Investigating the Interplay of Toxic Metals and Essential Elements in Liver Disease
Abstract
1. Introduction
2. Methodologies
2.1. Study Design
2.2. US-FLI and NAFLD
2.3. Variables and Covariates for Model Adjustment
2.4. Statistical Analysis
2.4.1. Descriptive Statistics and Regression Analysis
2.4.2. Bayesian Kernel Machine Regression (BKMR)
3. Results
3.1. Descriptive Analysis of Sex, Ethnicity, and Age
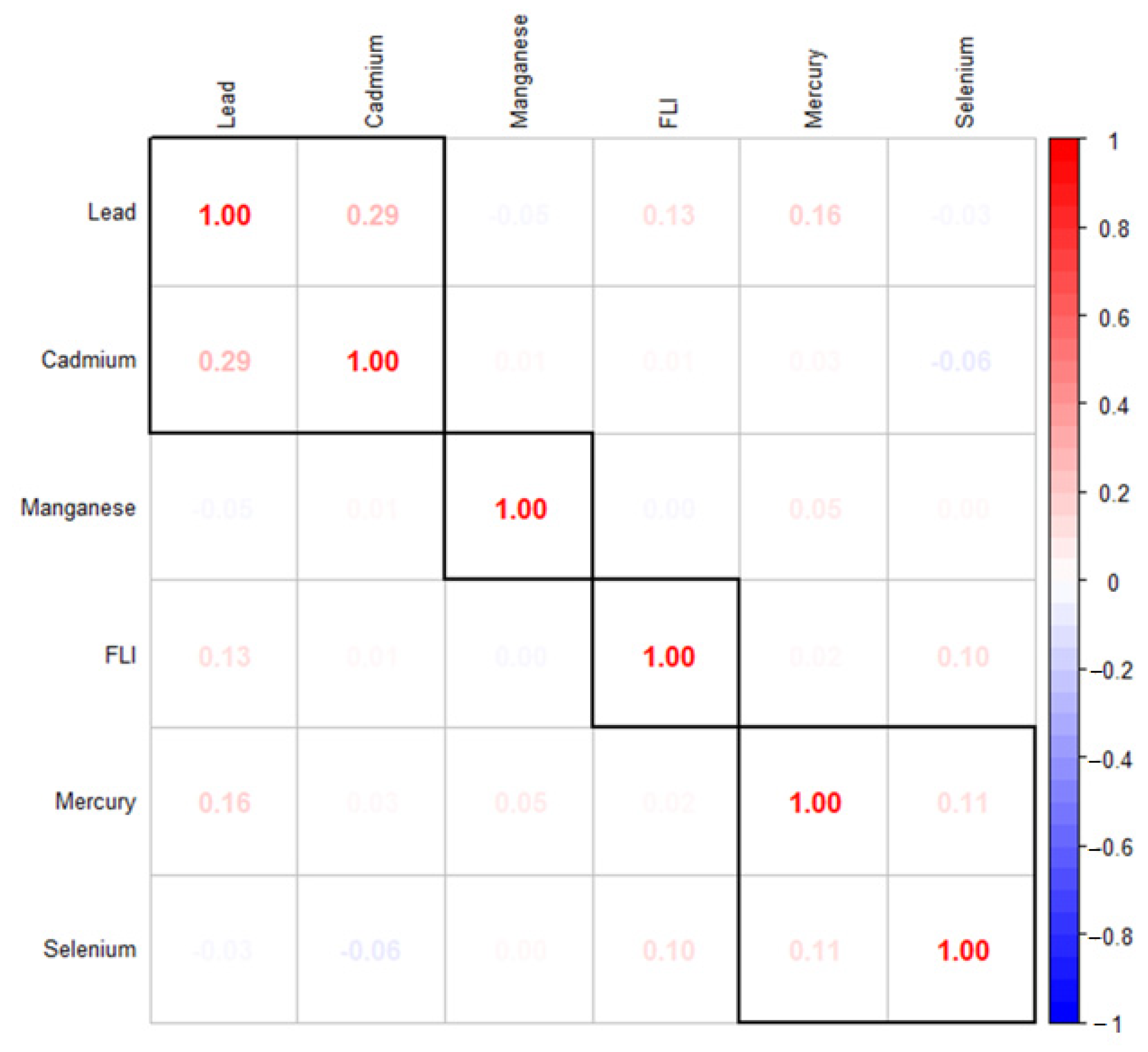
Correlation between Variables of Interest
3.2. Linear Regression for Association of Combined Toxic Metals and Essential Element on Liver Dysfunction
3.3. BKMR Results
3.3.1. Analysis of Posterior Inclusion Probabilities (PIPs) for Metals and Essential Elements in Relation to NAFLD Risk
3.3.2. Univariate Analysis: Examining the Isolated Effects of Pb, Cd, Hg, Se, and Mn on NAFLD
