Current Diagnostic and Therapeutic Challenges in Superficial Venous Thrombosis
Abstract
:1. Introduction
2. Definition and Epidemiology
3. Etiology and Risk Factors
| Etiology and risk factors |
| History of venous thromboembolism |
| Malignancy |
| Recent surgery or trauma |
| Immobilization |
| Inherited or acquired thrombophilia |
| Use of oral contraceptives |
| Infectious disease |
| Obesity |
| Cardiac of respiratory failure |
| Varicose veins |
| Burger’s disease |
| Natural coagulation inhibitory deficiency |
| Mutation factor V Leiden |
| Antiphospholipid syndrome |
| Increased levels of factor VIII |
4. Diagnosis
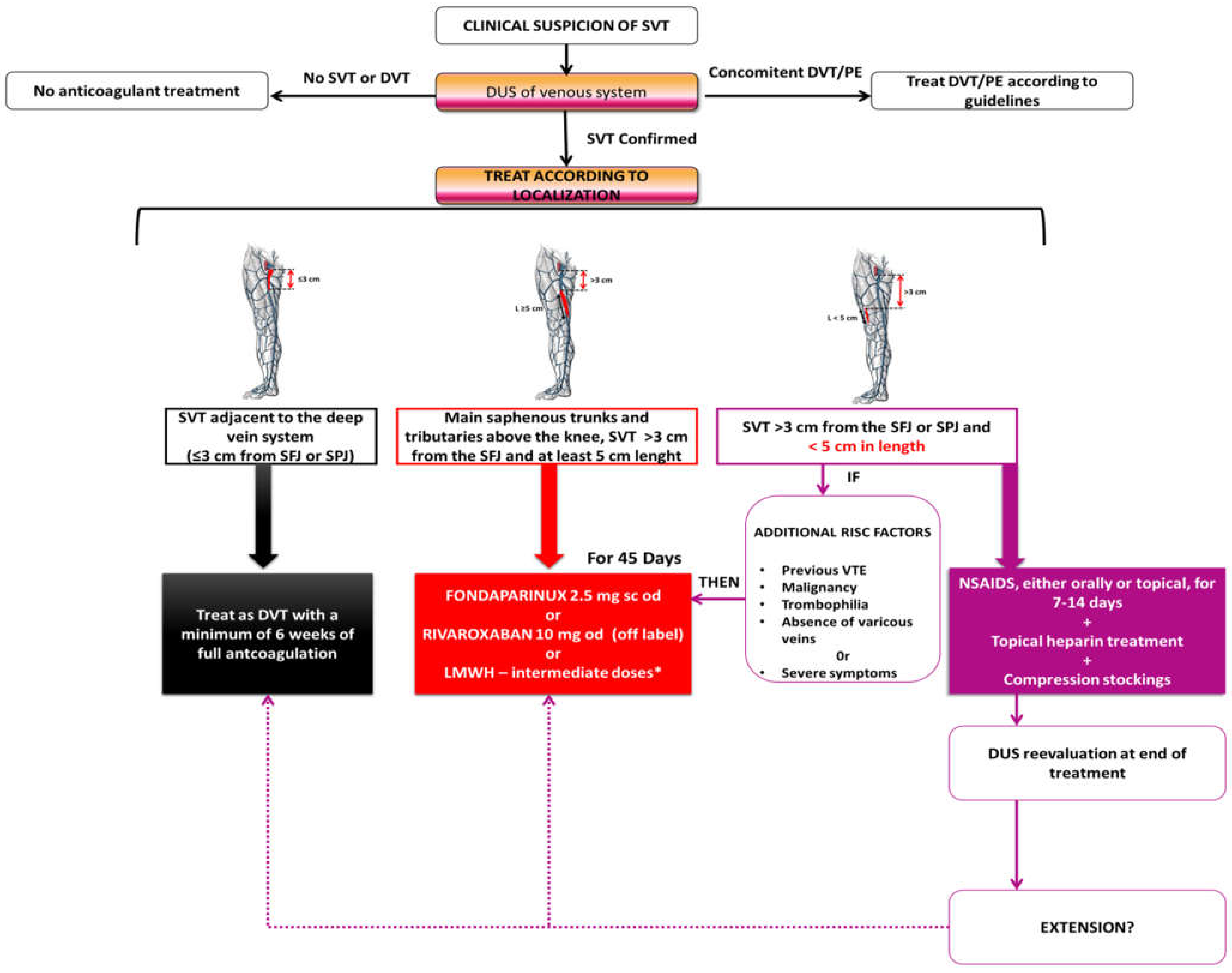
5. Management
5.1. Anticoagulation
| Anticoagulant | Studies | No. Patients | Conclusions |
|---|---|---|---|
| Fondaparinux | CALISTO trial, 2010 (RCT) | 3002 | Fondaparinux 2.5 mg sc daily is superior to placebo |
| Duffet et al., 2019 (meta-analysis) | 6862 | Fondaparinux is associated with the lowest VTE event rate during follow-up between patients with SVT treated with NSAIDs/anticoagulant therapies/surgical therapies/observation or placebo | |
| Di Nisio et al., 2018 (systematic review) | 7296 | This study supports fondaparinux in prophylactic dose for 45 days as efficient in superficial vein thrombosis | |
| Rivaroxaban | SURPRISE trial, 2017 (RCT) | 472 | Rivaroxaban (10 mg od) is non-inferior to fondaparinux (2.5 mg od sc) |
| Single center retrospective case record review at King’s College Hospital, 2021 | 54 | Rivaroxaban is effective and safe for the treatment of SVT | |
| Rivaroxaban compared to placebo for the treatment of leg superficial vein thrombosis: A randomized trial, 2020 (RCT) | 85 | The conclusion derived from the study is that rivaroxaban is effective in the treatment of SVT, although based on limited data | |
| LMWH | STEFLUX trial, 2012 (RCT) | 664 | A 30-day regimen of an intermediate dose of parnaparin is more effective than both a 30-day regimen of a prophylactic dose and a 10-day regimen of an intermediate dose. |
| REVETR study, 2014 (RCT) | 68 | Patients were assigned to either a prophylactic dose of dalteparin (5000 IU od) or to a double dose (10,000 IU od); the conclusion was that the dosage of anticoagulant does not impact the rate of thrombus resolution | |
| Rathbun et al., 2012 (RCT) | 72 | Dalteparin (intermediate dose) is superior to the NSAID ibuprofen | |
| Prandoni et al., 2005 (RCT) | 164 | Therapeutic dose of nadroparin, administered for 1 month in patients with SVT, is not superior to prophylactic dose | |
