Isolation and Optimisation of Culture Conditions for a Marine Bioflocculant-Producing Bacterium and Application of Its Bioflocculant in Wastewater Treatment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Production Medium
2.2. Sample Collection
2.3. Isolation of the Bioflocculant-Producing Bacteria
2.4. Activation of the Isolates
2.5. Cultivation Medium for Bioflocculant Production
2.6. Determination of Flocculating Activity of Isolates
2.7. Identification of the Bacterium
2.8. Optimization of the Culture Conditions
2.9. Extraction and Purification of the Bioflocculant
2.10. Characterizations of the Bioflocculant
2.11. Biosafety of the Bioflocculant
2.12. Effect Dosages, Cations and pH on the Flocculating Activity
2.13. Application of the Bioflocculant in Wastewater Treatment
2.14. Statistical Analysis
3. Results and Discussion
3.1. Physiochemical Parameters of Water
3.2. Isolation and Identification of the Bioflocculant-Producing Bacterium
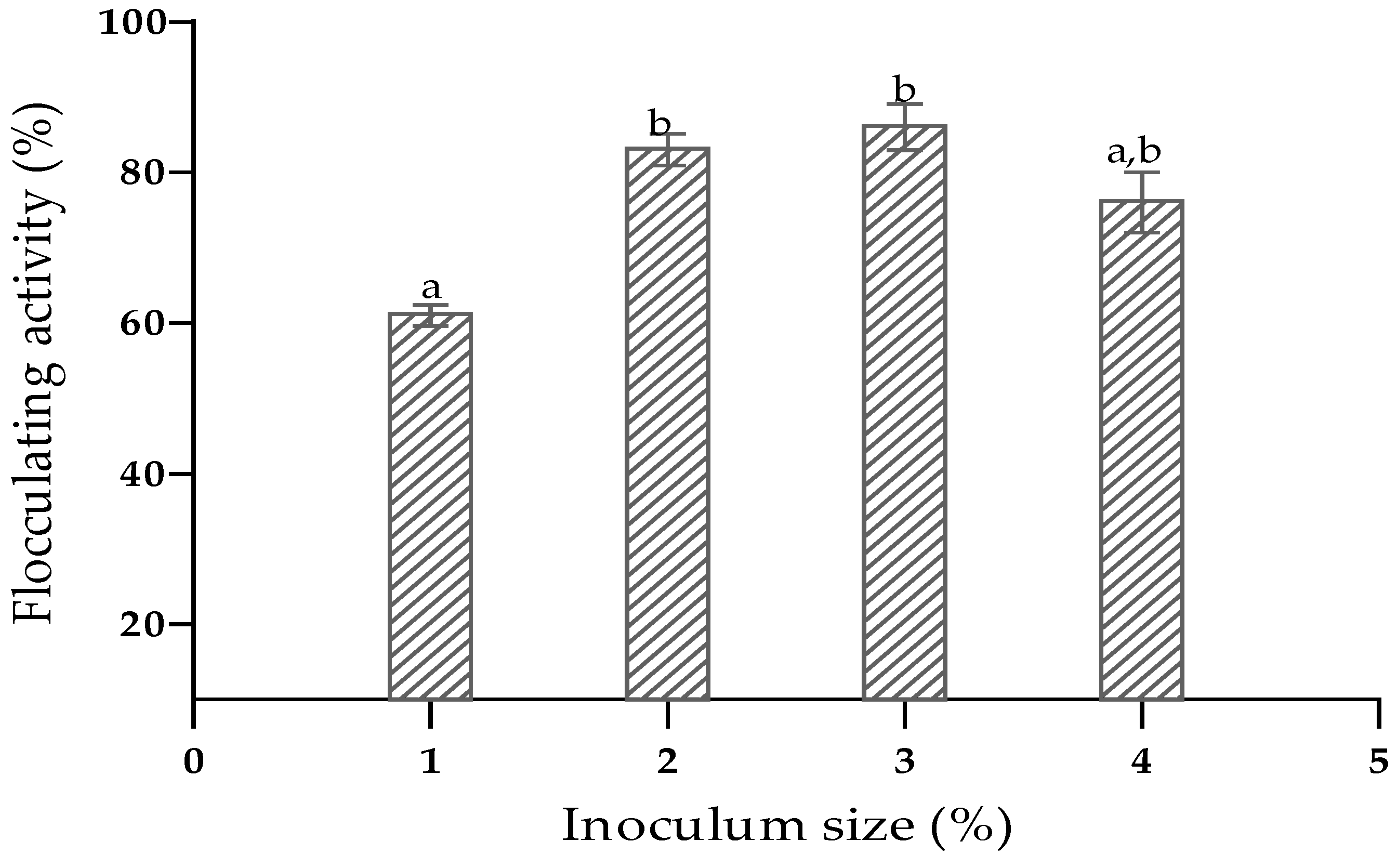
3.3. Optimisation of Medium Composition and Culture Conditions
3.4. Extraction and Purification of Bioflocculant
3.5. Characterization of the Purified Bioflocculant
3.6. Biosafety of the Bioflocculant from O. oryzae AB841138
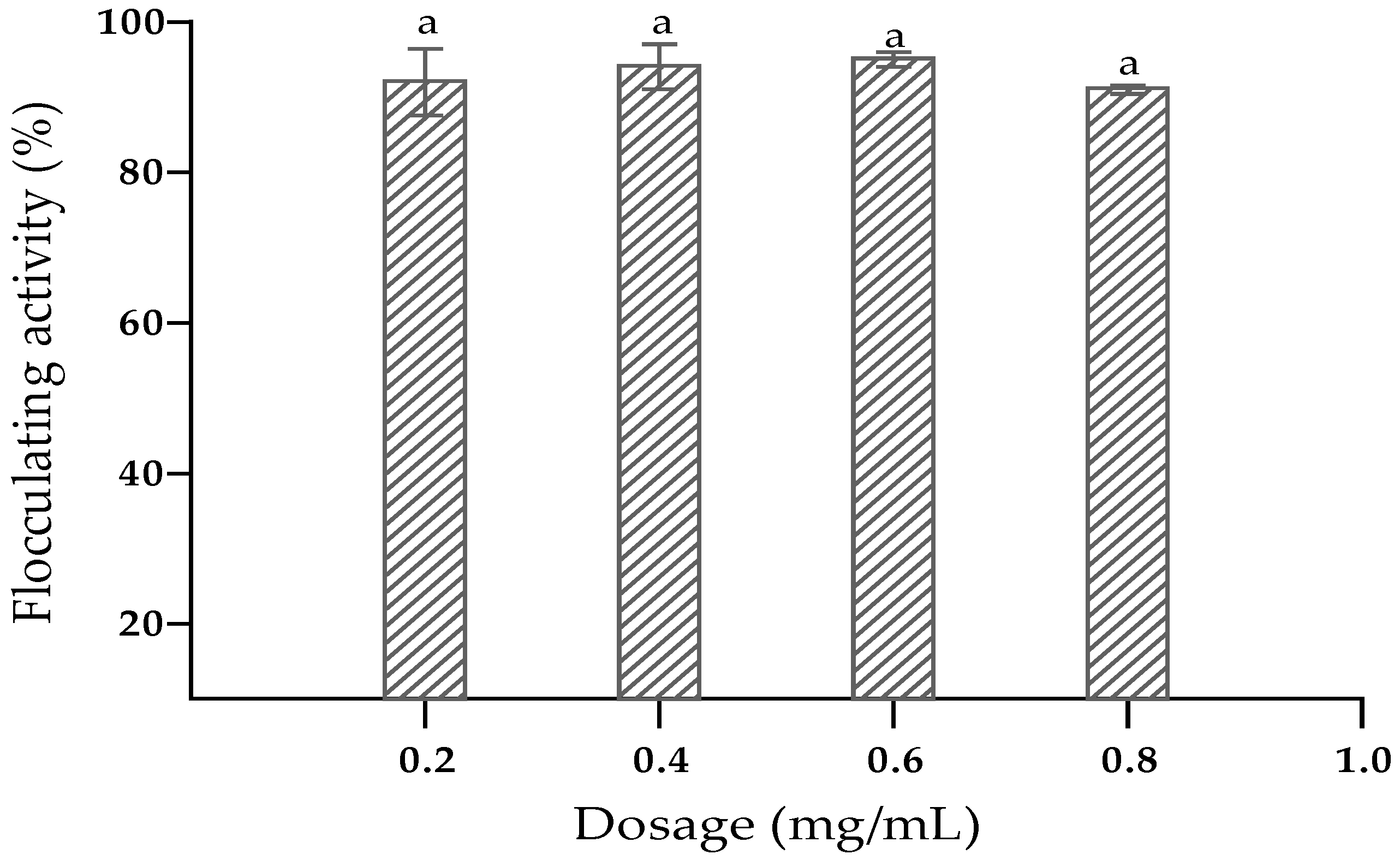
3.7. Effect of Dosage, Cations and pH on Flocculating Activity
3.8. Removal Efficiency of the Bioflocculant
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gayathiri, E.; Prakash, P.; Selvam, K.; Awasthi, M.K.; Gobinath, R.; Karri, R.R.; Ragunathan, M.G.; Jayanthi, J.; Mani, V.; Poudineh, M.A. Plant microbe based remediation approaches in dye removal: A review. Bioengineered 2022, 13, 7798–7828. [Google Scholar] [CrossRef]
- Devda, V.; Chaudhary, K.; Varjani, S.; Pathak, B.; Patel, A.K.; Singhania, R.R.; Taherzadeh, M.J.; Ngo, H.H.; Wong, J.W.; Guo, W. Recovery of resources from industrial wastewater employing electrochemical technologies: Status, advancements and perspectives. Bioengineered 2021, 12, 4697–4718. [Google Scholar] [CrossRef] [PubMed]
- Ma, F.; Zheng, L.; Chi, Y. Applications of biological flocculants (BFs) for coagulation treatment in water purification: Turbidity elimination. Chem. Biochem. Eng. Q. 2008, 22, 321–326. [Google Scholar]
- Programme, W.W.A.; UN-Water. Water in a Changing World; Wiley: New York, NY, USA, 2009. [Google Scholar]
- Maruyama, H.; Seki, H. Evaluation of flocculation performance of polysaccharide-protamine complex flocculant by flocculation model. Biochem. Eng. J. 2022, 180, 108356. [Google Scholar] [CrossRef]
- Akinola, O.; Adebayo, E. Screening and characterization of bioflocculant producing bacteria isolated from domestic waste water in Bowen University, Iwo, Osun State, Nigeria. Niger. J. Biotechnol. 2018, 35, 159–166. [Google Scholar] [CrossRef]
