Can Intra-Oral Qualitative Sensory Testing Foretell Postoperative Dental Pain? A Preliminary Report
Abstract
:1. Introduction
2. Materials and Methods
2.1. Population
2.2. Tools
2.2.1. Assessment of Dental Anxiety
2.2.2. Intra-Oral Thermal Test (IT-QualST)
- On the treated side, immediately prior to the planned restorative treatment. Patients rated the sensation of pain (Pain-treated1), sensation of cold (Cold-treated1), and duration (in seconds) of the cold sensation (Time-treated1).
- On the contralateral (CL) side, immediately prior to treatment. Patients rated the sensation of pain (Pain-CL1), sensation of cold (Cold-CL1), and duration (in seconds) of the cold sensation (Time-CL1).
- On the treated side at a follow-up meeting, 1–3 weeks later. Patients rated the sensation of pain (Pain-treated2), sensation of cold (Cold-treated2), and duration (in seconds) of the cold sensation (Time-treated2).
- On the contralateral side at a follow-up meeting. Patients rated the sensation of pain (Pain-CL2), sensation of cold (Cold-CL2), and duration (in seconds) of the cold sensation (Time-CL2).
2.2.3. Postoperative Dental Pain (PDP), Evaluated through Numeric Rating Scales (NRSs)
- What is the level of pain you feel at the treated side at the moment? (NRS ranging from 0—no pain whatsoever to 10—unbearable pain). Results were marked as: 24currentPDP, 48currentPDP, and 72currentPDP.
- What was the level of the worst pain you experienced in the treated area in the last 24 h? (NRS as above). Results were marked as: 24worstPDP, 48worstPDP, and 72worstPDP.
2.3. Statistical Analyses
3. Results
3.1. Descriptive Data
