Malaria and Helminthic Co-Infection during Pregnancy in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
1.1. Clinical Implications of Concurrent Malaria and Helminthic Infection in Pregnancy
1.2. Operational Definitions
2. Materials and Methods
2.1. Reporting
2.2. Search Strategy and Information Sources
2.3. Study Selection
2.4. Eligibility Criteria
2.5. Quality Assessment
2.6. Data Extraction
2.7. Outcome Measurement
2.8. Statistical Analysis
3. Results
3.1. Literature Search
3.2. Characteristics of Included Studies
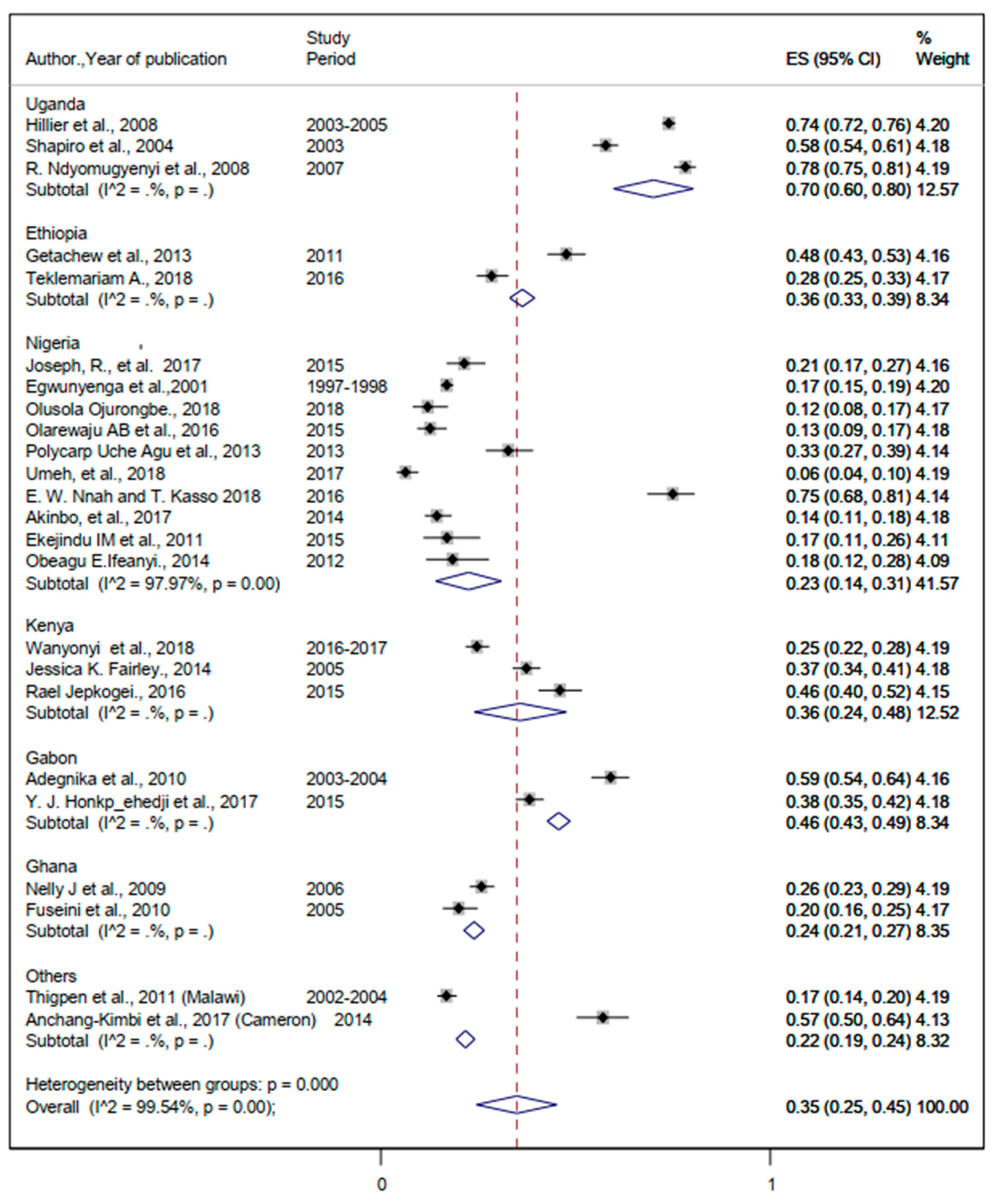
3.3. Meta-Analysis
3.3.1. The Burden of Malaria Infection
3.3.2. The Burden of Helminthic Infection
3.3.3. The Burden of Malaria and Helminthic Co-Infection
4. Discussion
4.1. Optimisation of Anti-Malarial and Anti-Helminthic Infections in Endemic Areas
4.2. Ending Preventable Maternal Mortality due to Malarial and Helminthic Co-Infection
4.3. Implications for Practice, Policy, and Future Research and Innovation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. World Malaria Report 2020—20 Years of Global Progress & Challenges; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- World Malaria Report. 2020. Available online: https://www.wipo.int/amc/en/ (accessed on 11 March 2022).
- Lubinda, J.; Bi, Y.; Hamainza, B.; Haque, U.; Moore, A.J. Modelling of malaria risk, rates, and trends: A spatiotemporal approach for identifying and targeting sub-national areas of high and low burden. PLoS Comput. Biol. 2021, 17, e1008669. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Report of the WHO Strategic Advisory Group on Malaria Eradication i Malaria Eradication: Benefits, Future Scenarios & Feasibility A Report of the Strategic Advisory Group on Malaria Eradication; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- World Health Organization. Guideline WHO Guidelines for Malaria—16 February 2021. Available online: http://apps.who.int/bookorders (accessed on 11 March 2022).
- Kumari, R.; Jayswar, H.; Dhingra, N. High Burden to High Impact (HBHI) approaches—Country perspective for adoption and adaptation in India. J. Commun. Dis. 2020, 52, 5–16. [Google Scholar] [CrossRef]
- World Health Organization. A Strategic Framework for Malaria Prevention and Control During Pregnancy in the African Region; WHO Regional Office for Africa: Brazzaville, Congo, 2004. [Google Scholar]
- World Health Organization. UHC in Africa: A Framework for Action. 2016. Available online: https://apps.who.int/iris/handle/10665/341157 (accessed on 11 March 2022).
- Rogerson, S.J.; Beeson, J.G.; Laman, M.; Poespoprodjo, J.R.; William, T.; Simpson, J.A.; Price, R.N.; Anstey, N.; Fowkes, F.; McCarthy, J.; et al. Identifying and combating the impacts of COVID-19 on malaria. BMC Med. 2020, 18, 239. [Google Scholar] [CrossRef] [PubMed]
- Feleke, S.M.; Reichert, E.N.; Mohammed, H.; Brhane, B.G.; Mekete, K.; Mamo, H.; Petros, B.; Solomon, H.; Abate, E.; Hennelly, C.; et al. Plasmodium falciparum is evolving to escape malaria rapid diagnostic tests in Ethiopia. Nat. Microbiol. 2021, 6, 1289–1299. [Google Scholar] [CrossRef]
- World Malaria Report 2021; World Health Organization: Geneva, Switzerland, 2021.