| Lozano et al., 2003 (RCT) | 84 | No statistically significant differences between enoxaparin (1 mg/kg b.i.d. for the first week, then 1 mg/kg od for 3 weeks) and saphenofemoral disconnection; however, enoxaparin group had socioeconomic advantages | |
| STENOX group, 2003 (double-blind trial) | 427 | Comparison between enoxaparin 40 mg od, enoxaparin 1.5 mg/kg od, oral tenoxicam, and placebo for 8–12 days; there was observed a benefit of the active treatment over placebo, but the results were not statistically significant | |
| Titon et al., 1994 (multicenter, randomized, open trial) | 117 | Calcium nadroparin proved a better efficacy in improving symptoms and signs comparative to naproxen; a higher effect of nadroparin on repermeabilization of the thrombosed vein was also observed, but the result was not statistically significant | |
| Gouveia et al., 2018 (retrospective cohort) | 60 | Patients were treated with enoxaparin 40 mg od (and 80 mg od for obese patients > 100 kg); the findings support LMWH usage in SVT |
| Guidelines | Recommendations |
|---|---|
| ESVS 2021 Clinical Practice Guidelines on the Management of Venous Thrombosis | A 45-day regimen of anticoagulation is recommended in the following situations (Class I B): Superficial vein thrombosis ≥ 3 cm away from the deep venous system and ≥ 5 cm in length:
|
| The 2023 Society for Vascular Surgery, American Venous Forum, and American Vein and Lymphatic Society clinical practice guidelines for the management of varicose veins of the lower extremities | SVT of the main saphenous trunks and tributaries above the knee, >3 cm from the SFJ, and >5 cm in length (with or without varicose veins):
SVT of the main saphenous trunks, <3 cm from the SFJ:
|
| Guidelines on the investigation and management of venous thrombosis at unusual sites (British Society for Haematology; last reviewed in 2022) | Confirmed SVT <3 cm to the sapheno–femoral junction:
|
| Thrombosis Canada Guide (last revised 2023) | Isolated SVT <3 cm to the sapheno–femoral junction (SFJ) or sapheno–popliteal (SPJ) junction:
|
5.2. Symptomatic Relief
5.3. Role of Surgery in SVT
5.4. Suggestions Regarding Treatment Options in Low-Income Countries
6. Superficial Vein Thrombosis in Pregnancy
- the first line of treatment is LMWH (fondaparinux 2.5 mg daily only in allergic or intolerant patients; however, the use of fondaparinux in pregnant women is off-label, as this drug can cross the placenta and the experience using it is limited, especially in the first trimester; certainly, there is need for more data about the safety of fondaparinux during pregnancy) [51,52]
