Abstract
The main objective of the present study was to assess culturable airborne fungal concentrations, and types in different seasons. Two-stage viable impactor samplers were used with malt extract agar medium as the collection media. Culturable airborne fungal concentrations were collected indoors and outdoors of 43 homes in urban and rural environments from November 2008 to October 2009 in Egypt. Fungal concentrations were significantly higher in the rural environment than the urban environment. The median indoor and outdoor total fungal concentrations were 608 and 675 CFU/m3 in the urban environment and 1,932 and 1,872 CFU/m3 in the rural environment, respectively. The greatest concentrations were found in the autumn and spring season. Indoor and outdoor concentrations were significantly correlated (P < 0.001). The highest concentrations were observed in the fungal size range of <8 µm (fine fraction). The indoor/outdoor (I/O) ratios were not statistically different between seasons. Alternaria, Aspergillus, Cladosporium, Penicillium and yeasts were the predominant genera indoors and outdoors, and the abundance of genera varied by season and region. This study is of a potential interest as little reported research on the indoor fungal air quality from Egypt.
1. Introduction
Fungal organisms are common in indoor and outdoor air environments. They are known to induce irritation, infection, allergy, asthma, and toxic effects [1,2]. Fungi may produce mycotoxins that may cause several diseases in humans and animals. Knowledge of indoor airborne fungi is important, as various fungi could be common in indoor air of occupational and non-occupational buildings.
In the recent years, assessment of buildings for evidence of indoor fungal growth has dramatically increased [3,4]. Many studies have focused on fungal concentrations in problematic or moldy contaminated buildings [5,6], and few references for normal buildings without mold damage [7,8]. Nevalainen et al. reported that the presence of indoor fungi and air quality problems could still occur in buildings without moisture or mold damage [9]. Fungal organisms are known to induce irritation, infection, allergy, asthma, and toxic effects [5,6].
The concentrations and types of airborne fungi depend on time of day, climatic conditions, geographical location and type of vegetation [10]. Airborne fungal concentrations in homes and schools ranged between 102 and 104 CFU/m3 in warm climates [11], and averaged 102 CFU/m3 in cold subarctic climates [12]. Airborne fungal concentrations exceeded 104 CFU/m3 in the moldy homes, and <200 CFU/m3 in the reference dwellings (without mold growth), with Penicillium, Aspergillus, Cladosporium and Alternaria being the common genera found [13]. In Poland, fungal concentrations averaged 60 CFU/m3 in healthy homes in winter, and >800 CFU/m3 in moldy homes in summer, with the maximum concentration reaching 17,000 CFU/m3 [14]. There is a growing interest in indoor air quality as the concentrations and prevalence of fungi in a particular region help identify the association between residential exposure, clinical diagnosis, and in the prevention of seasonal allergic diseases [15]. The aim of the present study was to assess concentrations, types and frequency of distributions of indoor and outdoor fungi in the air of urban and rural homes without a suspected or known problem, in order to gain data on home-indoor air quality in Egypt.
2. Experimental Section
2.1. Sampling Strategy and Sites
Air samples were collected indoors and outdoors, from 43 homes with no obviously visible fungal growth or existing water leaks (within the past six months). The samples were taken between 12.00 a.m. and 6.00 p.m. over a period of 12 months between November 2008 and October 2009.
Table 1 shows the housing characteristics of the investigated homes. Air samples were taken from 26 homes with an average size of 100 m2 in the urban environment across Greater Cairo (30°02" N and 31°20" E), and from 17 homes, with an average size of 130 m2, in the rural village located in "Dakahlia governorate" (30.5°–31.5° N and 30°–32° E), 150 km northeast of Cairo.

Table 1.
Characteristics of the homes under the investigation.
| Characteristic | Urban (%) | Rural (%) | |
|---|---|---|---|
| Age of building | 1–5 years | 7 | 0 |
| 5–10 years | 22 | 6 | |
| 10–15 years | 15 | 6 | |
| >15 years | 56 | 88 | |
| Number of occupants | 1–2 person | 11 | 19 |
| 3 person | 4 | 0 | |
| 4 person | 41 | 13 | |
| ≥5 person | 44 | 69 | |
| Type of building | Single house | 0 | 19 |
| Small complex (2–3 apartments) | 0 | 81 | |
| Big complex (>5 apartments) | 100 | 0 | |
| Pets | Yes | 4 | 50 |
| No | 96 | 50 |
Temperature and relative humidity were measured during the course of the study. All homes had natural ventilation and no home had air-conditioning. Temperatures ranged between 20–42 °C indoors and 13–42 °C outdoors in the urban area, and 15–31 °C indoors and 12–33 °C outdoors in the rural area. Indoor and outdoor relative humidity ranged between 25–90% and 28–84%, respectively, in urban areas and 31–82% and 31–81% in the rural area. Egypt is a desert climate with very low rainfall (http://www.climate-zone.com/climate/egypt/). Egypt is characterized with moderate winter months, (December–February), warm spring (March–May) and autumn (September–November) months, and hot summer (June–August) months. Greater information on the environmental conditions can be found in the companion publication to this study that focused on bacterial bioaerosols [16].