- Montresor, A.; Mupfasoni, D.; Mikhailov, A.; Mwinzi, P.; Lucianez, A.; Mekasha, S.; Woyessa, A.; Shafi, O.; Vercruysse, J.; Grimes, J.E.T.; et al. The global progress of soil-transmitted helminthiases control in 2020 and world health organization targets for 2030. PLoS Negl. Trop. Dis. 2020, 14, e0008505. [Google Scholar] [CrossRef]
- Leta, G.T.; Mekete, K.; Wuletaw, Y.; Gebretsadik, A.; Sime, H.; Mekasha, S.; Woyessa, A.; Shafi, O.; Vercruysse, J.; Grimes, J.E.T.; et al. National mapping of soil-transmitted helminth and schistosome infections in Ethiopia. Parasites Vectors 2020, 13, 437. [Google Scholar] [CrossRef]
- van Eijk, A.M.; Lindblade, K.A.; Odhiambo, F.; Peterson, E.; Rosen, D.H.; Karanja, D.; Ayisi, J.G.; Shi, Y.P.; Adazu, K.; Slutsker, L. Geohelminth infections among pregnant women in rural western Kenya: A cross-sectional study. PLoS Negl. Trop. Dis. 2009, 3, e370. [Google Scholar] [CrossRef]
- Bolka, A.; Gebremedhin, S. Prevalence of intestinal parasitic infection and its association with anemia among pregnant women in Wondo Genet district, Southern Ethiopia: A cross-sectional study. BMC Infect. Dis. 2019, 19, 483. [Google Scholar] [CrossRef] [Green Version]
- Adam, I.; ALhabardi, N.A.; Al-Wutayd, O.; Khamis, A.H. Prevalence of schistosomiasis and its association with anemia among pregnant women: A systematic review and meta-analysis. Parasites Vectors 2021, 14, 133. [Google Scholar] [CrossRef]
- Kamau, A.; Mogeni, P.; Okiro, E.A.; Snow, R.W.; Bejon, P. A systematic review of changing malaria disease burden in sub-Saharan Africa since 2000: Comparing model predictions and empirical observations. BMC Med. 2020, 18, 94. [Google Scholar] [CrossRef]
- Moore, K.A.; Fowkes, F.J.I.; Wiladphaingern, J.; Wai, N.S.; Paw, M.K.; Pimanpanarak, M.; Carrara, V.I.; Raksuansak, J.; Simpson, J.A.; White, N.J.; et al. Mediation of the effect of malaria in pregnancy on stillbirth and neonatal death in an area of low transmission: Observational data analysis. BMC Med. 2017, 15, 98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khurana, S.; Singh, S.; Mewara, A. Diagnostic Techniques for Soil-Transmitted Helminths—Recent Advances. Res. Rep. Trop. Med. 2021, 12, 181–196. [Google Scholar] [CrossRef] [PubMed]
- Bangert, M.; Bancalari, P.; Mupfasoni, D.; Mikhailov, A.; Gabrielli, A.F.; Montresor, A. Provision of deworming intervention to pregnant women by antenatal services in countries endemic for soil-transmitted helminthiasis. PLoS Negl. Trop. Dis. 2019, 13, e0007406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makaula, P.; Sadalaki, J.R.; Muula, A.S.; Kayuni, S.; Jemu, S.; Bloch, P. Schistosomiasis in Malawi: A systematic review. Parasites Vectors 2014, 7, 570. [Google Scholar] [CrossRef]
- Mwinnyaa, G.; Hazel, E.; Maïga, A.; Amouzou, A. Estimating population-based coverage of reproductive, maternal, newborn, and child health (RMNCH) interventions from health management information systems: A comprehensive review. BMC Health Serv. Res. 2021, 21, 1083. [Google Scholar] [CrossRef]
- Ferreira, L.Z.; Blumenberg, C.; Utazi, C.E.; Nilsen, K.; Hartwig, F.P.; Tatem, A.J.; Barros, A.J.D. Geospatial estimation of reproductive, maternal, newborn and child health indicators: A systematic review of methodological aspects of studies based on household surveys. Int. J. Health Geogr. 2020, 19, 41. [Google Scholar] [CrossRef]
- Rios Quituizaca, P.; Gatica-Domínguez, G.; Nambiar, D.; Ferreira Santos, J.L.; Brück, S.; Ruas, L.V.; Barros, A.J.D. National and subnational coverage and inequalities in reproductive, maternal, newborn, child, and sanitary health interventions in Ecuador: A comparative study between 1994 and 2012. Int. J. Equity Health 2021, 20, 48. [Google Scholar] [CrossRef]
- Boerma, T.; Requejo, J.; Victora, C.G.; Amouzou, A.; George, A.; Taylor, C.M.; Amouzou, A.; Jiwani, S.S.; da Silva, I.C.M.; Sidze, E.M.; et al. Countdown to 2030: Tracking progress towards universal coverage for reproductive, maternal, newborn, and child health. Lancet 2018, 391, 1538–1548. [Google Scholar] [CrossRef]
- Faye, C.M.; Wehrmeister, F.C.; Melesse, D.Y.; Mutua, M.K.K.; Maïga, A.; Taylor, C.M.; Amouzou, A.; Jiwani, S.S.; da Silva, I.C.M.; Sidze, E.M.; et al. Large and persistent subnational inequalities in reproductive, maternal, newborn and child health intervention coverage in sub-Saharan Africa. BMJ Glob. Health 2020, 5, e002232. [Google Scholar] [CrossRef] [Green Version]
- Barros, A.J.D.; Wehrmeister, F.C.; Ferreira, L.Z.; Vidaletti, L.P.; Hosseinpoor, A.R.; Victora, C.G. Are the poorest poor being left behind? Estimating global inequalities in reproductive, maternal, newborn and child health. BMJ Glob. Health 2020, 5, e002229. [Google Scholar] [CrossRef] [Green Version]
- Molyneux, D.H.; Lindsay, S.W.; Fitzpatrick, C.; Engels, D. The cross-cutting contribution of the end of neglected tropical diseases to the sustainable development goals. Infect. Dis. Poverty 2017, 6, 73. [Google Scholar]
- WHO. Monitoring the Health-Related Sustainable Development Goals (SDGs); WHO: Geneva, Switzerland, 2017; pp. 9–10. [Google Scholar]
- Bakken, L.; Iversen, P.O. The impact of malaria during pregnancy on low birth weight in East-Africa: A topical review. Malar. J. 2021, 20, 348. [Google Scholar] [CrossRef] [PubMed]
- Chetty, A.; Omondi, M.A.; Butters, C.; Smith, K.A.; Katawa, G.; Ritter, M.; Layland, L.; Horsnell, W. Impact of Helminth Infections on Female Reproductive Health and Associated Diseases. Front. Immunol. 2020, 11, 577516. [Google Scholar] [CrossRef] [PubMed]
- Animaw, Z.; Melese, A.; Demelash, H.; Seyoum, G.; Abebe, A. Intestinal parasitic infections and associated factors among pregnant women in Ethiopia: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2021, 21, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Demeke, G.; Mengistu, G.; Abebaw, A.; Toru, M.; Yigzaw, M.; Shiferaw, A.; Mengist, H.M.; Dilnessa, T. Effects of intestinal parasite infection on hematological profiles of pregnant women attending antenatal care at Debre Markos Referral Hospital, Northwest Ethiopia: Institution based prospective cohort study. PLoS ONE 2021, 16, e0250990. [Google Scholar] [CrossRef]
- Brooker, S.; Hotez, P.J.; Bundy, D.A.P. Hookworm-related anaemia among pregnant women: A systematic review. PLoS Negl. Trop. Dis. 2008, 2, e291. [Google Scholar] [CrossRef] [Green Version]
- Mosawi, S.H.; Dalimi, A.; Charkhi, M.A.; Baarae, O.; Darman, A.; Mosavi, M.; Baryal, M.W.; Stanikzai, H. Gallbladder perforation due to Ascaris lumbricoides in a pregnant woman and 6 year old girl from afghanistan: Case report. Iran. J. Parasitol. 2019, 14, 477–481. [Google Scholar]
- Righetti, A.A.; Glinz, D.; Adiossan, L.G.; Koua, A.Y.G.; Niamké, S.; Hurrell, R.F.; Wegmuller, R.; N’Goran, E.K.; Utzinger, J. Interactions and Potential Implications of Plasmodium falciparum-Hookworm Coinfection in Different Age Groups in South-Central Côte d’Ivoire. PLoS Negl. Trop. Dis. 2012, 6, e1889. [Google Scholar] [CrossRef] [Green Version]
- Tuasha, N.; Hailemeskel, E.; Erko, B.; Petros, B. Comorbidity of intestinal helminthiases among malaria outpatients of Wondo Genet health centers, southern Ethiopia: Implications for integrated control. BMC Infect. Dis. 2019, 19, 659. [Google Scholar] [CrossRef] [Green Version]
- Hirst, J.; Villar, J.; Victora, C.; Papageorghiou, A.; Finkton, D.; Barros, F.; Gravett, M.; Giuliani, F.; Purwar, M.; Frederick, I.; et al. The antepartum stillbirth syndrome: Risk factors and pregnancy conditions identified from the INTERGROWTH-21 st Project. BJOG Int. J. Obstet. Gynaecol. 2018, 125, 1145–1153. [Google Scholar] [CrossRef]
- Boel, M.; Carrara, V.I.; Rijken, M.; Proux, S.; Nacher, M.; Pimanpanarak, M.; Paw, M.K.; Moo, O.; Gay, H.; Bailey, W.; et al. Complex interactions between soil-transmitted helminths and malaria in pregnant women on the thai-burmese border. PLoS Negl. Trop. Dis. 2010, 4, e887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adam, I.; Salih, M.M.; Mohmmed, A.A.; Rayis, D.A.; Elbashir, M.I. Pregnant women carrying female fetuses are at higher risk of placental malaria infection. PLoS ONE 2017, 12, e0182394. [Google Scholar] [CrossRef] [PubMed]
- Mpairwe, H.; Tweyongyere, R.; Elliott, A. Pregnancy and helminth infections. Parasite Immunol. 2014, 36, 328–337. [Google Scholar] [CrossRef] [Green Version]
- Ndibazza, J.; Webb, E.L.; Lule, S.; Mpairwe, H.; Akello, M.; Oduru, G.; Kizza, M.; Akurut, H.; Muhangi, L.; Magnussen, P.; et al. Associations between maternal helminth and malaria infections in pregnancy and clinical malaria in the offspring: A birth cohort in Entebbe, Uganda. J. Infect. Dis. 2013, 208, 2007–2016. [Google Scholar] [CrossRef] [PubMed]
- Alarcón de Noya, B.; Ruiz Guevara, R. Pregnancy as a risk factor to disease and the vertical transmission to the fetus, of a host of parasitic ailments. CientMed 2020, 1, 1–16. [Google Scholar] [CrossRef]
- Maestre, A.; Carmona-Fonseca, J. Immune responses during gestational malaria: A review of the current knowledge and future trend of research. J. Infect. Dev. Ctries. 2014, 8, 391–402. [Google Scholar] [CrossRef] [Green Version]
- Wilairatana, P.; Mahannop, P.; Tussato, T.; Hayeedoloh, I.M.; Boonhok, R.; Klangbud, W.K.; Mala, W.; Kotepui, K.U.; Kotepui, M. C-reactive protein as an early biomarker for malaria infection and monitoring of malaria severity: A meta-analysis. Sci. Rep. 2021, 11, 22033. [Google Scholar] [CrossRef]
- Sarfo, B.O.; Hahn, A.; Schwarz, N.G.; Jaeger, A.; Sarpong, N.; Marks, F.; Adu-Sarkodie, Y.; Tamminga, T.; May, J. The usefulness of c-reactive protein in predicting malaria parasitemia in a sub-saharan african region. PLoS ONE 2018, 13, e0201693. [Google Scholar] [CrossRef] [Green Version]
- Abrams, E.T.; Kwiek, J.J.; Mwapasa, V.; Kamwendo, D.D.; Tadesse, E.; Lema, V.M.; Molyneux, M.E.; Rogerson, S.J.; Meshnick, S.R. Malaria during pregnancy and foetal haematological status in Blantyre, Malawi. Malar. J. 2005, 4, 39. [Google Scholar] [CrossRef] [Green Version]
- Sharma, L.; Shukla, G. Placental Malaria: A new insight into the pathophysiology. Front. Med. 2017, 4, 117. [Google Scholar] [CrossRef] [Green Version]
