Abstract
In people with Alzheimer’s disease (PwAD), there is a need for specific tools for the timely diagnosis, management, and treatment of symptoms. New technological solutions, including digital devices, application programs (apps), sensors and virtual reality, represent promising possibilities for objective and reliable assessment, monitoring and intervention strategies in this field. Our structured review presents an up-to-date summary of the technological solutions for the (i) diagnosis, (ii) management and (iii) treatment of AD-related symptoms. To this end, we searched electronic databases (i.e., PubMed, Web of Science, and Cochrane Library) for studies published over the last 10 years. Two authors of the review extracted data of interest. A total of eight manuscripts were included. In the last decade, a series of technological solutions across AD stages have been proposed. These include: (i) innovative strategies for the early detection of deficits in finger dexterity, visuo-spatial abilities (including spatial navigation), divided attention and instrumental autonomy; (ii) tools to activate the patient’s responsiveness in terms of alertness and mood improvement; and (iii) useful interventions for retrieving memories, increasing body movements and improving spatial cognition. Methodological limitations, mainly pertaining to the paucity of randomized controlled trials and comprehensive assessments, were observed. Advances in technology currently provide the potential for designing innovative methods for evaluating, controlling and handling AD-related symptoms. The co-creation of technological solutions with all stakeholders represents the best way to design effective strategies for PwAD.
1. Introduction
Dementia is a neurological condition affecting approximately 50 million people worldwide [1]. Because of the aging population, it currently represents a social and health emergency. Dementia is usually characterized by loss of cognitive function and decreased behavioral abilities to such an extent that it interferes with a person’s activities in daily living. Moreover, behavioral and psychological symptoms of dementia (BPSD) afflict the vast majority of patients, especially those with Alzheimer’s disease (AD) [2]. AD represents 60–80% of all dementia types [3] and is the sixth leading cause of death in the United States [4]. The presence of extracellular senile plaques of insoluble β-amyloid peptide (Aβ) and neurofibrillary tangles composed of phosphorylated tau protein (P-tau) in neuronal cytoplasm constitute the hallmarks of this neurological condition [5]. Pathogenesis is characterized by an insidious onset; degeneration tends to develop slowly and gradually and worsens over many years. Over 90% of people with AD (PwAD) manifest BPSD in mild stages of the disease, including depression, anxiety, apathy, agitation and aggression; disinhibition, delusion, hallucinations, irritability and emotional lability; euphoria; and aberrant motor, sleep and eating behaviors [2,6].
Symptoms depend on the progression of the disease, which is divided into five stages: preclinical; mild cognitive impairment (MCI) due to AD; and mild, moderate and severe dementia due to AD. Preclinical AD is usually identified only in clinical settings thanks to the use of neuroimaging techniques able to detect amyloid-beta deposits without overt cognitive deficits. Conversely, individuals with MCI usually show low performance in objective neuropsychological testing of episodic memory, with difficulties that may also encompass other cognitive domains (e.g., executive functioning, visuo-constructive abilities, gnosis, praxis and language), without the impairment of personal and instrumental autonomy. In the mild stage, memory deterioration and difficulties in problem solving, complex tasks and judgment (e.g., making financial decisions) become more pronounced, and changes in personality (uncharacteristic irritability or anger, reduced motivation, etc.), difficulty in organizing and in expressing thoughts and getting lost or misplacing belongings may occur. During the moderate stage, PwAD display confusion and need more assistance in their daily activities. They especially show substantial difficulties in temporal orientation and a reduction in cognitive efficiency, require assistance for self-care and undergo significant changes in personality and behavior (e.g., wandering). Finally, at the severe stage, PwAD lose the ability to communicate coherently and require total assistance for eating, dressing, using the bathroom and performing self-care tasks. They may experience a deficit in motor abilities pertaining to walking and lose the ability to swallow and control bladder and bowel functions [7,8].
Physicians of PwAD thus have to deal with a complex spectrum of patient symptoms during the course of the disease, and taking care of demented patients is difficult and often results in depression, burden and compromised health for caregivers because of continuous and stressful daily care and support [6].