- in SVT located below the knee: prophylactic doses of LMWH for 6 weeks (for example, enoxaparin 40 mg SC daily or 60 mg SC daily in obese patients); in patients with additional thromboembolic factors, continuation after 6 weeks should be considered (known significant thrombophilia, immobilization, infections, inflammatory or autoimmune conditions, cancer, or history of VTE)
- in SVT located above the knee, >10 cm from the sapheno–femoral junction or below the knee, >5 cm from the sapheno–popliteal junction: intermediate dose of LMWH for the entire pregnancy period and 6 weeks post-partum (for example, enoxaparin 40 mg SC b.i.d.)
- SVT located <10 cm from the sapheno–femoral junction or <5 cm from the sapheno–popliteal junction: therapeutic dose of LMWH for the entire pregnancy period and 6 weeks post-partum (for example, enoxaparin 1 mg/kgc SC b.i.d.)
- elastic compression stockings could be a viable option, but mainly in cases of chronic venous insufficiency [53].
7. Atypical Localization in SVT
8. Superficial Vein Thrombosis: A Distinct Entity or a Manifestation of Venous Thromboembolic Disease Spectrum
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schönauer, V.; Kyrle, P.A.; Weltermann, A.; Minar, E.; Bialonczyk, C.; Hirschl, M.; Quehenberger, P.; Schneider, B.; Partsch, H.; Eichinger, S. Superficial thrombophlebitis and risk for recurrent venous thromboembolism. J. Vasc. Surg. 2003, 37, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Di Minno, M.N.D.; Ambrosino, P.; Ambrosini, F.; Tremoli, E.; Di Minno, G.; Dentali, F. Prevalence of deep vein thrombosis and pulmonary embolism in patients with superficial vein thrombosis: A systematic review and meta-analysis. J. Thromb. Haemost. 2016, 14, 964–972. [Google Scholar] [CrossRef] [PubMed]
- Otten, T.R.; Stein, P.D.; Patel, K.C.; Mustafa, S.; Silbergleit, A. Thromboembolic Disease Involving the Superior Vena Cava and Brachiocephalic Veinsa. Chest 2003, 123, 809–812. [Google Scholar] [CrossRef]
- Kakkos, S.K.; Gohel, M.; Baekgaard, N.; Bauersachs, R.; Bellmunt-Montoya, S.; Black, S.A.; Ten Cate-Hoek, A.J.; Elalamy, I.; Enzmann, F.K.; Geroulakos, G.; et al. Editor’s Choice—European Society for Vascular Surgery (ESVS) 2021 Clinical Practice Guidelines on the Management of Venous Thrombosis. Eur. J. Vasc. Endovasc. Surg. 2021, 61, 9–82. [Google Scholar] [CrossRef]
- Gorty, S.; Patton-Adkins, J.; DaLanno, M.; Starr, J.; Dean, S.; Satiani, B. Superficial venous thrombosis of the lower extremities: Analysis of risk factors, and recurrence and role of anticoagulation. Vasc. Med. 2004, 9, 1–6. [Google Scholar] [CrossRef]