3.2. IT-QualST-Pain, Cold, and Cold Duration Assessments
- (i)
- Pain (Pain-treated1, Pain-CL1, Pain-treated2, Pain-CL2): Cronbach alpha = 0.84.
- (ii)
- Cold (Cold-treated1, Cold-CL1, Cold-treated2, Cold-CL2): Cronbach alpha = 0.93.
- (iii)
- Cold duration (Time-treated1, Time-CL1, Time-treated2, Time-CL2): Pearson correlation coefficients ranging 0.75–0.80.
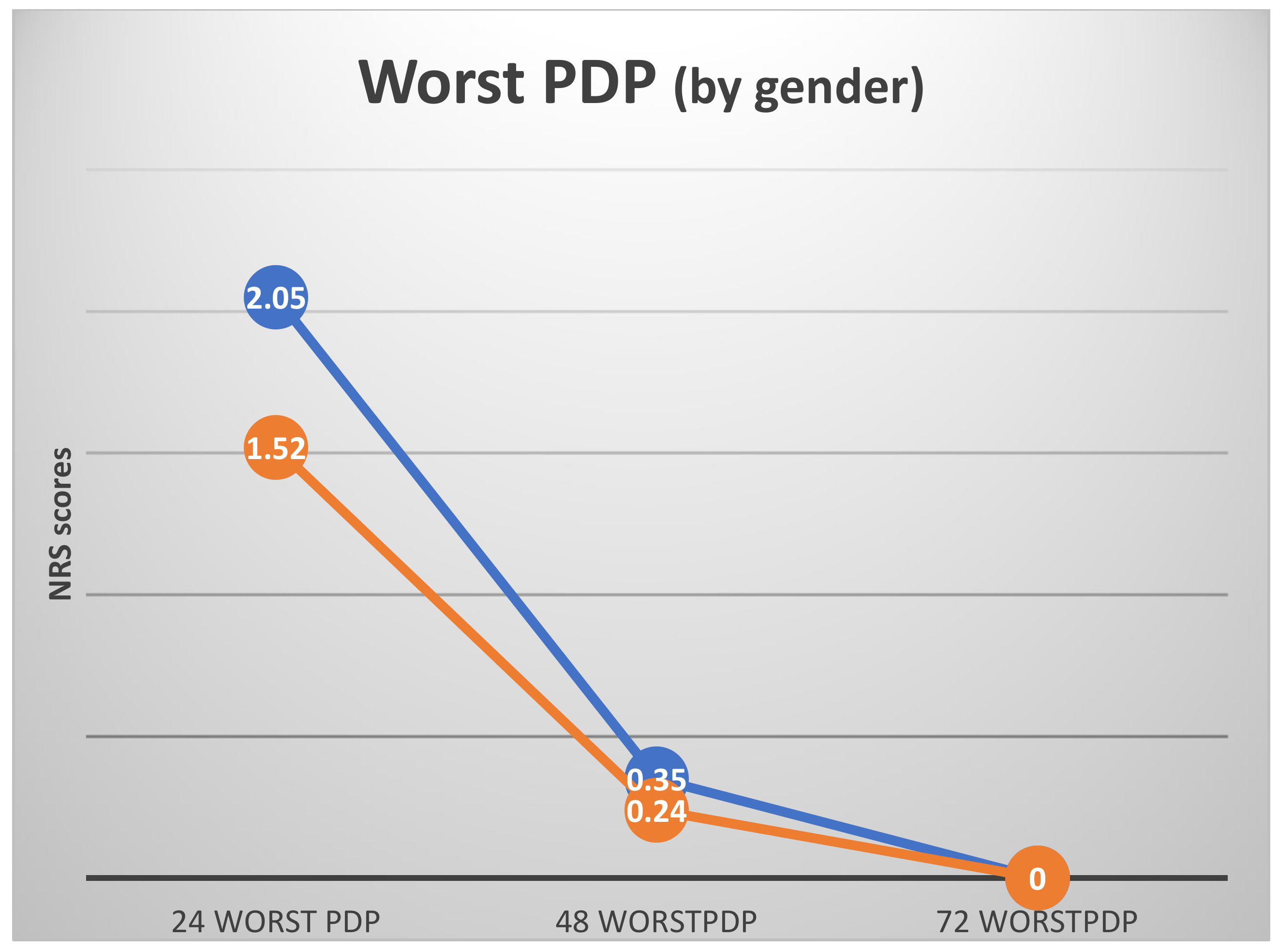
3.3. Postoperative Dental Pain (PDP)
3.4. Stepwise Linear Regressions Analyses
4. Discussion
5. Study Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhatti, U.A. The phenomenon of postoperative sensitivity and composite restorations—A review. J. Pak. Dent. Assoc. 2019, 28, 33–40. [Google Scholar] [CrossRef]
- Al-Omari, Q.D.; Al-Omari, W.M.; Omar, R. Factors associated with postoperative sensitivity of amalgam restorations. J. Ir. Dent. Assoc. 2009, 55, 87–91. [Google Scholar] [PubMed]
- Johnson, G.H.; Gordon, G.E.; Bales, D.J. Postoperative sensitivity associated with posterior composite and amalgam restorations. Oper. Dent. 1988, 13, 66–73. [Google Scholar] [PubMed]
- Ashkenazi, M.; Blumer, S.; Eli, I. Post-operative pain and use of analgesic agents in children following intrasulcular anaesthesia and various operative procedures. Br. Dent. J. 2007, 202, E13. [Google Scholar] [CrossRef]
- Isabel, C.C. Post-Operative sensitivity in direct resin composite restorations: Clinical practice guidelines. IJRD 2012, 1, 1–12. Available online: https://www.academia.edu/28415706/Post_operative_sensitivity_in_direct_resin_composite_restorations_Clinical_practice_guidelines (accessed on 23 July 2021).
