1. Introduction
Salmon muscle is rich in ω3 fatty acids such as docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA). Consumption of ω3 fatty acids leads to a variety of health benefits such as reducing cardiovascular disease (CVD) risk, rheumatoid arthritis, and behavioral disorders [
1]. Moreover, ω3 fatty acids are heavily involved in a broad spectrum of physiological roles such as neurogenesis, neuroinflammation, and neurotransmission, indicating their essential roles in the development and aging of the brain. An insufficient supply of ω3 fatty acids results in severe mental disorders such as depression, schizophrenia, bipolar disorder, autism, dementia, and attention-deficit/hyperactivity disorder [
2].
Shotgun lipidomics is characterized by direct infusion into the mass spectrometer without an HPLC system, and it has been used for the identification and quantification of various lipid species in biological samples. Shotgun lipidomics enables the acquisition of a mass spectrum of samples at a constant concentration of the solution during direct infusion [
3]. Thus, it provides unlimited time to operate multiple fragmentation strategies, including precursor ion, product ion, neutral loss (NL) scanning modes, and selected reaction monitoring (SRM) for the identification of target compounds. Moreover, this system provides abundant opportunities to adjust mass analysis conditions such as the ionization condition, gas pressures, and collision energies, which are constrained in the HPLC-MS/MS system due to one-time injection. Due to this advantage, shotgun-based lipidomics has been widely applied in a variety of research areas such as seed lipidomes, cellular lipidomes, plant lipids, lipid peroxidation in redox biology, and others [
4,
5]. Meanwhile, multiple NL scans enable the confirmation of each segment of structural components of lipid molecules possessing similar substructures such as triacylglycerols (TAGs) and phospholipids (PhLs) [
6]. Considerable research has demonstrated the effectiveness of multiple NL and precursor ion scanning for the identification and quantification of PhL and TAG molecules of diverse sample origins, including vegetable oil, ginseng, and turmeric [
6,
7,
8].
Fishmeal and fish oil are traditional ingredients in salmonid feeds, but now they are becoming unsustainable due to their high cost following their limited supply, leading to research and development of appropriate alternatives to overcome this challenge, including heterotrophic protists [
9]. Thraustochytrids, which have a uniform spherical shape between 30 and 100 μm in diameter, are classified into a marine group of eukaryotic protists called Labyrinthulomycetes and are abundant in marine ecosystems [
10]. Increasing evidence has been reported on the health-promoting potential of microbial DHA and EPA in many clinical trials [
11]. Thus, microbial oil from thraustochytrids could be a promising alternative to fish oil due to their high level of ω3 fatty acids. Meanwhile, eicosanoids, derived by enzymatic or non-enzymatic oxidation of arachidonic acid or other PUFAs, are responsible for intercellular signaling, sustaining homeostatic functions, and mediating pathogenic mechanisms in animals [
12]. Fish leukocytes synthesize and release prostaglandins (PGs) on immunostimulation and they play a key role in regulating the primary biological functions of fish leukocytes, affecting the innate immune system of fish [
13]. Recent research has reported many health-promoting abilities of eicosanoids including suppressing inflammation, neurodegenerative diseases, cancer, central nervous system injury, and neuropsychiatric conditions [
14,
15,
16].
Recently, much research has been carried out on alterations in the nutritional components such as lipids (by focusing on fatty acid profiles), proteins, and micromolecules in salmon muscle tissue as affected by different feeding sources. However, monitoring lipid species such as TAGs, PhLs, and eicosanoids after feeding a different dietary source has rarely been investigated. This is important for consumers because ω3 PUFA bound to phospholipids seems to exert differential bioavailability and biological effects on consumption in comparison to neutral forms of ω3 PUFA [
17]. In previous work from our research group, the total lipid content, total TAG, and total PhL in salmon muscle after feeding different diets were measured, and as an extension of that research, this study was designed to observe the detailed composition of TAG and PhL species. In terms of PhLs, alkyl acyl PhLs were not analyzed as they are minor components in salmon muscle tissue [
18].
Thus, the objective of the present study was to observe the alteration in the levels of PhL and TAG molecules as well as prostaglandins PGE2 and PGF3α in salmon muscle tissue after feeding four different diets using shotgun-based mass spectrometry (MS) analysis. Here, we focused on tracing the flow of DHA, which is rich in microbial oil, into PhL and TAG in salmon muscle tissue.
3. Discussion
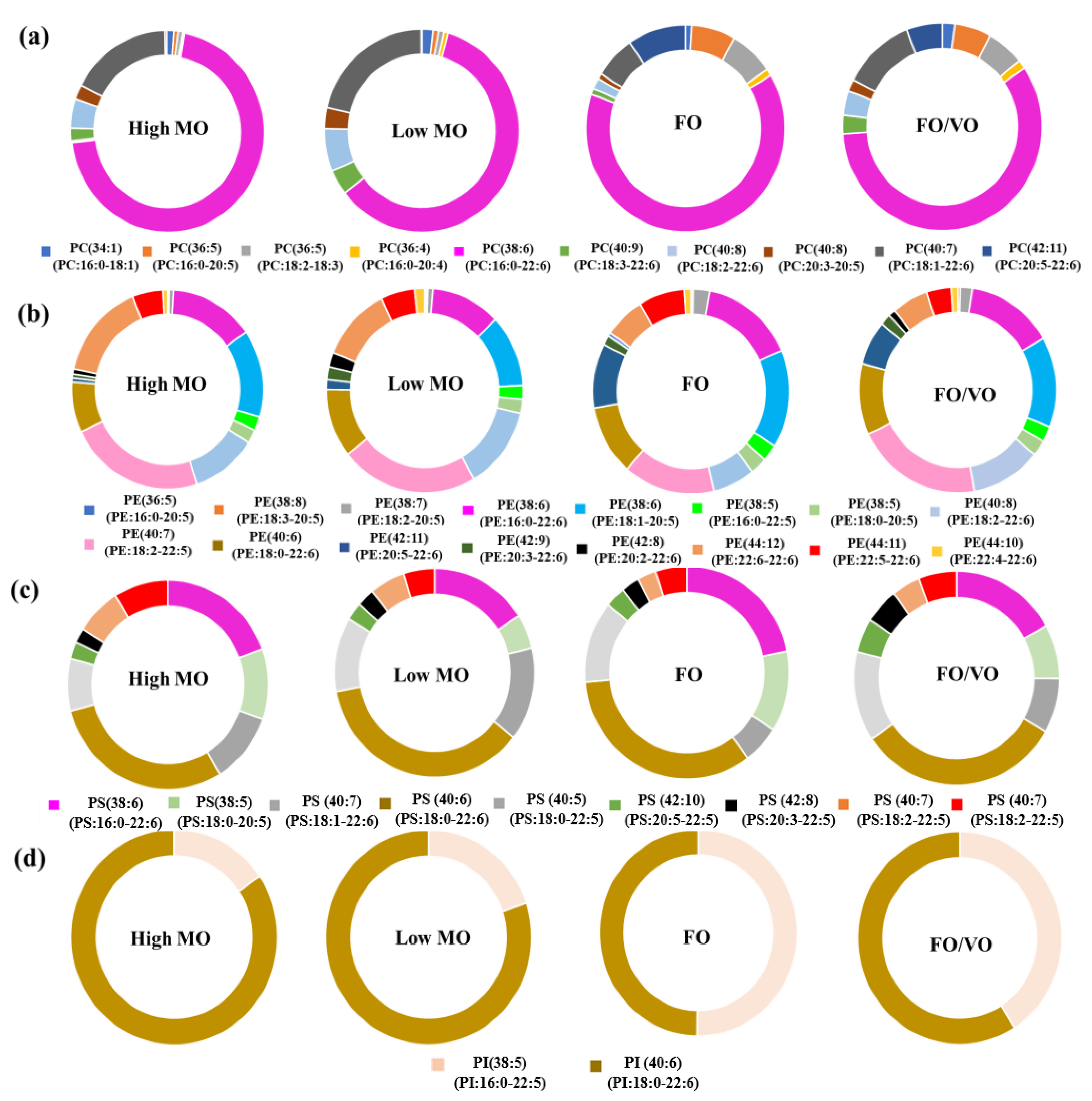
In the present study, the changes in PhL species in salmon muscle tissue were investigated using MS analysis. In particular, the alteration of DHA-containing PhLs using the MO diets was the focus. Overall, DHA in the MO diets was efficiently incorporated into the PhLs, especially in PC species, in the salmon muscle tissue as shown in the results.
Broughton et al. [
22] reported that feeding genetically modified (GM) oilseeds to Atlantic salmon significantly changed lipid profiles in their brain, eyes, gills, intestines, and liver. Tissue-dependent trends were found in the incorporation of long chain-polyunsaturated fatty acid (LC-PUFA) as well as the putative alteration in stereospecific isomer numbering (
sn). This indicates that changes in oil sources for fish feeding could lead to notable alterations in lipid composition in fish tissues, which agrees with the present study. Jin et al. [
23] investigated how diet affects lipid profiles in salmon muscle tissues and found comparable changes in PhL profiles driven by the switch in diet, but more dramatic alteration was observed in the gut and liver.
Meanwhile, Wang and Zhang [
23] investigated the phospholipid profiles of muscle from
Ctennopharyngodon idellus and they found that it contains 16:0_18:1-PC, 16:0_20:4-PC, and 18:0_20:5-PC as the predominant PC species, which is different to salmon muscle with 16:0-22:6-PC as the primary PC species. Moreover, Inhamuns and Bueno Franco [
24] reported that phospholipids in Mapara muscle tissue possess high levels of 16:0 and 18:1. Thus, the above findings reveal significant differences in fatty acid and phospholipid compositions of muscle tissue of different fishes.
Lipids in dietary sources may change their structural formation in the digestion and absorption process, that is, TAG molecules in the dietary source are digested into
sn-2-monoacylglycerol and free fatty acids, followed by the absorption and re-synthesize into PhL or other lipids [
25]. Thus, the remodeling of lipidomes in salmon muscle tissue on feeding different diets may occur in this process. The alteration of lipidomes in salmon muscle can also be caused by the influence of feeding diets on membrane-associated protein. Jeromson et al. [
26] reported that EPA influences the alterations of membrane-associated proteomes, which are related to the synthesis and modification of lipids, in muscle cell models, followed by changes in membrane lipid species. Thus, the EPA content in four different diets may change the composition of lipid molecules in salmon muscle tissue by affecting membrane-associated proteomes.
The levels of DHA and EPA may play crucial roles in maintaining a well-balanced metabolism and suppressing severe diseases in the human body. Thus, the incorporation of DHA in salmon muscle tissue via MO diets would be an excellent strategy for enhancing the nutritional and functional value of Atlantic salmon for consumers. For instance, DHA has been proven to be an effective bioactive compound in enhancing heart and brain function as well as retarding the rate of cognitive decline [
27]. Meanwhile, a substantial body of evidence has reported the beneficial health effects of EPA such as reducing cardiovascular disease (CVD), asthma, rheumatoid arthritis, behavioral disorders, cystic fibrosis, etc. [
28]. Some animal studies demonstrated that marine ω3 fatty acids elevated the level of the anti-inflammatory cytokine IL-10 [
29]. Several studies show that marine ω3 fatty acid supplements reduced the production of TNF, IL-6, and IL-1β by endotoxin-stimulated monocytes in healthy humans [
30]. Robinson and Stone [
31] summarized the remarkable antiatherosclerotic and antithrombotic activities of ω3 fatty acids in animal and human studies.
The forms in which DHA and EPA are consumed are of particular interest. The elevated level of DHA in salmon muscle tissue, as found in the PhL profile, on feeding a high DHA diet such as high MO may provide a variety of beneficial effects on human health for consumers. Calder [
32] reported the anti-inflammatory effect of ω3 fatty acids in the form of phospholipids in marine species such as salmon. Hals et al. [
33] demonstrated that purified phospholipids rich in ω3 fatty acids significantly reduced cardiovascular disease risk factors. Shiels et al. [
34] reported that phospholipids containing ω3 fatty acids exhibit strong anti-inflammatory and antithrombotic effects. PhLs containing ω3 fatty acids derived from salmon displayed high antithrombotic activities against the platelet-activating factor (PAF) and thrombin pathways [
18]. Further, recent studies demonstrated that DHA-PhLs in particular show a higher absorption and bioavailability in the brain compared to that of DHA-TAG, leading to the prevention of Alzheimer’s disease [
35]. Thus, it is important to achieve such enrichment in the PhLs of the fish by feeding them appropriate diets such as MO.
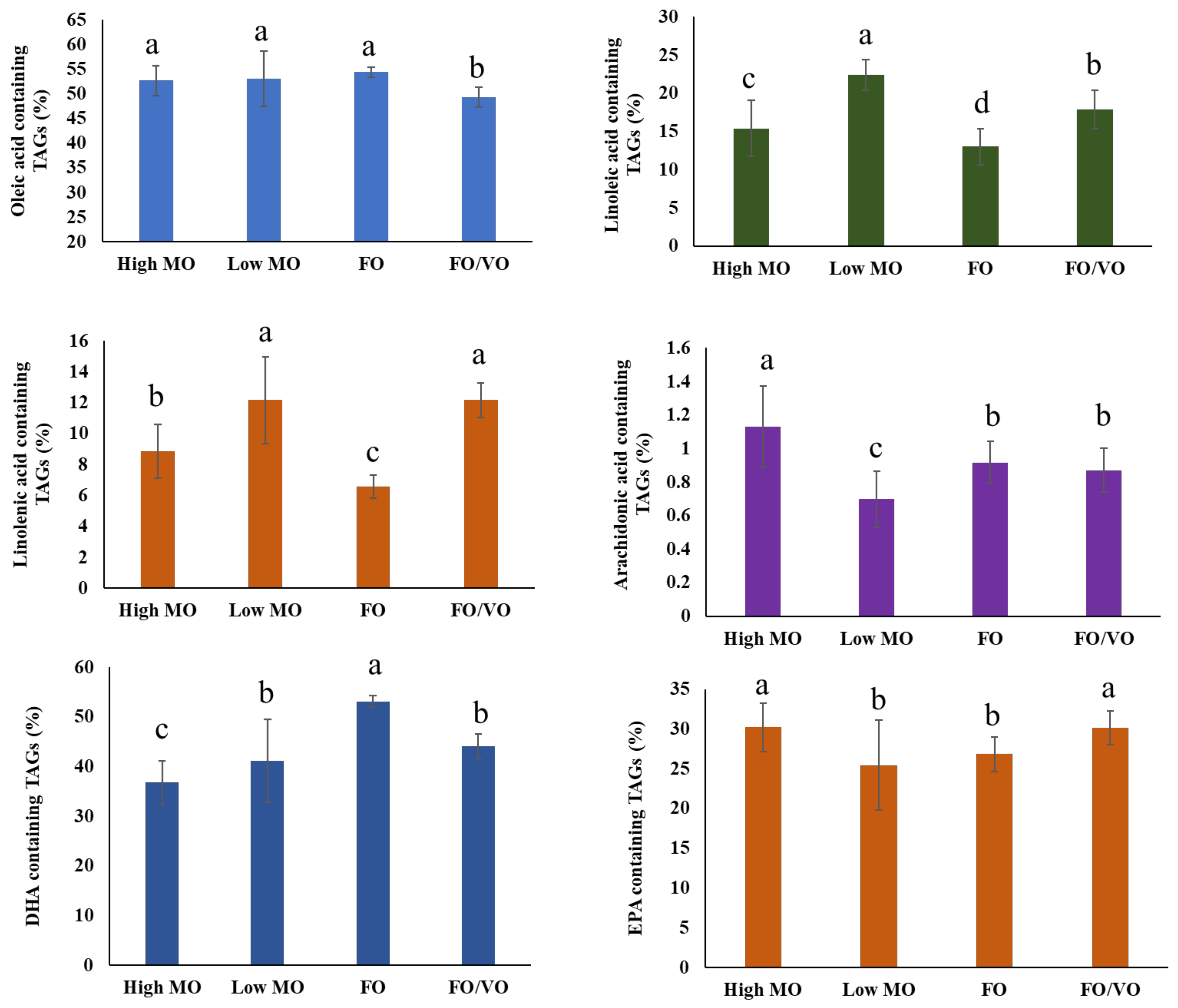
Meanwhile, DHA and EPA levels in the diets did not significantly affect the levels of DHA- and EPA-containing TAGs in salmon muscle tissue. Hixson et al. [
36] demonstrated that salmon fed with camelina oil, which contains a low DHA content (0.5–1.1% of total fatty acids), sustained DHA levels in salmon muscle tissue and led to the increase in ω3 and ω3 intermediary biosynthesis products. This fact indicates the endogenous production of 22:6ω3 from dietary 18:3ω3 in salmon muscle, and it supports the results of the present study. PCA analysis displayed a similar distribution of TAG molecules with each dietary treatment, although high MO samples appeared to be more concentrated on the positive side of the first axis, while low MO samples appeared more concentrated in the upper left quadrant (PCA results not shown). Jin et al. [
