Phenomenological and Thermodynamic Model of Gas Exchanges in the Placenta during Pregnancy: A Case Study of Intoxication of Carbon Monoxide
Abstract
1. Introduction
2. Methods
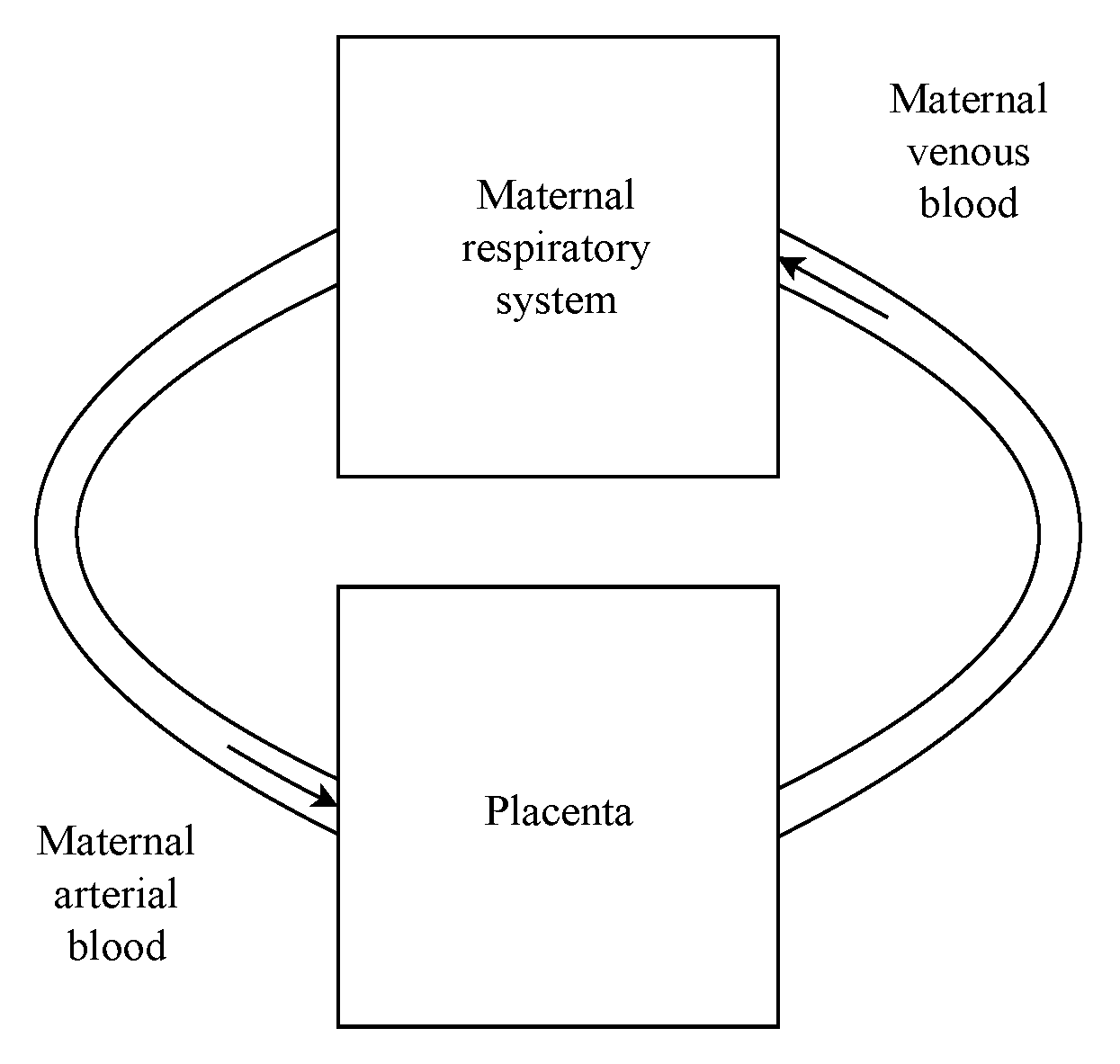
2.1. Maternal Respiratory System
2.2. Placenta Gases Exchange
2.3. Exergy Analysis
2.3.1. Respiratory Exergy and Entropy Generation Analyzes
- Model 1: O, CO and CO dissolved in blood as ideal gases.Destroyed exergy rate is obtained by is given by Equation (10), which is an adaptation of the general case (Equation (7)) [7].Because each stream has its exergy specified, only in this model, it is possible to evaluate an exergy efficiency [7], as in Equation (11).In these equations, refers to exergy flow rate associated with the blood. The Indices and refer to arterial and venous, respectively. is the exergy flow rate associated with air, denotes expired air and inspired air. is heat transfer from the lungs to the tissues surrounding it. The temperature is the reference temperature and is lung’s temperature. The term is the metabolic exergy reaction rate associated with lungs’ metabolism, evaluated in [6,34]. is the power done by the intercostal muscles into the respiratory system [35].
- Model 2: Considering the bound of O and CO with the hemoglobin.In this situation, only the destroyed exergy rate is calculated, which is justified in [7], where the methodology used to evaluate the bound to hemoglobin only allow to determine an exergy difference after and before the bounding () between the arterial and venous blood. These exergy variations were obtained from the Gibbs variation of the reactions, better described in [7].
- Model 3: Entropy generation caused by spontaneous diffusion.This method does not take into consideration the bounds of the gases with hemoglobin since it isolated the effects of the transport mechanism. Therefore, it is possible to calculate the irreversibilities (or entropy generation solely on the mass diffusion. The destroyed exergy rate for this system is given by Equation (13):where is:where for each gas is the mass flow rate of the gas in arterial blood flow and in the venous blood. is the pressure of the gas in the expired air, while is the pressure in the arterial blood.
2.3.2. Respiratory Exergy and Entropy Generation Analyzes
- Model 1: O, CO and CO dissolved in blood as ideal gases.In the following equations, m refers to maternal blood, while f to fetal. In addition, the subscript refers to venous blood and to arterial. is the exergy rate associated with the metabolism and is heat transferred from the placenta to it surroundings. is the reference temperature and is the placenta temperature. The efficiency, based on the discussions carried out in [3,30], is defined as:Destroyed exergy rate is given by Equation (19):
- Model 2: Considering the bound of O and CO with the hemoglobin.To apply this model to the placenta, it was necessary to evaluate the Gibbs free energy related to the gases binding to fetal hemoglobin. They were found using the equilibrium constants of fetal hemoglobin in an analogous manner to the one described in [7] for the adult blood. These values are summarized in Table 3.Hemoglobin bounding with CO and O is cooperative, meaning that, once the first ligation happens, the following ones occurs more easily. Following that, a hemoglobin molecule is commonly unbounded or bounded with four other molecules. The exergy destruction rate is described as follow:is given by:
- Model 3: Entropy generation caused by spontaneous diffusion.The entropy variation for each gas in the placenta is calculated by:
3. Results and Discussion
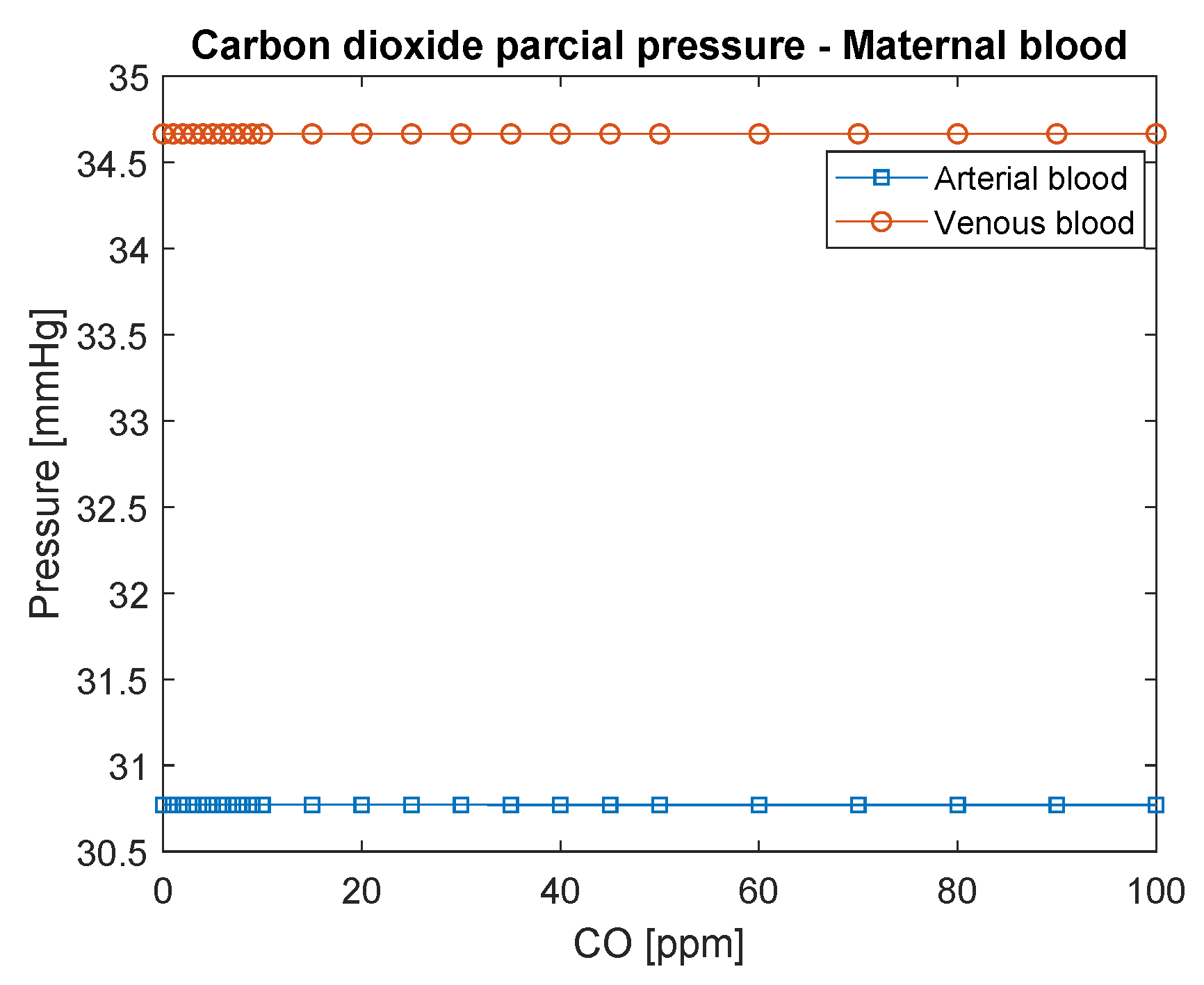
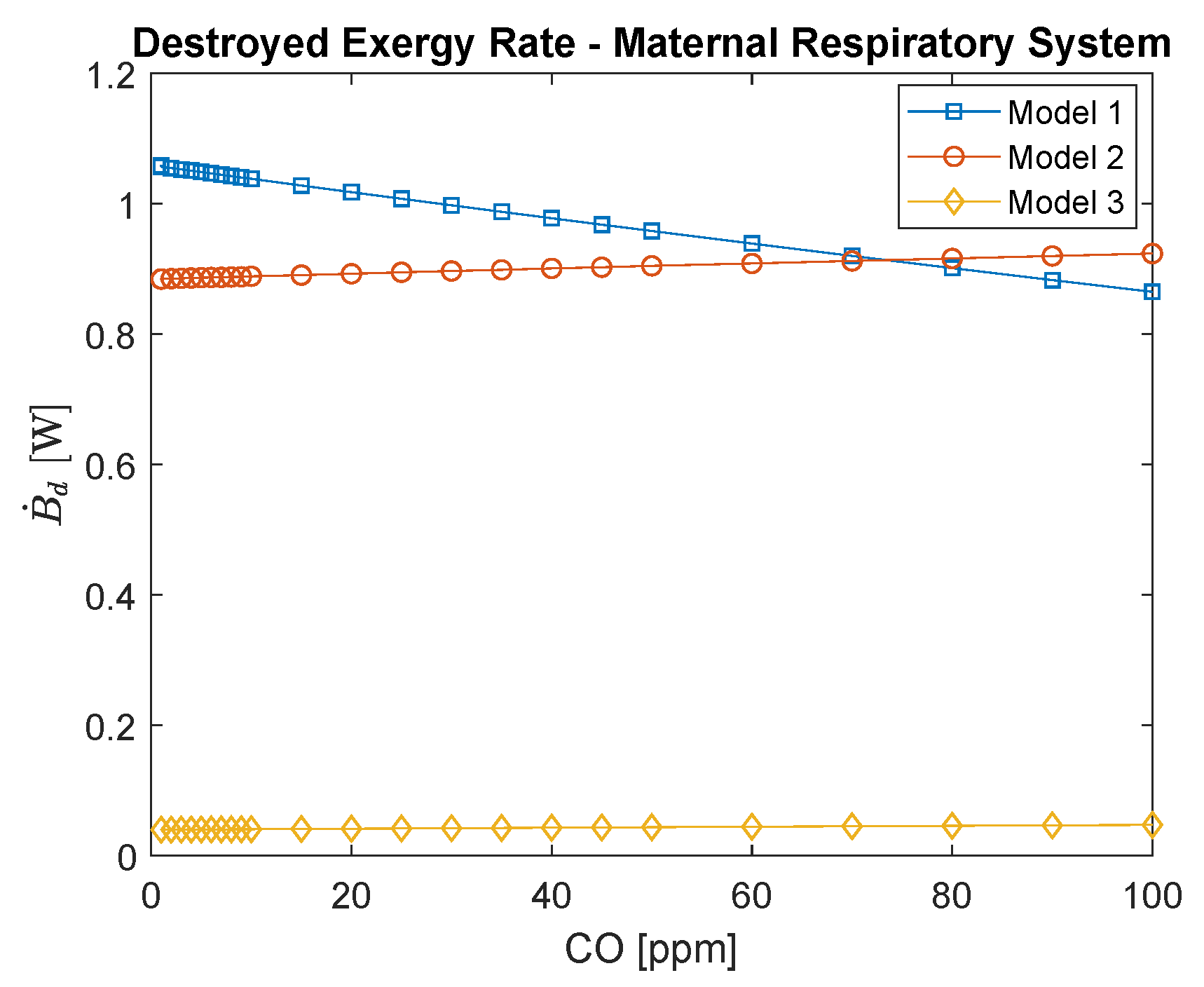
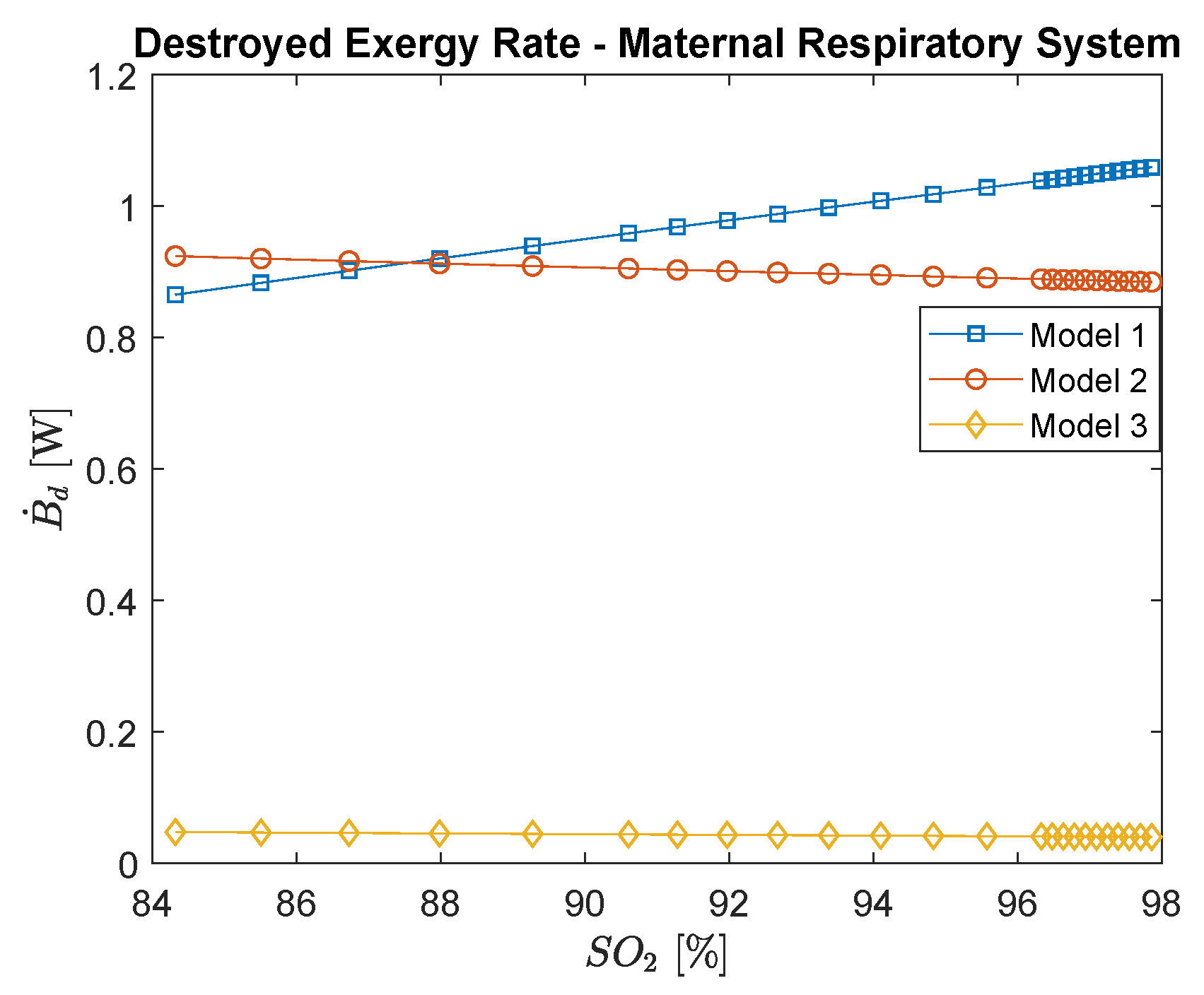
3.1. Maternal
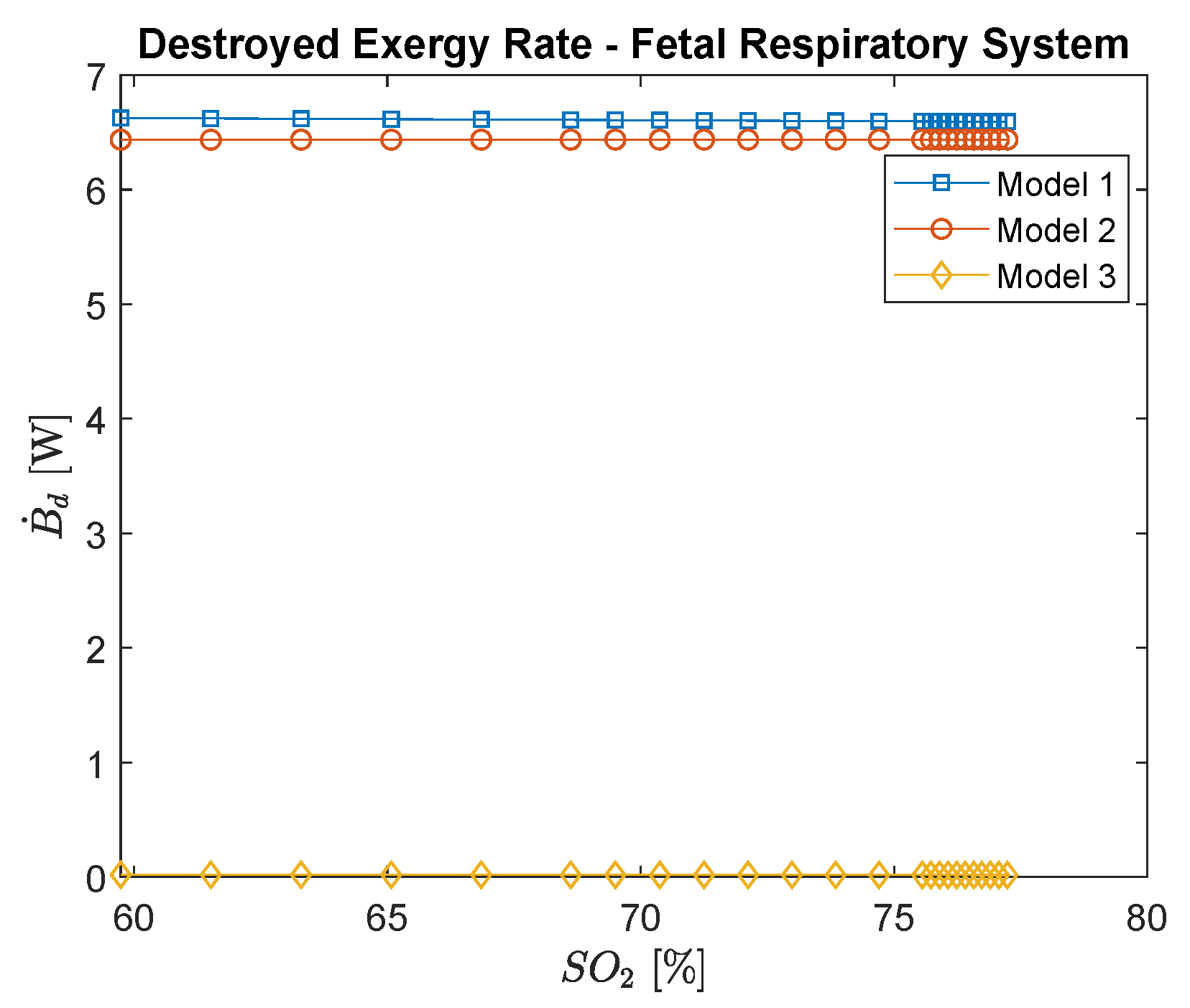
3.2. Fetal Exergy Behavior
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Symbols | |
| B | Exergy of the control volume (J) |
| Exergy rate (W) | |
| b | Specific exergy (kJ/kg) |
| C | Concentration (kg/m) |
| D | Diffusion coefficient (mL/(kg.min)) |
| G | Gibbs free energy (kJ) |
| g | specific gibbs energy (kJ/kg) |
| Enthalpy rate (W) | |
| h | Specific enthalpy (kJ/kg) |
| Mass flow rate (kg/s) | |
| Energy metabolism (W) | |
| P | Pressure (kPa, mmHg) |
| Heat transfer rate (W) | |
| R | Gas constant (kJ/kgK) |
| respiratory quotient (-) | |
| Entropy rate (W/K) | |
| s | Specific entropy (kJ/kgK) |
| T | Temperature (C) |
| t | Time (s) |
| Volumetric flow rate (m/s) | |
| V | Volume (m) |
| v | Specific volume (m/kg) |
| Power output (W) |
| Greek letters | |
| solubility coefficient of the gas in the blood (ml) | |
| efficiency (-) | |
| specific mass (kg/m) | |
| entropy generation rate (kW/K) |
