The Efficacy of Brief School-Based Exercise Programs in Improving Pubertal Bone Mass and Physical Fitness: A Randomized Controlled Trial
Abstract
:1. Introduction
2. Method
2.1. Study Oversight
2.2. Participants
2.3. Design
2.4. Anthropometry
2.5. Extracurricular Physical Activity
2.6. Total Energy and Calcium Intake
2.7. BMD Measure
2.8. Physical Fitness Measure
2.9. Statistics
3. Result
3.1. Baseline Characteristics of Participants
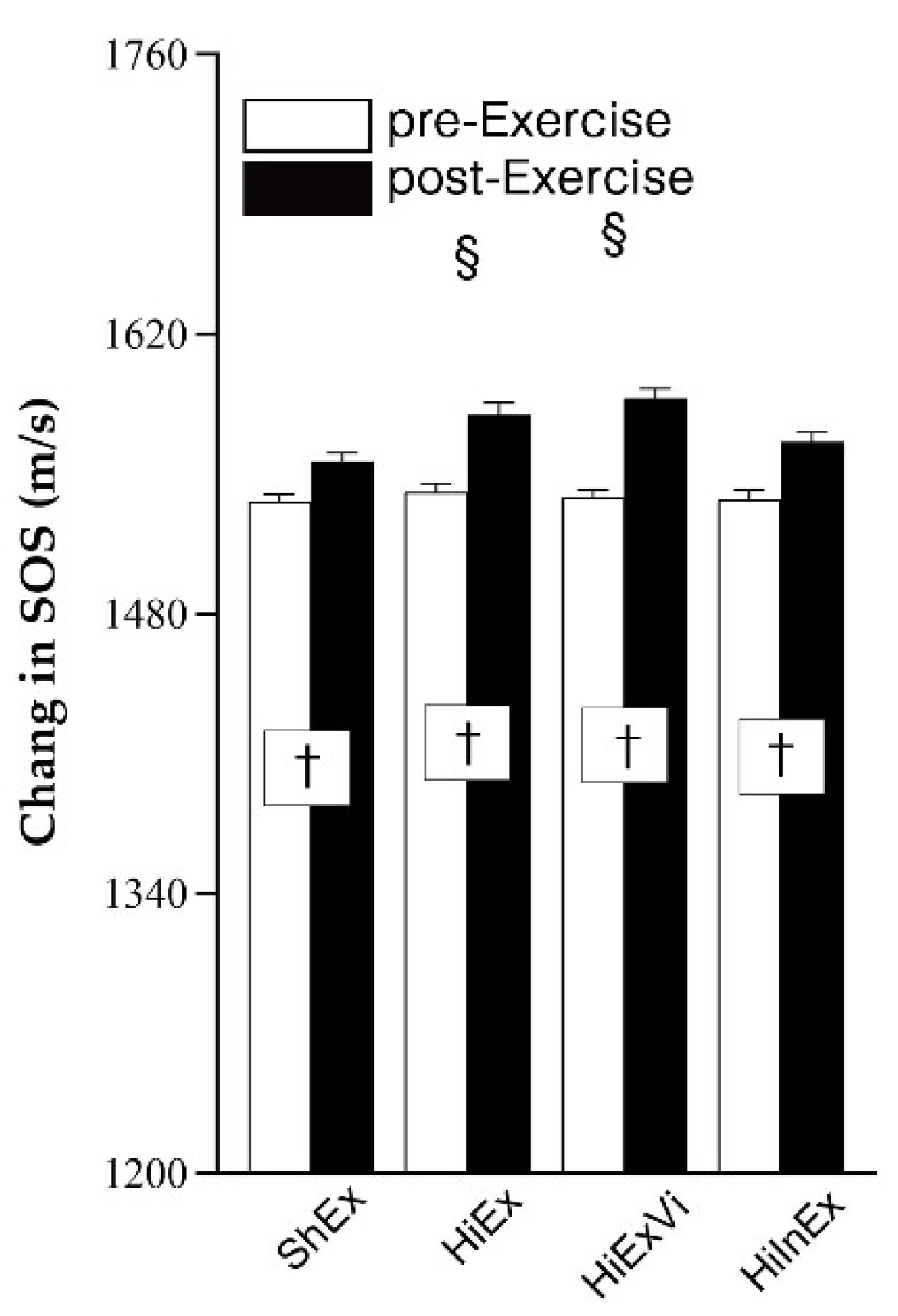
3.2. Changes in SOS Evaluation
3.3. Physical Fitness Evaluation
4. Discussion
Limitation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dorn, L.D.; Hostinar, C.E.; Susman, E.J.; Pervanidou, P. Conceptualizing puberty as a window of opportunity for impacting health and well-being across the life span. J. Res. Adolesc. 2019, 29, 155–176. [Google Scholar] [CrossRef] [Green Version]
- Styne, D.; Grumbach, M. Physiology and disorders of puberty. In Williams Textbook of Endocrinology, 12th ed.; Melmed, S., Polonsky, K., Larsen, P., Kronenberg, H., Eds.; Elsevier: Philadelphia, PA, USA, 2015; pp. 1074–1298. [Google Scholar]
- Kohl, H.W.; Craig, C.L.; Lambert, E.V.; Inoue, S.; Alkandari, J.R.; Leetongin, G.; Kahlmeier, S. Lancet Phys Activity Series W: The pandemic of physical inactivity: Global action for public health. Lancet 2012, 380, 294–305. [Google Scholar] [CrossRef] [Green Version]
- Lee, I.M.; Shiroma, E.J.; Lobelo, F.; Puska, P.; Blair, S.N.; Katzmarzyk, P.T. Effect of physical inactivity on major non-communicable diseases worldwide: An analysis of burden of disease and life expectancy. Lancet 2012, 380, 219–229. [Google Scholar] [CrossRef] [Green Version]
- Blair, S.N.; Brodney, S. Effects of physical inactivity and obesity on morbidity and mortality: Current evidence and research issues. Med. Sci. Sports Exerc. 1999, 31, S646–S662. [Google Scholar] [CrossRef]
- Li, J.; Siegrist, J. Physical activity and risk of cardiovascular disease—A meta-analysis of prospective cohort studies. Int. J. Environ. Res. Public Health 2012, 9, 391–407. [Google Scholar] [CrossRef]