2.2. Sampling of Fungi
Air samples were collected using the two-stage viable cascade impactor sampler (TE-10-160, Tisch Environmental, Cleves, OH, USA), separating particles into two size ranges: fine (<8 µm) and coarse (>8 µm) [2]. The particles ≥8 μm are usually deposited in the upper respiratory tract and can contribute to allergic rhinitis and asthma [15], however particles ≤8 μm can penetrate into the alveoli contributing to allergies [17].
Indoor samples were collected at a height of 1.5 m, the human breathing zone, above the floor level in the middle of the main common room or living room while outdoor comparison samples were taken approximately 5–10 m outside the entrance doors or on the roofs according to the location of homes inside the buildings. Sampling was limited so that no samples were collected within 48 h of a rain event or if daily wind speeds were predicted to exceed 15 km/h. The sampler was operated at the manufacturer recommended flow rate of 28.3 L/min for 2 min, to prevent overloading the collection plates. Petri dishes containing malt extract agar (BD, Sparks, MD, USA) supplemented with 50 ppm chloramphenicol (Oxoid, Hampshire, UK) were used to collect fungi. Each home was scheduled for sampling twice per season, and because of the short sampling time two consecutive indoor samples, at 12.00 and 15.00 p.m. where taken in parallel to the outdoor samples during every sampling event [2].
The Petri dishes were incubated at 28 °C for 5–7 days and colonies were counted with replicate plates used to calculate a mean. Positive-hole correction [18] was applied to the raw CFU recovered on each plate then used along with the sampling time and flow rate to calculate the concentration with final concentration expressed as colony forming units per cubic meter of air (CFU/m3). The recovery range for fungal organisms in this study ranged from a minimum of 17 to a maximum of 20,770 CFU/m3. Fungal isolates were purified and identified by direct observation on the basis of micro and macro-morphological features, reverse and surface coloration of colonies on Sabouraud dextrose agar, Czapek dox agar and Malt extract agar [2,19,20,21,22].
2.3. Statistical Analysis
The data were analyzed using descriptive statistics, such as median, mean, and percentiles. In addition, data analyses were conducted using non-parametric statistics, which do not require distributional assumptions (normal distribution). The Kruskal–Wallis test of the equality of medians is a non-parametric method was used to compare two or more populations (P < 0.05). The assumption for this test is that the samples from the different populations are independent random samples from continuous distributions with the distribution having a similar shape. Spearman’s rank (rs) correlation test was used to determine the linear relationships between the indoor and outdoor concentrations.
3. Results
3.1. Overall Concentrations
Table 2 shows the range, median, mean and median I/O ratios of fungal concentrations in the urban and rural homes. The concentrations ranged between 65 and 34,784 CFU/m3 in the urban area, and 67 and 16,492 CFU/m3 in the rural area. Fungal concentrations were to be higher in the rural environment than in the urban environment (P < 0.0002). Significant positive correlations (P ≤ 0.001) were found between the indoor and outdoor concentrations in both the urban (rs = 0.79) and the rural (rs = 0.81), and no significant differences were detected between both areas.
Fungal concentrations in the size fraction < 8 µm were higher than those of >8 µm, and constituted ~84% of the total organisms recovered. The median I/O ratios of fine and coarse fractions, respectively, were 0.789 and 0.91 in the urban area, and 1.14 and 1.07 in the rural area. I/O ratios for total fungal counts were 0.908 in the urban and 1.03 in the rural areas (Table 2). Generally, indoor fungal concentrations exceeded outdoor concentrations in the rural area.

Table 2.
Indoor and outdoor range, mean and median fungal concentrations (colony forming units per cubic meter of air), and Indoor/Outdoor ratio for coarse, fine, and total organism sizes.
| Coarse (> 8µm) | Fine (< 8µm) | |||||
|---|---|---|---|---|---|---|
| Indoor | Outdoor | Indoor | Outdoor | Indoor | ||
| Urban (n = 26) | ||||||
| Range | 1–641 | 8–742 | 43–4,108 | 32–3,076 | 65–4,523 | 67–3,697 |
| Mean | 129 | 174 | 765 | 810 | 894 | 984 |
| Median | 101 | 128 | 495 | 541 | 608 | 675 |
| I/O ratio a | 0.789 | 0.91 | 0.908 | |||
| Rural (n = 17) | ||||||
| Range | 40–2,744 | 1–4,604 | 67–32,040 | 120–14,264 | 111–34,784 | 360–16,492 |
| Mean | 572 | 613 | 3,273 | 2,854 | 3845 | 3467 |
| Median | 404 | 353 | 1,537 | 1,441 | 1,932 | 1,872 |
| I/O ratio a | 1.14 | 1.07 | 1.03 | |||
a the ratio was calculated by dividing the median of indoor concentration by the corresponding median of the outdoor concentration.
3.2. Seasonal Concentrations
The range, median, mean (CFU/m3) and I/O ratios of the total fungal concentrations in the different seasons are shown in Table 3.
