Clinical Effectiveness of Herbal Oral Care Products in Periodontitis Patients: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. PICO Question
- Population: Adult human subjects diagnosed with periodontitis based on clinical examination (probing pocket depth and/or clinical attachment loss) and/or radiographic evaluation
- Intervention: Use of herbal dental products (mouthwash, dentifrice, gel) following SRP or supragingival debridement (SPD) or no additional periodontal treatment
- Comparison/Control: Use of non-herbal dental products (mouthwash, dentifrice, gel) or placebo following SRP or SPD or no additional periodontal treatment
- Outcome: Clinical periodontal parameters including probing pocket depth, gingival inflammation, dental plaque, clinical attachment loss.
2.2. Eligibility Criteria
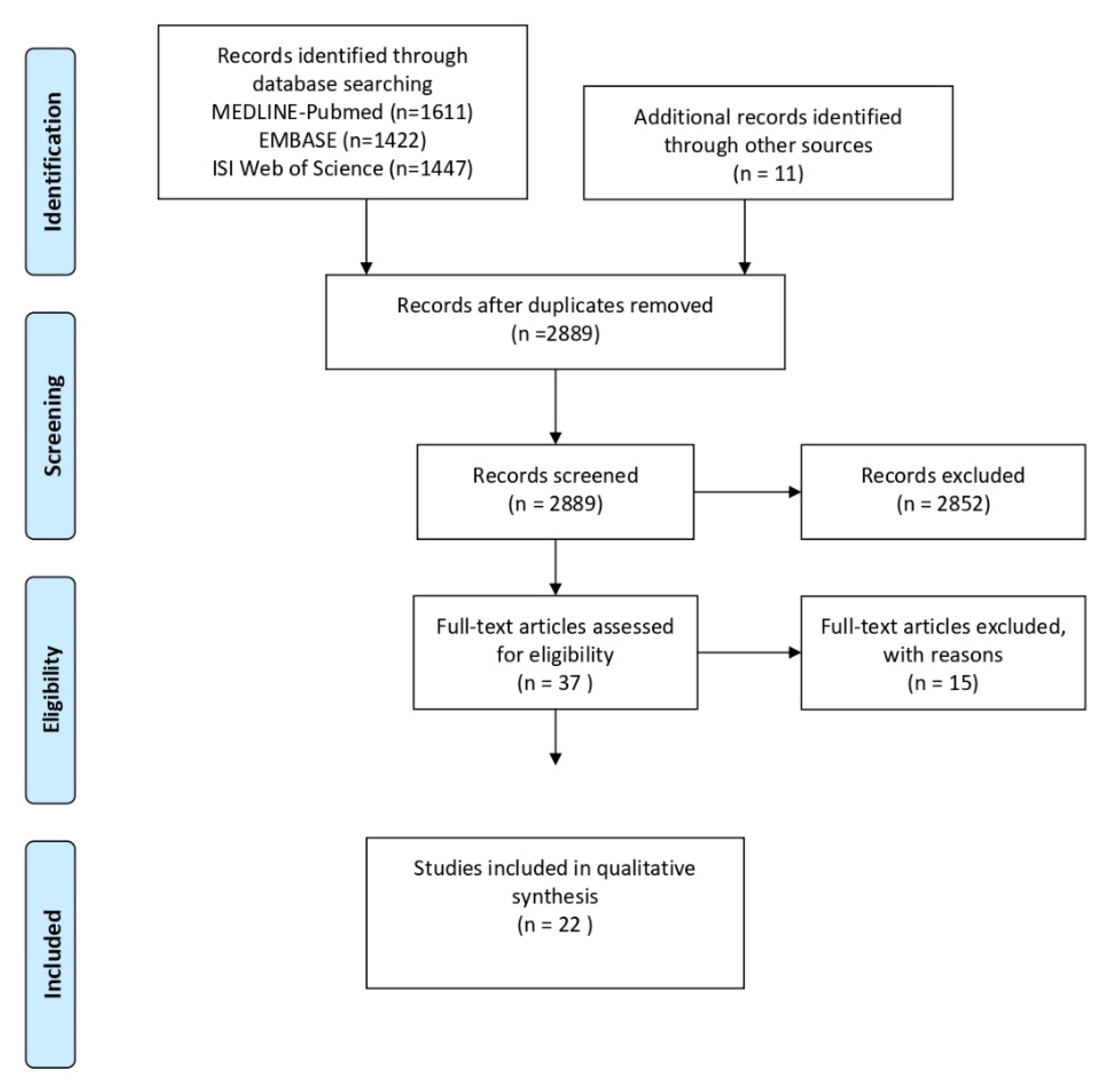
2.3. Search and Screening Strategy
2.4. Data Extraction
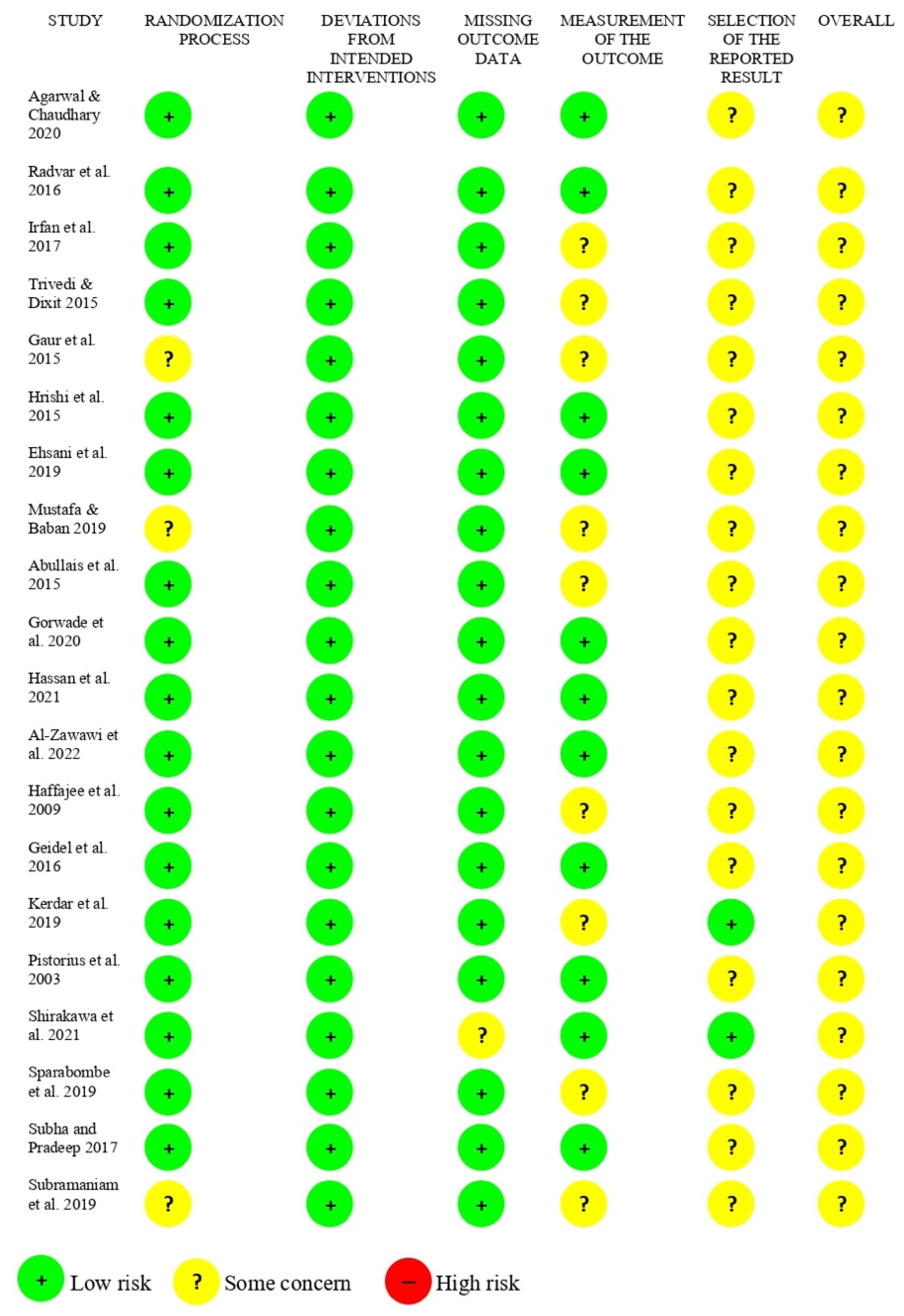
2.5. Risk of Bias Assessment
3. Results
4. Discussion
- Herbal products (mouthwash and solution for irrigation) used in conjunction with SRP or SPD led to superior clinical outcomes when compared to placebo or no adjuncts (8 studies).
- Herbal oral care products (mouthwash, solution for irrigation and dentifrice) including 1% Matricaria chamomilla, S. lappa extracts, Triphala, 4% Ocimum sanctum and polyherbal mouthwashes/solutions used in conjunction with SRP shown comparable outcomes with CHX (6 studies) or significantly better (4 studies).
- Herbal dental products including lemongrass and red ginseng used as supplements of SPD resulted in comparable outcomes with CHX and conventional products (4 studies).
- None of the included studies investigated the effect of the use of supragingival application of herbal gels in periodontitis.
- Research regarding side effects is lacking.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Offenbacher, S. Periodontal Diseases: Pathogenesis. Ann. Periodontol. 1996, 1, 821–878. [Google Scholar] [CrossRef] [PubMed]
- Kinane, D.F.; Stathopoulou, P.G.; Papapanou, P.N. Periodontal diseases. Nat. Rev. Dis. Primers 2017, 3, 17038. [Google Scholar] [CrossRef]
- Schaudinn, C.; Gorur, A.; Keller, D.; Sedghizadeh, P.P.; Costerton, J.W. Periodontitis: An archetypical biofilm disease. J. Am. Dent. Assoc. 2009, 140, 978–986. [Google Scholar] [CrossRef]
- Sanz, M.; Herrera, D.; Kebschull, M.; Chapple, I.; Jepsen, S.; Berglundh, T.; Sculean, A.; Tolleti, M.S.; On behalf of the EFP Workshop Participants and Methodological Consultants. Treatment of stage I–III periodontitis—The EFP S3 level clinical practice guideline. J. Clin. Periodontol. 2020, 47, 4–60. [Google Scholar] [CrossRef] [PubMed]
- Chapple, I.L.C.; Van Der Weijden, F.; Doerfer, C.; Herrera, D.; Shapira, L.; Polak, D.; Madianos, P.; Louropoulou, A.; Machtei, E.; Donos, N.; et al. Primary prevention of periodontitis: Managing gingivitis. J. Clin. Periodontol. 2015, 42 (Suppl. S16), S71–S76. [Google Scholar] [CrossRef] [PubMed]
- Van der Weijden, F.A.; Van der Sluijs, E.; Ciancio, S.G.; Slot, D.E. Can Chemical Mouthwash Agents Achieve Plaque/Gingivitis Control? Dent. Clin. N. Am. 2015, 59, 799–829. [Google Scholar] [CrossRef] [PubMed]
- Herrera, D.; Matesanz, P.; Martín, C.; Oud, V.; Feres, M.; Teughels, W. Adjunctive effect of locally delivered antimicrobials in periodontitis therapy: A systematic review and meta-analysis. J. Clin. Periodontol. 2020, 47, 239–256. [Google Scholar] [CrossRef]
- Herrera, D.; Sanz, M.; Kebschull, M.; Jepsen, S.; Sculean, A.; Berglundh, T.; Papapanou, P.N.; Chapple, I.; Tonetti, M.S.; On behalf of the EFP Workshop Participants and Methodological Consultant. Treatment of stage IV periodontitis: The EFP S3 level clinical practice guideline. J. Clin. Periodontol. 2022, 49 (Suppl. S24), 4–71. [Google Scholar] [CrossRef]
- Lindhe, J.; Nyman, S. Long-term maintenance of patients treated for advanced periodontal disease. J. Clin. Periodontol. 1984, 11, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Cobb, C.M. Non-Surgical Pocket Therapy: Mechanical. Ann. Periodontol. 1996, 1, 443–490. [Google Scholar] [CrossRef]