- Hamasaki, H. Daily physical activity and type 2 diabetes: A review. World J. Diabetes 2016, 7, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Sanchis-Gomar, F.; Lucia, A.; Yvert, T.; Ruiz-Casado, A.; Pareja-Galeano, H.; Santos-Lozano, A.; Fiuza-Luces, C.; Garatachea, N.; Lippi, G.; Bouchard, C.; et al. Physical inactivity and low fitness deserve more attention to alter cancer risk and prognosis. Cancer Prev. Res. 2015, 8, 105–110. [Google Scholar] [CrossRef] [Green Version]
- Berenson, G.S.; Srinivasan, S.R.; Bao, W.; Newman, W.P.; Tracy, R.E.; Wattigney, W.A. Association between multiple cardiovascular risk factors and atherosclerosis in children and young adults. N. Engl. J. Med. 1998, 338, 1650–1656. [Google Scholar] [CrossRef] [PubMed]
- Katzmarzyk, P.T.; Pérusse, L.; Malina, R.M.; Bergeron, J.; Després, J.-P.; Bouchard, C. Stability of indicators of the metabolic syndrome from childhood and adolescence to young adulthood. J. Clin. Epidemiol. 2001, 54, 190–195. [Google Scholar] [CrossRef]
- Marques, A.; Henriques-Neto, D.; Peralta, M.; Martins, J.; Demetriou, Y.; Schonbach, D.M.I.; de Matos, M.G. Prevalence of physical activity among adolescents from 105 low, middle, and high-income countries. Int. J. Environ. Res. Public Health 2020, 17, 11. [Google Scholar] [CrossRef] [PubMed]
- Trost, S.G.; McCoy, T.A.; Vander Veur, S.S.; Mallya, G.; Duffy, M.L.; Foster, G.D. Physical activity patterns of inner-city elementary schoolchildren. Med. Sci. Sports Exerc. 2013, 45, 470–474. [Google Scholar] [CrossRef]
- Tan, L.; Yu, F.; Zhang, Z.; Chen, Q.; Hu, X.; Zhang, J. An obesity epidemic among primary and middle school students in Minhang district, Shanghai: A survey of Body Mass Index and distribution characteristics. Chin. Gen. Pract. 2020, 23, 1936–1941. [Google Scholar] [CrossRef]
- Liang, X. The Digital Gap behind the Physical Fitness of Chinese and Japanese Adolescents; China Youth Daily: Beijing, China, 2017. [Google Scholar]
- Zhou, Y. Reflection on the Incident in Nanjing No. 1 Middle School: Quality Education and Exam-Oriented Education, Who Can Achieve the Future of Children? Available online: https://new.qq.com/rain/a/20200817A0EX8L00 (accessed on 17 August 2021).