- González-Roldán, A.M.; Terrasa, J.L.; Sitges, C.; van der Meulen, M.; Anton, F.; Montoya, P. Age-related changes in pain perception are associated with altered functional connectivity during resting state. Front. Aging Neurosci. 2020, 12, 116. [Google Scholar] [CrossRef]
- Ip, H.Y.; Abrishami, A.; Peng, P.W.; Wong, J.; Chung, F. Predictors of postoperative pain and analgesic consumption: A qualitative systematic review. Anesthesiology 2009, 111, 657–677. [Google Scholar] [CrossRef] [Green Version]
- Raja, S.N.; Carr, D.B.; Cohen, M.; Finnerup, N.B.; Flor, H.; Gibson, S.; Keefe, F.J.; Mogil, J.S.; Ringkamp, M.; Sluka, K.A.; et al. The revised International Association for the Study of Pain definition of pain: Concepts, challenges, and compromises. Pain 2020, 161, 1976–1982. [Google Scholar] [CrossRef]
- Zhou, P.; Chen, Y.; Zhang, J.; Wang, K.; Svensson, P. Quantitative sensory testing for assessment of somatosensory function in human oral mucosa: A review. Acta Odontol. Scand. 2018, 76, 13–20. [Google Scholar] [CrossRef]
- Jacobs, R.; Wu, C.H.; Goossens, K.; Van Loven, K.; Van Hees, J.; Van Steenberghe, D. Oral mucosal versus cutaneous sensory testing: A review of the literature. J. Oral Rehabil. 2002, 29, 923–950. [Google Scholar] [CrossRef]
- Hansson, P.; Backonja, M.; Bouhassira, D. Usefulness and limitations of quantitative sensory testing: Clinical and research application in neuropathic pain states. Pain 2007, 129, 256–259. [Google Scholar] [CrossRef]
- Zagury, J.G.; Eliav, E.; Heir, G.M.; Nasri-Heir, C.; Ananthan, S.; Pertes, R.; Sharav, Y.; Benoliel, R. Prolonged gingival cold allodynia: A novel finding in patients with atypical odontalgia. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2011, 111, 312–319. [Google Scholar] [CrossRef]
- Cruz-Almeida, Y.; Fillingim, R.B. Can quantitative sensory testing move us closer to mechanism-based pain management? Pain Med. 2014, 15, 61–72. [Google Scholar] [CrossRef] [Green Version]
- Rolke, R.; Baron, R.; Maier, C.; Tölle, T.R.; Treede, R.-D.; Beyer, A.; Binder, A.; Birbaumer, N.; Birklein, F.; Bötefür, I.C.; et al. Quantitative sensory testing in the German Research Network on Neuropathic Pain (DFNS): Standardized protocol and reference values. Pain 2006, 123, 231–243, Erratum in Pain 2006, 125, 197. [Google Scholar] [CrossRef]
- Svensson, P.; Baad-Hansen, L.; Pigg, M.; List, T.; Eliav, E.; Ettlin, D.; Michelotti, A.; Tsukiyama, Y.; Matsuka, Y.; Jääskeläinen, S.K.; et al. Guidelines and recommendations for assessment of somatosensory function in oro-facial pain conditions—A taskforce report. J. Oral Rehabil. 2011, 38, 366–394. [Google Scholar] [CrossRef] [Green Version]
- Pigg, M.; Baad-Hansen, L.; Svensson, P.; Drangsholt, M.; List, T. Reliability of intraoral quantitative sensory testing (QST). Pain 2010, 148, 220–226. [Google Scholar] [CrossRef] [Green Version]
- Baad-Hansen, L.; Pigg, M.; Ivanovic, S.E.; Faris, H.; List, T.; Drangsholt, M.; Svensson, P. Chairside intraoral qualitative somatosensory testing: Reliability and comparison between patients with atypical odontalgia and healthy controls. J. Orofac. Pain. 2013, 27, 165–170. [Google Scholar] [CrossRef]
- Nasri-Heir, C.; Khan, J.; Benoliel, R.; Feng, C.; Yarnitsky, D.; Kuo, F.; Hirschberg, C.; Hartwell, G.; Huang, C.-Y.; Heir, G.; et al. Altered pain modulation in patients with persistent postendodontic pain. Pain 2015, 156, 2032–2041. [Google Scholar] [CrossRef] [Green Version]