- Mangiafico, M.; Costanzo, L. Superficial Venous Thrombosis: A Comprehensive Review. Healthcare 2024, 12, 500. [Google Scholar] [CrossRef]
- Nikolakopoulos, K.M.; Kakkos, S.K.; Papageorgopoulou, C.P.; Tsolakis, I.A. Extended-Duration Treatment of Superficial Vein Thrombosis of the Lower Limbs with Tinzaparin. Vasc. Spec. Int. 2018, 34, 1–9. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- van Weert, H.; Dolan, G.; Wichers, I.; de Vries, C.; ter Riet, G.; Buller, H. Spontaneous superficial venous thrombophlebitis: Does it increase risk for thromboembolism? A historic follow-up study in primary care. J. Fam. Pract. 2006, 55, 52–57. [Google Scholar]
- Frappé, P.; Buchmuller-Cordier, A.; Bertoletti, L.; Bonithon-Kopp, C.; Couzan, S.; Lafond, P.; Leizorovicz, A.; Merah, A.; Presles, E.; Preynat, P.; et al. Annual diagnosis rate of superficial vein thrombosis of the lower limbs: The STEPH community-based study. J. Thromb. Haemost. 2014, 12, 831–838. [Google Scholar] [CrossRef]
- Gillet, J.L.; Allaert, F.A.; Perrin, M. Thromboses veineuses superficielles des veines non variqueuses des membres inférieurs. J. Mal. Vasc. 2004, 29, 263–272. [Google Scholar] [CrossRef]
- Saber, W.; Moua, T.; Williams, E.C.; Verso, M.; Agnelli, G.; Couban, S.; Young, A.; De Cicco, M.; Biffi, R.; van Rooden, C.J.; et al. Risk factors for catheter-related thrombosis (CRT) in cancer patients: A patient-level data (IPD) meta-analysis of clinical trials and prospective studies. J. Thromb. Haemost. 2011, 9, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Hirmerová, J.; Seidlerová, J.; Šubrt, I.; Hajšmanová, Z. Prevalence of cancer in patients with superficial vein thrombosis and its clinical importance. J. Vasc. Surg. Venous Lymphat. Disord. 2022, 10, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Langer, F.; Gerlach, H.E.; Schimke, A.; Heinken, A.; Hoffmann, U.; Noppeney, T.; Pittrow, D.; Klotsche, J.; Rabe, E.; Bauersachs, R.; et al. Clinical outcomes of cancer-associated isolated superficial vein thrombosis in daily practice. Thromb. Res. 2022, 220, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Lyman, G.H.; Carrier, M.; Ay, C.; Di Nisio, M.; Hicks, L.K.; Khorana, A.A.; Leavitt, A.D.; Lee, A.Y.Y.; Macbeth, F.; Morgan, R.L.; et al. American Society of Hematology 2021 guidelines for management of venous thromboembolism: Prevention and treatment in patients with cancer. Blood Adv. 2021, 5, 927–974. [Google Scholar] [CrossRef]
- Czysz, A.; Higbee, S.L. Superficial Thrombophlebitis. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2024. Available online: https://www.ncbi.nlm.nih.gov/books/NBK556017 (accessed on 10 May 2024).