- Heradstveit, O.; Haugland, S.; Hysing, M.; Stormark, K.M.; Sivertsen, B.; Boe, T. Physical inactivity, non-participation in sports and socioeconomic status: A large population-based study among Norwegian adolescents. BMC Public Health 2020, 20, 1010. [Google Scholar] [CrossRef]
- Moradi, G.; Mostafavi, F.; Piroozi, B.; Zareie, B.; Mahboobi, M.; Rasouli, M.A. The prevalence of physical inactivity in Iranian adolescents and the impact of economic and social inequalities on it: Results of a National Study in 2018. BMC Public Health 2020, 20, 1499. [Google Scholar] [CrossRef] [PubMed]
- El-ammari, A.; El Kazdouh, H.; Bouftini, S.; El Fakir, S.; El Achhab, Y. Level and potential social-ecological factors associated with physical inactivity and sedentary behavior among Moroccan school-age adolescents: A cross-sectional study. Environ. Health Prev. 2017, 22, 9. [Google Scholar] [CrossRef] [Green Version]
- Friel, C.P.; Duran, A.T.; Shechter, A.; Diaz, K.M. US children meeting physical activity, screen time, and sleep guidelines. Am. J. Prev. Med. 2020, 59, 513–521. [Google Scholar] [CrossRef]
- Ghekiere, A.; Van Cauwenberg, J.; Vandendriessche, A.; Inchley, J.; de Matos, M.G.; Borraccino, A.; Gobina, I.; Tynjala, J.; Deforche, B.; De Clercq, B. Trends in sleeping difficulties among European adolescents: Are these associated with physical inactivity and excessive screen time? Int. J. Public Health 2019, 64, 487–498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin-Smith, R.; Cox, A.; Buchan, D.S.; Baker, J.S.; Grace, F.; Sculthorpe, N. High Intensity Interval Training (HIIT) improves Cardiorespiratory Fitness (CRF) in healthy, overweight and obese adolescents: A systematic review and meta-analysis of controlled studies. Int J Env. Res. Public Health 2020, 17, 2955. [Google Scholar] [CrossRef] [PubMed]
- Costigan, S.A.; Eather, N.; Plotnikoff, R.C.; Taaffe, D.R.; Lubans, D.R. High-intensity interval training for improving health-related fitness in adolescents: A systematic review and meta-analysis. Br. J. Sports Med. 2015, 49, 9. [Google Scholar] [CrossRef]
- Barry, D.W.; Kohrt, W.M. BMD decreases over the course of a year in competitive male cyclists. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2008, 23, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Robling, A.G.; Castillo, A.B.; Turner, C.H. Biomechanical and molecular regulation of bone remodeling. Annu. Rev. Biomed. Eng. 2006, 8, 455–498. [Google Scholar] [CrossRef]
