Abstract
Breast cancer is the most common malignancy in women, with a complex clinical path that involves several professionals and that requires a multidisciplinary approach. However, the effectiveness of breast cancer multidisciplinary care and the processes that contribute to its effectiveness have not yet been firmly determined. This study aims to evaluate the impact of multidisciplinary tumor boards on breast cancer care outcomes. A systematic literature review was carried out through Scopus, Web of Science and Pubmed databases. The search was restricted to articles assessing the impact of MTB implementation on breast cancer care. Fourteen studies were included in the review. The most analyzed outcomes were diagnosis, therapy and survival. Four out of four studies showed that, with implementation of an MTB, there was a change in diagnosis, and all reported changes in the treatment plan after MTB implementation. A pooled analysis of three studies reporting results on the outcome “mortality” showed a statistically significant 14% reduction in mortality relative risk for patients enrolled versus not enrolled in an MTB. This study shows that MTB implementation is a valuable approach to deliver appropriate and effective care to patients affected by breast cancer and to improve their outcomes.
Keywords:
tumor board; multidisciplinary team; breast cancer; healthcare; outcomes; diagnosis; treatment; survival 1. Introduction
Breast cancer is the most common malignancy in women [1]. In 2020, there were 2.3 million women diagnosed with breast cancer and 685,000 deaths globally. At the end of 2020, there were 7.8 million women alive who had been diagnosed with breast cancer in the past five years, making it the world’s most prevalent cancer [2]. The risk factors include age, reproductive factors, hormonal factors, dietary and metabolic factors, previous thoracic radiotherapy, previous dysplasia or breast cancer, family history and genetic predisposition [3]. Due to screening and greater awareness amongst women, most breast malignancies are diagnosed at an early stage when surgical treatment can more often be conservative and the therapy adopted more effective, with, in consequence, higher five-year survival rates [4].
As with other cancers, breast cancer treatment requires a highly complex approach, and quality care is dependent on coordinated multidisciplinary input [5,6].
The path of the patient diagnosed with neoplasia is complex and involves several professionals, from the surgeon to the psychotherapist. In this context, structured healthcare is necessary to improve outcomes and promote the coordinated management of the patient’s care. Therefore, to pursue quality objectives in healthcare, a multidisciplinary approach has become essential and represents the current trend in the United Kingdom, Europe, the United States, Asia and Australia [7]. Multidisciplinarity represents an organisational response to the need to make increasingly complex clinical decisions and, consequently, represents an essential aspect of oncological treatment. Moreover, many scientific studies reported in the literature describe multidisciplinary tumor boards (MTBs) as the therapeutic standard in oncology [8]. MTBs consist of multidisciplinary teams composed of different clinical specialists working together to make shared decisions regarding the clinical pathway of cancer patients. This approach ensures that all professionals involved in the oncological clinical pathway take part in the process of diagnosis, planning, assessment and treatment [9]. MTBs have also been reported to have an impact on cancer care, in terms of change to treatment strategies and processes, clinical results improvement, reduction in clinical outcomes variability, better use of economic resources and improvement in healthcare personnel training [10].
With respect to breast cancer, in many countries, multidisciplinary care has been formally established as an essential practice in its management and has been used as a benchmark for accreditation and funding [11]. Research into multidisciplinary breast cancer care varies by design, clinical context and study outcomes considered [12]. According to J. Shao et al., 2019, there is growing emphasis on the application of multidisciplinary approaches to breast cancer care. However, because of heterogeneous definitions and contexts, the effectiveness of multidisciplinary care and the processes that contribute to its effectiveness have not yet been determined with certainty [12].
Starting from an umbrella review published by Specchia ML et al. in 2020 [9], regarding the impact of MTBs on oncological patients’ care in general, the present systematic review aims to evaluate the impact of MTBs on breast cancer care outcomes.
2. Materials and Methods
A systematic literature review was performed based on a structured search process using a specific search algorithm, an accurate study selection process, data extraction and quality assessment of the included studies.