- Leon, L.; Giannoukas, A.D.; Dodd, D.; Chan, P.; Labropoulos, N. Clinical Significance of Superficial Vein Thrombosis. Eur. J. Vasc. Endovasc. Surg. 2005, 29, 10–17. [Google Scholar] [CrossRef]
- Di Nisio, M.; Van Sluis, G.L.; Bossuyt, P.M.M.; Büller, H.R.; Porreca, E.; Rutjes, A.W.S. Accuracy of diagnostic tests for clinically suspected upper extremity deep vein thrombosis: A systematic review. J. Thromb. Haemost. 2010, 8, 684–692. [Google Scholar] [CrossRef]
- Varrias, D.; Palaiodimos, L.; Balasubramanian, P.; Barrera, C.; Nauka, P.; Arfaras-Melainis, A.; Zamora, C.; Zavras, P.; Napolitano, M.; Gulani, P.; et al. The Use of Point-of-Care Ultrasound (POCUS) in the Diagnosis of Deep Vein Thrombosis. J. Clin. Med. 2021, 10, 3903. [Google Scholar] [CrossRef]
- Górski, G.; Noszczyk, W.; Kostewicz, W.; Szopiński, P.; Kielar, M.; Michalak, J.; Marianowska, A.; Wilkoswski, D. Progress of local symptoms of superficial vein thrombosis vs. duplex findings. Vasa 2004, 33, 219–225. [Google Scholar] [CrossRef]
- Beyer-Westendorf, J. Controversies in venous thromboembolism: To treat or not to treat superficial vein thrombosis. Hematol. Am. Soc. Hematol. Educ. Program 2017, 2017, 223–230. [Google Scholar] [CrossRef]
- Scovell, S. Superficial vein thrombosis and phlebitis of the lower extremity veins. In UpToDate; Connor, R.F., Ed.; Wolters Kluwer: Alphen aan den Rijn, The Netherlands, 2022; Available online: https://www.uptodate.com/contents/superficial-vein-thrombosis-and-phlebitis-of-the-lower-extremity-veins (accessed on 6 May 2024).
- Gloviczki, P.; Lawrence, P.F.; Wasan, S.M.; Meissner, M.H.; Almeida, J.; Brown, K.R.; Bush, R.L.; Di Iorio, M.; Fish, J.; Fukaya, E.; et al. The 2023 Society for Vascular Surgery, American Venous Forum, and American Vein and Lymphatic Society clinical practice guidelines for the management of varicose veins of the lower extremities. Part II: Endorsed by the Society of Interventional Radiology and the Society for Vascular Medicine. J. Vasc. Surg. Venous Lymphat. Disord. 2023, 12, 101670. [Google Scholar]
- Cosmi, B. Management of superficial vein thrombosis. J. Thromb. Haemost. 2015, 13, 1175–1183. [Google Scholar] [CrossRef]
- Prandoni, P.; Pesavento, R.; Bilora, F.; Fernández Reyes, J.L.; Madridano, O.; Soler, S.; Monreal, M. No difference in outcome between therapeutic and preventive anticoagulation in patients with superficial vein thrombosis involving the saphenous–femoral junction. Vasc. Med. 2022, 27, 290–292. [Google Scholar] [CrossRef] [PubMed]
- Leizorovicz, A.; Becker, F.; Buchmüller, A.; Quéré, I.; Prandoni, P.; Decousus, H. Clinical relevance of symptomatic superficial-vein thrombosis extension: Lessons from the CALISTO study. Blood 2013, 122, 1724–1729. [Google Scholar] [CrossRef] [PubMed]
- Decousus, H.; Prandoni, P.; Mismetti, P.; Bauersachs, R.M.; Boda, Z.; Brenner, B.; Laporte, S.; Matyas, L.; Middeldorp, S.; Sokurenko, G.; et al. Fondaparinux for the Treatment of Superficial-Vein Thrombosis in the Legs. N. Engl. J. Med. 2010, 363, 1222–1232. [Google Scholar] [CrossRef]
- Beyer-Westendorf, J.; Schellong, S.; Gerlach, H.; Jersemann, K.; Rabe, E.; Sahin, K.; Bauersachs, R. Rivaroxaban Versus Fondaparinux in the Treatment of Superficial Vein Thrombosis—The Surprise Trial. Blood 2016, 128, 85. [Google Scholar] [CrossRef]
- Beyer-Westendorf, J.; Schellong, S.M.; Gerlach, H.; Rabe, E.; Weitz, J.I.; Jersemann, K.; Sahin, K.; Bauersachs, R. Prevention of thromboembolic complications in patients with superficial-vein thrombosis given rivaroxaban or fondaparinux: The open-label, randomised, non-inferiority SURPRISE phase 3b trial. Lancet Haematol. 2017, 4, e105–e113. [Google Scholar] [CrossRef]
- Di Nisio, M.; Wichers, I.M.; Middeldorp, S. Treatment for superficial thrombophlebitis of the leg. Cochrane Vascular Group, editor. Cochrane Database Syst. Rev. 2018, 2018, CD004982. [Google Scholar] [CrossRef]
