Abstract
Different forms of physical activity—endurance, resistance or dynamic power—stimulate cytokine release from various tissues to the bloodstream. Receptors for exercise-induced cytokines are present in muscle tissue, adipose tissue, liver, brain, bones, cardiovascular system, immune system, pancreas, and skin. They have autocrine, paracrine and endocrine activities. Many of them regulate the myocyte growth and differentiation necessary for muscle hypertrophy and myogenesis. They also modify energy homeostasis, lipid, carbohydrate, and protein metabolism, regulate inflammation and exchange information (crosstalk) between remote organs. So far, interleukin 6 and irisin have been the best studied exercise-induced cytokines. However, many more can be grouped into myokines, hepatokines and adipomyokines. This review focuses on the less known exercise-induced cytokines such as myostatin, follistatin, decorin, brain-derived neurotrophic factor, fibroblast growth factor 21 and interleukin 15, and their relation to various forms of exercise, i.e., acute vs. chronic, regular training in healthy people.
1. Introduction
In 2003, Pedersen et al. introduced the term “myokines’’ to describe cytokines produced and released by skeletal muscles in response to contraction [1]. Later, Seldin et al. highlighted that some of the myokines are secreted in response to exercise even in higher amounts by non-muscle tissues than by the muscles [2]. Still, the authors of the term “myokines” state that potential cytokines, to be classified as myokines, must be derived from skeletal muscle, must be secreted and must carry out a biological function in an endocrine, paracrine or autocrine mode [3,4]. Due to the main site of origin of particular cytokines and the proposed myokine classification criteria, the nomenclature among literature can be confusing. It is further complicated by the emergence of new names for groups of cytokines, such as hepatokines (e.g., follistatin, fibroblast growth factor 21) or adipomyokines (e.g., interleukin 15, irisin). So far, myokines have been considered as cytokines secreted in response to exercise. However, whether only cytokines produced by muscle cells can be called myokines is widely discussed and unresolved. Irrespective of the classification, the common feature of such cytokines is a change in their concentrations in response to exercise. For the need of this review, we assume that cytokines secreted in response to exercise (including myokines, hepatokines or adipomyokines) can also generally be referred to as myokines, however they are most commonly classified due to their main origin and can be also called “exercise-induced cytokines’’ family.
The first cytokine classified as a myokine was interleukin 6 (IL-6) [1,5]. It is expressed acutely during exercise, promotes glycogen breakdown, increases glucose transporter type 4 (GLUT4) expression and glucose uptake in myocytes, enhancing insulin sensitivity [6,7]. In adipocytes, IL-6 induces lipolysis and free fatty acid oxidation [8,9]. IL-6 also facilitates muscle growth, regulating the function of satellite cells [10,11]. The first evidence of IL-6 response to exercise was published by Northoff et al. in 1991, who reported an increase in IL-6 concentration after a marathon run [12]. Further studies demonstrated that the IL-6 increase after exercise is attributable predominantly to skeletal muscle production [13,14,15,16,17,18,19]. We refer readers interested in IL-6 to the literature reviews by Pedersen et al. and Febbraio and Pedersen [20,21].
Irisin is another well studied exercise-induced cytokine, although classified mainly as a myokine or adipomyokine [22,23,24,25,26]. The muscles and adipose tissue are considered the principal source of irisin [27,28,29,30]. It has been hypothesized that irisin secreted from muscles after exercise causes so-called “browning” of the white adipose tissue. The resulting conversion of white into the beige adipose tissue, with similar thermogenic features to brown adipose tissue, raises energy expenditure [22]. Up to date, irisin has been summarized in two meta-analyses—one (Fox et al.) considering an acute bout of exercise and another (Qiu et al.) regular exercise training [23,24]. Immediately following an acute bout of exercise, irisin concentration increases substantially in adults. [23]. In contrast, randomized control trials have shown that regular training reduces the circulating irisin concentration. Evidence from non-randomized studies is inconclusive [24].
Myokines, which are perhaps the most recognizable subgroup of exercise-induced cytokines, have been recently reviewed in several aspects. Lee and Jun et al. focused on myokine activity’s molecular basis and their general functions in humans [31]. Eckel summarized the muscle-adipose, muscle-liver, muscle-bone, muscle-beta cell crosstalks, and myokines’ involvement in the anti-inflammatory effects of physical activity counteracting insulin resistance and metabolic abnormalities caused by obesity and type 2 diabetes [32]. Fiuza-Luces et al. presented an impact of physical activity on the cardiovascular system, including myokines as possible mediators of beneficial physical exercise effects in people [33]. Chen et al. focused on tissue crosstalk mechanisms, highlighting that myokines mediate the effects of muscle work on whole-body metabolism [34].
New exercise-induced cytokines and exercise-related activity of already known proteins are being discovered continuously. We selected the cytokines with evidence-based relation to exercise. Myostatin (MSTN) is considered to be the most potent negative regulator of muscle mass. Conversely, follistatin (FST) and decorin (DCN) are direct antagonists of MSTN. Although the brain-derived neurotrophic factor (BDNF) concentration is strongly associated with cognitive function, it is also linked with regular physical activity. Fibroblast growth factor 21 (FGF21) and FST are produced mainly by the liver and both are considered hepatokines, nonetheless, their concentration alters in response to exercise classifying them as exercise-induced cytokines [35]. For this reason, we have included FGF21 and FST in our review as well. Finally, Interleukin 15 (IL-15) is a cytokine from the same class as myokines protoplast—IL-6; however, it is less known and deserves attention. A more detailed list of different functions and locations of expression of MSTN, FST, DCN, BDNF, FGF21, and IL-15 is given in Table 1. Figure 1 illustrates the potential relations of exercise-induced cytokines to physical activity and accompanying phenomena.

Table 1.
Potential functions of selected cytokines and location of expression.
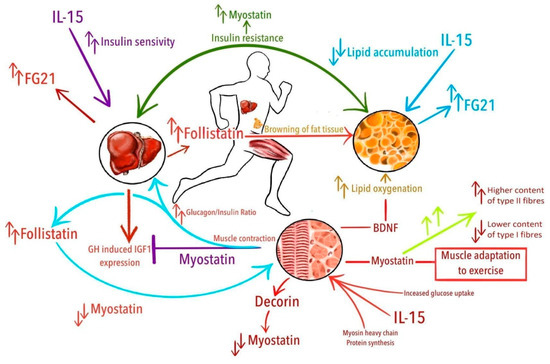
Figure 1.
Potential relations of exercise-induced cytokines with physical activity and accompanying phenomena. From the top in clockwise manner: Overall insulin resistance is related with increased levels of myostatin; IL-15 decreases lipid accumulation; muscle-contraction induced increase of brain-derived neurotrophic factor (BDNF) drives lipid oxygenation; increased levels of myostatin are related with higher content of type II fibers and lower content of type I fibers and probably are associated with muscle adaptation to exercise; interleukin 15 (IL-15) stimulates synthesis of myosin heavy chain proteins and increase glucose uptake in the muscle; decorin binds with myostatin and downregulates its activity; in the liver, myostatin suppresses growth hormone (GH)—induced insulin-like growth factor 1 (IGF-1) expression; increased glucagon/insulin ratio after exercise leads to increased production of follistatin in the liver, which suppress myostatin activity; fibroblast growth factor 21 (FGF21) is produced mainly by the liver; IL-15 improves insulin-sensitivity; follistatin promotes browning of white adipose tissue.
In this review, we summarize the physiological role of MSTN, FST, DCN, BDNF, FGF21, and IL-15, emphasizing the effect of various exercise and training protocols on their serum/plasma concentrations in healthy people.
2. Materials and Methods
Using the search engines PubMed and Google Scholar, we searched for the following phrases: “myokines and exercise”, “myokines and physical activity”, “myostatin and exercise”, “follistatin and exercise”, “decorin and exercise”, “brain-derived neurotrophic factor and exercise”, “interleukin 15 and exercise”, “fibroblast growth factor 21 and exercise”, “myostatin and physical activity”, “follistatin and physical activity”, “decorin and physical activity”, “brain-derived neurotrophic and physical activity”, “interleukin 15 and physical activity”, “fibroblast growth factor 21 and physical activity”.
The final selection was non-systematic. We reviewed papers that have been published only in peer-reviewed English-language journals over the last decade. Studies performed in healthy people were our primary focus. As the data presentation differed between the selected studies (absolute values, % of change, area under the curve, charts), the tables’ data differ in terms of units. If possible, we included the results in absolute values.
3. Results and Discussion
3.1. Myostatin
Over 20 years ago, McPherron et al. discovered myostatin (MSTN), earlier referred to as growth differentiation factor 8 (GDF-8) [66]. MSTN belongs to the transforming growth factor β (TGF-b) superfamily that controls growth and differentiation. In humans, the TGF-b superfamily comprises over 30 members that act through cell membrane receptors and further induce and integrate pathways regulating transcription in the nuclei [67]. Elliot et al. have reported that MSTN circulates in the bloodstream either in a bound (to its own propeptide or inhibiting molecules like FST) or an unbound (free) form. These forms determine biological activity of MSTN (unbound = active; bound = inactive) [68].
Schuelke et al. have identified a loss-of-function mutation in the MSTN in a child with muscle hypertrophy and shown for the first time that MSTN is relevant to muscle mass regulation in humans [69]. Lee et al. (2016) and Lee et al. (2010) described both paracrine and endocrine MSTN influence on muscle mass. The subtle endocrine effect of MSTN modulated whole-body metabolism [40,70]. In response to exercise, MSTN binds to the cell membrane receptors initiating the microtubule-associated protein kinase (MAPK) cascade. By causing the nuclear accumulation of SMAD (main signal transducers for receptors of the TGF-B superfamily) proteins, it activates genes involved in skeletal muscle adaptation to exercise [71,72]. Covington et al. found an association between high MSTN concentration, lower content of type I slow-twitch muscle fibers and higher content of type II fast-twitch fibers in people [73]. MSTN is involved in muscle atrophy, satellite cell activation, proliferation and differentiation, cell survival, and protein synthesis [37]. Hittel et al. have observed a positive association between MSTN concentration and insulin resistance [74]. However, MSTN’s influence on carbohydrate metabolism remains unclear and requires further investigation. Studies examining the effects of exercise on MSTN are inconclusive—Table 2.