3.3.3. Bivariate Exposure–Response Function of Toxic Metals and Essential Elements with NAFLD
3.4. Overall Risk Summary of NAFLD Levels in Relation to Exposure Percentiles
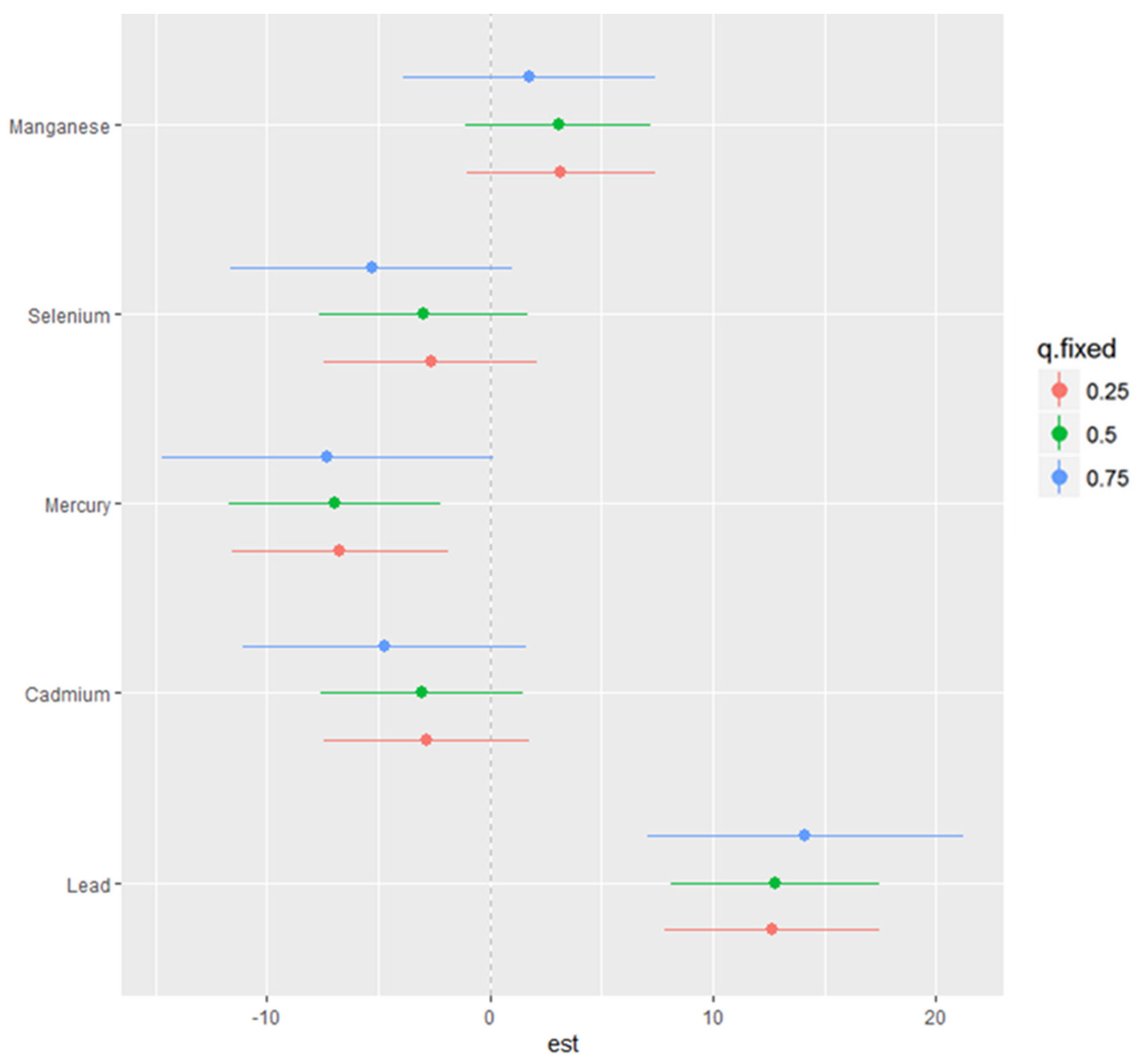
3.5. Single-Variable Effects of Toxic Metals and Essential Elements on NAFLD
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Asrani, S.K.; Devarbhavi, H.; Eaton, J.; Kamath, P.S. Burden of liver diseases in the world. J. Hepatol. 2019, 70, 151–171. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Sounds Alarm on Viral Hepatitis Infections Claiming 3500 Lives Each Day. 2024. Available online: https://www.who.int/news/item/09-04-2024-who-sounds-alarm-on-viral-hepatitis-infections-claiming-3500-lives-each-day (accessed on 11 June 2024).
