Abstract
With decreasing mortality, the quality of life, spiritual needs, and mental health of breast cancer patients have become increasingly important. Demoralization is a poor prognostic factor for cancer patients. The extent of demoralization in breast cancer patients and its association with these factors remains unclear. This cross-sectional study was conducted at a Taiwanese medical center. We enrolled 121 participants (34 with high demoralization and 87 with low demoralization, as per the Mandarin Version of Demoralization Scale). High demoralization was associated with reduced quality of life, sleep quality, and spiritual interests. Multivariate analyses revealed that the scores of the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire ≥ 62.5 (OR = 0.21, p = 0.002) and Spiritual Interests Related to Illness Tool Chinese Version ≥ 3.66 (OR = 0.11, p < 0.001) were associated with low demoralization. Demoralized patients with depression had a poorer quality of life and sleep quality. Although not statistically significant, depressed and demoralized participants were at a higher risk of suicide. Cancer patients with both depression and demoralization had the worst prognosis. Breast cancer patients exhibited demoralization when they had unmet bio-psycho-social-spiritual needs. An early assessment of demoralization may improve holistic healthcare for breast cancer patients.
1. Introduction
Breast cancer accounts for 11.7% of all new cancer cases [1]. Female breast cancer has surpassed lung cancer as the most commonly diagnosed cancer, with an estimated 2.3 million new cases (11.7%) and a 6.9% mortality rate [2]. By 2040, the burden of breast cancer is predicted to increase to over 3 million new cases and 1 million deaths every year owing to population growth and aging alone [3]. In 2019, data from the Taiwan Cancer Registry Center showed that breast cancer is fourth in the whole cause of cancer mortality, and the second leading cause of cancer death in women. Breast cancer is also the most common cancer among women and the whole population [4]. The literature shows that breast cancer screening and adjuvant therapy have important roles in reducing breast cancer mortality [5,6]. Research shows that multidisciplinary teams can help reduce breast cancer mortality by integrating surgery, radiation, and medical oncology [7]. Breast cancer mortality rates declined between 1975 and 2011 [8]. With this decreasing mortality, the quality of life, spiritual needs, and mental health [9] of breast cancer patients have become increasingly important. However, aside from the biological aspect of breast cancer, the psychological and spiritual needs of patients with breast cancer are often not met, and related research is lacking.
1.1. Demoralization and Breast Cancer
Demoralization is a normal psychological response to painful, advanced, terminal diseases [10,11,12,13], and/or heroin addiction [14]. Demoralization includes symptoms in five domains: loss of meaning, dysphoria, disheartenment, helplessness, and sense of failure, which can be assessed using rating scales [15,16]. The specific manifestation of demoralization differs from the American Psychiatric Association’s diagnostic criteria for major depressive disorder. In the diagnosis, there is a focus on depression and anhedonia. Currently, demoralization does not refer to a specific brain pathology, and the understanding of demoralization is not comprehensive. Fang et al. reported that 42% (n = 200) of cancer patients in Taiwan were highly demoralized [17]. Nevertheless, the incidence and severity of different types of cancer remain unclear. Previous studies on demoralization and breast cancer treatment strategies have been limited to countries such as Taiwan, China, Turkey, and Lebanon [18,19,20,21]. They have found that demoralization in breast cancer is related to breast and hair loss [20,21]. Triple-negative breast cancer (TNBC) is a poor prognostic factor for breast cancer [22]. Patients with breast cancer and TNBC may have a higher risk of demoralization. However, the association between demoralization and TNBC is not well understood.
1.2. Demoralization and Its Association with Quality of Life, Sleep Quality, Spiritual Interests and Suicide
Quality of life is an important factor that may have a bi-directional relationship with demoralization in cancer patients [23,24]. However, the negative associations with breast cancer remain unclear. Patients with breast cancer may suffer from REM or NREM sleep problems [25,26]. A previous study revealed that demoralization mediates the effect of stress on sleep disturbances in patients with breast cancer [18]. An assessment of the association between demoralization and sleep quality is important. Furthermore, spiritual care for cancer patients must be interdisciplinary and culturally appropriate. Research exploring spirituality and spiritual care among cancer patients in Asian countries or globally is scarce [27]. Demoralization is associated with spiritual interests in oral cancer [23]. An assessment of demoralization may help in understanding spiritual care in breast cancer.
Depression and suicide rates are high among breast cancer survivors [28]. The increased risk of suicide in breast cancer survivors is likely underestimated, as suicide is often classified under “other causes of death”, and this may occur more often in women who have cancer. Suicide almost always occurs among individuals with mental health disorders, particularly depression [29]. Extant literature distinguishes between demoralization and depression; however, the two are strongly correlated in cancer patients [17,30,31,32,33,34]. Depression and demoralization are both high-risk factors for suicide in cancer patients, and patients with both are at higher risk [23,24,35]. Demoralization is independently associated with suicide after controlling for the severity of depression [36]. Nevertheless, the association between demoralization and depression in patients with breast cancer remains understudied.
1.3. The Present Study
There are several research gaps concerning the demoralization of breast cancer. First, previous studies did not confirm the severity and prevalence of demoralization in patients with breast cancer. Second, demoralization is a psychological response of patients with breast cancer; its associations with quality of life, sleep quality, and spiritual interests are not well understood. Third, the topic of whether quality of life, sleep quality, and spiritual interests can predict demoralization in patients with breast cancer has not been determined. Fourth, depression and its association with demoralization, quality of life, sleep quality, spiritual interest, and suicide in patients with highly demoralized breast cancer remain unclear. To evaluate these issues in inpatients settings, this cross-sectional study assessed demoralization in breast cancer inpatients and its relationship with demographic factors, quality of life, sleep quality, spiritual fulfillment, and suicide risk. Additionally, the study analyzed the differences between depressed and non-depressed patients with demoralization.
2. Materials and Methods
2.1. Participants
Convenience sampling was used to recruit participants (N = 121). Researchers expected to collect more measures in the inpatient settings. Breast cancer patients hospitalized in medical care for surgery, chemotherapy, symptom relief, and radiotherapy at Taichung Veterans General Hospital (Taichung, Taiwan) from August to December 2019 were invited to participate. The inclusion criteria were a diagnosis of breast cancer, age ≥20 years, and the ability to communicate in Mandarin.
2.2. Measures
Trained research assistants administered the following surveys and assessed relevant sociodemographic factors (age, sex, education, marital status, religious affiliation, employment status, and monthly income), cancer status (stage, location, and TNBC), treatment in current hospitalization, and time since diagnosis.
2.2.1. Mandarin Version of Demoralization Scale (DS-MV)
DS-MV is a demoralization scale developed by Kissane [16]. It spans five distinct dimensions: loss of meaning (five items), disheartenment (six items), dysphoria (five items), sense of failure (four items), and helplessness (four items). The DS-MV demonstrated high reliability (overall Cronbach’s alpha = 0.928; subscale Cronbach’s alpha range = 0.63–0.85). Research has also shown a positive correlation (γ = 0.703, p < 0.001) between DS-MV and Beck Hopelessness Scale scores, and a negative correlation (γ = −0.680, p < 0.001) between DS-MV and McGill Quality of Life Questionnaire scores. DS-MV scores greater than 30 indicate high demoralization [15,16].
2.2.2. Chinese Version of European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30)
The European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30) has been translated and validated in over 100 languages and used in numerous studies worldwide. We obtained the Chinese version of the EORTC QLQ-C30 in Taiwan with an agreement license from the Quality of Life Unit of the EORTC Data Center. The questionnaire contained 30 items evaluating general health, with specific physical, emotional, and social domains. Items were grouped into five functional scales: physical, role, cognitive, emotional, and social. It also has three symptom scales (fatigue, nausea, and pain), as well as six individual items evaluating the intensity of the following symptoms: dyspnea, sleeplessness, lack of appetite, constipation, diarrhea, and financial problems. The last two items constitute the overall health assessment. Items use a 4-point Likert response scale (1 = never to 4 = often) [37], with higher scores indicating higher levels of the given construct. A high global health status represents a high quality of life, and a high symptom score represents a high level of symptoms.
2.2.3. Chinese Version of the Pittsburgh Sleep Quality Index (CPSQI)
The Pittsburgh Sleep Quality Index (PSQI) evaluates subjective sleep quality. There are 19 items assessing sleep patterns and quality over the past month. Four items concern sleep timing, with multiple-choice questions based on sleep quality. The 19 items span seven dimensions: sleep quality, habitual sleep efficiency, sleep latency, daytime dysfunction, sleep duration, sleep disturbances, and the use of sleep medications. The sum of the responses on these seven components constitutes a global score ranging from 0 to 21, with lower scores indicating better sleep quality. Scores greater than 5 indicate possible sleep pathology [38]. Subsequently, the CPSQI was developed and properly translated, becoming a reliable and valid tool to screen for sleep pathology. It is a useful self-administered tool with high sensitivity. Scores greater than 5 on the CPSQI yield 98% sensitivity and 55% specificity [39].
2.2.4. Spiritual Interests Related to Illness Tool Chinese Version (C-SpIRIT)
Taylor developed the 44-item SpIRIT to measure the fulfillment of spiritual interests among cancer patients and their families [40]. It spans eight dimensions: possessing a positive perspective, having a relationship with God, giving love to others, receiving love from others, revaluating beliefs, seeking meaning in life, practicing religion, and preparing for death. Items use a 5-point Likert response scale, with higher scores indicating stronger spiritual interests in a specific category. Lin [41] modified the Chinese version of the scale for Taiwanese patients. This version contains 21 items spanning five dimensions: beliefs/religion, positive attitude toward life, love to/from others, seeking the meaning of life, and a peaceful mind.
2.2.5. Patient Health Questionnaire (PHQ-9)
The PHQ-9 is a nine-item instrument administered to patients in primary care settings to screen for depression (presence and severity). This can be used to establish a diagnosis of depression according to the DSM-5 criteria. Test–retest reliability was assessed by computing the correlation (0.84) between the PHQ-9 scores obtained from in-person and phone interviews with the same patients [42]. In an assessment of construct validity, the correlation between the scores on the PHQ-9 and SF-20 mental health scales was 0.73. To assess criterion validity, a mental health professional validated depression diagnoses using PHQ-9 scores from 580 participants, resulting in 88% sensitivity and 88% specificity [42].
2.2.6. Questionnaire on Suicide Risk
The DS-MV and PHQ-9 define suicide risk in distinct ways. First, items from the DS-MV that indicate suicide risk include items 14 (“Life is no longer worth living”) and 20 (“I would rather not be alive”). Answering “yes” to at least one of these statements indicated a higher risk of suicide. Second, responses greater than 0 on item 9 of the PHQ-9 (“Thoughts that you would be better off dead, or thoughts of hurting yourself in some way?”) indicated a higher risk of suicide.
2.3. Data Analysis
We divided participants into high- and low-demoralization groups based on the DS-MV cutoff score of 30 [15,16]. We used the receiver operating characteristic (ROC) curve classification to demoralize the associations between quality of life and spiritual interests. Furthermore, the high-demoralization group completed the PHQ-9, and was subdivided into depressed and non-depressed groups based on a cutoff score of 10 [42]. Sociodemographic and cancer status data were expressed as frequencies and percentages. We evaluated between-group differences using chi-square tests, Fisher’s exact tests, Mann-Whitney tests, and Spearman’s rank correlation coefficients. We then estimated the EORTC QLQ-C30, Global CPSQI, and C-SpIRIT scores associated with demoralization using logistic regression. All tests were set at a significance level of 0.05. All analyses were performed using IBM SPSS (version 22.0, IBM, New York, USA).
3. Results
3.1. Sociodemographic Characteristics and Demoralization in Breast Cancer Patients
There were 34 participants (28.10%) in the high-demoralization group and 87 (71.90%) in the low-demoralization group (Table 1). Sociodemographic and cancer status variables, such as age, tumor stage, TNBC, time since diagnosis, recurrence, marital status, religious affiliation, employment status, and income, did not differ significantly between the groups. Treatment during hospitalization did not differ significantly, except for participants hospitalized for surgery (low demoralization: 25/87 (28.74%) and high demoralization: 3/34 (8.82%) (p = 0.036)). Education levels were significantly different between the groups (p = 0.024). Those in the low-demoralization group had completed mainly senior high school (40/87) (45.98%) and college (36/87) (41.38%), while those in the high-demoralization group had completed college (19/34) (55.88%), senior high school (8/34) (23.53%), and elementary school (4/34) (11.76%).

Table 1.
The associations between sociodemographic characteristics and demoralization.
3.2. Demoralization’s Relationship with Quality of Life, Sleep Quality, and Spiritual Interests
The mean DS-MV scores were 25.12 ± 14.89, 18.02 ± 9.34, and 43.29 ± 10.29 for all participants, the low-demoralization group, and the high-demoralization group, respectively. The mean EORTC QLQ-C30 scores were 60.20 ± 24.03, 65.80 ± 22.17, and 45.85 ± 22.88 for all participants, the low-demoralization group, and the high-demoralization group, respectively (p < 0.001). The mean CPSQI scores were 8.45 ± 4.51, 7.86 ± 4.28, and 10.06 ± 4.75 for all participants, the low-demoralization group, and the high-demoralization group, respectively (p = 0.022). The mean C-SpIRIT scores were 3.85 ± 0.58, 3.98 ± 0.48, and 3.50 ± 0.66 for all participants, the low-demoralization group, and the high-demoralization group, respectively (p < 0.001). For quality of life, high demoralization was associated with lower scores on the EORTC QLQ-C30; role, emotional, cognitive, and social functioning; fatigue; pain; insomnia; and appetite loss. High demoralization was associated with lower global CPSQI scores, poorer subjective sleep quality, higher sleep latency, and daytime dysfunction. High demoralization was associated with low spiritual fulfillment in all the domains (Table 2).

Table 2.
The associations between variables of rating scales and demoralization.
3.3. ROC Curve Analysis and Univariate and Multivariate Analyses of Variables Associated with Demoralization
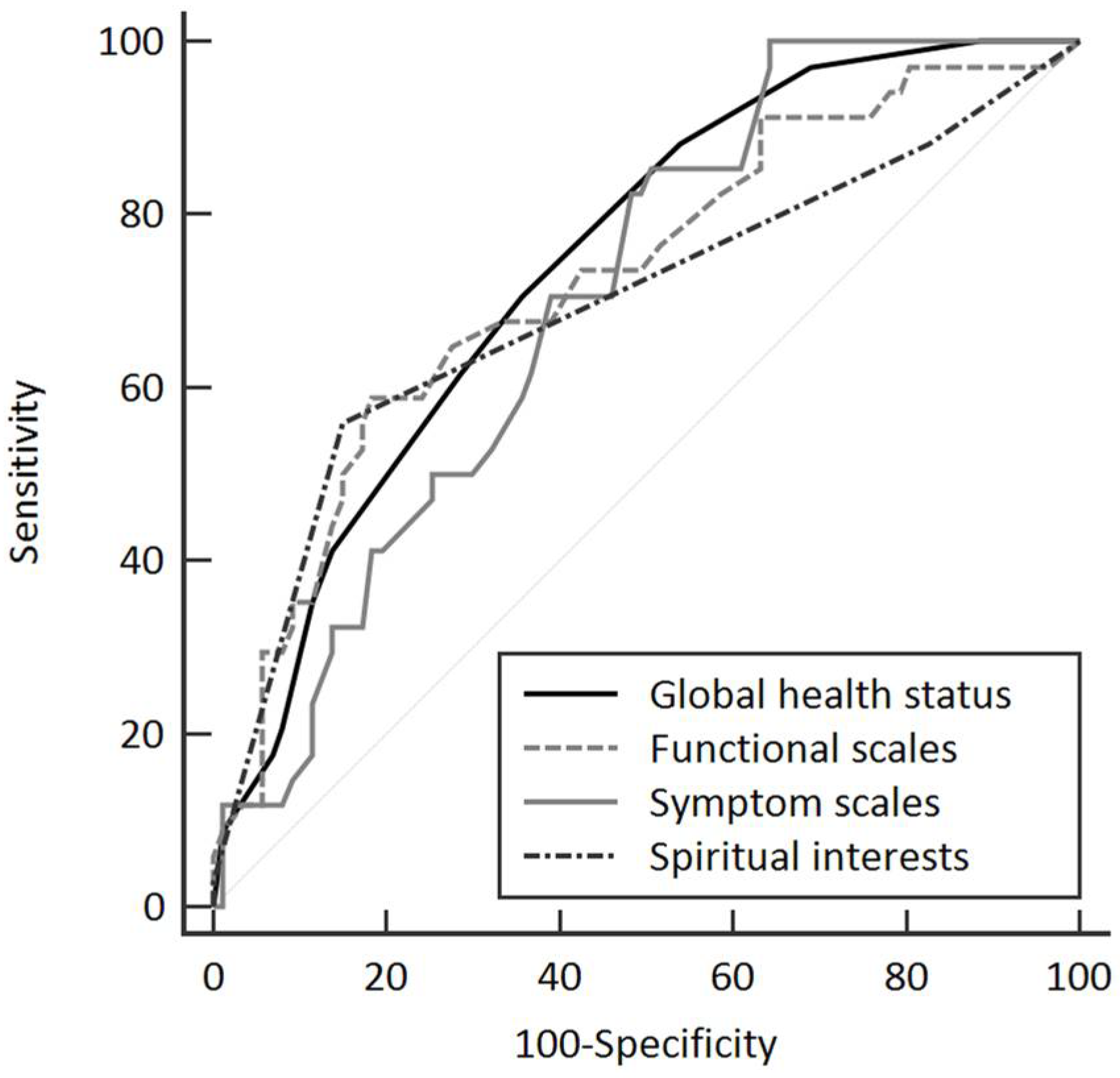
Logistic regression and ROC curves were used to determine correlations between the overall quality of life, sleep quality, spiritual interests, and demoralization status. Scores on the EORTC QLQ-C30 ≥ 62.5, functional scales ≥ 72.7, symptom scales ≥ 20.3, and C-SpIRIT ≥ 3.66 were associated with demoralization (Figure 1; Table 2). The results of univariate and multivariate analyses showed that the scores for C-SpIRIT (OR = 0.15, p < 0.001), EORTC QLQ-C30 ≥ 62.5 (OR = 0.21, p = 0.002), and C-SpIRIT ≥ 3.66 (OR = 0.11, p < 0.001) were strongly associated with demoralization (Table 3).
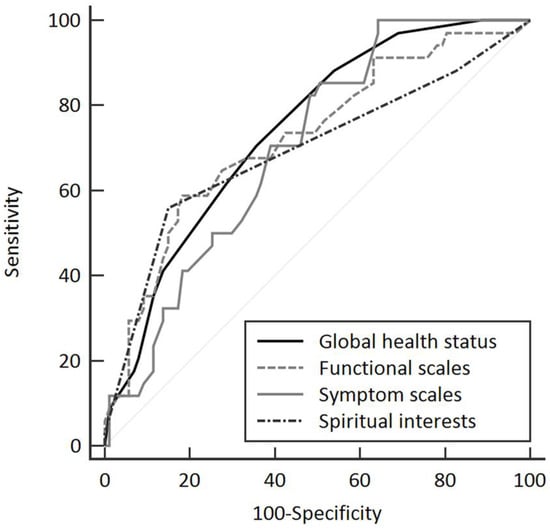
Figure 1.
Receiver operating characteristic curve analysis of the scores of European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30) and spiritual interests (total scores of Spiritual Interests Related to Illness Tool Chinese Version) for demoralized patients. Global health status is the total scores of EORTC QLQ-C30.