The greatest concentrations were found in the autumn (P < 0.05). The greatest I/O ratios were found in the summer in both the rural and urban environment. No significant differences (P > 0.05) were found between the I/O ratios between any season. The Spearman rank correlation analyses was utilized to evaluate correlations between environmental conditions (temperature and relative humidity) and fungal concentrations; however, no correlation was found.
The seasonal distributions of the fine and coarse fractions are shown in Figure 1. Fine fractions were generally greater than those of coarse fractions in all seasons in both urban and rural areas. The lowest concentration of the coarse fraction was found in the urban area. The highest median indoor and outdoor concentrations for the size fraction >8 µm were found in the winter (Figure 1(a)) and <8 µm in the autumn (Figure 1(b)), in the urban area, however the highest median concentrations for both size fractions were found in the spring (Figure 1(c,d)) in the rural area.

Table 3.
Total Indoor and outdoor range, mean and median fungal concentrations (colony forming units per cubic meter of air), and Indoor/Outdoor ratio by season.
| Winter | Spring | Summer | Autumn | |||||
|---|---|---|---|---|---|---|---|---|
| Outdoor | Outdoor | Outdoor | Outdoor | |||||
| Urban (n = 26) | ||||||||
| Range | 317–2,868 | 283–3,697 | 67–2,600 | 194–3,236 | ||||
| Mean | 887 | 829 | 706 | 1,500 | ||||
| Median | 763 | 516 | 511 | 1,225 | ||||
| I/O ratio a | 0.81 | 0.88 | 1.06 | 0.746 | ||||
| Rural (n = 17) | ||||||||
| Range | 449–6,323 | 3,052–16,492 | 360–3,200 | 704–2,712 | ||||
| Median | 2,028 | 7,636 | 988 | 1,509 | ||||
| Mean | 20,730 | 21,073 | 9,782 | 17,143 | ||||
| I/O ratio a | 0.84 | 1.13 | 1.49 | 1.21 | ||||
a the ratio was calculated by dividing the median of indoor concentration by the corresponding median of the outdoor concentration.
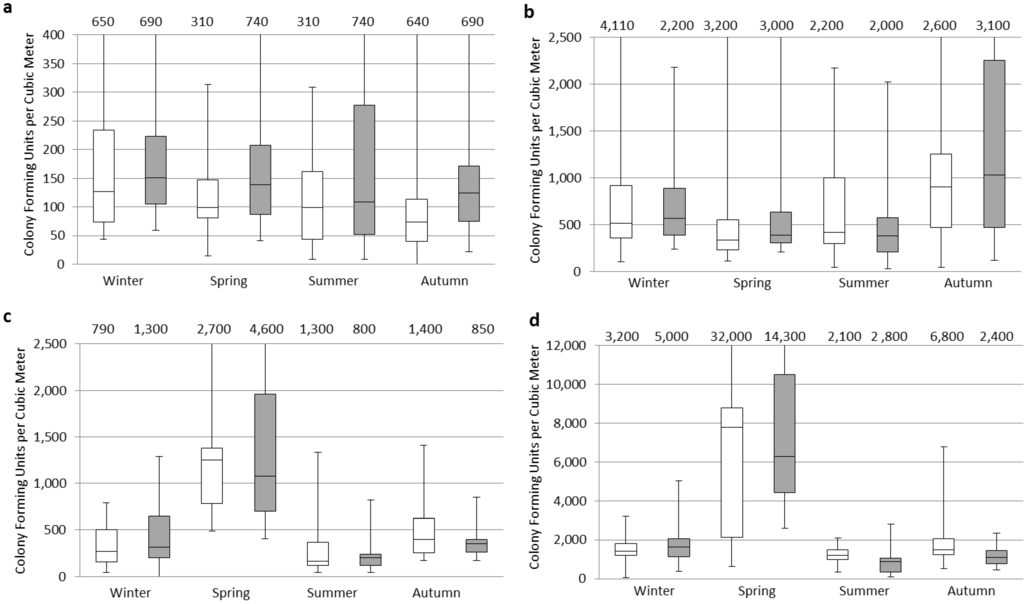
Figure 1.
Culturable airborne fungal concentrations measured indoors and outdoors in the rural and urban sites in Egypt. The box plots represents indoors (white) and outdoors (grey) where: (a) Urban > 8 µm; (b) Urban < 8 µm; (c) Rural > 8 µm; and (d) Rural < 8 µm. Note: Y-axis is scaled differently in each graph.
Figure 1.
Culturable airborne fungal concentrations measured indoors and outdoors in the rural and urban sites in Egypt. The box plots represents indoors (white) and outdoors (grey) where: (a) Urban > 8 µm; (b) Urban < 8 µm; (c) Rural > 8 µm; and (d) Rural < 8 µm. Note: Y-axis is scaled differently in each graph.
3.3. Identification of Fungi
The counts and percentages of the predominant identified fungi are found in Table 4. The ranking of the most numerous genera were similar in both the urban and rural areas, with Alternaria, Aspergillus, Cladosporium, Penicillium and yeasts as the predominant types indoors and outdoors.

