Redox Regulation and Oxidative Stress in Mammalian Oocytes and Embryos Developed In Vivo and In Vitro
Abstract
:1. Introduction
2. Cellular ROS
2.1. Sources of Cellular ROS

2.2. The (Patho)Physiological Roles of ROS
2.2.1. Oxidative Eustress
2.2.2. Oxidative Distress
3. The Effect of Oxidative Eustress and Distress on Oocyte Maturation, Fertilisation, and Embryo Development

3.1. Oocyte Maturation and Fertilisation
3.2. The Preimplantation Embryo
3.2.1. Use of Antioxidants for In Vitro Culture

3.2.2. Amino Acids and Polyamines
3.2.3. O2 Tension and Oxidative Stress
3.2.4. Laboratory Light
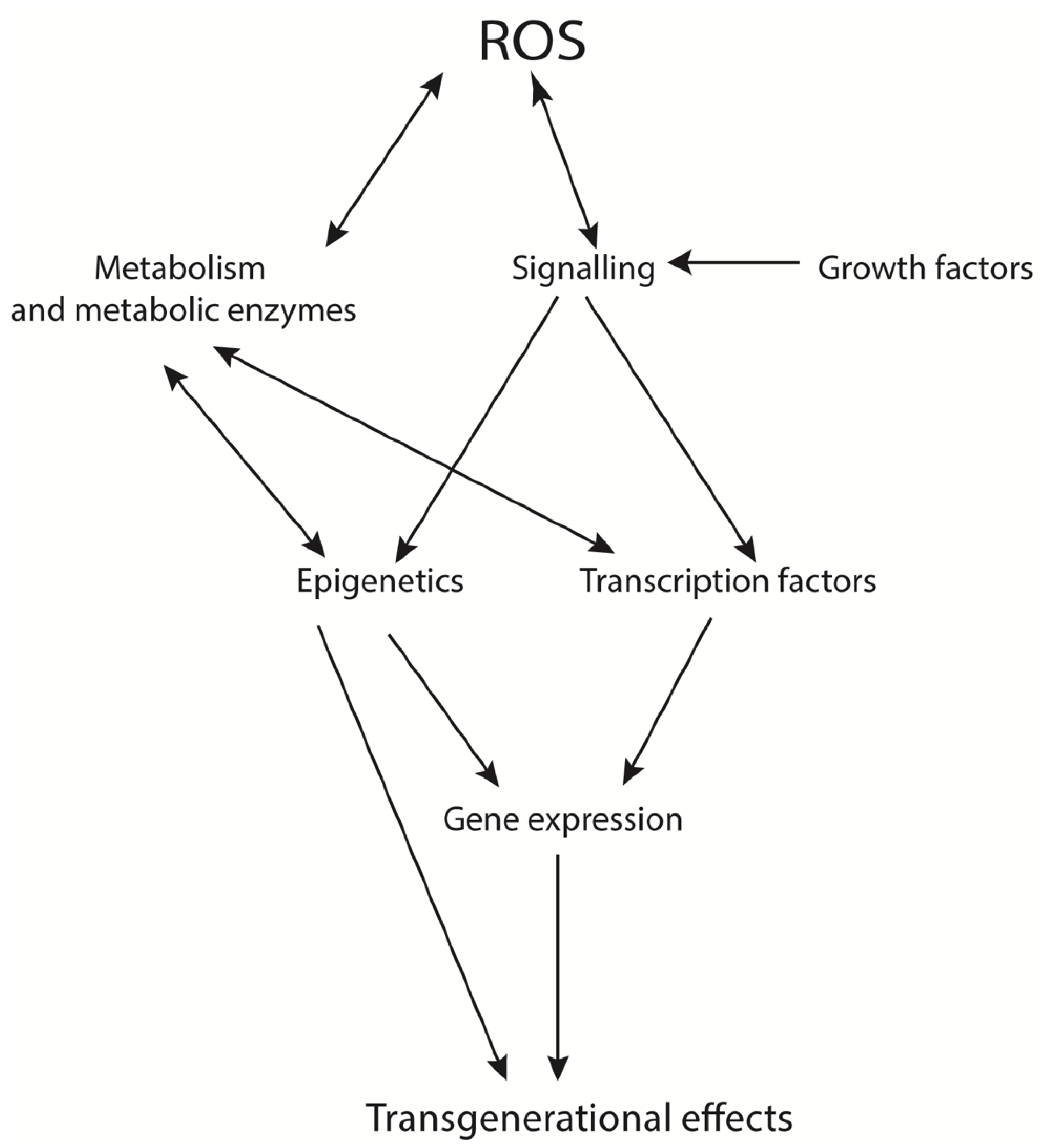
4. Transgenerational Effects of ROS
5. Emerging Therapies and Trends in ART
| Antioxidant | Trial Type | Population | Method | Results | Reference |
|---|---|---|---|---|---|
| Melatonin | Retrospective | Women with poor oocyte quality or low embryo quality in previous cycles. | 3 mg/day oral melatonin for ≥2 weeks until the day of hCG trigger dose. | Improved fertilisation rates and improved embryo quality. No effect on oocyte maturation or percentage of blastocyst development. | [209] |
| Melatonin | Randomised clinical trial | Women 20–45 years undergoing IVF. | 3 mg/day oral melatonin from the day of GnRH antagonist until the day of embryo transfer. | Increased percentage of mature oocytes and grade 1 embryos. No effect on pregnancy rates | [210] |
| Melatonin | Randomised pilot study | Women with unexplained infertility undergoing a second IVF cycle. | Groups allocated to 0, 3, or 6 mg/day oral melatonin from first appointment to start of ovarian stimulation (i.e., 40 days). | Both doses of melatonin increased levels of melatonin, TAC, and lipid peroxidation in follicular fluid; 6 mg/day melatonin increased SOD. Both 3 and 6 mg/day melatonin increased the number of oocytes retrieved, fertilisation percentage, and number of transferable embryos. | [211] |
| Myoinositol and melatonin | Randomised double-blind clinical trial | Women with PCOS undergoing IVF treatment. | Women were allocated to the following groups: Control, 4 g myoinositol or 4 g myoinositol + 3 mg melatonin, orally twice per day, from cycle day 1 to 14 days post embryo transfer. | Melatonin increased the percentage of mature oocytes and number of high-grade embryos. No effect on pregnancy rate. | [212] |
| Myoinositol and melatonin | Prospective clinical trial | Women aged 30–40 with one or more unsuccessful IVF cycles due to poor oocyte quality. | Daily oral supplementation with 4 g myoinositol + 1.8 mg melatonin for 3 months prior to IVF cycle. | Increased number of mature oocytes. No effect on the percentage of mature oocytes, fertilised embryos, or grade of embryos. | [213] |
| Coenzyme Q10 | Randomised control trial | Women aged <35, with poor ovarian response to stimulation undergoing IVF/ICSI. | Oral administration of 200 mg CoQ10 3 times per day for 60 days prior to IVF/ICSI cycle. | Decreased day 3 FSH, increased peak E2 concentration, number of oocytes retrieved, fertilisation rate, and embryo quality. No effect on clinical pregnancy rate. | [214] |
| Coenzyme Q10 | Controlled clinical study | Women undergoing IVF-ET for unexplained or tubal disease-related infertility. | Oral supplementation of 200 mg CoQ10 daily for 30 days before oocyte pick up. | Increased follicular fluid CoQ10 in its reduced form. Decreased TAC in patients aged >35 years. | [215] |
| Coenzyme Q10 | Retrospective study | Women with poor ovarian reserve undergoing IUI or IVF cycles. | Daily oral administration of either 75 mg DHEA alone or 75 mg DHEA + 600 mg CoQ10. | Improved ovarian responsiveness with an increase in antral follicular count and number of mature follicles. No change in blastocyst development or pregnancy rates. | [216] |
| Growth hormone | Randomised control clinical trial | Women with poor ovarian reserve undergoing IVF. | 4 IU/day growth hormone injected subcutaneously from day 2 of the previous menstrual cycle until trigger day (36–48 days). | Increased endometrial thickness, implantation rate, and clinical pregnancy. Increased TAC, decreased total oxidative stress index in follicular fluid. Decreased ROS in granulosa cells. Increased embryo quality, implantation rate, and clinical pregnancies. | [217] |
| N-acetyl cysteine (NAC) | Placebo-controlled double-blind, randomised clinical trial | Women with PCOS undergoing IVF. | Oral administration of 1.2 g NAC on days 3–7 of the menstrual cycle. | Increased number of follicles, ovulation rate, pregnancy rate, and endometrial thickness. | [218] |
| Pentoxifylline and vitamin E | Randomised clinical trial | Women <39 years of age with various forms of infertility undergoing ICSI-ZIFT. | Daily oral administration of 400 mg vitamin E and 400 mg pentoxifylline for two cycles before ZIFT. | Improved clinical pregnancy rate. | [224,229] |
| Multivitamin and mineral | Controlled clinical trial | Women undergoing infertility treatment. | Oral multivitamin and mineral supplementation for 45 days before oocyte collection. | Decreased lipid peroxidase levels in follicular fluid and serum. Increased GSH and vitamins C and E in follicular fluid. | [219] |
| FertiMax2 (Vitamins C and E, zinc, selenium, L-carnitine, folic acid, and coenzyme Q10) | Preliminary clinical study | Males with male factor infertility undergoing IVF/ICSI. | Oral administration of Fertimax2 for 2–5 months prior to partner’s IVF/ICSI cycle. | Increased fertilisation, cleavage, embryo quality, implantation, and clinical pregnancy rate. No effect on semen parameters. | [220] |
| Menevit (Lycopene, vitamins C and E, zinc, selenium, folate, garlic oil) | Retrospective cohort analysis | Males with male factor infertility undergoing IVF/ICSI. | Single daily capsule for an unrecorded amount of time. | Increased clinical pregnancy and live birth rate. | [225] |
| Micronutrient antioxidants (Vitamins, folates and minerals) | Preliminary study | Women aged >39 years undergoing infertility treatments with one failed embryo transfer. | After one typical GnRH antagonist cycle, failed patients were prescribed a daily capsule of micronutrient antioxidants for three months before embryo transfer. | Increased TAC and free thiol availability in follicular fluid and serum. Decreased number of poor grade embryos. No change in fertilisation or cleavage rates. | [222] |
| Mixed antioxidant formulation (Vitamins C and E, selenium, L-carnitine, zinc, folic acid, lycopene) | Randomised controlled trial | Males with either low sperm concentration, motility, morphology or high DNA fragmentation. | Allocated antioxidant or placebo for 3–6 months. Semen parameters including concentration, motility, morphology, and DNA fragmentation measured. | Decreased sperm concentration, total sperm count, and total motile sperm. No change in morphology, motility, or DNA fragmentation. No change in pregnancy/live birth rates. | [223] |
| Antioxidant | Trial Type | Population | Method | Results | Reference |
|---|---|---|---|---|---|
| Mixed antioxidant formulation (acetyl-L-carnitine, N-acetyl cysteine, alpha-lipoic acid) | Blinded randomised control sibling oocyte study | IVF/ICSI patients under 40 years undergoing fertility treatment. | Formulation added to G-series medium, including 10 μM acetyl-L-carnitine, 10 μM N-acetylcysteine, and 5 μM alpha-lipoic acid added to both fertilisation and culture media. | No effect on fertilisation. Increased percentage of good quality embryos on day 3 (patients <35 years). Increased number of patients (35–40 years) receiving a positive pregnancy test, increased percentage with fetal heart beat and ongoing pregnancy. | [230] |
| L-carnitine | Retrospective clinical trial | Patients <40 years undergoing infertility treatment. | 1 mM L-carnitine added to embryo culture medium from day 1 to day 6. | No effect on percentage of embryos developed to blastocyst stage but increased percentage of good quality embryos on days 2, 3, and 5. Increased blastocyst ICM and TE cell numbers and increased clinical and ongoing pregnancies. | [231] |