Table 3.
Univariate and multivariate analyses of demoralization in all patients.
3.4. Depression in the High-Demoralization Group
There were 15/34 (44.12%) participants in the high-demoralization group with PHQ-9 scores of ≥ 10. Sociodemographic and cancer status variables, such as age, tumor stage, TNBC, treatment, recurrence, marital status, religious affiliation, employment status, and income, did not differ significantly between the subgroups. The time since diagnosis was significantly different between the depression and non-depression subgroups (p = 0.040). The time since diagnosis was 3 months to 1 year (10/19) (52.63%) in the non-depression subgroup and <3 months (9/15) (60.00%) or 3 months to 1 year (5/15) (33.33%) in the depression subgroup. Education level was significantly different between the depression and non-depression subgroups (p = 0.042). The majority of those in the non-depression group had completed college (14/19) (73.68%), while those in the depression group had completed senior high school (6/15) (40.00%) and college (5/15) (33.33%). Work (part-time or full-time) was significantly different between the depression (46.67%) and non-depression (84.21%) subgroups (p = 0.030) (Table 4).

Table 4.
The associations between sociodemographic characteristics and depression in the high-demoralization group.
The mean DS-MV scores were 48.20 ± 11.43 and 39.42 ± 7.52 for the depression and non-depression subgroups, respectively (p = 0.010). The mean EORTC QLQ-C30 scores were 30.53 ± 16.24 and 57.95 ± 20.11 for the depression and non-depression group subgroups, respectively (p < 0.001). The mean CPSQI scores were 12.27 ± 4.57 and 8.32 ± 4.22 for the depression and non-depression subgroups, respectively (p = 0.017). The mean C-SpIRIT scores did not differ significantly between the depression and non-depression subgroups (Table 5).

Table 5.
The associations between the variables of rating scales and depression in the high-demoralization group.
3.5. Suicide Risk in Breast Cancer Patients
Suicide risk did not differ significantly between the high (5.88%) and low (1.65%) demoralization groups or between the depressed (6.67%) and non-depressed (5.26%) subgroups when defining suicide risk per responses to items 14 and 20 on the DS-MV; it was also not significantly different between the depressed (20.00%) and non-depressed (10.52%) subgroups when defining suicide risk per responses to item 9 of the PHQ-9 (Table 2 and Table 5).
4. Discussion
This study addresses the research gap concerning spiritual and mental healthcare in patients with breast cancer. First, demoralization is common in patients with breast cancer. High demoralization in breast cancer was associated with reduced quality of life, sleep quality, and spiritual interests. Reduced quality of life and spiritual interests might predict demoralization in patients with breast cancer, and strategies that improve quality of life and spiritual interests may prevent demoralization. We found that depression is common in highly demoralized breast cancer patients. Demoralized breast cancer patients with depression may have the worst quality of life and sleep quality. Depression was not associated with spiritual interests in demoralized breast cancer patients; thus, improving depression may not improve spiritual care in highly demoralized patients. Therefore, specific treatment that focuses on demoralization is required. Furthermore, demoralized patients with depression may have a higher suicide risk than non-depressed patients.
In this study, the educational level was associated with demoralization. Previous studies have shown that more educated women may experience higher levels of distress owing to increased knowledge about their disease and associated therapies [43]. However, they may also have more opportunities to learn coping strategies and receive psychosocial support and treatment resources. Past research has also shown that providing appropriate educational models can improve palliative care for cancer patients [44], and that education, monthly income, and family support are protective factors for demoralization [45]. Clinicians must provide straightforward and specific coping strategies, and psychotherapy for less educated patients.
In this study, demoralization was not associated with TNBC or the tumor stage. We found that demoralization was more significantly associated with mental health, spiritual interests, and quality of life than biological factors, such as TNBC or tumor stage. Although TNBC is not associated with demoralization, it has a particularly poor prognosis [22]. Future studies should explore demoralization in patients with TNBC. In this study, participants hospitalized for surgery were, on average, less demoralized. According to a recent study, demoralization fluctuates during the treatment period [46]. Participants may perceive surgery as preferable to chemotherapy/radiotherapy, which causes long-term side effects and complications. The treatment course and cancer stage are important factors in demoralization. Patients with cancer at end-of-life stages may have higher levels of demoralization [47]. However, a longitudinal study is required in this field.
4.1. Demoralization and Quality of Life, Sleep Quality, and Spiritual Interests
This study found that demoralized breast cancer patients may have reduced quality of life, sleep quality, and spiritual interests. The results of this study are consistent with those of a Taiwanese study on the prevalence and severity of demoralization in breast cancer patients [18]. Demoralization is a poor prognostic factor for breast cancer care; this finding is consistent with those of a similar study on oral cancer [23], although patients with oral cancer had a higher prevalence and severity of demoralization than patients with breast cancer [17,23]. The prevalence and severity of demoralization differ among cancer types. Clinicians should provide individual assessments and psychosocial interventions tailored to different cancer patients.
This study suggests that spiritual interests and quality of life are strong predictors of demoralization in patients with breast cancer. Research on spiritual care of patients with breast cancer is limited. Further evaluation of demoralization may lead to a deeper understanding of spiritual care for breast cancer. Demoralization should be examined in a specific population of patients with breast cancer. We found that demoralization was associated with poor sleep quality, specifically subjective sleep quality, sleep latency, and daytime dysfunction. However, sleep disturbances were not a strong predictor of demoralization in multivariate analyses. Previous studies have shown that demoralization mediates the relationship between stress, sleep disturbances, and mental health [18]. However, the relationship between sleep quality and demoralization remains unclear. Demoralization and its causal or bidirectional relationship with quality of life, sleep quality, and spiritual interests should be established in future studies. Furthermore, demoralization may be a prognostic factor or predictor of quality of cancer care because of its association with indicators of holistic cancer care.
4.2. Quality of Life and Sleep Quality in Depressed, Demoralized Breast Cancer Patients
In this study, demoralized patients with depression had poorer quality of life and sleep quality, and 60% of the patients with depression had received their diagnosis within the last 3 months. Therefore, clinicians must assess and treat depression at an early stage. We confirmed that the proportion of employed individuals in the depression subgroup was lower than that in the non-depressed subgroup. An impaired ability to work is another important issue in cancer care. In a study by Kim et al., nearly one-third of participants reported that their work conditions had changed following cancer treatment; in the depressed group, the prevalence of decreased productivity, which is associated with depression, demoralization, and anxiety, was four times that in the non-depressed group [48]. Additionally, depression was associated with higher PSQI scores (poorer subjective sleep quality) among highly demoralized patients. These results align with those of a previous longitudinal study on depression as a predictor of sleep disturbance in women with metastatic breast cancer [49]. Depressed and demoralized patients experienced a more severe loss of meaning and helplessness, and had the worst quality of life compared to non-depressed patients. Depression was not significantly associated with spiritual interests. Our research highlights the need for spiritual care in breast cancer patients with high levels of demoralization. Treating depression alone does not constitute adequate holistic care.
4.3. Suicide Risk in Depressed, Demoralized Patients
Suicide risk, as defined by items 14 and 20 of the DS-MV, was relatively lower among all our participants than among oral cancer patients [23]. A previous cohort study showed that increased suicide risk was particularly prominent in cancer patients with poor prognoses [50]. Although breast cancer is associated with a lower suicide rate than other cancers, suicide risk increases as the stage of cancer increases [51]. We found that, as defined by item 9 of the PHQ-9, 20% of the depressed subgroup of highly demoralized patients was at risk of suicide. This subgroup also had a worse quality of life and sleep quality than the non-depressed subgroup. The risk of suicide may be underestimated in patients with breast cancer. Depressed and demoralized patients have a higher suicide risk than non-depressed patients. The combination of demoralization and depression is a poor prognostic factor for patients with breast cancer. Clinicians must carefully assess the suicide risk, demoralization, and depression in this population.
4.4. Limitations
First, this study was cross-sectional and based on inpatients from a single institute in Taiwan, and the results may only be applied to the population of inpatients breast cancer patients. Further large-scale, longitudinal, and multicenter studies are needed to clarify these findings. Second, other physical or psychiatric diseases, treatment history, family history, treatment methods, side effects, and other relevant information concerning patients must be evaluated to improve the quality of the research. Third, this study only assessed depression in the high-demoralization group. The comorbidities of demoralization and depression in patients with breast cancer require further study. Fourth, the reliability and validity of the DS-MV and PHQ-9 in assessing suicide risk have not yet been established, which may affect the interpretability of the results. Standard suicide assessment tools, such as the Beck Scale for Suicide Ideation, should be considered in future studies. In addition, although both men and women could participate in the study, no male participants participated, and the results may only be applied to the population of female breast cancer patients.