Table 4.
The total number of organisms recovered (CFU/m3) and percentages of the most prevalent fungal organisms recovered in urban and rural areas.
| Urban | |||||
|---|---|---|---|---|---|
| Indoor | Outdoor | Indoor | |||
| % | % | % | |||
| Alternaria | 1.4 | 2.2 | 2.4 | ||
| Aspergillus spp. | 33.9 | 27.8 | 7.9 | ||
| Cladosporium | 28.3 | 36.3 | 67.2 | ||
| Fusarium | 1.2 | 1.0 | 2.3 | ||
| Penicillium | 27.9 | 25.6 | 12.0 | ||
| Yeasts | 2.7 | 2.3 | 2.4 | ||
| Other | 4.5 | 4.8 | 5.7 | ||
| Sum of all Samples | 100 | 100 | 100 | ||
Aspergillus flavus and Aspergillus niger were the dominant Aspergillus species. Aspergillus flavus and Penicillium were higher in the urban homes, while Alternaria and Cladosporium in the rural homes. Aspergillus spp., including Aspergillus flavus and Aspergillus niger, Fusarium, Penicillium, and yeasts were more numerous indoors, while Alternaria and Cladosporium were more numerous outdoors. Microsporium, Trichophyton, and water indicator fungi (Stachybotrys and Ulocladium) were found in low counts. Aspergillus candidus, Aspergillus clavatus, Aspergillus lucknowensis, Aspergillus parasiticus, Drechslera and Trichoderma as well as Aspergillus egypticus, Gliocladium, Monilia, Stachybotrys and Trichophyton were found indoors and not outdoors in the urban and rural homes, rerspectively.
Table 5 shows the counts of the predominant identified fungal types recovered by season. Aspergillus spp., including Aspergillus fumigatus dominated in the summer, Claodsporium in the autumn and Penicillium in the winter. Yeasts were found in high counts in the summer. Fusarium was found in higher counts in the summer in the rural area. Although not in Table 5, Drechslera and Epicoccum were detected in the warm months, and Ulocladium and Stachybotrys in the wet months.
3.4. Frequency of the Occurrence of Fungal Genera by Season
The percentages of homes positive for identified fungal genera by season are shown in Table 6. Alternaria, Aspergillus spp, Penicillium, and Cladosporium were frequently found in all homes in every season. Aspergillus flavus was recovered indoor in percentages ranged from 73–96% in the urban, and 41–70% in the rural area, in different seasons, with the maximum in the spring and summer. Aspergillus niger was recovered in 100% and Alternaria in 15% of the homes investigated in the winter in the urban area. Alternaria was recovered in 82% of samples indoors and outdoors in the spring, Cladosporium in 100% in the winter, spring and autumn, and Penicillium in all homes in the autumn in the rural area. Although not shown in Table 5, Stachybotrys was found indoors in the winter (11.7%) in the rural area.
4. Discussion
Many studies have been performed to identify the concentrations and types of airborne fungal organisms in buildings all over the world. Almost all studies have focused on problematic buildings; however few reported research non-problematic building or on indoor air quality from Egypt.
In the present study, fungal concentrations were higher in the rural environment and may be a result of outdoor fungal sources, such as “animal shelters, solid-waste, composts, plant debris and vegetation”. The rate of air exchange may be higher in the rural homes where the rate of air ventilation from outdoor to indoor may help increase the indoor fungal concentrations. In milder climates the outdoor fungal organisms are the largest source of indoor concentrations of fungi and the outdoor concentration can routinely exceed of 103 CFU/m3 [23]. In addition indoor fungi are directly dependent on building type, hygienic rules and ventilation [24]; life style and residential characteristics [15,25], and the rural homes in this study were characterized by higher ventilation rates, larger areas and dusty floors.

Table 5.
The total counts (CFU/m3) for the indoor (In) and outdoor (Out) most prevalent fungal organisms recovered by season.
| Urban | Rural | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Winter | Spring | Summer | Autumn | Winter | Spring | Summer | Autumn | |||||||||
| Out | Out | Out | Out | Out | Out | Out | Out | |||||||||
| Alternaria | 135 | 959 | 975 | 30 | 819 | 6,380 | 880 | 291 | ||||||||
| Aspergillus spp. | 6,804 | 3,860 | 7,989 | 8,136 | 3,896 | 800 | 5,104 | 3,779 | ||||||||
| Cladosporium | 65 | 7,145 | 4,150 | 19,086 | 31,689 | 277,714 | 4,148 | 11,117 | ||||||||
| Fusarium | 104 | 1,560 | 288 | 0 | 599 | 1,560 | 2,924 | 639 | ||||||||
| Penicillium | 8,340 | 6,021 | 1,750 | 8,568 | 4,805 | 45,428 | 3,176 | 4,421 | ||||||||
| Yeasts | 194 | 1,123 | 884 | 37 | 555 | 4,144 | 1,160 | 316 | ||||||||
| Other | 1,154 | 8,181 | 5,318 | 20,394 | 35,863 | 278,914 | 5,358 | 14,161 | ||||||||
| Total | 16,731 | 21,704 | 17,204 | 37,165 | 46,537 | 337,226 | 18,602 | 23,607 | ||||||||

Table 6.