- Brabin, B.; Tinto, H.; Roberts, S.A. Testing an infection model to explain excess risk of preterm birth with long-term iron supplementation in a malaria endemic area. Malar. J. 2019, 18, 374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haque, M.; Koski, K.G.; Scott, M.E. Maternal Gastrointestinal Nematode Infection Up-regulates Expression of Genes Associated with Long-Term Potentiation in Perinatal Brains of Uninfected Developing Pups. Sci. Rep. 2019, 9, 4165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, J.; Baisley, K.; Kavishe, B.; Changalucha, J.; Andreasen, A.; Mayaud, P.; Gumodoka, B.; Kapiga, S.; Hayes, R.; Watson-Jones, D. Impact of malaria and helminth infections on immunogenicity of the human papillomavirus-16/18 AS04-adjuvanted vaccine in Tanzania. Vaccine 2014, 32, 611–617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mabbott, N.A. The Influence of Parasite Infections on Host Immunity to Co-infection with Other Pathogens. Front. Immunol. 2018, 9, 2579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menon, S.; Rodolfo, R.; Akudibillah, G.; Dusabimana, A.; Harmon, S.; Mabeya, H. Effects of malaria/helminthic coinfections on cervical cancer progression among sub Saharan African women on highly active antiretroviral therapy: A scoping review. Gynecol. Oncol. Rep. 2019, 29, 64–69. [Google Scholar] [CrossRef]
- Mwangi, T.W.; Bethony, J.M.; Brooker, S. Malaria and helminth interactions in humans: An epidemiological viewpoint. Ann. Trop. Med. Parasitol. 2006, 100, 551–570. [Google Scholar] [CrossRef]
- Boltena, M.T.; El-Khatib, Z.; Kebede, A.S.; Asamoah, B.O.; Boltena, A.T.; Yeshambaw, M.; Biru, M. Comorbidity of geo-helminthes among malaria outpatients of the health facilities in Ethiopia: Systematic review and meta-analysis. Int. J. Environ. Res. Public Health 2021, 18, 862. [Google Scholar] [CrossRef]
- Degarege, A.; Erko, B. Epidemiology of Plasmodium and helminth coinfection and possible reasons for heterogeneity. BioMed Res. Int. 2016, 2016, 3083568. [Google Scholar] [CrossRef] [Green Version]
- Makenga, G.; Menon, S.; Baraka, V.; Minja, D.T.R.; Nakato, S.; Delgado-Ratto, C.; Francis, F.; Lusingu, J.P.; Van Geertruyden, J.-P. Prevalence of malaria parasitaemia in school-aged children and pregnant women in endemic settings of sub-Saharan Africa: A systematic review and meta-analysis. Parasite Epidemiol. Control 2020, 11, e00188. [Google Scholar] [CrossRef]
- Karshima, S.N. Prevalence and distribution of soil-transmitted helminth infections in Nigerian children: A systematic review and meta-analysis. Infect. Dis. Poverty 2018, 7, 1118–1132. [Google Scholar] [CrossRef] [Green Version]
- Bahati, Y.L.; Delanghe, J.; Balaluka, G.B.; Kishabongo, A.S.; Philippé, J. Asymptomatic submicroscopic Plasmodium infection is highly prevalent and is associated with anemia in children younger than 5 years in South Kivu/Democratic Republic of Congo. Am. J. Trop. Med. Hyg. 2020, 102, 1048–1055. [Google Scholar] [CrossRef] [PubMed]
- Degarege, A.; Veledar, E.; Degarege, D.; Erko, B.; Nacher, M.; Madhivanan, P. Plasmodium falciparum and soil-transmitted helminth co-infections among children in sub-Saharan Africa: A systematic review and meta-analysis. Parasites Vectors 2016, 9, 344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osakunor, D.N.M.; Sengeh, D.M.; Mutapi, F. Coinfections and comorbidities in African health systems: At the interface of infectious and noninfectious diseases. PLoS Negl. Trop. Dis. 2018, 12, e0006711. [Google Scholar] [CrossRef] [PubMed]
- Snow, R.W. Global malaria eradication and the importance of Plasmodium falciparum epidemiology in Africa. BMC Med. 2015, 13, 23. [Google Scholar] [CrossRef] [Green Version]
- Eisele, T.P.; Larsen, D.A.; Anglewicz, P.A.; Keating, J.; Yukich, J.; Bennett, A.; Hutchinson, P.; Steketee, R.W. Malaria prevention in pregnancy, birthweight, and neonatal mortality: A meta-analysis of 32 national cross-sectional datasets in Africa. Lancet Infect. Dis. 2012, 12, 942–949. [Google Scholar] [CrossRef]
- Carrasco-Escobar, G.; Fornace, K.; Benmarhnia, T. Mapping socioeconomic inequalities in malaria in Sub-Sahara African countries. Sci. Rep. 2021, 11, 15121. [Google Scholar] [CrossRef]
- Osungbade, K.O.; Oladunjoye, O.O. Prevention of congenital transmission of malaria in sub-Saharan African Countries: Challenges and implications for health system strengthening. J. Trop. Med. 2012, 2012, 648456. [Google Scholar] [CrossRef] [Green Version]
- Zeukeng, F.; Tchinda, V.H.M.; Bigoga, J.D.; Seumen, C.H.T.; Ndzi, E.S.; Abonweh, G.; Makoge, V.; Motsebo, A.; Moyou, R.S. Co-infections of Malaria and Geohelminthiasis in Two Rural Communities of Nkassomo and Vian in the Mfou Health District, Cameroon. PLoS Negl. Trop. Dis. 2014, 8, e3236. [Google Scholar] [CrossRef]
- Mahande, A.M.; Mahande, M.J. Prevalence of parasitic infections and associations with pregnancy complications and outcomes in northern Tanzania: A registry-based cross-sectional study. BMC Infect. Dis. 2016, 16, 78. [Google Scholar] [CrossRef] [Green Version]
- Fourie, C. The trouble with inequalities in global health partnerships. Med. Anthr. Theory 2018, 5, 142–155. [Google Scholar] [CrossRef] [Green Version]
- Accrombessi, M.; Issifou, S. Malaria control and elimination in sub-Saharan Africa: Data from antenatal care centres. Lancet Glob. Health 2019, 7, e1595–e1596. [Google Scholar] [CrossRef] [Green Version]
- Zerbo, A.; Castro Delgado, R.; Arcos González, P. Water sanitation and hygiene in Sub-Saharan Africa: Coverage, risks of diarrheal diseases, and urbanization. J. Biosaf. Biosecur. 2021, 3, 41–45. [Google Scholar] [CrossRef]
- European Union. Tackling Infectious Disease in Sub-Saharan Africa; European Union: Maastricht, The Netherlands, 2018. [Google Scholar]
- Gosling, R.; Chimumbwa, J.; Uusiku, P.; Rossi, S.; Ntuku, H.; Harvard, K.; White, C.; Tatarsky, A.; Chandramohan, D.; Chen, I. District-level approach for tailoring and targeting interventions: A new path for malaria control and elimination. Malar. J. 2020, 19, 125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kayentao, K.; Garner, P.; van Eijk, A.M.; Naidoo, I.; Roper, C.; Mulokozi, A.; MacArthur, J.R.; Luntamo, M.; Ashorn, P.; Doumbo, O.K.; et al. Intermittent preventive therapy for malaria during pregnancy using 2 vs. 3 or more doses of sulfadoxine-pyrimethamine and risk of low birth weight in Africa: Systematic review and meta-analysis. JAMA J. Am. Med. Assoc. 2013, 309, 594–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosenthal, J.; Arku, R.E.; Baumgartner, J.; Brown, J.; Clasen, T.; Eisenberg, J.N.; Hovmand, P.; Jagger, P.; Luke, D.A.; Quinn, A.; et al. Systems science approaches for global environmental health research: Enhancing intervention design and implementation for household air pollution (hap) and water, sanitation, and hygiene (wash) programs. Environ. Health Perspect. 2020, 128, 105001. [Google Scholar] [CrossRef]
- Haque, S.S.; Freeman, M.C. Erratum: ‘The Applications of Implementation Science in Water, Sanitation, and Hygiene (WASH) Research and Practice’. Environ. Health Perspect. 2021, 129, 89001. [Google Scholar] [CrossRef]
- Walker, M.; Freitas, L.T.; Halder, J.B.; Brack, M.; Keiser, J.; King, C.H.; Levecke, B.; Lim, Y.A.-L.; Pieri, O.; Sow, D.; et al. Improving anthelmintic treatment for schistosomiasis and soil-transmitted helminthiases through sharing and reuse of individual participant.-Lata [version 1; peer review: Awaiting peer review]. Wellcome Open Res. 2022, 7, 5. [Google Scholar] [CrossRef]
- Sanders, D.M.; Todd, C.; Chopra, M. Confronting Africa’s health crisis: More of the same will not be enough. Br. Med. J. 2005, 331, 755–758. [Google Scholar] [CrossRef] [Green Version]
- Juma, P.A.; Jones, C.M.; Mijumbi-Deve, R.; Wenham, C.; Masupe, T.; Sobngwi-Tambekou, J.; Biemba, G.; Mtombo, N.; Parkhurst, J. Governance of health research in four eastern and southern African countries. Health Res. Policy Syst. 2021, 19, 132. [Google Scholar] [CrossRef]
- Azevedo, M.J. Historical Perspectives on the State of Health and Health Systems in Africa; Palgrave Macmillan: London, UK, 2017; Volume II. [Google Scholar] [CrossRef]
- Oleribe, O.O.; Momoh, J.; Uzochukwu, B.S.C.; Mbofana, F.; Adebiyi, A.; Barbera, T.; Williams, R.; Robinson, S.D.T. Identifying key challenges facing healthcare systems in Africa and potential solutions. Int. J. Gen. Med. 2019, 12, 395–403. [Google Scholar] [CrossRef] [Green Version]
- Nabyonga-Orem, J.; Asamani, J.A.; Makanga, M. The state of health research governance in Africa: What do we know and how can we improve? Health Res. Policy Syst. 2021, 19, 11. [Google Scholar] [CrossRef] [PubMed]
- Kasprowicz, V.O.; Chopera, D.; Waddilove, K.D.; Brockman, M.A.; Gilmour, J.; Hunter, E.; Kilembe, W.; Karita, E.; Gaseitsiwe, S.; Sanders, E.J.; et al. African-led health research and capacity building—Is it working? BMC Public Health 2020, 20, 1104. [Google Scholar] [CrossRef] [PubMed]
- Bakibinga, P.; Kamande, E.; Kisia, L.; Omuya, M.; Matanda, D.J.; Kyobutungi, C. Challenges and prospects for implementation of community health volunteers’ digital health solutions in Kenya: A qualitative study. BMC Health Serv. Res. 2020, 20, 888. [Google Scholar] [CrossRef] [PubMed]
- Manyazewal, T.; Woldeamanuel, Y.; Blumberg, H.M.; Fekadu, A.; Marconi, V.C. The potential use of digital health technologies in the African context: A systematic review of evidence from Ethiopia. NPJ Digit. Med. 2021, 4, 125. [Google Scholar] [CrossRef]
- Beyene, J.; Harrar, S.W.; Altaye, M.; Astatkie, T.; Awoke, T.; Shkedy, Z.; Mersha, T.B. A Roadmap for Building Data Science Capacity for Health Discovery and Innovation in Africa. Front. Public Health 2021, 9, 1435. [Google Scholar] [CrossRef]
- Bader, E.; Alhaj, A.M.; Hussan, A.A.; Adam, I. Malaria and stillbirth in Omdurman Maternity Hospital, Sudan. Int. J. Gynecol. Obstet. 2010, 109, 144–146. [Google Scholar] [CrossRef]
- Moore, K.A.; Simpson, J.A.; Scoullar, M.J.L.; McGready, R.; Fowkes, F.J.I. Quantification of the association between malaria in pregnancy and stillbirth: A systematic review and meta-analysis. Lancet Glob. Health 2017, 5, e1101–e1112. [Google Scholar] [CrossRef] [Green Version]
- Say, L.; Donner, A.; Gülmezoglu, A.M.; Taljaard, M.; Piaggio, G. The prevalence of stillbirths: A systematic review. Reprod. Health 2006, 3, 1. [Google Scholar] [CrossRef] [Green Version]
- Global Technical Strategy for Malaria 2016–2030; 2021 update; World Health Organization: Geneva, Switzerland, 2021.