A systematic search of studies was carried out through Scopus, Web of Science and Pubmed databases using the following search string: (“breast neoplasms”[Mesh] OR “breast cancer*” OR “breast neoplasm*” OR “breast tumor*” OR “breast carcinoma”) AND (“patient care team”[Mesh] OR “patient care team*” OR “tumor board*” OR “multidisciplinary team*” OR “multidisciplinarity” OR “MDT” OR “multidisciplinary team meeting*” OR “multidisciplinary meeting*” OR “cancer conference*” OR “multidisciplinary care conference*” OR “MCC” OR “cancer meeting*” OR “healthcare team*” OR “interdisciplinary health team*” OR “clinical board*” OR “multidisciplinary treatment” OR “multidisciplinary management” OR “multidisciplinary care” OR “teamwork” OR “team work”) AND (“outcome*” OR “process” OR “assessment” OR “evaluation*” OR “efficacy” OR “efficiency” OR “effectiveness” OR “cost*” OR “caseload” OR “workload” OR “plan” OR “planning” OR “decision making” OR “impact” OR “personal*” OR “precision medicine”).
The review considered all studies published from 1995 to 28 June 2021. This year range was set since the multidisciplinary approach in cancer care was not regularly implemented in clinical practice until the late 1990’s, more specifically, from 1997, though it has been described in the literature from 1975 [9]. The retrieved studies were independently assessed by two researchers by first reading the title, and, if deemed suitable, then reading the abstract, before finally reading the full text based on the inclusion and exclusion criteria described below. Uncertainties arising regarding the inclusion of articles were overcome by discussion amongst the research members.
Inclusion and exclusion criteria were defined according to the PICO (Population, Intervention, Comparison, Outcome) model as follows: (P) The studies considered were focused on breast cancer patients; (I) The intervention was represented by multidisciplinary teams in a hospital setting. A multidisciplinary team was defined as a team composed of professionals from different clinical backgrounds who make decisions together to recommend the clinical pathway of an individual patient [9]; (C) The comparison was with lack of MTB implementation (patients’ clinical management without the intervention of multidisciplinary teams); (O) Both clinical outcomes (e.g., measurable change in symptoms, overall health, quality of life, or survival/mortality) and process-related outcomes (regarding the decisions made about the clinical journey in terms of diagnosis and therapy) were taken into account. The search was restricted to articles assessing the impact of MTB implementation, written in the English or Italian language, published between 1995 and 2021, and for which full text was available. Reviews, letters and commentaries were excluded because they do not report original data.
Articles were selected according to the PRISMA statement criteria and the data were extracted independently by two researchers. For each study the researchers retrieved and summarized data related to the following: authors, year of publication and country of origin, study population, study design, method/intervention, main outcomes, main findings and quality assessment.
To assess the quality of the included studies the Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies and the Quality Assessment Tool for Before-After (Pre-Post) Studies With No Control Group [13] from the National Heart, Lung and Blood Institute Study Quality Assessment Tools were used. The quality assessment was carried out by two researchers independently with disagreements discussed until consensus was reached. The Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies consists of fourteen questions with three possible answers for each item: ‘Yes’, ‘No’ or ‘Other’ (cannot determine, not applicable, not reported). Observational studies were labeled as of ‘Good’, ‘Fair’, or ‘Poor’ quality based on the cut-off points assigned: ‘Good’ quality (above 9 points), ‘Fair’ quality (between 9 and 5 points), ‘Poor’ quality (below 5 points). The Quality Assessment Tool for Before-After (Pre-Post) Studies With No Control Group consists of twelve items and the possible answers for each item are: ‘Yes’, ‘No’ or ‘Other’. With the same methodology, based on points assignment, before-after (pre-post) studies were labeled as of ‘Good’ quality (above 8 points), ‘Fair’ quality (between 8 and 4 points), or ‘Poor’ quality (below 4 points).
To combine non-homogeneous results, when appropriate (at least two studies reporting comparable outcomes), a meta-analysis was performed. Data regarding the detailed inclusion criteria, duration of follow-up and outcome (adjusted hazard risks (HRs and 95% confidence intervals (CIs)) for patients enrolled and not enrolled in MTBs) were extracted from each individual study. Study-specific estimates were combined using inverse variance-weighted averages of logarithmic HRs in both fixed and random-effects models (primary meta-analysis). Heterogeneity was assessed by Chi2 and I2 test. If no significant heterogeneity was observed (I2 ≤ 50%, p > 0.1), a fixed-effects model was adopted; otherwise, a random-effects model was applied [14]. Meta-analysis was performed using the Review Manager software (version 5.4.1).
