Increased Compulsivity in Adulthood after Early Adolescence Immune Activation: Preclinical Evidence
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Experimental Design
2.2.1. Lipopolysaccharide Immune Challenge
2.2.2. Exposure to Stress
2.3. Behavioral Assessment
2.3.1. 5-Choice Serial Reaction Time (5-CSRT) Task
2.3.2. Schedule-Induced Polydipsia (SIP) Procedure
2.3.3. Spontaneous Locomotor Activity and Novelty Reactivity
2.4. Data Analysis
3. Results
3.1. 5-CSRT Task
3.1.1. Acquisition and Baseline Performance
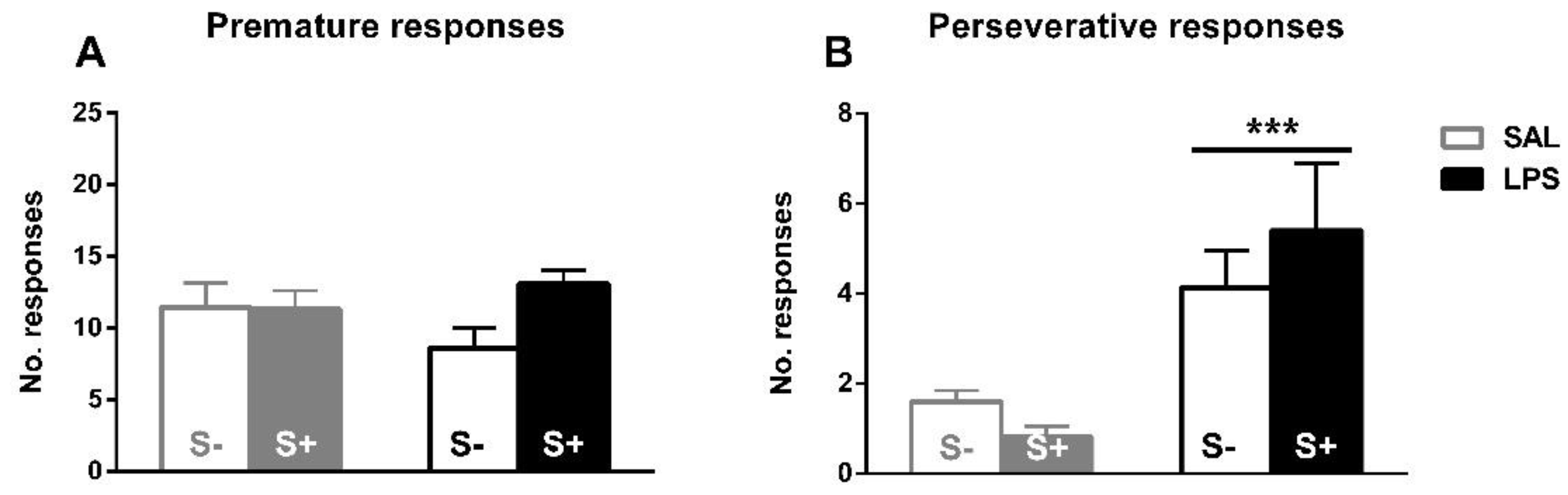
3.1.2. Inhibitory Control Assessment
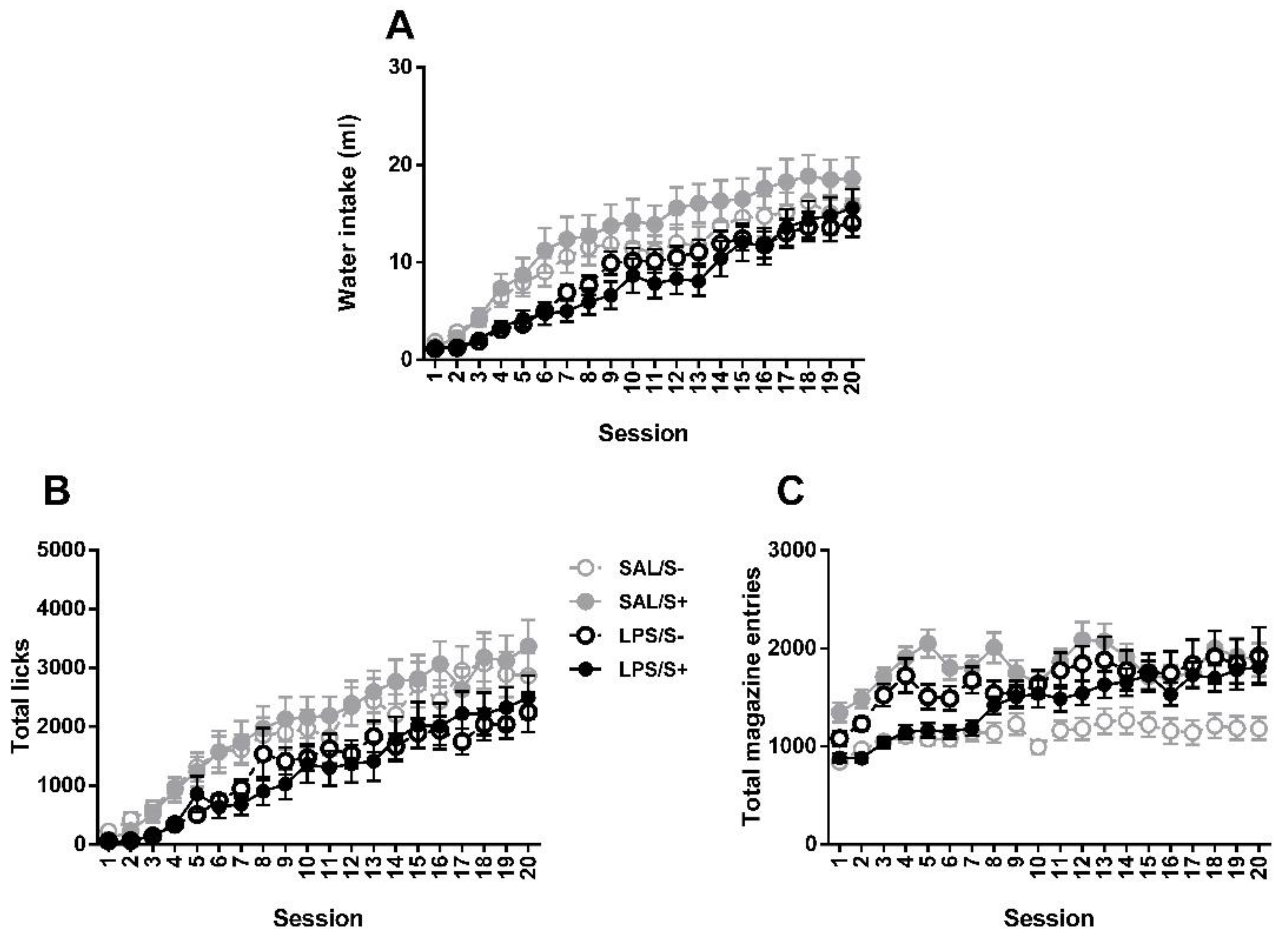
3.2. Schedule-Induced Polydipsia
3.3. Spontaneous Locomotor Activity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| SAL/S- | SAL/S | LPS/S- | LPS/S+ | |
|---|---|---|---|---|
| Latency to Correct | 54.1 ± 4.7 | 57.1 ± 3.8 | 56.4 ± 2.0 | 50.8 ± 2.9 |
| Latency to Incorrect | 25.0 ± 5.4 | 24.5 ± 3.1 | 20.8 ± 4.4 | 20.4 ± 4.6 |
| Latency to Reward | 72.4 ± 3.6 | 71.8 ± 4.9 | 70.2 ± 3.8 | 79.7 ± 6.1 |
References
- Bari, A.; Dalley, J.W.; Robbins, T.W. The application of the 5-choice serial reaction time task for the assessment of visual attentional processes and impulse control in rats. Nat. Protoc. 2008, 3, 759–767. [Google Scholar] [CrossRef]
- Fineberg, N.A.; Potenza, M.N.; Chamberlain, S.R.; Berlin, H.A.; Menzies, L.; Bechara, A.; Sahakian, B.J.; Robbins, T.W.; Bullmore, E.T.; Hollander, E. Probing compulsive and impulsive behaviors, from animal models to endophenotypes: A narrative review. Neuropsychopharmacology 2010, 35, 592–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robbins, T.W.; Gillan, C.M.; Smith, D.G.; de Wit, S.; Ersche, K.D. Neurocognitive endophenotypes of impulsivity and compulsivity: Towards dimensional psychiatry. Trends Cogn. Sci. 2012, 16, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Winstanley, C.A. The utility of rat models of impulsivity in developing pharmacotherapies for impulse control disorders. Br. J. Pharmacol. 2011, 164, 1301–1321. [Google Scholar] [CrossRef] [PubMed]
- Figee, M.; Pattij, T.; Willuhn, I.; Luigjes, J.; van den Brink, W.; Goudriaan, A.; Potenza, M.N.; Robbins, T.W.; Denys, D. Compulsivity in obsessive-compulsive disorder and addictions. Eur. Neuropsychopharmacol. 2016, 26, 856–868. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Press: Washington, DC, USA, 2013. [Google Scholar]
- Hollander, E.; Kim, S.; Khanna, S.; Pallanti, S. Obsessive-compulsive disorder and obsessive-compulsive spectrum disorders: Diagnostic and dimensional issues. CNS Spectr. 2007, 12, 5–13. [Google Scholar] [CrossRef]
- Robbins, T.W.; Crockett, M.J. Role of Central Serotonin in Impulsivity and Compulsivity: Comparative Studies in Experimental Animals and Humans. In Handbook of Behavioral Neuroscience; Müller, C., Jacobs, B., Eds.; Academic Press: London, UK, 2010. [Google Scholar]
- Van den Heuvel, O.A.; van Wingen, G.; Soriano-Mas, C.; Alonso, P.; Chamberlain, S.R.; Nakamae, T.; Denys, D.; Goudriaan, A.E.; Veltman, D.J. Brain circuitry of compulsivity. Eur. Neuropsychopharmacol. 2016, 26, 810–827. [Google Scholar] [CrossRef] [Green Version]
- Orefici, G.; Cardona, F.; Cox, C.J.; Cunningham, M.W. Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS). In Streptococcus Pyogenes: Basic Biology to Clinical Manifestations; University of Oklahoma Health Sciences Center: Oklahoma City, OK, USA, 2016. [Google Scholar] [CrossRef]