5. Conclusions
This study addressed a research gap in the field of holistic breast cancer care, and provided evidence that demoralization in breast cancer is associated with reduced quality of life, sleep quality, and spiritual interests. Quality of life and spiritual interests could be predictors of demoralization. In this study, highly demoralized breast cancer patients with depression had the worst quality of life and sleep quality. Breast cancer patients with demoralization had higher suicidal ideation (2.94–5.88%), although the difference was not significant. In participants with high demoralization and depression, suicidal ideation increased to 6.67–20.00%. In the future, demoralization and its causal or bidirectional relationship with quality of life, sleep quality, and spiritual interests should be established. The DS-MV is a self-report questionnaire that can be used as a routine screening tool to predict the quality of life, suicidal ideation, and spiritual interests among cancer patients. Therefore, further large-scale longitudinal studies are warranted.
Author Contributions
Conceptualization: T.-G.C., C.-C.H. and T.-T.Y.; methodology: T.-G.C. and T.-T.Y.; data curation: T.-G.C., P.-C.H. and C.-C.H.; writing—original draft, T.-G.C.; supervision: T.-T.Y.; formal analysis: T.-T.Y. and C.-Y.H.; writing—reviewing and editing: T.-T.Y.; funding acquisition: T.-T.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Taichung Veterans General Hospital, Taiwan (serial number: TCVGH-YM107034).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of Taichung Veterans General Hospital, Taiwan (serial number: CE18244B).
Informed Consent Statement
Informed consent was obtained from all the subjects involved in the study.
Data Availability Statement
Not applicable.
Acknowledgments
We wish to acknowledge Taichung Veterans General Hospital and all patients who participated in this study.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have influenced this study. The funders had no role in the study design; the collection, analyses, or interpretation of data; the writing of the manuscript; or the decision to publish the results.
References
- Wild, C.P.; Weiderpass, E.; Stewart, B.W. World Cancer Report: Cancer Research for Cancer Prevention; The International Agency for Research on Cancer (IARC): Lyon, France, 2020. [Google Scholar]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.; Morgan, E.; Rumgay, H.; Mafra, A.; Singh, D.; Laversanne, M.; Vignat, J.; Gralow, J.R.; Cardoso, F.; Siesling, S.; et al. Current and future burden of breast cancer: Global statistics for 2020 and 2040. Breast 2022, 66, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Taiwan Cancer Registery Center. Top 10 cancers in Taiwan, 2019. In Taiwan Cancer Registery Center; Taiwan Cancer Registery Center: Taipei, Taiwan, 2019. [Google Scholar]
- De Gelder, R.; Heijnsdijk, E.A.; Fracheboud, J.; Draisma, G.; de Koning, H.J. The effects of population-based mammography screening starting between age 40 and 50 in the presence of adjuvant systemic therapy. Int. J. Cancer 2015, 137, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Munoz, D.; Near, A.M.; van Ravesteyn, N.T.; Lee, S.J.; Schechter, C.B.; Alagoz, O.; Berry, D.A.; Burnside, E.S.; Chang, Y.; Chisholm, G.; et al. Effects of screening and systemic adjuvant therapy on ER-specific US breast cancer mortality. J. Natl. Cancer Inst. 2014, 106, dju289. [Google Scholar] [CrossRef] [PubMed]
- Kesson, E.M.; Allardice, G.M.; George, W.D.; Burns, H.J.; Morrison, D.S. Effects of multidisciplinary team working on breast cancer survival: Retrospective, comparative, interventional cohort study of 13 722 women. BMJ 2012, 344, e2718. [Google Scholar] [CrossRef]
- Kohler, B.A.; Sherman, R.L.; Howlader, N.; Jemal, A.; Ryerson, A.B.; Henry, K.A.; Boscoe, F.P.; Cronin, K.A.; Lake, A.; Noone, A.M.; et al. Annual Report to the Nation on the Status of Cancer, 1975–2011, Featuring Incidence of Breast Cancer Subtypes by Race/Ethnicity, Poverty, and State. J. Natl. Cancer Inst. 2015, 107, djv048. [Google Scholar] [CrossRef]
- Kornblith, A.B.; Herndon, J.E., 2nd; Weiss, R.B.; Zhang, C.; Zuckerman, E.L.; Rosenberg, S.; Mertz, M.; Payne, D.; Jane Massie, M.; Holland, J.F.; et al. Long-term adjustment of survivors of early-stage breast carcinoma, 20 years after adjuvant chemotherapy. Cancer 2003, 98, 679–689. [Google Scholar] [CrossRef]
- Mehnert, A.; Vehling, S.; Höcker, A.; Lehmann, C.; Koch, U. Demoralization and depression in patients with advanced cancer: Validation of the German version of the demoralization scale. J. Pain Symptom. Manage. 2011, 42, 768–776. [Google Scholar] [CrossRef]
- Robinson, S.; Kissane, D.W.; Brooker, J.; Burney, S. A Review of the Construct of Demoralization: History, Definitions, and Future Directions for Palliative Care. Am. J. Hosp. Palliat. Care 2016, 33, 93–101. [Google Scholar] [CrossRef]
- Kissane, D.W.; Clarke, D.M.; Street, A.F. Demoralization syndrome—A relevant psychiatric diagnosis for palliative care. J. Palliat. Care 2001, 17, 12–21. [Google Scholar] [CrossRef]
- Townsend, K. Demoralisation in palliative care. Lancet Oncol. 2018, 19, 168. [Google Scholar] [CrossRef]
- Chang, T.G.; Yen, T.T.; Hsu, W.Y.; Chang, S.M. Frontal Lobe Functions, Demoralization, Depression and Craving as Prognostic Factors and Positive Outcomes of Patients with Heroin Use Disorder Receiving 6 Months of Methadone Maintenance Treatment. Int. J. Environ. Res. Public Health 2022, 19, 3703. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.H.; Chang, Y.F.; Yang, Y.C.; Liu, C.L.; Hsieh, R.K.; Leu, Y.S.; Chen, Y.J.; Wang, T.E.; Tsai, L.Y.; Liu, S.I.; et al. Evaluation of the Reliability and Validity of the Mandarin Version of Demoralization Scale for Cancer Patients. J. Intern. Med. Taiwan 2010, 21, 427–435. (In Chinese) [Google Scholar]
- Kissane, D.W.; Wein, S.; Love, A.; Lee, X.Q.; Kee, P.L.; Clarke, D.M. The Demoralization Scale: A report of its development and preliminary validation. J. Palliat. Care 2004, 20, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.K.; Chang, M.C.; Chen, P.J.; Lin, C.C.; Chen, G.S.; Lin, J.; Hsieh, R.K.; Chang, Y.F.; Chen, H.W.; Wu, C.L.; et al. A correlational study of suicidal ideation with psychological distress, depression, and demoralization in patients with cancer. Support. Care Cancer 2014, 22, 3165–3174. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.L.; Hsueh, H.W.; Chang, Y.H.; Li, R.H. The Mediation and Suppression Effect of Demoralization in Breast Cancer Patients After Primary Therapy: A Structural Equation Model. J. Nurs. Res. 2021, 29, e144. [Google Scholar] [CrossRef]
- Sun, F.K.; Hung, C.M.; Yao, Y.; Fu, C.F.; Tsai, P.J.; Chiang, C.Y. The Effects of Logotherapy on Distress, Depression, and Demoralization in Breast Cancer and Gynecological Cancer Patients: A Preliminary Study. Cancer Nurs. 2021, 44, 53–61. [Google Scholar] [CrossRef]
- Doumit, M.A.; El Saghir, N.; Abu-Saad Huijer, H.; Kelley, J.H.; Nassar, N. Living with breast cancer, a Lebanese experience. Eur. J. Oncol. Nurs. 2010, 14, 42–48. [Google Scholar] [CrossRef]
- Cebeci, F.; Yangin, H.B.; Tekeli, A. Life experiences of women with breast cancer in south western Turkey: A qualitative study. Eur. J. Oncol. Nurs. 2012, 16, 406–412. [Google Scholar] [CrossRef]
- Foulkes, W.D.; Smith, I.E.; Reis-Filho, J.S. Triple-negative breast cancer. N. Engl. J. Med. 2010, 363, 1938–1948. [Google Scholar] [CrossRef]
- Chang, T.G.; Huang, P.C.; Hsu, C.Y.; Yen, T.T. Demoralization in oral cancer inpatients and its association with spiritual needs, quality of life, and suicidal ideation: A cross-sectional study. Health Qual. Life Outcomes 2022, 20, 60. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.T.; Lin, Y.A.; Pan, Y.X.; Lin, J.L.; Lin, X.J.; Zhang, J.; Huang, F.F. Understanding factors influencing demoralization among cancer patients based on the Bio-Psycho-Social Model: A systematic review. Psychooncology, 2022; in press. [Google Scholar] [CrossRef] [PubMed]
- Savard, J.; Ivers, H.; Villa, J.; Caplette-Gingras, A.; Morin, C.M. Natural course of insomnia comorbid with cancer: An 18-month longitudinal study. J. Clin. Oncol. 2011, 29, 3580–3586. [Google Scholar] [CrossRef] [PubMed]
- Perrier, J.; Duivon, M.; Clochon, P.; Rehel, S.; Doidy, F.; Grellard, J.M.; Segura-Djezzar, C.; Geffrelot, J.; Emile, G.; Allouache, D.; et al. Sleep macro- and microstructure in breast cancer survivors. Sci. Rep. 2022, 12, 2557. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H. Spiritual Care for Cancer Patients. Asia Pac. J. Oncol. Nurs. 2019, 6, 101–103. [Google Scholar] [CrossRef]
- Carreira, H.; Williams, R.; Funston, G.; Stanway, S.; Bhaskaran, K. Associations between breast cancer survivorship and adverse mental health outcomes: A matched population-based cohort study in the United Kingdom. PLoS Med. 2021, 18, e1003504. [Google Scholar] [CrossRef]
- Barraclough, B.; Bunch, J.; Nelson, B.; Sainsbury, P. A hundred cases of suicide: Clinical aspects. Br. J. Psychiatry 1974, 125, 355–373. [Google Scholar] [CrossRef]
- Tang, P.L.; Wang, H.H.; Chou, F.H. A Systematic Review and Meta-Analysis of Demoralization and Depression in Patients with Cancer. Psychosomatics 2015, 56, 634–643. [Google Scholar] [CrossRef]
- Tang, L.; Li, Z.; Pang, Y. The differences and the relationship between demoralization and depression in Chinese cancer patients. Psychooncology 2020, 29, 532–538. [Google Scholar] [CrossRef]
- Scandurra, C.; Mangiapia, F.; La Rocca, R.; Di Bello, F.; De Lucia, N.; Muzii, B.; Cantone, M.; Zampi, R.; Califano, G.; Maldonato, N.M.; et al. A cross-sectional study on demoralization in prostate cancer patients: The role of masculine self-esteem, depression, and resilience. Support. Care Cancer 2022, 30, 7021–7030. [Google Scholar] [CrossRef]
- Nanni, M.G.; Caruso, R.; Travado, L.; Ventura, C.; Palma, A.; Berardi, A.M.; Meggiolaro, E.; Ruffilli, F.; Martins, C.; Kissane, D.; et al. Relationship of demoralization with anxiety, depression, and quality of life: A Southern European study of Italian and Portuguese cancer patients. Psychooncology 2018, 27, 2616–2622. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.J.; Quan, M.M.; Gao, L.; Li, Q.; Yan, C.X.; Zhang, Q.; Shi, B.X. Demoralization and depression in Chinese cancer patients. Support. Care Cancer 2021, 29, 6211–6216. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Hu, D.; Liu, Y.; Han, Y.; Guo, X.; Teng, F.; Zhou, Y. Relationship of Suicidal Ideation With Demoralization, Depression, and Anxiety: A Study of Cancer Patients in Mainland China. J. Nerv. Ment. Dis. 2019, 207, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Vehling, S.; Kissane, D.W.; Lo, C.; Glaesmer, H.; Hartung, T.J.; Rodin, G.; Mehnert, A. The association of demoralization with mental disorders and suicidal ideation in patients with cancer. Cancer 2017, 123, 3394–3401. [Google Scholar] [CrossRef]
- Kaasa, S.; Bjordal, K.; Aaronson, N.; Moum, T.; Wist, E.; Hagen, S.; Kvikstad, A. The EORTC core quality of life questionnaire (QLQ-C30): Validity and reliability when analysed with patients treated with palliative radiotherapy. Eur. J. Cancer 1995, 31A, 2260–2263. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F., 3rd; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Tsai, P.S.; Wang, S.Y.; Wang, M.Y.; Su, C.T.; Yang, T.T.; Huang, C.J.; Fang, S.C. Psychometric evaluation of the Chinese version of the Pittsburgh Sleep Quality Index (CPSQI) in primary insomnia and control subjects. Qual. Life Res. 2005, 14, 1943–1952. [Google Scholar] [CrossRef]
- Taylor, E.J. Spiritual needs of patients with cancer and family caregivers. Cancer Nurs. 2003, 26, 260–266. [Google Scholar] [CrossRef]
- Lin, Y.L.; Rau, K.M.; Liu, Y.H.; Lin, Y.H.; Ying, J.; Kao, C.C. Development and validation of the Chinese Version of Spiritual Interests Related Illness Tool for patients with cancer in Taiwan. Eur. J. Oncol. Nurs. 2015, 19, 589–594. [Google Scholar] [CrossRef]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B. The PHQ-9: Validity of a brief depression severity measure. J. Gen. Intern. Med. 2001, 16, 606–613. [Google Scholar] [CrossRef]
- Van Onselen, C.; Paul, S.M.; Lee, K.; Dunn, L.; Aouizerat, B.E.; West, C.; Dodd, M.; Cooper, B.; Miaskowski, C. Trajectories of sleep disturbance and daytime sleepiness in women before and after surgery for breast cancer. J. Pain Symptom Manag. 2013, 45, 244–260. [Google Scholar] [CrossRef] [PubMed]
- Prince, H.; Nadin, S.; Crow, M.; Maki, L.; Monture, L.; Smith, J.; Kelley, M.L. If you understand you cope better with it: The role of education in building palliative care capacity in four First Nations communities in Canada. BMC Public Health 2019, 19, 768. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Ho, C.H.; Wang, H.H. Protective Factors of Demoralization among Cancer Patients in Taiwan: An Age-matched and Gender-matched Study. Asian Nurs. Res. Korean Soc. Nurs. Sci. 2017, 11, 174–179. [Google Scholar] [CrossRef]
- Tang, P.L.; Lin, H.S.; Wang, H.H.; Hu, L.Y.; Chou, F.H. Demoralization level fluctuated at various time frame of radiotherapy for patients with different cancers: A longitudinal observational study. BMC Palliat. Care 2022, 21, 143. [Google Scholar] [CrossRef] [PubMed]
- Bovero, A.; Botto, R.; Adriano, B.; Opezzo, M.; Tesio, V.; Torta, R. Exploring demoralization in end-of-life cancer patients: Prevalence, latent dimensions, and associations with other psychosocial variables. Palliat. Support. Care 2019, 17, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Kissane, D.W.; Richardson, G.; Senior, J.; Morgan, J.; Gregory, P.; Birks, S.; Ooi, C.; Lipton, L.; Antill, Y.; et al. The role of depression and other psychological factors in work ability among breast cancer survivors in Australia. Psychooncology 2022, 31, 167–175. [Google Scholar] [CrossRef]
- Palesh, O.G.; Collie, K.; Batiuchok, D.; Tilston, J.; Koopman, C.; Perlis, M.L.; Butler, L.D.; Carlson, R.; Spiegel, D. A longitudinal study of depression, pain, and stress as predictors of sleep disturbance among women with metastatic breast cancer. Biol. Psychol. 2007, 75, 37–44. [Google Scholar] [CrossRef]
- Fang, F.; Fall, K.; Mittleman, M.A.; Sparen, P.; Ye, W.; Adami, H.O.; Valdimarsdottir, U. Suicide and cardiovascular death after a cancer diagnosis. N. Engl. J. Med. 2012, 366, 1310–1318. [Google Scholar] [CrossRef]
- Schairer, C.; Brown, L.M.; Chen, B.E.; Howard, R.; Lynch, C.F.; Hall, P.; Storm, H.; Pukkala, E.; Anderson, A.; Kaijser, M.; et al. Suicide after breast cancer: An international population-based study of 723,810 women. J. Natl. Cancer Inst. 2006, 98, 1416–1419. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).