3. Results
A total of 8998 records were retrieved from the search, of which 5303 were on Pubmed, 2014 on Scopus and 1691 on the Web of Science. After removal of duplicates, the final number of records retrieved was 6658. Selection by title and abstract reduced the total to 71 studies that were assessed for eligibility for reading the full text. Of these, 14 studies, satisfying the stated inclusion/exclusion criteria, were eligible for the study and were included in the review. A PRISMA flowchart provides details of the study selection process (Figure 1).
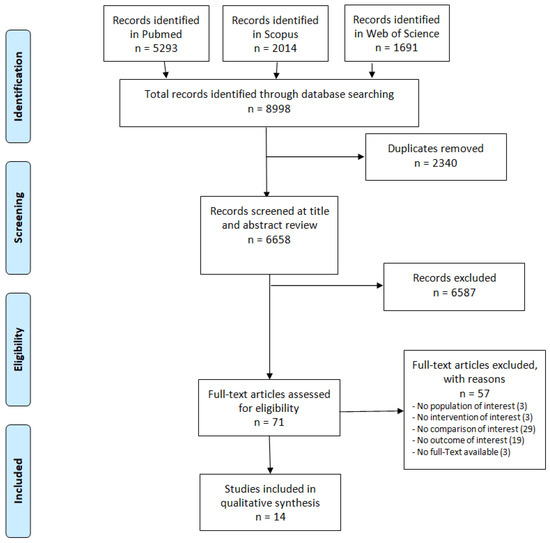
Figure 1.
PRISMA flowchart of the included studies.
The studies included were published between 2006 and 2021 [5,8,15,16,17,18,19,20,21,22,23,24,25,26].
Four out of 14 studies (29%) were from the USA [5,15,22,24] and three (21%) from the UK [16,17,21]. The remaining seven studies (50%) were from seven different countries (one from Canada [18], one from Lebanon [19], one from Australia [20], one from China [8], one from Taiwan [23], one from Romania [25], and one from Mozambique [26].
The sample size was from 20 [25] to 61039 patients [5]; in 50% of the studies the sample size was between 149 and 657 [15,17,19,20,21,24,26].
Regarding the study design: six (43%) were historical cohort studies [5,8,20,23,24,26], five (36%) were before and after studies [15,18,19,21,22], one (7%) was a cohort study [25], one (7%) was a retrospective interventional cohort study [16] and one (7%) was an observational retrospective study [17].
Within the selected articles, the following outcomes were identified: diagnosis [15,18,19,22], therapy [5,15,18,19,20,21,25], survival [8,26], mortality [16,20,23], recurrence [23], time from visit to chemotherapy/surgery [24], prophylactic mastectomy [17], physical recovery [25], therapeutic compliance [25] and MTB cost-effectiveness [26].
The most analyzed outcomes were therapy and diagnosis, evaluated in seven (50%) [5,15,18,19,20,21,25] and four (29%) [15,18,19,22] studies, respectively.
Eight out of 14 studies (57%) analyzed more than one (from two to three) outcome [15,18,19,20,23,24,25,26] and, of these, three studies [15,18,19] analyzed both diagnosis and therapy outcomes.
In line with the search string, all the studies assessed the impact of MTBs on breast cancer care. The findings are detailed below.
3.1. Diagnosis
In terms of diagnosis, four out of four studies showed that, with implementation of the MTB, there was a change in this outcome [15,18,19,22]. In particular, three studies reported a change both in imaging and anatomo-pathologic interpretation [15,18,22].
3.2. Therapy
Changes in the treatment plan after MTB implementation were reported by seven out of seven studies [5,15,18,19,20,21,25]. With respect to the surgical treatment plan, as reported by Foster et al., 2016, patients followed by the MTB received a treatment recommendation different from that previously proposed by a single professional [18]. In one study [20], patients discussed at the MTB were found to undergo surgery significantly more than patients not discussed. A statistically significant increase in reconstruction was also highlighted [21].
3.3. Mortality
In two studies, the implementation of the MTB was associated with a reduced risk of mortality [16,23], while in one study [20], no statistically significant difference in mortality was found between patients discussed and patients not discussed.
A pooled analysis of three studies [16,20,23] reporting results on the outcome “breast cancer mortality, showed, in the fixed-effects model (p = 0.43), a statistically significant 14% reduction in mortality relative risk for patients enrolled versus not enrolled in MTBs (pooled HR = 0.86; 95% CI: 0.81–0.97) (Figure 2).
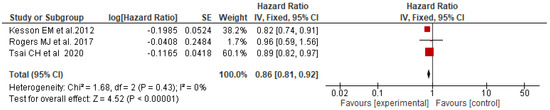
Figure 2.
Forest plot of adjusted HRs for breast cancer mortality among patients enrolled (experimental) vs. not enrolled (control) in MTBs [16,20,23].
3.4. Survival
In terms of survival, the study outcomes were not unanimous. In one study, implementation of the MTB was associated with a higher five-year overall survival rate [8]. According to Brandao et al., 2021, a higher three-year overall survival rate was shown among patients with early breast cancer discussed at MTB but, with respect to three-year disease-free survival and general survival rate in patients with metastatic breast cancer, no significant differences were found between the pre-MTB and the post-MTB sub-cohorts [26].
- Other outcomes
The recurrence rate, analyzed in one study only, was significantly lower for patients enrolled in an MTB than for those in a not-enrolled group [23].
The median time from first visit to neo-adjuvant chemotherapy for patients managed at the multidisciplinary clinic was significantly shorter than for those seen on different days by specialists from different disciplines, while the median time to definitive surgery was not significantly different between the two groups [24]. One study also reported a reduction in the use of prophylactic mastectomy following an open forum for clinical debate [17].
Furthermore, patients undergoing multidisciplinary clinical management showed faster physical recovery and better therapeutic compliance after mastectomy and breast reconstruction compared to the control group [25]. In addition, in one study, the implementation of the MTB was found to be a cost-effective measure [26].
From quality assessment of the studies, seven [15,17,18,19,21,22,25] were found to be of fair quality and seven [5,8,16,20,23,24,26] of good quality.
The main characteristics of the included studies are summarized in Table 1.

Table 1.
Characteristics of the included studies.
4. Discussion
In line with other findings in the literature [9,27], this study showed that the implementation of MTBs significantly impacted breast cancer care outcomes from diagnosis, to treatment, to survivorship. Out of 14 studies included, only three [20,24,26] did not show a significant association between the implementation of an MTB and outcome indicators. More specifically, they did not find a statistically significant difference between patient groups discussed or not at an MTB, respectively, with respect to mortality [20], median time to definitive surgery [24], general survival rate in patients with metastatic breast cancer or three-year disease-free survival [26]. All the other studies included found significant associations between MTB implementation and breast cancer care outcomes. As was also found in a study by Specchia and colleagues [9], the most analyzed outcomes were diagnosis, treatment and survival. Other variables impacted by the implementation of the MTBs were considered in single studies, with a high level of heterogeneity and a weaker evidence base.
In terms of diagnosis, four out of four studies [15,18,19,22], in line with others from the literature [28,29,30], showed that, with implementation of an MTB, there was a change in this outcome, especially with respect to imaging and anatomo-pathologic interpretation.
In terms of treatment, our results also showed that, after discussion at the MTB, patients received a different recommendation from the one previously proposed by a single professional. In particular, a reduction in the use of prophylactic mastectomy was observed [17] and an increase in immediate breast reconstruction [21]. Patient survival was the most heterogeneous variable analysed, with varying outcomes observed. In some cases [8,16,23], survival rates improved after implementation of MTBs, in line with another study in the literature [28]. However, as emerged in the studies published by Rogers et al., 2017 [20] and Brandao et al., 2021 [26], observed improvements were not significant, which was in line with other studies from the literature [29,31,32]. The lack of sufficient evidence on survival, as noted by Coory et al., 2008 [32], may be attributed to the difficulty of conducting randomized clinical trials to demonstrate the potential impact of MTB on its improvement, free from confounding factors. Moreover, it is important to highlight that, of the included studies, most were retrospective in design and limited in their ability to attribute change in outcomes to multidisciplinary processes. Therefore, prospective studies could be useful to reliably assess any benefits in clinical care and care processes.
Despite this, we were able to perform a pooled analysis of three studies [16,20,23], all of good quality, which reported results for the outcome “breast cancer mortality”. This analysis showed, in the fixed-effects model (p = 0.43), a statistically significant 14% reduction in mortality relative risk for patients enrolled compared to those not enrolled in MTBs.