Table 2.
Effects of exercise on myostatin.
MSTN serum/plasma concentration increases after acute exercise [75,78,79,80]. A decrease in serum MSTN was reported in one study, but this could be the effect of late blood sampling (24 h after the exercise), i.e., much later than in other studies. [77]. No exercise-induced change in MSTN was reported in a study with apparently insufficient muscle stimulus during an exercise, as participants trained only with their body mass. [76].
MSTN is also expressed in non-muscle cells and tissues. Kerschan et al. examined a group of highly trained long-distance runners after completing an ultramarathon [78]. They proposed that increased serum MSTN be a consequence of general inflammation accompanied by a release of other cytokines that might be responsible for the stimulation of MSTN synthesis. Another hypothesis would be the release of MSTN by injured muscles. Nevertheless, we have not found any reports supporting an association between MSTN concentration and muscle injury markers (e.g., myoglobin, creatine kinase, lactate dehydrogenase). MSTN concentration increases immediately after exercise and returns to baseline within 24 h of recovery.
Regular exercise effects were investigated in a few studies, ranging from five weeks to six months [68,76,81,82]. Studies reported a decrease [76,81], an increase [68] or no change [82] in MSTN basal concentrations. Contradictory results may be due to the differences in study groups, including age and sex, or the type, duration, and intensity of analyzed training.
3.2. Follistatin
Follistatin (FST), discovered by Robertson et al. in 1987, is a glycoprotein belonging to the TGF-b superfamily [83]. FST can suppress the follicle-stimulating hormone (FSH). In humans, FST is secreted by several organs and tissues, including gonads, skeletal muscles, liver, and adipose tissue. It circulates in three isoforms: Fst288, Fst303 and Fst301 [84,85]. According to Hansen et al., FST is mainly attributable to liver secretion and regulated by the glucagon-to-insulin ratio in response to exercise [43]. Even though FST liver secretion is induced by muscle contraction and, therefore, is considered a hepatokine, some authors also list it as a myokine [80,86]. Regardless of the label, FST belongs to the exercise-induced cytokines family.
FST is an antagonist of MSTN and other TGF-b superfamily members and prevents these proteins from binding to the activin IIb receptor [47,87]. FST regulates muscle cell metabolism, differentiation, and growth indirectly through inhibition of MSTN effects or, as reported by Winbanks et al., directly by mechanisms independent from MSTN [44,45]. In humans under increased energy demand, for example, after acute exercise or prolonged fasting, FST is probably stimulated by an increased glucagon-to-insulin ratio [86,88]. Studies examining the effect of physical exercise on FST are consistent, demonstrating its increase after exercise—Table 3.

Table 3.
Effects of exercise on follistatin.
The studies investigating relations between exercise and FST focus primarily on acute rather than long-term effects. All types of exercise, including resistance [79,81,82,89,90], endurance [43,78,80,89,91,92] and HIIT [68,79,89] raised plasma/serum FST after an acute bout, despite the variety of used protocols. An increase in FST concentration ranges from around 5% to 500%. The strongest response has been found in younger subjects performing the exercise with higher intensity. These features may be the factors limiting an increase in FST serum/plasma concentration in other studies. FST concentration peaks at 3–4 h post-exercise, and then it decreases; however, in some studies, an elevated concentration was observed for 72 h.
Effects of regular exercise were investigated in studies based on training lasting from 2 weeks to 6 months. In the studies considering serum/plasma FST, regular physical activity increases its basal concentration, regardless of the training duration and the type of performed activity. However, it must be highlighted that the participants in these studies were predominantly middle-aged and elderly.
3.3. Decorin
DCN is a small, leucine-rich proteoglycan encoded by the DCN gene involved in the collagen-fibril assembly. It binds to multiple cell surface receptors, modulates tumor suppression, stimulates autophagy and inflammation, and inhibits angiogenesis and tumorigenesis [50]. DCN is secreted by skeletal muscle cells. As a myokine, it promotes skeletal muscle hypertrophy by binding with MSTN and regulates muscle growth upon physical exercise [93]. A summary of studies on DCN is presented in Table 4.

Table 4.
Effects of exercise on decorin.
Few studies have explored the potential role of DCN in physical exercise. Studies presented in Table 4 were performed in small groups of subjects (<15 individuals), except for the investigation by Micielska et al. [76]. Kanzleiter et al. reported that plasma DCN concentration increased in humans performing acute resistance exercises. They also observed an enhanced secretion of DCN from in vitro contracting human myotubes in response to chronic endurance training. [93]. Knuiman et al. studied 13 young and healthy adults who underwent endurance and resistance exercise sessions [94]. DCN concentration did not differ significantly between the baseline and after the training. However, although not significant, an increasing trend in the plasma DCN concentration was found at 1, 2, and 3 h after exercise, compared with the baseline values. Knuiman et al. drew blood from subjects one hour after exercise, whereas the peak release of protein is right after the exercise [93]. Bugera et al. found that plasma concentration of DCN immediately post-exercise was higher than immediately pre-, 1 h post-exercise, and 24 h post-exercise. [95] On the other hand, Micielska et al. reported that high-intensity circuit training for five weeks did not change the resting plasma concentration of DCN in women [76]. This discrepancy might be explained by differences in applied exercise protocol and timing for drawing blood for DCN measurement [76].
In conclusion, the effects of exercise on DCN are not well explored, and their understanding requires future research on larger groups of people. It is, however, known that DCN binds extracellularly to MSTN. Consequently, MSTN’s regulatory effects are inhibited, leading to muscle fiber hypertrophy [93,96].
3.4. BDNF
Brain-derived neurotrophic factor (BDNF) is a protein encoded by the BDNF gene. BDNF, first isolated in 1898 by Yves-Alain Barde and Hans Thoenen [97], is part of a larger family of neurotrophins primarily responsible for developing neurons [51]. BDNF is essential in the autocrine mediation of adult dorsal root ganglion neuron survival [98]. This neurotrophin is involved in numerous neurophysiological processes, including the control of neuronal and glial cell development and the activity-dependent regulation of the synaptic structure and its maintenance, i.e., processes critical for memory and cognition [52].
The origin of BDNF in human serum is still not fully understood. Recent studies have found that megakaryocytes are the primary source of BDNF in platelets and serum [99], suggesting that serum BDNF concentration does not reflect its brain level. The variations in BDNF concentration during physical exercise may reflect different degrees of platelet activation [100]. On the other hand, Rasmussen et al. reported that the brain contributed 70–80% of circulating BDNF both at rest and during exercise [101]. Non-neurogenic tissues, including skeletal muscle, also release this protein. Exercise increases BDNF in the brain, plasma, and skeletal muscles. However, muscle-derived BDNF appears not to be released into the circulation [53]. BDNF is a contraction-inducible protein enhancing lipid oxidation in skeletal muscles via AMP-activated kinase activation [53]. The relation of BDNF to exercise is shown in Table 5.

Table 5.
Effects of exercise on BDNF.
Most of the analyzed studies report an increase in serum BDNF after bouts of exercise, including aerobic, anaerobic, or more prolonged efforts. In some studies, however, BDNF concentration remained unchanged or decreased after exercise. Studies analyzed BDNF concentration changes after an acute bout of exercise and a more extended training protocol, whereas some only one of these factors. Most studies measured serum BDNF concentration, and one study measured plasma BDNF concentration. [108]. Serum BDNF measurement is favoured over the EDTA-plasma for its higher reliability [114].
Szuhany et al. performed a meta-analysis [102]. Fourteen studies examined BDNF concentration changes after a single session of exercise, eight studies immediately post-exercise in a design evaluating the effects of a regular training program, and 13 studies at rest but during a longer regular training. Every bout of exercise causes an increase in BDNF concentration, and the magnitude of this effect can be enhanced by prolonged training. Resting BDNF has been reported to rise after an exercise program. Szuhany et al. found that BDNF after exercise increased to a lesser extent in women than in men. Hakansson et al. found that BDNF increased after exercise in older adults aged 65–85 years [103]. Tsai et al. highlighted that serum BDNF secretion after a bout of exercise depended on the exercise intensity, as more forceful effort caused a higher BDNF increase [107]. On the other hand, Church et al. reported that BDNF increased after an acute bout of resistance exercise in a group of experienced weight lifters regardless of the training protocol (high-intensity, low-volume training, or high-volume moderate-intensity training). No significant differences in BDNF concentration were found between the training protocols [108]. Moreover, BDNF concentration increased after a 7-week training [108]. Roh et al. [113] observed that serum BDNF increased after a 16-week taekwondo training program.
Murawska et al. reported that resting BDNF increased after the 12-week CrossFit program in both men and women. However, before the 12-week CrossFit program, BDNF increased directly after the Wingate and progressive tests, while after the training was completed, BDNF decreased right after the Wingate and progressive tests [106]. Different physiological adaptation levels to exercise before and after the training may play a vital part in BDNF concentration changes after a single exercise.
Marquez et al. reported that BDNF concentration increased similarly during high-intensity interval training and a continuous exercise protocol. However, BDNF increase during exercise was higher in the high-intensity protocol. In this study, BDNF concentration was measured eight times during exercise, giving a unique insight into this protein’s exercise-related kinetics. BDNF gradually increased during the 20 min exercise, with maximal concentrations found at the end of the activity. It quickly decreased to basal values after 20 min of rest [109]. Fortunato et al. [105] confirmed that BDNF peak after exercise is relatively short, and BDNF returned to normal values within 2 h of recovery.
Antunes et al. [111] observed that exercise raised BDNF independently of the intensity protocol; however, the largest BDNF increase was noted after the high-intensity training of >90% VO2max. Additionally, they reported that BDNF increase was higher in subjects with lower than higher physical fitness levels during the short-time high-intensity exercise. This finding suggests that post-exercise growth in BDNF depends on the physical fitness level. This finding can be a possible explanation for some of the previous results of some reduction in BDNF after exercise (Wagner et al. [110], Murawska et al. [106]). In contrast to most reports showing a post-exercise/training increase in BDNF, Wagner et al. found a significant exercise-induced decline in BDNF both before and after a 6-week aerobic physical training [110]. No response of baseline BDNF to longer physical exercise protocols has been noted by Antunes et al. [111] and Wagner et al. [110].