- Buxton, M.; Machekano, H.; Gotcha, N.; Nyamukondiwa, C.; Wasserman, R.J. Are vulnerable communities thoroughly informed on mosquito bio-ecology and burden? Int. J. Environ. Res. Public Health 2020, 17, 8196. [Google Scholar] [CrossRef]
- Tegegne, Y.; Worede, A.; Derso, A.; Ambachew, S. The Prevalence of Malaria among Children in Ethiopia: A Systematic Review and Meta-Analysis. J. Parasitol. Res. 2021, 2021, 6697294. [Google Scholar] [CrossRef]
- Imhoff-Kunsch, B.; Briggs, V. Antihelminthics in pregnancy and maternal, newborn and child health. Paediatr. Perinat. Epidemiol. 2012, 26, 223–238. [Google Scholar] [CrossRef] [PubMed]
- Barua, P.; Beeson, J.G.; Maleta, K.; Ashorn, P.; Rogerson, S.J. The impact of early life exposure to Plasmodium falciparum on the development of naturally acquired immunity to malaria in young Malawian children. Malar. J. 2019, 18, 11. [Google Scholar] [CrossRef] [PubMed]
- Hartgers, F.C.; Yazdanbakhsh, M. Co-infection of helminths and malaria: Modulation of the immune responses to malaria. Parasite Immunol. 2006, 28, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Fairley, J.K.; Bisanzio, N.; King, C.H.; Kitron, U.; Mungai, P.; Muchiri, E.; King, C.L.; Malhotra, I. Birthweight in offspring of mothers with high prevalence of helminth and malaria infection in coastal Kenya. Am. J. Trop. Med. Hyg. 2013, 88, 48–53. [Google Scholar] [CrossRef] [Green Version]
- Woolhouse, M.E.J.; Thumbi, S.M.; Jennings, A.; Chase-Topping, M.; Callaby, R.; Kiara, H.; Oosthuizen, M.C.; Mbole-Kariuki, M.N.; Conradie, I.; Handel, I.G.; et al. Co-infections determine patterns of mortality in a population exposed to parasite infection. Sci. Adv. 2015, 1, e1400026. [Google Scholar] [CrossRef] [Green Version]
- Singer, M. Development, coinfection, and the syndemics of pregnancy in sub-Saharan Africa. Infect. Dis. Poverty 2013, 2, 26. [Google Scholar] [CrossRef] [Green Version]
- Wolday, D.; Gebrecherkos, T.; Arefaine, Z.G.; Kiros, Y.K.; Gebreegzabher, A.; Tasew, G.; Abdulkader, M.; Abraha, H.E.; Desta, A.A.; Hailu, A.; et al. Effect of co-infection with intestinal parasites on COVID-19 severity: A prospective observational cohort study. EClinicalMedicine 2021, 39, 101054. [Google Scholar] [CrossRef]
- Candela, E.; Goizueta, C.; Periago, M.V.; Muñoz-Antoli, C. Prevalence of intestinal parasites and molecular characterization of Giardia intestinalis, Blastocystis spp. and Entamoeba histolytica in the village of Fortín Mbororé (Puerto Iguazú, Misiones, Argentina). Parasites Vectors 2021, 14, 510. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 51, 264–269. [Google Scholar] [CrossRef] [Green Version]
- NIHR. International Prospective Register of Systematic Reviews Registering a Systematic Review on PROSPERO What Does Registration on PROSPERO Involve? Inclusion Criteria When to Register Your Review PROSPERO International Prospective Register of Systematic Rev; NIHR: Southampton, UK, 2019; pp. 1–12. [Google Scholar]
- Tawfik, G.M.; Dila, K.A.S.; Mohamed, M.Y.F.; Tam, D.N.H.; Kien, N.D.; Ahmed, A.M.; Huy, N.T. A step by step guide for conducting a systematic review and meta-analysis with simulation data. Trop. Med. Health 2019, 47, 46. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 2020–2021. [Google Scholar]
- Joanna Briggs Institute. Checklist for Prevalence Studies; Joanna Briggs Institute: Adelaide, Australia, 2016; p. 7. [Google Scholar]
- Naing, C.; Whittaker, M.; Nyunt-Wai, V.; Reid, S.; Wong, S.F.; Mak, J.W.; Tanner, M. Malaria and soil-transmitted intestinal helminth co-infection and its effect on anemia: A meta-analysis. Trans. R. Soc. Trop. Med. Hyg. 2013, 107, 672–683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pei, Q.; Qiao, H.; Zhang, M.; Wang, G.; Feng, H.; Pan, J.; Shi, Y. Pocket-creation method versus conventional method of endoscopic submucosal dissection for superficial colorectal neoplasms: A meta-analysis. Gastrointest. Endosc. 2021, 93, 1038–1046. [Google Scholar] [CrossRef] [PubMed]
- Tseng, T.Y.; Dahm, P.; Poolman, R.W.; Preminger, G.M.; Canales, B.J.; Montori, V.M. How to Use a Systematic Literature Review and Meta-Analysis. J. Urol. 2008, 180, 1249–1256. [Google Scholar] [CrossRef]
- Ahn, E.; Kang, H. Introduction to systematic review and meta-analysis. Korean J. Anesthesiol. 2018, 71, 103–112. [Google Scholar] [CrossRef] [Green Version]
- Phillips, C.V. Publication bias in situ. BMC Med. Res. Methodol. 2004, 4, 20. [Google Scholar] [CrossRef] [Green Version]
- Ioannidis, J.P.A. Interpretation of tests of heterogeneity and bias in meta-analysis. J. Eval. Clin. Pract. 2008, 14, 951–957. [Google Scholar] [CrossRef]
- Lin, L.; Xu, C. Arcsine-based transformations for meta-analysis of proportions: Pros, cons, and alternatives. Health Sci. Rep. 2020, 3, e178. [Google Scholar] [CrossRef]
- Copas, J.B.; Shi, J.Q. A sensitivity analysis for publication bias in systematic reviews. Stat. Methods Med. Res. 2001, 10, 251–265. [Google Scholar] [CrossRef]
- Joseph, R.; Chessed, G.; Daniel, L.; Haruna, Y.; Demshemino, M.; Bagula, R. Prevalence of malaria and geohelminth co-infection among antenatal women at the Federal Medical Centre and Specialist Hospital, Yola, Adamawa Sate, Nigeria. J. Appl. Sci. Environ. Manag. 2017, 21, 469. [Google Scholar] [CrossRef] [Green Version]
- Egwunyenga, A.; Ajayi, J.; Nmorsi, O.; Duhlinska-Popova, D. Plasmodium/intestinal Helminth Co-infections among Pregnant Nigerian Women. Memórias Do Inst. Oswaldo Cruz. 2001, 96, 1055–1059. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ojurongbe, O.; Okorie, P.N.; Opatokun, R.L.; Ojurongbe, T.A.; Mabayoje, V.O.; Olowe, O.A.; Adeyeba, O.A. Prevalence and associated factors of Plasmodium falciparum and soil transmitted helminth infections among pregnant women in Osun state, Nigeria. Afr. Health Sci. 2018, 18, 542–551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Babamale, O.A.; Shittu, O.; Danladi, Y.K.; Abdulraheem, J.Y.; Ugbomoiko, U.S. Pattern of Plasmodium-intestinal helminth co-infection among pregnant women in a high transmission zone of malaria in Nigeria. Asian Pac. J. Trop. Dis. 2016, 6, 424–428. [Google Scholar] [CrossRef]
- Agu, P.U.; Ogboi, J.S.; Akpoigbe, K.; Okeke, T.; Ezugwu, E. Impact of plasmodium falciparum and hookworm infections on the frequency of anaemia in pregnant women of rural communities in Enugu, South East Nigeria. Pan Afr. Med. J. 2013, 14, 27. [Google Scholar] [CrossRef] [PubMed]
- Umeh, J.C.; Inyang-Etoh, P.C.; Okpokam, D.C.; Otu-Bassey, I.B. Malaria and hookworm co-infection and their effects on anaemia of pregnant women attending ante-natal clinic in University Teaching Hospital, Calabar, Nigeria. Asian J. Med. Sci. 2018, 9, 27–35. [Google Scholar] [CrossRef] [Green Version]
- Nnah, E.; Kasso, T. The Prevalence of Malaria and Helminth Infection in Pregnancy at Booking and Their Relationship to Anaemia at the University of Port Harcourt Teaching Hospital, Southern Nigeria. Int. J. Trop. Dis. Health 2018, 28, 1–9. [Google Scholar] [CrossRef]
- Oyefabi, A.; Adetiba, E.; Leeshak, E.A.O. Origina l Artic l e Co-infection of malaria and intestinal parasites among pregnant women in Edo State, Nigeria ABSTRACT. J. Med. Trop. 2019, 19, 116–122. [Google Scholar] [CrossRef]
- Ekejindu, I.M.; Akah, B.; Okpala, E.C.; Onwurah, O. Malaria and Hookworm Co-Infection among Pregnant and Non-Pregnant Women in a Semi-Urban Area in Anambra State, Nigeria. J. Med. Sci. 2011, 6, 33–35. [Google Scholar]