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global Epidemiology of Nonalcoholic Fatty Liver Disease—Meta-Analytic Assessment of Prevalence, Incidence, and Outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef]
- Perumpail, B.J.; Khan, M.A.; Yoo, E.R.; Cholankeril, G.; Kim, D.; Ahmed, A. Clinical Epidemiology and Disease Burden of Nonalcoholic Fatty Liver Disease. World J. Gastroenterol. 2017, 23, 8263. [Google Scholar] [CrossRef]
- Seitz, H.K.; Bataller, R.; Cortez-Pinto, H.; Gao, B.; Gual, A.; Lackner, C.; Mathurin, P.; Mueller, S.; Szabo, G.; Tsukamoto, H. Alcoholic Liver Disease. Nat. Rev. Dis. Primers 2018, 4, 16. [Google Scholar] [CrossRef] [PubMed]
- Dolce, A.; Della Torre, S. Sex, Nutrition, and NAFLD: Relevance of Environmental Pollution. Nutrients 2023, 15, 2335. [Google Scholar] [CrossRef] [PubMed]
- Romero-Gómez, M.; Zelber-Sagi, S.; Trenell, M. Treatment of NAFLD with Diet, Physical Activity, and Exercise. J. Hepatol. 2017, 67, 829–846. [Google Scholar] [CrossRef]
- Machado, M.V.; Cortez-Pinto, H. Diet, Microbiota, Obesity, and NAFLD: A Dangerous Quartet. Int. J. Mol. Sci. 2016, 17, 481. [Google Scholar] [CrossRef]
- Ozougwu, J.C. Physiology of the Liver. Int. J. Res. Pharm. Biosci. 2017, 4, 13–24. [Google Scholar]
- Himoto, T.; Masaki, T. Associations between Zinc Deficiency and Metabolic Abnormalities in Patients with Chronic Liver Disease. Nutrients 2018, 10, 88. [Google Scholar] [CrossRef]
- Mohammad, M.K.; Zhou, Z.; Cave, M.; Barve, A.; McClain, C.J. Zinc and Liver Disease. Nutr. Clin. Pract. 2012, 27, 8–20. [Google Scholar] [CrossRef]
- Nordberg, M.; Nordberg, G.F. Trace Element Research—Historical and Future Aspects. J. Trace Elem. Med. Biol. 2016, 38, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Järup, L. Hazards of Heavy Metal Contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Tao, R.; Huang, J.; He, X.; Qu, L.; Jin, Y.; Zhang, S.; Fu, Z. Hepatic Oxidative Stress and Inflammatory Responses with Cadmium Exposure in Male Mice. Environ. Toxicol. Pharmacol. 2015, 39, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Das, S.C.; Al-Naemi, H.A. Cadmium Toxicity: Oxidative Stress, Inflammation, and Tissue Injury. Occup. Dis. Environ. Med. 2019, 7, 144–163. [Google Scholar] [CrossRef]
- Cave, M.; Appana, S.; Patel, M.; Falkner, K.C.; McClain, C.J.; Brock, G. Polychlorinated Biphenyls, Lead, and Mercury Are Associated with Liver Disease in American Adults: NHANES 2003–2004. Environ. Health Perspect. 2010, 118, 1735–1742. [Google Scholar] [CrossRef] [PubMed]
- Hyder, O.; Chung, M.; Cosgrove, D.; Herman, J.M.; Li, Z.; Firoozmand, A.; Gurakar, A.; Koteish, A.; Pawlik, T.M. Cadmium Exposure and Liver Disease among US Adults. J. Gastrointest. Surg. 2013, 17, 1265–1273. [Google Scholar] [CrossRef]
- Taylor, K.W.; Joubert, B.R.; Braun, J.M.; Dilworth, C.; Gennings, C.; Hauser, R.; Heindel, J.J.; Rider, C.V.; Webster, T.F.; Carlin, D.J. Statistical Approaches for Assessing Health Effects of Environmental Chemical Mixtures in Epidemiology: Lessons from an Innovative Workshop. Environ. Health Perspect. 2016, 124, A227–A229. [Google Scholar] [CrossRef] [PubMed]