- Sigra, S.; Hesselmark, E.; Bejerot, S. Treatment of PANDAS and PANS: A systematic review. Neurosci. Biobehav. Rev. 2018, 86, 51–65. [Google Scholar] [CrossRef]
- Snider, L.A.; Swedo, S.E. PANDAS: Current status and directions for research. Mol. Psychiatry 2004, 9, 900–907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benros, M.E.; Eaton, W.W.; Mortensen, P.B. The epidemiologic evidence linking autoimmune diseases and psychosis. Biol. Psychiatry 2014, 75, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Mell, L.K.; Davis, R.L.; Owens, D. Association between streptococcal infection and obsessive-compulsive disorder, Tourette’s syndrome, and tic disorder. Pediatrics 2005, 116, 56–60. [Google Scholar] [CrossRef] [Green Version]
- Hirschtritt, M.E.; Hammond, C.J.; Luckenbaugh, D.; Buhle, J.; Thurm, A.E.; Casey, B.J.; Swedo, S.E. Executive and attention functioning among children in the PANDAS subgroup. Child. Neuropsychol. 2009, 15, 179–194. [Google Scholar] [CrossRef]
- Peterson, B.S.; Leckman, J.F.; Tucker, D.; Scahill, L.; Staib, L.; Zhang, H.; King, R.; Cohen, D.J.; Gore, J.C.; Lombroso, P. Preliminary findings of antistreptococcal antibody titers and basal ganglia volumes in tic, obsessive-compulsive, and attention deficit/hyperactivity disorders. Arch. Gen. Psychiatry 2000, 57, 364–372. [Google Scholar] [CrossRef] [Green Version]
- Sokol, M.S. Infection-triggered anorexia nervosa in children: Clinical description of four cases. J. Child. Adolesc. Psychopharmacol. 2000, 10, 133–145. [Google Scholar] [CrossRef]
- Sokol, M.S.; Ward, P.E.; Tamiya, H.; Kondo, D.G.; Houston, D.; Zabriskie, J.B. D8/17 expression on B lymphocytes in anorexia nervosa. Am. J. Psychiatry. 2002, 159, 1430–1432. [Google Scholar] [CrossRef]
- Leslie, D.L.; Kozma, L.; Martin, A.; Landeros, A.; Katsovich, L.; King, R.A.; Leckman, J.F. Neuropsychiatric disorders associated with streptococcal infection: A case-control study among privately insured children. J. Am. Acad. Child. Adolesc. Psychiatry 2008, 47, 1166–1172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hollander, E.; DelGiudice-Asch, G.; Simon, L.; Schmeidler, J.; Cartwright, C.; DeCaria, C.M.; Kwon, J.; Cunningham-Rundles, C.; Chapman, F.; Zabriskie, J.B. B lymphocyte antigen D8/17 and repetitive behaviors in autism. Am. J. Psychiatry 1999, 156, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Margutti, P.; Delunardo, F.; Ortona, E. Autoantibodies associated with psychiatric disorders. Curr. Neurovasc. Res. 2006, 3, 149–157. [Google Scholar] [CrossRef]
- Mora, S.; Martín-González, E.; Flores, P.; Moreno, M. Neuropsychiatric consequences of childhood group A streptococcal infection: A systematic review of preclinical models. Brain Behav. Immun. 2020, 86, 53–62. [Google Scholar] [CrossRef]
- Basta-Kaim, A.; Szczȩeny, E.; Leśkiewicz, M.; Głombik, K.; Slusarczyk, J.; Budziszewska, B.; Regulska, M.; Kubera, M.; Nowak, W.; Wędzony, K.; et al. Maternal immune activation leads to age-related behavioral and immunological changes in male rat offspring-The effect of antipsychotic drugs. Pharmacol. Rep. 2012, 64, 1400–1410. [Google Scholar] [CrossRef]
- Borrell, J.; Vela, J.M.; Arévalo-Martin, A.; Molina-Holgado, E.; Guaza, C. Prenatal immune challenge disrupts sensorimotor gating in adult rats: Implications for the etiopathogenesis of schizophrenia. Neuropsychopharmacology 2002, 26, 204–215. [Google Scholar] [CrossRef] [Green Version]