- Ifeanyi, O.E.; Chibunna, O.M.; Braxton, N.A.Q.; Uche, E.C. Impact of Plasmodium falciparum malaria and hookworm infection on anaemia among pregnant women of ikwuano local government area, Abia state, Nigeria. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 104–111. [Google Scholar]
- Wanyonyi, W.A.; Mulambalah, C.S.; Mulama, D.H.; Omukunda, E.; Siteti, D.I. Malaria and Geohelminthiasis Coinfections in Expectant Women: Effect on Maternal Health and Birth Outcomes in a Malaria Endemic Region in Kenya. J. Parasitol. Res. 2018, 2018, 2613484. [Google Scholar] [CrossRef] [Green Version]
- Jepkogei, M.R.; Moses, N.; Judith, M. Socio-Demographic Factors Associated with Malaria-Geohelminth Co-Infection and Syndemics in Pregnancy: A Cross Sectional Study of Pregnant Women Attending Ante Natal Care at Nandi Hills Sub County Hospital, Kenya. Available online: www.iiste.org (accessed on 9 March 2022).
- Hillier, S.D.; Booth, M.; Muhangi, L.; Nkurunziza, P.; Khihembo, M.; Kakande, M.; Sewankambo, M.; Kizindo, R.; Kizza, M.; Muwanga, M.; et al. Plasmodium falciparum and helminth coinfection in a semiurban population of pregnant women in Uganda. J. Infect. Dis. 2008, 198, 920–927. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ndyomugyenyi, R.; Kabatereine, N.; Olsen, A.; Magnussen, P. Malaria and hookworm infections in relation to haemoglobin and serum ferritin levels in pregnancy in Masindi district, western Uganda. Trans. R Soc. Trop. Med. Hyg. 2008, 102, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, A.E.; Tukahebwa, E.M.; Kasten, J.; Clarke, S.E.; Magnussen, P.; Olsen, A.; Kabatereine, N.B.; Ndyomugyenyi, R.; Brooker, S. Epidemiology of helminth infections and their relationship to clinical malaria in southwest Uganda. Trans. R. Soc. Trop. Med. Hyg. 2005, 99, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Getachew, M.; Tafess, K.; Zeynudin, A.; Yewhalaw, D. Prevalence Soil Transmitted Helminthiasis and malaria co-infection among pregnant women and risk factors in Gilgel Gibe dam Area, Southwest Ethiopia. BMC Res. Note 2013, 6, 263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teklemariam, A.; Alemseged, M.; Adugna, S. Malaria-intestinal helminthes co-infection among patients in Wolkite Health Center and Attat Hospital, Gurage Zone, Southern Ethiopia. J. Parasitol. Vector Biol. 2018, 10, 26–32. [Google Scholar]
- Adegnika, A.A.; Ramharter, M.; Agnandji, S.T.; Ngoa, U.A.; Issifou, S.; Yazdanbahksh, M.; Kremsner, P.G. Epidemiology of parasitic co-infections during pregnancy in Lambaréné, Gabon. Trop. Med. Int. Health 2010, 15, 1204–1209. [Google Scholar] [CrossRef]
- Honkpéhèdji, Y.J.; Adegbite, B.R.; Zinsou, J.F.; Dejon-Agobé, J.C.; Edoa, J.-R.; Manego, R.Z.; McCall, M.; Ngwese, M.M.; Mougeni, F.L.; Mombo-Ngoma, G.; et al. Association of low birth weight and polyparasitic infection during pregnancy in Lambaréné, Gabon. Trop. Med. Int. Health 2021, 26, 973–981. [Google Scholar] [CrossRef]
- Yatich, N.J.; Rayner, J.C.; Turpin, A.; Jolly, P.E.; Ellis, W.O.; Stiles, J.; Agbenyega, T.; Ehiri, J.E.; Funkhouser, E.; Williams, J.H.; et al. Malaria and Intestinal Helminth Co-infection Among Pregnant Women in Ghana: Prevalence and Risk Factors. Am. J. Trop. Med. Hyg. 2009, 80, 896–901. [Google Scholar] [CrossRef] [Green Version]
- Tay, S.C.K.; Nani, E.A.; Walana, W. Parasitic infections and maternal anaemia among expectant mothers in the Dangme East District of Ghana. BMC Res. Notes 2017, 10, 3. [Google Scholar] [CrossRef] [Green Version]
- Thigpen, M.C.; Filler, S.J.; Kazembe, P.N.; Parise, M.E.; Macheso, A.; Campbell, C.H.; Newman, R.D.; Steketee, R.W.; Hamel, M. Associations between peripheral Plasmodium falciparum malaria parasitemia, human immunodeficiency virus, and concurrent helminthic infection among pregnant women in Malawi. Am. J. Trop. Med. Hyg. 2011, 84, 379–385. [Google Scholar] [CrossRef] [Green Version]
- Anchang-Kimbi, J.K.; Elad, D.M.; Sotoing, G.T.; Achidi, E.A. Coinfection with Schistosoma haematobium and Plasmodium falciparum and Anaemia Severity among Pregnant Women in Munyenge, Mount Cameroon Area: A Cross-Sectional Study. J. Parasitol. Res. 2017, 2017, 61734650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhiman, S. Are malaria elimination efforts on right track? An analysis of gains achieved and challenges ahead. Infect. Dis. Poverty 2019, 8, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alemu, F.; Kumie, A.; Medhin, G.; Gebre, T.; Godfrey, P. A socio-ecological analysis of barriers to the adoption, sustainablity and consistent use of sanitation facilities in rural Ethiopia. BMC Public Health 2017, 17, 706. [Google Scholar] [CrossRef] [PubMed]
- Mwendera, C.A.; De Jager, C.; Longwe, H.; Phiri, K.; Hongoro, C.; Mutero, C.M. Changing the policy for intermittent preventive treatment with sulfadoxine-pyrimethamine during pregnancy in Malawi. Malar. J. 2017, 16, 84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muhammad, F.M.; Nedjat, S.; Sajadi, H.S.; Parsaeian, M.; Assan, A.; Majdzadeh, R. Malaria intermittent preventive treatment in Nigeria: A qualitative study to explore barriers. BMC Infect. Dis. 2021, 21, 438. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Regional Office for Europe. Framework for Control and Prevention of Soil-Transmitted Helminthiases in the WHO European Region. Available online: http://www.euro.who.int/ (accessed on 9 March 2022).
- Maskin, E.; Monga, C.; Thuilliez, J.; Berthélemy, J.C. The economics of malaria control in an age of declining aid. Nat. Commun. 2019, 10, 2269. [Google Scholar] [CrossRef] [Green Version]
- Walker, P.G.T.; Griffin, J.T.; Ferguson, N.M.; Ghani, A.C. Estimating the most efficient allocation of interventions to achieve reductions in Plasmodium falciparum malaria burden and transmission in Africa: A modelling study. Lancet Glob. Health 2016, 4, e474–e484. [Google Scholar] [CrossRef] [Green Version]
- Head, M.G.; Goss, S.; Gelister, Y.; Alegana, V.; Brown, R.J.; Clarke, S.C.; Fitchett, J.R.A.; Atun, R.; Scott, J.A.G.; Newell, M.-L.; et al. Global funding trends for malaria research in sub-Saharan Africa: A systematic analysis. Lancet Glob. Health 2017, 5, e772–e781. [Google Scholar] [CrossRef] [Green Version]
- Cardona-Arias, J.A.; Carmona-Fonseca, J. Meta-analysis of the prevalence of malaria associated with pregnancy in Colombia 2000–2020. PLoS ONE 2021, 16, e0255028. [Google Scholar] [CrossRef]
- Tegegne, Y.; Asmelash, D.; Ambachew, S.; Eshetie, S.; Addisu, A.; Zeleke, A.J. The Prevalence of Malaria among Pregnant Women in Ethiopia: A Systematic Review and Meta-Analysis. J. Parasitol. Res. 2019, 2019, 8396091. [Google Scholar] [CrossRef]
- Bilal, J.A.; Malik, E.E.; Al-Nafeesah, A.; Adam, I. Global prevalence of congenital malaria: A systematic review and meta-analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 252, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Tediosi, F.; Penny, M. Evidence for optimal allocation of malaria interventions in Africa. Lancet Glob. Health 2016, 4, e432–e433. [Google Scholar] [CrossRef] [Green Version]
- The malERA Refresh Consultative Panel on Combination Interventions and Modelling. malERA: An updated research agenda for combination interventions and modelling in malaria elimination and eradication. PLoS Med. 2017, 14, e1002453. [Google Scholar]
- Blas, E.; Multisectoral Action Framework for Malaria. Roll Back Malar Partnership/UNDP. Available online: http://www.rollbackmalaria.org/files/files/resources/Multisectoral-Action-Framework-for-Malaria.pdf (accessed on 9 March 2022).
