Nanometer Titanium Dioxide Mediated High Efficiency Photodegradation of Fluazifop-p-Butyl
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Equipment
2.2. Analytical Instruments
2.3. Experimental Procedure
2.3.1. Optimization of Nano-TiO2 Dosage
2.3.2. Photolytic Kinetics
2.3.3. Identification of Photolysis Products
3. Results and Discussion
3.1. Optimized Nano-TiO2 Dosage
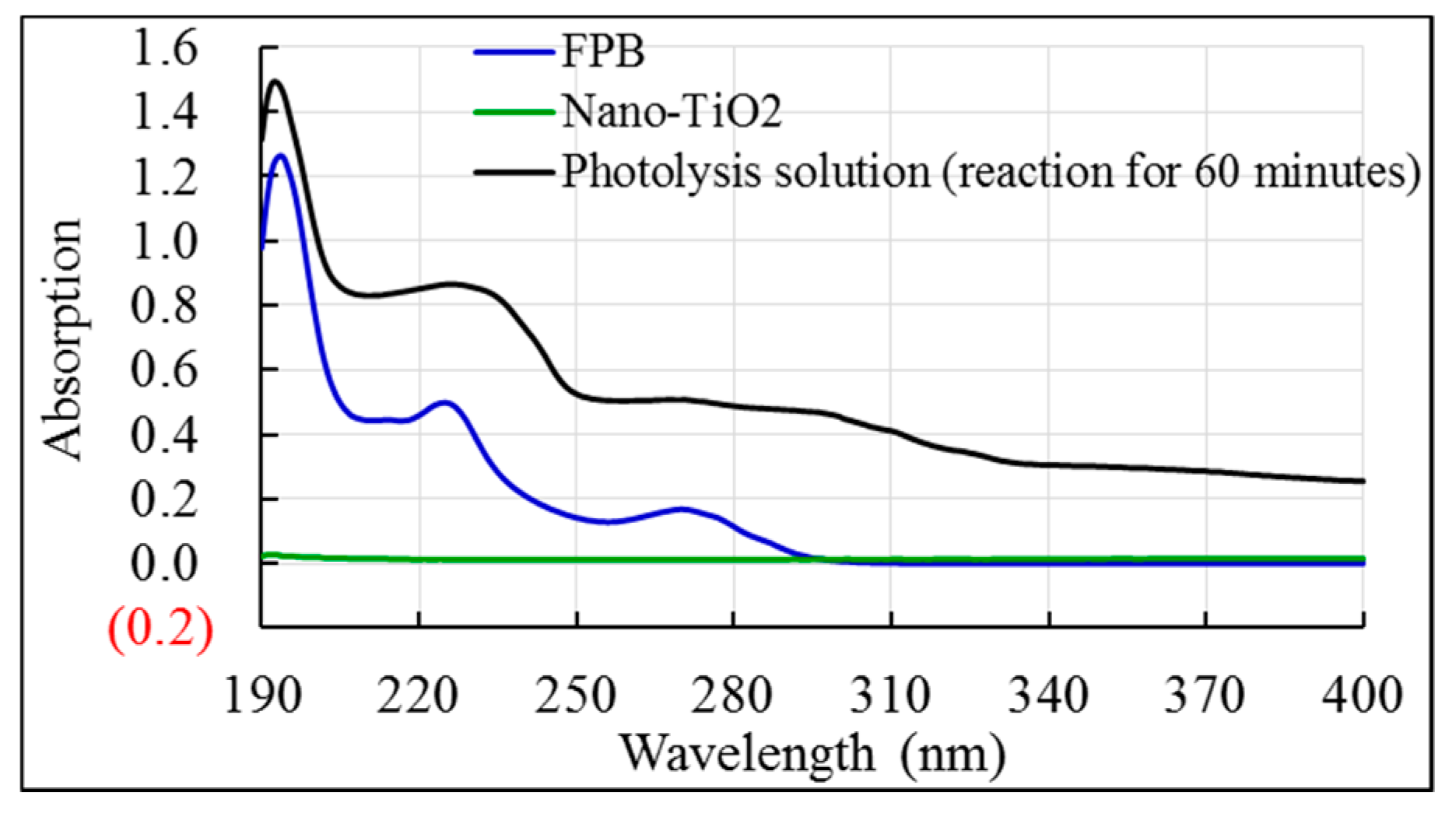
3.2. Photolysis Kinetics and Photosesitization Efficiency
3.3. Identification of Photolysis Products
3.4. Photodegradation Pathway
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Blake, R.J.; Westbury, D.B.; Woodcock, B.A.; Sutton, P.; Potts, S.G. Investigating the phytotoxicity of the graminicide fluazifop-P-butyl against native UK wildflower species. Pest Manag. Sci. 2012, 68, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Tu, M.; Hurd, C.; Randall, J.M. Weed Control Methods Handbook: Tools & Techniques for Use in Natural Areas. Available online: https://digitalcommons.usu.edu/govdocs/533/ (accessed on 20 May 2019).