With respect to its weaknesses, this review relied on a relatively limited number of eligible studies, some of which had very small sample sizes. In addition, we were not able to perform a meta-analysis that included all the studies considered in the review due to the heterogeneity observed in the evaluation of different outcomes. Heterogeneity in outcomes was observed even among studies investigating the same cancer type because they involved patients with different subtypes of the cancer or who were at different stages of the same disease. These two factors could have influenced patient assessment and management. Another weakness results from the variety of studies included in the review. Most were observational studies and therefore susceptible to bias because of the study design. This was reflected in the results of the methodological assessment, which led to labeling of half of the studies as “fair”. However, although only half of the studies were labeled as “good”, none of them were found to be of “poor” quality. Finally, our review was limited to studies written in English or Italian only, and it may be that additional evidence of MTB impacts from studies written in other languages was missed.
Despite these limitations, the present review represents one of the few studies that has attempted to synthesize the research available on the topic of MTBs and their impact on healthcare outcomes, focusing on patients affected by breast cancer in a comprehensive manner and using a strict methodology. Moreover, the analysis focusing on the outcome “patient mortality” provided a strong quantitative evaluation of the topic that could represent a starting point for future investigations.
5. Conclusions
There is growing emphasis on the application of multidisciplinary approaches to breast cancer care. However, because of heterogeneous characterizations and contexts, the effectiveness of multidisciplinary care and of the processes that contribute to its effectiveness cannot be firmly determined.
In terms of clinical management, this study showed that a multidisciplinary approach is a valuable way to deliver appropriate and effective care to patients affected by breast cancer, with significant gains in survival. The complex and various needs that characterize these patients’ experience require organisational improvements with enhanced coordination of care and the implementation of well-structured protocols and pathways.
In terms of future research, further studies are needed to enrich the evidence available to date, especially with respect to outcomes which have been less investigated but which are important for the improvement of clinical care and care processes. Evidence on the impact of MTBs in clinical practice is still lacking regarding many aspects of breast cancer care. Further studies should aim to evaluate the impact on survival rates, quality of life, patient satisfaction and cost-effectiveness. The importance of the latter should not be underestimated. In a world where economic factors strongly impact the healthcare sector, the implementation of multidisciplinary teams composed of different specialists could represent a new approach to the healthcare process that could lead to improvement in diagnosis and treatment and, in turn, to cost reduction.
Author Contributions
Conceptualization and methodology, M.L.S. and G.D.; software and formal analysis, F.C., F.S. and E.C.; investigation and data curation, M.R.C., R.M. and A.G.; writing—original draft preparation, M.R.C. and A.D.P.; writing—review and editing, M.L.S., G.D., A.D.P., A.M. and M.R.C.; supervision, M.L.S. and G.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- World Cancer Research Fund International. Available online: https://www.wcrf.org/dietandcancer/worldwide-cancer-data/ (accessed on 7 February 2022).
- World Health Organization Website. Available online: https://www.who.int/news-room/fact-sheets/detail/breast-cancer#:~:text=In%202020%2C%20there%20were%202.3 (accessed on 7 February 2022).