Most of the studies were performed in small groups of less than 20 participants [103,105,106,109,113], usually with no or just a few women [105,107,108,109,110,111,112,113]. Various types of exercise with BDNF have been analyzed so far. Subjects exercised on a cycle ergometer [104,109,111], treadmill [112], underwent strength exercises [105], high intensity interval training [106,108,109] or other types of exercises like general workout or taekwondo [102,103,107,110,113]. The exercise duration was also diverse ranging from 20 to 35 min for acute exercise to 6 to 16 weeks for the regular training programs. Different exercise loads during endurance exercise were applied from 40% to 95% VO2max. These differences make it difficult to compare the observed effects of the studies. On the other hand, different exercise types and their duration evoked similar effects—an increase in BDNF concentration after exercise, with few exceptions in rare situations [106,110,111].
In conclusion, BDNF rises after physical exercise in healthy adults after acute bouts of exercise and after regular exercise [103,104,105,106,107,108,109,112,113]. A more significant increase in BDNF concentration is demonstrated after high-intensity training [104,107,109] and more extended training lasting at least 12 weeks [106,112,113]. However, a single bout of strength exercise also increases BDNF [103,104,105,106,107,108,111]. Fitness level influences BDNF concentration changes, but results are inconsistent. Most recently, authors report that subjects with lower physical fitness levels had a more significant increment in BDNF than higher fitness level subjects [106,110,111].
3.5. Fibroblast Growth Factor 21 (FGF21)
Fibroblast growth factors (FGFs) comprise a group of cell-signaling proteins with mitogenic activity. They facilitate cell differentiation, migration, and survival [54]. Fibroblast growth factor 21 (FGF21) belongs to the subfamily of so-called endocrine FGFs secreted into the bloodstream and expressing metabolic and endocrine functions [115]. The liver secretes most of FGF21 in response to exercise. However, FGF21 is also expressed in adipose tissue, skeletal muscle, and pancreas [116,117,118]. Similar to FST, FGF21 is an exercise-induced cytokine which primarily originates from the liver. Therefore, FGF21 is considered as a hepatokine, though due to its exercise-dependent character and activity, it can also be referred to as either adipokine or myokine [119]. It acts through the FGFR receptor in the presence of the cofactor beta-klotho—its tissue expression determines the target organs for the FGF21 [120]. FGF21 modulates metabolic adaptation to stress [117,121,122,123], decreases serum glucose and lipid concentrations, and improves insulin sensitivity [124,125]. Data on the relation between FGF21 and exercise are summarized in Table 6.

Table 6.
Effect of exercise on FGF21.
In general, serum FGF21 concentration increases in response to acute bouts of endurance exercise [79,91,92,126,127,128,129]. However, Cuevas-Ramos et al. and Taniguchi et al. observed no significant changes in serum FGF21 after a single session of endurance exercise [130,131]. These discrepancies may result from lower training intensity and shorter duration compared to other studies. According to Kim et al. and Wills et al., FGF21 rises proportionally to the training intensity [92,126]. In the study by Cuevas-Ramos et al., the average training duration was approximately 14 min, whereas, in Taniguchi et al., the exercise lasted 30 min at 70% VO2max [130,131]. Other factors potentially influencing the obtained results are the participants’ age and gender. Most studies in which IGF21 increased were carried out in young men. By contrast, Cuevas-Ramos et al. investigated women and Taniguchi et al. older men.
Moreover, FGF21 serum concentration rises gradually after acute endurance exercise with no or slight alterations observed immediately after exercise and peak values measured one-hour post-exercise [79,91,92,126,127,128,129]. Few studies assessed further time points, though FGF21 concentration tends to normalize quickly in the resting period [128], reaching even lower than baseline values 24 h post-exercise [131]. In a recent meta-analysis, circulating FGF21 increased immediately after exercise, reached maximal concentration at one hour of recovery and returned to baseline within three hours of recovery [132].
Janssen Duijghuijsen et al. studied well-trained individuals who cycled on two occasions separated by one week [129]. The first exercise increased serum FGF21, whereas the retest performed after a week did not affect FGF21 concentration. The authors suggested this may reflect an adaptation to exercise-induced cellular stress that stimulated FGF21 during the first trial.
The data on the influence of regular endurance training on serum FGF21 are scarce. Cuevas-Ramos et al. observed a significant increase in FGF21 concentration after two weeks of regular physical activity. However, their study lacks information on the exact time point of blood sampling after a complete intervention [130]. It is unclear whether blood samples were taken before or after the last training session, and consequently if acute exercise might have influenced the results. By contrast, Taniguchi et al. reported a significant decrease in serum FGF21 in older men after five weeks of regular cycling [133]. The authors hypothesize that prolonged exercise reduced the so-called FGF21 resistance, similarly to insulin resistance, and consequently decreased FGF21 serum concentration. The presented studies on the effects of regular physical activity on FGF21 differed significantly in the training duration, intensity, and participants’ age and sex, which might influence the obtained results.
Three of the reviewed studies focused on resistance training [79,128,134]. Results indicate that serum FGF21 remains unchanged shortly after acute resistance exercise, regardless of its intensity and involved muscle mass [128,134]. However, it may increase in the late phase of recovery [79].
3.6. Interleukin 15
Interleukin 15 (IL-15) was discovered in 1994 as a T-cell growth factor [58,135]. Its expression was identified in various cells and tissues, including cells of monocyte/macrophage lineage and other antigen-presenting cells, skeletal and heart muscle, placenta, kidney, lung, bone marrow, and thymus [59,135]. IL-15 regulates cell proliferation, maturation, and the extent of the cellular response in the immune system. It also exerts pleiotropic functions in lipid and glucose metabolism [62,63]. IL-15 administration prevents fatty liver development induced by a high-fat-fed diet and reduces serum glucose and insulin concentrations leading to improved insulin sensitivity [64,65]. In myocytes, IL-15 enhances basal and insulin-induced glucose uptake and GLUT4 translocation to the cell membrane [136,137]. IL-15 is engaged in transducing the beneficial effects of exercise on health. IL-15 promotes muscle anabolism through stimulation of myosin heavy-chain protein synthesis in mature muscle fibers [61]. Although earlier studies found that exercise interventions did not elicit IL-15 serum concentration changes, [138,139,140] recently demonstrated that an acute bout of resistance exercise and regular endurance training affect IL-15 [141,142,143]. Summary of studies on IL-15 and exercise or training are presented in Table 7.

Table 7.
Effects of exercise on IL-15.
Acute resistance exercise stimulates IL-15 production [94,144,145]. Its increase occurred within the first hour of recovery and was not affected by pre-exercise availability of carbohydrates or fat. However, Perez-Lopez et al. noted that an increase in serum IL-15 was not associated with an increased muscle protein synthesis, questioning the role of this myokine in exercise-induced muscle hypertrophy [145].
In contrast, Bugera et al. and Fortunato et al. reported no influence of a single session of resistance exercise of legs on serum IL-15 [95,105]. They speculated that exercise limited to lower extremities might be insufficient to induce myokine secretion. However, other studies demonstrated that IL-15 raised in response to similar resistance exercise [94,145]. He et al. investigated whole-body resistance training and reported no changes in IL-15 concentration. Only one study focused on the long-term effects of regular resistance training and demonstrated an increase in IL-15 concentration after completing a 12-week whole-body resistance training protocol [146].
The mentioned studies differed in subjects’ fitness level, which possibly affected the IL-15 response to exercise. Bazghir et al. and Kapilevich et al. acknowledged that fitness level might modulate IL-15 concentration [147,148]. Higher baseline IL-15 was observed in both resistance and endurance-trained athletes compared to untrained individuals [148]. A higher increment of serum IL-15 upon exercise was observed in athletes after the eccentric than concentric phase emphasized resistance exercises. The IL-15 increment found in athletes doing resistance exercises was also higher than in untrained subjects [147]. On the other hand, Kapilevich et al. reported that endurance training did not influence IL-15 concentration in either trained or untrained participants.
Perez-Lopez et al., Yargic et al. and Micielska et al. studied cycling sprint and long-distance trail running and found upregulated IL-15 secretion [76,149,150]. Micielska et al. demonstrated an altered response of IL-15 to high-intensity circuit training following 5-week regular training [76]. After the first training session, serum IL-15 declined below its baseline values. After the last training, IL-15 increased. These results support the hypothesis that IL-15 increases along with an improving fitness level. An acute bout of high-intensity interval endurance training did not change IL-15 concentration in sedentary subjects [79].
Nishida et al. demonstrated no change in IL-15 concentration in older women after 12 weeks of regular aerobic activity, namely, bench step exercise [151]. However, the training was not supervised, which might have influenced the results. Similarly, Roh et al. reported no change in IL-15 concentration in overweight and obese adolescents undergoing a 16-week taekwondo training [113]. Participants’ body composition might have affected this study results as IL-15 concentration correlates with fat mass, and its response to exercise in overweight and obese individuals may be altered [152].
4. Conclusions
Research on exercise-induced cytokines has many limitations and thus is not easy. The most common limitation is a small number of studied individuals who usually have distinct baseline characteristics like sex (mainly men), age (usually younger participants), or race. Another is the use of various protocols for physical activity—it complicates the comparison of obtained results between different studies. The next limitation is related to the technical specifications of cytokine measurement. Some are measured in either blood plasma or serum, whereas others in muscle biopsies. Regardless of the mentioned problems, a certain generalization of the exercise effects on cytokines is possible.
There is no doubt that exercise affects many organs and tissues, which release various cytokines in response. Some are directly secreted by contracting muscles (MSTN, DCN, BDNF, IL-15) while others (FST, FGF21) by non-myocyte cells, for example, hepatocytes or adipocytes. These exercise-induced cytokines regulate muscle differentiation, growth, and remodeling in response to the whole body’s exercise and metabolism.