- Axelsson, P.; Lindhe, J. The significance of maintenance care in the treatment of periodontal disease. J. Clin. Periodontol. 1981, 8, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Bernimoulin, J.-P. Recent concepts in plaque formation. J. Clin. Periodontol. 2003, 30 (Suppl. S5), 7–9. [Google Scholar] [CrossRef] [PubMed]
- Löe, H.; Schiött, C.R.; Karring, G.; Karring, T.; Löue, H.; Schiöutt, C.R.; Glavind, L. Two years oral use of chlorhexidine in man. J. Periodontal Res. 1976, 11, 135–144. [Google Scholar] [CrossRef]
- Jones, G.G. Chlorhexidine is it still the gold standard? Periodontology 2000 1997, 15, 55–62. [Google Scholar] [CrossRef]
- Gjermo, P.; Bonesvoll, P.; Rölla, G. Relationship between plaque-inhibiting effect and retention of chlorhexidine in the human oral cavity. Arch. Oral Biol. 1974, 19, 1031–1034. [Google Scholar] [CrossRef]
- James, P.; Worthington, H.V.; Parnell, C.; Harding, M.; Lamont, T.; Cheung, A.; Whelton, H.; Riley, P. Chlorhexidine mouthrinse as an adjunctive treatment for gingival health. Cochrane Database Syst. Rev. 2017, 3, CD008676. [Google Scholar]
- Zhao, H.; Hu, J.; Zhao, L. Adjunctive subgingival application of Chlorhexidine gel in nonsurgical periodontal treatment for chronic periodontitis: A systematic review and meta-analysis. BMC Oral Health 2020, 20, 34. [Google Scholar] [CrossRef]
- Addy, M. Oral hygiene products: Potential for harm to oral and systemic health? Periodontology 2000 2008, 48, 54–65. [Google Scholar] [CrossRef]
- Laleman, I.; Teughels, W. Novel natural product-based oral topical rinses and toothpastes to prevent periodontal diseases. Periodontology 2000 2020, 84, 102–123. [Google Scholar] [CrossRef]
- Rashrash, M.; Schommer, J.C.; Brown, L.M. Prevalence and Predictors of Herbal Medicine Use Among Adults in the United States. J. Patient Exp. 2017, 4, 108–113. [Google Scholar] [CrossRef]
- Chen, Y.; Wong, R.W.; McGrath, C.; Hagg, U.; Seneviratne, C.J. Natural compounds containing mouthrinses in the management of dental plaque and gingivitis: A systematic review. Clin. Oral Investig. 2014, 18, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Kharaeva, Z.F.; Mustafaev, M.S.; Khazhmetov, A.V.; Gazaev, I.H.; Blieva, L.Z.; Steiner, L.; Mayer, W.; Luca, C.; Korkina, L.G. Anti-Bacterial and Anti-Inflammatory Effects of Toothpaste with Swiss Medicinal Herbs towards Patients Suffering from Gingivitis and Initial Stage of Periodontitis: From Clinical Efficacy to Mechanisms. Dent. J. 2020, 8, 10. [Google Scholar] [CrossRef] [PubMed]
- Kommuri, K.; Michelogiannakis, D.; Barmak, B.A.; Rossouw, P.E.; Javed, F. Efficacy of herbal- versus chlorhexidine-based mouthwashes towards oral hygiene maintenance in patients undergoing fixed orthodontic therapy: A systematic review and meta-analysis. Int. J. Dent. Hyg. 2022, 20, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Janakiram, C.; Venkitachalam, R.; Fontelo, P.; Iafolla, T.J.; Dye, B.A. Effectiveness of herbal oral care products in reducing dental plaque & gingivitis—A systematic review and meta-analysis. BMC Complement. Med. Ther. 2020, 20, 43. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. J. Clin. Epidemiol. 2009, 62, 1006–1012. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Slim, K.; Nini, E.; Forestier, D.; Kwiatkowski, F.; Panis, Y.; Chipponi, J. Methodological index for non-randomized studies (MINORS): Development and validation of a new instrument. ANZ J. Surg. 2003, 73, 712–716. [Google Scholar] [CrossRef]
- Khan, W.; Khan, M.; Alradwan, H.; Williams, R.; Simunovic, N.; Ayeni, O.R. Utility of intra-articular hip injections for femoroacetabular impingement: A systematic review. Orthop. J. Sports Med. 2015, 3, 2325967115601030. [Google Scholar] [CrossRef]
- NaikTari, R.S.; Gaonkar, P.; Gurav, A.N.; Khiste, S.V. A randomized clinical trial to evaluate and compare the efficacy of triphala mouthwash with 0.2% chlorhexidine in hospitalized patients with periodontal diseases. J. Periodontal Implant. Sci. 2014, 44, 134–140. [Google Scholar] [CrossRef]