| Coenzyme Q10 | Randomised clinical trial | Women 38–46 years and ≤30 years undergoing IVF. | GV stage oocytes randomly allocated to no treatment or antioxidant treatment where oocytes were cultured ±50 μM CoQ10 for 24 h. | For patients 38–46 years, CoQ10 increased oocyte maturation and decreased oocyte aneuploidy. No effect on oocyte maturation or aneuploidy in ≤30 years group. | [232] |
| Mixed antioxidants (L-carnitine, taurine vitamin B5, vitamin C; other vitamins that are not antioxidants were also added to this formulation) | Non-interventional sibling oocyte study | Women ≤42 years old undergoing ICSI cycles. | Oocytes randomly allocated to medium containing mixed antioxidants or standard continuous single culture medium. | Antioxidant containing medium had no effect on blastulation but showed slower compaction and blastulation rates, and blastocysts were of poorer quality. | [233] |
| Alpha-lipoic acid | Randomised clinical trial | Normozoospermic men undergoing IVF/ICSI cycles. | Semen samples were randomly allocated to sperm wash medium ±0.02 mM alpha-lipoic acid during centrifugation and incubation for 1 h. | Sperm viability and motility increased while DNA damage and ROS decreased when prepared in wash medium containing alpha-lipoic acid. | [234] |
| L-carnitine | Randomised clinical trial | Infertile men with normospermia or asthenozoospermia. | Cryopreservation of semen samples in medium containing 1 g/L L-carnitine. | Improved sperm parameters after thawing including motility and viability as well as decreased DNA fragmentation. | [235] |
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dumollard, R.; Duchen, M.; Carroll, J. The role of mitochondrial function in the oocyte and embryo. Curr. Top. Dev. Biol. 2007, 77, 21–49. [Google Scholar] [CrossRef]
- Harvey, A.J.; Kind, K.L.; Thompson, J.G. REDOX regulation of early embryo development. Reproduction 2002, 123, 479–486. [Google Scholar] [CrossRef]
- Schafer, F.Q.; Buettner, G.R. Redox environment of the cell as viewed through the redox state of the glutathione disulfide/glutathione couple. Free Radic. Biol. Med. 2001, 30, 1191–1212. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef]
- Sies, H. Oxidative Stress. In Stress: Physiology, Biochemistry, and Pathology; Fink, G., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 153–163. [Google Scholar] [CrossRef]
- Sies, H.; Chance, B. The steady state level of catalase compound I in isolated hemoglobin-free perfused rat liver. FEBS Lett. 1970, 11, 172–176. [Google Scholar] [CrossRef] [Green Version]
- Harper, M.E.; Bevilacqua, L.; Hagopian, K.; Weindruch, R.; Ramsey, J.J. Ageing, oxidative stress, and mitochondrial uncoupling. Acta Physiol. Scand. 2004, 182, 321–331. [Google Scholar] [CrossRef]
- Mailloux, R.J.; Jin, X.; Willmore, W.G. Redox regulation of mitochondrial function with emphasis on cysteine oxidation reactions. Redox Biol. 2014, 2, 123–139. [Google Scholar] [CrossRef] [Green Version]
- Boveris, A.; Chance, B. The mitochondrial generation of hydrogen peroxide. General properties and effect of hyperbaric oxygen. Biochem. J. 1973, 134, 707–716. [Google Scholar] [CrossRef]
- Arrigo, A.P. Gene expression and the thiol redox state. Free Radic. Biol. Med. 1999, 27, 936–944. [Google Scholar] [CrossRef]
- Guerin, P.; El Mouatassim, S.; Menezo, Y. Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Hum. Reprod. Update 2001, 7, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Huo, Y.; Qin, Q.; Zhang, L.; Kuo, Y.; Wang, H.; Yan, L.; Li, R.; Zhang, X.; Yan, J.; Qiao, J. Effects of oocyte vitrification on the behaviors and physiological indexes of aged first filial generation mice. Cryobiology 2020, 95, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Calle, A.; Fernandez-Gonzalez, R.; Ramos-Ibeas, P.; Laguna-Barraza, R.; Perez-Cerezales, S.; Bermejo-Alvarez, P.; Ramirez, M.A.; Gutierrez-Adan, A. Long-term and transgenerational effects of in vitro culture on mouse embryos. Theriogenology 2012, 77, 785–793. [Google Scholar] [CrossRef]
- Ruebel, M.L.; Latham, K.E. Listening to mother: Long-term maternal effects in mammalian development. Mol. Reprod. Dev. 2020, 87, 399–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, R.; Jia, Z.; Trush, M.A. Defining ROS in Biology and Medicine. React. Oxyg. Species (Apex) 2016, 1, 9–21. [Google Scholar] [CrossRef] [Green Version]
- Onyango, A.N. Endogenous Generation of Singlet Oxygen and Ozone in Human and Animal Tissues: Mechanisms, Biological Significance, and Influence of Dietary Components. Oxid. Med. Cell. Longev. 2016, 2016, 2398573. [Google Scholar] [CrossRef] [Green Version]
- Pisoschi, A.M.; Pop, A. The role of antioxidants in the chemistry of oxidative stress: A review. Eur. J. Med. Chem. 2015, 97, 55–74. [Google Scholar] [CrossRef]
- He, A.; Dean, J.M.; Lodhi, I.J. Peroxisomes as cellular adaptors to metabolic and environmental stress. Trends Cell Biol. 2021, 31, 656–670. [Google Scholar] [CrossRef]
- Ryter, S.W.; Kim, H.P.; Hoetzel, A.; Park, J.W.; Nakahira, K.; Wang, X.; Choi, A.M. Mechanisms of cell death in oxidative stress. Antioxid. Redox Signal. 2007, 9, 49–89. [Google Scholar] [CrossRef]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid. Med. Cell. Longev. 2016, 2016, 1245049. [Google Scholar] [CrossRef]
- Panday, A.; Sahoo, M.K.; Osorio, D.; Batra, S. NADPH oxidases: An overview from structure to innate immunity-associated pathologies. Cell. Mol. Immunol. 2015, 12, 5–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kodaman, P.H.; Behrman, H.R. Endocrine-regulated and protein kinase C-dependent generation of superoxide by rat preovulatory follicles. Endocrinology 2001, 142, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Ufer, C.; Wang, C.C.; Borchert, A.; Heydeck, D.; Kuhn, H. Redox control in mammalian embryo development. Antioxid. Redox Signal. 2010, 13, 833–875. [Google Scholar] [CrossRef]
- Liu, Y.; Fiskum, G.; Schubert, D. Generation of reactive oxygen species by the mitochondrial electron transport chain. J. Neurochem. 2002, 80, 780–787. [Google Scholar] [CrossRef]
- Oshino, N.; Chance, B.; Sies, H.; Bücher, T. The role of H2O2 generation in perfused rat liver and the reaction of catalase compound I and hydrogen donors. Arch. Biochem. Biophys. 1973, 154, 117–131. [Google Scholar] [CrossRef]
- Murphy, M.P. How mitochondria produce reactive oxygen species. Biochem. J. 2009, 417, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Cadenas, E.; Davies, K.J. Mitochondrial free radical generation, oxidative stress, and aging. Free Radic. Biol. Med. 2000, 29, 222–230. [Google Scholar] [CrossRef]
- Hancock, C.N.; Liu, W.; Alvord, W.G.; Phang, J.M. Co-regulation of mitochondrial respiration by proline dehydrogenase/oxidase and succinate. Amino Acids 2016, 48, 859–872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goncalves, R.L.; Rothschild, D.E.; Quinlan, C.L.; Scott, G.K.; Benz, C.C.; Brand, M.D. Sources of superoxide/H2O2 during mitochondrial proline oxidation. Redox Biol. 2014, 2, 901–909. [Google Scholar] [CrossRef] [Green Version]
- Pandhare, J.; Donald, S.P.; Cooper, S.K.; Phang, J.M. Regulation and function of proline oxidase under nutrient stress. J. Cell. Biochem. 2009, 107, 759–768. [Google Scholar] [CrossRef] [Green Version]
- Phang, J.M.; Donald, S.P.; Pandhare, J.; Liu, Y. The metabolism of proline, a stress substrate, modulates carcinogenic pathways. Amino Acids 2008, 35, 681–690. [Google Scholar] [CrossRef] [Green Version]
- Phang, J. The regulatory functions of proline and pyrroline-5-carboxylic acid. Curr. Top. Cell. Regul. 1985, 25, 91–132. [Google Scholar] [PubMed]
- Phang, J.M.; Yeh, G.C.; Hagedorn, C.H. The intercellular proline cycle. Life Sci. 1981, 28, 53–58. [Google Scholar] [CrossRef]
- Natarajan, S.K.; Zhu, W.; Liang, X.; Zhang, L.; Demers, A.J.; Zimmerman, M.C.; Simpson, M.A.; Becker, D.F. Proline dehydrogenase is essential for proline protection against hydrogen peroxide-induced cell death. Free Radic. Biol. Med. 2012, 53, 1181–1191. [Google Scholar] [CrossRef] [Green Version]
- Wong, H.S.; Benoit, B.; Brand, M.D. Mitochondrial and cytosolic sources of hydrogen peroxide in resting C2C12 myoblasts. Free Radic. Biol. Med. 2019, 130, 140–150. [Google Scholar] [CrossRef]