The change in the concentration is cytokine-specific and also depends on various features of the exercise. An important feature is a type of exercise like endurance, resistance, dynamic power, or HIIT. Another is related to the metabolism mode, such as aerobic, anaerobic, or mixed. An additional feature is the exercise’s intensity. It can be measured as the percentage of VO2max for the endurance activity, the percentage of the one-repetition maximum for the resistance exercise, or the percentage for the estimated maximal heart rate for any form of physical activity. The amount and duration of exercise impact the release of the cytokines as well. Short-lasting or a single bout of an acute exercise has a different effect on cytokines than prolonged activity or repeated regular training. The baseline fitness level may modify the type, and extent of cytokine response to physical activity as some of these proteins are secreted or not, depending on whether someone is a worse or better fit.
In this review, we have focused mainly on the physiology of exercise-induced cytokines. Studying these proteins and their relation to exercise, although challenging, is very interesting. It helps to understand many aspects of exercise physiology and should be useful in the research on muscle pathology or limited exercise tolerance. As some of the cytokines are involved in muscle growth and differentiation of progenitor cells into myocytes, studying them is promising in regenerative medicine. Investigation of other exercise-related cytokines regulating energy, lipid, carbohydrate, or protein metabolism might be valuable in researching several metabolic diseases and conditions like diabetes, obesity, malnutrition, or cachexia. Still, many gaps in the knowledge about exercise-induced cytokines (i.e., myokines, hepatokines, adipomyokines) exist. New studies and findings gradually fill these gaps.
Author Contributions
P.G. and R.D. are originators of the paper; R.D., D.D., and M.P. prepared the draft and final version of the manuscript and should be considered as equal first authors; A.Z.-K., P.G., and M.R. made major revisions and proofreading; P.G. and M.R. are senior authors. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
The authors would like to appreciate the work of Wiktoria Domin, who prepared Figure 1.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pedersen, B.K.; Steensberg, A.; Fischer, C.; Keller, C.; Keller, P.; Plomgaard, P.; Febbraio, M.; Saltin, B. Searching for the exercise factor: Is IL-6 a candidate? J. Muscle Res. Cell Motil. 2003, 24, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Seldin, M.M.; Peterson, J.M.; Byerly, M.S.; Wei, Z.; Wong, G.W. Myonectin (CTRP15), a Novel Myokine That Links Skeletal Muscle to Systemic Lipid Homeostasis. J. Biol. Chem. 2012, 287, 11968–11980. [Google Scholar] [CrossRef] [PubMed]
- Whitham, M.; Febbraio, M.A. The ever-expanding myokinome: Discovery challenges and therapeutic implications. Nat. Rev. Drug Discov. 2016, 15, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an Endocrine Organ: Focus on Muscle-Derived Interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Yasukawa, K.; Harada, H.; Taga, T.; Watanabe, S.; Matsuda, T.; Kashiwamura, S.-I.; Nakajima, K.; Koyama, K.; Iwamatsu, A.; et al. Complementary DNA for a novel human interleukin (BSF-2) that induces B lymphocytes to produce immunoglobulin. Nat. Cell Biol. 1986, 324, 73–76. [Google Scholar] [CrossRef]
- Kelly, M.; Gauthier, M.-S.; Saha, A.K.; Ruderman, N.B.; Hong, E.-G.; Ko, H.J.; Cho, Y.-R.; Kim, H.-J.; Yu, T.Y.; Friedline, R.H.; et al. Activation of AMP-Activated Protein Kinase by Interleukin-6 in Rat Skeletal Muscle: Association With Changes in cAMP, Energy State, and Endogenous Fuel Mobilization. Diabetes 2009, 58, 1953–1960. [Google Scholar] [CrossRef]
- Ikeda, S.-I.; Tamura, Y.; Kakehi, S.; Sanada, H.; Kawamori, R.; Watada, H. Exercise-induced increase in IL-6 level enhances GLUT4 expression and insulin sensitivity in mouse skeletal muscle. Biochem. Biophys. Res. Commun. 2016, 473, 947–952. [Google Scholar] [CrossRef]
- Van Hall, G.; Steensberg, A.; Sacchetti, M.; Fischer, C.; Keller, C.; Schjerling, P.; Hiscock, N.; Møller, K.; Saltin, B.; Febbraio, M.A.; et al. Interleukin-6 Stimulates Lipolysis and Fat Oxidation in Humans. J. Clin. Endocrinol. Metab. 2003, 88, 3005–3010. [Google Scholar] [CrossRef]
- Petersen, E.W.; Carey, A.L.; Sacchetti, M.; Steinberg, G.R.; Macaulay, S.L.; Febbraio, M.A.; Pedersen, B.K. Acute IL-6 treatment increases fatty acid turnover in elderly humans in vivo and in tissue culture in vitro. Am. J. Physiol. Metab. 2005, 288, E155–E162. [Google Scholar] [CrossRef]
- Mitchell, C.J.; Churchward-Venne, T.A.; Bellamy, L.; Parise, G.; Baker, S.K.; Phillips, S.M. Muscular and Systemic Correlates of Resistance Training-Induced Muscle Hypertrophy. PLoS ONE 2013, 8, e78636. [Google Scholar] [CrossRef]
- Begue, G.; Douillard, A.; Galbes, O.; Rossano, B.; Vernus, B.; Candau, R.; Py, G. Early Activation of Rat Skeletal Muscle IL-6/STAT1/STAT3 Dependent Gene Expression in Resistance Exercise Linked to Hypertrophy. PLoS ONE 2013, 8, e57141. [Google Scholar] [CrossRef] [PubMed]
- Northoff, H.; Berg, A. Immunologic Mediators as Parameters of the Reaction to Strenuous Exercise. Int. J. Sports Med. 1991, 12, S9–S15. [Google Scholar] [CrossRef] [PubMed]
- Ostrowski, K.; Rohde, T.; Zacho, M.; Asp, S.; Pedersen, B.K. Evidence that interleukin-6 is produced in human skeletal muscle during prolonged running. J. Physiol. 1998, 508, 949–953. [Google Scholar] [CrossRef] [PubMed]
- Starkie, R.L.; Arkinstall, M.J.; Koukoulas, I.; Hawley, J.A.; Febbraio, M.A. Carbohydrate ingestion attenuates the increase in plasma interleukin-6, but not skeletal muscle interleukin-6 mRNA, during exercise in humans. J. Physiol. 2001, 533, 585–591. [Google Scholar] [CrossRef]
- Steensberg, A.; Keller, C.; Starkie, R.L.; Osada, T.; Febbraio, M.A.; Pedersen, B.K. IL-6 and TNF-α expression in, and release from, contracting human skeletal muscle. Am. J. Physiol. Metab. 2002, 283, E1272–E1278. [Google Scholar] [CrossRef]
- Febbraio, M.A.; Pedersen, B.K. Contraction-Induced Myokine Production and Release: Is Skeletal Muscle an Endocrine Organ? Exerc. Sport Sci. Rev. 2005, 33, 114–119. [Google Scholar] [CrossRef]
- Leggate, M.; Nowell, M.A.; A Jones, S.; Nimmo, M.A. The response of interleukin-6 and soluble interleukin-6 receptor isoforms following intermittent high intensity and continuous moderate intensity cycling. Cell Stress Chaperon. 2010, 15, 827–833. [Google Scholar] [CrossRef]
- Zwetsloot, K.A.; John, C.S.; Lawrence, M.M.; A Battista, R.; Shanely, R.A. High-intensity interval training induces a modest systemic inflammatory response in active, young men. J. Inflamm. Res. 2014, 7, 9–17. [Google Scholar] [CrossRef]
- Steensberg, A.; Van Hall, G.; Osada, T.; Sacchetti, M.; Saltin, B.; Pedersen, B.K. Production of interleukin-6 in contracting human skeletal muscles can account for the exercise-induced increase in plasma interleukin-6. J. Physiol. 2000, 529, 237–242. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Steensberg, A.; Schjerling, P. Exercise and interleukin-6. Curr. Opin. Hematol. 2001, 8, 137–141. [Google Scholar] [CrossRef]
- Febbraio, M.A.; Pedersen, B.K. Muscle-derived interleukin-6: Mechanisms for activation and possible biological roles. FASEB J. 2002, 16, 1335–1347. [Google Scholar] [CrossRef] [PubMed]
- Bostroem, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Bostroem, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nat. Cell Biol. 2012, 481, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.; Rioux, B.V.; Goulet, E.D.; Johanssen, N.M.; Swift, D.L.; Bouchard, D.; Loewen, H.; Sénéchal, M. Effect of an acute exercise bout on immediate post-exercise irisin concentration in adults: A meta-analysis. Scand. J. Med. Sci. Sports 2017, 28, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Cai, X.; Sun, Z.; Schumann, U.; Zügel, M.; Steinacker, J.M. Chronic Exercise Training and Circulating Irisin in Adults: A Meta-Analysis. Sports Med. 2015, 45, 1577–1588. [Google Scholar] [CrossRef]
- Grygiel-Górniak, B.; Puszczewicz, M. A review on irisin, a new protagonist that mediates muscle-adipose-bone-neuron connectivity. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4687–4693. [Google Scholar]
- Pukajło, K.; Kolackov, K.; Laczmanski, L.; Daroszewski, J. Irisin—A new mediator of energy homeostasis. Postępy Hig. Med. Dośw. 2015, 69, 233–242. [Google Scholar] [CrossRef]
- Roca-Rivada, A.; Castelao, C.; Senin, L.L.; Landrove, M.O.; Baltar, J.; Crujeiras, A.B.; Seoane, L.M.; Casanueva, F.F.; María, P. FNDC5/Irisin Is Not Only a Myokine but Also an Adipokine. PLoS ONE 2013, 8, e60563. [Google Scholar] [CrossRef]
- Huh, J.Y.; Panagiotou, G.; Mougios, V.; Brinkoetter, M.; Vamvini, M.T.; Schneider, B.E.; Mantzoros, C.S. FNDC5 and irisin in humans: I. Predictors of circulating concentrations in serum and plasma and II. mRNA expression and circulating concentrations in response to weight loss and exercise. Metabolism 2012, 61, 1725–1738. [Google Scholar] [CrossRef]
- Aydin, S.; Kuloglu, T.; Aydin, S.; Kalayci, M.; Yilmaz, M.; Cakmak, T.; Albayrak, S.; Gungor, S.; Colakoglu, N.; Ozercan, I.H. A comprehensive immunohistochemical examination of the distribution of the fat-burning protein irisin in biological tissues. Peptides 2014, 61, 130–136. [Google Scholar] [CrossRef]
- Kuloglu, T.; Aydin, S.; Eren, M.N.; Yilmaz, M.; Sahin, I.; Kalayci, M.; Sarman, E.; Kaya, N.; Yilmaz, O.F.; Turk, A.; et al. Irisin: A potentially candidate marker for myocardial infarction. Peptides 2014, 55, 85–91. [Google Scholar] [CrossRef]
- Lee, J.H.; Jun, H.-S. Role of Myokines in Regulating Skeletal Muscle Mass and Function. Front. Physiol. 2019, 10, 42. [Google Scholar] [CrossRef] [PubMed]
- Eckel, J. Myokines in metabolic homeostasis and diabetes. Diabetologia 2019, 62, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Fiuza-Luces, C.; Santos-Lozano, A.; Joyner, M.; Carrera-Bastos, P.; Picazo, O.; Zugaza, J.L.; Izquierdo, M.; Ruilope, L.M.; Lucia, A. Exercise benefits in cardiovascular disease: Beyond attenuation of traditional risk factors. Nat. Rev. Cardiol. 2018, 15, 731–743. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Wang, L.; You, W.; Shan, T. Myokines mediate the cross talk between skeletal muscle and other organs. J. Cell. Physiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.S.; Pedersen, B.K.; Xu, G.; Lehmann, R.; Weigert, C.; Plomgaard, P. Exercise-Induced Secretion of FGF21 and Follistatin Are Blocked by Pancreatic Clamp and Impaired in Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2016, 101, 2816–2825. [Google Scholar] [CrossRef] [PubMed]
- The Human Protein Atlas. Available online: https://www.proteinatlas.org/ (accessed on 10 December 2020).