- Custódio, C.S.; Mello, B.S.F.; Filho, A.J.M.C.; de Carvalho Lima, C.N.; Cordeiro, R.C.; Miyajima, F.; Réus, G.Z.; Vasconcelos, S.M.M.; Barichello, T.; Quevedo, J.; et al. Neonatal Immune Challenge with Lipopolysaccharide Triggers Long-lasting Sex- and Age-related Behavioral and Immune/Neurotrophic Alterations in Mice: Relevance to Autism Spectrum Disorders. Mol. Neurobiol. 2017, 55, 3775–3788. [Google Scholar] [CrossRef] [PubMed]
- Fortier, M.E.; Luheshe, G.N.; Boksa, P. Effects of prenatal infection on prepulse inhibition in the rat depend on the nature of the infectious agent and the stage of pregnancy. Behav. Brain Res. 2007, 181, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Romero, E.; Guaza, C.; Castellano, B.; Borrell, J. Ontogeny of sensorimotor gating and immune impairment induced by prenatal immune challenge in rats: Implications for the etiopathology of schizophrenia. Mol. Psychiatry 2010, 15, 372–383. [Google Scholar] [CrossRef] [Green Version]
- Wischhof, L.; Irrsack, E.; Osorio, C.; Koch, M. Prenatal LPS-exposure-a neurodevelopmental rat model of schizophrenia-differentially affects cognitive functions, myelination and parvalbumin expression in male and female offspring. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2015, 57, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Babri, S.; Doosti, M.H.; Salari, A.A. Strain-dependent effects of prenatal maternal immune activation on anxiety- and depression-like behaviors in offspring. Brain Behav. Immun. 2014, 37, 164–176. [Google Scholar] [CrossRef]
- Enayati, M.; Solati, J.; Hosseini, M.H.; Shahi, H.R.; Saki, G.; Salari, A.A. Maternal infection during late pregnancy increases anxiety- and depression-like behaviors with increasing age in male offspring. Brain Res. Bull. 2012, 87, 295–302. [Google Scholar] [CrossRef]
- Tenk, C.M.; Kavaliers, M.; Ossenkopp, K.P. Neonatal treatment with lipopolysaccharide differentially affects adult anxiety responses in the light-dark test and taste neophobia test in male and female rats. Int. J. Dev. Neurosci. 2013, 31, 171–180. [Google Scholar] [CrossRef]
- De Cossío, L.F.; Fourrier, C.; Sauvant, J.; Everard, A.; Capuron, L.; Cani, P.D.; Layé, S.; Castanon, N. Impact of prebiotics on metabolic and behavioral alterations in a mouse model of metabolic syndrome. Brain Behav. Immun. 2017, 64, 33–49. [Google Scholar] [CrossRef]
- Fortunato, J.J.; da Rosa, N.; Martins Laurentino, A.O.; Goulart, M.; Michalak, C.; Borges, L.P.; da Cruz Cittadin Soares, E.; Reis, P.A.; de Castro Faria Neto, H.C.; Petronilho, F. Effects of ω-3 fatty acids on stereotypical behavior and social interactions in Wistar rats prenatally exposed to lipopolysaccarides. Nutrition 2017, 35, 119–127. [Google Scholar] [CrossRef]
- Kirsten, T.B.; Taricano, M.; Flório, J.C.; Palermo-Neto, J.; Bernardi, M.M. Prenatal lipopolysaccharide reduces motor activity after an immune challenge in adult male offspring. Behav. Brain Res. 2010, 211, 77–82. [Google Scholar] [CrossRef]
- Batinić, B.; Santrač, A.; Divović, B.; Timić, T.; Stanković, T.; Obradović ALj Joksimović, S.; Savić, M.M. Lipopolysaccharide exposure during late embryogenesis results in diminished locomotor activity and amphetamine response in females and spatial cognition impairment in males in adult, but not adolescent rat offspring. Behav. Brain Res. 2016, 299, 72–80. [Google Scholar] [CrossRef]
- Harvey, L.; Boksa, P. Do prenatal immune activation and maternal iron deficiency interact to affect neurodevelopment and early behavior in rat offspring? Brain Behav. Immun. 2014, 35, 144–154. [Google Scholar] [CrossRef]