- Bergmann, P.; Body, J.J.; Boonen, S.; Boutsen, Y.; Devogelaer, J.P.; Goemaere, S.; Kaufman, J.; Reginster, J.Y.; Rozenberg, S. Loading and skeletal development and maintenance. J. Osteoporos. 2010, 2011, 786752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weeks, B.K.; Young, C.M.; Beck, B.R. Eight months of regular in-school jumping improves indices of bone strength in adolescent boys and girls: The POWER PE Study. J. Bone Miner. Res. 2008, 23, 1002–1011. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nogueira, R.C.; Weeks, B.K.; Beck, B.R. An in-school exercise intervention to enhance bone and reduce fat in girls: The CAPO Kids trial. Bone 2014, 68, 92–99. [Google Scholar] [CrossRef] [Green Version]
- MacKelvie, K.J.; Petit, M.A.; Khan, K.M.; Beck, T.J.; McKay, H.A. Bone mass and structure are enhanced following a 2-year randomized controlled trial of exercise in prepubertal boys. Bone 2004, 34, 755–764. [Google Scholar] [CrossRef]
- Granacher, U.; Prieske, O.; Majewski, M.; Büsch, D.; Muehlbauer, T. The role of instability with plyometric training in sub-elite adolescent soccer players. Int. J. Sports Med. 2015, 36, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Jlid, M.C.; Racil, G.; Coquart, J.; Paillard, T.; Bisciotti, G.N.; Chamari, K. Multidirectional plyometric training: Very efficient way to improve vertical jump performance, change of direction performance and dynamic postural control in young soccer players. Front. Physiol. 2019, 10, 9. [Google Scholar] [CrossRef]
- Lange, A.K.; Vanwanseele, B.; Foroughi, N.; Baker, M.K.; Shnier, R.; Smith, R.M.; Singh, M.A.F. Resistive Exercise for Arthritic Cartilage Health (REACH): A randomized double-blind, sham-exercise controlled trial. BMC Geriatr. 2009, 9. [Google Scholar] [CrossRef] [Green Version]
- Ainsworth, B.E.; Haskell, W.L.; Whitt, M.C.; Irwin, M.L.; Swartz, A.M.; Strath, S.J.; O’Brien, W.L.; Bassett, D.R., Jr.; Schmitz, K.H.; Emplaincourt, P.O.; et al. Compendium of physical activities: An update of activity codes and MET intensities. Med. Sci. Sports Exerc. 2000, 32, S498–S504. [Google Scholar] [CrossRef] [Green Version]
- Egger, T.; Flueck, J.L. Energy availability in male and female elite wheelchair athletes over seven consecutive training days. Nutrients 2020, 12, 3262. [Google Scholar] [CrossRef]
- Delshad, M.; Beck, K.L.; Conlon, C.A.; Mugridge, O.; Kruger, M.C.; Hurst, P.R.V. Validity of quantitative ultrasound and bioelectrical impedance analysis against dual x-ray absorptiometry for measuring bone quality and body composition in children. Proceedings 2019, 8, 16. [Google Scholar] [CrossRef] [Green Version]
- Kishimoto, H. CM-100. Nihon Rinsho. Jpn. J. Clin. Med. 2004, 62 (Suppl. S2), 305–308. [Google Scholar]
- Ministry of Education of the People’s Republic of China. Compulsory Schooling Physical Education and Health Curriculum Standard; Ministry of Education of the People’s Republic of China: Beijing, China, 2012.