- Corah, N.L. Development of a dental anxiety scale. J. Dent. Res. 1969, 48, 596. [Google Scholar] [CrossRef]
- Corah, N.L.; Gale, E.N.; Illig, S.J. Assessment of a dental anxiety scale. J. Am. Dent. Assoc. 1978, 97, 816–819. [Google Scholar] [CrossRef]
- Dailey, Y.M.; Humphris, G.M.; Lennon, M.A. The use of dental anxiety questionnaires: A survey of a group of UK dental practitioners. Br. Dent. J. 2001, 190, 450–453. [Google Scholar] [CrossRef] [Green Version]
- Höglund, M.; Bågesund, M.; Shahnavaz, S.; Wårdh, I. Evaluation of the ability of dental clinicians to rate dental anxiety. Eur. J. Oral Sci. 2019, 127, 455–461. [Google Scholar] [CrossRef] [Green Version]
- Amir, A.; Kamate, S.; Gupta, P.; Gupta, A.; Singh, J.; Singh, S. Assessment of dental anxiety using MDAS (Modified Dental Anxiety Scale) among students in bareilly city—A cross sectional study. Int. J. Contemp. Med. Res. 2018, 5, 2454–7379. [Google Scholar]
- Newton, J.T.; Buck, D.J. Anxiety and pain measures in dentistry: A guide to their quality and application. J. Am. Dent. Assoc. 2000, 131, 1449–1457. [Google Scholar] [CrossRef]
- Kassem El Hajj, H.; Fares, Y.; Abou-Abbas, L. Assessment of dental anxiety and dental phobia among adults in Lebanon. BMC Oral Health 2021, 21, 48. [Google Scholar] [CrossRef]
- Appukuttan, D.P. Strategies to manage patients with dental anxiety and dental phobia: Literature review. Clin. Cosmet. Investig. Dent. 2016, 8, 35–50. [Google Scholar] [CrossRef] [Green Version]
- Nicolas, E.; Collado, V.; Faulks, D.; Bullier, B.; Hennequin, M. A national cross-sectional survey of dental anxiety in the French adult population. BMC Oral Health 2007, 7, 12. [Google Scholar] [CrossRef] [Green Version]
- Uziel, N.; Bronner, G.; Elran, E.; Eli, I. Sexual correlates of gagging and dental anxiety. Community Dent Health 2012, 29, 243–247. [Google Scholar]
- Peretz, B.; Zadik, D. Dental anxiety of parents in an Israeli kibbutz population. Int. J. Paediatr. Dent. 1994, 4, 87–92. [Google Scholar] [CrossRef]
- Stabholz, A.; Peretz, B. Dental anxiety among patients prior to different dental treatments. Int. Dent. J. 1999, 49, 90–94. [Google Scholar] [CrossRef]
- Ratson, T.; Blumer, S.; Peretz, B. Dental anxiety of parents in an israeli kibbutz population and their prediction of their children’s behavior in the dental office. J. Clin. Pediatr. Dent. 2016, 40, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Schwartz-Arad, D.; Bar-Tal, Y.; Eli, I. The effect of stress on information processing in the dental implant surgery setting. Clin. Oral Implant. Res. 2007, 18, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Bucci, R.; Michelotti, A. Somatosensory changes in orthodontics—findings from quantitative sensory testing (QST) studies. Semin. Orthod. 2018, 24, 225–232. [Google Scholar] [CrossRef]
- Bakkers, M.; Faber, C.G.; Peters, M.J.H.; Reulen, J.P.H.; Franssen, H.; Fischer, T.Z.; Merkies, I.S.J. Temperature threshold testing: A systematic review. J. Peripher. Nerv. Syst. 2013, 18, 7–18. [Google Scholar] [CrossRef]
- Eliav, E.; Gracely, R.H.; Nahlieli, O.; Benoliel, R. Quantitative sensory testing in trigeminal nerve damage assessment. J. Orofac. Pain 2004, 18, 339–344. [Google Scholar]
- Baad-Hansen, L.; Pigg, M.; Yang, G.; List, T.; Svensson, P.; Drangsholt, M. Reliability of intra-oral quantitative sensory testing (QST) in patients with atypical odontalgia and healthy controls—A multicentre study. J. Oral Rehabil. 2015, 42, 127–135. [Google Scholar] [CrossRef] [Green Version]