- Tait, C.; Baglin, T.; Watson, H.; Laffan, M.; Makris, M.; Perry, D.; Keeling, D. British Committee for Standards in Haematology. Guidelines on the investigation and management of venous thrombosis at unusual sites. Br. J. Haematol. 2012, 159, 28–38. [Google Scholar] [CrossRef]
- Karathanos, C.; Kakkos, S.K.; Georgiadis, G.; Ioannou, C.; Vasdekis, S.; Chatzis, D.; Latzios, P.; Giannoukas, A.D. Risk of recurrent thromboembolic events according to treatment duration in patients with superficial vein thrombosis treated with intermediate dose of tinzaparin. Phlebol. J. Venous Dis. 2023, 38, 141–149. [Google Scholar] [CrossRef]
- Duffett, L.; Kearon, C.; Rodger, M.; Carrier, M. Treatment of Superficial Vein Thrombosis: A Systematic Review and Meta-Analysis. Thromb. Haemost. 2019, 119, 479–489. [Google Scholar] [CrossRef]
- Clapham, R.E.; Speed, V.; Czuprynska, J.; Gazes, A.; Guppy, S.; Patel, R.K.; Rea, C.; Vadher, B.; Arya, R.; Roberts, L.N. Rivaroxaban for the treatment of superficial vein thrombosis, experience at King’s College Hospital. Br. J. Haematol. 2022, 196, e3–e6. [Google Scholar] [CrossRef] [PubMed]
- Kearon, C.; Carrier, M.; Gu, C.S.; Schulman, S.; Bates, S.M.; Kahn, S.R.; Chagnon, I.; Nguyen, D.T.; Wu, C.; Rudd-Scott, L.; et al. Rivaroxaban Compared to Placebo for the Treatment of Leg Superficial Vein Thrombosis: A Randomized Trial. Semin. Thromb. Hemost. 2020, 46, 977–985. [Google Scholar] [CrossRef]
- Cosmi, B.; Filippini, M.; Tonti, D.; Avruscio, G.; Ghirarduzzi, A.; Bucherini, E.; Camporese, G.; Imberti, D.; Palareti, G. A randomized double-blind study of low-molecular-weight heparin (parnaparin) for superficial vein thrombosis: STEFLUX (Superficial ThromboEmbolism and Fluxum). J. Thromb. Haemost. 2012, 10, 1026–1035. [Google Scholar] [CrossRef]
- Rathbun, S.W.; Aston, C.E.; Whitsett, T.L. A randomized trial of dalteparin compared with ibuprofen for the treatment of superficial thrombophlebitis. J. Thromb. Haemost. JTH 2012, 10, 833–839. [Google Scholar] [CrossRef]
- Siddiqua, N.; Mathew, R.; Sahu, A.K.; Jamshed, N.; Bhaskararayuni, J.; Aggarwal, P.; Kumar, A.; Khan, M.A. High vs. low doses of low-molecular-weight heparin for the treatment of superficial vein thrombosis of the legs: A double-blind, randomized trial. J. Thromb. Haemost. 2005, 3, 1152–1157. [Google Scholar]
- Lozano, F.S.; Almazan, A. Low-Molecular-Weight Heparin Versus Saphenofemoral Disconnection for the Treatment of Above-Knee Greater Saphenous Thrombophlebitis: A Prospective Study. Vasc. Endovasc. Surg. 2003, 37, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Spirkoska, A.; Jezovnik, M.K.; Poredos, P. Time Course and the Recanalization Rate of Superficial Vein Thrombosis Treated With Low-Molecular-Weight Heparin. Angiology 2015, 66, 381–386. [Google Scholar] [CrossRef]
- The Superficial Thrombophlebitis Treated by Enoxaparin Study Group. A Pilot Randomized Double-blind Comparison of a Low-Molecular-Weight Heparin, a Nonsteroidal Anti-inflammatory Agent, and Placebo in the Treatment of Superficial Vein Thrombosis. Arch. Intern. Med. 2003, 163, 1657–1663. Available online: https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/215878 (accessed on 15 May 2024). [CrossRef]
- Titon, J.P.; Auger, D.; Grange, P.; Hecquet, J.P.; Remond, A.; Ulliac, P.; Vaissie, J.J. Therapeutic management of superficial venous thrombosis with calcium nadroparin. Dosage testing and comparison with a non-steroidal anti-inflammatory agent. Ann. Cardiol. Angeiol. 1994, 43, 160–166. [Google Scholar]
- Gouveia, S.; Roberts, L.N.; Czuprynska, J.; Patel, R.K.; Arya, R. Single centre experience of the management of superficial vein thrombosis with prophylactic low-molecular-weight heparin. Br. J. Haematol. 2018, 181, 682–684. [Google Scholar] [CrossRef]
- Thrombosis Canada. Superficial Thrombophlebitis, Superficial Vein Thrombosis. Available online: https://thrombosiscanada.ca/hcp/practice/clinical_guides?language=enca&guideID=90 (accessed on 2 May 2024).