- McLester, C.N.; Nickerson, B.S.; Kliszczewicz, B.M.; McLester, J.R. Reliability and agreement of various InBody body composition analyzers as compared to dual-energy X-ray absorptiometry in healthy men and women. J. Clin. Densitom. 2020, 23, 443–450. [Google Scholar] [CrossRef]
- Liu, X.; Song, X.; Meng, Z. Effects of airborne pollution on cardiopulmonary function of healthy person. Wei Sheng Yan Jiu J. Hyg. Res. 2008, 37, 429–432. [Google Scholar]
- Vellas, B.J.; Rubenstein, L.Z.; Ousset, P.J.; Faisant, C.; Kostek, V.; Nourhashemi, F.; Allard, M.; Albarede, J.L. One-leg standing balance and functional status in a population of 512 community-living elderly persons. Aging 1997, 9, 95–98. [Google Scholar] [CrossRef]
- Aoyama, Y.; Uchida, H.; Sugi, Y.; Kawakami, A.; Fujii, M.; Kiso, K.; Kono, R.; Takebayashi, T.; Hirao, K.M. Immediate effect of subliminal priming with positive reward stimuli on standing balance in healthy individuals: A randomized controlled trial. J. Med. 2017, 96, e7494. [Google Scholar] [CrossRef]
- Ireland, A.; Ferretti, J.L.; Rittweger, J. Imaging of the muscle-bone relationship. Curr. Osteoporos Rep. 2014, 12, 486–495. [Google Scholar] [CrossRef] [PubMed]
- Hart, N.H.; Nimphius, S.; Rantalainen, T.; Ireland, A.; Siafarikas, A.; Newton, R.U. Mechanical basis of bone strength: Influence of bone material, bone structure and muscle action. J. Musculoskelet. Neuronal Interact. 2017, 17, 114–139. [Google Scholar] [PubMed]
- Honda, A.; Matsumoto, M.; Kato, T.; Umemura, Y. Exercise characteristics influence femoral cross-sectional geometry: A magnetic resonance imaging study in elite female athletes. Osteoporos. Int. 2015, 26, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Petit, M.A.; McKay, H.A.; MacKelvie, K.J.; Heinonen, A.; Khan, K.M.; Beck, T.J. A randomized school-based jumping intervention confers site and maturity-specific benefits on bone structural properties in girls: A hip structural analysis study. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2002, 17, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, D.; Barker, A.R.; Ubago-Guisado, E.; Williams, C.A.; Gracia-Marco, L. A 9-month Jumping intervention to improve bone geometry in adolescent male athletes. Med. Sci. Sports. Exerc. 2018, 50, 2544–2554. [Google Scholar] [CrossRef]
- Vlachopoulos, D.; Barker, A.R.; Ubago-Guisado, E.; Williams, C.A.; Gracia-Marco, L. The effect of a high-impact jumping intervention on bone mass, bone stiffness and fitness parameters in adolescent athletes. Arch. Osteoporos. 2018, 13, 128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Min, S.K.; Oh, T.; Kim, S.H.; Cho, J.; Chung, H.Y.; Park, D.H.; Kim, C.S. Position statement: Exercise guidelines to increase peak bone mass in adolescents. J. Bone Metab. 2019, 26, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Umemura, Y.; Nagasawa, S.; Honda, A.; Singh, R. High-impact exercise frequency per week or day for osteogenic response in rats. J. Bone Min. Metab. 2008, 26, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Burr, D.B.; Robling, A.G.; Turner, C.H. Effects of biomechanical stress on bones in animals. Bone 2002, 30, 781–786. [Google Scholar] [CrossRef]
- Umemura, Y.; Ishiko, T.; Yamauchi, T.; Kurono, M.; Mashiko, S. Five jumps per day increase bone mass and breaking force in rats. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1997, 12, 1480–1485. [Google Scholar] [CrossRef]
- Robling, A.G.; Burr, D.B.; Turner, C.H. Recovery periods restore mechanosensitivity to dynamically loaded bone. J. Exp. Biol. 2001, 204, 3389–3399. [Google Scholar] [CrossRef]