- Rampon, C.; Volovitch, M.; Joliot, A.; Vriz, S. Hydrogen Peroxide and Redox Regulation of Developments. Antioxidants 2018, 7, 159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dutta, S.; Henkel, R.; Sengupta, P.; Agarwal, A. Physiological role of ROS in sperm function. In Male Infertility; Springer: Cham, Switzerland, 2020; pp. 337–345. [Google Scholar]
- Ray, P.D.; Huang, B.W.; Tsuji, Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell. Signal. 2012, 24, 981–990. [Google Scholar] [CrossRef] [Green Version]
- Mailloux, R.J.; McBride, S.L.; Harper, M.E. Unearthing the secrets of mitochondrial ROS and glutathione in bioenergetics. Trends Biochem. Sci. 2013, 38, 592–602. [Google Scholar] [CrossRef] [PubMed]
- Harris, C.; Hansen, J.M. Oxidative stress, thiols, and redox profiles. In Developmental Toxicology; Humana Press: Totowa, NJ, USA, 2012; pp. 325–346. [Google Scholar]
- Reczek, C.R.; Chandel, N.S. ROS-dependent signal transduction. Curr. Opin. Cell Biol. 2015, 33, 8–13. [Google Scholar] [CrossRef] [Green Version]
- Essers, M.A.; Weijzen, S.; de Vries-Smits, A.M.; Saarloos, I.; de Ruiter, N.D.; Bos, J.L.; Burgering, B.M. FOXO transcription factor activation by oxidative stress mediated by the small GTPase Ral and JNK. EMBO J. 2004, 23, 4802–4812. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burhans, W.C.; Heintz, N.H. The cell cycle is a redox cycle: Linking phase-specific targets to cell fate. Free Radic. Biol. Med. 2009, 47, 1282–1293. [Google Scholar] [CrossRef] [PubMed]
- Klotz, L.O.; Sanchez-Ramos, C.; Prieto-Arroyo, I.; Urbanek, P.; Steinbrenner, H.; Monsalve, M. Redox regulation of FoxO transcription factors. Redox Biol. 2015, 6, 51–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Veal, E.A.; Day, A.M.; Morgan, B.A. Hydrogen peroxide sensing and signaling. Mol. Cell 2007, 26, 1–14. [Google Scholar] [CrossRef]
- Fourquet, S.; Guerois, R.; Biard, D.; Toledano, M.B. Activation of NRF2 by nitrosative agents and H2O2 involves KEAP1 disulfide formation. J. Biol. Chem. 2010, 285, 8463–8471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kobayashi, Y.; Oguro, A.; Imaoka, S. Feedback of hypoxia-inducible factor-1alpha (HIF-1alpha) transcriptional activity via redox factor-1 (Ref-1) induction by reactive oxygen species (ROS). Free Radic. Res. 2021, 55, 154–164. [Google Scholar] [CrossRef]
- Chan, D.A.; Sutphin, P.D.; Yen, S.-E.; Giaccia, A.J. Coordinate regulation of the oxygen-dependent degradation domains of hypoxia-inducible factor 1α. Mol. Cell. Biol. 2005, 25, 6415–6426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dengler, V.L.; Galbraith, M.; Espinosa, J.M. Transcriptional regulation by hypoxia inducible factors. Crit. Rev. Biochem. Mol. Biol. 2014, 49, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Colavitti, R.; Rovira, I.I.; Finkel, T. Redox-dependent transcriptional regulation. Circ. Res. 2005, 97, 967–974. [Google Scholar] [CrossRef] [Green Version]
- Kumari, S.; Badana, A.K.; G, M.M.; G, S.; Malla, R. Reactive Oxygen Species: A Key Constituent in Cancer Survival. Biomark. Insights 2018, 13, 1177271918755391. [Google Scholar] [CrossRef] [Green Version]
- Moloney, J.N.; Cotter, T.G. ROS signalling in the biology of cancer. Semin. Cell Dev. Biol. 2018, 80, 50–64. [Google Scholar] [CrossRef] [PubMed]
- Roy, K.; Wu, Y.; Meitzler, J.L.; Juhasz, A.; Liu, H.; Jiang, G.; Lu, J.; Antony, S.; Doroshow, J.H. NADPH oxidases and cancer. Clin. Sci. 2015, 128, 863–875. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.; Apostolova, N.; Herance, J.R.; Rovira-Llopis, S.; Hernandez-Mijares, A.; Victor, V.M. Perspectives and potential applications of mitochondria-targeted antioxidants in cardiometabolic diseases and type 2 diabetes. Med. Res. Rev. 2014, 34, 160–189. [Google Scholar] [CrossRef] [Green Version]
- Fiaschi, T.; Chiarugi, P. Oxidative stress, tumor microenvironment, and metabolic reprogramming: A diabolic liaison. Int. J. Cell Biol. 2012, 2012, 762825. [Google Scholar] [CrossRef] [Green Version]
- Giorgi, C.; Danese, A.; Missiroli, S.; Patergnani, S.; Pinton, P. Calcium dynamics as a machine for decoding signals. Trends Cell Biol. 2018, 28, 258–273. [Google Scholar] [CrossRef] [PubMed]
- Gorlach, A.; Bertram, K.; Hudecova, S.; Krizanova, O. Calcium and ROS: A mutual interplay. Redox Biol. 2015, 6, 260–271. [Google Scholar] [CrossRef] [Green Version]
- Sharov, V.S.; Dremina, E.S.; Galeva, N.A.; Williams, T.D.; Schoneich, C. Quantitative mapping of oxidation-sensitive cysteine residues in SERCA in vivo and in vitro by HPLC-electrospray-tandem MS: Selective protein oxidation during biological aging. Biochem. J. 2006, 394, 605–615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Higaki, Y.; Mikami, T.; Fujii, N.; Hirshman, M.F.; Koyama, K.; Seino, T.; Tanaka, K.; Goodyear, L.J. Oxidative stress stimulates skeletal muscle glucose uptake through a phosphatidylinositol 3-kinase-dependent pathway. Am. J. Physiol. Endocrinol. Metab. 2008, 294, E889–E897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozlovsky, N.; Rudich, A.; Potashnik, R.; Ebina, Y.; Murakami, T.; Bashan, N. Transcriptional activation of the Glut1 gene in response to oxidative stress in L6 myotubes. J. Biol. Chem. 1997, 272, 33367–33372. [Google Scholar] [CrossRef] [Green Version]
- Fischer, B.; Kunzel, W.; Kleinstein, J.; Gips, H. Oxygen tension in follicular fluid falls with follicle maturation. Eur. J. Obstet. Gynecol. Reprod. Biol. 1992, 43, 39–43. [Google Scholar] [CrossRef]
- Fischer, B.; Bavister, B.D. Oxygen tension in the oviduct and uterus of rhesus monkeys, hamsters and rabbits. J. Reprod. Fertil. 1993, 99, 673–679. [Google Scholar] [CrossRef]
- Lim, M.; Thompson, J.G.; Dunning, K.R. Hypoxia and ovarian function: Follicle development, ovulation, oocyte maturation. Reproduction 2021, 161, F33–F40. [Google Scholar] [CrossRef] [PubMed]
- Morin, S.J. Oxygen tension in embryo culture: Does a shift to 2% O 2 in extended culture represent the most physiologic system? J. Assist. Reprod. Genet. 2017, 34, 309–314. [Google Scholar] [CrossRef]
- El Mouatassim, S.; Guerin, P.; Menezo, Y. Mammalian oviduct and protection against free oxygen radicals: Expression of genes encoding antioxidant enzymes in human and mouse. Eur. J. Obstet. Gynecol. Reprod. Biol. 2000, 89, 1–6. [Google Scholar] [CrossRef]
- Ambekar, A.S.; Nirujogi, R.S.; Srikanth, S.M.; Chavan, S.; Kelkar, D.S.; Hinduja, I.; Zaveri, K.; Prasad, T.S.; Harsha, H.C.; Pandey, A.; et al. Proteomic analysis of human follicular fluid: A new perspective towards understanding folliculogenesis. J. Proteom. 2013, 87, 68–77. [Google Scholar] [CrossRef]
- Agarwal, A.; Aponte-Mellado, A.; Premkumar, B.J.; Shaman, A.; Gupta, S. The effects of oxidative stress on female reproduction: A review. Reprod. Biol. Endocrinol. 2012, 10, 49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Mouatassim, S.; Guerin, P.; Menezo, Y. Expression of genes encoding antioxidant enzymes in human and mouse oocytes during the final stages of maturation. Mol. Hum. Reprod. 1999, 5, 720–725. [Google Scholar] [CrossRef]
- Ozawa, M.; Nagai, T.; Somfai, T.; Nakai, M.; Maedomari, N.; Miyazaki, H.; Kaneko, H.; Noguchi, J.; Kikuchi, K. Cumulus cell-enclosed oocytes acquire a capacity to synthesize GSH by FSH stimulation during in vitro maturation in pigs. J. Cell. Physiol. 2010, 222, 294–301. [Google Scholar] [CrossRef]
- Nasr-Esfahani, M.H.; Aitken, J.R.; Johnson, M.H. Hydrogen peroxide levels in mouse oocytes and early cleavage stage embryos developed in vitro or in vivo. Development 1990, 109, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Lane, M.; Gardner, D.K. Embryo culture medium: Which is the best? Best Pract. Res. Clin. Obstet. Gynaecol. 2007, 21, 83–100. [Google Scholar] [CrossRef]
- Gardner, D.K.; Sakkas, D. Mouse embryo cleavage, metabolism and viability: Role of medium composition. Hum. Reprod. 1993, 8, 288–295. [Google Scholar] [CrossRef]
- Biggers, J.D. Thoughts on embryo culture conditions. Reprod. Biomed. Online 2002, 4 (Suppl. 1), 30–38. [Google Scholar] [CrossRef]
- Martin-Romero, F.J.; Miguel-Lasobras, E.M.; Dominguez-Arroyo, J.A.; Gonzalez-Carrera, E.; Alvarez, I.S. Contribution of culture media to oxidative stress and its effect on human oocytes. Reprod. Biomed. Online 2008, 17, 652–661. [Google Scholar] [CrossRef]