- WHO. The Global Global Malaria Malaria Action Plan For a malaria free world. In Guidel Malar Treat; WHO: Geneva, Switzerland, 2015; pp. 12–74. [Google Scholar]
- Danwang, C.; Bigna, J.J.; Nzalie, R.N.T.; Robert, A. Epidemiology of clinical congenital and neonatal malaria in endemic settings: A systematic review and meta-analysis. Malar. J. 2020, 19, 312. [Google Scholar] [CrossRef] [PubMed]
- Esopo, K.; Derby, L.; Haushofer, J. Interventions to improve adherence to antenatal and postnatal care regimens among pregnant women in sub-Saharan Africa: A systematic review. BMC Pregnancy Childbirth 2020, 20, 316. [Google Scholar] [CrossRef]
- Ippolito, M.M.; Moser, K.A.; Kabuya, J.-B.B.; Cunningham, C.; Juliano, J.J. Antimalarial Drug Resistance and Implications for the WHO Global Technical Strategy. Curr. Epidemiol. Rep. 2021, 8, 46–62. [Google Scholar] [CrossRef]
- Muanda, F.T.; Chaabane, S.; Boukhris, T.; Santos, F.; Sheehy, O.; Perreault, S.; Blais, L.; Bérard, A. Antimalarial drugs for preventing malaria during pregnancy and the risk of low birth weight: A systematic review and meta-analysis of randomized and quasi-randomized trials. BMC Med. 2015, 13, 193. [Google Scholar] [CrossRef] [Green Version]
- Blasco, B.; Leroy Di Fidock, D.A. Antimalarial drug resistance: Linking Plasmodium falciparum parasite biology to the clinic. Nat. Med. 2017, 23, 917–928. [Google Scholar] [CrossRef]
- Shibeshi, M.A.; Kifle, Z.D.; Atnafie, S.A. Antimalarial drug resistance and novel targets for antimalarial drug discovery. Infect. Drug Resist. 2020, 13, 4047–4060. [Google Scholar] [CrossRef]
- Ehrlich, H.Y.; Jones, J.; Parikh, S. Molecular surveillance of antimalarial partner drug resistance in sub-Saharan Africa: A spatial-temporal evidence mapping study. Lancet Microbe 2020, 1, e209–e217. [Google Scholar] [CrossRef]
- McMahon, A.; Mihretie, A.; Ahmed, A.A.; Lake, M.; Awoke, W.; Wimberly, M.C. Remote sensing of environmental risk factors for malaria in different geographic contexts. Int. J. Health Geogr. 2021, 20, 28. [Google Scholar] [CrossRef] [PubMed]
- Rogerson, S.J.; Aitken, E.H. Progress towards vaccines to protect pregnant women from malaria. EBioMedicine 2019, 42, 12–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mordmüller, B.; Sulyok, M.; Egger-Adam, D.; Resende, M.; De Jongh, W.A.; Jensen, M.H.; Smedegaard, H.H.; Ditlev, S.B.; Soegaard, M.; Poulsen, L.; et al. First-in-human, Randomized, Double-blind Clinical Trial of Differentially Adjuvanted PAMVAC, A Vaccine Candidate to Prevent Pregnancy-associated Malaria. Clin. Infect. Dis. 2019, 69, 1509–1516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chico, R.M.; Cano, J. Devising a strategy for prevention of malaria in pregnant women in the Asia Pacific. Lancet Infect. Dis. 2019, 19, 919–920. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Zeroing in on Malaria Elimination: Final Report of the E-2020 Initiative; WHO: Geneva, Switzerland, 2021; pp. 1–20. [Google Scholar]
- Russell, T.L.; Beebe, N.W.; Cooper, R.D.; Lobo, N.F.; Burkot, T.R. Successful malaria elimination strategies require interventions that target changing vector behaviours. Malar. J. 2013, 12, 56. [Google Scholar] [CrossRef] [Green Version]
- Lal, A.A.; Rajvanshi, H.; Jayswar, H.; Das, A.; Bharti, P. Malaria elimination: Using past and present experience to make malaria-free India by 2030. J. Vector Borne Dis. 2019, 56, 60–65. [Google Scholar] [CrossRef]
- World Health Organization. Eliminating Malaria: Learning from the Past, Looking Ahead; Progress and Impact Series; WHO: Geneva, Switzerland, 2011; Volume 8, pp. 1–85. [Google Scholar]
- Maat, H.; Balabanova, D.; Mokuwa, E.; Richards, P.; Mohan, V.; Ssengooba, F.; Twinomuhangi, R.; Woldie, M.; Mayhew, S. Towards sustainable community-based systems for infectious disease and disaster response; lessons from local initiatives in four African countries. Sustainability 2021, 13, 10083. [Google Scholar] [CrossRef]
- Ajayi, I.O.O.; Ajumobi, O.; Ogunwale, A.; Adewole, A.; Odeyinka, O.T.; Balogun, M.S.; Nguku, P.; Bamiselu, O.; Fellows, F.N. Is the malaria short course for program managers, a priority for malaria control effort in Nigeria? Evidence from a qualitative study. PLoS ONE 2020, 15, e0236576. [Google Scholar] [CrossRef]
- Orok, A.B.; Ajibaye, O.; Aina, O.O.; Iboma, G.; Oboshi, S.A.; Iwalokun, B. Malaria interventions and control programes in Sub-Saharan Africa: A narrative review. Cogent. Med. 2021, 8, 1940639. [Google Scholar] [CrossRef]
- Fernando, D.; Wijeyaratne, P.; Wickremasinghe, R.; Abeyasinghe, R.R.; Galappaththy, G.N.L.; Wickremasinghe, R.; Hapugoda, M.; Abeywickrema, W.A.; Rodrigo, C. Use of a public-private partnership in malaria elimination efforts in Sri Lanka; A case study. BMC Health Serv. Res. 2018, 18, 202. [Google Scholar] [CrossRef] [Green Version]
- Jones, R.T.; Tusting, L.S.; Smith, H.M.P.; Segbaya, S.; Macdonald, M.B.; Bangs, M.J.; Logan, J.G. The role of the private sector in supporting malaria control in resource development settings. J. Infect. Dis. 2020, 222, S701–S708. [Google Scholar] [CrossRef] [PubMed]
- Tizifa, T.A.; Kabaghe, A.N.; McCann, R.S.; Berg, H.V.D.; Van Vugt, M.; Phiri, K.S. Prevention Efforts for Malaria. Curr. Trop. Med. Rep. 2018, 5, 41–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Badmos, A.O.; Alaran, A.J.; Adebisi, Y.A.; Bouaddi, O.; Onibon, Z.; Dada, A.; Lin, X.; Lucero-Prisno, D.E. What sub-Saharan African countries can learn from malaria elimination in China. Trop. Med. Health 2021, 49, 86. [Google Scholar] [CrossRef] [PubMed]
- Taghipour, A.; Ghodsian, S.; Jabbari, M.; Olfatifar, M.; Abdoli, A.; Ghaffarifar, F. Global prevalence of intestinal parasitic infections and associated risk factors in pregnant women: A systematic review and meta-analysis. Trans. R. Soc. Trop. Med. Hyg. 2021, 115, 457–470. [Google Scholar] [CrossRef]
- Chami, G.F.; Kontoleon, A.A.; Bulte, E.; Fenwick, A.; Kabatereine, N.B.; Tukahebwa, E.M.; Dunne, D.W. Profiling Nonrecipients of Mass Drug Administration for Schistosomiasis and Hookworm Infections: A Comprehensive Analysis of Praziquantel and Albendazole Coverage in Community-Directed Treatment in Uganda. Clin. Infect. Dis. 2016, 62, 200–207. [Google Scholar] [CrossRef] [Green Version]
- Mutapi, F.; Maizels, R.; Fenwick, A.; Woolhouse, M. Human schistosomiasis in the post mass drug administration era. Lancet Infect. Dis. 2017, 17, e42–e48. [Google Scholar] [CrossRef] [Green Version]