| subscripts | |
| 0 | reference state |
| arterial blood | |
| environment air | |
| blood | |
| control volume | |
| d | destroyed |
| Diffusion | |
| e | exit |
| expired air | |
| f | fetus |
| reacted with hemoglobin | |
| i | inlet |
| lung | |
| m | mother |
| M | associated with the metabolic heat |
| venous blood |
References
- Schrödinger, E. What Is Life?: The Physical Aspect of the LIVING Cell; with, Mind and Matter; & Autobiographical Sketches; Cambridge University Press: Cambridge, UK, 1944. [Google Scholar]
- Prigogine, I.; Wiame, J. Biologie et thermodynamique des phénomènes irréversibles. Cell. Mol. Life Sci. 1946, 2, 451–453. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Junior, S. Exergy: Production, Cost and Renewability; Springer Science & Business Media: São Paulo, Brazil, 2012. [Google Scholar]
- Luo, L. Entropy production in a cell and reversal of entropy flow as an anticancer therapy. Front. Phys. China 2009, 4, 122–136. [Google Scholar] [CrossRef]
- Genc, S.; Sorguven, E.; Ozilgen, M.; Aksan Kurnaz, I. Unsteady exergy destruction of the neuron under dynamic stress conditions. Energy 2013, 59, 422–431. [Google Scholar] [CrossRef]
- Henriques, I.; Mady, C.; Neto, C.A.; Yanagihara, J.; Junior, S.O. The effect of altitude and intensity of physical activity on the exergy efficiency of respiratory system. Int. J. Thermodyn. 2014, 17, 265–273. [Google Scholar] [CrossRef]
- Cenzi, J.; Albuquerque, C.; Keutenedjian Mady, C. The Effect of Carbon Monoxide on the Exergy Behavior of the Lungs. Bioengineering 2018, 5, 108. [Google Scholar] [CrossRef]
- Henriques, I.; Mady, C.; de Oliveira-Junior, S. Exergy model of the human heart. Energy 2016, 117, 612–619. [Google Scholar] [CrossRef]
- Çatak, J.; Özilgen, M.; Olcay, A.B.; Yılmaz, B. Assessment of the work efficiency with exergy method in ageing muscles and healthy and enlarged hearts. Int. J. Exergy 2018, 25, 1–33. [Google Scholar] [CrossRef]
- Brandão Roll, J.; Leone Borges, M.; Keutenedjian Mady, C.E.; de Oliveira Junior, S. Exergy Analysis of the Heart with a Stenosis in the Arterial Valve. Entropy 2019, 21, 563. [Google Scholar] [CrossRef]
- Spanghero, G.M.; Albuquerque, C.; Lazzaretti Fernandes, T.; Hernandez, A.J.; Keutenedjian Mady, C.E. Exergy Analysis of the Musculoskeletal System Efficiency during Aerobic and Anaerobic Activities. Entropy 2018, 20, 119. [Google Scholar] [CrossRef]
- Mady, C.E.K.; Albuquerque, C.; Fernandes, T.L.; Hernandez, A.J.; Saldiva, P.H.N.; Yanagihara, J.I.; de Oliveira, S., Jr. Exergy performance of human body under physical activities. Energy 2013, 62, 370–378. [Google Scholar] [CrossRef]
- Mady, C.E.K.; Henriques, I.B.; de Oliveira, S., Jr. A thermodynamic assessment of therapeutic hypothermia techniques. Energy 2015, 85, 392–402. [Google Scholar] [CrossRef]
- Dovjak, M.; Shukuya, M.; Krainer, A. User-Centred Healing-Oriented Conditions in the Design of Hospital Environments. Int. J. Environ. Res. Public Health 2018, 15, 2140. [Google Scholar] [CrossRef] [PubMed]
- Prek, M. Exergy analysis of thermal comfort. Int. J. Exergy 2004, 1, 303–315. [Google Scholar] [CrossRef]
- Dovjak, M.; Shukuya, M.; Krainer, A. Connective thinking on building envelope–Human body exergy analysis. Int. J. Heat Mass Transf. 2015, 90, 1015–1025. [Google Scholar] [CrossRef]
- Mady, C.E.K.; Ferreira, M.S.; Yanagihara, J.I.; de Oliveira, S., Jr. Human body exergy analysis and the assessment of thermal comfort conditions. Int. J. Heat Mass Transf. 2014, 77, 577–584. [Google Scholar] [CrossRef]