- Litteljohn, D.; Rudyk, C.; Dwyer, Z.; Farmer, K.; Fortin, T.; Hayley, S.; Canadian Lrrk2 in Inflammation Team (CLINT). The impact of murine LRRK2 G2019S transgene overexpression on acute responses to inflammatory challenge. Brain Behav. Immun. 2018, 67, 246–256. [Google Scholar] [CrossRef]
- Straley, M.E.; Van Oeffelen, W.; Theze, S.; Sullivan, A.M.; O’Mahony, S.M.; Cryan, J.F.; O’Keeffe, G.W. Distinct alterations in motor & reward seeking behavior are dependent on the gestational age of exposure to LPS-induced maternal immune activation. Brain Behav. Immun. 2017, 63, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.H.; Ai, W.M.; Lei, D.L.; Luo, X.G.; Yan, X.X.; Li, Z. Lipopolysaccharide induces paired immunoglobulin-like receptor B (PirB) expression, synaptic alteration, and learning-memory deficit in rats. Neuroscience 2012, 209, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.K.; Hiles, S.A.; Sominsky, L.; McLaughlin, E.A.; Hodgson, D.M. Neonatal lipopolysaccharide exposure impairs sexual development and reproductive success in the Wistar rat. Brain Behav. Immun. 2011, 25, 674–684. [Google Scholar] [CrossRef] [PubMed]
- Kirsten, T.B.; Bernardi, M.M. Prenatal lipopolysaccharide induces hypothalamic dopaminergic hypoactivity and autistic-like behaviors: Repetitive self-grooming and stereotypies. Behav. Brain Res. 2017, 331, 25–29. [Google Scholar] [CrossRef]
- Mora, S.; Martín-González, E.; Prados-Pardo, Á.; Moreno, J.; López, M.J.; Pilar-Cuellar, F.; Castro, E.; Díaz, Á.; Flores, P.; Moreno, M. Increased vulnerability to impulsive behavior after streptococcal antigen exposure and antibiotic treatment in rats. Brain Behav. Immun. 2020, 89, 675–688. [Google Scholar] [CrossRef]
- Atrooz, F.; Liu, H.; Salim, S. Stress, psychiatric disorders, molecular targets, and more. Prog. Mol. Biol. Transl. Sci. 2019, 167, 77–105. [Google Scholar] [CrossRef]
- Fisher, H.L.; Craig, T.K.; Fearon, P.; Morgan, K.; Dazzan, P.; Lappin, J.; Hutchinson, G.; Doody, G.A.; Jones, P.B.; McGuffin, P.; et al. Reliability and comparability of psychosis patients’ retrospective reports of childhood abuse. Schizophr. Bull. 2010, 37, 546–553. [Google Scholar] [CrossRef] [Green Version]
- Giovanoli, S.; Engler, H.; Engler, A.; Richetto, J.; Voget, M.; Willi, R.; Winter, C.; Riva, M.A.; Mortensen, P.B.; Feldon, J.; et al. Stress in puberty unmasks latent neuropathological consequences of prenatal immune activation in mice. Science 2015, 339, 1095–1099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sengupta, P. The Laboratory Rat: Relating Its Age with Human’s. Int. J. Prev. Med. 2013, 4, 624–630. [Google Scholar] [PubMed]
- Ariza-Traslaviña, G.A.; de Oliveira, F.L.; Franci, C.R. Early adolescent stress alters behavior and the HPA axis response in male and female adult rats: The relevance of the nature and duration of the stressor. Physiol. Behav. 2015, 133, 178–189. [Google Scholar] [CrossRef]
- Moser, V.C. Comparisons of the acute effects of cholinesterase inhibitors using a neurobehavioral screening battery in rats. Neurotoxicol. Teratol. 1995, 17, 617–625. [Google Scholar] [CrossRef]
- Sánchez-Santed, F.; Cañadas, F.; Flores, P.; López-Grancha, M.; Cardona, D. Long-term functional neurotoxicity of paraoxon and chlorpyrifos: Behavioural and pharmacological evidence. Neurotoxicol. Teratol. 2004, 26, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Carli, M.; Robbins, T.W.; Evenden, J.L.; Everitt, B.J. Effects of lesions to ascending noradrenergic neurones on performance of a 5-choice serial reaction task in rats; implications for theories of dorsal noradrenergic bundle function based on selective attention and arousal. Behav. Brain Res. 1983, 9, 361–380. [Google Scholar] [CrossRef]
- Robbins, T.W. The 5-choice serial reaction time task: Behavioural pharmacology and functional neurochemistry. Psychopharmacology 2002, 163, 362–380. [Google Scholar] [CrossRef] [PubMed]
- Moreno, M.; Cardona, D.; Gómez, M.J.; Sánchez-Santed, F.; Tobeña, A.; Fernández-Teruel, A.; Campa, L.; Suñol, C.; Escarabajal, M.D.; Torres, C.; et al. Impulsivity characterization in the roman high-and low-avoidance rat strains: Behavioral and neurochemical differences. Neuropsychopharmacology 2010, 35, 1198–1208. [Google Scholar] [CrossRef]
- Moreno, M.; Gutiérrez-Ferre, V.E.; Ruedas, L.; Campa, L.; Suñol, C.; Flores, P. Poor inhibitory control and neurochemical differences in high compulsive drinker rats selected by schedule-induced polydipsia. Psychopharmacology 2012, 219, 661–672. [Google Scholar] [CrossRef]
- Dalley, J.W.; Lääne, K.; Pena, Y.; Theobald, D.E.; Everitt, B.J.; Robbins, T.W. Attentional and motivational deficits in rats withdrawn from intravenous self-administration of cocaine or heroin. Psychopharmacology 2005, 182, 579–587. [Google Scholar] [CrossRef]
- Jupp, B.; Pitzoi, S.; Petretto, E.; Mar, A.C.; Oliver, Y.P.; Jordan, E.R.; Taylor, S.; Atanur, S.S.; Srivastava, P.K.; Saar, K.; et al. Impulsivity is a heritable trait in rodents and associated with a novel quantitative trait locus on chromosome 1. Sci. Rep. 2020, 10, 6684. [Google Scholar] [CrossRef] [Green Version]
- López-Grancha, M.; Lopez-Crespo, G.; Sanchez-Amate, M.C.; Flores, P. Individual differences in schedule-induced polydipsia and the role of gabaergic and dopaminergic systems. Psychopharmacology 2008, 197, 487–498. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Kelley, K.W.; Bluthé, R.M.; Dantzer, R.; Zhou, J.H.; Shen, W.H.; Johnson, R.W.; Broussard, S.R. Cytokine-induced sickness behavior. Brain Behav. Imm. 2003, 17, 112–118. [Google Scholar] [CrossRef]
- Cossio-Bolaños, M.; Campos, R.G.; Vitoria, R.V.; Hochmuller Fogaça, R.T.; de Arruda, M. Reference curves for assessing the physical growth of male Wistar rats. Nutr. Hosp. 2013, 28, 2151–2156. [Google Scholar] [CrossRef] [PubMed]
- Bannert, E.; Tesch, T.; Kluess, J.; Frahm, J.; Kersten, S.; Kahlert, S.; Renner, L.; Rothkötter, H.J.; Dänicke, S. Metabolic and hematological consequences of dietary deoxynivalenol interacting with systemic Escherichia coli lipopolysaccharide. Toxins 2015, 7, 4773–4796. [Google Scholar] [CrossRef] [Green Version]
- IUPS Thermal Commission. Glossary of terms for thermal physiology. Pflug. Arch. 1987, 410, 567–587. [Google Scholar] [CrossRef] [PubMed]
- Mackowiak, P.A. Concepts of fever. Arch. Intern. Med. 1998, 158, 1870–1881. [Google Scholar] [CrossRef] [Green Version]
- Roth, J.; Blatteis, C.M. Mechanisms of fever production and lysis: Lessons from experimental LPS fever. Compr. Physiol. 2014, 4, 1563–1604. [Google Scholar] [CrossRef] [PubMed]
- Tesch, T.; Bannert, E.; Kluess, J.; Frahm, J.; Kersten, S.; Breves, G.; Renner, L.; Kahlert, S.; Rothkötter, H.J.; Dänicke, S. Does dietary deoxynivalenol modulate the acute phase reaction in endotoxaemic pigs?—Lessons from clinical signs, white blood cell counts, and TNF-Alpha. Toxins 2016, 8, 3. [Google Scholar] [CrossRef]
- Tesch, T.; Bannert, E.; Kluess, J.; Frahm, J.; Hüther, L.; Kersten, S.; Breves, G.; Renner, L.; Kahlert, S.; Rothkötter, H.J.; et al. Relationships between body temperatures and inflammation indicators under physiological and pathophysiological conditions in pigs exposed to systemic lipopolysaccharide and dietary deoxynivalenol. J. Anim. Physiol. Anim. Nutr. 2017, 102, 241–251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dantzer, R. Cytokine-induced sickness behavior: Mechanisms and implications. Ann. N. Y. Acad. Sci. 2001, 933, 222–234. [Google Scholar] [CrossRef]
- Tansey, E.A.; Johnson, C.D. Recent advances in thermoregulation. Adv. Physiol. Educ. 2015, 39, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Dalley, J.W.; Everitt, B.J.; Robbins, T.W. Impulsivity, Compulsivity, and Top-Down Cognitive Control. Neuron 2011, 69, 680–694. [Google Scholar] [CrossRef] [Green Version]
- Kohl, S.; Heekeren, K.; Klosterkötter, J.; Kuhn, J. Prepulse inhibition in psychiatric disorders--apart from schizophrenia. J. Psychiatr. Res. 2013, 47, 445–452. [Google Scholar] [CrossRef]
- Thompson, S.L.; Welch, A.C.; Iourinets, J.; Dulawa, S.C. Ketamine induces immediate and delayed alterations of OCD-like behavior. Psychopharmacology 2020, 237, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Yaddanapudi, K.; Hornig, M.; Serge, R.; De Miranda, J.; Baghban a Villar, G.; Lipkin, W.I. Passive transfer of streptococcus-induced antibodies reproduces behavioral disturbances in a mouse model of pediatric autoimmune neuropsychiatric disorders associated with streptococcal infection. Mol. Psychiatry 2010, 15, 712–726. [Google Scholar] [CrossRef]
- Brimberg, L.; Benhar, I.; Mascaro-Blanco, A.; Alvarez, K.; Lotan, D.; Winter, C.; Klein, J.; Moses, A.E.; Somnier, F.E.; Leckman, J.F.; et al. Behavioral, pharmacological, and immunological abnormalities after streptococcal exposure: A novel rat model of Sydenham chorea and related neuropsychiatric disorders. Neuropsychopharmacology 2012, 37, 2076–2087. [Google Scholar] [CrossRef]
- Lotan, D.; Cunningham, M.; Joel, D. Antibiotic treatment attenuates behavioral and neurochemical changes induced by exposure of rats to group a streptococcal antigen. PLoS ONE 2014, 9, e101257. [Google Scholar] [CrossRef]
- Zhang, D.; Patel, A.; Zhu, Y.; Siegel, A.; Zalcman, S.S. Anti-streptococcus IgM antibodies induce repetitive stereotyped movements: Cell activation and co-localization with Fcalpha/mu receptors in the striatum and motor cortex. Brain Behav. Immun. 2012, 26, 521–533. [Google Scholar] [CrossRef] [Green Version]
- Holloway, T.; Moreno, J.L.; Umali, A.; Rayannavar, V.; Hodes, G.E.; Russo, S.J.; González-Maeso, J. Prenatal stress induces schizophrenia-like alterations of serotonin 2A and metabotropic glutamate 2 receptors in the adult offspring: Role of maternal immune system. J. Neurosci. 2013, 33, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Moreno, M.; Flores, P. Schedule-induced polydipsia as a model of compulsive behavior: Neuropharmacological and neuroendocrine bases. Psychopharmacology 2012, 219, 647–659. [Google Scholar] [CrossRef]
- Cardona, D.; López-Grancha, M.; López-Crespo, G.; Nieto-Escamez, F.; Sánchez-Santed, F.; Flores, P. Vulnerability of long-term neurotoxicity of chlorpyrifos: Effect on schedule-induced polydipsia and a delay discounting task. Psychopharmacology 2006, 189, 47–57. [Google Scholar] [CrossRef]