- Torres-Vitolas, C.A.; Dhanani, N.; Fleming, F.M. Factors affecting the uptake of preventive chemotherapy treatment for schistosomiasis in sub-saharan africa: A systematic review. PLoS Negl. Trop. Dis. 2021, 15, e0009017. [Google Scholar] [CrossRef]
- Kanyangarara, M.; Allen, S.; Jiwani, S.S.; Fuente, D. Access to water, sanitation and hygiene services in health facilities in sub-Saharan Africa 2013-2018: Results of health facility surveys and implications for COVID-19 transmission. BMC Health Serv. Res. 2021, 21, 601. [Google Scholar] [CrossRef]
- WHO Evidence Review Group on Malaria in Pregnancy. Malaria Policy Advisory Committee Meeting Meeting Report of the WHO Evidence Review Group on Malaria in Pregnancy; WHO/HTM/GMP/MPAC/2017.18; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- de Neve, J.-W.; Andriantavison, R.L.; Croke, K.; Krisam, J.; Rajoela, V.H.; Rakotoarivony, R.A.; Rambeloson, V.; Schultz, L.; Qamruddin, J.; Verguet, S. Health, financial, and education gains of investing in preventive chemotherapy for schistosomiasis, soil-transmitted helminthiases, and lymphatic filariasis in Madagascar: A modeling study. PLoS Negl. Trop. Dis. 2018, 12, e0007002. [Google Scholar] [CrossRef] [Green Version]
- Salari, P.; Fürst, T.; Knopp, S.; Utzinger, J.; Tediosi, F. Cost of interventions to control schistosomiasis: A systematic review of the literature. PLoS Negl. Trop. Dis. 2020, 14, e0008098. [Google Scholar] [CrossRef] [Green Version]
- Price, A.; Verma, A.; Welfare, W. Are health education interventions effective for the control and prevention of urogenital schistosomiasis in sub-Saharan Africa? A systematic review. Trans. R. Soc. Trop. Med. Hyg. 2015, 109, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Zegeye, B.; Ahinkorah, B.O.; Ameyaw, E.K.; Seidu, A.A.; Yaya, S. Utilization of Deworming Drugs and Its Individual and Community Level Predictors among Pregnant Married Women in Cameroon: A Multilevel Modeling. BioMed Res. Int. 2021, 2021, 6645336. [Google Scholar] [CrossRef] [PubMed]
- Fenwick, A.; Zhang, Y.; Stoever, K. Control of the Neglected Tropical Diseases in sub-Saharan Africa: The unmet needs. Int. Health 2009, 1, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Zegeye, B.; Keetile, M.; Ahinkorah, B.O.; Ameyaw, E.K.; Seidu, A.A.; Yaya, S. Utilization of deworming medication and its associated factors among pregnant married women in 26 sub-Saharan African countries: A multi-country analysis. Trop. Med. Health 2021, 49, 53. [Google Scholar] [CrossRef]
- Sustainable Development Goal (SDG) 6. Synthesis Report 2018 on Water and Sanitation; United Nations: Geneva, Switzerland, 2018. [CrossRef]
- WHO. Global Progress Report on Wash in Health Care Facilities; WHO: Geneva, Switzerland, 2020. [Google Scholar]
- Moser, W.; Schindler, C.; Keiser, J. Efficacy of recommended drugs against soil transmitted helminths: Systematic review and network meta-analysis. BMJ 2017, 358, j4307. [Google Scholar] [CrossRef] [Green Version]
- Kabore, A.; Ibikounle, M.; Tougoue, J.J.; Mupoyi, S.; Ndombe, M.; Shannon, S.; Ottesen, E.A.; Mukunda, F.; Awaca, N. Initiating NTD programs targeting schistosomiasis and soil-transmitted helminthiasis in two provinces of the Democratic Republic of the Congo: Establishment of baseline prevalence for mass drug administration. Acta Trop. 2017, 166, 177–185. [Google Scholar] [CrossRef]
- Craig, P.; Dieppe, P.; Macintyre, S.; Mitchie, S.; Nazareth, I.; Petticrew, M. Developing and evaluating complex interventions: The new Medical Research Council guidance. BMJ 2008, 337, 979–983. [Google Scholar] [CrossRef] [Green Version]
- Hawadak, J.; Dongang Nana, R.R.; Singh, V. Global trend of Plasmodium malariae and Plasmodium ovale spp. malaria infections in the last two decades (2000–2020): A systematic review and meta-analysis. Parasites Vectors 2021, 14, 297. [Google Scholar] [CrossRef]
- Turner, H.C.; Stolk, W.A.; Solomon, A.W.; King, J.D.; Montresor, A.; Molyneux, D.H.; Toor, J. Are current preventive chemotherapy strategies for controlling and eliminating neglected tropical diseases cost-effective? BMJ Glob. Health 2021, 6, e005456. [Google Scholar] [CrossRef]
- Jacobsen, A.; Schmiegelow, C.; Sørensen, B.; Msemo, O.A.; Nielsen, K.; Nielsen, B.; Møller, S.; Lusingu, J.; Minja, D.; Hedegaard, M.; et al. Biosensor for Detecting Fetal Growth Restriction in a Low-Resource Setting. Reprod. Med. 2021, 2, 57–67. [Google Scholar] [CrossRef]
- Lau, R.; Chris, R.B.; Phuong, M.S.; Khatib, A.; Kopalakrishnan, S.; Bhasker, S.; Raheel, H.; Lecce, C.; Yegorov, S.; Mishra, S.; et al. Treatment of soil-transmitted helminth infections in pregnancy: A systematic review and meta-analysis of maternal outcomes. J. Travel Med. 2020, 27, taz079. [Google Scholar] [CrossRef] [PubMed]
- Zawawi, A.; Else, K.J. Soil-Transmitted Helminth Vaccines: Are We Getting Closer? Front. Immunol. 2020, 11, 2426. [Google Scholar] [CrossRef] [PubMed]
- Perera, D.J.; Ndao, M. Promising Technologies in the Field of Helminth Vaccines. Front. Immunol. 2021, 12, 711650. [Google Scholar] [CrossRef] [PubMed]
- Filbey, K.J.; Finney, C.A.M.; Giacomin, P.R.; Siracusa, M.C. Editorial: Recent Advances in the Immunology of Helminth Infection—Protection, Pathogenesis and Panaceas. Front. Immunol. 2021, 12, 10–12. [Google Scholar] [CrossRef]
- Tran, V.T.; Ravaud, P. Frugal innovation in medicine for low resource settings. BMC Med. 2016, 14, 102–104. [Google Scholar] [CrossRef] [Green Version]
- Pacifico Silva, H.; Lehoux, P.; Miller, F.A.; Denis, J.L. Introducing responsible innovation in health: A policy-oriented framework. Health Res. Policy Syst. 2018, 16, 90. [Google Scholar] [CrossRef]
- Mundle, D.; Davis Pluess, J. Innovative Finance to Expand access to Healthcare: Opportunities for Business; BSR: New York, NY, USA, 2017; pp. 1–54. [Google Scholar]
- Bloom, D.E.; Michael, K.; Klaus, P. Modern Infectious Diseases: Macroeconomic Impacts and Policy Responses; IZA Discussion Paper No. 13625; NBE: Cambridge, MA, USA, 2020. [Google Scholar] [CrossRef]
- Nkengasong, J.N.; Tessema, S.K. Africa Needs a New Public Health Order to Tackle Infectious Disease Threats. Cell 2020, 183, 296–300. [Google Scholar] [CrossRef]
- Quinn, S.C.; Kumar, S. Health inequalities and infectious disease epidemics: A challenge for global health security. Biosecurity Bioterrorism 2014, 12, 263–273. [Google Scholar] [CrossRef] [Green Version]
- Laxminarayan, R.; Malani, A. Economics of Infectious Diseases. In The Oxford Handbook of Health; Oxford University Press: Oxford, UK, 2012; pp. 1–20. [Google Scholar]
- Co-infections and co-morbidities. In Proceedings of the EDCTP Stakeholder Meeting, The Hague, the Netherlands, 13 September 2017.