- Aminoshariae, A.; Kulild, J.C.; Donaldson, M. Short-term use of nonsteroidal anti-inflammatory drugs and adverse effects. J. Am. Dent. Assoc. 2016, 147, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Boehler, K.; Kittler, H.; Stolkovich, S.; Tzaneva, S. Therapeutic Effect of Compression Stockings Versus no Compression on Isolated Superficial Vein Thrombosis of the Legs: A Randomized Clinical Trial. Eur. J. Vasc. Endovasc. Surg. 2014, 48, 465–471. [Google Scholar] [CrossRef]
- Décousus, H.; Bertoletti, L.; Frappé, P. Spontaneous acute superficial vein thrombosis of the legs: Do we really need to treat? J. Thromb. Haemost. 2015, 3, S230–S237. [Google Scholar] [CrossRef]
- Cabré, F.; Camacho, J.A.; Rodríguez-Garcés, C.A.; Breier, D.V.; Ballarin, M. Review of Topical Sodium Heparin 1000 IU/g Gel in Symptomatic Uncomplicated Superficial Thrombophlebitis. Cureus 2023, 5, e47418. [Google Scholar] [CrossRef] [PubMed]
- Belcaro, G.; Nicolaides, A.N.; Errichi, B.M.; Cesarone, M.R.; De Sanctis, M.T.; Incandela, L.; Venniker, R. Superficial Thrombophlebitis of the Legs: A Randomized, Controlled, Follow-up Study. Angiology 1999, 50, 523–529. [Google Scholar] [CrossRef]
- Casian, D.; Bzovii, F.; Culiuc, V.; Gutu, E. Urgent surgery versus anticoagulation for treatment of superficial vein thrombosis in patients with varicose veins: A prospective observational cohort study. Vasa 2022, 51, 174–181. [Google Scholar] [CrossRef]
- Kourlaba, G.; Relakis, J.; Kontodimas, S.; Holm, M.V.; Maniadakis, N. A systematic review and meta-analysis of the epidemiology and burden of venous thromboembolism among pregnant women. Int. J. Gynecol. Obstet. 2016, 132, 4–10. [Google Scholar] [CrossRef]
- Bates, S.M.; Rajasekhar, A.; Middeldorp, S.; McLintock, C.; Rodger, M.A.; James, A.H.; Vazquez, S.R.; Greer, I.A.; Riva, J.J.; Bhatt, M.; et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: Venous thromboembolism in the context of pregnancy. Blood Adv. 2018, 2, 3317–3359. [Google Scholar] [CrossRef]
- Bauersachs, R.M. Fondaparinux Sodium: Recent Advances in the Management of Thrombosis. J. Cardiovasc. Pharmacol. Ther. 2023, 28, 107424842211450. [Google Scholar] [CrossRef] [PubMed]
- Antic, D.; Lefkou, E.; Otasevic, V.; Banfic, L.; Dimakakos, E.; Olinic, D.; Milić, D.; Miljić, P.; Xhepa, S.; Stojkovski, I.; et al. Position Paper on the Management of Pregnancy-Associated Superficial Venous Thrombosis. Balkan Working Group for Prevention and Treatment of Venous Thromboembolism. Clin. Appl. Thromb. 2022, 28, 107602962093918. [Google Scholar] [CrossRef]
- Maki, D.G.; Ringer, M. Risk Factors for Infusion-related Phlebitis with Small Peripheral Venous Catheters: A Randomized Controlled Trial. Ann. Intern. Med. 1991, 114, 845–854. [Google Scholar] [CrossRef]