Frequency of occurrence (n = 26-urban, n = 17-rural) as percent of homes positive for most prevalent fungal organisms for the indoor (In) and outdoor (Out) by season.
| Winter | Spring | Summer | Autumn | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Urban | Rural | Urban | Rural | Urban | Rural | Urban | Rural | |||||||||
| Out | Out | Out | Out | Out | Out | Out | Out | |||||||||
| Alternaria | 30 | 58.8 | 65 | 82 | 61.5 | 53 | 3.8 | 29 | ||||||||
| Aspergillus spp. | 100 | 82.4 | 100 | 64.7 | 92.3 | 88.2 | 88.5 | 100 | ||||||||
| Cladosporium | 88.4 | 100 | 53.8 | 100 | 42 | 53 | 88 | 100 | ||||||||
| Fusarium | 15 | 53 | 53 | 76 | 38 | 88 | 0 | 64 | ||||||||
| Penicillium | 100 | 100 | 84 | 94 | 53 | 88 | 88 | 100 | ||||||||
| Yeasts | 15 | 47 | 69 | 100 | 50 | 64 | 2 | 41 | ||||||||
| Other | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | ||||||||
The indoor to outdoor ratio is used to compare the distribution and assess the potential sources of fungi. In the present study, most of the I/O ratios were close to one, with the slightly higher values in the rural area (Table 3) suggesting that not all indoor fungi originated from outdoors, which could be an indication of lesser indoor air quality. Hidden mold conditions, grains storage, dusty air and building materials may play a role in increased indoor fungal concentrations in the rural homes. The lowest I/O ratio that was found in the winter in the rural area may be attributed to less opening of windows that reduces outdoor infiltration of fungal organisms and decreases the presence of indoor dusty air from outdoor agricultural activities. On the other hand the lowest I/O ratio in urban area detected in autumn could be a result of increasing plant debris in the outdoor environment contributing to fungal concentrations outdoors.
In the present study, fungal concentrations were found to be higher than those reported in previous studies around the world. Indoor and outdoor concentrations, respectively, averaged 82 CFU/m3 and 540 CFU/m3 across the US [26]; 89 CFU/m3 and 68 CFU/m3 in Cincinnati, OH, USA [27]; and 812 CFU/m3 and 1,042 CFU/m3 in Latrobe Valley, Australia [28]. Although results of several studies are similar to those of our study with indoor and outdoor concentrations, respectively, in 820 healthy (or reference homes) residential buildings ranged between 68–2,307 CFU/m3 and 400–80,000 CFU/m3 in the USA [29] and 264–17,788 CFU/m3 and 123–5,771 CFU/m3 in Mexico [30]. Moreover, in Egypt, fungal concentrations were found in the range of 10–102 CFU/m3 in the indoor air of the Church of Saint Katherine Monastery in Sinai [31], and 52–124 CFU/m3 indoors and 25–222 CFU/m3 outdoors at the coastal buildings in Domitta [32].
There are no guidelines for airborne fungi, however a number of numeric guidelines have been proposed throughout the years [29,33,34,35,36], but none of them are currently widely accepted by the scientific community [7]. In comparison with the previously suggested guidelines, our findings indicated that the Egyptian’s homes had much higher than acceptable levels of fungi. The indoor air quality of the Egyptian's homes may be considered a poor since fungal concentrations exceeded 500 CFU/m3 suggested by the World Health Organization [35] and Singapore [36] as well as 200 CFU/m3 by the American Conference of Governmental Industrial Hygienists [37]. In the present study, fungal concentrations in the size range of ≤8 µm (84.26%) was the predominant fraction. This was similar to the respirable fungal fraction concentrations that accounted for 70–80% of the total fungi in American and Taiwanese homes [2,38,39], and 79–98% in Mexico [30].
In most parts of the world the main core of fungal aerosols is likely to be similar, but the dominance of genera may differ from one area to another depending on geographical location, local sources, and climatic conditions [40]. Qualitative determination of fungi may be more useful than determining concentrations, as many species may have health effects. The frequent detection of Aspergillus spp., Penicillium, and Cladosporium is attributed to their ready dissemination into the air. These findings in our study are similar to those observed in other geographical locations in Italy [41], Egypt [32,42], and Florida, USA [7].
Horner et al. [4] grouped fungi into three categories with different ecological relevance: (1) phyloplane fungi (Cladosporium, Curvularia and Alternaria); (2) soil fungi (Penicillium, Paecilomyces and Aspergillus), and (3), water indicator fungi (Chaetomium, Stachybotrys and Ulocladium). In the present study water indicator fungi were found in low counts, and their presence associated with rain time or the presence of damped materials, however phyloplane and soil fungi were found in higher counts in the rural environment.