- Bobb, J.F.; Valeri, L.; Henn, B.C.; Christiani, D.C.; Wright, R.O.; Mazumdar, M.; Godleski, J.J.; Coull, B.A. Bayesian Kernel Machine Regression for Estimating the Health Effects of Multi-Pollutant Mixtures. Biostatistics 2015, 16, 493–508. [Google Scholar] [CrossRef] [PubMed]
- Coull, B.A.; Bobb, J.F.; Wellenius, G.A.; Kioumourtzoglou, M.-A.; Mittleman, M.A.; Koutrakis, P.; Godleski, J.J. Part 1. Statistical Learning Methods for the Effects of Multiple Air Pollution Constituents. Res. Rep. Health Eff. Inst. 2015, 183, 1–2. [Google Scholar]
- Liu, S.H.; Bobb, J.F.; Lee, K.H.; Gennings, C.; Henn, B.C.; Bellinger, D.; Austin, C.; Schnaas, L.; Tellez-Rojo, M.M.; Hu, H.; et al. Lagged Kernel Machine Regression for Identifying Time Windows of Susceptibility to Exposures of Complex Mixtures. Biostatistics 2018, 19, 325–341. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Alanine Aminotransferase (ALT) Laboratory Procedure Manual. 2020. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2017-2018/labmethods/BIOPRO-J-MET-ALT-508.pdf (accessed on 11 June 2024).
- Centers for Disease Control and Prevention. Aspartate Aminotransferase (AST) Laboratory Procedure Manual. 2020. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2017-2018/labmethods/BIOPRO-J-MET-AST-508.pdf (accessed on 11 June 2024).
- Centers for Disease Control and Prevention. Alkaline Phosphatase (ALP) Laboratory Procedure Manual. 2020. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2017-2018/labmethods/BIOPRO-J-MET-ALP-508.pdf (accessed on 11 June 2024).
- Centers for Disease Control and Prevention. Gamma-Glutamyltransferase (GGT) Laboratory Procedure Manual. 2020. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2017-2018/labmethods/BIOPRO-J-MET-GGT-508.pdf (accessed on 11 June 2024).
- Centers for Disease Control and Prevention. Laboratory Procedure Manual. 2020. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2017-2018/labmethods/BIOPRO-J-MET-Total-Bilirubin-508.pdf (accessed on 11 June 2024).
- R Core Team. R: A Language and Environment for Statistical Computing. 2020. Available online: https://www.r-project.org/ (accessed on 11 June 2024).
- Vesey, D.A. Transport Pathways for Cadmium in the Intestine and Kidney Proximal Tubule: Focus on the Interaction with Essential Metals. Toxicol. Lett. 2010, 198, 13–19. [Google Scholar] [CrossRef]
- Reeves, P.G.; Chaney, R.L. Marginal Nutritional Status of Zinc, Iron, and Calcium Increases Cadmium Retention in the Duodenum and Other Organs of Rats Fed Rice-Based Diets. Environ. Res. 2004, 96, 311–322. [Google Scholar] [CrossRef]
- Hammad, T.A.; Sexton, M.; Langenberg, P. Relationship between Blood Lead and Dietary Iron Intake in Preschool Children: A Cross-Sectional Study. Ann. Epidemiol. 1996, 6, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Duruibe, J.O.; Ogwuegbu, M.; Egwurugwu, J. Heavy Metal Pollution and Human Biotoxic Effects. Int. J. Phys. Sci. 2007, 2, 112–118. [Google Scholar]
- Markiewicz-Górka, I.; Januszewska, L.; Michalak, A.; Prokopowicz, A.; Januszewska, E.; Pawlas, N.; Pawlas, K. Effects of Chronic Exposure to Lead, Cadmium, and Manganese Mixtures on Oxidative Stress in Rat Liver and Heart. Arh. Hig. Rada Toksikol. 2015, 66, 51–61. [Google Scholar] [CrossRef]