- Rodriguez, J.; Vernus, B.; Chelh, I.; Cassar-Malek, I.; Gabillard, J.-C.; Sassi, A.H.; Seiliez, I.; Picard, B.; Bonnieu, A. Myostatin and the skeletal muscle atrophy and hypertrophy signaling pathways. Cell. Mol. Life Sci. 2014, 71, 4361–4371. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Peng, Y.; Zhao, W.; Pan, J.; Ksiezak-Reding, H.; Cardozo, C.; Wu, Y.; Pajevic, P.D.; Bonewald, L.F.; Bauman, W.A.; et al. Myostatin inhibits osteoblastic differentiation by suppressing osteocyte-derived exosomal microRNA-218: A novel mechanism in muscle-bone communication. J. Biol. Chem. 2017, 292, 11021–11033. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.-H.; Bauman, W.A.; Cardozo, C. Myostatin inhibits glucose uptake via suppression of insulin-dependent and -independent signaling pathways in myoblasts. Physiol. Rep. 2018, 6, e13837. [Google Scholar] [CrossRef]
- Lee, Y.-S.; Huynh, T.V.; Lee, S.-J. Paracrine and endocrine modes of myostatin action. J. Appl. Physiol. 2016, 120, 592–598. [Google Scholar] [CrossRef]
- Czaja, W.; Nakamura, Y.K.; Li, N.; Eldridge, J.A.; DeAvila, D.M.; Thompson, T.B.; Rodgers, B.D. Myostatin regulates pituitary development and hepatic IGF1. Am. J. Physiol. Metab. 2019, 316, E1036–E1049. [Google Scholar] [CrossRef]
- Sriram, S.; Subramanian, S.; Sathiakumar, D.; Venkatesh, R.; Salerno, M.S.; McFarlane, C.D.; Kambadur, R.; Sharma, M. Modulation of reactive oxygen species in skeletal muscle by myostatin is mediated through NF-κB. Aging Cell 2011, 10, 931–948. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.S.; Rutti, S.; Arous, C.; Clemmesen, J.O.; Secher, N.H.; Drescher, A.; Gonelle-Gispert, C.; Halban, P.A.; Pedersen, B.K.; Weigert, C.; et al. Circulating Follistatin Is Liver-Derived and Regulated by the Glucagon-to-Insulin Ratio. J. Clin. Endocrinol. Metab. 2016, 101, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Zhou, X.; Mitch, W.E.; Goldberg, A.L. Myostatin/activin pathway antagonism: Molecular basis and therapeutic potential. Int. J. Biochem. Cell Biol. 2013, 45, 2333–2347. [Google Scholar] [CrossRef] [PubMed]
- Winbanks, C.E.; Weeks, K.L.; Thomson, R.E.; Sepulveda, P.V.; Beyer, C.; Qian, H.; Chen, J.L.; Allen, J.M.; Lancaster, G.I.; Febbraio, M.A.; et al. Follistatin-mediated skeletal muscle hypertrophy is regulated by Smad3 and mTOR independently of myostatin. J. Cell Biol. 2012, 197, 997–1008. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Pervin, S.; Lee, S.-J.; Kuo, A.; Grijalva, V.; David, J.; Vergnes, L.; Reddy, S.T. Metabolic profiling of follistatin overexpression: A novel therapeutic strategy for metabolic diseases. Diabetes Metab. Syndr. Obes. 2018, 11, 65–84. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, K.; Han, B.; Xu, Z.; Gao, X. The emerging role of follistatin under stresses and its implications in diseases. Gene 2018, 639, 111–116. [Google Scholar] [CrossRef]
- Kang, Y.-M.; Lee, S.-K.; Chun, Y.-M.; Choi, Y.; Moon, S.-H.; Lee, H.-M.; Kang, H.J. Follistatin Mitigates Myofibroblast Differentiation and Collagen Synthesis of Fibroblasts from Scar Tissue around Injured Flexor Tendons. Yonsei Med. J. 2020, 61, 85–93. [Google Scholar] [CrossRef]
- Han, X.; Møller, L.L.V.; De Groote, E.; Bojsen-Møller, K.N.; Davey, J.; Henríquez-Olguin, C.; Li, Z.; Knudsen, J.R.; Jensen, T.E.; Madsbad, S.; et al. Mechanisms involved in follistatin-induced hypertrophy and increased insulin action in skeletal muscle. J. Cachex. Sarcopenia Muscle 2019, 10, 1241–1257. [Google Scholar] [CrossRef]
- DCN Decorin [Homo sapiens (Human)]—Gene—NCBI. Available online: https://www.ncbi.nlm.nih.gov/gene?Db=gene&Cmd=ShowDetailView&TermToSearch=1634 (accessed on 22 December 2019).
- Weishaupt, N.; Blesch, A.; Fouad, K. BDNF: The career of a multifaceted neurotrophin in spinal cord injury. Exp. Neurol. 2012, 238, 254–264. [Google Scholar] [CrossRef]
- Kowiański, P.; Lietzau, G.; Czuba, E.; Waśkow, M.; Steliga, A.; Moryś, J. BDNF: A Key Factor with Multipotent Impact on Brain Signaling and Synaptic Plasticity. Cell. Mol. Neurobiol. 2018, 38, 579–593. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Pedersen, M.; Krabbe, K.S.; Bruunsgaard, H.; Matthews, V.B.; Febbraio, M.A. Role of exercise-induced brain-derived neurotrophic factor production in the regulation of energy homeostasis in mammals. Exp. Physiol. 2009, 94, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Oulion, S.; Bertrand, S.; Escriva, H. Evolution of the FGF Gene Family. Int. J. Evol. Biol. 2012, 2012, 298147. [Google Scholar] [CrossRef] [PubMed]
- Tezze, C.; Romanello, V.; Sandri, M. FGF21 as Modulator of Metabolism in Health and Disease. Front. Physiol. 2019, 10, 419. [Google Scholar] [CrossRef] [PubMed]
- Potthoff, M.J.; Inagaki, T.; Satapati, S.; Ding, X.; He, T.; Goetz, R.; Mohammadi, M.; Finck, B.N.; Mangelsdorf, D.J.; Kliewer, S.A.; et al. FGF21 induces PGC-1 and regulates carbohydrate and fatty acid metabolism during the adaptive starvation response. Proc. Natl. Acad. Sci. USA 2009, 106, 10853–10858. [Google Scholar] [CrossRef] [PubMed]
- Fisher, F.M.; Kleiner, S.; Douris, N.; Fox, E.C.; Mepani, R.J.; Verdeguer, F.; Wu, J.; Kharitonenkov, A.; Flier, J.S.; Maratos-Flier, E.; et al. FGF21 regulates PGC-1 and browning of white adipose tissues in adaptive thermogenesis. Genes Dev. 2012, 26, 271–281. [Google Scholar] [CrossRef]
- Burton, J.D.; Bamford, R.N.; Peters, C.; Grant, A.J.; Kurys, G.; Goldman, C.K.; Brennan, J.; Roessler, E.; Waldmann, T.A. A lymphokine, provisionally designated interleukin T and produced by a human adult T-cell leukemia line, stimulates T-cell proliferation and the induction of lymphokine-activated killer cells. Proc. Natl. Acad. Sci. USA 1994, 91, 4935–4939. [Google Scholar] [CrossRef]
- Leclercq, G.; Debacker, V.; De Smedt, M.; Plum, J. Differential effects of interleukin-15 and interleukin-2 on differentiation of bipotential T/natural killer progenitor cells. J. Exp. Med. 1996, 184, 325–336. [Google Scholar] [CrossRef]
- Nolz, J.C.; Richer, M.J. Control of memory CD8+ T cell longevity and effector functions by IL-15. Mol. Immunol. 2020, 117, 180–188. [Google Scholar] [CrossRef]
- Furmanczyk, P.S. Interleukin-15 increases myosin accretion in human skeletal myogenic cultures. Cell Biol. Int. 2003, 27, 845–851. [Google Scholar] [CrossRef]
- Quinn, L.S.; Strait-Bodey, L.; Anderson, B.G.; Argilés, J.M.; Havel, P.J. Interleukin-15 stimulates adiponectin secretion by 3T3-L1 adipocytes: Evidence for a skeletal muscle-to-fat signaling pathway. Cell Biol. Int. 2005, 29, 449–457. [Google Scholar] [CrossRef]
- Quinn, L.S.; Anderson, B.G.; Strait-Bodey, L.; Stroud, A.M.; Argilés, J.M. Oversecretion of interleukin-15 from skeletal muscle reduces adiposity. Am. J. Physiol. Metab. 2009, 296, E191–E202. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Liu, D. Hydrodynamic delivery of interleukin 15 gene promotes resistance to high fat diet-induced obesity, fatty liver and improves glucose homeostasis. Gene Ther. 2014, 22, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Ma, Y.; Gao, M.; Liu, D. IL-15/sIL-15Rα gene transfer induces weight loss and improves glucose homeostasis in obese mice. Gene Ther. 2016, 23, 349–356. [Google Scholar] [CrossRef] [PubMed]
- McPherron, A.C.; Lawler, A.M.; Lee, S.-J. Regulation of skeletal muscle mass in mice by a new TGF-p superfamily member. Nat. Cell Biol. 1997, 387, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Wrana, J.L. Signaling by the TGF Superfamily. Cold Spring Harb. Perspect. Biol. 2013, 5, a011197. [Google Scholar] [CrossRef] [PubMed]