- Rosato, V.; Bosetti, C.; Negri, E.; Talamini, R.; Maso, L.D.; Malvezzi, M.; Falcini, F.; Montella, M.; La Vecchia, C. Reproductive and hormonal factors, family history, and breast cancer according to the hormonal receptor status. Eur. J. Cancer Prev. 2014, 23, 412–417. [Google Scholar] [CrossRef]
- Wöckel, A.; Festl, J.; Stüber, T.; Brust, K.; Stangl, S.; Heuschmann, P.U.; Albert, U.-S.; Budach, W.; Follmann, M.; Janni, W.; et al. Interdisciplinary Screening, Diagnosis, Therapy and Follow-up of Breast Cancer. Guideline of the DGGG and the DKG Part 1 with Recommendations for the Screening, Diagnosis and Therapy of Breast Cancer. Geburtshilfe Frauenheilkd. 2018, 78, 927–948. [Google Scholar] [PubMed]
- Quyyumi, F.F.; Wright, J.D.; Accordino, M.K.; Buono, D.; Law, C.W.; Hillyer, G.C.; Neugut, A.I.; Hershman, D.L. Factors Associated with Multidisciplinary Consultations in Patients with Early Stage Breast Cancer. Cancer Investig. 2019, 37, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Levin, M. Multidisciplinary approach to breast cancer management. Drug News Perspect. 2004, 17, 206–212. [Google Scholar] [PubMed]
- Berardi, R.; Morgese, F.; Rinaldi, S.; Torniai, M.; Mentrasti, G.; Scortichini, L.; Giampieri, R. Benefits and Limitations of a Multidisciplinary Approach in Cancer Patient Management. Cancer Manag. Res. 2020, 12, 9363–9374. [Google Scholar] [CrossRef]
- Lu, J.; Jiang, Y.; Qian, M.; Lv, L.; Ying, X. The Improved Effects of a Multidisciplinary Team on the Survival of Breast Cancer Patients: Experiences from China. Int. J. Environ. Res. Public Health 2019, 17, 277. [Google Scholar] [CrossRef]
- Specchia, M.L.; Frisicale, E.M.; Carini, E.; Di Pilla, A.; Cappa, D.; Barbara, A.; Ricciardi, W.; Damiani, G. The impact of tumor board on cancer care: Evidence from an umbrella review. BMC Heal. Serv. Res. 2020, 20, 73. [Google Scholar] [CrossRef]
- El Saghir, N.S.; Keating, N.L.; Carlson, R.W.; Khoury, K.E.; Fallowfield, L. Tumor Boards: Optimizing the Structure and Improving Efficiency of Multidisciplinary Management of Patients with Cancer Worldwide. Am. Soc. Clin. Oncol. Educ. Book 2014, 34, e461–e466. [Google Scholar] [CrossRef]
- Janusch-Roi, A.; Neamţiu, L.; Dimitrova, N.; Ulutürk Tekin, A.; Parmelli, E.; Garcia Escribano, M.; Mansel, R. Manual for Breast Cancer Services—European Quality Assurance Scheme for Breast Cancer Services, Version 1; Publications Office of the European Union: Luxembourg, 2021. [Google Scholar]
- Shao, J.; Rodrigues, M.; Corter, A.L.; Baxter, N.N. Multidisciplinary Care of Breast Cancer Patients: A Scoping Review of Multidisciplinary Styles, Processes, and Outcomes. Curr. Oncol. 2019, 26, 385–397. [Google Scholar] [CrossRef]
- NIH. Study Quality Assessment Tools. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 7 February 2022).
- Greenland, S. Quantitative methods in the review of epidemiologic literature. Epidemiol. Rev. 1987, 9, 1–30. [Google Scholar] [CrossRef]
- Newman, E.A.; Guest, A.B.; Helvie, M.A.; Roubidoux, M.A.; Chang, A.E.; Kleer, C.G.; Diehl, K.M.; Cimmino, V.M.; Pierce, L.J.; Hayes, D.; et al. Changes in surgical management resulting from case review at a breast cancer multidisciplinary tumor board. Cancer 2006, 107, 2346–2351. [Google Scholar] [CrossRef]
- Kesson, E.M.; Allardice, G.M.; George, W.D.; Burns, H.J.G.; Morrison, D. Effects of multidisciplinary team working on breast cancer survival: Retrospective, comparative, interventional cohort study of 13,722 women. BMJ 2012, 344, e2718. [Google Scholar] [CrossRef]
- Leff, D.; Ho, C.; Thomas, H.; Daniels, R.; Side, L.; Lambert, F.; Knight, J.; Griffiths, M.; Banwell, M.; Aitken, J.; et al. A multidisciplinary team approach minimises prophylactic mastectomy rates. Eur. J. Surg. Oncol. (EJSO) 2015, 41, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Foster, T.J.; Bouchard-Fortier, A.; Olivotto, I.A.; Quan, M.L. Effect of Multidisciplinary Case Conferences on Physician Decision Making: Breast Diagnostic Rounds. Cureus 2016, 8, e895. [Google Scholar] [CrossRef] [PubMed]