- Jomova, K.; Makova, M.; Alomar, S.Y.; Alwasel, S.H.; Nepovimova, E.; Kuca, K.; Rhodes, C.J.; Valko, M. Essential Metals in Health and Disease. Chem. Biol. Interact. 2022, 367, 110173. [Google Scholar] [CrossRef]
- Niemelä, O.; Alatalo, P.J.S.J.C. Biomarkers of Alcohol Consumption and Related Liver Disease. Chem.-Biol. Interact. 2010, 70, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Conway, M. Aminotransferases. In Amino Acids in Human Nutrition and Health; CABI: Wallingford, UK, 2012; pp. 24–50. [Google Scholar]
- Obeng-Gyasi, E. Sources of Lead Exposure in Various Countries. Rev. Environ. Health 2019, 34, 25–34. [Google Scholar] [CrossRef]
- Vaziri, N.D. Mechanisms of Lead-Induced Hypertension and Cardiovascular Disease. Am. J. Physiol. Heart Circ. Physiol. 2008, 295, H454–H465. [Google Scholar] [CrossRef]
- Huang, X.-J.; Choi, Y.-K.; Im, H.-S.; Yarimaga, O.; Yoon, E.; Kim, H.-S. Aspartate Aminotransferase (AST/GOT) and Alanine Aminotransferase (ALT/GPT) Detection Techniques. Sensors 2006, 6, 756–782. [Google Scholar] [CrossRef]
- Makris, K.; Mousa, C.; Cavalier, E. Alkaline Phosphatases: Biochemistry, Functions, and Measurement. Calcif. Tissue Int. 2023, 112, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Lowe, D.; Sanvictores, T.; Zubair, M.; John, S. Alkaline Phosphatase. StatPearls. 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK459201/ (accessed on 11 June 2024).
- Li, L.; Yang, X. The Essential Element Manganese, Oxidative Stress, and Metabolic Diseases: Links and Interactions. Oxid. Med. Cell. Longev. 2018, 2018, 7580707. [Google Scholar] [CrossRef]
- Santamaria, A. Manganese Exposure, Essentiality & Toxicity. Indian J. Med. Res. 2008, 128, 484–500. [Google Scholar] [PubMed]
- Obeng-Gyasi, E.; Armijos, R.X.; Weigel, M.M.; Filippelli, G.; Sayegh, M.A. Hepatobiliary-Related Outcomes in US Adults Exposed to Lead. Environments 2018, 5, 46. [Google Scholar] [CrossRef]
- Kunutsor, S.K. Gamma-Glutamyltransferase—Friend or Foe within? Liver Int. 2016, 36, 1723–1734. [Google Scholar] [CrossRef]
- Jansen, P.L.M.; Bittar, E.E. Bilirubin Metabolism. Princ. Med. Biol. 2004, 15, 257–289. [Google Scholar]
- Guerra Ruiz, A.R.; Crespo, J.; López Martínez, R.M.; Iruzubieta, P.; Casals Mercadal, G.; Lalana Garcés, M.; Lavin, B.; Morales Ruiz, M. Measurement and Clinical Usefulness of Bilirubin in Liver Disease. Adv. Lab. Med./Av. En Med. De Lab. 2021, 2, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Lockitch, G. Selenium: Clinical Significance and Analytical Concepts. Crit. Rev. Clin. Lab. Sci. 1989, 27, 483–541. [Google Scholar] [CrossRef]
- Dehnavi, Z.; Razmpour, F.; Belghaisi Naseri, M.; Nematy, M.; Alamdaran, S.A.; Vatanparast, H.A.; Azimi Nezhad, M.; Abbasi, B.; Ganji, A. Fatty Liver Index (FLI) in Predicting Non-Alcoholic Fatty Liver Disease (NAFLD). Hepat. Mon. 2018, 18, e63227. [Google Scholar] [CrossRef]
- Wu, J.; Zeng, C.; Yang, Z.; Li, X.; Lei, G.; Xie, D.; Wang, Y.; Wei, J.; Yang, T. Association between Dietary Selenium Intake and the Prevalence of Nonalcoholic Fatty Liver Disease: A Cross-Sectional Study. J. Am. Coll. Nutr. 2020, 39, 103–111. [Google Scholar] [CrossRef]