- Cardona, D.; López-Crespo, G.; Sánchez-Amate, M.C.; Flores, P.; Sánchez-Santed, F. Impulsivity as long-term sequelae after chlorpyrifos intoxication: Time course and individual differences. Neurotox. Res. 2011, 19, 128–137. [Google Scholar] [CrossRef] [PubMed]
- DeCarolis, N.A.; Myracle, A.; Erbach, J.; Glowa, J.; Flores, P.; Riley, A.L. Strain-dependent differences in schedule-induced polydipsia: An assessment in Lewis and Fischer rats. Pharmacol. Biochem. Behav. 2003, 74, 755–763. [Google Scholar] [CrossRef]
- Ibias, J.; Pellón, R. Schedule-induced polydipsia in the spontaneously hypertensive rat and its relation to impulsive behaviour. Behav. Brain Res. 2011, 223, 58–69. [Google Scholar] [CrossRef]
- Stöhr, T.; Szuran, T.; Welzl, H.; Pliska, V.; Feldon, J.; Pryce, C.R. Lewis/Fischer rat strain differences in endocrine and behavioural responses to environmental challenge. Pharmacol. Biochem. Behav. 2000, 67, 809–819. [Google Scholar] [CrossRef]
- Merchán, A.; Mora, S.; Gago, B.; Rodriguez-Ortega, E.; Fernández-Teruel, A.; Puga, J.L.; Sánchez-Santed, F.; Moreno, M.; Flores, P. Excessive habit formation in schedule-induced polydipsia: Microstructural analysis of licking among rat strains and involvement of the orbitofrontal cortex. Genes. Brain Behav. 2018, 18, e12489. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, K.L.; Hornig, M.; Yaddanapudi, K.; Jabado, O.; Lipkin, W.I. A murine model for neuropsychiatric disorders associated with group A beta-hemolytic streptococcal infection. J. Neurosci 2004, 24, 1780–1791. [Google Scholar] [CrossRef]
- Macri, S.; Ceci, C.; Onori, M.P.; Invernizzi, R.W.; Bartolini, E.; Altabella, L.; Canese, R.; Imperi, M.; Orefici, G.; Creti, R.; et al. Mice repeatedly exposed to Group-A beta-Haemolytic Streptococcus show perseverative behaviors, impaired sensorimotor gating, and immune activation in rostral diencephalon. Sci. Rep. 2015, 5, 13257. [Google Scholar] [CrossRef] [Green Version]
- Bernstein, G.A.; Victor, A.M.; Pipal, A.J.; Williams, K.A. Comparison of clinical characteristics of pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections and childhood obsessive-compulsive disorder. J. Child. Adolesc. Psychopharmacol. 2010, 20, 333–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shulman, S.T. Pediatric autoimmune neuropsychiatric disorders associated with streptococci (PANDAS). Pediatr. Infect. Dis. J. 1999, 18, 281–282. [Google Scholar] [CrossRef]
- Flagel, S.B.; Robinson, T.E.; Clark, J.J.; Clinton, S.M.; Watson, S.J.; Seeman, P.; Phillips, P.E.; Akil, H. An animal model of genetic vulnerability to behavioral disinhibition and responsiveness to reward-related cues: Implications for addiction. Neuropsychopharmacology 2010, 35, 388–400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qian, Y.; Lei, G.; Castellanos, F.X.; Forssberg, H.; Heijtz, R.D. Deficits in fine motor skills in a genetic animal model of ADHD. Behav. Brain Funct. 2010, 6, 51. [Google Scholar] [CrossRef] [Green Version]
- Sagvolden, T.; Johansen, E.B.; Wøien, G.; Walaas, S.I.; Storm-Mathisen, J.; Bergersen, L.H.; Hvalby, O.; Jensen, V.; Aase, H.; Russell, V.A.; et al. The spontaneously hypertensive rat model of ADHD--the importance of selecting the appropriate reference strain. Neuropharmacology 2009, 57, 619–626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cordero, M.I.; Merino, J.J.; Sandi, C. Correlational relationship between shock intensity and corticosterone secretion on the establishment and subsequent expression of contextual fear conditioning. Behav. Neurosci. 1998, 112, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Kang, L.; Yao, H.; He, Y.; Wang, X.; Xu, W.; Song, Z.; Yin, Y.; Zhang, X. Streptococcus pneumoniae Endopeptidase O (PepO) Elicits a Strong Innate Immune Response in Mice via TLR2 and TLR4 Signaling Pathways. Front. Cell. Infect. Microbiol. 2016, 6, 23. [Google Scholar] [CrossRef]