- Okaiyeto, K.; Nwodo, U.U.; Mabinya, L.V.; Okoli, A.S.; Okoh, A.I. Evaluation of flocculating performance of a thermostable bioflocculant produced by marine Bacillus sp. Environ. Technol. 2016, 37, 1829–1842. [Google Scholar] [CrossRef]
- Salehizadeh, H.; Yan, N. Recent advances in extracellular biopolymer flocculants. Biotechnol. Adv. 2014, 32, 1506–1522. [Google Scholar] [CrossRef]
- Agunbiade, M.; Pohl, C.; Ashafa, O. Bioflocculant production from Streptomyces platensis and its potential for river and waste water treatment. Braz. J. Microbiol. 2018, 49, 731–741. [Google Scholar] [CrossRef]
- Maliehe, T.; Basson, A.; Singh, M. Wastewater treatment by a novel bioflocculant from a consortium of Bacillus pumilus JX860616 and Bacillus subtilis CSM5. Biosci. Res. 2020, 17, 1610–1625. [Google Scholar]
- Zaki, S.A.; Elkady, M.F.; Farag, S.; Abd-El-Haleem, D. Characterization and flocculation properties of a carbohydrate bioflocculant from a newly isolated Bacillus velezensis 40B. J. Environ. Biol. 2013, 34, 51–58. [Google Scholar]
- Ajao, V.; Fokkink, R.; Leermakers, F.; Bruning, H.; Rijnaarts, H.; Temmink, H. Bioflocculants from wastewater: Insights into adsorption affinity, flocculation mechanisms and mixed particle flocculation based on biopolymer size-fractionation. J. Colloid Interface Sci. 2021, 581, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Kurniawan, S.B.; Abdullah, S.R.S.; Imron, M.F.; Ahmad, A.; Mohd Said, N.S.; Mohd Rahim, N.F.; Mohammad Alnawajha, M.; Abu Hasan, H.; Othman, A.R.; Purwanti, I.F. Potential of valuable materials recovery from aquaculture wastewater: An introduction to resource reclamation. Aquac. Res. 2021, 52, 2954–2962. [Google Scholar] [CrossRef]
- Zafar, L.; Khan, A.; Kamran, U.; Park, S.-J.; Bhatti, H.N. Eucalyptus (camaldulensis) bark-based composites for efficient Basic Blue 41 dye biosorption from aqueous stream: Kinetics, isothermal, and thermodynamic studies. Surf. Interfaces 2022, 31, 101897. [Google Scholar] [CrossRef]
- Mu, J.; Cui, X.; Shao, M.; Wang, Y.; Yang, Q.; Yang, G.; Zheng, L. Microbial origin of bioflocculation components within a promising natural bioflocculant resource of Ruditapes philippinarum conglutination mud from an aquaculture farm in Zhoushan, China. PLoS ONE 2019, 14, e0217679. [Google Scholar] [CrossRef] [PubMed]
- Grenda, K.; Gamelas, J.A.; Arnold, J.; Pellizzer, L.; Cayre, O.J.; Rasteiro, M.G. Evaluation of anionic eco-friendly flocculants prepared from Eucalyptus pulps with diverse lignin contents for application in effluent treatment. Polymers 2020, 13, 25. [Google Scholar] [CrossRef]
- Nxumalo, C.I.; Ngidi, L.S.; Shandu, J.S.E.; Maliehe, T.S. Isolation of endophytic bacteria from the leaves of Anredera cordifolia CIX1 for metabolites and their biological activities. BMC Complement. Med. Ther. 2020, 20, 300. [Google Scholar] [CrossRef]
- Kurniawan, S.B.; Imron, M.F.; Chik, C.E.N.C.E.; Owodunni, A.A.; Ahmad, A.; Alnawajha, M.M.; Rahim, N.F.M.; Said, N.S.M.; Abdullah, S.R.S.; Kasan, N.A. What compound inside biocoagulants/bioflocculants is contributing the most to the coagulation and flocculation processes? Sci. Total Environ. 2022, 806, 150902. [Google Scholar] [CrossRef]
- Ben Rebah, F.; Mnif, W.; Siddeeg, S.M. Microbial flocculants as an alternative to synthetic polymers for wastewater treatment: A review. Symmetry 2018, 10, 556. [Google Scholar] [CrossRef]
- Bisht, V.; Lal, B. Exploration of performance kinetics and mechanism of action of a potential novel bioflocculant BF-VB2 on clay and dye wastewater flocculation. Front. Microbiol. 2019, 10, 1288. [Google Scholar] [CrossRef]
- Yin, Y.-J.; Tian, Z.-M.; Tang, W.; Li, L.; Song, L.-Y.; McElmurry, S.P. Production and characterization of high efficiency bioflocculant isolated from Klebsiella sp. ZZ-3. Bioresour. Technol. 2014, 171, 336–342. [Google Scholar] [CrossRef]
- Arafa, R.A.; El-Rouby, M.N.; Abass, H.A.; El-Khier, Z. Bioflocculants produced by bacterial isolates from Egyptian soil 1-characterization and application of extracellular bioflocculants and nanoparticles for treatment of river Nile water. J. Pharm. Biol. Sci. 2014, 9, 103–114. [Google Scholar] [CrossRef]
- Mabinya, L.V.; Cosa, S.; Mkwetshana, N.; Okoh, A.I. Halomonas sp. OKOH—A marine bacterium isolated from the bottom sediment of Algoa Bay—Produces a polysaccharide bioflocculant: Partial characterization and biochemical analysis of its properties. Molecules 2011, 16, 4358–4370. [Google Scholar] [CrossRef] [PubMed]
- Tsilo, P.H.; Basson, A.K.; Ntombela, Z.G.; Maliehe, T.S.; Pullabhotla, R.V. Isolation and optimization of culture conditions of a bioflocculant-producing fungi from Kombucha tea SCOBY. Microbiol. Res. 2021, 12, 950–966. [Google Scholar] [CrossRef]
- Kurane, R.; Hatamochi, K.; Kakuno, T.; Kiyohara, M.; Hirano, M.; Taniguchi, Y. Production of a bioflocculant by Rhodococcus erythropolis S-1 grown on alcohols. Biosci. Biotechnol. Biochem. 1994, 58, 428–429. [Google Scholar] [CrossRef]
- Shida, O.; Takagi, H.; Kadowaki, K.; Nakamura, L.K.; Komagata, K. Transfer of Bacillus alginolyticus, Bacillus chondroitinus, Bacillus curdlanolyticus, Bacillus glucanolyticus, Bacillus kobensis, and Bacillus thiaminolyticus to the genus Paenibacillus and emended description of the genus Paenibacillus. Int. J. Syst. Evol. Microbiol. 1997, 47, 289–298. [Google Scholar] [CrossRef]
- Zhang, Z.-Q.; Bo, L.; Xia, S.-Q.; Wang, X.-J.; Yang, A.-M. Production and application of a novel bioflocculant by multiple-microorganism consortia using brewery wastewater as carbon source. J. Environ. Sci. 2007, 19, 667–673. [Google Scholar] [CrossRef]
- Luo, Z.; Chen, L.; Chen, C.; Zhang, W.; Liu, M.; Han, Y.; Zhou, J. Production and characteristics of a bioflocculant by Klebsiella pneumoniae YZ-6 isolated from human saliva. Appl. Biochem. Biotechnol. 2014, 172, 1282–1292. [Google Scholar] [CrossRef] [PubMed]
- Ugbenyen, A.M.; Simonis, J.J.; Basson, A.K. Screening for bioflocculant-producing bacteria from the marine environment of Sodwana Bay, South Africa. Ann. Sci. Technol. 2018, 3, 16–20. [Google Scholar] [CrossRef]
- Abdel-Aziz, S.M.; Hamed, H.A.; Mouafi, F.E.; Abdelwahed, N.A. Extracellular metabolites produced by a novel strain, Bacillus alvei NRC-14:3. Synthesis of a bioflocculant that has chitosan-like structure. Life Sci. J. 2011, 8, 883–890. [Google Scholar]
- He, P.; Geng, L.; Wang, Z.; Mao, D.; Wang, J.; Xu, C. Fermentation optimization, characterization and bioactivity of exopolysaccharides from Funalia trogii. Carbohydr. Polym. 2012, 89, 17–23. [Google Scholar] [CrossRef]
- Xia, S.; Zhang, Z.; Wang, X.; Yang, A.; Chen, L.; Zhao, J.; Leonard, D.; Jaffrezic-Renault, N. Production and characterization of a bioflocculant by Proteus mirabilis TJ-1. Bioresour. Technol. 2008, 99, 6520–6527. [Google Scholar] [CrossRef] [PubMed]
- Maliehe, T.; Simonis, J.; Basson, A.; Reve, M.; Ngema, S.; Xaba, P. Production, characterisation and flocculation mechanism of bioflocculant TMT-1 from marine Bacillus pumilus JX860616. Afr. J. Biotechnol. 2016, 15, 2352–2367. [Google Scholar]
- Ugbenyen, A.; Cosa, S.; Mabinya, L.; Okoh, A. Bioflocculant production by Bacillus sp. Gilbert isolated from a marine environment in South Africa. Appl. Biochem. Microbiol. 2014, 50, 49–54. [Google Scholar] [CrossRef]
- Dlamini, N.G. Biosynthesis of Copper Nanoparticles Using a Bioflocculant from Alcaligenis faecalis, Characterization and Its Application; University of Zululand: Richards Bay, South Africa, 2017. [Google Scholar]
- Ngema, S.; Basson, A.; Maliehe, T. Synthesis, characterization and application of polyacrylamide grafted bioflocculant. Phys. Chem. Earth Parts A/B/C 2020, 115, 102821. [Google Scholar] [CrossRef]
- Dlamini, N.G.; Basson, A.K.; Pullabhotla, V. Biosynthesis and characterization of copper nanoparticles using a bioflocculant extracted from Alcaligenis faecalis HCB2. Adv. Sci. Eng. Med. 2019, 11, 1064–1070. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Maliehe, T.S.; Basson, A.K.; Dlamini, N.G. Removal of pollutants in mine wastewater by a non-cytotoxic polymeric bioflocculant from Alcaligenes faecalis HCB2. Int. J. Environ. Res. Public Health 2019, 16, 4001. [Google Scholar] [CrossRef]
- Tsilo, P.H.; Basson, A.K.; Ntombela, Z.G.; Maliehe, T.S.; Pullabhotla, V.R. Production and characterization of a bioflocculant from Pichia kudriavzevii MH545928. 1 and Its Application in Wastewater Treatment. Int. J. Environ. Res. Public Health 2022, 19, 3148. [Google Scholar] [CrossRef]
- Masuku, S.K. Synthesis and Application of a Grafted Flocculant Produced from a Chemical Combination of a Bioflocculant TKT and Acrylamide (AM). Ph.D. Thesis, University of Zululand, Richards Bay, South Africa, 2019. [Google Scholar]
- Gohil, K.; Rajput, V.; Dharne, M. Pan-genomics of Ochrobactrum species from clinical and environmental origins reveals distinct populations and possible links. Genomics 2020, 112, 3003–3012. [Google Scholar] [CrossRef]
- Murínová, S.; Dercová, K. Potential use of newly isolated bacterial strain Ochrobactrum anthropi in bioremediation of polychlorinated biphenyls. Water Air Soil Pollut. 2014, 225, 1980. [Google Scholar] [CrossRef]
- Bezza, F.A.; Beukes, M.; Chirwa, E.M.N. Application of biosurfactant produced by Ochrobactrum intermedium CN3 for enhancing petroleum sludge bioremediation. Process. Biochem. 2015, 50, 1911–1922. [Google Scholar] [CrossRef]
- Kumar, S.S.; Kumar, M.S.; Siddavattam, D.; Karegoudar, T. Generation of continuous packed bed reactor with PVA–Alginate blend immobilized Ochrobactrum sp. DGVK1 cells for effective removal of N,N-dimethylformamide from industrial effluents. J. Hazard. Mater. 2012, 199, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Samaei, M.R.; Mortazavi, S.B.; Bakhshi, B.; Jafari, A.J.; Shamsedini, N.; Mehrazmay, H.; Ansarizadeh, M. Investigating the effects of combined bio-enhancement and bio-stimulation on the cleaning of hexadecane-contaminated soils. J. Environ. Chem. Eng. 2022, 10, 106914. [Google Scholar] [CrossRef]
- Tsegaye, B.; Balomajumder, C.; Roy, P. Biodegradation of wheat straw by Ochrobactrum oryzae BMP03 and Bacillus sp. BMP01 bacteria to enhance biofuel production by increasing total reducing sugars yield. Environ. Sci. Pollut. Res. 2018, 25, 30585–30596. [Google Scholar] [CrossRef]
- Loan, L.T.; Phuong, T.V.; Diep, C.N. Isolation and identification of bioflocculant-producing bacteria, heterotrophic nitrogen removal bacteria and poly-phosphate bacteria in wastewater from “My Tho rice noodle” factories, Tien Giang province, Vietnam. Int. J. Innov. Eng. Technol. 2018, 10, 185–198. [Google Scholar]
- Shahadat, M.; Teng, T.T.; Rafatullah, M.; Shaikh, Z.; Sreekrishnan, T.; Ali, S.W. Bacterial bioflocculants: A review of recent advances and perspectives. Chem. Eng. J. 2017, 328, 1139–1152. [Google Scholar] [CrossRef]
- Makapela, B.; Okaiyeto, K.; Ntozonke, N.; Nwodo, U.U.; Green, E.; Mabinya, L.V.; Okoh, A.I. Assessment of Bacillus pumilus isolated from fresh water milieu for bioflocculant production. Appl. Sci. 2016, 6, 211. [Google Scholar] [CrossRef]
- Deng, S.; Yu, G.; Ting, Y.P. Production of a bioflocculant by Aspergillus parasiticus and its application in dye removal. Colloids Surf. B Biointerfaces 2005, 44, 179–186. [Google Scholar] [CrossRef]
- Sheng, Y.; Zhang, Q.; Sheng, Y.; Li, C.; Wang, H. Screening and flocculating properties of bioflocculant-producing microorganisms. J. Univ. Sci. Technol. Beijing Miner. Metall. Mater. 2006, 13, 289–292. [Google Scholar] [CrossRef]
- Deng, S.; Bai, R.; Hu, X.; Luo, Q. Characteristics of a bioflocculant produced by Bacillus mucilaginosus and its use in starch wastewater treatment. Appl. Microbiol. Biotechnol. 2003, 60, 588–593. [Google Scholar] [CrossRef]
- Wang, S.-G.; Gong, W.-X.; Liu, X.-W.; Tian, L.; Yue, Q.-Y.; Gao, B.-Y. Production of a novel bioflocculant by culture of Klebsiella mobilis using dairy wastewater. Biochem. Eng. J. 2007, 36, 81–86. [Google Scholar] [CrossRef]
- Giri, S.S.; Harshiny, M.; Sen, S.S.; Sukumaran, V.; Park, S.C. Production and characterization of a thermostable bioflocculant from Bacillus subtilis F9, isolated from wastewater sludge. Ecotoxicol. Environ. Saf. 2015, 121, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Salehizadeh, H.; Shojaosadati, S. Isolation and characterisation a bioflocculant produced by Bacillus firmus. Biotechnol. Lett. 2002, 24, 35–40. [Google Scholar] [CrossRef]
- Mabrouk, M.E. Production of bioflocculant by the marine actinomycete Nocardiopsis aegyptia sp. nov. Life Sci. J. 2014, 11, 27–35. [Google Scholar]
- Mabinya, L.V.; Cosa, S.; Nwodo, U.; Okoh, A.I. Studies on bioflocculant production by Arthrobacter sp. Raats, a freshwater bacteria isolated from Tyume River, South Africa. Int. J. Mol. Sci. 2012, 13, 1054–1065. [Google Scholar] [CrossRef]
- Gong, W.-X.; Wang, S.-G.; Sun, X.-F.; Liu, X.-W.; Yue, Q.-Y.; Gao, B.-Y. Bioflocculant production by culture of Serratia ficaria and its application in wastewater treatment. Bioresour. Technol. 2008, 99, 4668–4674. [Google Scholar] [CrossRef]
- Nkosi, N.C.; Basson, A.K.; Ntombela, Z.G.; Maliehe, T.S.; Pullabhotla, R.V. Isolation, identification and characterization of bioflocculant-producing bacteria from activated sludge of Vulindlela Wastewater Treatment Plant. Appl. Microbiol. 2021, 1, 586–606. [Google Scholar] [CrossRef]
- Natarajan, K. Production and characterization of bioflocculants for mineral processing applications. Int. J. Miner. Process. 2015, 137, 15–25. [Google Scholar]
- Okaiyeto, K.; Nwodo, U.U.; Mabinya, L.V.; Okoh, A.I. Characterization and flocculating properties of a biopolymer produced by Halomonas sp. Okoh. Water Environ. Res. 2015, 87, 298–303. [Google Scholar] [CrossRef]
- Li, Z.; Chen, R.-W.; Lei, H.-Y.; Shan, Z.; Bai, T.; Yu, Q.; Li, H.-L. Characterization and flocculating properties of a novel bioflocculant produced by Bacillus circulans. World J. Microbiol. Biotechnol. 2009, 25, 745–752. [Google Scholar] [CrossRef]
- Cosa, S.; Mabinya, L.V.; Olaniran, A.O.; Okoh, O.O.; Bernard, K.; Deyzel, S.; Okoh, A.I. Bioflocculant production by Virgibacillus sp. Rob isolated from the bottom sediment of Algoa Bay in the Eastern Cape, South Africa. Molecules 2011, 16, 2431–2442. [Google Scholar] [CrossRef]
- Ajao, V.O. Flocculants from Wastewater; Wageningen University and Research: Wageningen, The Netherlands, 2020. [Google Scholar]
- Elkady, M.; Farag, S.; Zaki, S.; Abu-Elreesh, G.; Abd-El-Haleem, D. Bacillus mojavensis strain 32A, a bioflocculant-producing bacterium isolated from an Egyptian salt production pond. Bioresour. Technol. 2011, 102, 8143–8151. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Yu, J.; Xin, X.; Zou, C.; Cheng, Q.; Yang, H.; Nengzi, L. Characterization and flocculation mechanism of a bioflocculant from hydrolyzate of rice stover. Bioresour. Technol. 2015, 177, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Pathak, M.; Devi, A.; Bhattacharyya, K.; Sarma, H.; Subudhi, S.; Lal, B. Production of a non-cytotoxic bioflocculant by a bacterium utilizing a petroleum hydrocarbon source and its application in heavy metal removal. RSC Adv. 2015, 5, 66037–66046. [Google Scholar] [CrossRef]
- Sharma, V.; Kaur, T.; Bridle, H.; Ghosh, M. Antimicrobial efficacy and safety of mucoadhesive exopolymer produced by Acinetobacter haemolyticus. Int. J. Biol. Macromol. 2017, 94, 187–193. [Google Scholar] [CrossRef]
- Hu, Y.; Gu, Z.; He, J.; Li, Q. Novel strategy for controlling colloidal instability during the flocculation pretreatment of landfill leachate. Chemosphere 2022, 287, 132051. [Google Scholar] [CrossRef]
- Aljuboori, A.H.R.; Uemura, Y.; Osman, N.B.; Yusup, S. Production of a bioflocculant from Aspergillus niger using palm oil mill effluent as carbon source. Bioresour. Technol. 2014, 171, 66–70. [Google Scholar] [CrossRef]
- Zhao, H.; Liu, H.; Zhou, J. Characterization of a bioflocculant MBF-5 by Klebsiella pneumoniae and its application in Acanthamoeba cysts removal. Bioresour. Technol. 2013, 137, 226–232. [Google Scholar] [CrossRef]
- Kaur, R.; Roy, D.; Yellapu, S.K.; Tyagi, R.D.; Drogui, P.; Surampalli, R.Y. Enhanced composting leachate treatment using extracellular polymeric substances as bioflocculant. J. Environ. Eng. 2019, 145, 04019075. [Google Scholar] [CrossRef]





| Carbon Sources | FA(%) ± SD | Nitrogen Sources | FA(%) ± SD | Temperature (°C) | FA(%) ± SD |
|---|---|---|---|---|---|
| Sucrose | 81 ± 2.1 a | Casein | 89 ± 0.5 a | 20 | 77 ± 0.1 a |
| Molasses | 70 ± 2.2 a | Peptone | 88 ± 2.2 a | 25 | 84 ± 0.2 b |
| Lactose | 66 ± 4.6 b | Ammonia | 91 ± 4.7 a | 30 | 93 ± 0.4 c |
| Xylose | 76 ± 1.2 a | Urea | 85 ± 3.2 a | 35 | 82 ± 0.6 b |
| Sucrose | 43 ± 0.6 c | Yeast extract | 92 ± 3.8 a | 40 | 80 ± 0.1 b |
| Glucose | 86 ± 7.1 a | ||||
| Starch | 92 ± 1.7 a |
| Element | % (w/w) |
|---|---|
| Potassium | 0.2 |
| Oxygen | 43.3 |
| Magnesium | 1.7 |
| Sodium | 2.3 |
| Sulphur | 0.2 |
| Nitrogen | 2.5 |
| Chlorine | 1.3 |
| Calcium | 0.8 |
| Carbon | 45.6 |
| Phosphorus | 1.9 |
| pH | FA(%) ± SD | Cations | FA(%) ± SD |
|---|---|---|---|
| 3 | 74 ± 0.2 a | Control | 93 ± 3.5 a |
| 4 | 79 ± 0.9 a | Li+ | 88 ± 3.5 a |
| 5 | 90 ± 4.5 b | Na+ | 97 ± 2.7 a |
| 6 | 89 ± 0.2 a | K+ | 89 ± 3.8 a |
| 7 | 85 ± 0.4 b | Ba2+ | 96 ± 4.4 a |
| 8 | 85 ± 1.0 b | Mn2+ | 90±11.3 a |
| 9 | 84 ± 1.3 b | Mg2+ | 97 ± 1.5 a |
| 10 | 81 ± 1.7 a,b | Ca2+ | 96 ± 2.1 a |
| 11 | 80 ± 1.3 a,b | Fe3+ | 47 ± 1.0 b |
| 12 | 75 ± 0.4 a |
| Flocculants | COD (%) | BOD (%) | Sulphur |
|---|---|---|---|
| Bioflocculant | 98 ± 2.0 a | 91 ± 1.4 a | 86 ± 0.5 a |
| Aluminium sulphate | 97 ± 0.1 a | 82 ± 3.1 a | 90 ± 1.2 a |
| FeCl3 | 98 ± 1.8 a | 86 ± 0.5 a | 90 ± 0.1 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selepe, T.N.; Maliehe, T.S.; Moganedi, K.; Masoko, P.; Mulaudzi, V. Isolation and Optimisation of Culture Conditions for a Marine Bioflocculant-Producing Bacterium and Application of Its Bioflocculant in Wastewater Treatment. Int. J. Environ. Res. Public Health 2022, 19, 10237. https://doi.org/10.3390/ijerph191610237
Selepe TN, Maliehe TS, Moganedi K, Masoko P, Mulaudzi V. Isolation and Optimisation of Culture Conditions for a Marine Bioflocculant-Producing Bacterium and Application of Its Bioflocculant in Wastewater Treatment. International Journal of Environmental Research and Public Health. 2022; 19(16):10237. https://doi.org/10.3390/ijerph191610237
Chicago/Turabian StyleSelepe, Tlou Nelson, Tsolanku Sidney Maliehe, Kgabo Moganedi, Peter Masoko, and Vusimuzi Mulaudzi. 2022. "Isolation and Optimisation of Culture Conditions for a Marine Bioflocculant-Producing Bacterium and Application of Its Bioflocculant in Wastewater Treatment" International Journal of Environmental Research and Public Health 19, no. 16: 10237. https://doi.org/10.3390/ijerph191610237






