Novel Options to Counteract Oral Biofilm Formation: In Vitro Evidence
Abstract
:1. Introduction
2. Materials and Methods
2.1. Compounds
2.2. Microbial Strains and Growth Condition
2.3. Assessment of MicroR and PomeGr Effects on Microbial Biofilm Formation
2.4. Statistical Analysis
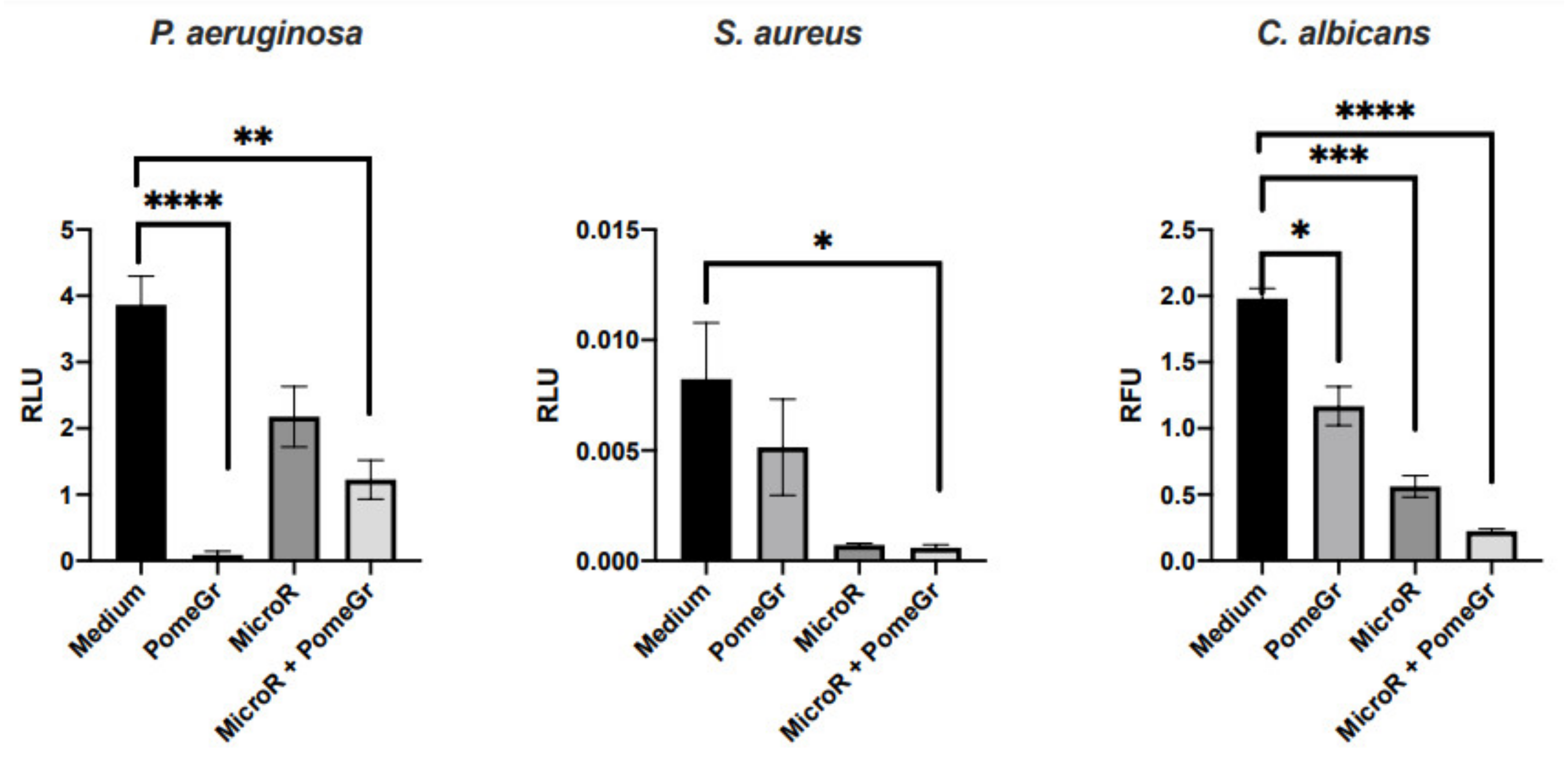
3. Results
Antibiofilm Efficacy of MicroR and/or PomeGr
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wade, W.G. The oral microbiome in health and disease. Pharmacol. Res. 2013, 69, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Butera, A.; Pascadopoli, M.; Gallo, S.; Lelli, M.; Tarterini, F.; Giglia, F.; Scribante, A. SEM/EDS Evaluation of the Mineral Deposition on a Polymeric Composite Resin of a Toothpaste Containing Biomimetic Zn-Carbonate Hydroxyapatite (microRepair®) in Oral Environment: A Randomized Clinical Trial. Polymers 2021, 13, 2740. [Google Scholar] [CrossRef] [PubMed]
- Meto, A.; Colombari, B.; Odorici, A.; Giva, L.B.; Pericolini, E.; Regina, A.L.; Blasi, E. Antibacterial Effects of MicroRepair®BIOMA-Based Toothpaste and Chewing Gum on Orthodontic Elastics Contaminated In Vitro with Saliva from Healthy Donors: A Pilot Study. Appl. Sci. 2020, 10, 6721. [Google Scholar] [CrossRef]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277–283. [Google Scholar]
- World Health Organization (WHO). Antimicrobial Resistance. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance (accessed on 12 December 2021).
