Association of Vegetable and Fruit Consumption with Urinary Oxidative Biomarkers in Teenaged Girls: A School-Based Pilot Study in Japan
Abstract
:1. Introduction
2. Subjects and Methods
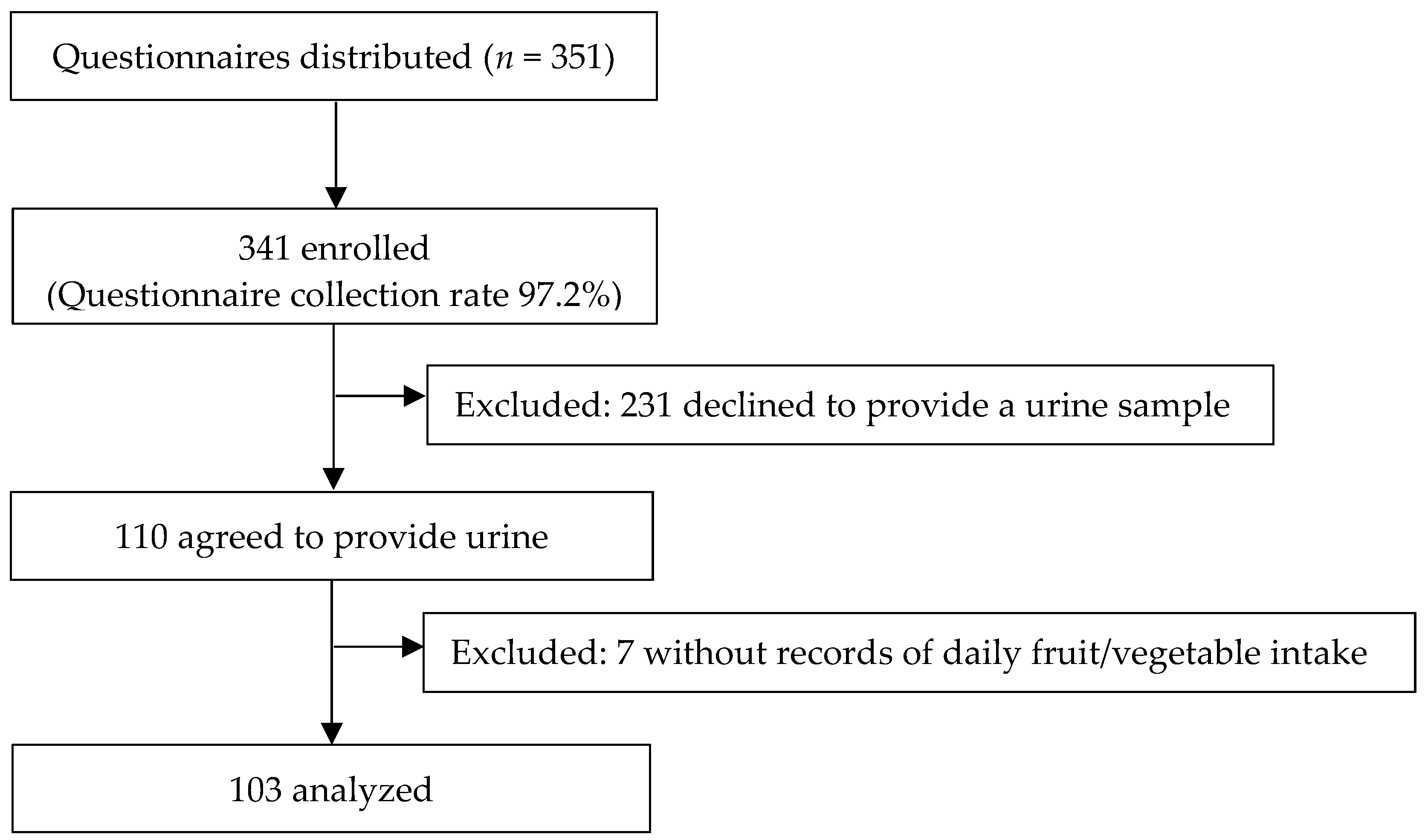
2.1. Study Participants and Study Design
2.2. Estimation of Usual Vegetable and Fruit Intake
2.3. Urine Sampling and Determination of Oxidative Biomarkers in Urine
2.4. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Halliwell, B.; Gutteridge, J.M.C. Oxidative stress. In Free Radicals in Biology and Medicine, 3rd ed.; Halliwell, B., Gutteridge, J.M.C., Eds.; Oxford University Press: New York, NY, USA, 1999; pp. 246–350. [Google Scholar]
- Sies, H. Oxidative stress: From basic research to clinical application. Am. J. Med. 1991, 91, 31S–38S. [Google Scholar] [CrossRef]
- Committee on Biological Markers of the National Research Council. Biological markers in environmental health research. Environ. Health Perspect. 1987, 74, 3–9. [Google Scholar]
- Bonassi, S.; Au, W.W. Biomarkers in molecular epidemiology studies for health risk prediction. Mutat. Res. 2002, 511, 73–86. [Google Scholar] [CrossRef]
- Ogino, K.; Wang, D.-H. Biomarkers of Oxidative/Nitrosative Stress: An Approach to Disease Prevention. Acta Med. Okayama 2007, 61, 181–189. [Google Scholar]
- Giulivi, C.; Davies, K.J.A. Dityrosine: A marker for oxidatively modified proteins and selective proteolysis. In Methods in Enzymology, Oxygen Radicals in Biological Systems Part C; Packer, L., Ed.; Academic Press Inc.: San Diego, CA, USA, 1994; Volume 233, pp. 363–367. [Google Scholar]
- Kato, Y.; Mori, Y.; Makino, Y.; Morimitsu, Y.; Hiroi, S.; Ishikawa, T.; Osawa, T. Formation of Nepsilon-(hexanonyl)lysine in protein exposed to lipid hydroperoxide. A plausible marker for lipid hydroperoxide-derived protein modification. J. Biol. Chem. 1999, 274, 20406–20414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cooke, M.S.; Evans, M.D.; Herbert, K.E.; Lunec, J. Urinary 8-oxo-2′-deoxyguanosine—Source, significance and supplements. Free Radic. Res. 2000, 32, 381–397. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Wu, X.; Naito, M.; Nomura, H.; Kitamoto, N.; Osawa, T. Immunochemical detection of protein dityrosine in atherosclerotic lesion of apo-E-deficient mice using a novel monoclonal antibody. Biochem. Biophys. Res. Commun. 2000, 275, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Naito, M.; Wu, X.; Nomura, H.; Kodama, M.; Kato, Y.; Kato, Y.; Osawa, T. The protective effects of tetrahydrocurcumin on oxidative stress in cholesterol-fed rabbits. J. Atheroscler. Thromb. 2002, 9, 243–250. [Google Scholar] [CrossRef] [Green Version]
- Ghezzo, A.; Visconti, P.; Abruzzo, P.M.; Bolotta, A.; Ferreri, C.; Gobbi, G.; Malisardi, G.; Manfredini, S.; Marini, M.; Nanetti, L.; et al. Oxidative stress and erythrocyte membrane alterations in children with autism: Correlation with clinical features. PLoS ONE 2013, 8, e66418. [Google Scholar] [CrossRef] [Green Version]
- Heinecke, J.W.; Li, W.; Daehnke, H.L.; Goldstein, J.A. Dityrosine, a specific marker of oxidation, is synthesized by the myeloperoxidase-hydrogen peroxide system of human neutrophils and macrophages. J. Biol. Chem. 1993, 268, 4069–4077. [Google Scholar] [CrossRef]
- Kanauchi, M.; Nishioka, H.; Hashimoto, T. Oxidative DNA damage and tubulointerstitial injury in diabetic nephropathy. Nephron 2002, 91, 327–329. [Google Scholar] [CrossRef] [PubMed]
- Akagi, S.; Nagake, Y.; Kasahara, J.; Sarai, A.; Kihara, T.; Morimoto, H.; Yano, A.; Nakao, K.; Nanba, K.; Ichikawa, H.; et al. Significance of 8-hydroxy-2′-deoxyguanosine levels in patients with chronic renal failure. Nephrology 2003, 8, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Chiou, C.C.; Chang, P.Y.; Chan, E.C.; Wu, T.L.; Tsao, K.C.; Wu, J.T. Urinary 8-hydroxydeoxyguanosine and its analogs as DNA marker of oxidative stress: Development of an ELISA and measurement in both bladder and prostate cancers. Clin. Chim. Acta 2003, 334, 87–94. [Google Scholar] [CrossRef]
- Loft, S.; Poulsen, H.E.; Vistisen, K.; Knudsen, L.E. Increased urinary excretion of 8-oxo-2′-deoxyguanosine, a biomarker of oxidative DNA damage, in urban bus drivers. Mutat. Res. 1999, 441, 11–19. [Google Scholar] [CrossRef]
- Barbato, D.L.; Tomei, G.; Tomei, F.; Sancini, A. Traffic air pollution and oxidatively generated DNA damage: Can urinary 8-oxo-7,8-dihydro-2-deoxiguanosine be considered a good biomarker? A meta-analysis. Biomarkers 2010, 15, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.W.; Chen, M.L.; Lung, S.C.; Tsai, C.J.; Yin, X.J.; Mao, I.F. Exposure assessment of PM2.5 and urinary 8-OHdG for diesel exhaust emission inspector. Sci. Total Environ. 2010, 408, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteride, J.M.C. Oxidative stress and redox regulation: Adaptation, damage, repair, senescence, and death. In Free Radicals in Biology and Medicine, 5th ed.; Halliwell, B., Gutteride, J.M.C., Eds.; Oxford University Press Inc.: New York, NY, USA, 2015; pp. 199–201. [Google Scholar]
- Schreck, R.; Rieber, P.; Baeuerle, P.A. Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kappa B transcription factor and HIV-1. EMBO J. 1991, 10, 2247–2258. [Google Scholar] [CrossRef]
- Veal, E.A.; Day, A.M.; Morgan, B.A. Hydrogen Peroxide Sensing and Signaling. Mol. Cell 2007, 26, 1–14. [Google Scholar] [CrossRef]
- Mathru, M.; Rooney, M.W.; Dries, D.J.; Hirsch, L.J.; Barnes, L.; Tobin, M.J. Urine hydrogen peroxide during adult respiratory distress syndrome in patients with and without sepsis. Chest 1994, 105, 232–236. [Google Scholar] [CrossRef] [Green Version]
- Campos, C.; Guzmán, R.; López-Fernández, E.; Casado, A. Evaluation of urinary biomarkers of oxidative/nitrosative stress in adolescents and adults with Down syndrome. Biochim. Biophys. Acta. 2011, 1812, 760–768. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, D.; Jacob, J.; Kunjamma, G.; Madhusoodanan, U.K.; Ghosh, S. Measurement of urinary hydrogen peroxide by FOX-1 method in conjunction with catalase in diabetes mellitus—A sensitive and specific approach. Clin. Chim. Acta 2004, 350, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Bailey, R.L.; Blumberg, J.B.; Burton-Freeman, B.; Chen, C.O.; Crowe-White, K.M.; Drewnowski, A.; Hooshmand, S.; Johnson, E.; Lewis, R.; et al. Fruits, vegetables, and health: A comprehensive narrative, umbrella review of the science and recommendations for enhanced public policy to improve intake. Crit. Rev. Food Sci. Nutr. 2020, 60, 2174–2211. [Google Scholar] [CrossRef] [Green Version]
- Lampe, J.W. Health effects of vegetables and fruit: Assessing mechanisms of action in human experimental studies. Am. J. Clin. Nutr. 1999, 70, 475–490. [Google Scholar] [CrossRef] [PubMed]
- Boeing, H.; Bechthold, A.; Bub, A.; Ellinger, S.; Haller, D.; Kroke, A.; Leschik-Bonnet, E.; Müller, M.J.; Oberritter, H.; Schulze, M.; et al. Critical review: Vegetables and fruit in the prevention of chronic diseases. Eur. J. Nutr. 2012, 51, 637–663. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Institute of Health and Nutrition. Outline for the Results of the National Health and Nutrition Survey Japan. Available online: https://www.mhlw.go.jp/content/10904750/000351576.pdf (accessed on 18 October 2021). (In Japanese).
- Ministry of Health, Labour and Welfare, Japan. Health Japan 21 (The National Health Promotion Movement in 21st Century). 2000. Available online: https://www.mhlw.go.jp/www1/topics/kenko21_11/pdf/all.pdf (accessed on 15 October 2021). (In Japanese).
- Ministry of Agriculture, Forestry and Fisheries, Japan. Guidelines for Balanced Diet. 2005. Available online: https://www.mhlw.go.jp/bunya/kenkou/pdf/eiyou-syokuji8.pdf (accessed on 15 October 2021).
- Verhagen, H.; de Vries, A.; Nijhoff, W.A.; Schouten, A.; van Poppel, G.; Peters, W.H.; van den Berg, H. Effect of Brussels sprouts on oxidative DNA-damage in man. Cancer Lett. 1997, 114, 127–130. [Google Scholar] [CrossRef]
- Young, J.F.; Nielsen, S.E.; Haraldsdóttir, J.; Daneshvar, B.; Lauridsen, S.T.; Knuthsen, P.; Crozier, A.; Sandström, B.; Dragsted, L.O. Effect of fruit juice intake on urinary quercetin excretion and biomarkers of antioxidative status. Am. J. Clin. Nutr. 1999, 69, 87–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iwasawa, H.; Morita, E.; Yui, S.; Yamasaki, M. Anti-oxidant Effects of Kiwi Fruit in Vitro and in Vivo. Biol. Pharm. Bull. 2011, 34, 128–134. [Google Scholar] [CrossRef] [Green Version]
- Duthie, S.J.; Duthie, G.G.; Russell, W.R.; Kyle, J.A.M.; Macdiarmid, J.I.; Rungapamestry, V.; Stephen, S.; Megias-Baeza, C.; Kaniewska, J.J.; Shaw, L.; et al. Effect of increasing fruit and vegetable intake by dietary intervention on nutritional biomarkers and attitudes to dietary change: A randomised trial. Eur. J. Nutr. 2018, 57, 1855–1872. [Google Scholar] [CrossRef] [Green Version]
- Francina, R.; Baldrick, J.; Elborn, S.; Woodside, J.V.; Treacy, K.; Bradley, J.M.; Patterson, C.C.; Schock, B.C.; Ennis, M.; Young, I.S.; et al. Effect of fruit and vegetable intake on oxidative stress and inflammation in COPD: A randomised controlled trial. Eur. Respir. J. 2012, 39, 1377–1384. [Google Scholar]
- Havas, S.; Treiman, K.; Langenberg, P.; Ballesteros, M.; Anliker, J.; Damron, D.; Feldman, R. Factors associated with fruit and vegetable consumption among women participating in WIC. J. Am. Diet. Assoc. 1998, 98, 1141–1148. [Google Scholar] [CrossRef]
- Wang, D.-H.; Kogashiwa, M.; Mori, N.; Yamashita, S.; Fujii, W.; Ueda, N.; Homma, H.; Suzuki, H.; Masuoka, N. Psychosocial Determinants of Fruit and Vegetable Consumption in a Japanese Population. Int. J. Environ. Res. Public Health 2016, 13, 786. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saito, S.; Yamauchi, H.; Hasui, Y.; Kurashige, J.; Ochi, H.; Yoshida, K. Quantitative determination of urinary 8-hydroxydeoxyguanosine (8-OH-dg) by using ELISA. Res. Commun. Mol. Pathol. Pharmacol. 2000, 107, 39–44. [Google Scholar] [PubMed]
- Sato, Y.; Ogino, K.; Sakano, N.; Wang, D.-H.; Yoshida, J.; Akazawa, Y.; Kanbara, S.; Inoue, K.; Kubo, M.; Takahashi, H. Evaluation of urinary hydrogen peroxide as an oxidative stress biomarker in a healthy Japanese population. Free Radic. Res. 2013, 47, 181–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strong, J.P.; McGill, H.C., Jr. The nature history of aortic atherosclerosis: Relationship to race, sex, and coronary lesions in New Orleans. Exp. Mol. Pathol. 1963, 52 (Suppl. 1), 15–27. [Google Scholar]
- McGill, H.C., Jr. Fatty streaks in the coronary arteries and aorta. Lab. Investig. 1968, 18, 560–564. [Google Scholar]
- Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group. Relationship of atherosclerosis in young men to serum lipoprotein cholesterol concentrations and smoking. A preliminary report from the Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group. JAMA 1990, 264, 3018–3024. [Google Scholar] [CrossRef]
- Sakai, K.; Kino, S.; Masuda, A.; Takeuchi, M.; Ochi, T.; Osredkar, J.; Rejc, B.; Gersak, K.; Ramarathnam, N.; Kato, Y. Determination of HEL (Hexanoyl-Lysine Adduct): A novel biomarker for omega-6 PUFA oxidation. In Lipid Hydroperoxide-Derived Modification of Biomolecules; Kato, Y., Ed.; Springer: Dordrecht, The Netherlands, 2014; Volume 77, pp. 61–72. [Google Scholar]