- Lautenbacher, S. Experimental approaches in the study of pain in the elderly. Pain Med. 2012, 13 (Suppl. 2), S44–S50. [Google Scholar] [CrossRef] [Green Version]
- Lautenbacher, S.; Peters, J.H.; Heesen, M.; Scheel, J.; Kunz, M. Age changes in pain perception: A systematic-review and meta-analysis of age effects on pain and tolerance thresholds. Neurosci. Biobehav. Rev. 2017, 75, 104–113. [Google Scholar] [CrossRef]
- El Tumi, H.; Johnson, M.I.; Dantas, P.B.F.; Maynard, M.J.; Tashani, O.A. Age-related changes in pain sensitivity in healthy humans: A systematic review with meta-analysis. Eur. J. Pain 2017, 21, 955–964. [Google Scholar] [CrossRef] [Green Version]
- Briso, A.L.F.; Mestrener, S.R.; Delício, G.; Sundfeld, R.H.; Bedran-Russo, A.K.; de Alexandre, R.S.; Ambrosano, G.M.B. Clinical assessment of postoperative sensitivity in posterior composite restorations. Oper. Dent. 2007, 32, 421–426. [Google Scholar] [CrossRef] [Green Version]
- Berkowitz, G.S.; Horowitz, A.J.; Curro, F.A.; Craig, R.G.; Ship, J.A.; Vena, D.; Thompson, V.P. Postoperative hypersensitivity in class I resin-based composite restorations in general practice: Interim results. Compend. Contin. Educ. Dent. 2009, 30, 356–363. [Google Scholar]
- Robinson, M.E.; Bialosky, J.E.; Bishop, M.D.; Price, D.D.; George, S.Z. Supra-threshold scaling, temporal summation, and after-sensation: Relationships to each other and anxiety/fear. J. Pain Res. 2010, 3, 25–32. [Google Scholar] [CrossRef] [Green Version]
- Sanikop, S.; Agrawal, P.; Patil, S. Relationship between dental anxiety and pain perception during scaling. J. Oral Sci. 2011, 53, 341–348. [Google Scholar] [CrossRef] [Green Version]
- Van Wijk, A.J.; Hoogstraten, J. Experience with dental pain and fear of dental pain. J. Dent. Res. 2005, 84, 947–950. [Google Scholar] [CrossRef]
- Maggirias, J.; Locker, D. Psychological factors and perceptions of pain associated with dental treatment. Community Dent. Oral Epidemiol. 2002, 30, 151–159. [Google Scholar] [CrossRef]
- Canakçi, C.F.; Canakçi, V. Pain experienced by patients undergoing different periodontal therapies. J. Am. Dent. Assoc. 2007, 138, 1563–1573. [Google Scholar] [CrossRef]
- Settineri, S.; Tati, F.; Fanara, G. Gender differences in dental anxiety: Is the chair position important? J. Contemp. Dent. Pract. 2005, 6, 115–122. [Google Scholar] [CrossRef] [Green Version]
- Blumer, S.; Ram, D.; Costa, L.; Peretz, B. Dental Anxiety among Israeli Postgraduate Pediatric Dental Students and their Instructors. J. Clin. Pediatr. Dent. 2019, 43, 161–166. [Google Scholar] [CrossRef]
- Koutantji, M.; Pearce, S.A.; Oakley, D.A. The relationship between gender and family history of pain with current pain experience and awareness of pain in others. Pain 1998, 77, 25–31. [Google Scholar] [CrossRef]
- McGrath, P.A. Psychological aspects of pain perception. Arch. Oral Biol. 1994, 39, 55S–62S. [Google Scholar] [CrossRef]
- Defrin, R.; Shramm, L.; Eli, I. Gender role expectations of pain is associated with pain tolerance limit but not with pain threshold. Pain 2009, 145, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Linden, R.W. Periodontal mechanoreceptors and their function. In Neurophysiology of the Jaws and Teeth; Taylor, A., Ed.; The Macmillan Press Ltd., Scientific and Medical: London, UK, 1990; pp. 52–95. [Google Scholar]
- De Munck, J.; Van Landuyt, K.; Peumans, M.; Poitevin, A.; Lambrechts, P.; Braem, M.; Van Meerbeek, B. A critical review of the durability of adhesion to tooth tissue: Methods and results. J. Dent. Res. 2005, 84, 118–132. [Google Scholar] [CrossRef] [PubMed]