Cladosporium contains species that commonly grow indoors (C. sphaerospermum) and outdoors (C. cladosporioides and C. herbarum) [43]. Yeasts and Cladosporium dominate healthy homes while Absidia and Alternaria are more likely in moldy homes, and Penicillium and Cladosporium in both healthy and moldy homes [14]. Alternaria has an affinity for outdoor substrates, and when its I/O ratio exceeds one, this could indicate the presence of abnormal indoor conditions. The presence of Aureobasidium and Eurotium in rural homes is an indication of the presence of cellulotic materials and bad storage conditions, respectively. Moreover, the presence of some fungal species in indoor not outdoors mainly Stachybotrys may indicate hidden mold conditions particularly in the rural homes.
Regarding season, Cladosporium, Penicillium and yeasts dominated in the winter, while Aspergillus spp. in the summer. High concentrations of Aspergillus spp. and Alternaria in the autumn are associated with the decaying vegetable materials [44]. Penicillium was found in the highest concentration in the months with low temperatures, as precipitation seemed to optimize their sporulation [45]. Hargreaves et al. [46] isolated maximum concentrations of Alternaria in the spring, as the suitability of humidity and temperature and vegetation debris [25].
Cladosporium, Penicillium and Alternaria could increase the risk of asthma and allergic rhinitis and allergic alveolitis [47]. Acremonium, Alternaria, Aspergillus, Cladosporium, Fusarium, Paecilomyces, Penicillium, Stachybotrys and Trichoderma are well known mycotoxin producers [48]. Aspergillus flavus and Aspergillus fumigatus can lead to aspergillosis [49], and Microsporium and Trichophyton are agents of dermatophytoses [50]. As the potential implications of the fungal contamination on health have not been studied in Egypt, it should to be reported that exposure to such concentrations and types is a risk factor for resident’s respiratory symptoms.
In the present study, the results represent the normal mycoflora in non-problem homes and describe the typical background concentrations in the indoor air environment. It should be mentioned that, the main limitations in the present study were: (1) short indoor sampling times that may not provide representative samples or accurately reflect exposures; (2) outdoor samples may not be representative since the apartment complexes restricted the airflow; and (3) geographical regions were large and could contain localized sources, and (4) explanations in discussion section regarding fungal concentration variations are speculation and not supported by local measurements. However, we believe that all buildings should be thoroughly inspected for visible signs of moisture. In addition good ventilation, household maintenance, minimizing dust generation and applying a proper and routine cleaning should be applied.
5. Conclusions
Indoor culturable airborne fungi were found in concentrations relatively similar to those outdoors of the homes. Fungal concentration and composition varied by season and no significant differences were found between the rural and urban homes and indoors and outdoors concentrations. Fungal organisms of the size fraction <8 µm represented 84.26% of the total fungal counts with the greatest counts found in the spring and autumn. I/O ratios slightly exceeded one for coarse, fine and total fungi in the rural area, and the highest ratios were found in the summer season for both rural and urban environments. Knowledge on airborne fungi is important as it is considered a potential public health problem and data can be used as a base to develop criteria for assessing indoor air quality in Egypt. Studies regarding fungal concentrations and types in buildings with moisture and damp materials are needed in the future.
Acknowledgment
This study was partially funded by Dual-Cooperator Research Grant No: 58-3148-8-176 from the USA and Egypt (Science and Technology Development Fund).
References
- Ozkara, A.; Ocak, I.; Korcan, S.; Konuk, M. Determination of fungal air spora in Afyonkarahisar, Turkey. Mycotaxon 2007, 102, 199–202. [Google Scholar]
- Mota, L.; Gibbs, S.; Green, C.; Flores, C.; Tarwater, P.; Ortiz, M. Seasonal fine and coarse culturable fungal constituents and concentrations from indoor and outdoor air samples taken from an arid environment. J. Occup. Environ. Hyg. 2008, 5, 511–518. [Google Scholar] [CrossRef]
- D’Arcy, N.; Canales, M; Spratt, D.; Lai, K. Healthy schools: Standardization of culturing methods for seeking airborne pathogens in bioaerosols emitted from human sources. Aerobiologia 2012, 28, 413–422. [Google Scholar] [CrossRef]