- Číž, M.; Čížová, H.; Denev, P.; Kratchanova, M.; Slavov, A.; Lojek, A. Different methods for control and comparison of the antioxidant properties of vegetables. Food Control 2010, 21, 518–523. [Google Scholar] [CrossRef]
- Lotito, S.B.; Frei, B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radic. Biol. Med. 2006, 41, 1727–1746. [Google Scholar] [CrossRef]
- Shan, B.; Cai, Y.Z.; Sun, M.; Corke, H. Antioxidant capacity of 26 spice extracts and characterization of their phenolic constituents. J. Agric. Food Chem. 2005, 53, 7749–7759. [Google Scholar] [CrossRef]
- Ping, Z.; Liu, W.; Kang, Z.; Cai, J.; Wang, Q.; Cheng, N.; Wang, S.; Wang, S.; Zhang, J.H.; Sun, X. Sulforaphane protects brains against hypoxic-ischemic injury through induction of Nrf2-dependent phase 2 enzyme. Brain Res. 2010, 1343, 178–185. [Google Scholar] [CrossRef]
- Li, B.; Tian, S.; Liu, X.; He, C.; Ding, Z.; Shan, Y. Sulforaphane protected the injury of human vascular endothelial cell induced by LPC through up-regulating endogenous antioxidants and phase II enzymes. Food Funct. 2015, 6, 1984–1991. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality—A systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef]
- Xie, Z.; Lin, H.; Fang, R.; Shen, W.; Li, S.; Chen, B. Effects of a fruit-vegetable dietary pattern on oxidative stress and genetic damage in coke oven workers: A cross-sectional study. Environ. Health 2015, 6, 40. [Google Scholar] [CrossRef] [Green Version]
- Briviba, K.; Bub, A.; Möseneder, J.; Schwerdtle, T.; Hartwig, A.; Kulling, S.; Watzl, B. No differences in DNA damage and antioxidant capacity between intervention groups of healthy, nonsmoking men receiving 2, 5, or 8 servings/day of vegetables and fruit. Nutr. Cancer 2008, 60, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Akagawa, M.; Shigemitsu, T.; Suyama, K. Production of hydrogen peroxide by polyphenols and polyphenol-rich beverages under quasi-physiological conditions. Biosci. Biotechnol. Biochem. 2003, 67, 2632–2640. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, H.; Honda, Y.; Nakagawa, S.; Ashida, H.; Kanazawa, K. Simultaneous determination of all polyphenols in vegetables, fruits, and teas. J. Agric. Food Chem. 2003, 51, 571–581. [Google Scholar] [CrossRef]
- Long, L.H.; Halliwell, B. Coffee drinking increases levels of urinary hydrogen peroxide detected in healthy human volunteers. Free Radic. Res. 2000, 32, 463–467. [Google Scholar] [CrossRef]
- Hiramoto, K.; Kida, T.; Kikugawa, K. Increased urinary hydrogen peroxide levels caused by coffee drinking. Biol. Pharm. Bull. 2002, 25, 1467–1471. [Google Scholar] [CrossRef] [Green Version]
- Halliwell, B.; Long, L.H.; Yee, T.P.; Lim, S.; Kelly, R. Establishing biomarkers of oxidative stress: The measurement of hydrogen peroxide in human urine. Curr. Med. Chem. 2004, 11, 1085–1092. [Google Scholar] [CrossRef]
- Giulivi, C.; Traaseth, N.J.; Davies, K.J.A. Tyrosine oxidation products: Analysis and biological relevance. Amino Acids 2003, 25, 227–232. [Google Scholar] [CrossRef]
- Giulivi, C.; Davies, K.J. Dityrosine and tyrosine oxidation products are endogenous markers for the selective proteolysis of oxidatively modified red blood cell hemoglobin by (the 19 S) proteasome. J. Biol. Chem. 1993, 268, 8752–8759. [Google Scholar] [CrossRef]
- Kato, Y.; Nagao, A.; Terao, J.; Osawa, T. Inhibition of myeloperoxidase-catalyzed tyrosylation by phenolic antioxidants in vitro. Biosci. Biotechnol. Biochem. 2003, 67, 1136–1139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Izabela, S.-B.; Sabina, G.; Grzegorz, B. P78—Polyphenols protect against protein glycoxidation. Free Radic. Biol. Med. 2014, 75 (Suppl. 1). [Google Scholar]
- Baba, S.; Osakabe, N.; Kato, Y.; Natsume, M.; Yasuda, A.; Kido, T.; Fukuda, K.; Muto, Y.; Kondo, K. Continuous intake of polyphenolic compounds containing cocoa powder reduces LDL oxidative susceptibility and has beneficial effects on plasma HDL-cholesterol concentrations in humans. Am. J. Clin. Nutr. 2007, 85, 709–717. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pennathur, S.; Wagner, J.D.; Leeuwenburgh, C.; Litwak, K.N.; Heinecke, J.W. A hydroxyl radical-like species oxidizes cynomolgus monkey artery wall proteins in early diabetic vascular disease. J. Clin. Investig. 2001, 107, 853–860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kato, Y.; Dozaki, N.; Nakamura, T.; . Kitamoto, N.; Yoshida, A.; Naito, M.; Kitamura, M.; Osawa, T. Quantification of modified tyrosines in healthy and diabetic human urine using liquid chromatography/tandem mass spectrometry. J. Clin. Biochem. Nutr. 2009, 44, 67–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anwar, A.; Abruzzo, P.M.; Pasha, S.; Rajpoot, K.; Bolotta, A.; Ghezzo, A.; Marini, M.; Posar, A.; Visconti, P.; Thornalley, P.J.; et al. Advanced glycation endproducts, dityrosine and arginine transporter dysfunction in autism—Aa source of biomarkers for clinical diagnosis. Mol. Autism. 2018, 9, 3. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteride, J.M.C. Measurement of reactive species. In Free Radicals in Biology and Medicine, 5th ed.; Halliwell, B., Gutteride, J.M.C., Eds.; Oxford University Press Inc.: New York, NY, USA, 2015; pp. 284–353. [Google Scholar]
- Leeuwenburgh, C.; Hansen, P.A.; Holloszy, J.O.; Heinecke, J.W. Oxidized amino acids in the urine of aging rats: Potential markers for assessing oxidative stress in vivo. Am. J. Physiol. 1999, 276, 128. [Google Scholar] [CrossRef]
- Orhan, H.; van Holland, B.; Krab, B.; Moeken, J.; Vermeulen, N.P.; Hollander, P.; Meerman, J.H. Evaluation of a multi-parameter biomarker set for oxidative damage in man: Increased urinary excretion of lipid, protein and DNA oxidation products after one hour of exercise. Free Radic. Res. 2004, 38, 1269–1279. [Google Scholar] [CrossRef]
- Leeuwenburgh, C.; Heinecke, J.W. Oxidative stress and antioxidants in exercise. Curr. Med. Chem. 2001, 8, 829–838. [Google Scholar] [CrossRef] [Green Version]
- Ristow, M.; Zarse, K.; Oberbach, A.; Klöting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.R.; Blüher, M. Antioxidants prevent health-promoting effects of physical exercise in humans. Proc. Natl. Acad. Sci. USA 2009, 106, 8665–8670. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, F.; Li, J.; Liu, Z.; Chuang, C.-C.; Yang, W.; Zuo, L. Redox mechanism of reactive oxygen species in exercise. Front. Physiol. 2016, 7, 486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| mean ± SD (n) | |||
|---|---|---|---|
| Age | 15.6 ± 1.7 (103) | ||
| BMI | 20.7 ± 2.3 (103) | ||
| n (%) | |||
| Education | |||
| Junior high school students | 33 (32.0) | ||
| High school students | 70 (68.0) | ||
| Awareness of the recommended amount of daily vegetable intake | |||
| Yes | 6 (5.8) | ||
| No | 97 (94.2) | ||
| Awareness of the recommended amount of daily fruit intake | |||
| Yes | 2 (1.9) | ||
| No | 101 (98.1) | ||
| Self–reported daily vegetable intake (g/day) | |||
| <250 | 72 (69.9) | ||
| 250–349 | 11 (10.7) | ||
| ≥350 | 20 (19.4) | ||
| Self–reported daily fruit intake (g/day) | |||
| <100 | 32 (31.1) | ||
| 100–199 | 47 (45.6) | ||
| ≥200 | 24 (23.3) | ||
| Physical exercise at least once a week | |||
| Yes | 72 (69.9) | ||
| No | 31 (30.1) | ||
| Oxidative biomarkers in urine | median (min, max) | ||
| HEL (nmol/g Cr) | 86.22 (10.38, 884.71) | ||
| DT (µmol/g Cr) | 2.55 (0.56, 11.2) | ||
| 8-OHdG (ng/mg Cr) | 7.29 (2.22, 23.21) | ||
| H2O2 (µmol/g Cr) | 45.85 (0.5, 281.06) | ||
| Fruit | |||
| <100 g/d (n = 32) | ≥100 g/d (n = 71) | p Value | |
| HEL (nmol/g Cr) | 95.09 (835.22) | 79.24 (379.04) | 0.033 |
| 8-OHdG (ng/mg Cr) | 7.90 (15.46) | 7.07 (20.81) | 0.127 |
| DT (μmol/g Cr) | 2.81 (6.18) | 2.41 (10.42) | 0.385 |
| H2O2 (µmol/g Cr) | 40.14 (249.33) | 57.88 (277.55) | 0.092 |
| Vegetable | |||
| <250 g/d (n = 72) | ≥250 g/d (n = 31) | p Value | |
| HEL (nmol/g Cr) | 89.86 (874.33) | 70.33 (222.69) | 0.052 |
| 8-OHdG (ng/mg Cr) | 7.41 (20.99) | 7.01 (15.29) | 0.278 |
| DT (μmol/g Cr) | 2.45 (10.63) | 2.80 (5.82) | 0.774 |
| H2O2 (µmol/g Cr) | 43.27 (280.56) | 69.31 (198.99) | 0.349 |
| Vegetable Intake | Fruit Intake | Adjusted R2 a | |||
|---|---|---|---|---|---|
| β | p | β | p | ||
| HEL | −0.332 | 0.014 | 0.171 | 0.213 | 0.060 |
| 8-OHdG | 0.002 | 0.987 | 0.106 | 0.437 | 0.067 |
| DT | 0.302 | 0.026 | −0.019 | 0.890 | 0.047 |
| H2O2 | 0.121 | 0.253 | 0.039 | 0.715 | 0.089 |
| Physical Exercise at Least Once a Week | p Value | ||
|---|---|---|---|
| No (n = 31) | Yes (n = 72) | ||
| Urinary HEL (nmol/g Cr) | 140.7 (32.3, 352.6) | 71.2(10.4, 884.7) | 0.002 |
| Urinary 8-OHdG (ng/mg Cr) | 10.0 (4.2, 23.2) | 6.6 (2.2, 15.4) | <0.001 |
| Urinary DT (µmol/g Cr) | 3.6 (0.6, 11.2) | 2.2 (0.8, 5.5) | <0.001 |
| Urinary H2O2 (µmol/g Cr) | 42.3 (0.5, 249.8) | 47.2 (9.7, 281.1) | 0.930 |
| BMI (kg/m2) | ||
|---|---|---|
| r | p Value | |
| Fruit intake (g/day) | −0.015 | 0.878 |
| Vegetable intake (g/day) | −0.125 | 0.211 |
| HEL (nmol/g Cr) | −0.094 | 0.348 |
| 8-OHdG (ng/mg Cr) | −0.149 | 0.136 |
| DT (μmol/g Cr) | −0.056 | 0.573 |
| H2O2 (µmol/g Cr) | −0.033 | 0.745 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sato, Y.; Yamada, A.; Miyanaga, M.; Wang, D.-H. Association of Vegetable and Fruit Consumption with Urinary Oxidative Biomarkers in Teenaged Girls: A School-Based Pilot Study in Japan. Int. J. Environ. Res. Public Health 2022, 19, 10474. https://doi.org/10.3390/ijerph191710474
Sato Y, Yamada A, Miyanaga M, Wang D-H. Association of Vegetable and Fruit Consumption with Urinary Oxidative Biomarkers in Teenaged Girls: A School-Based Pilot Study in Japan. International Journal of Environmental Research and Public Health. 2022; 19(17):10474. https://doi.org/10.3390/ijerph191710474
Chicago/Turabian StyleSato, Yoshiko, Ai Yamada, Masamitsu Miyanaga, and Da-Hong Wang. 2022. "Association of Vegetable and Fruit Consumption with Urinary Oxidative Biomarkers in Teenaged Girls: A School-Based Pilot Study in Japan" International Journal of Environmental Research and Public Health 19, no. 17: 10474. https://doi.org/10.3390/ijerph191710474