- Charara, R.N.; Kreidieh, F.Y.; Farhat, R.A.; Al-Feghali, K.A.; Khoury, K.E.; Haydar, A.; Nassar, L.; Berjawi, G.; Shamseddine, A.; El Saghir, N.S. Practice and Impact of Multidisciplinary Tumor Boards on Patient Management: A Prospective Study. J. Glob. Oncol. 2017, 3, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.; Matheson, L.; Garrard, B.; Maher, B.; Cowdery, S.; Luo, W.; Reed, M.; Riches, S.; Pitson, G.; Ashley, D. Comparison of outcomes for cancer patients discussed and not discussed at a multidisciplinary meeting. Public Health 2017, 149, 74–80. [Google Scholar] [CrossRef]
- El Gammal, M.M.; Lim, M.; Uppal, R.; Sainsbury, R. Improved immediate breast reconstruction as a result of oncoplastic multidisciplinary meeting. Breast Cancer 2017, 9, 293–296. [Google Scholar] [CrossRef] [PubMed]
- Garcia, D.; Spruill, L.S.; Irshad, A.; Wood, J.; Kepecs, D.; Klauber-Demore, N. The Value of a Second Opinion for Breast Cancer Patients Referred to a National Cancer Institute (NCI)-Designated Cancer Center with a Multidisciplinary Breast Tumor Board. Ann. Surg. Oncol. 2018, 25, 2953–2957. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-H.; Hsieh, H.-F.; Lai, T.-W.; Kung, P.-T.; Kuo, W.-Y.; Tsai, W.-C. Effect of multidisciplinary team care on the risk of recurrence in breast cancer patients: A national matched cohort study. Breast 2020, 53, 68–76. [Google Scholar] [CrossRef]
- Akhtar, Z.; Stearns, V.; Cartwright, P.; Blackford, A.L.; Prasath, V.; Klein, C.; Jelovac, D.; Asrari, F.; Habibi, M. The effect of 1-day multidisciplinary clinic on breast cancer treatment. Breast Cancer Res. Treat. 2020, 182, 623–629. [Google Scholar] [CrossRef]
- Baciu, A.C.; Clinic, C.-N.B.C.; Marcu, O.A.; Pop, M.S.; Debucean, D.; Clinic, O.L.M. A multidisciplinary approach to breast cancer – introducing a management file for breast cancer patients. Arch. Balk. Med. Union 2020, 55, 80–86. [Google Scholar] [CrossRef]
- Brandão, M.; Guisseve, A.; Bata, G.; Firmino-Machado, J.; Alberto, M.; Ferro, J.; Garcia, C.; Zaque, C.; Jamisse, A.; Lorenzoni, C.; et al. Survival Impact and Cost-Effectiveness of a Multidisciplinary Tumor Board for Breast Cancer in Mozambique, Sub-Saharan Africa. Oncologist 2021, 26, e996–e1008. [Google Scholar] [CrossRef] [PubMed]
- Biganzoli, L.; Cardoso, F.; Beishon, M.; Cameron, D.; Cataliotti, L.; Coles, C.E.; Bolton, R.C.D.; Trill, M.D.; Erdem, S.; Fjell, M.; et al. The requirements of a specialist breast centre. Breast 2020, 51, 65–84. [Google Scholar] [CrossRef]
- Prades, J.; Remue, E.; Van Hoof, E.; Borras, J.M. Is it worth reorganising cancer services on the basis of multidisciplinary teams (MDTs)? A systematic review of the objectives and organisation of MDTs and their impact on patient outcomes. Health Policy 2015, 119, 464–474. [Google Scholar] [CrossRef]
- Lamb, B.W.; Brown, K.F.; Nagpal, K.; Vincent, C.; Green, J.S.A.; Sevdalis, N. Quality of Care Management Decisions by Multidisciplinary Cancer Teams: A Systematic Review. Ann. Surg. Oncol. 2011, 18, 2116–2125. [Google Scholar] [CrossRef]
- Basta, Y.L.; Bolle, S.; Fockens, P.; Tytgat, K.M.A.J. The Value of Multidisciplinary Team Meetings for Patients with Gastrointestinal Malignancies: A Systematic Review. Ann. Surg. Oncol. 2017, 24, 2669–2678. [Google Scholar] [CrossRef] [PubMed]
- Pillay, B.; Wootten, A.C.; Crowe, H.; Corcoran, N.; Tran, B.; Bowden, P.; Crowe, J.; Costello, A.J. The impact of multidisciplinary team meetings on patient assessment, management and outcomes in oncology settings: A systematic review of the literature. Cancer Treat. Rev. 2016, 42, 56–72. [Google Scholar] [CrossRef]
- Coory, M.; Gkolia, P.; Yang, I.; Bowman, R.; Fong, K. Systematic review of multidisciplinary teams in the management of lung cancer. Lung Cancer 2008, 60, 14–21. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).