- Chen, X.; Daliri, E.B.; Kim, N.; Kim, J.R.; Yoo, D.; Oh, D.H. Microbial etiology and prevention of dental caries: Exploiting natural products to inhibit cariogenic biofilms. Pathogens 2020, 9, 569. [Google Scholar] [CrossRef] [PubMed]
- Al-Zoreky, N.S. Antimicrobial activity of pomegranate (Punica granatum L.) fruit peels. Int. J. Food Microbiol. 2009, 134, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, P.R. Pomegranate (Punica granatum L). Ancient seeds for modern cure? Review of potential therapeutic applications. Int. J. Nutr. Pharmacol. Neurol. Dis. 2012, 2, 171–184. [Google Scholar] [CrossRef]
- Rodríguez-Rentería, M.; Márquez-Preciado, R.; Ortiz-Magdaleno, M.; Bermeo-Escalona, J.; Sánchez-Vargas, L.O. Frequency of Pathogenic Microorganisms in Removable Orthodontic Appliances and Oral Mucosa in Children. J. Clin. Pediatr. Dent. 2021, 45, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Ciofu, O.; Moser, C.; Jensen, P.Ø.; Høiby, N. Tolerance and resistance of microbial biofilms. Nat. Rev. Microbiol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Nord, C.E.; Sjöberg, L.; Wadström, T. Pseudomonas aeruginosa in oral infections. Acta Odontol. Scand. 1972, 30, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, P.V.A.; Amêndola, I.; Oliveira, L.D.; Silva, C.R.G.E.; Leão, M.V.P.; Santos, S.S.F.D. Prevalence and Sensitivity of Bacilli and Pseudomonas in the Newborn’s Oral Cavity. Braz. Dent. J. 2017, 28, 423–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koukos, G.; Sakellari, D.; Arsenakis, M.; Tsalikis, L.; Slini, T.; Konstantinidis, A. Prevalence of Staphylococcus aureus and methicillin resistant Staphylococcus aureus (MRSA) in the oral cavity. Arch. Oral Biol. 2015, 60, 1410–1415. [Google Scholar] [CrossRef] [PubMed]
- Bertolini, M.; Ranjan, A.; Thompson, A.; Diaz, P.I.; Sobue, T.; Maas, K.; Dongari-Bagtzoglou, A. Candida albicans induces mucosal bacterial dysbiosis that promotes invasive infection. PLoS Pathog. 2019, 15, e1007717. [Google Scholar] [CrossRef] [PubMed]
- Colombari, B.; Alfano, G.; Gamberini, C.; Cappelli, G.; Blasi, E. EDTA and Taurolidine Affect Pseudomonas aeruginosa Virulence In Vitro-Impairment of Secretory Profile and Biofilm Production onto Peritoneal Dialysis Catheters. Microbiol. Spectr. 2021, 9, e0104721. [Google Scholar] [CrossRef] [PubMed]
- Meto, A.; Colombari, B.; Sala, A.; Pericolini, E.; Meto, A.; Peppoloni, S.; Blasi, E. Antimicrobial and antibiofilm efficacy of a copper/calcium hydroxide-based endodontic paste against Staphylococcus aureus, Pseudomonas aeruginosa and Candida albicans. Dent. Mater. J. 2019, 38, 591–603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krzyściak, W.; Jurczak, A.; Kościelniak, D.; Bystrowska, B.; Skalniak, A. The virulence of Streptococcus mutans and the ability to form biofilms. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 499–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Treatments | Biofilm Decrease (%) | ||
|---|---|---|---|
| P. aeruginosa | S. aureus | C. albicans | |
| PomeGr | 98 | 37.5 | 41 |
| MicroR | 43 | 91 | 72 |
| MicroR + PomeGr | 68 | 93 | 89 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Odorici, A.; Colombari, B.; Bellini, P.; Meto, A.; Venturelli, I.; Blasi, E. Novel Options to Counteract Oral Biofilm Formation: In Vitro Evidence. Int. J. Environ. Res. Public Health 2022, 19, 8056. https://doi.org/10.3390/ijerph19138056
Odorici A, Colombari B, Bellini P, Meto A, Venturelli I, Blasi E. Novel Options to Counteract Oral Biofilm Formation: In Vitro Evidence. International Journal of Environmental Research and Public Health. 2022; 19(13):8056. https://doi.org/10.3390/ijerph19138056
Chicago/Turabian StyleOdorici, Alessandra, Bruna Colombari, Pierantonio Bellini, Aida Meto, Irene Venturelli, and Elisabetta Blasi. 2022. "Novel Options to Counteract Oral Biofilm Formation: In Vitro Evidence" International Journal of Environmental Research and Public Health 19, no. 13: 8056. https://doi.org/10.3390/ijerph19138056