AD can be especially challenging to detect at an early stage. In addition to physical and neurological examination and a review of medical history, assessment includes neuropsychological tests, laboratory exams and neuroimaging investigations, resulting in a multidimensional and time-consuming practice including different clinical competences. To this end, researchers are working on new diagnostic tools that may enable clinicians to accurately diagnose AD earlier in the course of the disease when symptoms are very mild or even before symptoms appear [9], with positive implications for therapeutic purposes.
Caregiver burden is associated with the higher dependence level of the patient and loss of autonomy, as well as BPDS severity [10,11]. Due to cognitive and progressive behavioral deterioration, a number of safety issues have been reported in the literature regarding PwAD, including injuries (e.g., falls, ingestion of dangerous substances, accidents with sharp objects, fires and burns) requiring hospital admission to the emergency department, abnormal behavior and misuse of medication [12]. Moreover, language skills as well as interactive communication decrease during AD progression, making patients unable to relate their needs to caregivers effectively [7,8]. To this end, beyond diagnosis, increasing attention has also been given to developing technologies for management (e.g., safety and security, mobility aids, support in everyday living tasks and engagement in familial and social relationships) and treatment (e.g., medication reminders and improvement in cognitive function and physical mobility) [13] of PwAD.
Technology can be used to empower PwAD, allowing them to live a more satisfying and meaningful life. People should be empowered to promote their own health, interact effectively with health services and be active partners in making decisions. Empowerment is a multidimensional community process through which individuals and groups are able to better understand, control and manage symptoms throughout their lives. It represents a key concept in the World Health Organization’s vision of health promotion. WHO consensus documents [14,15] on mental health identify the empowerment of people with mental difficulties (including cognitive ones) and those who care for them as key priorities in the health agenda for the next decades. Research should provide a deep understanding for mental health practitioners, decision-makers and urban service organizations about how PwAD and their caregivers can benefit from new technology products in order to improve cognitive health.
Our structured review aims to present an up-to-date overview of technological solutions in terms of digital devices, applications (apps), sensors and virtual reality (VR) options by highlighting their applicability for the diagnosis, management and treatment of AD-related symptoms. Implications for the empowerment of patients and their caregivers by designing effective strategies for AD care are discussed.
2. Materials and Methods
The literature reviewed in this research was obtained by searching PubMed, Cochrane Library and Web of Science. A search strategy limited to the last 10 years (from 1 January 2012 to 19 January 2022) was performed using the following combination of terms: “Alzheimer’s Disease” AND “mental health technology” AND “digital devices” OR “app” OR “sensors” OR “virtual reality” AND “empowerment”. The search was restricted to a limited time period (i.e., 10 years) because technological solutions—including those designed for PwAD—have quickly improved over time. A total of 574 manuscripts were selected. Studies were eligible for inclusion if they reported effective technological solutions in terms of digital devices, apps, sensors or virtual reality (VR) for the diagnosis, management and treatment of AD-related symptoms. Exclusionary criteria encompassed the following: (i) non-primary research (i.e., scoping reviews, systematic reviews, meta-analyses, perspectives or case studies and research protocols); (ii) studies that analyzed results by qualitative methodologies; (iii) studies that included combined interventions (e.g., technological solutions plus pharmacological/non-pharmacological interventions); (iv) studies that included patients other than AD (i.e., other dementia types) or generally referred to subjects as ‘‘demented patients’’; and (v) studies written in languages other than English. This screening finally yielded 8 articles for evaluation (Figure 1). Two independent reviewers (D.M.C. and G.C.) extracted data of interest, and disagreements were discussed until a consensus was reached.
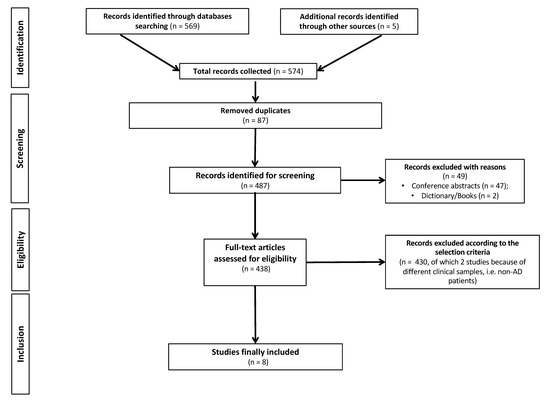
Figure 1.