- Orsi, N.M.; Leese, H.J. Amino acid metabolism of preimplantation bovine embryos cultured with bovine serum albumin or polyvinyl alcohol. Theriogenology 2004, 61, 561–572. [Google Scholar] [CrossRef]
- Palasz, A.; Tornesi, M.; Archer, J.; Mapletoft, R. Media alternatives for the collection, culture and freezing of mouse and cattle embryos. Theriogenology 1995, 44, 705–714. [Google Scholar] [CrossRef]
- O’Flaherty, C.; de Lamirande, E.; Gagnon, C. Reactive oxygen species modulate independent protein phosphorylation pathways during human sperm capacitation. Free Radic. Biol. Med. 2006, 40, 1045–1055. [Google Scholar] [CrossRef]
- Freitas, C.; Neto, A.C.; Matos, L.; Silva, E.; Ribeiro, A.; Silva-Carvalho, J.L.; Almeida, H. Follicular Fluid redox involvement for ovarian follicle growth. J. Ovarian Res. 2017, 10, 44. [Google Scholar] [CrossRef]
- de Matos, D.G.; Furnus, C.C.; Moses, D.F. Glutathione Synthesis During in Vitro Maturation of Bovine Oocytes: Role of Cumulus Cells1. Biol. Reprod. 1997, 57, 1420–1425. [Google Scholar] [CrossRef] [Green Version]
- Agarwal, A.; Gupta, S.; Sekhon, L.; Shah, R. Redox considerations in female reproductive function and assisted reproduction: From molecular mechanisms to health implications. Antioxid. Redox Signal. 2008, 10, 1375–1403. [Google Scholar] [CrossRef] [PubMed]
- Kala, M.; Shaikh, M.V.; Nivsarkar, M. Equilibrium between anti-oxidants and reactive oxygen species: A requisite for oocyte development and maturation. Reprod. Med. Biol. 2017, 16, 28–35. [Google Scholar] [CrossRef]
- Tripathi, A.; Khatun, S.; Pandey, A.N.; Mishra, S.K.; Chaube, R.; Shrivastav, T.G.; Chaube, S.K. Intracellular levels of hydrogen peroxide and nitric oxide in oocytes at various stages of meiotic cell cycle and apoptosis. Free Radic. Res. 2009, 43, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Choi, A.; Yu, H.Y.; Czerniak, S.M.; Holick, E.A.; Paolella, L.J.; Agarwal, A.; Combelles, C.M. Fluctuations in total antioxidant capacity, catalase activity and hydrogen peroxide levels of follicular fluid during bovine folliculogenesis. Reprod. Fertil. Dev. 2011, 23, 673–680. [Google Scholar] [CrossRef] [Green Version]
- Rubio, C.P.; Hernandez-Ruiz, J.; Martinez-Subiela, S.; Tvarijonaviciute, A.; Ceron, J.J. Spectrophotometric assays for total antioxidant capacity (TAC) in dog serum: An update. BMC Vet. Res. 2016, 12, 166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Das, S.; Chattopadhyay, R.; Ghosh, S.; Ghosh, S.; Goswami, S.K.; Chakravarty, B.N.; Chaudhury, K. Reactive oxygen species level in follicular fluid--embryo quality marker in IVF? Hum. Reprod. 2006, 21, 2403–2407. [Google Scholar] [CrossRef] [Green Version]
- Jancar, N.; Kopitar, A.N.; Ihan, A.; Virant Klun, I.; Bokal, E.V. Effect of apoptosis and reactive oxygen species production in human granulosa cells on oocyte fertilization and blastocyst development. J. Assist. Reprod. Genet. 2007, 24, 91–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jana, S.K.; Babu, N.; Chattopadhyay, R.; Chakravarty, B.; Chaudhury, K. Upper control limit of reactive oxygen species in follicular fluid beyond which viable embryo formation is not favorable. Reprod. Toxicol. 2010, 29, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Salimi, M.; Mozdarani, H. Effect of vitamin E on preovulatory stage irradiated female mouse expressed as chromosomal abnormalities in generated embryos. Iran. J. Radiat. Res 2006, 4, 35–40. [Google Scholar]
- Baumber, J.; Ball, B.A.; Linfor, J.J.; Meyers, S.A. Reactive oxygen species and cryopreservation promote DNA fragmentation in equine spermatozoa. J. Androl. 2003, 24, 621–628. [Google Scholar] [CrossRef]
- Lane, M.; McPherson, N.O.; Fullston, T.; Spillane, M.; Sandeman, L.; Kang, W.X.; Zander-Fox, D.L. Oxidative stress in mouse sperm impairs embryo development, fetal growth and alters adiposity and glucose regulation in female offspring. PLoS ONE 2014, 9, e100832. [Google Scholar] [CrossRef] [Green Version]
- Ruihuan, G.; Zhichao, L.; Song, G.; Jing, F.; Yijuan, S.; Xiaoxi, S. Oocyte Vitrification Temporarily Turns on Oxidation-Reduction Process Genes in Mouse Preimplantation Embryos. Reprod. Sci. 2021, 28, 1307–1315. [Google Scholar] [CrossRef]
- Zhang, L.; Xue, X.; Yan, J.; Yan, L.Y.; Jin, X.H.; Zhu, X.H.; He, Z.Z.; Liu, J.; Li, R.; Qiao, J. L-proline: A highly effective cryoprotectant for mouse oocyte vitrification. Sci. Rep. 2016, 6, 26326. [Google Scholar] [CrossRef]
- Wang, F.; Tian, X.; Zhou, Y.; Tan, D.; Zhu, S.; Dai, Y.; Liu, G. Melatonin improves the quality of in vitro produced (IVP) bovine embryos: Implications for blastocyst development, cryotolerance, and modifications of relevant gene expression. PLoS ONE 2014, 9, e93641. [Google Scholar] [CrossRef]
- Gupta, M.K.; Uhm, S.J.; Lee, H.T. Effect of vitrification and beta-mercaptoethanol on reactive oxygen species activity and in vitro development of oocytes vitrified before or after in vitro fertilization. Fertil. Steril. 2010, 93, 2602–2607. [Google Scholar] [CrossRef]
- Kattera, S.; Chen, C. Short coincubation of gametes in in vitro fertilization improves implantation and pregnancy rates: A prospective, randomized, controlled study. Fertil. Steril. 2003, 80, 1017–1021. [Google Scholar] [CrossRef]
- Harris, S.E.; Leese, H.J.; Gosden, R.G.; Picton, H.M. Pyruvate and oxygen consumption throughout the growth and development of murine oocytes. Mol. Reprod. Dev. 2009, 76, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Rieger, D.; Loskutoff, N. Changes in the metabolism of glucose, pyruvate, glutamine and glycine during maturation of cattle oocytes in vitro. Reproduction 1994, 100, 257–262. [Google Scholar] [CrossRef] [Green Version]
- Tiwari, M.; Chaube, S.K. Moderate increase of reactive oxygen species triggers meiotic resumption in rat follicular oocytes. J. Obstet. Gynaecol. Res. 2016, 42, 536–546. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.N.; Chaube, S.K. A moderate increase of hydrogen peroxide level is beneficial for spontaneous resumption of meiosis from diplotene arrest in rat oocytes cultured in vitro. BioResearch Open Access 2014, 3, 183–191. [Google Scholar] [CrossRef]
- Park, Y.S.; You, S.Y.; Cho, S.; Jeon, H.-J.; Lee, S.; Cho, D.-H.; Kim, J.-S.; Oh, J.S. Eccentric localization of catalase to protect chromosomes from oxidative damages during meiotic maturation in mouse oocytes. Histochem. Cell Biol. 2016, 146, 281–288. [Google Scholar] [CrossRef]
- Shkolnik, K.; Tadmor, A.; Ben-Dor, S.; Nevo, N.; Galiani, D.; Dekel, N. Reactive oxygen species are indispensable in ovulation. Proc. Natl. Acad. Sci. USA 2011, 108, 1462–1467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takami, M.; Preston, S.L.; Toyloy, V.A.; Behrman, H.R. Antioxidants reversibly inhibit the spontaneous resumption of meiosis. Am. J. Physiol. 1999, 276, E684–E688. [Google Scholar] [CrossRef] [PubMed]
- Lopes, A.; Lane, M.; Thompson, J. Oxygen consumption and ROS production are increased at the time of fertilization and cell cleavage in bovine zygotes. Hum. Reprod. 2010, 25, 2762–2773. [Google Scholar] [CrossRef] [Green Version]
- Aitken, R.J. Reactive oxygen species as mediators of sperm capacitation and pathological damage. Mol. Reprod. Dev. 2017, 84, 1039–1052. [Google Scholar] [CrossRef] [PubMed]
- O’Flaherty, C. Redox regulation of mammalian sperm capacitation. Asian J. Androl. 2015, 17, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Aitken, R.; Harkiss, D.; Knox, W.; Paterson, M.; Irvine, D. A novel signal transduction cascade in capacitating human spermatozoa characterised by a redox-regulated, cAMP-mediated induction of tyrosine phosphorylation. J. Cell Sci. 1998, 111, 645–656. [Google Scholar] [CrossRef] [PubMed]
- Visconti, P.E.; Bailey, J.L.; Moore, G.D.; Pan, D.; Olds-Clarke, P.; Kopf, G.S. Capacitation of mouse spermatozoa. I. Correlation between the capacitation state and protein tyrosine phosphorylation. Development 1995, 121, 1129–1137. [Google Scholar] [CrossRef]
- Aitken, R.J.; Ryan, A.L.; Baker, M.A.; McLaughlin, E.A. Redox activity associated with the maturation and capacitation of mammalian spermatozoa. Free Radic. Biol. Med. 2004, 36, 994–1010. [Google Scholar] [CrossRef]
- Morado, S.; Cetica, P.; Beconi, M.; Thompson, J.G.; Dalvit, G. Reactive oxygen species production and redox state in parthenogenetic and sperm-mediated bovine oocyte activation. Reproduction 2013, 145, 471–478. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Winuthayanon, W. Oviduct: Roles in fertilization and early embryo development. J. Endocrinol. 2017, 232, R1–R26. [Google Scholar] [CrossRef] [Green Version]
- Saleh, R.A.; Agarwal, A.; Nada, E.A.; El-Tonsy, M.H.; Sharma, R.K.; Meyer, A.; Nelson, D.R.; Thomas, A.J. Negative effects of increased sperm DNA damage in relation to seminal oxidative stress in men with idiopathic and male factor infertility. Fertil. Steril. 2003, 79 (Suppl. 3), 1597–1605. [Google Scholar] [CrossRef]
- Agarwal, A.; Said, T.M. Oxidative stress, DNA damage and apoptosis in male infertility: A clinical approach. BJU Int. 2005, 95, 503–507. [Google Scholar] [CrossRef]
- Sharma, R.; Said, T.; Agarwal, A. Sperm DNA damage and its clinical relevance in assessing reproductive outcome. Asian J. Androl. 2004, 6, 139–148. [Google Scholar]
- McPherson, N.O.; Vincent, A.D.; Pacella-Ince, L.; Tremellen, K. Comparison of in vitro fertilisation/intracytoplasmic sperm injection on live birth rates in couples with non-male factor infertility and advanced maternal age. J. Assist. Reprod. Genet. 2021, 38, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Saleh, R.A.; Bedaiwy, M.A. Role of reactive oxygen species in the pathophysiology of human reproduction. Fertil. Steril. 2003, 79, 829–843. [Google Scholar] [CrossRef] [Green Version]
- Tatone, C.; Carbone, M.; Falone, S.; Aimola, P.; Giardinelli, A.; Caserta, D.; Marci, R.; Pandolfi, A.; Ragnelli, A.; Amicarelli, F. Age-dependent changes in the expression of superoxide dismutases and catalase are associated with ultrastructural modifications in human granulosa cells. Mol. Hum. Reprod. 2006, 12, 655–660. [Google Scholar] [CrossRef] [Green Version]
- de Bruin, J.P.; Dorland, M.; Spek, E.R.; Posthuma, G.; van Haaften, M.; Looman, C.W.N.; te Velde, E.R. Age-Related Changes in the Ultrastructure of the Resting Follicle Pool in Human Ovaries1. Biol. Reprod. 2004, 70, 419–424. [Google Scholar] [CrossRef] [Green Version]
- Te Velde, E.R.; Pearson, P.L. The variability of female reproductive ageing. Hum. Reprod. Update 2002, 8, 141–154. [Google Scholar] [CrossRef]
- Miyamoto, K.; Sato, E.F.; Kasahara, E.; Jikumaru, M.; Hiramoto, K.; Tabata, H.; Katsuragi, M.; Odo, S.; Utsumi, K.; Inoue, M. Effect of oxidative stress during repeated ovulation on the structure and functions of the ovary, oocytes, and their mitochondria. Free Radic. Biol. Med. 2010, 49, 674–681. [Google Scholar] [CrossRef]
- Martin, K.L.; Leese, H.J. Role of developmental factors in the switch from pyruvate to glucose as the major exogenous energy substrate in the preimplantation mouse embryo. Reprod. Fertil. Dev. 1999, 11, 425–433. [Google Scholar] [CrossRef]
- Houghton, F.D.; Thompson, J.G.; Kennedy, C.J.; Leese, H.J. Oxygen consumption and energy metabolism of the early mouse embryo. Mol. Reprod. Dev. Inc. Gamete Res. 1996, 44, 476–485. [Google Scholar] [CrossRef]
- Houghton, F.D.; Leese, H.J. Metabolism and developmental competence of the preimplantation embryo. Eur. J. Obstet. Gynecol. Reprod. Biol. 2004, 115 (Suppl. 1), S92–S96. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.; Jain, S.; Kumar, G.P.; Laloraya, M. A programmed oxyradical burst causes hatching of mouse blastocysts. J. Cell Sci. 1997, 110 Pt 14, 1597–1602. [Google Scholar] [CrossRef]
- Bedaiwy, M.; Agarwal, A.; Said, T.M.; Goldberg, J.M.; Sharma, R.K.; Worley, S.; Falcone, T. Role of total antioxidant capacity in the differential growth of human embryos in vitro. Fertil. Steril. 2006, 86, 304–309. [Google Scholar] [CrossRef]
- Swain, J.E. Controversies in ART: Can the IVF laboratory influence preimplantation embryo aneuploidy? Reprod. Biomed. Online 2019, 39, 599–607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munné, S.; Alikani, M. Culture-induced chromosome abnormalities: The canary in the mine. Reprod. Biomed. Online 2011, 22, 506–508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joo, B.S.; Kim, M.K.; Na, Y.J.; Moon, H.S.; Lee, K.S.; Do Kim, H. The mechanism of action of coculture on embryo development in the mouse model: Direct embryo-to-cell contact and the removal of deleterious components. Fertil. Steril. 2001, 75, 193–199. [Google Scholar] [CrossRef]
- Cheong, A.W.; Lee, Y.L.; Liu, W.M.; Yeung, W.S.; Lee, K.F. Oviductal microsomal epoxide hydrolase (EPHX1) reduces reactive oxygen species (ROS) level and enhances preimplantation mouse embryo development. Biol. Reprod. 2009, 81, 126–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsui, M.; Oshima, M.; Oshima, H.; Takaku, K.; Maruyama, T.; Yodoi, J.; Taketo, M.M. Early embryonic lethality caused by targeted disruption of the mouse thioredoxin gene. Dev. Biol. 1996, 178, 179–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xanthoudakis, S.; Smeyne, R.J.; Wallace, J.D.; Curran, T. The redox/DNA repair protein, Ref-1, is essential for early embryonic development in mice. Proc. Natl. Acad. Sci. USA 1996, 93, 8919–8923. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuscu, N.; Gungor-Ordueri, N.E.; Sozen, B.; Adiguzel, D.; Celik-Ozenci, C. FoxO transcription factors 1 regulate mouse preimplantation embryo development. J. Assist. Reprod. Genet. 2019, 36, 2121–2133. [Google Scholar] [CrossRef] [PubMed]
- Kehrer, J.P. The Haber–Weiss reaction and mechanisms of toxicity. Toxicology 2000, 149, 43–50. [Google Scholar] [CrossRef]
- Noda, Y.; Matsumoto, H.; Umaoka, Y.; Tatsumi, K.; Kishi, J.; Mori, T. Involvement of superoxide radicals in the mouse two-cell block. Mol. Reprod. Dev. 1991, 28, 356–360. [Google Scholar] [CrossRef]
- Legge, M.; Sellens, M.H. Free radical scavengers ameliorate the 2-cell block in mouse embryo culture. Hum. Reprod. 1991, 6, 867–871. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.K.; Lane, M. Alleviation of the’2-cell block’and development to the blastocyst of CF1 mouse embryos: Role of amino acids, EDTA and physical parameters. Hum. Reprod. 1996, 11, 2703–2712. [Google Scholar] [CrossRef] [Green Version]
- Hu, D.B.; Li, Z.S.; Ali, I.; Xu, L.J.; Fang, N.Z. Effect of potential role of p53 on embryo development arrest induced by H2O2 in mouse. In Vitro Cell. Dev. Biol. Anim. 2017, 53, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Cebral, E.; Carrasco, I.; Vantman, D.; Smith, R. Preimplantation embryotoxicity after mouse embryo exposition to reactive oxygen species. Biocell 2007, 31, 51–59. [Google Scholar] [CrossRef]
- Gardiner, C.S.; Reed, D.J. Status of glutathione during oxidant-induced oxidative stress in the preimplantation mouse embryo. Biol. Reprod. 1994, 51, 1307–1314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozawa, M.; Nagai, T.; Fahrudin, M.; Karja, N.W.; Kaneko, H.; Noguchi, J.; Ohnuma, K.; Kikuchi, K. Addition of glutathione or thioredoxin to culture medium reduces intracellular redox status of porcine IVM/IVF embryos, resulting in improved development to the blastocyst stage. Mol. Reprod. Dev. 2006, 73, 998–1007. [Google Scholar] [CrossRef]
- Sun, W.J.; Pang, Y.W.; Liu, Y.; Hao, H.S.; Zhao, X.M.; Qin, T.; Zhu, H.B.; Du, W.H. Exogenous glutathione supplementation in culture medium improves the bovine embryo development after in vitro fertilization. Theriogenology 2015, 84, 716–723. [Google Scholar] [CrossRef]
- Brütsch, S.H.; Wang, C.C.; Li, L.; Stender, H.; Neziroglu, N.; Richter, C.; Kuhn, H.; Borchert, A. Expression of inactive glutathione peroxidase 4 leads to embryonic lethality, and inactivation of the Alox15 gene does not rescue such knock-in mice. Antioxid. Redox Signal. 2015, 22, 281–293. [Google Scholar] [CrossRef]
- Dalton, T.P.; Dieter, M.Z.; Yang, Y.; Shertzer, H.G.; Nebert, D.W. Knockout of the mouse glutamate cysteine ligase catalytic subunit (Gclc) gene: Embryonic lethal when homozygous, and proposed model for moderate glutathione deficiency when heterozygous. Biochem. Biophys. Res. Commun. 2000, 279, 324–329. [Google Scholar] [CrossRef]
- Winkler, A.; Njålsson, R.; Carlsson, K.; Elgadi, A.; Rozell, B.; Abraham, L.; Ercal, N.; Shi, Z.-Z.; Lieberman, M.W.; Larsson, A. Glutathione is essential for early embryogenesis–analysis of a glutathione synthetase knockout mouse. Biochem. Biophys. Res. Commun. 2011, 412, 121–126. [Google Scholar] [CrossRef]
- Zhang, H.M.; Zhang, Y. Melatonin: A well-documented antioxidant with conditional pro-oxidant actions. J. Pineal Res. 2014, 57, 131–146. [Google Scholar] [CrossRef]
- Pang, Y.-w.; Jiang, X.-l.; Zhao, S.-j.; Huang, Z.-q.; Zhu, H.-b. Beneficial role of melatonin in protecting mammalian gametes and embryos from oxidative damage. J. Integr. Agric. 2018, 17, 2320–2335. [Google Scholar] [CrossRef] [Green Version]
- Tamura, H.; Takasaki, A.; Taketani, T.; Tanabe, M.; Kizuka, F.; Lee, L.; Tamura, I.; Maekawa, R.; Aasada, H.; Yamagata, Y.; et al. The role of melatonin as an antioxidant in the follicle. J. Ovarian Res. 2012, 5, 5. [Google Scholar] [CrossRef] [Green Version]
- Tamura, H.; Takasaki, A.; Miwa, I.; Taniguchi, K.; Maekawa, R.; Asada, H.; Taketani, T.; Matsuoka, A.; Yamagata, Y.; Shimamura, K. Oxidative stress impairs oocyte quality and melatonin protects oocytes from free radical damage and improves fertilization rate. J. Pineal Res. 2008, 44, 280–287. [Google Scholar] [CrossRef]
- Gao, C.; Han, H.B.; Tian, X.Z.; Tan, D.X.; Wang, L.; Zhou, G.B.; Zhu, S.E.; Liu, G.S. Melatonin promotes embryonic development and reduces reactive oxygen species in vitrified mouse 2-cell embryos. J. Pineal Res. 2012, 52, 305–311. [Google Scholar] [CrossRef]
- Kitagawa, Y.; Suzuki, K.; Yoneda, A.; Watanabe, T. Effects of oxygen concentration and antioxidants on the in vitro developmental ability, production of reactive oxygen species (ROS), and DNA fragmentation in porcine embryos. Theriogenology 2004, 62, 1186–1197. [Google Scholar] [CrossRef]
- Hossein, M.S.; Hashem, M.A.; Jeong, Y.W.; Lee, M.S.; Kim, S.; Kim, J.H.; Koo, O.J.; Park, S.M.; Lee, E.G.; Park, S.W.; et al. Temporal effects of alpha-tocopherol and L-ascorbic acid on in vitro fertilized porcine embryo development. Anim. Reprod. Sci. 2007, 100, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Falcone, T.; Attaran, M.; Goldberg, J.M.; Agarwal, A.; Sharma, R.K. Vitamin C and vitamin E supplementation reduce oxidative stress–induced embryo toxicity and improve the blastocyst development rate. Fertil. Steril. 2002, 78, 1272–1277. [Google Scholar] [CrossRef]
- Lin, T.; Lee, J.E.; Kang, J.W.; Shin, H.Y.; Lee, J.B.; Jin, D.I. Endoplasmic Reticulum (ER) Stress and Unfolded Protein Response (UPR) in Mammalian Oocyte Maturation and Preimplantation Embryo Development. Int. J. Mol. Sci. 2019, 20, 1–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.Y.; Diao, Y.F.; Kim, H.R.; Jin, D.I. Inhibition of endoplasmic reticulum stress improves mouse embryo development. PLoS ONE 2012, 7, e40433. [Google Scholar] [CrossRef] [Green Version]
- Kang, M.H.; Das, J.; Gurunathan, S.; Park, H.W.; Song, H.; Park, C.; Kim, J.H. The cytotoxic effects of dimethyl sulfoxide in mouse preimplantation embryos: A mechanistic study. Theranostics 2017, 7, 4735–4752. [Google Scholar] [CrossRef]
- Yoon, S.B.; Choi, S.A.; Sim, B.W.; Kim, J.S.; Mun, S.E.; Jeong, P.S.; Yang, H.J.; Lee, Y.; Park, Y.H.; Song, B.S.; et al. Developmental competence of bovine early embryos depends on the coupled response between oxidative and endoplasmic reticulum stress. Biol. Reprod. 2014, 90, 104. [Google Scholar] [CrossRef]
- Mao, T.; Han, C.; Wei, B.; Zhao, L.; Zhang, Q.; Deng, R.; Liu, J.; Luo, Y.; Zhang, Y. Protective effects of quercetin against cadmium chloride-induced oxidative injury in goat sperm and zygotes. Biol. Trace Elem. Res. 2018, 185, 344–355. [Google Scholar] [CrossRef] [PubMed]
- Fakruzzaman, M.; Ghanem, N.; Bang, J.-I.; Ha, A.-N.; Lee, K.-L.; Sohn, S.-H.; Wang, Z.; Lee, D.-S.; Kong, I.-K. Effect of peroxiredoxin II on the quality and mitochondrial activity of pre-implantation bovine embryos. Anim. Reprod. Sci. 2015, 159, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Lv, J.; Zhang, Y.; Jiang, Y.; Chu, C.; Wang, S. Epigallocatechin Gallate Promotes the Development of Mouse 2-Cell Embryos In Vitro by Regulating Mitochondrial Activity and Expression of Genes Related to p53 Signalling Pathway. Basic Clin. Pharmacol. Toxicol. 2014, 115, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Li, C.; Gao, F.; Fan, Y.; Zeng, F.; Meng, L.; Li, L.; Zhang, S.; Wei, H. Epigallocatechin-3-Gallate Promotes the in vitro Maturation and Embryo Development Following IVF of Porcine Oocytes. Drug Des. Dev. Ther. 2021, 15, 1013–1020. [Google Scholar] [CrossRef]
- Gardner, D.; Leese, H. Concentrations of nutrients in mouse oviduct fluid and their effects on embryo development and metabolism in vitro. Reproduction 1990, 88, 361–368. [Google Scholar] [CrossRef] [Green Version]
- Treleaven, T.; Hardy, M.L.M.; Guttman-Jones, M.; Morris, M.B.; Day, M.L. In Vitro Fertilisation of Mouse Oocytes in L-Proline and L-Pipecolic Acid Improves Subsequent Development. Cells 2021, 10, 1–16. [Google Scholar] [CrossRef]
- Morris, M.B.; Ozsoy, S.; Zada, M.; Zada, M.; Zamfirescu, R.C.; Todorova, M.G.; Day, M.L. Selected Amino Acids Promote Mouse Pre-implantation Embryo Development in a Growth Factor-Like Manner. Front. Physiol. 2020, 11, 140. [Google Scholar] [CrossRef]
- Morbeck, D.E.; Krisher, R.L.; Herrick, J.R.; Baumann, N.A.; Matern, D.; Moyer, T. Composition of commercial media used for human embryo culture. Fertil. Steril. 2014, 102, 759–766. [Google Scholar] [CrossRef]
- Krishnan, N.; Dickman, M.B.; Becker, D.F. Proline modulates the intracellular redox environment and protects mammalian cells against oxidative stress. Free Radic. Biol. Med. 2008, 44, 671–681. [Google Scholar] [CrossRef] [Green Version]
- Liang, X.; Zhang, L.; Natarajan, S.K.; Becker, D.F. Proline mechanisms of stress survival. Antioxid. Redox Signal. 2013, 19, 998–1011. [Google Scholar] [CrossRef] [Green Version]
- Holtzapple, P.; Genel, M.; Rea, C.; Segal, S. Metabolism and uptake of l-proline by human kidney cortex. Pediatr. Res. 1973, 7, 818–825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Xue, Z.; Hua, C.; Lin, J.; Shen, Z.; Song, Y.; Ying, H.; Lv, Q.; Wang, M.; Zhou, B. Metabolomic Analysis of the Ameliorative Effect of Enhanced Proline Metabolism on Hypoxia-Induced Injury in Cardiomyocytes. Oxid. Med. Cell. Longev. 2020, 2020, 8866946. [Google Scholar] [CrossRef]
- Liu, N.; Yang, Y.; Si, X.; Jia, H.; Zhang, Y.; Jiang, D.; Dai, Z.; Wu, Z. L-Proline Activates Mammalian Target of Rapamycin Complex 1 and Modulates Redox Environment in Porcine Trophectoderm Cells. Biomolecules 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Adams, E. Metabolism of proline and of hydroxyproline. Int. Rev. Connect. Tissue Res. 1970, 5, 1–91. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.; Zhu, Z.; Bai, W.; Li, R.; Zheng, Y.; Tian, X.e.; Wu, D.; Lu, H.; Wang, Y.; Zeng, W. Proline Protects Boar Sperm against Oxidative Stress through Proline Dehydrogenase-Mediated Metabolism and the Amine Structure of Pyrrolidine. Animals 2020, 10, 1–16. [Google Scholar] [CrossRef]
- Suzuki, C.; Yoshioka, K.; Sakatani, M.; Takahashi, M. Glutamine and hypotaurine improves intracellular oxidative status and in vitro development of porcine preimplantation embryos. Zygote 2007, 15, 317–324. [Google Scholar] [CrossRef]
- Guerin, P.; Menezo, Y. Hypotaurine and taurine in gamete and embryo environments: De novo synthesis via the cysteine sulfinic acid pathway in oviduct cells. Zygote 1995, 3, 333–343. [Google Scholar] [CrossRef]
- Anchordoquy, J.P.; Lizarraga, R.M.; Anchordoquy, J.M.; Nikoloff, N.; Rosa, D.E.; Fabra, M.C.; Peral-Garcia, P.; Furnus, C.C. Effect of cysteine, glutamate and glycine supplementation to in vitro fertilization medium during bovine early embryo development. Reprod. Biol. 2019, 19, 349–355. [Google Scholar] [CrossRef]
- Bahrami, M.; Morris, M.B.; Day, M.L. Amino acid supplementation of a simple inorganic salt solution supports efficient in vitro maturation (IVM) of bovine oocytes. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Das, K.C.; Misra, H.P. Hydroxyl radical scavenging and singlet oxygen quenching properties of polyamines. Mol. Cell. Biochem. 2004, 262, 127–133. [Google Scholar] [CrossRef]