- Cai, X.; Liu, W.; Sheng, G. Enantioselective degradation and ecotoxicity of the chiral herbicide diclofop in three freshwater alga cultures. J. Agric. Food Chem. 2008, 56, 2139–2146. [Google Scholar] [CrossRef] [PubMed]
- Extonet. Fluazifop-p-butyl pesticide information profile. Ext. Technol. Netw. 1996, 2, 216–219. [Google Scholar]
- Inyang, I.R.; Thomas, S. Toxicity of Fluazifop-p-butyl on blood cells and metabolites of a common African catfish (Clarias gariepinus). Niger. J. Agric. Food Environ. 2016, 12, 128–132. [Google Scholar]
- Elefsiniotis, I.S.; Liatsos, G.D.; Stamelakis, D.; Moulakakis, A. Case report: Mixed cholestatic/hepatocellular liver injury induced by the herbicide quizalofop-p-ethyl. Environ. Health Perspect. 2007, 115, 1479–1481. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Gu, L.; Chen, X.; Huang, Y.; Xu, J.; Cheng, Q.; Sun, J.; Wu, L.; Zhang, X. Study on subchronic toxicity of fluazifop-p-butyl to SD rats. J. Environ. Occup. Med. 2007, 24, 526–528. [Google Scholar]
- Bagheri, H.; Es’haghi, A.; Es-haghi, A. Immersed solvent microextraction of aryloxyphenoxypropionate herbicides from aquatic media. Int. J. Environ. Anal. Chem. 2013, 93, 450–460. [Google Scholar] [CrossRef]
- Curini, R.; Gentili, A.; Marchese, S.; Marino, A.; Perret, D. Solid-phase extraction followed by high-performance liquid chromatography–ionspray interface–mass spectrometry for monitoring of herbicides in environmental water. J. Chromatogr. A 2000, 874, 187–198. [Google Scholar] [CrossRef]
- Negre, M.; Gennari, M.; Andreoni, V.; Ambrosoli, R.; Celi, L. Microbial metabolism of fluazifop-butyl. J. Environ. Sci. Health B 1993, 28, 545–576. [Google Scholar] [CrossRef]
- Luo, M.; Liu, D.; Zhao, L.; Han, J.; Liang, Y.; Wang, P.; Zhou, Z. A novel magnetic ionic liquid modified carbon nanotube for the simultaneous determination of aryloxyphenoxypropionate herbicides and their metabolites in water. Anal. Chim. Acta 2014, 852, 88–96. [Google Scholar] [CrossRef]
- Liu, H.; Lou, X.; Ge, Z.; Yang, F.; Chen, D.; Zhu, J.; Xu, J.; Li, S.; Hong, Q. Isolation of an aryloxyphenoxy propanoate (AOPP) herbicide-degrading strain Rhodococcus ruber JPL-2 and the cloning of a novel carboxylesterase gene (feh). Braz. J. Microbiol. 2015, 46, 425–432. [Google Scholar] [CrossRef]
- Wang, C.; Qiu, J.; Yang, Y.; Zheng, J.; He, J.; Li, S. Identification and characterization of a novel carboxylesterase (FpbH) that hydrolyzes aryloxyphenoxypropionate herbicides. Biotechnol. Lett. 2017, 39, 553–560. [Google Scholar] [CrossRef]
- Chen, D.; Kong, X.; Wu, S.; Huang, J.; Chen, X.; Jiang, J. An esterase AppH for the hydrolysis of 2-(4-aryloxyphenoxy) propionate herbicides in Sphingobium sp. strain C3. Int. Biodeterior. Biodegrad. 2019, 136, 34–40. [Google Scholar] [CrossRef]
- Xu, X.; Wang, J.; Yu, T.; Nian, H.; Zhang, H.; Wang, G.; Li, F. Characterization of a novel aryloxyphenoxypropionate herbicide hydrolyzing carboxylesterase with R-enantiomer preference from Brevundimonas sp. QPT-2. Process Biochem. 2019, 82, 102–109. [Google Scholar] [CrossRef]
- Carp, O.; Huisman, C.L.; Reller, A. Photoinduced reactivity of titanium dioxide. Prog. Solid State Chem. 2004, 32, 33–177. [Google Scholar] [CrossRef]
- Asahi, R.; Taga, Y.; Mannstadt, W.; Freeman, A.J. Electronic and optical properties of anatase TiO2. Phys. Rev. B 2000, 61, 7459–7465. [Google Scholar] [CrossRef]