- Boafo, Y.S.; Mostafa, S.; Obeng-Gyasi, E. Association of Combined Metals and PFAS with Cardiovascular Disease Risk. Toxics 2023, 11, 979. [Google Scholar] [CrossRef] [PubMed]
- Rajak, S.; Raza, S.; Tewari, A.; Sinha, R.A. Environmental Toxicants and NAFLD: A Neglected yet Significant Relationship. Dig. Dis. Sci. 2022, 67, 3497–3507. [Google Scholar] [CrossRef]
- Wan, H.; Wang, Y.; Zhang, H.; Zhang, K.; Chen, Y.; Chen, C.; Zhang, W.; Xia, F.; Wang, N.; Lu, Y. Chronic Lead Exposure Induces Fatty Liver Disease Associated with the Variations of Gut Microbiota. Ecotoxicol. Environ. Saf. 2022, 232, 113257. [Google Scholar] [CrossRef] [PubMed]
- Haruna, I.; Obeng-Gyasi, E. Association of Combined Per-and Polyfluoroalkyl Substances and Metals with Chronic Kidney Disease. Int. J. Environ. Res. Public Health 2024, 21, 468. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Yang, Y.; Wen, C.; Liu, W.; Cao, L.; Feng, X.; Chen, J.; Wang, H.; Tang, Y.; Tian, L.; et al. Effects of Environmental Contaminants in Water Resources on Nonalcoholic Fatty Liver Disease. Environ. Int. 2021, 154, 106555. [Google Scholar] [CrossRef] [PubMed]
- Obeng-Gyasi, E. Cumulative Effects of Low-Level Lead Exposure and Chronic Physiological Stress on Hepatic Dysfunction—A Preliminary Study. Med. Sci. 2020, 8, 30. [Google Scholar] [CrossRef]
- Nassir, F. NAFLD: Mechanisms, Treatments, and Biomarkers. Biomolecules 2022, 12, 824. [Google Scholar] [CrossRef]



| (a) | ||||
|---|---|---|---|---|
| Sex | Mean | Linearized Std. Error | (95% Conf. Interval) | |
| Male | 37.427 | 0.493 | 36.375 | 38.478 |
| Female | 39.378 | 0.592 | 38.116 | 40.640 |
| (b) | ||||
| Ethnicity | Mean | Linearized Std. Error | (95% Conf. Interval) | |
| Mexican American | 29.319 | 0.852 | 27.504 | 31.134 |
| Other Hispanic | 33.685 | 0.900 | 31.766 | 35.604 |
| Non-Hispanic White | 41.670 | 0.785 | 39.996 | 43.343 |
| Non-Hispanic Black | 35.830 | 0.540 | 34.679 | 36.982 |
| Non-Hispanic Asian | 37.926 | 0.938 | 35.925 | 39.927 |
| Other Race—Including Multi-Racial | 33.475 | 1.822 | 29.591 | 37.359 |
| (a) | |||||
|---|---|---|---|---|---|
| ALT | * Coefficient | Linearized Std. Error | p-Value | (95% Conf. Interval) | |
| Pb | 0.355 | 0.433 | 0.425 | −0.568 | 1.277 |
| Cd | 0.101 | 0.924 | 0.915 | −1.869 | 2.071 |
| Hg | −0.401 | 0.344 | 0.262 | −1.133 | 0.332 |
| Mn | 0.364 | 0.120 | 0.005 | 0.130 | 0.598 |
| Se | 0.040 | 0.022 | 0.086 | −0.006 | 0.086 |
| (b) | |||||
| AST | * Coefficient | Linearized Std. Error | p-Value | (95% Conf. Interval) | |
| Pb | 0.908 | 0.347 | 0.019 | 0.169 | 1.647 |
| Cd | 0.577 | 1.268 | 0.656 | −2.125 | 3.279 |
| Hg | −0.197 | 0.232 | 0.409 | −0.691 | 0.297 |
| Mn | 0.324 | 0.153 | 0.052 | −0.003 | 0.652 |
| Se | −0.004 | 0.016 | 0.819 | −0.039 | 0.031 |
| (c) | |||||
| ALP | * Coefficient | Linearized Std. Error | p-Value | (95% Conf. Interval) | |
| Pb | 2.509 | 0.853 | 0.010 | 0.690 | 4.327 |
| Cd | 0.480 | 1.592 | 0.767 | −2.914 | 3.875 |
| Hg | −1.258 | 0.453 | 0.014 | −2.223 | −0.292 |
| Mn | 0.650 | 0.304 | 0.050 | 0.001 | 1.299 |
| Se | −0.059 | 0.028 | 0.048 | −0.118 | −0.000 |
| (d) | |||||
| GGT | * Coefficient | Linearized Std. Error | p-Value | (95% Conf. