Detection of SARS-CoV-2 in Wastewater Northeast of Mexico City: Strategy for Monitoring and Prevalence of COVID-19
Abstract
:1. Introduction
2. Case Study
3. Materials and Methods
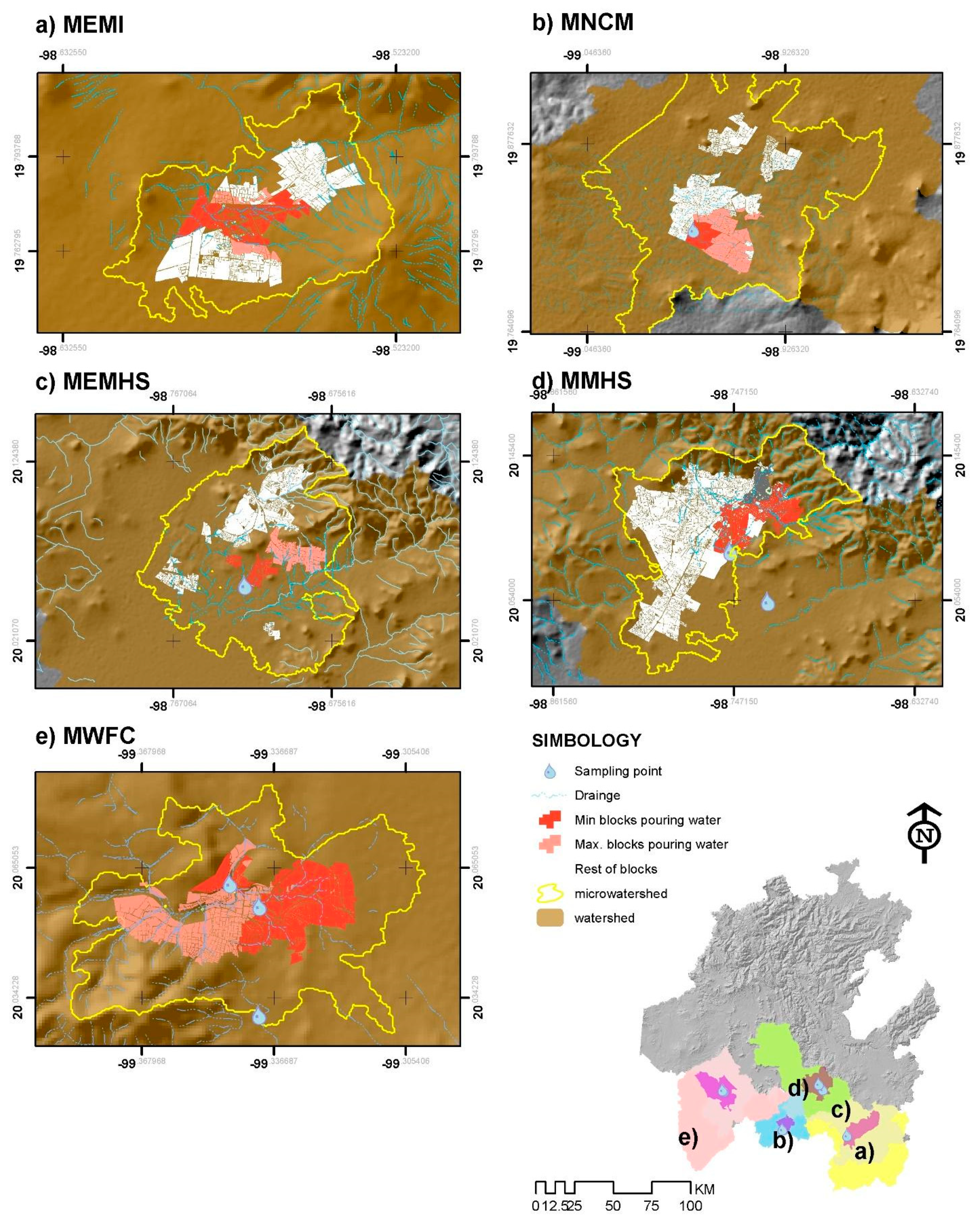
3.1. Micro-Basin Determination
3.2. Sampling Strategy
3.3. Viral RNA Residues Determination
3.4. Number of Infected People and Prevalence through Monte Carlo Model
4. Results
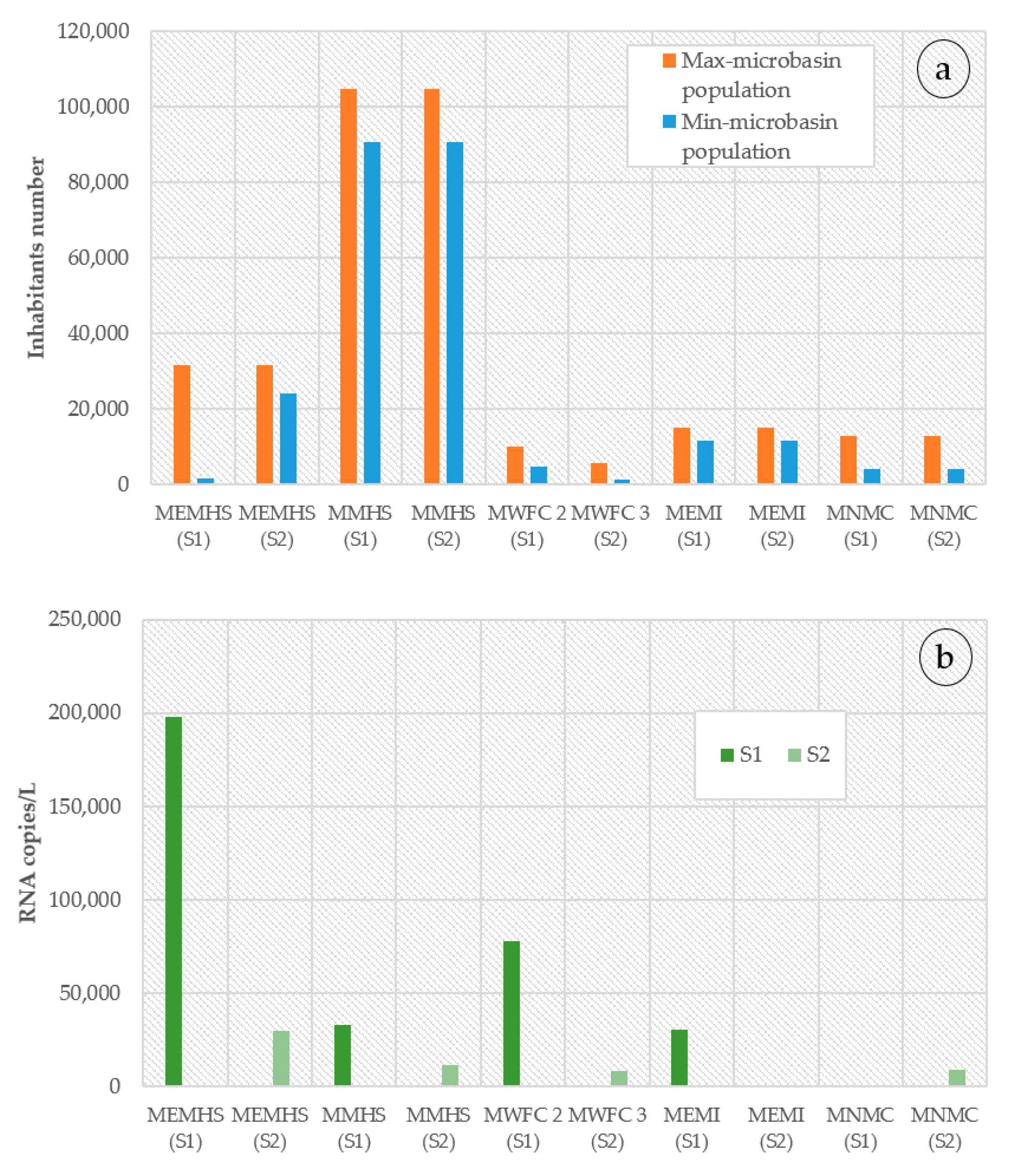
4.1. Micro-Basin Population and General Viral Waste Determination
4.2. Baseline Comparison in MWFC
4.3. Number of Infections Monte Carlo Model
5. Discussion
5.1. Micro-Basin Population and General Viral Waste Determination
5.2. Baseline Comparison in MWFC
5.3. Number of Infections Monte Carlo Model
5.4. Difference between Monte Carlo Model Results and Officially Reported Cases
5.5. Prevalence Monitoring
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Medema, G.; Heijnen, L.; Elsinga, G.; Italiaander, R.; Brouwer, A. Presence of SARS Coronavirus-2 R.N.A. in sewage and three correlations with reported COVID-19 prevalence in the early 4 stage of the epidemic in the Netherlands. Environ. Sci. Technol. Lett. 2020, 7, 511–516. [Google Scholar] [CrossRef]
- Arslan, M.; Xu, B.; El-Din, G. Transmission of SARS-CoV-2 via fecal-oral and aerosols–borne routes: Environmental dynamics and implications for wastewater management in underprivileged societies. Sci. Total Environ. 2020, 743, 140709. [Google Scholar] [CrossRef]
- Meo, S.A.; Alhowikan, A.M.; Al-Khlaiwi, T.; Meo, I.M.; Halepoto, D.M.; Iqbal, M.; Usmani, A.M.; Hajjar, W.; Ahmed, N. Novel coronavirus 2019-nCoV: Prevalence, biological and clinical characteristics comparison with SARS-CoV and MERS-CoV. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 2012–2019. [Google Scholar] [PubMed]
- Xu, H.; Zhong, L.; Deng, J.; Peng, J.; Dan, H.; Zeng, X.; Li, T.; Chen, Q. High expression of ACE2 receptor of the 2019-nCoV on the epithelial cells of oral mucosa. Int. J. Oral Sci. 2020, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- Harmer, D.; Gilbert, M.; Borman, R.; Clark, K.L. Quantitative mRNA expression profiling of ACE 2, a novel homologue of angiotensin converting enzyme. FEBS Lett. 2002, 532, 107–110. [Google Scholar] [CrossRef] [Green Version]
- Xiao, F.; Tang, M.; Zheng, X.; Li, C.; He, J. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 2020, 158, 1831–1833. [Google Scholar] [CrossRef] [PubMed]
- Hamming, I.; Timens, W.; Bulthuis, M.; Lely, A.; Navis, G.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, M.; Ahmed, W.; Bibby, K.; Carducci, A.; Gerba, C.P.; Hamillton, K.A.; Haramoto, E.; Rose, J.B. SARS.CoV-2 in wastewater: State of knowledge and research needs. Sci. Total Environ. 2020, 739, 1–19. [Google Scholar] [CrossRef]
- Wu, Y.; Guo, C.; Tang, L.; Hong, Z.; Zhou, J.; Dong, X.; Yin, H.; Xiao, Q.; Tang, Y.; Qu, X.; et al. Prolonged presence of SARS-CoV-2 viral RNA in fecal samples. Lancet Gastroenterol. Hepatol. 2020, 5, 434–435. [Google Scholar] [CrossRef]
- Shirazi, S.; Stanford, C.M.; Cooper, L.F. Characteristics and Detection Rate of SARS-CoV-2 in Alternative Sites and Specimens Pertaining to Dental Practice: An Evidence Summary. J. Clin. Med. 2021, 10, 1158. [Google Scholar] [CrossRef]
- Ling, Y.; Xu, S.-B.; Lin, Y.-X.; Tian, D.; Zhu, Z.-Q.; Dai, F.-H.; Wu, F.; Song, Z.-G.; Huang, W.; Chen, J.; et al. Persistence and clearance of viral RNA in 2019 novel coronavirus disease rehabilitation patients. Chin. Med. J. 2020, 133, 1039–1043. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Xu, Y.; Gao, R.; Lu, R.; Han, K.; Wu, G.; Tan, W. Detection of SARS-CoV-2 in Different Types of Clinical Specimens. JAMA 2020, 323, 1843–1844. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, W.; Angel, N.; Edson, J.; Bibby, K.; Bivins, A.; O’Brien, J.W.; Choi, P.M.; Kitajima, M.; Simpson, S.L.; Li, J.; et al. First confirmed detection of SARS-CoV-2 in untreated wastewater in Australia: A proof of concept for the wastewater surveillance of COVID-19 in the community. Sci. Total Environ. 2020, 728, 138764. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Ling, H.; Huang, X.; Li, J.; Li, W.; Yi, C.; Zhang, T.; Jiang, Y.; He, Y.; Deng, S.; et al. Potential spreading risks and disinfection challenges of medical wastewater by the presence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) viral RNA in septic tanks of Fangcang Hospital. Sci. Total Environ. 2020, 741, 140445. [Google Scholar] [CrossRef] [PubMed]
- Mercer, T.R.; Salit, M. Testing at scale during the COVID-19 pandemic. Nat. Rev. Genet. 2021, 22, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Oran, D.P.; Topol, E. Prevalence of Asymptomatic SARS-CoV-2 Infection. Ann. Intern. Med. 2020, 173, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Hillary, L.S.; Farkas, K.; Maher, K.H.; Lucaci, A.; Thorpe, J.; Distaso, M.A.; Gaze, W.H.; Paterson, S.; Burke, T.; Connor, T.R.; et al. Monitoring SARS-CoV-2 in municipal wastewater to evaluate the success of lockdown measures for controlling COVID-19 in the UK. Water Res. 2021, 200, 117214. [Google Scholar] [CrossRef] [PubMed]
- Gobierno del Estado de Hidalgo. Operativo Escudo. Available online: https://coronavirus.hidalgo.gob.mx/ (accessed on 19 November 2020).
- Jiménez, B.; Chávez, A. Low-cost technology for reliable use of Mexico City’s wastewater for agricultural irrigation. Technology 2002, 9, 84–107. [Google Scholar]
- Wu, F.; Zhang, J.; Xiao, A.; Gu, X.; Lee, W.L.; Armas, F.; Kauffman, K.; Hanage, W.; Matus, M.; Ghaeli, N.; et al. SARS-CoV-2 Titers in Wastewater Are Higher than Expected from Clinically Confirmed Cases. mSystems 2020, 5, e00614-20. [Google Scholar] [CrossRef]
- SEMARNATH. Secretaría de Medio Ambiente y Recursos Naturales de Hidalgo. Available online: https://smedioambiente (accessed on 24 November 2020).
- Rose, C.; Parker, A.; Jefferson, B.; Cartmell, E. The Characterization of Feces and Urine: A Review of the Literature to Inform Advanced Treatment Technology. Crit. Rev. Environ. Sci. Technol. 2015, 45, 1827–1879. [Google Scholar] [CrossRef] [Green Version]
- Instituto Nacional de Estadística y Geografía (INEGI). Encuesta Intercensal. 2015. Available online: https://www.inegi.org.mx/rnm/index.php/catalog/214/datacollection (accessed on 20 November 2020).
- Hart, O.E.; Halden, R.U. Computational analysis of SARS-CoV-2/COVID-19 surveillance by wastewater-based epidemiology locally and globally: Feasibility, economy, opportunities, and challenges. Sci. Total Environ. 2020, 730, 138875. [Google Scholar] [CrossRef]
- Fronza, R.; Lusic, M.; Schmidt, M.; Lucic, B. Spatial-temporal variations in atmospheric factors contribute to the SARS-CoV-2 outbreak. Viruses 2020, 12, 588. [Google Scholar] [CrossRef]
- Hasan, S.W.; Ibrahim, Y.; Daou, M.; Kannout, H.; Jan, N.; Lopes, A.; Alsafar, H.; Yousef, A.F. Detection and quantification of SARS-CoV-2 RNA in wastewater and treated effluents: Surveillance of COVID-19 epidemic in the United Arab Emirates. Sci. Total Environ. 2021, 764, 142929. [Google Scholar] [CrossRef] [PubMed]
- Randazzo, W.; Truchado, P.; Cuevas-Ferrando, E.; Simón, P.; Allende, A.; Sánchez, G. SARS-CoV-2 RNA in wastewater anticipated COVID-19 occurrence in a low prevalence area. Water Res. 2020, 181, 115942. [Google Scholar] [CrossRef] [PubMed]
- Sapula, S.A.; Whittall, J.J.; Pandopulos, A.J.; Gerber, C.; Venter, H. An optimized and robust PEG precipitation method for detection of SARS-CoV-2 in wastewater. Sci. Total Environ. 2021, 785, 147270. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, N.G.; Menon, G.; Mohapatra, G.; Pisharody, L.; Pattnaik, A.; Menon, N.G.; LalBhukya, P.; Srivastava, M.; Singh, M.; Barman, M.K.; et al. The Novel SARS-CoV-2 Pandemic: Possible Environmental Transmission, Detection, Persistence, and Fate during Wastewater and Water Treatmen. Sci. Total Environ. 2020, 765, 142746. [Google Scholar] [CrossRef] [PubMed]
- Our World in Data. Daily Covid-19 Tests per Thousand People. 2021. Available online: https://ourworldindata.org/grapher/full-list-daily-covid-19-tests-per-thousand?country=IND~KOR~USA~NZL~MEX~CAN~FRA~TWN (accessed on 19 November 2020).
- Li, X.; Zhang, S.; Shi, J.; Luby, S.P.; Jiang, G. Uncertainties in estimating SARS-CoV-2 prevalence by wastewater-based epidemiology. Chem. Eng. J. 2021, 415, 129039. [Google Scholar] [CrossRef]
- Siemens, J.; Huschek, G.; Siebe, C.; Kaupenjohann, M. Concentrations and mobility of human pharmaceuticals in the world’s largest wastewater irrigation system, Mexico City–Mezquital Valley. Water Res. 2008, 42, 2124–2134. [Google Scholar] [CrossRef]
- Carducci, A.; Morici, P.; Pizzi, F.; Battistini, R.; Rovini, E.; Verani, M. Study of the viral removal efficiency in a urban wastewater treatment plant. Water Sci. Technol. 2008, 58, 893–897. [Google Scholar] [CrossRef]
- Carducci, A.; Verani, M. Effects of Bacterial, Chemical, Physical and Meteorological Variables on Virus Removal by a Wastewater Treatment Plant. Food Environ. Virol. 2013, 5, 69–76. [Google Scholar] [CrossRef]
- Wigginton, K.; Ye, Y.; Ellenberg, R.M. Emerging investigators series: The source and fate of pandemic viruses in the urban water cycle. Environ. Sci. Water Res. Technol. 2015, 1, 735–746. [Google Scholar] [CrossRef]
- Farkas, K.; Cooper, D.M.; McDonald, J.E.; Malham, S.K.; de Rougemont, A.; Jones, D. Seasonal and spatial dynamics of enteric viruses in wastewater and in riverine and estuarine receiving waters. Sci. Total Environ. 2018, 634, 1174–1183. [Google Scholar] [CrossRef]
- Sbaoui, Y.; Bennis, F.; Chegdani, F. SARS-CoV-2 as Enteric Virus in Wastewater: Which Risk on the Environment and Human Behavior? Microbiol. Insights 2021, 14, 1178636121999673. [Google Scholar] [CrossRef] [PubMed]
- Amereh, F.; Negahban-Azar, M.; Isazadeh, S.; Dabiri, H.; Masihi, N.; Jahangiri-rad, M.; Rafiee, M. Sewage Systems Surveillance for SARS-CoV-2: Identification of Knowledge Gaps, Emerging Threats, and Future Research Needs. Pathogens 2021, 10, 946. [Google Scholar] [CrossRef]
- WHO. Water, Sanitation, Hygiene, and Waste Management for the COVID-19 Virus; WHO/2019-nCoV/IPC_WASH/2020.3; World Health Organization and the United Nations Children’s Fund (UNICEF): Geneva, Switzerland, 2020. [Google Scholar]


| Municipality | Confirmed Infected People Reported by the Government at Sampling Dates 1 | Sampling Date 2 | Municipality Place in Hidalgo State According to the Number of Confirmed Infected People Reported by the Government 1 | Municipality Population in 2020 |
|---|---|---|---|---|
| MEMHS | 319 | June 30th (S1) | 2nd place | 187,701 |
| 347 | July 7th (S2) | |||
| MMHS | 848 | June 30th (S1) | 1st place | 281,702 |
| 959 | July 7th (S2) | |||
| MWFC 2 | 124 | June 30th (S1) | 9th place | 117,083 |
| 142 | July 7th (S2) | |||
| MEMI | 158 | June 30th (S1) | 6th place | 55,061 |
| 177 | July 7th (S2) | |||
| MNMC | 283 | June 30th (S1) | 3rd place | 140,727 |
| 321 | July 7th (S2) |
| Sample Name | RNA Copies/L | Inhabitants Infected in the Micro-Basin 1 | Inhabitants Infected in the Municipality 2 | Prevalence of Infection (%) Median (90%CI) |
|---|---|---|---|---|
| MEMHS (S1) | 197,655 | 13,200 (3016–62,125) | 78,379 (17,909–368,888) | 0.417 (0.095–1.965) |
| MEMHS (S2) | 29,885 | 2044 (506–9843) | 12,137 (3005–66,365) | 0.064 (0.016–0.311) |
| MMHS (S1) | 32,909 | 7864 (2997–63,846) | 21,182 (8073–184,227) | 0.075 (0.028–0.61) |
| MMHS (S2) | 11,762 | 2824 (700–13,098) | 7607 (1885–37,794) | 0.027 (0.006–0.125) |
| MWFC 2 (S1) | 78,174 | 349 (209–6658) | 4076 (2441–106,647) | 0.034 (0.020–0.664) |
| MWFC 3 (S2) | 8159 | 977 (17–604) | 20,438 (356–20,392) | 0.174 (0.003–0.107) |
| MEMI (S1) | 30,230 | 251 (215–4553) | 917 (785–18,835) | 0.016 (0.014–0.302) |
| MEMI (S2) | Not detected | - | - | - |
| MNMC (S1) | Not detected | - | - | - |
| MNMC (S2) | 9234 | 189 (29–887) | 2055 (315–14,589) | 0.014 (0.002–0.068) |
| Sample Name | Hidalgo Government Reported Cases | Ratio 1 |
|---|---|---|
| MEMHS (S1) | 319 | 245.7 (56.1–1156.3) |
| MEMHS (S2) | 347 | 39.7(9.8–191.2) |
| MMHS (S1) | 848 | 26.7 (10.1–217.2) |
| MMHS(S2) | 959 | 8.4 (2.1–39.4) |
| MWFC 2 (S1) | 124 | 45(26.9–860) |
| MWFC 3 (S2) | 124 | 266 (4.6–164.4) |
| MEMI (S1) | 158 | 6.5(5.6–119.2) |
| MEMI (S2) | 177 | - |
| MNMC (S1) | 283 | - |
| MNMC (S2) | 321 | 9.6 (1.4–45.4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Reyes, J.R.; Hernández-Flores, M.d.l.L.; Paredes-Zarco, J.E.; Téllez-Jurado, A.; Fayad-Meneses, O.; Carranza-Ramírez, L. Detection of SARS-CoV-2 in Wastewater Northeast of Mexico City: Strategy for Monitoring and Prevalence of COVID-19. Int. J. Environ. Res. Public Health 2021, 18, 8547. https://doi.org/10.3390/ijerph18168547
González-Reyes JR, Hernández-Flores MdlL, Paredes-Zarco JE, Téllez-Jurado A, Fayad-Meneses O, Carranza-Ramírez L. Detection of SARS-CoV-2 in Wastewater Northeast of Mexico City: Strategy for Monitoring and Prevalence of COVID-19. International Journal of Environmental Research and Public Health. 2021; 18(16):8547. https://doi.org/10.3390/ijerph18168547
Chicago/Turabian StyleGonzález-Reyes, José Roberto, María de la Luz Hernández-Flores, Jesús Eduardo Paredes-Zarco, Alejandro Téllez-Jurado, Omar Fayad-Meneses, and Lamán Carranza-Ramírez. 2021. "Detection of SARS-CoV-2 in Wastewater Northeast of Mexico City: Strategy for Monitoring and Prevalence of COVID-19" International Journal of Environmental Research and Public Health 18, no. 16: 8547. https://doi.org/10.3390/ijerph18168547
APA StyleGonzález-Reyes, J. R., Hernández-Flores, M. d. l. L., Paredes-Zarco, J. E., Téllez-Jurado, A., Fayad-Meneses, O., & Carranza-Ramírez, L. (2021). Detection of SARS-CoV-2 in Wastewater Northeast of Mexico City: Strategy for Monitoring and Prevalence of COVID-19. International Journal of Environmental Research and Public Health, 18(16), 8547. https://doi.org/10.3390/ijerph18168547