- Pelaez, M.; Nolan, N.T.; Pillai, S.C.; Seery, M.K.; Falaras, P.; Kontos, A.G.; Dunlop, P.S.M.; Hamilton, J.W.J.; Byrne, J.A.; O’Shea, K.; et al. A review on the visible light active titanium dioxide photocatalyst for environmental applications. Appl. Catal. B-Environ. 2012, 125, 331–349. [Google Scholar] [CrossRef]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Carey, J.H.; Lawrence, J.; Tosine, H.M. Photodechlorination of PCB’s in the presence of titanium dioxide in aqueous suspensions. Bull. Environ. Contam. Toxicol. 1976, 16, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Asahi, R. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ma, Q.; Wang, S.; Song, S.; Li, B.; Guo, R.; Cheng, X.; Cheng, Q. Photocatalytic performance and degradation mechanism of aspirin by TiO2 through response surface methodology. Catalysts 2018, 8, 118–132. [Google Scholar] [CrossRef]
- Minabe, T.; Tryk, D.A.; Sawunyama, P.; Kikuchi, Y.; Hashimoto, K.; Fujishima, A. TiO2-mediated photodegradation of liquid and solid organic compounds. J. Photochem. Photobiol. A 2000, 137, 53–62. [Google Scholar] [CrossRef]
- Ohko, Y.; Ando, I.; Niwa, C.; Tatsuma, T.; Yamamura, T.; Nakashima, T.; Kubota, Y.; Fujishima, A. Degradation of bisphenol A in water by TiO2 photocatalyst. Environ. Sci. Technol. 2001, 35, 2365–2368. [Google Scholar] [CrossRef] [PubMed]
- Han, F.; Kambala, V.R.; Srinivasan, M.; Rajarathnam, D.; Naidu, R. Tailored titanium dioxide photocatalysts for the degradation of organic dyes in wastewater treatment: A review. Appl. Catal. A-Gen. 2009, 359, 25–40. [Google Scholar] [CrossRef]
- Zhang, J.; Fu, D.; Xu, Y.; Liu, C. Optimization of parameters on photocatalytic degradation of chloramphenicol using TiO2 as photocatalyst by response surface methodology. J. Environ. Sci-China 2010, 22, 1281–1289. [Google Scholar] [CrossRef]
- Debjani, M.; Shahzad, B.; Ajay, R. Preparation and characterization of the TiO2 immobilized polymeric photocatalyst for degradation of aspirin under UV and solar light. Processes 2013, 2, 12–23. [Google Scholar] [CrossRef]
- Amalraj, A.; Pius, A. Photocatalytic degradation of monocrotophos and chlorpyrifos in aqueous solution using TiO2, under UV radiation. J. Water Process Eng. 2015, 7, 94–101. [Google Scholar] [CrossRef]
- Le Cunff, J.; Tomašić, V.; Wittine, O. Photocatalytic degradation of the herbicide terbuthylazine: Preparation, characterization and photoactivity of the immobilized thin layer of TiO2/chitosan. J. Photochem. Photobiol. A 2015, 309, 22–29. [Google Scholar] [CrossRef]
- Atchudan, R.; Edison, T.N.J.I.; Perumal, S.; Karthikeyan, D.; Lee, Y.R. In-situ green synthesis of nitrogen-doped carbon dots for bioimaging and TiO2, nanoparticles@nitrogen-doped carbon composite for photocatalytic degradation of organic pollutants. J. Alloy Compd. 2018, 766, 12–24. [Google Scholar] [CrossRef]
- Atchudan, R.; Nesakumar, J.I.E.T.; Perumal, S.; Vinodh, R.; Lee, Y.R. Effective photocatalytic degradation of anthropogenic dyes using graphene oxide grafting titanium dioxide nanoparticles under UV-light irradiation. Photochem. Photobiol. 2017, 333, 92–104. [Google Scholar] [CrossRef]
- Christou, C.; Gika, H.G.; Raikos, N.; Theodoridis, G. GC-MS analysis of organic acids in human urine in clinical settings: A study of derivatization and other analytical parameters. J. Chromatogr. B 2014, 964, 195–201. [Google Scholar] [CrossRef]
- Rousová, J.; Chintapalli, M.R.; Lindahl, A.; Casey, J.; Kubátová, A. Simultaneous determination of trace concentrations of aldehydes and carboxylic acids in particulate matter. J. Chromatogr. A 2018, 1544, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, H.; Zhu, M. A sensitive GC/MS detection method for analyzing microbial metabolites short chain fatty acids in fecal and serum samples. Talanta 2019, 196, 249–254. [Google Scholar] [CrossRef]
- Yang, H.; Yang, X.; Zhan, M.; Zhang, A. Photodegradation of bisphenol A in presence of suwannee river fulvic acid. J. Environ. Sci-China 2005, 26, 40–44. [Google Scholar] [CrossRef]
- Caban, P.; Stepnowski, P. Silylation of acetaminophen by trifluoroacetamide-based silylation agents. J. Pharm. Biomed. Anal. 2018, 154, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Ahmida, H.; Bertucci, P.; Franzò, L.; Massoud, R.; Cortese, C.; Lala, A.; Federici, G. Simultaneous determination of plasmatic phytosterols and cholesterol precursors using gas chromatography–mass spectrometry (GC–MS) with selective ion monitoring (SIM). J. Chromatogr. B 2006, 842, 43–47. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, Z.; Jia, N. Formation of multiple trimethylsilyl derivatives in the derivatization of 17 α-ethinylestradiol with BSTFA or MSTFA followed by gas chromatography-mass spectrometry determination. J. Environ. Sci. 2007, 19, 879–884. [Google Scholar] [CrossRef]
- Ghorbani-Choghamarani, A.; Cheraghi-Fathabad, N. Chemoselective and catalytic trimethylsilylation of alcohols and phenols by 1,1,1,3,3,3-hexamethyldisilazane and catalytic amounts of PhMe3N+Br3−. Chin. J. Catal. 2010, 31, 1103–1106. [Google Scholar] [CrossRef]





| Compound No. | Retention Time (Minute) | Major Fragment Ion(m/z) | Speculative Structure and Precise Molecular Weight |
|---|---|---|---|
| 1 | 7.25 | 57, 75, 130 |  (130) |
| 2 | 8.40 | 69, 78, 127, 147 |  (147) |
| 3 | 9.85 | 73, 96, 146, 163, 235 |  (163 = 235–73 + 1) |
| 4 | 10.87 | 73, 66, 78, 94, 166 |  (93 = 166–73 + 1) |
| 5 | 14.63 | 121, 146, 238, 254, 283 |  (283) |
| 6 | 16.69 | 73, 74, 117, 146 |  (74 = 146–73 + 1) |
| 7 | 28.02 | 73, 81, 110, 165, 254 |  (110 = 254–73−73 + 2) |
| 8 | 28.82 | 73, 146, 235, 238, 327 |  (255 = 327–73 + 1) |
| 9 | 31.83 | 73, 146, 237, 282, 327, 399 |  (327 = 399–73 + 1) |
| 10 | 38.79 | 146, 238, 254, 282, 383 |  (383) |
| 11 | 40.40 | 73, 145, 254, 282, 310, 383, 471 |  (399 = 471–73 + 1) |
| 12 | 44.42 | 73, 131, 146, 238, 385 |  (313 = 385–73 + 1) |
| 13 | 47.77 | 73, 129, 165, 181, 310 |  (238 = 310–73 + 1) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, G.; Hou, Z.; Zhang, R.; Chen, X.; Lu, Z. Nanometer Titanium Dioxide Mediated High Efficiency Photodegradation of Fluazifop-p-Butyl. Int. J. Environ. Res. Public Health 2019, 16, 3600. https://doi.org/10.3390/ijerph16193600
Li G, Hou Z, Zhang R, Chen X, Lu Z. Nanometer Titanium Dioxide Mediated High Efficiency Photodegradation of Fluazifop-p-Butyl. International Journal of Environmental Research and Public Health. 2019; 16(19):3600. https://doi.org/10.3390/ijerph16193600
Chicago/Turabian StyleLi, Guangling, Zhiguang Hou, Ruihong Zhang, Xiling Chen, and Zhongbin Lu. 2019. "Nanometer Titanium Dioxide Mediated High Efficiency Photodegradation of Fluazifop-p-Butyl" International Journal of Environmental Research and Public Health 16, no. 19: 3600. https://doi.org/10.3390/ijerph16193600