- Hachiya, Y.; Miyata, R.; Tanuma, N.; Hongou, K.; Tanaka, K.; Shimoda, K.; Kanda, S.; Hoshino, A.; Hanafusa, Y.; Kumada, S.; et al. Autoimmune neurological disorders associated with group-A beta-hemolytic streptococcal infection. Brain Dev. 2013, 35, 670–674. [Google Scholar] [CrossRef]
- Walls, A.; Cubangbang, M.; Wang, H.; Raiji, M.; Knight, J.; Steehler, M.; Latimer, E.; Harley, E.H., Jr. Pediatric Autoimmune Neuropsychiatric Disorder Associated with Streptococcus Immunology: A Pilot Study. Otolaryngol. Head. Neck. Surg. 2015, 153, 130–136. [Google Scholar] [CrossRef]
- Kim, H.-S.; Park, H.; Cho, I.-Y.; Paik, H.-D.; Park, E. Dietary supplementation of probiotic Bacillus polyfermenticus, Bispan strain, modulates natural killer cell and T cell subset populations and immunoglobulin G levels in human subjects. J. Med. Food 2006, 9, 321–327. [Google Scholar] [CrossRef]
- Murphy, T.K.; Kurlan, R.; Leckman, J. The immunobiology of Tourette’s disorder, pediatric autoimmune neuropsychiatric disorders associated with Streptococcus, and related disorders: A way forward. J. Child. Adolesc. Psychopharmacol. 2010, 20, 317–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]

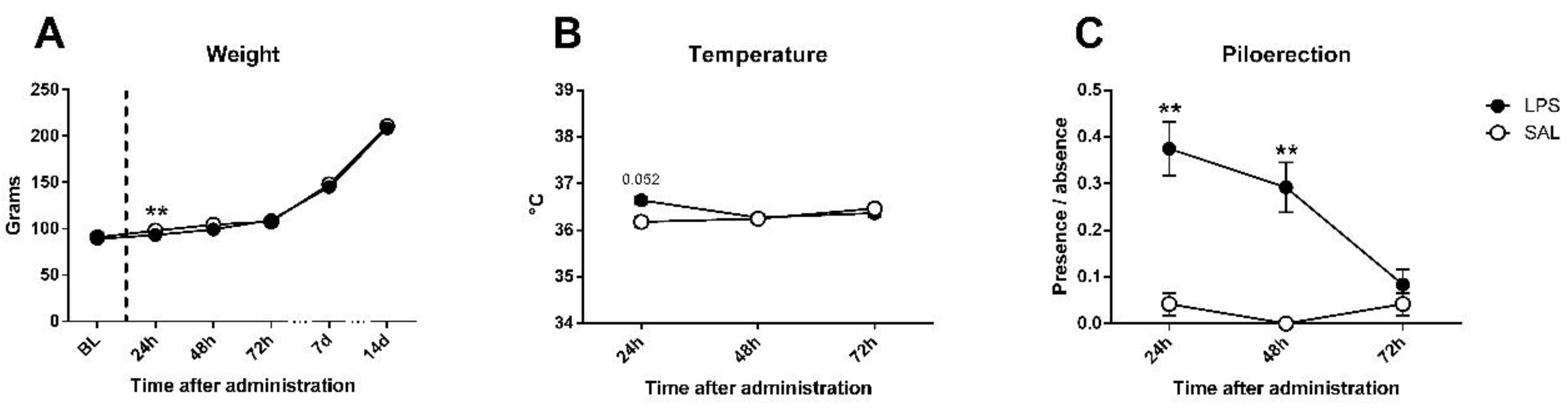




| SAL/S- | SAL/S+ | LPS/S- | LPS/S+ | |
|---|---|---|---|---|
| Weight 24 h | 98.0 ± 0.87 | 98.16 ± 0.8 | 93.5 ± 1.04 | 92.91 ± 0.87 |
| 48 h | 105.0 ± 0.7 | 103.75 ± 0.74 | 96.25 ± 3.47 | 102.16 ± 0.73 |
| 72 h | 104.75 ± 4.88 | 111.0 ± 0.89 | 108.58 ± 1.0 | 110.33 ± 0.89 |
| 7 d | 148.08 ± 1.12 | 147.75 ± 1.22 | 145.08 ± 1.17 | 145.0 ± 1.09 |
| 14 d | 211.5 ± 1.43 | 210.08 ± 1.53 | 209.08 ± 1.9 | 207.83 ± 1.61 |
| Temperature 24 h | 35.65 ± 0.13 | 36.7 ± 0.11 | 36.48 ± 0.1 | 36.8 ± 0.11 |
| 48 h | 35.92 ± 0.08 | 36.56 ± 0.09 | 36.54 ± 0.08 | 36.0 ± 0.08 |
| 72 h | 36.45 ± 0.07 | 36.46 ± 0.09 | 36.72 ± 0.08 | 6.0 ± 0.07 |
| Piloerection 24 h | 0.08 ± 0.04 | 0.0 ± 0.0 | 0.16 ± 0.06 | 0.58 ± 0.08 |
| 48 h | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.25 ± 0.07 | 0.33 ± 0.08 |
| 72 h | 0.0 ± 0.0 | 0.08 ± 0.04 | 0.08 ± 0.04 | 0.08 ± 0.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mora, S.; Martín-González, E.; Prados-Pardo, Á.; Flores, P.; Moreno, M. Increased Compulsivity in Adulthood after Early Adolescence Immune Activation: Preclinical Evidence. Int. J. Environ. Res. Public Health 2021, 18, 4684. https://doi.org/10.3390/ijerph18094684
Mora S, Martín-González E, Prados-Pardo Á, Flores P, Moreno M. Increased Compulsivity in Adulthood after Early Adolescence Immune Activation: Preclinical Evidence. International Journal of Environmental Research and Public Health. 2021; 18(9):4684. https://doi.org/10.3390/ijerph18094684
Chicago/Turabian StyleMora, Santiago, Elena Martín-González, Ángeles Prados-Pardo, Pilar Flores, and Margarita Moreno. 2021. "Increased Compulsivity in Adulthood after Early Adolescence Immune Activation: Preclinical Evidence" International Journal of Environmental Research and Public Health 18, no. 9: 4684. https://doi.org/10.3390/ijerph18094684