- Lonnie, W. Sustaining Workforce Engagement: How to Ensure Your Employees Are Healthy, Happy, and Productive, 1st ed.; Productivity Press: New York, NY, USA, 2019. [Google Scholar] [CrossRef]
- Mugabe, J.O. Science, Technology and Innovation in Africa’s Regional Integration: From Rhetoric to Practice; ACODE Policy Research Series; ACODE: Kampala, Uganda, 2011. [Google Scholar]
- Agaba, B.B.; Yeka, A.; Nsobya, S.; Arinaitwe, E.; Nankabirwa, J.; Opigo, J.; Mbaka, P.; Lim, C.S.; Kalyango, J.N.; Karamagi, C. Systematic review of the status of pfhrp2 and pfhrp3 gene deletion, approaches and methods used for its estimation and reporting in Plasmodium falciparum populations in Africa: Review of published studies 2010–2019. Malar. J. 2019, 18, 355. [Google Scholar] [CrossRef]
- Slater, H.C.; Ross, A.; Felger, I.; Hofmann, N.E.; Robinson, L.; Cook, J.; Gonçalves, B.P.; Björkman, A.; Ouedraogo, A.L.; Morris, U. The temporal dynamics and infectiousness of subpatent Plasmodium falciparum infections in relation to parasite density. Nat. Commun. 2019, 10, 1433. [Google Scholar] [CrossRef] [Green Version]







| Included Studies for Meta-Analysis | Study Level Bias Score | ||
|---|---|---|---|
| S. No | Author, Publication year | Total No. Yes (Y) | Percentage of Yes (Y) |
| 1 | Hillier et al., 2008 | 9 | 100.00% |
| 2 | Getachew et al., 2013 | 8 | 89.00% |
| 3 | Joseph et al., 2017 | 9 | 100.00% |
| 4 | Wanyonyi et al., 2018 | 8 | 89.00% |
| 5 | Teklemariam A., 2018 | 8 | 89.00% |
| 6 | Egwunyenga et al., 2001 | 8 | 89.00% |
| 7 | Adegnika et al., 2010 | 9 | 100.00% |
| 8 | Nelly et al., 2009 | 9 | 100.00% |
| 9 | Shapiro et al., 2004 | 9 | 100.00% |
| 10 | Thigpen et al., 2011 | 9 | 100.00% |
| 11 | Olusola Ojurongbe | 8 | 89.00% |
| 12 | Olarewaju et al., 2016 | 9 | 100.00% |
| 13 | Polycarp Uche Agu et al., 2013 | 9 | 100.00% |
| 14 | Ndyomugyenyi et al., 2008 | 8 | 89.00% |
| 15 | Anchang-Kimbi et al., 2017 | 8 | 89.00% |
| 16 | Umeh et al., 2018 | 8 | 89.00% |
| 17 | Nnah and Kasso, 2018 | 8 | 89.00% |
| 18 | Akinbo et al., 2017 | 7 | 78.00% |
| 19 | Ekejindu et al., 2011 | 9 | 100.00% |
| 20 | Ifeanyi., 2014 | 9 | 100.00% |
| 21 | Fairley, 2014 | 9 | 100.00% |
| 22 | Fuseini et al., 2010 | 7 | 78.00% |
| 23 | Masai, Rael Jepkogei, 2016 | 8 | 89.00% |
| 24 | Honkpehedji et al., 2017 | 8 | 89.00% |
| Average bias score (%Yes) | 93.00% | ||
| S. No | Author, Year of Publication | Year Study Conducted | Country | Study Design | Sample Size | Trimester | Parity | Test Approach for Malaria Diagnosis | Test Approach for Helminthiases | Prevalence of Pf Infection | Prevalence of Pv Infection | Prevalence of Any Malaria Infection | Prevalence of Malaria Associated Anemia | Overall Prevalence of Helminthiasis | Overall Prevalence of Malaria-Helminthiases Co-infection | Hookworm | Ascaris lumbricoids | Trichuris trichuria | Shistosoma mansoni | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1st | 2nd | 3rd | Primigravida | Multigravida | ||||||||||||||||||
| 1 | Hillier et al., 2008 | 2003–2005 | Uganda | Cross-sectional | 2507 | Microscopy | Kato-Katz thick smear | 268 (11%) | 268 (11%) | 1693 (68%) | 1112 (45%) | 58 (2%) | 226 (9%) | 458 (18%) | ||||||||
| 2 | Getachew et al., 2013 | 2011 | Ethiopia | Cross-sectional | 388 | 156 | 167 | 95 | 133 | 285 | Microscopy | McMaster concentration technique | 45 (11.6%) | 159 (41%) | 30 (7.7%) | 114 (29%) | 58 (15%) | 13 (3.4%) | ||||
| 3 | Joseph, R. et al., 2017 | 2015 | Nigeria | Cross-sectional | 252 | 63 | 169 | Microscopy | Formalin-ether concentration techniques+ wet mount | 51 (20.2%) | 54 (21.4%) | 16 (6.3%) | ||||||||||
| 4 | Wanyonyi et al., 2018 | 2016–2017 | Kenya | Cross-sectional | 750 | Microscopy | Kato-Katz thick smear | 21.60% | 367 (48.9%) | 24.70% | 6.8% | |||||||||||
| 5 | Teklemariam A., 2018 | 2016 | Ethiopia | Cross-sectional | 460 | Microscopy | Formalin-ether concentration techniques | 27 (5.9%) | 55 (12%) | 84 (18.3%) | 198 (43%) | 46 (10%) | 54 (11.7%) | 77 (16.7%) | ||||||||
| 6 | Egwunyenga et al.,2001 | 1997–1998 | Nigeria | Cross-sectional | 2104 | Microscopy | Formalin-ether concentration techniques | 762 (36.2%) | 816 (38.8%) | 394 (48.3%) | 116 (5.5%) | 156 (7.4%) | 57 (2.7%) | 28 (1.3%) | ||||||||
| 7 | Adegnika et al., 2010 | 2003–2004 | Gabon | Cross-sectional | 388 | 111 | 277 | Microscopy | Kato-Katz thick smear | 98 (25%) | 216 (64%) | 15% | 34 (8.8%) | 112 (28.9%) | 83 (21.4%) | |||||||
| 8 | Nelly J et al., 2009 | 2006 | Ghana | Cross-sectional | 746 | 390 | 324 | 26 | 255 | 521 | Malaria Antigen CELISA assay | Kato-Katz thick smear | 271 (36.3%) | 36.30% | 192 (25.7%) | 124 (16.6%) | 59 (7.5%) | 92 (12.3%) | 42 (5.6%) | |||
| 9 | Shapiro et al., 2004 | 2003 | Uganda | Cross-sectional | 856 | Microscopy | Kato-Katz thick smear | 217 (49.9% | 217 (49.9%) | 405 (47.3%) | 118 (54.8%) | 275 (32.1%) | 149 (17.4%) | 70 (8.1%) | ||||||||
| 10 | Thigpen et al., 2011 | 2002–2004 | Malawi | Cross-sectional | 848 | 412 | 436 | Microscopy | Kato-katz thick smear | 667 (37.6%) | 667 (37.6%) | 691 (81.5%) | 143 (16.8%) | 81 (9.7%) | 122 (14.4%) | 21 (2.5%) | ||||||
| 11 | Olusola Ojurongbe | 2018 | Nigeria | Cross-sectional | 200 | 90 | 178 | 25 | Microscopy | Formalin-ether concentration techniques | 29.5% (59/200) | 12% (24/200) | 5% (10/200) | 2.0% (4/200) | 10.0% (20/200) | |||||||
| 12 | Olarewaju AB et al., 2016 | 2015 | Nigeria | Cross-sectional | 300 | 32 | 116 | 152 | 185 | 115 | Microscopy | Kato-Katz techniques | 14 (4.6) | 12 (4.0) | 73.1% (219) | 11 (3.6) | 15 (5.0) | 12 (4.0) | ||||
| 13 | Polycarp Uche Agu et al., 2013 | 2013 | Nigeria | Cross-sectional | 226 | 65 | 113 | 47 | Microscopy | Kato-Katz techniques | 119 | 90 (40%) | 60 (26.5%) | 14 (6.2%) | ||||||||
| 14 | R. Ndyomugyenyi et al., 2008 | 2007 | Uganda | Cross-sectional | 802 | Microscopy | Kato-Katz techniques | 281 (35%) | 219 (16%) | 554 (69%) | 4 (0.5%) | 38 (4.74%) | 31 (3.87%) | |||||||||
| 15 | Judith K. Anchang-Kimbi et al., 2017 | 2014 | Cameroon | Cross-sectional | 205 | 10 (4%) | 125 (50%) | 115 (46%) | Microscopy | Kato-Katz techniques | 98 (39.2%) | 38 (15.2%) | 117 (46.8%) | |||||||||
| 16 | Umeh et al., 2018 | 2017 | Nigeria | Cross-sectional | 300 | Microscopy | Kato-Katz techniques | 45 (15.0%) | 9 (3%) | 19 (6.3%) | ||||||||||||
| 17 | E. W. Nnah and T. Kasso 2018 | 2016 | Nigeria | Cross-sectional | 192 | Microscopy | Kato-Katz techniques | 47 (24.5%) | 32 (16.7%) | 1 (0.5%) | 6 (3.1%) | 144 (75%) | ||||||||||
| 18 | Akinbo et al., 2017 | 2014 | Nigeria | Cross-sectional | 402 | Microscopy | Kato-Katz techniques | 100 (24.9%) | 73 (18.2%) | 173 (43.14%) | 12 (3%) | 36 (9%) | 10 (2.5%) | |||||||||
| 19 | Ekejindu IM et al., 2011 | 2015 | Nigeria | Cross-sectional | 100 | Microscopy | Kato-Katz techniques | 81 (81%) | 17 (13%) | 17 (17%) | ||||||||||||
| 20 | Obeagu E. Ifeanyi., 2014 | 2012 | Nigeria | Cross-sectional | 87 | Microscopy | Kato-Katz techniques | 44 (51%) | 11 (13%) | 16 (18%) | ||||||||||||
| 21 | Jessica K. Fairley., 2014 | 2005 | Kenya | Cross-sectional | 696 | Microscopy | Kato-Katz techniques | 297 (42.7%) | 205 (29.5%) | 219 (31.5%) | 41 (5.9%) | |||||||||||
| 22 | Fuseini et al., 2010 | 2005 | Ghana | Cross-sectional | 300 | Microscopy | Kato-Katz techniques | 174 (58%) | 69 (23%) | 21 (7%) | 2 (0.7%) | 37 (12.3%) | ||||||||||
| 23 | Masai, Rael Jepkogei, 2016 | 2015 | Kenya | Cross-sectional | 300 | Microscopy | Kato-Katz techniques | 24 (8%) | 39 (13%) | 45 (15%) | 90 (30%) | 3 (1%) | ||||||||||
| 24 | Y. J. Honkpehedji et al., 2017 | 2015 | Gabon | Cross-sectional | 678 | Microscopy | Kato-Katz techniques | 221 (33%) | 259 (38%) | 468 (69%) | ||||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tadesse Boltena, M.; El-Khatib, Z.; Kebede, A.S.; Asamoah, B.O.; Yaw, A.S.C.; Kamara, K.; Constant Assogba, P.; Tadesse Boltena, A.; Adane, H.T.; Hailemeskel, E.; et al. Malaria and Helminthic Co-Infection during Pregnancy in Sub-Saharan Africa: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2022, 19, 5444. https://doi.org/10.3390/ijerph19095444
Tadesse Boltena M, El-Khatib Z, Kebede AS, Asamoah BO, Yaw ASC, Kamara K, Constant Assogba P, Tadesse Boltena A, Adane HT, Hailemeskel E, et al. Malaria and Helminthic Co-Infection during Pregnancy in Sub-Saharan Africa: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2022; 19(9):5444. https://doi.org/10.3390/ijerph19095444
Chicago/Turabian StyleTadesse Boltena, Minyahil, Ziad El-Khatib, Abraham Sahilemichael Kebede, Benedict Oppong Asamoah, Appiah Seth Christopher Yaw, Kassim Kamara, Phénix Constant Assogba, Andualem Tadesse Boltena, Hawult Taye Adane, Elifaged Hailemeskel, and et al. 2022. "Malaria and Helminthic Co-Infection during Pregnancy in Sub-Saharan Africa: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 19, no. 9: 5444. https://doi.org/10.3390/ijerph19095444