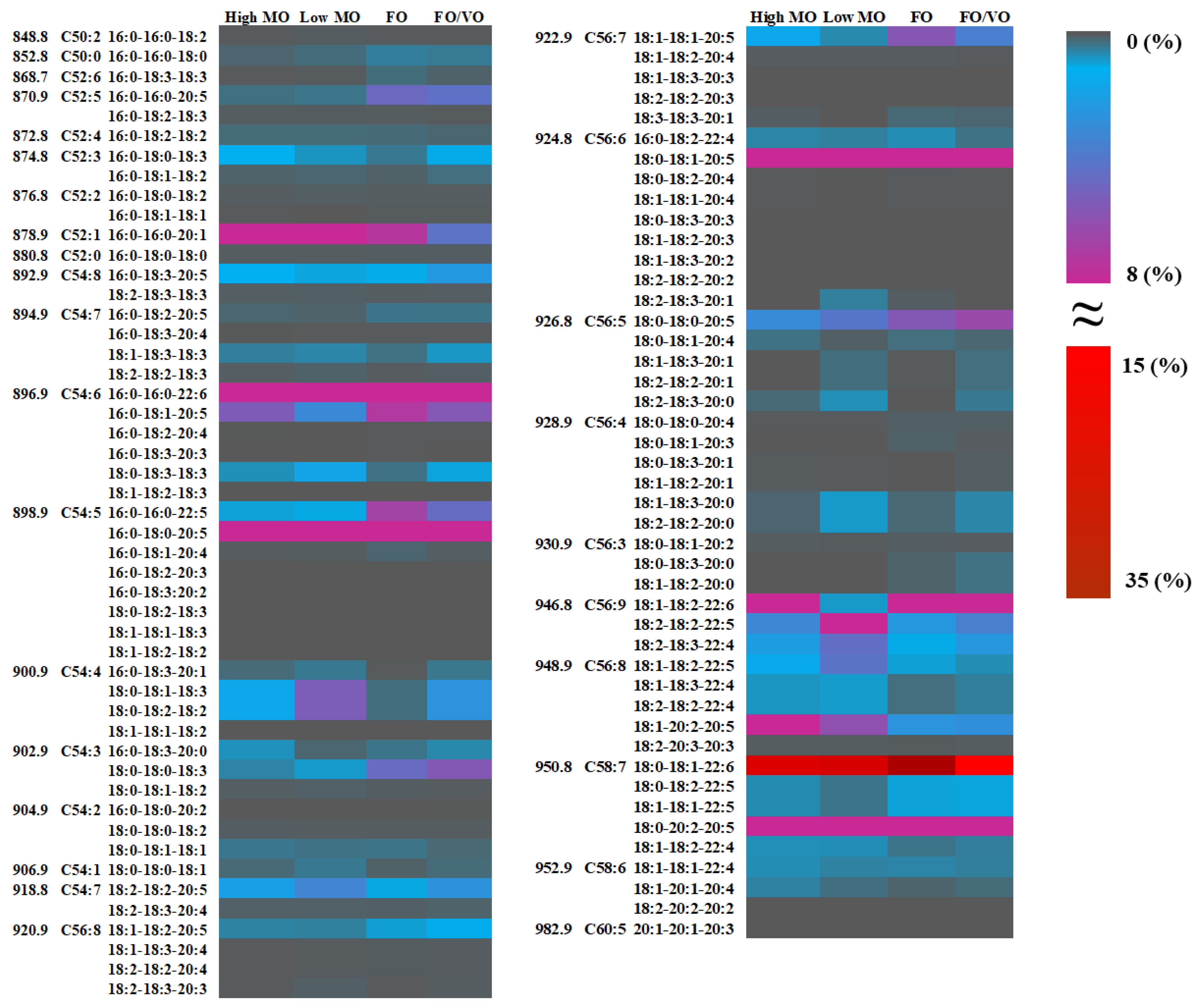
25] investigated the alteration of TAG molecules in salmon muscle tissue when fed different diets in which TAG molecules with higher chain lengths (more than 60) and double bonds between 12 and 18 were highly related to being fed fish oil. On the other hand, being fed vegetable oil (VO) was closely associated with the TAG species possessing shorter carbon chain lengths (less than 60) and low unsaturation (i.e., 56:10, 54:10, and 58:10). However, they did not report the alteration in lipid species (i.e., 18:0_18:2_22:6, 18:1_18:2_22:4, 16:0_18:2_20:5, etc.), limiting the comparison with the present study in monitoring TAG species during feeding trials. Until now, most research has not been focused on the analysis of TAG molecules; thus, the present study may fill the gaps in understanding lipid species in salmon muscles and their changes.
The discrepancy in PGE2 contents between FO and MO diets might be due to the difference in the levels of their precursor molecule, 20:4ω6, required for their synthesis. As shown in
Table S4, the levels of 20:4ω6 in high MO, low MO, FO, and FO/VO were 0.44, 0.43, 1.85, and 1.41 mg/g, respectively. Thus, the higher levels of PGE2 in the salmon muscle tissue fed with FO and FO/VO compared to MO diets might be due to higher contents in their precursor molecules (20:4ω6) in the diets.
PGE2 plays a significant role in modulating signaling, maintaining homeostasis, and facilitating pathogenic mechanisms in animals; there are health-beneficial effects in reducing neurodegenerative diseases, cancer, central nervous system injury, and neuropsychiatric conditions as well [
12]. Thus, PGE2 in salmon muscle tissue is a significant molecule in sustaining the metabolism and homeostasis of humans and animals.
In terms of PGF3α, given that the FO diet contains high EPA, the precursor molecule for the biosynthesis of PGF3α, as shown in
Table S4, the high level of PGF3α in salmon muscle tissue after FO feeding might be due to a direct dietary effect. However, this pattern was not well reflected in the high MO diet, which gave the second highest PGF3α concentration in salmon muscle tissue even with the low level of dietary EPA. Therefore, as mentioned in the PGE2 section, this result indicates that there are other indirect factors influencing the biosynthesis of PGF3α in salmon muscle tissue.
The techniques used in the present study would be a useful means for analyzing lipid species in a variety of foods, in particular, monitoring the changes in the PhL and TAG molecules as affected by food processing. As proven through the present study, shotgun-based mass spectrometry analysis displayed an effective performance in the identification and quantification of TAG species containing specific fatty acyl chains (i.e., DHA, EPA, ARA, etc.); thus, this high throughput can be widely utilized in exploring other lipid species such as sterol ester, wax ester, sphingomyelin (SM), cardiolipin (CL), and galactosylceramide (GalCer) in diverse plant- and animal-based foods.
Although shotgun-based lipidomics is a high throughput approach with numerous advantages, classical shotgun lipidomics has some limitations including ambiguous identification of isobaric lipid molecules, the presence of nominal mass, matrix effect, isomers that generate almost identical fragmentation patterns, and fatty acids with other linkages [
37,
38]. Thus, we are planning to carry out additional research to address the above drawbacks of shotgun-based lipidomics as well as TLC and chemical quantifications for PC, PE, PI, and PS and following comparisons with HPLC-MS/MS.
4. Materials and Methods
4.1. Materials
The microbial oil (MO) isolated from Schizochytrium sp. thraustochytrids was provided by Mara Renewables Corporation (Dartmouth, NS, Canada). Standards, including 18:1-18:1-phosphatidylcholine, 18:1-18:1-phosphatidylethanolamine, 16:0-16:0-phosphatidylserine, and 18:1-18:1-phosphatidylinositol, used for the identification and quantification of phospholipid species were obtained from Sigma-Aldrich Canada Ltd. (Oakville, ON, Canada). PGE2 and PGF3α standards were purchased from Cayman Chemical Company (Ann Arbor, MI, USA). Sodium hydroxide, chloroform (HPLC grade), and methanol (HPLC grade) were obtained from Fisher Scientific Co. (Nepean, Ottawa, ON, Canada).
4.2. Experimental Fish
Preparation of experimental diets including high MO, low MO, fish oil (FO), and the mixture of fish and canola oil (FO/VO), experimental design, growth performance, and sampling procedures were carried out as summarized by Wei et al. [
9] Briefly, salmon (~25 g initial weight) were fed experimental diets in triplicate tanks for 16 weeks in freshwater (12 °C, 100% oxygen saturation) and were hand fed to satiation twice daily. Their initial lengths were from 13.3 to 16.2 cm, and their final lengths ranged from 21.3 to 22.4 cm, showing approximately 8.0 cm of growth over 16 weeks. Two fish per tank, a total of six fish per group, were euthanized by injecting an overdose of buffered tricaine methane sulfonate (TMS) at a concentration of 400 mg/L, and their clinical death was ensured before sampling. The ethical treatment of salmon used in this research followed guidelines as described by the Canadian Council of Animal Care. The skin was removed on the left side and white dorsal muscle tissue was extracted and immediately frozen and stored at −80 °C. The fatty acid content of the experimental diets is provided in
Table S4.
4.3. Lipid Extraction
The procedure for the extraction of crude lipids in salmon muscle tissue was conducted as described by Parrish [
39], with minor modifications. Briefly, 250 mg of salmon muscle tissue, 2 mL of ice-cold chloroform, and 1 mL of methanol were mixed in a 15 mL glass vial and then homogenized using a blender. The metal rod of the blender was subsequently washed with 0.5 mL of chloroform-extracted water and 1 mL of chloroform: methanol (2:1). The mixture was then sonicated for 4 min in an ice bath and centrifuged at 3500×
g for 2 min. A double pipetting technique was used to transfer the organic layer (bottom) containing lipophilic molecules to a clean glass vial using glass Pasteur pipettes. This procedure was repeated three times, and all organic layers were combined into the vial, followed by removing organic solvent using nitrogen evaporation. Finally, the remaining crude lipid in the vial was re-dissolved with chloroform and kept at −20 °C until further use.
4.4. ESI-MS/MS Analysis for the Identification and Quantification of PhLs
The identification of PhLs and their semi-quantitative analysis in salmon muscle tissue was conducted using a triple quadrupole mass spectrometer (TSQ Quantis™ Triple Quadrupole Mass Spectrometer, Thermo Fisher Scientific, Waltham, MA, USA) after electrospray ionization (ESI) as described by Yeo and Parrish [
19]. Briefly, the lipid extract of salmon muscle tissue was diluted with 1:4 chloroform: methanol (
v/
v) to prepare the final solution at a concentration of 25 mg/mL, followed by the inclusion of 10 μL of sodium hydroxide in the sample solution for the formation of sodium adducts of PhLs. The sample solution was injected directly into the mass spectrometer using a high-pressure syringe pump (F100T2, Chemyx Inc., Stafford, TX, USA) at a flow of 10 μL/min. The analysis of phosphatidylcholine (PC) species was carried out in the positive mode, and the conditions of the mass spectrometer were set to 325 °C ion transfer tube temperature, 4000 (v) ion spray voltage, 10 (arbitrary) aux gas, 50 (arbitrary) sheath gas, 1 (arbitrary) sweep gas, and 30 °C vaporizer temperature. The identification of the PC moiety and two fatty acids of PCs was carried out in NL scanning mode, and for the collision energy, collision-induced dissociation (CID) gas was set at 37 eV and 1 mTorr. The identification of phosphatidylethanolamine (PE), phosphatidylserine (PS), and phosphatidylinositol (PI) was carried out in the negative mode along with multiple precursor ion scans. The conditions for mass spectrometry analysis were set as follows: 325 °C ion transfer tube temperature, 2500 (v) ion spray voltage, 13.8 (arbitrary) aux gas, 2.0 (arbitrary) sheath gas, 0.8 (arbitrary) sweep gas, and 30 °C vaporizer temperature. The same condition of collision energy and CID gas was used for the MS/MS analysis.
Multiple NL scans were utilized in the positive mode for the identification of PC species. The NL at 183.1 accounts for the loss of phosphatidylcholine moiety in the chemical structure of PCs, and other NL corresponding to different fatty acids (i.e., NL 256.4 for palmitic acid, NL 328.4 for DHA, NL 302.4 for EPA, etc.) were employed for the identification of target PC species in the salmon muscle tissue. On the other hand, the identification of PEs, PSs, and PIs was carried out through the precursor ion scan mode in the negative mode.
For the semi-quantitative analysis, isotopic deconvolution was carried out to obtain the corrected peak area of lipids as described by Li [
4], that is, it was carried out by applying the following formula Z
M = [(NC × 1.12) × (NC × 1.12)/200 + (No × 0.204)]/100. Here, Z
M indicates the isotopic correction factor, which is further used to yield the corrected peak signal below. N
C and N
O denote the numbers of carbon and oxygen atoms in the product ion of the diacylglycerol generated in the fragmentation of molecule M, implying that the number of carbons and oxygens in the lipid species affects the intensities of isotopic signals. Then, the corrected peak signal (
CI
M+2), in which M + 2 indicates a molecule having two more molecular mass than molecule M and (
CI
M+2) means the corrected intensity of molecule M + 2, was deduced through the formula
CI
M+2 = I
M + 2 −
CI
M × Z
M, in which I
M+2 indicates the raw signal intensity (before isotopic deconvolution) of lipid species, and
CI
M denotes the corrected signal of lipid molecule M. The corrected peak signals of the PhL species were utilized for the calculation of their final quantification using external standards including 18:1_18:1-PC, 18:1_18:1-PE, 16:0_16:0-PS, and 18:1_18:1-PI. The equation deduced by a standard curve of 18:1_18:1-PC in an NL scan at 183.1 in the positive ion mode was used for the calculation of the level of PC species in salmon muscle tissue. The same approach was used for the calculation of the level of PE, PS, and PI by applying precursor ion scanning at 196.0, 153.0, and 241.0 in the negative ion mode, respectively.
4.5. ESI-MS/MS Analysis for the Identification and Quantification of TAG Species
The identification and semi-quantitative analysis of TAGs using a triple quadrupole mass spectrometer were carried out as described by Yeo and Parrish [
19]. Briefly, 16 mM ammonium acetate was added to the lipid extract (final concentration) to form an ammonium adduct, and then 500 µL of the lipid extracts were introduced directly into the mass spectrometer at a flow rate of 10 μL/min. The identification of TAG species and their quantitative analysis were conducted in the positive mode with the following mass spectrometer conditions: 5000 (v) ion spray voltage, 0.4 (arbitrary) sweep gas, 16.3 (arbitrary) aux gas, 30 °C vaporizer temperature, and 325 °C ion transfer tube temperature. The collision energy for the fragmentation of molecular ions was set at 28 eV.
The identification of TAG molecules was carried out by confirming three fatty acid moieties of TAGs using the multiple NL scans, followed by data processing using Xcalibur Software (Version 4.3, Thermo Fisher Scientific, Waltham, MA, USA).
A standard curve of triolein (18:1_18:1_18:1) was created for the quantification of the TAG species identified. The correction of each peak area was carried out by calculating isotopic deconvolution as described by Li [
4] as explained in the PhL semi-quantification section. Then, those corrected peak areas were applied to the equations of the standard curve of triolein, followed by the quantification of TAG species. In NL scanning mode, fatty acids have different instrumental responses, in particular the higher signal intensities for DHA and EPA. Thus, in the present study, triolein was chosen for the quantitative analysis due to its average NL response among other TAG molecules. The identification of TAG species was carried out as described in the phospholipid section using multiple NL scans.
4.6. ESI-MS/MS Analysis for the Identification and Quantification of PGE2 and PGF3α
Prior to the analysis of PGE2 and PGF3α for creating a standard curve and running samples, the optimization process for searching for the best analysis parameters of the mass spectrometer was carried out. Then, the optimized conditions were applied to the analysis of standards and samples under the same environment. The identification of PGE2 and PGF3α was carried out in the negative mode along with the following analysis conditions: 4490 (v) ion spray voltage, 4.0 (arbitrary) sheath gas, 6.1 (arbitrary) aux gas, 7.1 (arbitrary) sweep gas, 18.26 eV of collision energy, ion transfer tube temperature (325 °C), and vaporizer temperature (30 °C). The quantitative analysis of PGE2 and PGF3α was conducted by using external standards.
4.7. Statistical Analysis
Independent replicates (duplicate) for three tanks, representing six salmon per group, were used to carry out statistical analyses, and those values were used for normality tests and one-way ANOVA. Each mean was for a total of six samples per treatment and was analyzed using Tukey’s HSD test (p < 0.05) in SPSS 16.0 (SPSS Inc., Chicago, IL, USA). PCA analysis was also carried out using the SPSS package.