- Elliott, B.; Herbert, P.; Sculthorpe, N.; Grace, F.M.; Stratton, D.; Hayes, L.D. Lifelong exercise, but not short-term high-intensity interval training, increases GDF11, a marker of successful aging: A preliminary investigation. Physiol. Rep. 2017, 5, e13343. [Google Scholar] [CrossRef] [PubMed]
- Schuelke, M.; Wagner, K.R.; Stolz, L.E.; Hübner, C.; Riebel, T.; Kömen, W.; Braun, T.; Tobin, J.F.; Lee, S.-J. Myostatin Mutation Associated with Gross Muscle Hypertrophy in a Child. N. Engl. J. Med. 2004, 350, 2682–2688. [Google Scholar] [CrossRef]
- Lee, S.-J. Extracellular Regulation of Myostatin: A Molecular Rheostat for Muscle Mass. Immunol. Endocr. Metab. Agents Med. Chem. 2010, 10, 183–194. [Google Scholar] [CrossRef]
- Lessard, S.J.; Rivas, D.A.; Alves-Wagner, A.B.; Hirshman, M.F.; Gallagher, I.J.; Constantin-Teodosiu, T.; Atkins, R.; Greenhaff, P.L.; Qi, N.R.; Gustafsson, T.; et al. Resistance to Aerobic Exercise Training Causes Metabolic Dysfunction and Reveals Novel Exercise-Regulated Signaling Networks. Diabetes 2013, 62, 2717–2727. [Google Scholar] [CrossRef]
- Massagué, J. TGFβ signalling in context. Nat. Rev. Mol. Cell Biol. 2012, 13, 616–630. [Google Scholar] [CrossRef]
- Covington, J.D.; Tam, C.S.; Bajpeyi, S.; Galgani, J.E.; Noland, R.C.; Smith, S.R.; Redman, L.M.; Ravussin, E. Myokine Expression in Muscle and Myotubes in Response to Exercise Stimulation. Med. Sci. Sports Exerc. 2016, 48, 384–390. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hittel, D.S.; Axelson, M.; Sarna, N.; Shearer, J.; Huffman, K.M.; Kraus, W.E. Myostatin Decreases with Aerobic Exercise and Associates with Insulin Resistance. Med. Sci. Sports Exerc. 2010, 42, 2023–2029. [Google Scholar] [CrossRef] [PubMed]
- Kabak, B.; Belviranlı, M.; Okudan, N. Irisin and myostatin responses to acute high-intensity interval exercise in humans. Horm. Mol. Biol. Clin. Investig. 2018, 35. [Google Scholar] [CrossRef] [PubMed]
- Micielska, K.; Gmiat, A.; Żychowska, M.; Kozlowska, M.; Walentukiewicz, A.; Lysak-Radomska, A.; Jaworska, J.; Rodziewicz, E.; Duda-Biernacka, B.; Ziemann, E. The beneficial effects of 15 units of high-intensity circuit training in women is modified by age, baseline insulin resistance and physical capacity. Diabetes Res. Clin. Pract. 2019, 152, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Kazemi, F. The correlation of resistance exercise-induced myostatin with insulin resistance and plasma cytokines in healthy young men. J. Endocrinol. Investig. 2015, 39, 383–388. [Google Scholar] [CrossRef]
- Kerschan-Schindl, K.; Thalmann, M.M.; Weiss, E.; Tsironi, M.; Föger-Samwald, U.; Meinhart, J.; Skenderi, K.; Pietschmann, P. Changes in Serum Levels of Myokines and Wnt-Antagonists after an Ultramarathon Race. PLoS ONE 2015, 10, e0132478. [Google Scholar] [CrossRef]
- He, Z.; Tian, Y.; Valenzuela, P.L.; Huang, C.; Zhao, J.; Hong, P.; He, Z.; Yin, S.; Lucia, A. Myokine Response to High-Intensity Interval vs. Resistance Exercise: An Individual Approach. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef]
- He, Z.; Tian, Y.; Valenzuela, P.L.; Huang, C.; Zhao, J.; Hong, P.; He, Z.; Yin, S.; Lucia, A. Myokine/Adipokine Response to “Aerobic” Exercise: Is It Just a Matter of Exercise Load? Front. Physiol. 2019, 10, 691. [Google Scholar] [CrossRef]
- Bagheri, R.; Rashidlamir, A.; Motevalli, M.S.; Elliott, B.T.; Mehrabani, J.; Wong, A. Effects of upper-body, lower-body, or combined resistance training on the ratio of follistatin and myostatin in middle-aged men. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 119, 1921–1931. [Google Scholar] [CrossRef]
- Hofmann, M.; Schober-Halper, B.; Oesen, S.; Franzke, B.; Tschan, H.; Bachl, N.; Strasser, E.-M.; Quittan, M.; Wagner, K.-H.; Wessner, B. Effects of elastic band resistance training and nutritional supplementation on muscle quality and circulating muscle growth and degradation factors of institutionalized elderly women: The Vienna Active Ageing Study (VAAS). Graefe’s Arch. Clin. Exp. Ophthalmol. 2016, 116, 885–897. [Google Scholar] [CrossRef]
- Robertson, D.; Klein, R.; De Vos, F.; McLachlan, R.; Wettenhall, R.; Hearn, M.; Burger, H.; De Kretser, D. The isolation of polypeptides with FSH suppressing activity from bovine follicular fluid which are structurally different to inhibin. Biochem. Biophys. Res. Commun. 1987, 149, 744–749. [Google Scholar] [CrossRef]
- Makanji, Y.; Zhu, J.; Mishra, R.; Holmquist, C.; Wong, W.P.S.; Schwartz, N.B.; Mayo, K.E.; Woodruff, T.K. Inhibin at 90: From Discovery to Clinical Application, a Historical Review. Endocr. Rev. 2014, 35, 747–794. [Google Scholar] [CrossRef] [PubMed]
- Schneyer, A.; Wang, Q.; Sidis, Y.; Sluss, P.M. Differential Distribution of Follistatin Isoforms: Application of a New FS315-Specific Immunoassay. J. Clin. Endocrinol. Metab. 2004, 89, 5067–5075. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.; Brandt, C.; Nielsen, A.R.; Hojman, P.; Whitham, M.; Febbraio, M.A.; Pedersen, B.K.; Plomgaard, P. Exercise Induces a Marked Increase in Plasma Follistatin: Evidence That Follistatin Is a Contraction-Induced Hepatokine. Endocrinology 2011, 152, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Schneyer, A.; Sidis, Y.; Gulati, A.; Sun, J.L.; Keutmann, H.; Krasney, P.A. Differential Antagonism of Activin, Myostatin and Growth and Differentiation Factor 11 by Wild-Type and Mutant Follistatin. Endocrinology 2008, 149, 4589–4595. [Google Scholar] [CrossRef]
- Hansen, J.S.; Plomgaard, P. Circulating follistatin in relation to energy metabolism. Mol. Cell. Endocrinol. 2016, 433, 87–93. [Google Scholar] [CrossRef]
- Perakakis, N.; Mougios, V.; Fatouros, I.; Siopi, A.; Draganidis, D.; Peradze, N.; Ghaly, W.; Mantzoros, C.S. Physiology of Activins/Follistatins: Associations With Metabolic and Anthropometric Variables and Response to Exercise. J. Clin. Endocrinol. Metab. 2018, 103, 3890–3899. [Google Scholar] [CrossRef]
- Hormonal responses following eccentric exercise in humans. Hormones 2018, 16, 405–413. [CrossRef]
- Sargeant, J.A.; Aithal, G.P.; Takamura, T.; Misu, H.; Takayama, H.; Douglas, J.A.; Turner, M.C.; Stensel, D.J.; Nimmo, M.A.; Webb, D.R.; et al. The influence of adiposity and acute exercise on circulating hepatokines in normal-weight and overweight/obese men. Appl. Physiol. Nutr. Metab. 2018, 43, 482–490. [Google Scholar] [CrossRef]
- Willis, S.A.; Sargeant, J.A.; Thackray, A.E.; Yates, T.; Stensel, D.J.; Aithal, G.P.; King, J.A. Effect of exercise intensity on circulating hepatokine concentrations in healthy men. Appl. Physiol. Nutr. Metab. 2019, 44, 1065–1072. [Google Scholar] [CrossRef]
- Kanzleiter, T.; Rath, M.; Görgens, S.W.; Jensen, J.; Tangen, D.S.; Kolnes, A.J.; Kolnes, K.J.; Lee, S.; Eckel, J.; Schürmann, A.; et al. The myokine decorin is regulated by contraction and involved in muscle hypertrophy. Biochem. Biophys. Res. Commun. 2014, 450, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Knuiman, P.; Hopman, M.T.E.; Hangelbroek, R.; Mensink, M. Plasma cytokine responses to resistance exercise with different nutrient availability on a concurrent exercise day in trained healthy males. Physiol. Rep. 2018, 6, e13708. [Google Scholar] [CrossRef] [PubMed]
- Bugera, E.M.; Duhamel, T.A.; Peeler, J.D.; Cornish, S.M. The systemic myokine response of decorin, interleukin-6 (IL-6) and interleukin-15 (IL-15) to an acute bout of blood flow restricted exercise. Graefe’s Arch. Clin. Exp. Ophthalmol. 2018, 118, 2679–2686. [Google Scholar] [CrossRef] [PubMed]
- Lightfoot, A.P.; Cooper, R.G. The role of myokines in muscle health and disease. Curr. Opin. Rheumatol. 2016, 28, 661–666. [Google Scholar] [CrossRef]
- Leibrock, J.; Lottspeich, F.; Hohn, A.; Hofer, M.; Hengerer, B.; Masiakowski, P.; Thoenen, H.; Barde, Y.-A. Molecular cloning and expression of brain-derived neurotrophic factor. Nat. Cell Biol. 1989, 341, 149–152. [Google Scholar] [CrossRef]
- Acheson, A.; Conover, J.C.; Fandl, J.P.; DeChiara, T.M.; Russell, M.; Thadani, A.; Squinto, S.P.; Yancopoulos, G.D.; Lindsay, R.M. A BDNF autocrine loop in adult sensory neurons prevents cell death. Nat. Cell Biol. 1995, 374, 450–453. [Google Scholar] [CrossRef]
- Chacón-Fernández, P.; Säuberli, K.; Colzani, M.; Moreau, T.; Ghevaert, C.; Barde, Y.-A. Brain-derived Neurotrophic Factor in Megakaryocytes. J. Biol. Chem. 2016, 291, 9872–9881. [Google Scholar] [CrossRef]
- Kestin, A.S.; Ellis, P.A.; Barnard, M.R.; Errichetti, A.; Rosner, B.A.; Michelson, A.D. Effect of strenuous exercise on platelet activation state and reactivity. Circulation 1993, 88, 1502–1511. [Google Scholar] [CrossRef]
- Krabbe, K.S.; Nielsen, A.R.; Krogh-Madsen, R.; Plomgaard, P.; Rasmussen, P.; Erikstrup, C.; Fischer, C.P.; Lindegaard, B.; Petersen, A.M.W.; Taudorf, S.; et al. Brain-derived neurotrophic factor (BDNF) and type 2 diabetes. Diabetologia 2007, 50, 431–438. [Google Scholar] [CrossRef]
- Szuhany, K.L.; Bugatti, M.; Otto, M.W. A meta-analytic review of the effects of exercise on brain-derived neurotrophic factor. J. Psychiatr. Res. 2015, 60, 56–64. [Google Scholar] [CrossRef]
- Håkansson, K.; Ledreux, A.; Daffner, K.; Terjestam, Y.; Bergman, P.; Carlsson, R.; Kivipelto, M.; Winblad, B.; Granholm, A.-C.; Mohammed, A.K.H. BDNF Responses in Healthy Older Persons to 35 Minutes of Physical Exercise, Cognitive Training, and Mindfulness: Associations with Working Memory Function. J. Alzheimer’s Dis. 2016, 55, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Hötting, K.; Schickert, N.; Kaiser, J.; Röder, B.; Schmidt-Kassow, M. The Effects of Acute Physical Exercise on Memory, Peripheral BDNF, and Cortisol in Young Adults. Neural Plast. 2016, 2016, 6860573. [Google Scholar] [CrossRef] [PubMed]
- Fortunato, A.K.; Pontes, W.M.; De Souza, D.M.S.; Prazeres, J.S.F.; Marcucci-Barbosa, L.S.; Santos, J.M.M.; Veira, É.L.M.; Bearzoti, E.; Pinto, K.M.D.C.; Talvani, A.; et al. Strength Training Session Induces Important Changes on Physiological, Immunological, and Inflammatory Biomarkers. J. Immunol. Res. 2018, 2018, 9675216. [Google Scholar] [CrossRef] [PubMed]
- Murawska-Cialowicz, E.; Wojna, J.; Zuwala-Jagiello, J. Crossfit training changes brain-derived neurotrophic factor and irisin levels at rest, after wingate and progressive tests, and improves aerobic capacity and body composition of young physically active men and women. J. Physiol. Pharmacol. 2015, 66, 811–821. [Google Scholar] [PubMed]
- Tsai, C.; Pan, C.; Chen, F.; Wang, C.-H.; Chou, F. The effects of acute aerobic exercise on a task-switching paradigm and BDNF levels in young adults with different levels of cardiorespiratory fitness. Exp. Physiol. 2016, 101, 836–850. [Google Scholar] [CrossRef]
- Church, D.D.; Hoffman, J.R.; Mangine, G.T.; Jajtner, A.R.; Townsend, J.R.; Beyer, K.S.; Wang, R.; La Monica, M.B.; Fukuda, D.H.; Stout, J.R. Comparison of high-intensity vs. high-volume resistance training on the BDNF response to exercise. J. Appl. Physiol. 2016, 121, 123–128. [Google Scholar] [CrossRef]
- Marquez, C.M.S.; Vanaudenaerde, B.; Troosters, T.; Wenderoth, N. High-intensity interval training evokes larger serum BDNF levels compared with intense continuous exercise. J. Appl. Physiol. 2015, 119, 1363–1373. [Google Scholar] [CrossRef]
- Wagner, G.; Herbsleb, M.; De La Cruz, F.; Schumann, A.; Brünner, F.; Schachtzabel, C.; Gussew, A.; Puta, C.; Smesny, S.; Gabriel, H.W.; et al. Hippocampal Structure, Metabolism, and Inflammatory Response after a 6-Week Intense Aerobic Exercise in Healthy Young Adults: A Controlled Trial. Br. J. Pharmacol. 2015, 35, 1570–1578. [Google Scholar] [CrossRef]
- Antunes, B.D.M.M.; Rossi, F.E.; Teixeira, A.M.; Lira, F.S. Short-time high-intensity exercise increases peripheral BDNF in a physical fitness-dependent way in healthy men. Eur. J. Sport Sci. 2019, 20, 43–50. [Google Scholar] [CrossRef]
- Jeon, Y.K.; Ha, C.H. The effect of exercise intensity on brain derived neurotrophic factor and memory in adolescents. Environ. Health Prev. Med. 2017, 22, 1–6. [Google Scholar] [CrossRef]
- Roh, H.-T.; Cho, S.-Y.; So, W.-Y. Effects of Regular Taekwondo Intervention on Oxidative Stress Biomarkers and Myokines in Overweight and Obese Adolescents. Int. J. Environ. Res. Public Health 2020, 17, 2505. [Google Scholar] [CrossRef] [PubMed]
- Polyakova, M.; Schlögl, H.; Sacher, J.; Schmidt-Kassow, M.; Kaiser, J.; Stumvoll, M.; Kratzsch, J.; Schroeter, M.L. Stability of BDNF in Human Samples Stored Up to 6 Months and Correlations of Serum and EDTA-Plasma Concentrations. Int. J. Mol. Sci. 2017, 18, 1189. [Google Scholar] [CrossRef] [PubMed]
- Itoh, N. Hormone-like (endocrine) Fgfs: Their evolutionary history and roles in development, metabolism, and disease. Cell Tissue Res. 2010, 342, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mraz, M.; Bartlova, M.; Lacinova, Z.; Michalsky, D.; Kasalicky, M.; Haluzikova, D.; Matoulek, M.; Dostalova, I.; Humenanska, V.; Haluzik, M. Serum concentrations and tissue expression of a novel endocrine regulator fibroblast growth factor-21 in patients with type 2 diabetes and obesity. Clin. Endocrinol. 2009, 71, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yeung, D.C.; Karpisek, M.; Stejskal, D.; Zhou, Z.-G.; Liu, F.; Wong, R.L.; Chow, W.-S.; Tso, A.W.; Lam, K.S.; et al. Serum FGF21 Levels Are Increased in Obesity and Are Independently Associated With the Metabolic Syndrome in Humans. Diabetes 2008, 57, 1246–1253. [Google Scholar] [CrossRef] [PubMed]
- Tacer, K.F.; Bookout, A.L.; Ding, X.; Kurosu, H.; John, G.B.; Wang, L.; Goetz, R.; Mohammadi, M.; Kuro-O, M.; Mangelsdorf, D.J.; et al. Research Resource: Comprehensive Expression Atlas of the Fibroblast Growth Factor System in Adult Mouse. Mol. Endocrinol. 2010, 24, 2050–2064. [Google Scholar] [CrossRef] [PubMed]
- Itoh, N. FGF21 as a Hepatokine, Adipokine, and Myokine in Metabolism and Diseases. Front. Endocrinol. 2014, 5, 107. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Kurosu, H.; Yamamoto, M.; Nandi, A.; Rosenblatt, K.P.; Goetz, R.; Eliseenkova, A.V.; Mohammadi, M.; Kuro-O, M. βKlotho is required for metabolic activity of fibroblast growth factor 21. Proc. Natl. Acad. Sci. USA 2007, 104, 7432–7437. [Google Scholar] [CrossRef]
- Hanks, L.J.; Gutierrez, O.M.; Bamman, M.M.; Ashraf, A.P.; McCormick, K.; Casazza, K. Circulating levels of fibroblast growth factor-21 increase with age independently of body composition indices among healthy individuals. J. Clin. Transl. Endocrinol. 2015, 2, 77–82. [Google Scholar] [CrossRef]
- Mashili, F.L.; Austin, R.L.; Deshmukh, A.S.; Fritz, T.; Caidahl, K.; Bergdahl, K.; Zierath, J.R.; Chibalin, A.V.; Moller, D.E.; Kharitonenkov, A.; et al. Direct effects of FGF21 on glucose uptake in human skeletal muscle: Implications for type 2 diabetes and obesity. Diabetes Metab. Res. Rev. 2011, 27, 286–297. [Google Scholar] [CrossRef]
- Shen, Y.; Ma, X.; Zhou, J.; Pan, X.; Hao, Y.; Zhou, M.; Lu, Z.; Gao, M.; Bao, Y.; Jia, W. Additive relationship between serum fibroblast growth factor 21 level and coronary artery disease. Cardiovasc. Diabetol. 2013, 12, 124. [Google Scholar] [CrossRef] [PubMed]
- Kharitonenkov, A.; Wroblewski, V.J.; Koester, A.; Chen, Y.-F.; Clutinger, C.K.; Tigno, X.T.; Hansen, B.C.; Shanafelt, A.B.; Etgen, G.J. The Metabolic State of Diabetic Monkeys Is Regulated by Fibroblast Growth Factor-21. Endocrinology 2007, 148, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Stanislaus, S.; Chinookoswong, N.; Lau, Y.Y.; Hager, T.; Patel, J.; Ge, H.; Weiszmann, J.; Lu, S.-C.; Graham, M.; et al. Acute glucose-lowering and insulin-sensitizing action of FGF21 in insulin-resistant mouse models—Association with liver and adipose tissue effects. Am. J. Physiol. Metab. 2009, 297, E1105–E1114. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Kim, S.H.; Min, Y.-K.; Yang, H.-M.; Lee, J.-B.; Lee, M.-S. Acute Exercise Induces FGF21 Expression in Mice and in Healthy Humans. PLoS ONE 2013, 8, e63517. [Google Scholar] [CrossRef]
- Tanimura, Y.; Aoi, W.; Takanami, Y.; Kawai, Y.; Mizushima, K.; Naito, Y.; Yoshikawa, T. Acute exercise increases fibroblast growth factor 21 in metabolic organs and circulation. Physiol. Rep. 2016, 4, e12828. [Google Scholar] [CrossRef] [PubMed]
- Morville, T.; Sahl, R.E.; Trammell, S.A.; Svenningsen, J.S.; Gillum, M.P.; Helge, J.W.; Clemmensen, C. Divergent effects of resistance and endurance exercise on plasma bile acids, FGF19, and FGF21 in humans. JCI Insight 2018, 3. [Google Scholar] [CrossRef]
- JanssenDuijghuijsen, L.M.; Keijer, J.; Mensink, M.; Lenaerts, K.; Ridder, L.; Nierkens, S.; Kartaram, S.W.; Verschuren, M.C.M.; Pieters, R.H.H.; Bas, R.; et al. Adaptation of exercise-induced stress in well-trained healthy young men. Exp. Physiol. 2017, 102, 86–99. [Google Scholar] [CrossRef]
- Cuevas-Ramos, D.; Paloma, A.-V.; Meza-Arana, C.E.; Brito-Córdova, G.; Gómez-Pérez, F.J.; Mehta, R.; Oseguera-Moguel, J.; Aguilar-Salinas, C.A. Exercise Increases Serum Fibroblast Growth Factor 21 (FGF21) Levels. PLoS ONE 2012, 7, e38022. [Google Scholar] [CrossRef]
- Taniguchi, H.; Tanisawa, K.; Sun, X.; Higuchi, M. Acute endurance exercise lowers serum fibroblast growth factor 21 levels in Japanese men. Clin. Endocrinol. 2016, 85, 861–867. [Google Scholar] [CrossRef]
- Khalafi, M.; Alamdari, K.A.; Symonds, M.; Nobari, H.; Carlos-Vivas, J. Impact of acute exercise on immediate and following early post-exercise FGF-21 concentration in adults: Systematic review and meta-analysis. Hormones 2020, 1–11. [Google Scholar] [CrossRef]
- Taniguchi, H.; Tanisawa, K.; Sun, X.; Kubo, T.; Higuchi, M. Endurance exercise reduces hepatic fat content and serum fibroblast growth factor 21 levels in elderly men. J. Clin. Endocrinol. Metab. 2016, 101, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Parmar, B.; Lewis, J.E.; Samms, R.J.; Ebling, F.J.P.; Cheng, C.C.; Adams, A.C.; Mallinson, J.; Cooper, S.; Taylor, T.; Ghasemi, R.; et al. Eccentric exercise increases circulating fibroblast activation protein α but not bioactive fibroblast growth factor 21 in healthy humans. Exp. Physiol. 2018, 103, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Grabstein, K.H.; Eisenman, J.; Shanebeck, K.; Rauch, C.; Srinivasan, S.; Fung, V.; Beers, C.; Richardson, J.; A Schoenborn, M.; Ahdieh, M.; et al. Cloning of a T cell growth factor that interacts with the beta chain of the interleukin-2 receptor. Science 1994, 264, 965–968. [Google Scholar] [CrossRef]
- Krolopp, J.E.; Thornton, S.M.; Abbott, M.J. IL-15 Activates the Jak3/STAT3 Signaling Pathway to Mediate Glucose Uptake in Skeletal Muscle Cells. Front. Physiol. 2016, 7, 626. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, L.; Patten, D.; Caron, A.; Garneau, L.; Pinault-Masson, E.; Foretz, M.; Haddad, P.; Anderson, B.; Quinn, L.; Jardine, K.; et al. IL-15 improves skeletal muscle oxidative metabolism and glucose uptake in association with increased respiratory chain supercomplex formation and AMPK pathway activation. Biochim. Biophys. Acta Gen. Subj. 2019, 1863, 395–407. [Google Scholar] [CrossRef]
- Ostrowski, K.; Hermann, C.; Bangash, A.; Schjerling, P.; Nielsen, J.N.; Pedersen, B.K. A trauma-like elevation of plasma cytokines in humans in response to treadmill running. J. Physiol. 1998, 513, 889–894. [Google Scholar] [CrossRef]
- Nieman, D.; Davis, J.M.; Henson, D.A.; Walberg-Rankin, J.; Shute, M.; Dumke, C.L.; Utter, A.C.; Vinci, D.M.; Carson, J.A.; Brown, A.; et al. Carbohydrate ingestion influences skeletal muscle cytokine mRNA and plasma cytokine levels after a 3-h run. J. Appl. Physiol. 2003, 94, 1917–1925. [Google Scholar] [CrossRef]
- Nieman, D.; Davis, J.M.; Brown, V.A.; Henson, D.A.; Dumke, C.L.; Utter, A.C.; Vinci, D.M.; Downs, M.F.; Smith, J.C.; Carson, J.A.; et al. Influence of carbohydrate ingestion on immune changes after 2 h of intensive resistance training. J. Appl. Physiol. 2004, 96, 1292–1298. [Google Scholar] [CrossRef]
- Riechman, S.E.; Balasekaran, G.; Roth, S.M.; Ferrell, R.E. Association of interleukin-15 protein and interleukin-15 receptor genetic variation with resistance exercise training responses. J. Appl. Physiol. 2004, 97, 2214–2219. [Google Scholar] [CrossRef]
- Nielsen, A.R.; Mounier, R.; Plomgaard, P.; Mortensen, O.H.; Penkowa, M.; Speerschneider, T.; Pilegaard, H.; Pedersen, B.K. Expression of interleukin-15 in human skeletal muscle—Effect of exercise and muscle fibre type composition. J. Physiol. 2007, 584, 305–312. [Google Scholar] [CrossRef]
- Nielsen, A.R.; Yfanti, C.; Nielsen, S.; Åkerström, T.C.A.; Peijs, L.; Zankari, A.; Fischer, C.P.; Pedersen, B.K. Endurance training enhances skeletal muscle interleukin-15 in human male subjects. Endocrine 2013, 45, 271–278. [Google Scholar] [CrossRef]
- Oliver, J.M.; Jenke, S.C.; Mata, J.D.; Kreutzer, A.; Jones, M.T. Acute Effect of Cluster and Traditional Set Configurations on Myokines Associated with Hypertrophy. Int. J. Sports. Med. 2016, 37, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Pérez-López, A.; McKendry, J.; Martin-Rincon, M.; Morales-Alamo, D.; Pérez-Köhler, B.; Valadés, D.; Buján, J.; Calbet, J.A.L.; Breen, L. Skeletal muscle IL-15/IL-15Rα and myofibrillar protein synthesis after resistance exercise. Scand. J. Med. Sci. Sports 2017, 28, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Park, K.M.; Park, S.C.; Kang, S. Effects of resistance exercise on adipokine factors and body composition in pre- and postmenopausal women. J. Exerc. Rehabil. 2019, 15, 676–682. [Google Scholar] [CrossRef]
- Bazgir, B.; Salesi, M.; Koushki, M.; Amirghofran, Z. Effects of Eccentric and Concentric Emphasized Resistance Exercise on IL-15 Serum Levels and Its Relation to Inflammatory Markers in Athletes and Non-Athletes. Asian J. Sports Med. 2015, 6, e27980. [Google Scholar] [CrossRef]
- Kapilevich, L.V.; Zakharova, A.N.; Kabachkova, A.V.; Kironenko, T.A.; Orlov, S.N. Dynamic and Static Exercises Differentially Affect Plasma Cytokine Content in Elite Endurance- and Strength-Trained Athletes and Untrained Volunteers. Front. Physiol. 2017, 8. [Google Scholar] [CrossRef]
- Pérez-López, A.; Martin-Rincon, M.; Santana, A.; Perez-Suarez, I.; Dorado, C.; Calbet, J.A.L.; Morales-Alamo, D. Antioxidants Facilitate High-intensity Exercise IL-15 Expression in Skeletal Muscle. Int. J. Sports Med. 2018, 40, 16–22. [Google Scholar] [CrossRef]
- Yargic, M.P.; Torgutalp, S.; Akin, S.; Babayeva, N.; Torgutalp, M.; Demirel, H.A.; Demirel, A.H. Acute long-distance trail running increases serum IL-6, IL-15, and Hsp72 levels. Appl. Physiol. Nutr. Metab. 2019, 44, 627–631. [Google Scholar] [CrossRef]
- Nishida, Y.; Tanaka, K.; Hara, M.; Hirao, N.; Tanaka, H.; Tobina, T.; Ikeda, M.; Yamato, H.; Ohta, M. Effects of home-based bench step exercise on inflammatory cytokines and lipid profiles in elderly Japanese females: A randomized controlled trial. Arch. Gerontol. Geriatr. 2015, 61, 443–451. [Google Scholar] [CrossRef]
- Duan, Y.; Li, F.; Wang, W.L.; Guo, Q.; Wen, C.; Li, Y.; Yin, Y. Interleukin-15 in obesity and metabolic dysfunction: Current understanding and future perspectives. Obes. Rev. 2017, 18, 1147–1158. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).