| Variable | Men (No. = 20) | Women (No. = 22) | |
|---|---|---|---|
| Age (years *) | 35.30 ± 14.54 | 35.86 ± 15.59 | |
| Type of restoration ** | Cl I CL V | 52.6% 47.4% | 40.9% 59.1% |
| Jaw treated **: | Upper: Lower | 40% 60% | 40.9% 59.1% |
| QST Measures | Mean | Std. Deviation | |
|---|---|---|---|
| Cold evaluation-1st meeting (VAS) | Cold-treated1 * | 3.71 | 2.61 |
| Cold-CL1 * | 3.43 | 2.57 | |
| Cold evaluation-2nd meeting (VAS) | Cold-treated2 * | 3.57 | 2.27 |
| Cold-CL2 | 3.42 | 2.51 | |
| Pain-1st meeting (VAS) | Pain-treated1 * | 0.71 | 1.61 |
| Pain-CL1 * | 0.83 | 1.96 | |
| Pain-2nd meeting (VAS) | Pain-trated2 * | 1.02 | 2.11 |
| Pain-CL2 * | 0.76 | 1.75 | |
| Cold duration-1st meeting (seconds) | Time-treated1 ** | 13.38 | 13.36 |
| Time-CL1 ** | 14.17 | 15.58 | |
| Cold duration-2nd meeting (seconds) | Time-treated2 ** | 15.23 | 12.64 |
| Time CL2 ** | 14.11 | 12.97 | |
| (a) Regression model including the following variables: type of restoration, jaw treated, Pain-treated1, Cold-treated1, Time-treated1, age, gender, DAS and the interactions DAS*gender, DAS*Pain-treated1, DAS*Cold-treated1, and DAS*Time-treated1 | ||||||||
| t | p | 95.0% Confidence Interval for B | ||||||
| Variable | B | Std. Error | Beta | Lower Bound | Upper Bound | |||
| Pain-treated1 | 0.578 | 0.167 | 0.491 | 3.452 | 0.001 | 0.239 | 0.917 | |
| DAS*gender (male) | 0.166 | 0.065 | 0.365 | 2.571 | 0.014 | 0.035 | 0.297 | |
| (b) Regression model including the following variables: type of restoration, jaw treated, Pain-treated1, Cold-treated1, Time-treated1, age, gender, DAS, and the DAS*gender interaction (final model marked in bold) | ||||||||
| Model | t | Sig. | 95.0% Confidence Interval for B | |||||
| B | Std. Error | Beta | Lower Bound | Upper Bound | ||||
| 1 | (Constant) | 1.391 | 0.311 | 4.470 | 0.000 | 0.761 | 2.021 | |
| Pain-treated1 | 0.479 | 0.174 | 0.408 | 2.752 | 0.009 | 0.127 | 0.831 | |
| 2 | (Constant) | 0.775 | 0.389 | 1.991 | 0.054 | −0.014 | 1.564 | |
| Pain-treated1 | 0.579 | 0.169 | 0.493 | 3.424 | 0.002 | 0.236 | 0.922 | |
| DAS*gender1 | 0.160 | 0.067 | 0.347 | 2.406 | 0.021 | 0.025 | 0.295 | |
| 3 | (Constant) | 0.236 | 0.453 | 0.522 | 0.605 | −0.683 | 1.156 | |
| Pain-treated1 | 0.645 | 0.165 | 0.549 | 3.910 | 0.000 | 0.311 | 0.980 | |
| DAS*gender (male) | 0.136 | 0.065 | 0.293 | 2.092 | 0.044 | 0.004 | 0.267 | |
| Time-treated1 | 0.042 | 0.020 | 0.292 | 2.088 | 0.044 | 0.001 | 0.082 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Emodi-Perlman, A.; Altarescu, D.; Frideman-Rubin, P.; Eli, I. Can Intra-Oral Qualitative Sensory Testing Foretell Postoperative Dental Pain? A Preliminary Report. Int. J. Environ. Res. Public Health 2022, 19, 8059. https://doi.org/10.3390/ijerph19138059
Emodi-Perlman A, Altarescu D, Frideman-Rubin P, Eli I. Can Intra-Oral Qualitative Sensory Testing Foretell Postoperative Dental Pain? A Preliminary Report. International Journal of Environmental Research and Public Health. 2022; 19(13):8059. https://doi.org/10.3390/ijerph19138059
Chicago/Turabian StyleEmodi-Perlman, Alona, Deia Altarescu, Pessia Frideman-Rubin, and Ilana Eli. 2022. "Can Intra-Oral Qualitative Sensory Testing Foretell Postoperative Dental Pain? A Preliminary Report" International Journal of Environmental Research and Public Health 19, no. 13: 8059. https://doi.org/10.3390/ijerph19138059