- Minett, M.M.; Binkley, T.B.; Weidauer, L.A.; Specker, B.L. Changes in body composition and bone of female collegiate soccer players through the competitive season and off-season. J. Musculoskelet. Neuronal Interact. 2017, 17, 386–398. [Google Scholar]
- Allison, S.J.; Poole, K.E.S.; Treece, G.M.; Gee, A.H.; Tonkin, C.; Rennie, W.J.; Folland, J.P.; Summers, G.D.; Brooke-Wavell, K. The influence of high-impact exercise on cortical and trabecular bone mineral content and 3D distribution across the proximal femur in older men: A randomized controlled unilateral intervention. J. Bone Miner. Res. 2015, 30, 1709–1716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hind, K.; Gannon, L.; Whatley, E.; Cooke, C.; Truscott, J. Bone cross-sectional geometry in male runners, gymnasts, swimmers and non-athletic controls: A hip-structural analysis study. Eur. J. Appl. Physiol. 2012, 112, 535–541. [Google Scholar] [CrossRef] [Green Version]
- Nakazono, E.; Miyazaki, H.; Abe, S.; Imai, K.; Masuda, T.; Iwamoto, M.; Moriguchi, R.; Ueno, H.; Ono, M.; Yazumi, K.; et al. Discontinuation of leisure time impact-loading exercise is related to reduction of a calcaneus quantitative ultrasound parameter in young adult Japanese females: A 3-year follow-up study. Osteoporos Int. 2014, 25, 485–495. [Google Scholar] [CrossRef]
- Allison, S.J.; Brooke-Wavell, K.; Folland, J. High and odd impact exercise training improved physical function and fall risk factors in community-dwelling older men. J. Musculoskelet. Neuronal Interact. 2018, 18, 100–107. [Google Scholar]
- Petersen, B.A.; Hastings, B.; Gottschall, J.S. Low load, high repetition resistance training program increases bone mineral density in untrained adults. J. Sports Med. Phys. Fit. 2017, 57, 70–76. [Google Scholar] [CrossRef]
- Vetrovsky, T.; Steffl, M.; Stastny, P.; Tufano, J.J. The efficacy and safety of lower-limb plyometric training in older adults: A systematic review. Sports Med. 2019, 49, 113–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dai, B.; Garrett, W.E.; Gross, M.T.; Padua, D.A.; Queen, R.M.; Yu, B.J.J.O.S. The effect of performance demands on lower extremity biomechanics during landing and cutting tasks. J. Sport Health Sci. 2019, 8, 228–234. [Google Scholar] [CrossRef] [Green Version]
- Van Melick, N.; Meddeler, B.M.; Hoogeboom, T.J.; Nijhuis-van der Sanden, M.W.G.; van Cingel, R.E.H. How to determine leg dominance: The agreement between self-reported and observed performance in healthy adults. PLoS ONE 2017, 12, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Granacher, U.; Muehlbauer, T.; Gruber, M. A qualitative review of balance and strength performance in healthy older adults: Impact for testing and training. J. Aging Res. 2012, 2012, 708905. [Google Scholar] [CrossRef]
- Brooke-Wavell, K.; Stensel, D.J. Exercise and children’s bone health. J. Fam. Health Care 2008, 18, 205–208. [Google Scholar] [PubMed]
- Azuma, K.; Osawa, Y.; Tabata, S.; Katsukawa, F.; Ishida, H.; Oguma, Y.; Kawai, T.; Itoh, H.; Okuda, S.; Oguchi, S. Decrease in regional body fat after long-term high-intensity interval training. J. Phys. Fit. Sports Med. 2017, 6, 103–110. [Google Scholar] [CrossRef] [Green Version]
- Trapp, E.G.; Chisholm, D.J.; Freund, J.; Boutcher, S.H. The effects of high-intensity intermittent exercise training on fat loss and fasting insulin levels of young women. Int. J. Obes. 2008, 32, 684–691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boudou, P.; Sobngwi, E.; Mauvais-Jarvis, F.; Vexiau, P.; Gautier, J.F. Absence of exercise-induced variations in adiponectin levels despite decreased abdominal adiposity and improved insulin sensitivity in type 2 diabetic men. Eur. J. Endocrinol. 2003, 149, 421–424. [Google Scholar] [CrossRef]
- Maillard, F.; Pereira, B.; Boisseau, N. Effect of high-intensity interval training on total, abdominal and visceral fat mass: A meta-analysis. Sports Med. 2018, 48, 269–288. [Google Scholar] [CrossRef]
- Shing, C.M.; Webb, J.J.; Driller, M.W.; Williams, A.D.; Fell, J.W. Circulating adiponectin concentration and body composition are altered in response to high-intensity interval training. J. Strength. Cond. Res. 2013, 27, 2213–2218. [Google Scholar] [CrossRef]
- Racil, G.; Ben Ounis, O.; Hammouda, O.; Kallel, A.; Zouhal, H.; Chamari, K.; Amri, M. Effects of high vs. moderate exercise intensity during interval training on lipids and adiponectin levels in obese young females. Eur. J. Appl. Physiol. 2013, 113, 2531–2540. [Google Scholar] [CrossRef]
- Kishida, K.; Funahashi, T.; Shimomura, I. Adiponectin as a routine clinical biomarker. Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 119–130. [Google Scholar] [CrossRef]
- Keating, S.E.; Johnson, N.A.; Mielke, G.I.; Coombes, J.S. A systematic review and meta-analysis of interval training versus moderate-intensity continuous training on body adiposity. Obes. Rev. 2017, 18, 943–964. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.; de Lannoy, L.; Stotz, P.J. Separate effects of intensity and amount of exercise on interindividual cardiorespiratory fitness response. Mayo. Clin. Proc. 2015, 90, 1506–1514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibala, M.J.; Gillen, J.B.; Percival, M.E. Physiological and health-related adaptations to low-volume interval training: Influences of nutrition and sex. Sports Med. 2014, 44 (Suppl. S2), S127–S137. [Google Scholar] [CrossRef] [Green Version]
- Wenger, H.A.; Bell, G.J. The interactions of intensity, frequency and duration of exercise training in altering cardiorespiratory fitness. Sports Med. 1986, 3, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, D.; Anderson, K.; Graff, M.; Strutz, V. The effect of high-intensity circuit training on physical fitness. J. Sports Med. Phys. Fit. 2016, 56, 534–540. [Google Scholar]
- Martland, R.; Mondelli, V.; Gaughran, F.; Stubbs, B. Can high-intensity interval training improve physical and mental health outcomes? A meta-review of 33 systematic reviews across the lifespan. J. Sports Sci. 2020, 38, 430–469. [Google Scholar] [CrossRef]
- Noll, M.; Mendonça, C.R.; Rodrigues, A.; de Almeida, A.A.; Noll, P. Narrative review of the influence of high-intensity interval training on adolescents’ bone health: Commentary and perspectives. Transl. Pediatr. 2021, 10, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Goodman, C.A.; Hornberger, T.A.; Robling, A.G. Bone and skeletal muscle: Key players in mechanotransduction and potential overlapping mechanisms. Bone 2015, 80, 24–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duncan, R.L.; Turner, C.H. Mechanotransduction and the functional response of bone to mechanical strain. Calcif. Tissue Int. 1995, 57, 344–358. [Google Scholar] [CrossRef]
- Meakin, L.B.; Delisser, P.J.; Galea, G.L.; Lanyon, L.E.; Price, J.S. Disuse rescues the age-impaired adaptive response to external loading in mice. Osteoporos Int. 2015, 26, 2703–2708. [Google Scholar] [CrossRef] [Green Version]
- Trimpou, P.; Bosaeus, I.; Bengtsson, B.A.; Landin-Wilhelmsen, K. High correlation between quantitative ultrasound and DXA during 7 years of follow-up. Eur. J. Radiol. 2010, 73, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Fricke, O.; Schoenau, E. The ‘Functional Muscle-Bone Unit’: Probing the relevance of mechanical signals for bone development in children and adolescents. Growth Horm. IGF Res. 2007, 17, 1–9. [Google Scholar] [CrossRef]
- Grimal, Q.; Grondin, J.; Guerard, S.; Barkmann, R.; Engelke, K.; Gluer, C.C.; Laugier, P. Quantitative Ultrasound of cortical bone in the femoral neck predicts femur strength: Results of a pilot study. J. Bone Miner. Res. 2013, 28, 302–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Y.; Guo, B.; Gong, J.; Xu, H.; Bai, Z. The correlation between calcaneus stiffness index calculated by QUS and total body BMD assessed by DXA in Chinese children and adolescents. J. Bone Miner. Metab. 2013, 32, 159–166. [Google Scholar] [CrossRef]
- Zha, X.-Y.; Hu, Y.; Pang, X.-N.; Chang, G.-L.; Li, L. Diagnostic value of Osteoporosis Self-Assessment Tool for Asians (OSTA) and quantitative bone ultrasound (QUS) in detecting high-risk populations for osteoporosis among elderly Chinese men. J. Bone Miner. Metab. 2014, 33, 230–238. [Google Scholar] [CrossRef]


| ShEx | HiEx | HiExVi | HiInEx | p-Value | |
|---|---|---|---|---|---|
| Age (year) | 12.3 ± 0.5 | 12.3 ± 0.5 | 12.3 ± 0.5 | 12.2 ± 0.4 | 0.763 |
| Height (cm) | 163.7 ± 8.2 | 163.1 ± 6.7 | 164.2 ± 7.3 | 163.8 ± 8.0 | 0.926 |
| Weight (kg) | 53.3 ± 11.1 | 52.9 ± 9.9 | 52.7 ± 9.5 | 54.0 ± 14.3 | 0.954 |
| BMI (kg/m2) | 19.8 ± 3.6 | 19.8 ± 2.9 | 19.4 ± 2.4 | 19.9 ± 4.1 | 0.919 |
| Calcium Intake (mg/week) | 2340 ± 566 | 2409 ± 724 | 2374 ± 803 | 2336 ± 729 | 0.963 |
| Energy Intake (kcal/day) | 1310 ± 161 | 1352 ± 195 | 1433 ± 167 | 1335 ± 292 | 0.053 |
| Extracurricular Exercise (MET-Hours/week) | 11.5 ± 11.5 | 11.0 ± 12.3 | 10.4 ± 14.1 | 11.7 ± 14.9 | 0.983 |
| F (hp2) | |||
|---|---|---|---|
| Time Effect | Time × Group Interaction | Group Effect | |
| SOS | 401.670 (0.715) * | 13.630 (0.204) * | 2.866 (0.051) * |
| PBF | 28.772 (0.152) * | 5.052 (0.087) * | 1.241 (0.023) |
| WHR | 45.561 (0.222) * | 6.640 (0.111) * | 0.499 (0.009) |
| Balance | 280.660 (0.637) * | 21.935 (0.291) * | 0.871 (0.016) |
| VC | 80.760 (0.335) * | 5.794 (0.098) * | 0.579 (0.011) |
| SLJ | 69.212 (0.302) * | 4.228 (0.073) * | 0.199 (0.004) |
| SU | 15.873 (0.090) * | 3.540 (0.062) * | 0.624 (0.012) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, X.; Fu, J.; Tian, J.; Yang, Y.; Liang, W.; Fan, W.; Zhao, R. The Efficacy of Brief School-Based Exercise Programs in Improving Pubertal Bone Mass and Physical Fitness: A Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2021, 18, 9648. https://doi.org/10.3390/ijerph18189648
Tian X, Fu J, Tian J, Yang Y, Liang W, Fan W, Zhao R. The Efficacy of Brief School-Based Exercise Programs in Improving Pubertal Bone Mass and Physical Fitness: A Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2021; 18(18):9648. https://doi.org/10.3390/ijerph18189648
Chicago/Turabian StyleTian, Xin, Jian Fu, Jiaxue Tian, Yu Yang, Wenjie Liang, Wencui Fan, and Renqing Zhao. 2021. "The Efficacy of Brief School-Based Exercise Programs in Improving Pubertal Bone Mass and Physical Fitness: A Randomized Controlled Trial" International Journal of Environmental Research and Public Health 18, no. 18: 9648. https://doi.org/10.3390/ijerph18189648