- Sukhabogi, J.R.; Shekar, B.R.C.; Ramana, I.V.; Yadav, S.S.; Kumar, G.S.; Harita, N. Antiplaque Efficacy of Tooth and Gums Tonic, Hiora-GA Gel, and Spirogyl Gum Paint in Comparison with Chlorhexidine M Gel: A Double-blind Randomized Control Trial. Contemp. Clin. Dent. 2017, 8, 42–47. [Google Scholar] [CrossRef]
- Nisha, S.; Pandey, V.; Kumar, D.; Gupta, A.K.; Verma, T.; Kumari, A. Evaluation of anti-plaque and anti-inflammatory effects of oral curcumin gel as adjunct to scaling and root planing: A clinical study. Int. J. Appl. Basic Med. Res. 2021, 11, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Giammarinaro, E.; Marconcini, S.; Genovesi, A.; Poli, G.; Lorenzi, C.; Covani, U. Propolis as an adjuvant to non-surgical periodontal treatment: A clinical study with salivary anti-oxidant capacity assessment. Minerva Stomatol. 2018, 67, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Puri, S.; Bele, R.; Shewale, A.; Malik, R. The effect of Topical Application of Honey Based Gel Containing Zingibar officinale for Non Surgical Periodontal Maintenance. J. Pharm. Res. Int. 2021, 33, 134–139. [Google Scholar] [CrossRef]
- Liu, J.; Huang, Y.; Lou, X.; Liu, B.; Liu, W.; An, N.; Wu, R.; Ouyang, X. Effect of PudilanKeyanning antibacterial mouthwash on dental plaque and gingival inflammation in patients during periodontal maintenance phase: Study protocol for double-blind, randomised clinical trial. BMJ Open 2021, 11, e048992. [Google Scholar] [CrossRef]
- Sreedhar, A.; Sarkar, I.; Rajan, P.; Pai, J.; Malagi, S.; Kamath, V.; Barmappa, R. Comparative evaluation of the efficacy of curcumin gel with and without photo activation as an adjunct to scaling and root planing in the treatment of chronic periodontitis: A split mouth clinical and microbiological study. J. Nat. Sci. Biol. Med. 2015, 6 (Suppl. S1), S102–S109. [Google Scholar] [CrossRef] [PubMed]
- Nagasri, M.; Madhulatha, M.; Musalaiah, S.V.V.S.; Kumar, P.M.; Krishna, C.M. Efficacy of curcumin as an adjunct to scaling and root planning in chronic periodontitis patients: A clinical and microbiological study. J. Pharm. Bioallied Sci. 2015, 7 (Suppl. S2), S554–S558. [Google Scholar] [CrossRef]
- Pappu, R.; Varghese, J.; Koteshwara, K.; Kamath, V.; Lobo, R.; Nimmy, K. Evaluation of Biodegradable Gel Containing Flax Seed Extract (Linum usitatissimum) as a Targeted Drug Delivery for Management of Chronic Periodontitis. J. Herb. Med. 2019, 15, 100254. [Google Scholar] [CrossRef]
- Awesi, M.M.; Aspalli, S.; Bhattacharjee, S.; Pal, M. The Non-Surgical Therapeutic Approach for the Treatment of Periodontitis by Using Ayurvedic Formulations. Int. J. Res. Ayurveda Pharm. 2018, 9, 99–106. [Google Scholar] [CrossRef]
- Bhatia, M.; Urolagin, S.S.; Pentyala, K.B.; Urolagin, S.B.; Menaka, K.B.; Bhoi, S. Novel therapeutic approach for the treatment of periodontitis by curcumin. J. Clin. Diagn. Res. 2014, 8, ZC65–ZC69. [Google Scholar] [CrossRef]
- Aljuboori, I.W.; Mahmood, M.S. The Effects of Salvia officinalis Gel as an Adjunct to Scaling and Root Planning in Patients with Periodontitis (Clinical and Immunological Study). Int. J. Drug Deliv. Technol. 2020, 10, 232–237. [Google Scholar] [CrossRef]
- Arteaga, S.; Davila, L.; Gutierrez, R.; Sosa, L.; Albarran, G.; Isla, M.; Diaz, N. Effectiveness of Manzanilla gel and look like coor-dinating therapy in the treatment of chronic periodontitis. Acta Bioclin. 2017, 7, 6–25. [Google Scholar]
- Madhumathi, D.; Varghese, S. Comparison of clinical and antimicrobial effects of Triphala mouthwash with Chlor-hexidine mouthwash in generalised chronic periodontitis patients—A clinical study. Biomedicine 2020, 39, 422–424. [Google Scholar]
- Agarwal, A.; Chaudhary, B. Clinical and microbiological effects of 1% Matricaria chamomilla mouth rinse on chronic periodontitis: A double-blind randomized placebo controlled trial. J. Indian Soc. Periodontol. 2020, 24, 354–361. [Google Scholar]
- Radvar, M.; Moeintaghavi, A.; Tafaghodi, M.; Ghanbaria, H.; Fatemic, K.; Mokhtaric, M.R.; Najafi, F.; Hoseinipour, Z.; Dastmalchi, P.; Farazi, F. Clinical efficacy of an herbal mouth wash composed of Salix alba, Malva sylvestrais and Althaea officinalis in chronic periodontitis patients. J. Herb. Med. 2016, 6, 24–27. [Google Scholar] [CrossRef]