- Lefevre, P.L.; Palin, M.F.; Murphy, B.D. Polyamines on the reproductive landscape. Endocr. Rev. 2011, 32, 694–712. [Google Scholar] [CrossRef] [Green Version]
- Chirino-Galindo, G.; Mejia-Zepeda, R.; Palomar-Morales, M. Change in lipoperoxidation but not in scavenging enzymes activity during polyamine embryoprotection in rat embryo cultured in hyperglycemic media. In Vitro Cell. Dev. Biol. Anim. 2012, 48, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.X.; Lee, S.; Khoirinaya, C.; Oh, A.; Kim, G.A.; Lee, B.C. Supplementation with spermine during in vitro maturation of porcine oocytes improves early embryonic development after parthenogenetic activation and somatic cell nuclear transfer. J. Anim. Sci. 2016, 94, 963–970. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Dai, Z.; Zhang, Y.; Jia, H.; Chen, J.; Sun, S.; Wu, G.; Wu, Z. Maternal l-proline supplementation during gestation alters amino acid and polyamine metabolism in the first generation female offspring of C57BL/6J mice. Amino Acids 2019, 51, 805–811. [Google Scholar] [CrossRef]
- Wu, G.; Bazer, F.W.; Hu, J.; Johnson, G.A.; Spencer, T.E. Polyamine synthesis from proline in the developing porcine placenta. Biol. Reprod. 2005, 72, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Leite, R.F.; Annes, K.; Ispada, J.; de Lima, C.B.; Dos Santos, E.C.; Fontes, P.K.; Nogueira, M.F.G.; Milazzotto, M.P. Oxidative Stress Alters the Profile of Transcription Factors Related to Early Development on In Vitro Produced Embryos. Oxid. Med. Cell. Longev. 2017, 2017, 1502489. [Google Scholar] [CrossRef] [Green Version]
- Amin, A.; Gad, A.; Salilew-Wondim, D.; Prastowo, S.; Held, E.; Hoelker, M.; Rings, F.; Tholen, E.; Neuhoff, C.; Looft, C. Bovine embryo survival under oxidative-stress conditions is associated with activity of the NRF2-mediated oxidative-stress-response pathway. Mol. Reprod. Dev. 2014, 81, 497–513. [Google Scholar] [CrossRef]
- Belli, M.; Zhang, L.; Liu, X.; Donjacour, A.; Ruggeri, E.; Palmerini, M.G.; Nottola, S.A.; Macchiarelli, G.; Rinaudo, P. Oxygen concentration alters mitochondrial structure and function in in vitro fertilized preimplantation mouse embryos. Hum. Reprod. 2019, 34, 601–611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qian, D.; Li, Z.; Zhang, Y.; Huang, Y.; Wu, Q.; Ru, G.; Chen, M.; Wang, B. Response of Mouse Zygotes Treated with Mild Hydrogen Peroxide as a Model to Reveal Novel Mechanisms of Oxidative Stress-Induced Injury in Early Embryos. Oxid. Med. Cell. Longev. 2016, 2016, 1521428. [Google Scholar] [CrossRef] [Green Version]
- Dennery, P.A. Effects of oxidative stress on embryonic development. Birth Defects Res. C Embryo Today 2007, 81, 155–162. [Google Scholar] [CrossRef] [PubMed]
- de Waal, E.; Mak, W.; Calhoun, S.; Stein, P.; Ord, T.; Krapp, C.; Coutifaris, C.; Schultz, R.M.; Bartolomei, M.S. In vitro culture increases the frequency of stochastic epigenetic errors at imprinted genes in placental tissues from mouse concepti produced through assisted reproductive technologies. Biol. Reprod. 2014, 90, 22. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.J.; Kind, K.L.; Pantaleon, M.; Armstrong, D.T.; Thompson, J.G. Oxygen-regulated gene expression in bovine blastocysts. Biol. Reprod. 2004, 71, 1108–1119. [Google Scholar] [CrossRef]
- Rinaudo, P.F.; Giritharan, G.; Talbi, S.; Dobson, A.T.; Schultz, R.M. Effects of oxygen tension on gene expression in preimplantation mouse embryos. Fertil. Steril. 2006, 86, 1252–1265. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.J. The role of oxygen in ruminant preimplantation embryo development and metabolism. Anim. Reprod. Sci. 2007, 98, 113–128. [Google Scholar] [CrossRef]
- Bontekoe, S.; Mantikou, E.; van Wely, M.; Seshadri, S.; Repping, S.; Mastenbroek, S. Low oxygen concentrations for embryo culture in assisted reproductive technologies. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- Christianson, M.S.; Zhao, Y.; Shoham, G.; Granot, I.; Safran, A.; Khafagy, A.; Leong, M.; Shoham, Z. Embryo catheter loading and embryo culture techniques: Results of a worldwide Web-based survey. J. Assist. Reprod. Genet. 2014, 31, 1029–1036. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.Y.; Chen, H.W.; Tzeng, C.R. Low oxygen tension increases mitochondrial membrane potential and enhances expression of antioxidant genes and implantation protein of mouse blastocyst cultured in vitro. J. Ovarian Res. 2017, 10, 47. [Google Scholar] [CrossRef]
- Peng, Z.F.; Shi, S.L.; Jin, H.X.; Yao, G.D.; Wang, E.Y.; Yang, H.Y.; Song, W.Y.; Sun, Y.P. Impact of oxygen concentrations on fertilization, cleavage, implantation, and pregnancy rates of in vitro generated human embryos. Int. J. Clin. Exp. Med. 2015, 8, 6179–6185. [Google Scholar]
- Du Plessis, S.S.; Makker, K.; Desai, N.R.; Agarwal, A. Impact of oxidative stress on IVF. Expert Rev. Obstet. Gynecol. 2008, 3, 539–554. [Google Scholar] [CrossRef]
- Pabon Jr, J.E.; Findley, W.E.; Gibbons, W.E. The toxic effect of short exposures to the atmospheric oxygen concentration on early mouse embryonic development. Fertil. Steril. 1989, 51, 896–900. [Google Scholar] [CrossRef]
- Takenaka, M.; Horiuchi, T.; Yanagimachi, R. Effects of light on development of mammalian zygotes. Proc. Natl. Acad. Sci. USA 2007, 104, 14289–14293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bognar, Z.; Csabai, T.J.; Pallinger, E.; Balassa, T.; Farkas, N.; Schmidt, J.; Gorgey, E.; Berta, G.; Szekeres-Bartho, J.; Bodis, J. The effect of light exposure on the cleavage rate and implantation capacity of preimplantation murine embryos. J. Reprod. Immunol. 2019, 132, 21–28. [Google Scholar] [CrossRef]
- Agarwal, A.; Durairajanayagam, D.; du Plessis, S.S. Utility of antioxidants during assisted reproductive techniques: An evidence based review. Reprod. Biol. Endocrinol. 2014, 12, 112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feuer, S.; Rinaudo, P. From embryos to adults: A DOHaD perspective on in vitro fertilization and other assisted reproductive technologies. Healthcare 2016, 4, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menezo, Y.; Clement, P.; Dale, B.; Elder, K. Modulating oxidative stress and epigenetic homeostasis in preimplantation IVF embryos. Zygote 2021, 1–10. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, X.; Vikash, V.; Ye, Q.; Wu, D.; Liu, Y.; Dong, W. ROS and ROS-Mediated Cellular Signaling. Oxid. Med. Cell. Longev. 2016, 2016, 4350965. [Google Scholar] [CrossRef] [Green Version]
- Delic, V.; Griffin, J.W.D.; Zivkovic, S.; Zhang, Y.; Phan, T.A.; Gong, H.; Chaput, D.; Reynes, C.; Dinh, V.B.; Cruz, J.; et al. Individual Amino Acid Supplementation Can Improve Energy Metabolism and Decrease ROS Production in Neuronal Cells Overexpressing Alpha-Synuclein. Neuromolecular Med. 2017, 19, 322–344. [Google Scholar] [CrossRef]
- Ding, Z.-M.; Jiao, X.-F.; Wu, D.; Zhang, J.-Y.; Chen, F.; Wang, Y.-S.; Huang, C.-J.; Zhang, S.-X.; Li, X.; Huo, L.-J. Bisphenol AF negatively affects oocyte maturation of mouse in vitro through increasing oxidative stress and DNA damage. Chem. Biol. Interact. 2017, 278, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Xin, F.; Susiarjo, M.; Bartolomei, M.S. Multigenerational and transgenerational effects of endocrine disrupting chemicals: A role for altered epigenetic regulation? Semin. Cell Dev. Biol. 2015, 43, 66–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chianese, R.; Pierantoni, R. Mitochondrial Reactive Oxygen Species (ROS) Production Alters Sperm Quality. Antioxidants 2021, 10, 92. [Google Scholar] [CrossRef] [PubMed]
- Drevet, J.R.; Aitken, R.J. Oxidation of Sperm Nucleus in Mammals: A Physiological Necessity to Some Extent with Adverse Impacts on Oocyte and Offspring. Antioxidants 2020, 9, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Edwards, R.G.; Ludwig, M. Are major defects in children conceived in vitro due to innate problems in patients or to induced genetic damage? Reprod. Biomed. Online 2003, 7, 131–138. [Google Scholar] [CrossRef]
- Nishihara, T.; Hashimoto, S.; Ito, K.; Nakaoka, Y.; Matsumoto, K.; Hosoi, Y.; Morimoto, Y. Oral melatonin supplementation improves oocyte and embryo quality in women undergoing in vitro fertilization-embryo transfer. Gynecol. Endocrinol. 2014, 30, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Batioglu, A.S.; Sahin, U.; Gurlek, B.; Ozturk, N.; Unsal, E. The efficacy of melatonin administration on oocyte quality. Gynecol. Endocrinol. 2012, 28, 91–93. [Google Scholar] [CrossRef]
- Espino, J.; Macedo, M.; Lozano, G.; Ortiz, A.; Rodriguez, C.; Rodriguez, A.B.; Bejarano, I. Impact of Melatonin Supplementation in Women with Unexplained Infertility Undergoing Fertility Treatment. Antioxidants 2019, 8, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Pacchiarotti, A.; Carlomagno, G.; Antonini, G.; Pacchiarotti, A. Effect of myo-inositol and melatonin versus myo-inositol, in a randomized controlled trial, for improving in vitro fertilization of patients with polycystic ovarian syndrome. Gynecol. Endocrinol. 2016, 32, 69–73. [Google Scholar] [CrossRef]
- Hasan, A.; Elena, A.-Q.; Ghassan, A.-Q.; Rimawi, T.; Nasrallah, S. Melatonin and Myoinositol: A Foreword Step Toward IVF Success, a Prospective Clinical Trial. J. Gynecol. Obstet. 2019, 7, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Nisenblat, V.; Lu, C.; Li, R.; Qiao, J.; Zhen, X.; Wang, S. Pretreatment with coenzyme Q10 improves ovarian response and embryo quality in low-prognosis young women with decreased ovarian reserve: A randomized controlled trial. Reprod. Biol. Endocrinol. 2018, 16, 29. [Google Scholar] [CrossRef] [PubMed]
- Giannubilo, S.R.; Orlando, P.; Silvestri, S.; Cirilli, I.; Marcheggiani, F.; Ciavattini, A.; Tiano, L. CoQ10 Supplementation in Patients Undergoing IVF-ET: The Relationship with Follicular Fluid Content and Oocyte Maturity. Antioxidants 2018, 7, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Gat, I.; Blanco Mejia, S.; Balakier, H.; Librach, C.L.; Claessens, A.; Ryan, E.A. The use of coenzyme Q10 and DHEA during IUI and IVF cycles in patients with decreased ovarian reserve. Gynecol. Endocrinol. 2016, 32, 534–537. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Luo, S.; Fan, P.; Jin, S.; Zhu, H.; Deng, T.; Quan, Y.; Huang, W. Growth hormone alleviates oxidative stress and improves oocyte quality in Chinese women with polycystic ovary syndrome: A randomized controlled trial. Sci. Rep. 2020, 10, 18769. [Google Scholar] [CrossRef]
- Salehpour, S.; Akbari Sene, A.; Saharkhiz, N.; Sohrabi, M.R.; Moghimian, F. N-acetylcysteine as an adjuvant to clomiphene citrate for successful induction of ovulation in infertile patients with polycystic ovary syndrome. J. Obstet. Gynaecol. Res. 2012, 38, 1182–1186. [Google Scholar] [CrossRef] [PubMed]
- Ozkaya, M.O.; Naziroglu, M. Multivitamin and mineral supplementation modulates oxidative stress and antioxidant vitamin levels in serum and follicular fluid of women undergoing in vitro fertilization. Fertil. Steril. 2010, 94, 2465–2466. [Google Scholar] [CrossRef] [PubMed]
- Kacem, O.; Harzallah, M.; Zedini, C.; Zidi, I.; Meddeb, S.; Fékih, M.; Saidi, H.; Chaib, A.; Boughizane, S.; Ali, H.B.; et al. Beneficial Effect of an Oral Antioxidant Supplementation (Fertimax2) on IVF-ICSI Outcomes: A Preliminary Clinical Study. Adv. Reprod. Sci. 2014, 2, 47–56. [Google Scholar] [CrossRef] [Green Version]
- Tremellen, K.; Miari, G.; Froiland, D.; Thompson, J. A randomised control trial examining the effect of an antioxidant (Menevit) on pregnancy outcome during IVF-ICSI treatment. Aust. N. Z. J. Obstet. Gynaecol. 2007, 47, 216–221. [Google Scholar] [CrossRef]
- Luddi, A.; Capaldo, A.; Focarelli, R.; Gori, M.; Morgante, G.; Piomboni, P.; De Leo, V. Antioxidants reduce oxidative stress in follicular fluid of aged women undergoing IVF. Reprod. Biol. Endocrinol. 2016, 14, 57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steiner, A.Z.; Hansen, K.R.; Barnhart, K.T.; Cedars, M.I.; Legro, R.S.; Diamond, M.P.; Krawetz, S.A.; Usadi, R.; Baker, V.L.; Coward, R.M.; et al. The effect of antioxidants on male factor infertility: The Males, Antioxidants, and Infertility (MOXI) randomized clinical trial. Fertil. Steril. 2020, 113, 552–560. [Google Scholar] [CrossRef]
- El Nashar, A.; Salama, K.; El-Deen, A.; Fayez, A. Effects of Pentoxifylline and Vitamin E on Pregnancy Rate in Infertile Women Treated By ICSI: A Randomized Clinical Trial. Benha J. Appl. Sci. 2020, 5, 1–6. [Google Scholar] [CrossRef]
- Tremellen, K.; Woodman, R.; Hill, A.; Shehadeh, H.; Lane, M.; Zander-Fox, D. Use of a male antioxidant nutraceutical is associated with superior live birth rates during IVF treatment. Asian J. Androl. 2021, 23, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.; Santamaria-Lopez, E.; Blasco, V.; Hernaez, M.J.; Caligara, C.; Pellicer, A.; Fernandez-Sanchez, M.; Prados, N. Effect of Group Embryo Culture under Low-Oxygen Tension in Benchtop Incubators on Human Embryo Culture: Prospective, Randomized, Controlled Trial. Reprod. Sci. 2020, 27, 1522–1533. [Google Scholar] [CrossRef] [PubMed]
- Van Montfoort, A.P.A.; Arts, E.; Wijnandts, L.; Sluijmer, A.; Pelinck, M.J.; Land, J.A.; Van Echten-Arends, J. Reduced oxygen concentration during human IVF culture improves embryo utilization and cumulative pregnancy rates per cycle. Hum. Reprod. Open 2020, 2020, hoz036. [Google Scholar] [CrossRef]
- Gelo, N.; Kirinec, G.; Baldani, D.P.; Vrcic, H.; Jezek, D.; Milosevic, M.; Stanic, P. Influence of human embryo cultivation in a classic CO2 incubator with 20% oxygen versus benchtop incubator with 5% oxygen on live births: The randomized prospective trial. Zygote 2019, 27, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Aleyasin, A.; Aghahosseini, M.; Mohseni, M.; Mahdavi, A. Effects of pentoxifylline and vitamin E on pregnancy rate in infertile women treated by ZIFT: A randomized clinical trial. Int. J. Reprod. BioMedicine 2009, 7, 170–175. [Google Scholar]
- Gardner, D.K.; Kuramoto, T.; Tanaka, M.; Mitzumoto, S.; Montag, M.; Yoshida, A. Prospective randomized multicentre comparison on sibling oocytes comparing G-Series media system with antioxidants versus standard G-Series media system. Reprod. Biomed. Online 2020, 40, 637–644. [Google Scholar] [CrossRef]
- Kim, M.K.; Park, J.K.; Paek, S.K.; Kim, J.W.; Kwak, I.P.; Lee, H.J.; Lyu, S.W.; Lee, W.S. Effects and pregnancy outcomes of L-carnitine supplementation in culture media for human embryo development from in vitro fertilization. J. Obstet. Gynaecol. Res. 2018, 44, 2059–2066. [Google Scholar] [CrossRef]
- Ma, L.; Cai, L.; Hu, M.; Wang, J.; Xie, J.; Xing, Y.; Shen, J.; Cui, Y.; Liu, X.J.; Liu, J. Coenzyme Q10 supplementation of human oocyte in vitro maturation reduces postmeiotic aneuploidies. Fertil. Steril. 2020, 114, 331–337. [Google Scholar] [CrossRef]
- Fabozzi, G.; Albricci, L.; Cimadomo, D.; Amendola, M.G.; Sanges, F.; Maggiulli, R.; Ubaldi, F.M.; Rienzi, L. Blastulation rates of sibling oocytes in two IVF culture media: An evidence-based workflow to implement newly commercialized products. Reprod. Biomed. Online 2021, 42, 311–322. [Google Scholar] [CrossRef]
- Taherian, S.S.; Khayamabed, R.; Tavalaee, M.; Nasr-Esfahani, M.H. Alpha-lipoic acid minimises reactive oxygen species-induced damages during sperm processing. Andrologia 2019, 51, e13314. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, F.; Cao, H.; Li, C.; Du, C.; Yao, L.; Mao, H.; Lin, W. Protective effects of l-carnitine on astheno- and normozoospermic human semen samples during cryopreservation. Zygote 2016, 24, 293–300. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hardy, M.L.M.; Day, M.L.; Morris, M.B. Redox Regulation and Oxidative Stress in Mammalian Oocytes and Embryos Developed In Vivo and In Vitro. Int. J. Environ. Res. Public Health 2021, 18, 11374. https://doi.org/10.3390/ijerph182111374
Hardy MLM, Day ML, Morris MB. Redox Regulation and Oxidative Stress in Mammalian Oocytes and Embryos Developed In Vivo and In Vitro. International Journal of Environmental Research and Public Health. 2021; 18(21):11374. https://doi.org/10.3390/ijerph182111374
Chicago/Turabian StyleHardy, Madeleine L. M., Margot L. Day, and Michael B. Morris. 2021. "Redox Regulation and Oxidative Stress in Mammalian Oocytes and Embryos Developed In Vivo and In Vitro" International Journal of Environmental Research and Public Health 18, no. 21: 11374. https://doi.org/10.3390/ijerph182111374