PRISMA (Preferred Reporting of Systematic Reviews and Meta-analysis) flowchart of search strategy and results.
3. Results
Over the last 10 years, advances in technology have led to innovative solutions for the diagnosis [16,17,18,19,20], management [21] and treatment [22,23] of PwAD. A summary of the main findings is shown in Table 1.

Table 1.
Summary of the main findings of selected studies.
The results generally provide support for the applicability of technological solutions for the early detection of deficits in finger dexterity, visuo-spatial abilities (including spatial navigation), divided attention and instrumental autonomy for managing the patient’s behavior (i.e., alertness and mood improvement) and treatment interventions for retrieving memories and increasing body movements, as well as improving spatial cognition. The applicability of technological solutions covers all AD stages, i.e., preclinical [18,19], mild-to-moderate [16,17,20,22] and moderate-to-severe [21,22], with different implications for clinicians and caregivers.
The analysis of the selected studies involved some methodological limitations. Diagnostic criteria differed among studies, and they were not reported in two cases [21,22]. Two studies [16,18] based the AD diagnosis on the National Institute on Aging/Alzheimer’s Association (NIA-AA) criteria [24], and three studies [17,19,23] based it on the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer’s Disease Related Disorders Association (NINCDS-ADRDA) criteria [25], with the supplement of the Diagnostic and Statistical Manual of Mental Disorders Fourth Edition (DSM-IV) [26] criteria in one case [20]. Heterogeneity among participants was also found. Enrolled individuals were outpatients in two cases [16,18]; in other cases, they were recruited from memory clinics [17], Alzheimer’s day centers [20,22] or social senior centers [19,23]. A convenience sample was used in another case [21]. Randomized controlled trials were not the rule, except for one investigation [23]; two studies compared experimental and control groups [16,19]. Three studies [17,18,20] aimed to reveal significant differences across the continuum of physiological/pathological aging among groups of healthy older adults, MCI and AD patients, while two studies drew conclusions from a single sample test [21,22]. In one study, the sample size was very small [22]. A comprehensive neuropsychological assessment was performed only in three studies [16,19,23], and no other neurophysiological or neuroimaging evaluation was adopted by researchers to corroborate their findings for the other selected studies. Pharmacological treatment able to influence patients’ performance was described only in one study [16], and recruited participants were generally poorly defined with regard to possible comorbidities they may have. Finally, compliance with technology was generally analyzed by using qualitative data (e.g., participants’ perception towards technology).
4. Discussion
Today, technological solutions represent useful tools to improve the diagnostic process, management and treatment of AD-related symptoms. Constant advances in technology provide the potential for designing innovative strategies for the diagnosis of AD-related symptoms. Intelligent and smart approaches for the diagnosis of AD are welcome, and technological innovations can be fast, low-cost and easy-to-administer solutions. Despite some methodological limitations of the selected studies, our review revealed positive findings in terms of technology applications to manage AD-related symptoms.
Given that not only higher brain functions but also fine motor skills are affected in AD [27], a finger-to-thumb tapping task represents a useful method for evaluating finger dexterity, which is believed to influence self-care and IADLs and may decline as AD progresses. Moreover, recent studies have shown that deterioration in performing IADLs may be an early predictor of cognitive deterioration and play a critical role in the progression of MCI to AD [28]. Intelligent and smart approaches for the diagnosis of IADLs are also represented by 3D cameras for video data analysis that are fast, low-cost and easy to install in different settings, including homes, with positive implications for the ecological validity of behavioral assessments. Our review also revealed that robotic interfaces and wearable sensors are valid tools for evaluating visuo-motor abilities and divided attention, respectively, offering more support for the neuropsychological assessment of PwAD.
