean (SD) | 8.7 (5.5) | 7.5 (5.2) | 6.7 (5.2) | 0.2 |
| (ClCr + KrU/2) (mL/min), mean (SD) | 6.4 (3.6) | 5.6 (3.6) | 5.3 (3.8) | 0.2 |
| MAU 24 h (mg/24 h), median [IQR] | 283 [65.2–553] | 139.5 [42.3–300] | 114.6 [56.9–428.4] | 0.9 |
| Proteinuria (mg/24 h), median [IQR] | 489.7 [192.6–1128] | 257.4 [180–500] | 328.6 [179–765.4] | 0.5 |
| SBP (mmHg), mean (SD) | 139.8 (10.2) | 129.5 (5.2) | 129.7 (7.0) | 0.003 |
| DBP (mmHg), mean (SD) | 71.4 (8.1) | 68.2 (6.8) | 71.8(5.9) | 0.06 |
| Bicarbonate (mEq/L), mean (SD) | 22.1 (2.1) | 21.8 (1.3) | 21.4 (0.8) | 0.5 |
| NoDM | DM | p-Value | |
|---|---|---|---|
| (N = 6) | (N = 6) | ||
| KrU (mL/min), median [IQR] | −0.1 [−0.4 to 0.2] | 0.1 [0–0.6] | 0.5 |
| ClCr (mL/min), median [IQR] | −1.1 [−1.6 to −0.7] | −0.3 [−3.1 to 2.1] | 0.5 |
| Diuresis (mL), median [IQR] | 0 [−150 to 200] | −125 [−600 to 130] | 0.5 |
| Ultrafiltration PD (mL/day), median [IQR] | 25 [−50 to 50] | 50 [0–200] | 0.5 |
| HbA1c (%), median [IQR] | 0 [−0.07 to 0] | −0.04 [−0.5 to −0.3] | 0.04 |
| Proteinuria (mg/24 h), median [IQR] | −15.3 [−40 to 440] | −266.3 [−2209.2 to −10.2] | 0.2 |
| MAU (mg/24 h), median [IQR] | −26.9 [−20 to 28.9] | −189 [−1172.6 to −79] | 0.05 |
| SBP (mmHg), median [IQR] | −15 [−20 to −7] | −5 [−8 to 2] | 0.1 |
| DBP (mmHg), median [IQR] | 51 [51–51] | 64 [53–68] | 0.07 |
| nPCR (g Urea/Kg/d), median [IQR] | −0.1 [−0.31 to 0.01] | −0.05 [−0.3 to 0.1] | 0.5 |
| KT/V weekly (L), median [IQR] | 0 [−0.02 to 0] | −0.05 [−0.1 to 0] | 0.3 |
| Dry weight (kg), median [IQR] | −0.8 [−2.2 to 0.4] | −2.4 [−3.6 to −1.1] | 0.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moral Berrio, E.; De La Flor, J.C.; Arambarri Segura, M.; Rodríguez-Doyágüez, P.; Martínez Calero, A.; Zamora, R.; Cieza-Terrones, M.; Yuste-Lozano, C.; Sánchez de la Nieta García, M.D.; Nieto Iglesias, J.; et al. Effects of Sodium–Glucose Cotransporter 2 Inhibitors in Diabetic and Non-Diabetic Patients with Advanced Chronic Kidney Disease in Peritoneal Dialysis on Residual Kidney Function: In Real-World Data. Medicina 2024, 60, 1198. https://doi.org/10.3390/medicina60081198
Moral Berrio E, De La Flor JC, Arambarri Segura M, Rodríguez-Doyágüez P, Martínez Calero A, Zamora R, Cieza-Terrones M, Yuste-Lozano C, Sánchez de la Nieta García MD, Nieto Iglesias J, et al. Effects of Sodium–Glucose Cotransporter 2 Inhibitors in Diabetic and Non-Diabetic Patients with Advanced Chronic Kidney Disease in Peritoneal Dialysis on Residual Kidney Function: In Real-World Data. Medicina. 2024; 60(8):1198. https://doi.org/10.3390/medicina60081198
Chicago/Turabian StyleMoral Berrio, Esperanza, José C. De La Flor, Minerva Arambarri Segura, Pablo Rodríguez-Doyágüez, Alberto Martínez Calero, Rocío Zamora, Michael Cieza-Terrones, Claudia Yuste-Lozano, María Dolores Sánchez de la Nieta García, Javier Nieto Iglesias, and et al. 2024. "Effects of Sodium–Glucose Cotransporter 2 Inhibitors in Diabetic and Non-Diabetic Patients with Advanced Chronic Kidney Disease in Peritoneal Dialysis on Residual Kidney Function: In Real-World Data" Medicina 60, no. 8: 1198. https://doi.org/10.3390/medicina60081198