- Horner, W.; Worthan, A.; Morey, P. Air and dustborne mycoflora in houses free of water damage and fungal growth. Appl. Environ. Microbiol. 2004, 70, 6394–6400. [Google Scholar] [CrossRef]
- Koskinen, O.; Husman, T.; Hyvärinen, A.; Reponen, T.; Nevalainen, A. Respiratory symptoms and infections among children in a day care centre with mold problems. Indoor Air 1995, 5, 3–9. [Google Scholar]
- Meklin, T.; Husman, T.; Vepsäläinen, A.; Vahteristo, M.; Koivisto, J.; Halla-Aho, J.; Hyvärinen, A.; Moschandreas, D.; Nevalainen, A. Indoor air microbes and respiratory symptoms of children in moisture damaged and reference schools. Indoor Air 2002, 12, 175–183. [Google Scholar] [CrossRef]
- Codina, R.; Fox, R.; Lockey, R.; DeMarco, P.; Bagg, A. Typical levels of airborne fungal spores in houses without obvious moisture problems during a rainy season in Florida, USA. J. Investig. Allergol. Clin. Immunol. 2008, 18, 156–162. [Google Scholar]
- Sessa, R.; di Pietro, M.; Sehiavoni, G.; Santino, I.; Altieri, A.; Pinelli, S.; del Piano, M. Microbiological indoor air quality in healthy buildings. New Microbiol. 2002, 25, 51–56. [Google Scholar]
- Nevalainen, A.; Hyvarinen, A.; Pasanen, A.L.; Reponen, T. Fungi and bacteria in normal and mouldy dwellings. In Health Implications of Fungi in Indoor Environments; Samson, R.A., Flannigan, B., Flannigan, M.E., Verhoeff, A.P., Adan, O.C.G., Hoekstra, E.S., Eds.; Elsevier: Amsterdam, The Netherlands, 1994; Volume 2, pp. 163–168. [Google Scholar]
- Pepeljnak, S.; Segvic, M. Occurrence of fungi in air and on plants investigation of different climatic regions in Croatia. Aerobiologia 2003, 19, 11–19. [Google Scholar] [CrossRef]
- Su, H.; Wu, P.; Lin, C. Fungal exposure of children at homes and schools: A health perspective. Arch. Environ. Health 2001, 56, 144–149. [Google Scholar] [CrossRef]
- Reponen, T.; Nevalainen, A.; Jantunen, M.; Pellikka, M.; Kalliokoski, P. Normal range for indoor air bacteria and fungal spores in a subarctic climate. Indoor Air 1992, 2, 26–31. [Google Scholar]
- Krikśtaponis, A. Diversity of Fungi Species in Occupational and Residential Environments and their Biological Peculiarities (Toxicity, Pathogenicity, Proteolytic, Lipolytic and Cellulolytic Activity). Ph.D. Thesis, Institute of Botany, Vilnius University, Vilnius, Lithuania, 2000. [Google Scholar]
- Pastuszka, J.; Paw, T.; Lis, O.; Wlazlo, A.; Ulfig, K. Bacterial and fungal bioaerosol in indoor environment in Upper Silesia, Poland. Atmos. Environ. 2000, 34, 3833–3842. [Google Scholar] [CrossRef]
- Seltzer, J.M. Biologic contamination. Occup. Med. State Art Rev. 1995, 10, 1–25. [Google Scholar]
- Abdel Hameed, A.A.; Gibbs, S.G.; Tarwater, P.M.; Casillas, M.E.; Green, C.F. Seasonal evaluation of fine and coarse culturable bacterial aerosols from residences within a rural and an urban city in Egypt. Int. J. Environ. Health 2012. [Google Scholar] [CrossRef]
- Mullins, J.; Seaton, A. Fungal spores in lung and sputum. Clin. Allergy 1978, 8, 525–533. [Google Scholar] [CrossRef]
- Andersen, A. New sampler for the collection, sizing and enumeration of viable airborne particles. J. Bacteriol. 1958, 76, 471–484. [Google Scholar]
- Raper, K.B.; Fennell, D.I. The Genus Aspergillus; Krieger: New York, NY, USA, 1973. [Google Scholar]
- Pitt, J.I. The Genus Penicillium and Its Teleomorphic States Eupenicillium and Talaromyces; Academic Press: London, UK, 1979. [Google Scholar]
- Carmichael, J.W.; Kendrick, W.B.; Conners, I.L.; Sigler, L. Genera of Hyphomycetes; The University of Alberta: Edmonton, Canada, 1980. [Google Scholar]
- Barnett, H.L.; Hunter, B.B. Illustrated Genera of Imperfect Fungi, 4th ed; The American Phytopathological Society (APS): St. Paul, MN, USA, 1999; p. 218. [Google Scholar]
- Levetin, E.; Shaughnessy, R.; Fisher, E.; Ligman, B.; Harrison, J.; Brennan, T. Indoor air quality in schools; exposure to fungal allergens. Aerobiologia 1995, 11, 27–34. [Google Scholar] [CrossRef]
- Jaffal, A.; Banat, I.; El Mogheth, A. Residential indoor airborne microbial populations in the United Arab Emirates. Environ. Int. 1997, 23, 529–533. [Google Scholar] [CrossRef]
- Sen, B.; Asan, A. Fungal flora in indoor and outdoor air of different residential homes in Tekirdag City (Turkey): Seasonal distribution and relationship with climatic factors. Environ. Monit. Assess. 2009, 15, 209–219. [Google Scholar]
- Shelton, G.; Kirkland, H.; Dana Flanders, W.; Morris, K. Profiles of airborne fungi in buildings and outdoor environments in the United States. Appl. Environ. Microbiol. 2002, 68, 1743–1753. [Google Scholar] [CrossRef]