- Reis, P.E.D.D.; De Campos Pereira Silveira, R.C.; Vasques, C.I.; De Carvalho, E.C. Pharmacological Interventions to Treat Phlebitis: Systematic Review. J. Infus. Nurs. 2009, 32, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Samlaska, C.P.; James, W.D. Superficial thrombophlebitis II. Secondary hypercoagulable states. J. Am. Acad. Dermatol. 1990, 23, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.; Garvin, D.; Poimboeuf, S. A common presentation to an uncommon disease. Penile Mondor’s disease: A case report and literature review. Int. Med. Case Rep. J. 2014, 7, 155–157. [Google Scholar] [PubMed]
- Ellis, M.H.; Fajer, S. A current approach to superficial vein thrombosis. Eur. J. Haematol. 2013, 90, 85–88. [Google Scholar] [CrossRef]
- Decousus, H.; Epinat, M.; Guillot, K.; Quenet, S.; Boissier, C.; Tardy, B. Superficial vein thrombosis: Risk factors, diagnosis, and treatment. Curr. Opin. Pulm. Med. 2003, 9, 393–397. [Google Scholar] [CrossRef]
- Costache, R.S.; Dragomirică, A.S.; Gheorghe, B.E.; Balaban, V.D.; Stanciu, S.M.; Jinga, M.; Costache, D.O. Oral Anticoagulation in Patients with Chronic Liver Disease. Medicina 2023, 59, 346. [Google Scholar] [CrossRef]
- Decousus, H.; Quéré, I.; Presles, E.; Becker, F.; Barrellier, M.T.; Chanut, M.; Gillet, J.L.; Guenneguez, H.; Leandri, C.; Mismetti, P.; et al. Superficial venous thrombosis and venous thromboembolism: A large, prospective epidemiologic study. Ann. Intern. Med. 2010, 152, 218–224. [Google Scholar] [CrossRef]
- Bell, L.N.; Berg, R.L.; Schmelzer, J.R.; Liang, H.; Mazza, J.J.; Kanth, R.; Bray, C.L.; Zaldivar, C.B.; Yale, S.H. Thromboembolic complications following a first isolated episode of superficial vein thrombosis: A cross-sectional retrospective study. J. Thromb. Thrombolysis 2017, 43, 31–37. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balahura, A.-M.; Florescu, A.-G.; Barboi, T.-M.; Weiss, E.; Miricescu, D.; Jurcuț, C.; Jinga, M.; Stanciu, S. Current Diagnostic and Therapeutic Challenges in Superficial Venous Thrombosis. Medicina 2024, 60, 1466. https://doi.org/10.3390/medicina60091466
Balahura A-M, Florescu A-G, Barboi T-M, Weiss E, Miricescu D, Jurcuț C, Jinga M, Stanciu S. Current Diagnostic and Therapeutic Challenges in Superficial Venous Thrombosis. Medicina. 2024; 60(9):1466. https://doi.org/10.3390/medicina60091466
Chicago/Turabian StyleBalahura, Ana-Maria, Adrian-Gabriel Florescu, Teodora-Maria Barboi, Emma Weiss, Daniela Miricescu, Ciprian Jurcuț, Mariana Jinga, and Silviu Stanciu. 2024. "Current Diagnostic and Therapeutic Challenges in Superficial Venous Thrombosis" Medicina 60, no. 9: 1466. https://doi.org/10.3390/medicina60091466
APA StyleBalahura, A.-M., Florescu, A.-G., Barboi, T.-M., Weiss, E., Miricescu, D., Jurcuț, C., Jinga, M., & Stanciu, S. (2024). Current Diagnostic and Therapeutic Challenges in Superficial Venous Thrombosis. Medicina, 60(9), 1466. https://doi.org/10.3390/medicina60091466