- Irfan, M.; Kumar, S.; Amin, V.; Cuevas-Suárez, C. Evaluation of the efficacy of triphala mouth rinse as coadjuvant in the treatment of chronic generalized periodontitis: A randomized clinical trial. Mouth Teeth 2017, 1, 1–6. [Google Scholar] [CrossRef]
- Trivedi, S.; Dixit, J. Clinical effects of irrigation with Saussurea lappa extract in the treatment of chronic periodontitis: A pilot randomized trial. J. Orofac. Sci. 2015, 7, 80. [Google Scholar] [CrossRef]
- Gaur, J.; Chandra, J.; Chaudhry, S.; Vaish, S.; Dodwad, V. Assessment of 4% ocimum sanctum and 0.2% chlorhexidine irrigation as an adjunct to scaling & root planing in management of chronic periodontitis—A randomized controlled trial. J. Dent. Spec. 2015, 3, 146–149. [Google Scholar]
- Hrishi, T.S.; Kundapur, P.P.; Naha, A.; Thomas, B.S.; Kamath, S.; Bhat, G.S. Effect of adjunctive use of green tea dentifrice in periodontitis patients—A Randomized Controlled Pilot Study. Int. J. Dent. Hyg. 2016, 14, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Ehsani, H.; Mehrani, J.; Jabbareh, L.; Habibi, E.; Mousazadeh, M.; Maleki, D.; Tayyeb e Khabbaz, S. Comparison of Oral Chlorhexidine with Green Tea Mouthwash in Treatment of Patients with Chronic Generalized Periodontitis: A Double-Blind Controlled Randomized Clinical Trial. J. Dentomaxillofac. Radiol. Pathol. Surg. 2019, 8, 7–12. [Google Scholar]
- Mustafa, S.Q.; Baban, D.A. Efficacy of green tea mouth wash as an adjunct to non-surgical periodontal treatment in patients with chronic periodontitis. Erbil Dent. J. EDJ 2019, 2, 180–187. [Google Scholar] [CrossRef]
- Abullais, S.S.; Dani, N.; Hamiduddin; Priyanka, N.; Kudyar, N.; Gore, A. Efficacy of irrigation with different antimicrobial agents on periodontal health in patients treated for chronic periodontitis: A randomized controlled clinical trial. AYU Int. Q. J. Res. Ayurveda 2015, 36, 380–386. [Google Scholar] [CrossRef]
- Gorwade, N.; Dhalkari, C.D.; Pol, D.G. Comparative evaluation of efficacy of three different mouthwashes as an adjunct to nonsurgical periodontal therapy—A double blind clinical study. J. Ayurveda Integr. Med. Sci. 2020, 2, 14–21. [Google Scholar] [CrossRef]
- Desai, A.; Anil, M.; Debnath, S. A clinical trial to evaluate the effects of triphala as a mouthwash in comparison with chlor-hexidine in chronic generalized periodontitis patient. Indian J. Dent. Adv. 2010, 2, 243. [Google Scholar]
- Hassan, G.; Ghafoor, S.; Atif, S.; Chaudhry, S.; Khan, Z.A. Non-Surgical periodontal therapy improves clinical outcomes in pa-tients with chronic periodontitis independent of the use of Nigella sativa oil or normal saline mouthwash; randomized controlled trial. J. Pak. Dent. Assoc. 2021, 30, 81–86. [Google Scholar] [CrossRef]
- Al-Zawawi, A.S.; Shaheen, M.Y.; Divakar, D.D.; Aldulaijan, H.A.; Basudan, A.M.; Devang, D.D. Postoperative anti-inflammatory efficacy of 2% saline rinses and a herbal-mouthwash after non-surgical periodontal therapy for the management of periodontal inflammation in young adults with chlorhexidine allergy: A randomized controlled trial. Int. J. Dent. Hyg. 2022, 20, 408–414. [Google Scholar] [CrossRef]
- Haffajee, A.D.; Roberts, C.; Murray, L.; Veiga, N.; Martin, L.; Teles, R.P.; Letteri, M.; Socransky, S.S. Effect of herbal, essential oil, and chlorhexidine mouthrinses on the composition of the subgingival microbiota and clinical periodontal parameters. J. Clin. Dent. 2009, 20, 211–217. [Google Scholar]
- Ali, A.; Mohammed, R. The Iraqi Method of Natural Liquorice as a Mouth Rinse and Its Effect in Patient with Chronic Peri-odontitis. Iraqi Dent. J. 2016, 38, 43–47. [Google Scholar] [CrossRef][Green Version]
- Geidel, A.; Krüger, M.; Schrödl, W.; Jentsch, H. Control of Plaque and Gingivitis by an Herbal Toothpaste—A Randomised Controlled Study. Oral Health Prev. Dent. 2017, 15, 407–413. [Google Scholar]