- Lee, T.; Grinshpun, S.; Martuzevicius, D.; Adhikari, A.; Crawford, C.; Reponen, T. Culturability in six single family homes. Atmos. Environ. 2006, 40, 2902–2910. [Google Scholar] [CrossRef]
- Garrett, M.; Hooper, B.; Cole, F.; Hooper, M. Airborne fungal spores in 80 homes in the Latrobe valley, Australia: Levels, seasonality and indoor-outdoor relationship. Aerobiologia 1997, 13, 121–126. [Google Scholar] [CrossRef]
- Gots, R.; Layton, N.; Pirages, S. Indoor health: Background levels of fungi. AIHA J. 2003, 64, 427–438. [Google Scholar] [CrossRef]
- Ponce-Caballero, C.; Ceron-Palma, I.; Lopez-Pacheco, M.; Gamboa-Marrufo, M.; Quintal-Franco, C. Indoor-outdoor fungal aerosols ratios of domestic homes in Merida, Mexico. Ingenierìa 2010, 14, 169–175. [Google Scholar]
- Abdulla, H.; Morshedy, H.; Dewedar, A. Characterization of actinomycetes isolated from the indoor air of the church of Saint Katherine Monastery, Egypt. Aerobiologia 2008, 24, 35–41. [Google Scholar] [CrossRef]
- El Sayed, M. Preliminary survey of indoor and outdoor airborne microfungi at coastal buildings in Egypt. Aerobiologia 2006, 22, 197–210. [Google Scholar] [CrossRef]
- Dutkiewicz, J.; Gôrny, R. Biological factors hazardous to human health: Classification and criteria of exposure assessment (in Polish). Med. Pr. 2002, 53, 29–39. [Google Scholar]
- Rao, C.; Burge, H.; Chang, J. Review of quantitative standards and guidelines for fungi in indoor air. J. Air Waste Manag. Assoc. 1996, 46, 899–908. [Google Scholar] [CrossRef]
- WHO Regional Publications European Series No 31: Indoor Air Quality: Biological Contaminants; World Health Organization: Copenhagen, Denmark, 1988.
- Singapore-Ministry of Environment. Guidelines for Good Indoor Air Quality in Office Premises. 1996. Available online: http://www.nea.gov.sg/cms/qed/indoor_air.pdf (accessed on 10 February 2012).
- Guidelines for the Assessment about Aerosols in the Indoor Environment; American Conference of Governmental Industrial Hygienists: OH, USA, 1989.
- Li, Ch.; Kuo, Y. Microbiological indoor air quality in subtropical areas. Environ. Int. 1993, 19, 233–239. [Google Scholar] [CrossRef]
- Wu, P-C.; Su, H-J.; Lin, C-Y. Characteristic of indoor and outdoor airborne fungi at suburban and urban homes in two seasons. Sci. Total Environ. 2000, 253, 111–118. [Google Scholar] [CrossRef]
- Abdel Wahid, O.; Moustafa, A.; Moustafa, A. Fungal population in the atmosphere of Ismailia city. Aerobiologia 1996, 12, 249–255. [Google Scholar]
- Dacarro, C.; Picco, M.; Grisoli, P.; Rodolfi, M. Determination of aerial microbiological contamination in scholastic sports environments. J. Appl. Microbiol. 2003, 95, 904–912. [Google Scholar] [CrossRef]
- Abdel Hameed, A.; El Hawarry, S.; Kamel, M. Prevalence and distribution of airborne and waterborne fungi and actinomycetes in the Nile River. Aerobiologia 2008, 24, 231–240. [Google Scholar] [CrossRef]
- Chao, H.; Milton, D.; Schwartz, J.; Burge, H. Dustborne fungi in large office buildings. Mycopathologia 2001, 154, 93–106. [Google Scholar]
- Chakraborty, S.; Sen, S.; Bhattacharya, K. Indoor and outdoor aeromycological survey in Burdwan, West Bengal, India. Aerobiologia 2000, 16, 211–219. [Google Scholar] [CrossRef]
- Dubey, S.; Lanjewar, S.; Sahu, M.; Pandey, K.; Kutti, U. The monitoring of filamentous fungi in the indoor, air quality and health. J. Phytol. 2011, 3, 13–14. [Google Scholar]
- Hargreaves, M.; Parappukkaran, S.; Morawska, L.; Hitchins, J.; He, C.; Gilbert, D. Pilot investigation into associations between indoor airborne fungal and non biological particle concentrations in residential houses in Brisbane, Australia. Sci. Total Environ. 2003, 312, 89–101. [Google Scholar] [CrossRef]
- Lacey, J.; Dutkiewicz, J. Bioaerosols and occupational lung disease. J. AerosolSci. 1994, 25, 1371–1404. [Google Scholar] [CrossRef]
- Davis, P.J. Molds, Toxic Molds, and Indoor Air Quality. California Research Bureau (CRB), California State Library: Sacramento, CA, USA. Available online: http://www.library.ca.gov/crb/01/notes/v8n1.pdf (accessed on 10 February 2012).
- Abarca, M. Taxonomy and identification of the species involved in nosocomial aspergillosis. Rev. Iberoam. Micol. 2000, 17, 79–84. [Google Scholar]
- Lugauskas, A.; Krikstaponis, A.; Seskauskas, V. Species of conditionally pathogenic micromycetes in the air of dwellings and occupational premises. Indoor Built Environ. 2003, 12, 167–177. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