Moreover, VR is becoming an established tool in AD clinical research, too [29]. Thanks to the ability of computer simulations to replace the external sensory world with an artificial environment or replicate the body movements of the user in an immersive way, the intuitive nature of this technology makes it appealing even to those with a significant burden of disease, such as PwAD. It is also useful both for the diagnosis and treatment of spatial navigation deficits that may occur in early stages [30]. Technical equipment integrating a computer system with a screen and sound amplifier, a pressure microswitch, a voice-detecting sensor with a throat microphone and basic software has also been shown to be consistent with patients’ behavior in the absence of engagement, producing encouraging results in fostering verbal abilities and arm raising in PwAD and offering new perspectives on the treatment of advanced stages of the disease.
AD is a chronic condition that causes the affected person to enter a state of progressive deterioration and become more dependent on others in order to continue performing personal and instrumental activities of daily living at a certain level. The usage of technology, such as tablet applications for the management of AD-related symptoms involving music therapy, may facilitate care planning and increase the empowerment of patients and their caregivers. It has been suggested that such an application allows caregivers to have a deep understanding of patients’ life experiences and may help them in maintaining their own health [31]. Empowerment in AD care can be defined as a confidence-building process whereby PwAD are respected, have a voice and are heard, are involved in making decisions about their lives and have the opportunity to create change through access to appropriate resources [32]. The process of designing technological solutions should be initiated as early as possible in collaboration with specialists as a comprehensive medical examination, including aspects that are important to PwAD, in order to clarify individual values, wishes and special needs for care.
5. Conclusions
According to the best practice guidelines for the involvement of people with dementia in developing technology-based solutions [33], a rigorous field-testing phase followed by observations by an expert team, exploration of the main methods for collecting feedback from PwAD and offering participants the opportunity to learn a new skill through their commitment may enhance patients’ wellbeing and improve empowerment. The co-creation of technological solutions in research by using brainstorming, focus groups, questionnaires, detailed interviews and surveys with all relevant stakeholders, including physicians, informal caregivers, staff members and technology developers, make the evaluation of the impact of the technology more effective and ensure that the system meets a high standard of sustainability and reliability in the diagnosis, management and treatment of PwAD. Having access to planning, information and resources and ensuring high-quality care represent key features of user empowerment in cognitive health, including in the case of PwAD.
Author Contributions
Conceptualization, D.M.C.; methodology, D.M.C. and G.C. (Gabriele Cipriani); validation, G.C. (Gianluca Castelnuovo); formal analysis, D.M.C. and G.C. (Gabriele Cipriani); investigation, D.M.C.; data curation, D.M.C.; writing—original draft preparation, D.M.C.; writing—review and editing G.C. (Gabriele Cipriani); visualization, G.C. (Gianluca Castelnuovo); supervision, G.C. (Gianluca Castelnuovo). All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Prince, M.J.; Wimo, A.; Guerchet, M.M.; Ali, G.C.; Wu, Y.T.; Prina, M. World Alzheimer Report 2015—The Global Impact of Dementia: An Analysis of Prevalence, Incidence, Cost and Trends; Alzheimer’s Disease International: London, UK, 2015. [Google Scholar]
- James, I.A.; Gray, K.; Moniz-Cook, E.; Lee, K.; Reichelt, K.; Richardson, J. Behavioural and psychological symptoms of dementia: A new framework for holistic understanding and non-pharmacological management. BJPsych Adv. 2021, 28, 11–20. [Google Scholar] [CrossRef]
- Sabbagh, M.; Decourt, B. Current and Emerging Therapeutics in AD. Curr. Alzheimer Res. 2017, 14, 354–355. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.H. Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2020, 16, 391–460. [Google Scholar]
- Kang, S.; Lee, Y.H.; Lee, J.E. Metabolism-centric overview of the pathogenesis of Alzheimer’s disease. Yonsei Med. J. 2017, 58, 479–488. [Google Scholar] [CrossRef]
- Kales, H.C.; Lyketsos, C.G.; Miller, E.M.; Ballard, C. Management of behavioral and psychological symptoms in people with Alzheimer’s disease: An international Delphi consensus. Int. Psychogeriatr. 2019, 31, 83–90. [Google Scholar] [CrossRef]
- Jack, C.R.; Knopman, D.S.; Jagust, W.J.; Shaw, L.M.; Aisen, P.S.; Weiner, M.W.; Petersen, R.C.; Trojanowski, J.Q. Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol. 2010, 9, 119–128. [Google Scholar] [CrossRef] [Green Version]
- Sperling, R.A.; Aisen, P.S.; Beckett, L.A.; Bennett, D.A.; Craft, S.; Fagan, A.M.; Iwatsubo, T.; Clifford, J.; Kaye, J.; Montine, T.J.; et al. Toward defining the preclinical stages of Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 280–292. [Google Scholar] [CrossRef] [Green Version]
- Laske, C.; Sohrabi, H.R.; Frost, S.M.; López-de-Ipiña, K.; Garrard, P.; Buscema, M.; Dauwels, J.; Soekard, S.R.; Mueller, S.; Linnemann, C.; et al. Innovative diagnostic tools for early detection of Alzheimer’s disease. Alzheimer’s Dement. 2015, 11, 561–578. [Google Scholar] [CrossRef]
- Sink, K.M.; Covinsky, K.E.; Barnes, D.E.; Newcomer, R.J.; Yaffe, K. Caregiver characteristics are associated with neuropsychiatric symptoms of dementia. J. Am. Geriatr. Soc. 2006, 54, 796–803. [Google Scholar] [CrossRef]
- Kim, B.; Noh, G.O.; Kim, K. Behavioural and psychological symptoms of dementia in patients with Alzheimer’s disease and family caregiver burden: A path analysis. BMC Geriatr. 2021, 21, 160. [Google Scholar] [CrossRef]
- Davies, K.; Cheraghi-Sohi, S.; Ong, B.N.; Perryman, K.; Sanders, C. Co-designing an Adaption of a Mobile App to Enhance Communication, Safety, and Well-being Among People Living at Home with Early-Stage Dementia: Protocol for an Exploratory Multiple Case Study. JMIR Res. Protoc. 2021, 10, e19543. [Google Scholar] [CrossRef] [PubMed]
- Fang, M.L.; Canham, S.L.; Battersby, L.; Sixsmith, J.; Wada, M.; Sixsmith, A. Exploring privilege in the digital divide: Implications for theory, policy, and practice. Gerontologist 2019, 59, e1–e15. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Mental Health Declaration for Europe: Facing the Challenges, Buildings Solutions. In Proceedings of the WHO European Ministerial Conference on Mental Health, Helsinki, Finland, 12–15 January 2005; Available online: https://www.euro.who.int/__data/assets/pdf_file/0008/88595/E85445.pdf (accessed on 20 January 2022).
- World Health Organization. The European Mental Health Action Plan; WHO Regional Office for Europe: Copenhagen, Denmark, 2015; Available online: https://www.euro.who.int/__data/assets/pdf_file/0020/280604/WHO-Europe-Mental-Health-Acion-Plan-2013–2020.pdf (accessed on 20 January 2022).
- Bartoli, E.; Caso, F.; Magnani, G.; Baud-Bovy, G. Low-cost robotic assessment of visuo-motor deficits in Alzheimer’s disease. IEEE Trans. Neural Syst. Rehabil. Eng. 2017, 25, 852–860. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Sabbagh, M.; Wyman, R.; Liebsack, C.; Kunik, M.E.; Najafi, B. Instrumented trail-making task to differentiate persons with no cognitive impairment, amnestic mild cognitive impairment, and Alzheimer disease: A proof of concept study. Gerontology 2017, 63, 189–200. [Google Scholar] [CrossRef] [Green Version]
- Suzumura, S.; Osawa, A.; Maeda, N.; Sano, Y.; Kandori, A.; Mizuguchi, T.; Yin, Y.; Kondo, I. Differences among patients with Alzheimer’s disease, older adults with mild cognitive impairment and healthy older adults in finger dexterity. Geriatr. Gerontol. Int. 2018, 18, 907–914. [Google Scholar] [CrossRef]