- Kerdar, T.; Rabienejad, N.; Alikhani, Y.; Moradkhani, S.; Dastan, D. Clinical, in vitro and phytochemical, studies of Scrophularia striata mouthwash on chronic periodontitis disease. J. Ethnopharmacol. 2019, 239, 111872. [Google Scholar] [CrossRef]
- Pistorius, A.; Willershausen, B.; Steinmeier, E.-M.; Kreisler, M. Efficacy of Subgingival Irrigation Using Herbal Extracts on Gingival Inflammation. J. Periodontol. 2003, 74, 616–622. [Google Scholar] [CrossRef]
- Shirakawa, S.; Matsushima, Y.; Kajiyama, S.; Suzuki, T.; Nagano, T.; Nikaidou, M.; Gomi, K. Effects of natural herb-containing dental toothpaste on periodontal pathogenic bacteria and clinical parameters: A randomized clinical trial. J. Herb. Med. 2021, 30, 100517. [Google Scholar] [CrossRef]
- Sparabombe, S.; Monterubbianesi, R.; Tosco, V.; Orilisi, G.; Hosein, A.; Ferrante, L.; Putignano, A.; Orsini, G. Efficacy of an All-Natural Polyherbal Mouthwash in Patients with Periodontitis: A Single-Blind Randomized Controlled Trial. Front. Physiol. 2019, 10, 632. [Google Scholar] [CrossRef] [PubMed]
- Subha, D.S.; Pradeep, T. Periodontal Therapy with 0.25% Lemongrass Oil Mouthwash in Reducing Risk of Cardiovascular Diseases: A 3-Arm Prospective Parallel Experimental Study. Ethiop. J. Health Sci. 2017, 27, 531–540. [Google Scholar] [CrossRef]
- Subramaniam, S.; Abirami, T.; Prakash, P.S.G.; Victor, D.J.; Bala, D. Comparison of clinical effectiveness of red ginseng mouthwash with chlorhexidine in generalized chronic periodontitis patients—A randomized controlled clinical trial. Pharm. Sci. Res. 2019, 11, 2570–2573. [Google Scholar]
- Van der Weijden, G.A.; Hioe, K.P. A systematic review of the effectiveness of self-performed mechanical plaque removal in adults with gingivitis using a manual toothbrush. J. Clin. Periodontol. 2005, 32 (Suppl. S6), 214–228. [Google Scholar] [CrossRef]
- Schätzle, M.; Löe, H.; Bürgin, W.; Ånerud, A.; Boysen, H.; Lang, N.P. Clinical course of chronic periodontitis. J. Clin. Periodontol. 2003, 30, 887–901. [Google Scholar] [CrossRef]
- Fardal, O.; Johannessen, A.C.; Linden, G. Tooth loss during maintenance following periodontal treatment in a periodontal practice in Norway. J. Clin. Periodontol. 2004, 31, 550–555. [Google Scholar] [CrossRef]
- Matuliene, G.; Pjetursson, B.E.; Salvi, G.E.; Schmidlin, K.; Brägger, U.; Zwahlen, M.; Lang, N.P. Influence of residual pockets on progression of periodontitis and tooth loss: Results after 11 years of maintenance. J. Clin. Periodontol. 2008, 35, 685–695. [Google Scholar] [CrossRef]
- Thangavelu, A.; Kaspar, S.S.; Kathirvelu, R.P.; Srinivasan, B.; Srinivasan, S.; Sundram, R. Chlorhexidine: An Elixir for Periodontics. J. Pharm. Bioallied. Sci. 2020, 12 (Suppl. S1), S57–S59. [Google Scholar] [CrossRef]
- Wu-Yuan, C.; Green, L.; Birch, W. In vitro Screening of Chinese Medicinal Toothpastes: Their Effects on Growth and Plaque Formation of Mutans Streptococci. Caries Res. 1990, 24, 198–202. [Google Scholar] [CrossRef]
- Chandra Shekar, B.R.; Nagarajappa, R.; Suma, S.; Thakur, R. Herbal extracts in oral health care—A review of the current scenario and its future needs. Pharm. Rev. 2015, 9, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, Y.; Kido, J.I.; Nishikawa, Y.; Kido, R.; Sakamoto, E.; Bando, M.; Naruishi, K.; Nagata, T.; Yumoto, H. Traditional Chinese Herbal Extracts, Reduces Osteoclast Differentiation In Vitro and Prevents Alveolar Bone Resorption in Rat Experimental Periodontitis. J. Clin. Med. 2021, 10, 386. [Google Scholar] [CrossRef]
- Song, H.K.; Noh, E.M.; Kim, J.M.; You, Y.O.; Kwon, K.B.; Lee, Y.R. Evodiae fructus Extract Inhibits Interleukin-1β-Induced MMP-1, MMP-3, and Inflammatory Cytokine Expression by Suppressing the Activation of MAPK and STAT-3 in Human Gingival Fi-broblasts In Vitro. Evid.-Based Complement. Altern. Med. 2021, 2021, 5858393. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, L.; Zhang, J.; Rastelli, A.N.D.S.; Yang, J.; Deng, D. Anti-Inflammatory Efficacy of Curcumin as an Adjunct to Non-Surgical Periodontal Treatment: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2022, 13, 808460. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, L.; Mazurel, D.; Zheng, H.; Yang, J.; Deng, D. Clinical efficacy of curcumin versus chlorhexidine as an adjunct to scaling and root planing for the treatment of periodontitis: A systematic review and meta-analysis. Phytother. Res. 2021, 35, 5980–5991. [Google Scholar] [CrossRef]