- Guo, H.; Luo, Y.; Meggers, F.; Simonetti, M. Human body exergy consumption evaluation methods evaluation and their sensitivities towards different environmental conditions. Energy 2019, 183, 1075–1088. [Google Scholar] [CrossRef]
- Özilgen, M. Review on biothermoydnamics applications: Timeline, challenges, and opportunities. Int. J. Energy Res. 2017, 41, 1513–1533. [Google Scholar] [CrossRef]
- Albuquerque Neto, C.; Yanagihara, J.I.; Turri, F. A carbon monoxide transport model of the human respiratory system applied to urban atmosphere exposure analysis. J. Braz. Soc. Mech. Sci. Eng. 2008, 30, 253–260. [Google Scholar] [CrossRef]
- Mountcastle, V.B. Medical Physiology; CV Mosby: Maryland Heights, MO, USA, 1974. [Google Scholar]
- Aubard, Y.; Magne, I. Carbon monoxide poisoning in pregnancy. BJOG Int. J. Obstet. Gynaecol. 2000, 107, 833–838. [Google Scholar] [CrossRef]
- Garcia, A.; Azoubel, R. Placenta Humana: Morfologia e Pathologia Fetal e Perinatal; Atheneu: Rio de Janeiro, Brazil, 1986. [Google Scholar]
- Hall, J.E. Guyton and Hall Textbook of Medical Physiology e-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Knuttgen, H.G.; Emerson, K., Jr. Physiological response to pregnancy at rest and during exercise. J. Appl. Physiol. 1974, 36, 549–553. [Google Scholar] [CrossRef]
- Robson, S.C.; Hunter, S.; Boys, R.J.; Dunlop, W. Serial study of factors influencing changes in cardiac output during human pregnancy. Am. J. Physiol. Heart Circ. Physiol. 1989, 256, H1060–H1065. [Google Scholar] [CrossRef] [PubMed]
- Goplerud, J.; Delivoria-Papadopoulos, M. Physiology of the placenta–gas exchange. Ann. Clin. Lab. Sci. 1985, 15, 270–278. [Google Scholar] [PubMed]
- Acharya, G.; Sitras, V. Oxygen uptake of the human fetus at term. Acta Obstet. Gynecol. Scand. 2009, 88, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Sharan, M.; Selvakumar, S.; Singh, M. Mathematical model for the computation of alveolar partial pressure of carbon monoxide. Int. J. Bio-Med Comput. 1990, 26, 135–147. [Google Scholar] [CrossRef]
- Szargut, J.; Morris, D.R.; Steward, F.R. Exergy Analysis of Thermal, Chemical, and Metallurgical Processes; Hemisphere Publishing Corporation: New York, NY, USA, 1987. [Google Scholar]
- Gallo, W.; Milanez, L. Choice of a reference state for exergetic analysis. Energy 1990, 15, 113–121. [Google Scholar] [CrossRef]
- Perrella, M.; Sabbioneda, L.; Samaja, M.; Rossi-Bernardi, L. The intermediate compounds between human hemoglobin and carbon monoxide at equilibrium and during approach to equilibrium. J. Biol. Chem. 1986, 261, 8391–8396. [Google Scholar]
- Wagman, D.D.; Evans, W.H.; Parker, V.B.; Schumm, R.H.; Halow, I.; Bailey, S.M.; Churney, K.L.; Nuttall, R.L. The NBS tables of chemical thermodynamic properties: Selected values for inorganic and C1 and C2 organic substances in SI units. J. Phys. Chem. Ref. Data 1989, 18, 1807. [Google Scholar] [CrossRef]
- Mady, C. Human body exergy metabolism. Int. J. Thermodyn. 2013, 16, 73–80. [Google Scholar] [CrossRef]
- Fritts, H.; Filler, J.; Fishman, A.; Cournand, A. The efficiency of ventilation during voluntary hyperpnea: Studies in normal subjects and in dyspneic patients with either chronic pulmonary emphysema or obesity. J. Clin. Investig. 1959, 38, 1339–1348. [Google Scholar] [CrossRef]







| Parameter | Value | Reference |
|---|---|---|
| Cardiac Output | 7.2 L·min | [25] |
| Blood Volume | 6.53 L | [26] |
| Oxygen Consumption | 249 mL·min | [26] |
| Ventilation | 8.51 L·min | [26] |
| Hemoglobin Concentration | 12.1 g·dL | [26] |
| Respiratory Quotient | 0.83 | [26] |
| Parameter | Value | Reference |
|---|---|---|
| Umbilical blood flow | 66 mL·min·kg | [28] |
| Fetal O | 6.53 L | [28] |
| Hemoglobin | 14.5 g·dL | [28] |
| PO venous umbilical blood | 28.8 mmHg | [28] |
| −150.09 | −18.34 | |
| −301.04 | −37.41 | |
| −464.96 | −59.86 | |
| −634.09 | −95.89 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rangel Cenzi, J.; Albuquerque, C.; Keutenedjian Mady, C.E. Phenomenological and Thermodynamic Model of Gas Exchanges in the Placenta during Pregnancy: A Case Study of Intoxication of Carbon Monoxide. Int. J. Environ. Res. Public Health 2019, 16, 4138. https://doi.org/10.3390/ijerph16214138
Rangel Cenzi J, Albuquerque C, Keutenedjian Mady CE. Phenomenological and Thermodynamic Model of Gas Exchanges in the Placenta during Pregnancy: A Case Study of Intoxication of Carbon Monoxide. International Journal of Environmental Research and Public Health. 2019; 16(21):4138. https://doi.org/10.3390/ijerph16214138
Chicago/Turabian StyleRangel Cenzi, Juliana, Cyro Albuquerque, and Carlos Eduardo Keutenedjian Mady. 2019. "Phenomenological and Thermodynamic Model of Gas Exchanges in the Placenta during Pregnancy: A Case Study of Intoxication of Carbon Monoxide" International Journal of Environmental Research and Public Health 16, no. 21: 4138. https://doi.org/10.3390/ijerph16214138
APA StyleRangel Cenzi, J., Albuquerque, C., & Keutenedjian Mady, C. E. (2019). Phenomenological and Thermodynamic Model of Gas Exchanges in the Placenta during Pregnancy: A Case Study of Intoxication of Carbon Monoxide. International Journal of Environmental Research and Public Health, 16(21), 4138. https://doi.org/10.3390/ijerph16214138