- Serino, S.; Morganti, F.; Colombo, D.; Pedroli, E.; Cipresso, P.; Riva, G. Disentangling the contribution of spatial reference frames to executive functioning in healthy and pathological aging: An experimental study with virtual reality. Sensors 2018, 18, 1783. [Google Scholar] [CrossRef] [Green Version]
- Karakostas, A.; König, A.; Crispim-Junior, C.F.; Bremond, F.; Derreumaux, A.; Lazarou, I.; Kompatsiaris, I.; Tsolaki, M.; Robert, P. A French-Greek Cross-Site Comparison Study of the Use of Automatic Video Analyses for the Assessment of Autonomy in Dementia Patients. Biosensors 2020, 10, 103. [Google Scholar] [CrossRef]
- Lancioni, G.; Singh, N.; O’Reilly, M.; Sigafoos, J.; D’Amico, F.; Laporta, D.; Scordamaglia, A.; Pinto, K. Tablet-based intervention to foster music-related hand responses and positive engagement in people with advanced Alzheimer’s disease. J. Enabling Technol. 2019, 13, 17–28. [Google Scholar] [CrossRef]
- Lancioni, G.E.; Singh, N.N.; O’Reilly, M.F.; Sigafoos, J.; D’Amico, F.; Renna, C.; Pinto, K. Technology-aided programs to support positive verbal and physical engagement in persons with moderate or severe Alzheimer’s disease. Front. Aging Neurosci. 2016, 8, 87. [Google Scholar] [CrossRef] [Green Version]
- Serino, S.; Pedroli, E.; Tuena, C.; De Leo, G.; Stramba-Badiale, M.; Goulene, K.; Mariotti, M.G.; Riva, G. A novel virtual reality-based training protocol for the enhancement of the “mental frame syncing” in individuals with Alzheimer’s disease: A development-of-concept trial. Front. Aging Neurosci. 2017, 9, 240. [Google Scholar] [CrossRef] [Green Version]
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Jack, C.R., Jr.; Kawas, C.H.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, E.; et al. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 263–269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKhann, G.; Drachman, D.; Folstein, M.; Katzman, R.; Price, D.; Stadlan, E.M. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group* under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984, 34, 939–944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington, DC, USA, 1994. [Google Scholar]
- Kluger, A.; Gianutsos, J.G.; Golomb, J.; Ferris, S.H.; George, A.E.; Franssen, E.; Reisberg, B. Patterns of motor impairment in normal aging, mild cognitive decline, and early alzheimer’s disease. J. Gerontol. Ser. B Psychol. Sci. Soc. Sci. 1997, 52, 28–39. [Google Scholar]
- Clare, L.; Woods, B. Cognitive rehabilitation and cognitive training for early-stage Alzheimer’s disease and vascular dementia. In Cochrane Database of Systematic Reviews; Clare, L., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2003. [Google Scholar]
- Clay, F.; Howett, D.; FitzGerald, J.; Fletcher, P.; Chan, D.; Price, A. Use of immersive virtual reality in the assessment and treatment of Alzheimer’s disease: A systematic review. J. Alzheimer’s Dis. 2020, 75, 23–43. [Google Scholar] [CrossRef] [Green Version]
- Allison, S.L.; Fagan, A.M.; Morris, J.C.; Head, D. Spatial navigation in preclinical Alzheimer’s disease. J. Alzheimer’s Dis. 2016, 52, 77–90. [Google Scholar] [CrossRef] [Green Version]
- Maiden, N.; D’Souza, S.; Jones, S.; Müller, L.; Pannese, L.; Pitts, K.; Prilla, M.; Pudney, K.; Rose, M.; Turner, I.; et al. Computing technologies for reflective, creative care of people with dementia. Commun. ACM 2013, 56, 60–67. [Google Scholar] [CrossRef]
- McConnell, T.; Sturm, T.; Stevenson, M.; McCorry, N.; Donnelly, M.; Taylor, B.J.; Best, P. Co-producing a shared understanding and definition of empowerment with people with dementia. Res. Involv. Engagem. 2019, 5, 19. [Google Scholar] [CrossRef]
- Rai, H.K.; Barroso, A.C.; Yates, L.; Schneider, J.; Orrell, M. Involvement of people with dementia in the development of technology-based interventions: Narrative synthesis review and best practice guidelines. J. Med. Internet Res. 2020, 22, e17531. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).