- Santi, S.S.; Casarin, M.; Grellmann, A.P.; Chambrone, L.; Zanatta, F.B. Effect of herbal mouthrinses on dental plaque formation and gingival inflammation: A systematic review. Oral Dis. 2021, 27, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Mehta, V.; Shetiya, S.H.; Kakodkar, P.; Janakiram, C.; Rizwan, S.A. Efficacy of herbal dentifrice on the prevention of plaque and gingivitis as compared to conventional dentifrice: A systematic review and meta-analysis. J. Indian Soc. Periodontol. 2018, 22, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Karygianni, L.; Al-Ahmad, A.; Argyropoulou, A.; Hellwig, E.; Anderson, A.C.; Skaltsounis, A.L. Natural Antimicrobials and Oral Microorganisms: A Systematic Review on Herbal Interventions for the Eradication of Multispecies Oral Biofilms. Front. Microbiol. 2016, 6, 1529. [Google Scholar] [CrossRef]
- Jassoma, E.; Baeesa, L.; Sabbagh, H. The antiplaque/anticariogenic efficacy of Salvadora persica (Miswak) mouthrinse in comparison to that of chlorhexidine: A systematic review and meta-analysis. BMC Oral Health 2019, 19, 64. [Google Scholar] [CrossRef]
- Srivastava, K.C.; Nimbulkar, G.; Garacha, V.; Shetty, V.; Bhor, K.; Shrivastava, D.; Sghaireen, M.G. Microbiological and Clinical evaluation of Neem gel and Chlorhexidine gel on dental plaque and gingivitis in 20–30 years old adults: A Randomized Parallel-Armed, Double-blinded Controlled Trial. J. Pharm. Bioallied Sci. 2020, 12 (Suppl. S1), S345–S351. [Google Scholar] [CrossRef]
- Freires, I.A.; Santaella, G.M.; Sardi, J.D.C.O.; Rosalen, P.L. The alveolar bone protective effects of natural products: A systematic review. Arch. Oral Biol. 2018, 87, 196–203. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, E.T.; de Araújo, J.S.M.; Pires, A.C.; Lira Dos Santos, E.J. Local delivery natural products to treat periodontitis: A systematic review and meta-analysis. Clin. Oral Investig. 2021, 25, 4599–4619. [Google Scholar] [CrossRef] [PubMed]
- Babrawala, I.; Venkatesh, P.M.L.; Varadhan, K.B. A Novel Approach Using 15% Natural Chitosan Gel in the Management of Intrabony Defects: A Pilot Study. Chin. J. Dent. Res. 2016, 19, 231–237. [Google Scholar]
- Moro, M.G.; Silveira Souto, M.L.; Franco, G.C.N.; Holzhausen, M.; Pannuti, C.M. Efficacy of local phytotherapy in the nonsurgical treatment of periodontal disease: A systematic review. J. Periodontal Res. 2018, 53, 288–297. [Google Scholar] [CrossRef] [PubMed]
| Study | Year of Publication | Country | Study Design | Adjunctive Therapy | Groups | Number of Subjects (Males/Females) | Periodontal Diagnosis | Parameters | Follow-Up | Conclusions |
|---|---|---|---|---|---|---|---|---|---|---|
| Agarwal & Chaudhary [43] | 2020 | India | RCT | Mouthwash | A. SRP + 1% Matricaria chamomilla (MTC) B. SRP + 0.12% CHX C. SRP + placebo | A. 25 (12/13) B. 25 (12/13) C. 25 (15/10) | Generalized chronic periodontitis | PI, GI, sulcus bleeding, PPD, CAL, GRE, SI | 6 weeks, 3 months | MTC significant benefits over placebo and comparable to CHX. |
| Radvar et al. [44] | 2016 | Iran | RCT | Mouthwash | A.SRP + herbal (Althaea officinalis, Salix alba and Malva Silvestris extracts) B. SRP + CHX C. SRP + placebo | 30 | Chronic periodontitis | PPD, BOP, CAL | 6 weeks | Herbal mouthwash significant benefits over placebo. CHX better than herbal. |
| Irfan et al. [45] | 2017 | India | RCT | Mouthwash | A. SRP + triphala B. SRP + 0.2% CHX | A. 25 B. 25 | Generalized chronic periodontitis | PI, GI | 7, 30, 45 days | Triphala mouthwash effective in reducing plaque and gingival inflammation. Triphala more effective in reducing inflammation. |
| Trivedi & Dixit [46] | 2015 | India | RCT | Subgingival irrigation with solution | A. SRP + S. lappa extracts B. SRP + 0.2% CHX C. SRP alone | 30 Periodontal pockets: A. 180 B. 180 C. 180 | Chronic periodontitis | Modified SBI, PPD, CAL | 7, 14, 28, 42 days | Irrigation with S. lappa significant benefits over SRP alone. S. lappa and CHX similar results. S. lappa better in 4–5 mm pockets. |
| Gaur et al. [47] | 2015 | India | RCT | Subgingival irrigation with solution | A. SRP + 4% Ocimum sanctum B. SRP + 0.2% CHX | 30 Periodontal pockets: A. 15 B. 15 | Chronic periodontitis | PI, GI, PPD, CAL | 30 days | Irrigation with Ocimum sanctum showed similar significant benefits in gingival inflammation and plaque with CHX. CHX was superior in PPD and CAL reduction. |
| Hrishi et al. [48] | 2015 | India | RCT | Dentifrice | A. SRP + green tea B. SRP + triclosan-containing | 30 (13/17) A. 15 B. 15 | Mild to moderate periodontitis | PI, GI, PPD, BOP, CAL | 4 weeks | Green tea showed greater reduction of gingival inflammation and CAL. |
| Ehsani et al. [49] | 2019 | Iran | RCT | Mouthwash | A. SRP + 0.05% green tea B. SRP + 0.2% CHX | 40 (19/21) A. 20 (4/16) B. 20 (15/5) | Generalized moderate chronic periodontitis | BOP, PI, GI, PPD, CAL | 7 and 21 days | Green tea was more effective to CHX at 3 weeks. |
| Mustafa &Baban [50] | 2019 | Iraq | RCT | Mouthwash | A. SRP + 5% green tea B. SRP + 0.12% CHX C. SRP only | 45 (27/18) A. 15 B. 15 C. 15 | Localized mild to moderate chronic periodontitis | GI, PI, GBI, PPD, CAL | 30 days | Green tea significantly improved all periodontal parameters compared to CHX or SRP only. |
| Abullais et al. [51] | 2015 | India | RCT | Subgingival irrigation with solution | A. Punica granatum Linn., Piper nigrum Linn., detoxified copper sulfate B. 0.2% CHX (SRP completed 1 month earlier) | 30 A. 15 B. 15 | Chronic periodontitis (residual pockets following SRP) | PI, SBI, PPD | 15, 30, 60, 90 days | Irrigation with herbal solution resulted in significant plaque reduction. CHX led to significant reduction in gingival inflammation. |
| Gorwade et al. [52] | 2020 | India | RCT | Mouthwash | A. SRP + Triphala B. SRP+ Bakul C. SRP + CHX | 90 A. 30 B. 30 C. 30 | Chronic periodontitis | GI, OHI, PI, PPD, CAL, papillary bleeding index | 2, 4, 6, 8, 12 weeks | Triphala resulted in significantly greater plaque reduction at 3 months thanBakul and CHX. |
| Desai & Debnath [53] | 2010 | India | Non-RCT | Mouthwash | A. SRP + Triphala B. SRP + CHX C. SRP only | 24 A. 8 B. 8 C. 8 | Generalized chronic periodontitis | PI, GI, OHI, Periodontal index | 7, 30, 45 days | Triphala showed significant reduction in periodontal indices compared to SRP alone. Similar outcome between Triphala and CHX. |
| Hassan et al. [54] | 2021 | Pakistan | RCT | Mouthwash | A. SRP + Nigella sativa B. SRP + saline | 50 A. 25 B. 25 | Chronic periodontitis | PPD, CAL, PI, BOP | 2 weeks | Both Nigella sativa and saline had a significant beneficial effect. No difference between the mouthwashes. |
| Al-Zawawi et al. [55] | 2022 | India | RCT | Mouthwash | A. SRP + Herbal (Himalaya Drug Company, HiOra®) B. SRP + 2% saline C. SRP + 0.12% CHX | 37 (27/10) A. 12 (9/3) B. 13 (8/5) C. 12 (10/2) | Generalized chronic periodontitis | PI, GI, PPD, CAL | 6 weeks | Significant improvement in PI, GI and PPD at 6 weeks. No differences between the mouthwashes. |
| Haffajee et al. [56] | 2009 | USA | RCT | Mouthwash | A. Listerine Cool Mint (essential oil) B. Peridex (CHX) C. The Natural Dentist Healthy Gums Oral rinse (herbal) D. The Natural Dentist Healthy Gums minus bloodroot (herbal) | 122 A. 28 B. 31 C. 29 D. 28 | Chronic periodontitis (residual pockets following SRP) | GI, PI, BOP, PD, CAL | 3 months | Both herbal mouthwashes reduced plaque significantly. No significant change in gingival inflammation. CHX significantly better results. |
| Ali & Mohammed [57] | 2016 | Iraq | Non-RCT | Mouthwash | A.SRP + liquorice mouthwash B.SRP | A. 15 B. 15 | Chronic periodontitis | PI, GI | 1 week | Decrease of plaque & gingival inflammation. No significant differences between SRP and SRP + liquorice. |
| Geidel et al. [58] | 2015 | Germany | RCT | Dentifrice | A. SPD + Herbal toothpaste B. SPD + triclosan/copolymer toothpaste C. SPD + amine/stannous fluoride toothpaste | A. 25 B. 26 C. 25 | Slight & moderate chronic periodontitis | OHI, API, SBI, BOP, PPD, CAL | 6, 12, 24 weeks | The herbal toothpaste as good as the control toothpastes. |
| Kerdar et al. [59] | 2019 | Iran | RCT | Mouthwash | A. Herbal (Hydro alcoholic extract of Stuckenia striata) mouthwash B.Irsha mouthwash No periodontal treatment | A. 25 B. 25 | Chronic periodontitis | PI, PD, BOP | 2, 4 weeks | Herbal mouthwash effective to chronic periodontitis and more potent compared to Irsha mouthwash. |
| Pistorius et al. [60] | 2003 | Germany | RCT | Mouthwash | A. SPD + oral irrigator + herbal mouthwash B. SPD + oral irrigator + conventional mouthwash C. SPD + conventional mouthwash | A. 34 B. 29 C. 26 | Chronic periodontitis | GI, SBI, PI, PD | 4, 8, 12 weeks | Herbal mouthwash group showed significantly higher SBI & GI reduction. |
| Shirakawa et al. [61] | 2021 | Japan | RCT | Dentifrice | A. Herbal toothpaste B. Control toothpaste No periodontal treatment | A. 37 B. 37 | Chronic periodontitis | GI, PD, BOP, Plaque control record | 2, 4 weeks | Significant improvement of clinical parameters when herbal toothpaste used. |
| Sparabombe et al. [62] | 2019 | Italy | RCT | Mouthwash | A. SPD + herbal mouthwash B. SPD + placebo mouthwash | A. 20 B. 20 | Moderate or severe periodontitis | FMBS, FMPS, PD and CAL | 12 weeks | Herbal mouthwash led tosignificantly higher reduction of bleeding score and plaque accumulation. |
| Subha and Pradeep [63] | 2017 | India | RCT | Mouthwash | A. SPD+ lemongrass oil mouthwash B. SPD + CHX mouthwash C. SPD only | A. 15 B. 15 C. 15 | Generalized severe periodontitis | PPD, CAL, c-reactive protein, total cholesterol, high and low density lipid, triglycerides | 12 weeks | Lemongrass oil mouthwash can be a good alternative in chronic periodontitis. Significantly higher PPD, CAL reduction in Herbal and CHX groups than SPD only. |
| Subramaniam et al. [64] | 2019 | India | RCT | Mouthwash | A. SPD + Red Ginseng mouthwash B. SPD + CHX mouthwash C. SPD+ placebo mouthwash | A. 10 B. 10 C. 10 | Generalized chronic periodontitis | GI, FMBS, FMPS, PPD, CAL | 4 weeks | Red Ginseng mouthwash has comparable effects to CHXand it is significantly better in GI and FMBS reduction than placebo. |
| Desai & Debnath [53] | Ali & Mohammed [57] | |
|---|---|---|
| Clearly stated aim | 2 | 2 |
| Inclusion of consecutive patients | 1 | 1 |
| Prospective data collection | 1 | 1 |
| Endpoints appropriate to study aim | 2 | 1 |
| Unbiased assessment of study endpoints | 1 | 1 |
| Follow-up period appropriate | 2 | 0 |
| <5% lost to follow-up | 2 | 2 |
| Prospective calculation of study size | 0 | 0 |
| Total | 11 | 8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chatzopoulos, G.S.; Karakostas, P.; Kavakloglou, S.; Assimopoulou, A.; Barmpalexis, P.; Tsalikis, L. Clinical Effectiveness of Herbal Oral Care Products in Periodontitis Patients: A Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 10061. https://doi.org/10.3390/ijerph191610061
Chatzopoulos GS, Karakostas P, Kavakloglou S, Assimopoulou A, Barmpalexis P, Tsalikis L. Clinical Effectiveness of Herbal Oral Care Products in Periodontitis Patients: A Systematic Review. International Journal of Environmental Research and Public Health. 2022; 19(16):10061. https://doi.org/10.3390/ijerph191610061
Chicago/Turabian StyleChatzopoulos, Georgios S., Panagiotis Karakostas, Stefania Kavakloglou, Andreana Assimopoulou, Panagiotis Barmpalexis, and Lazaros Tsalikis. 2022. "Clinical Effectiveness of Herbal Oral Care Products in Periodontitis Patients: A Systematic Review" International Journal of Environmental Research and Public Health 19, no. 16: 10061. https://doi.org/10.3390/ijerph191610061
APA StyleChatzopoulos, G. S., Karakostas, P., Kavakloglou, S., Assimopoulou, A., Barmpalexis, P., & Tsalikis, L. (2022). Clinical Effectiveness of Herbal Oral Care Products in Periodontitis Patients: A Systematic Review. International Journal of Environmental Research and Public Health, 19(16), 10061. https://doi.org/10.3390/ijerph191610061







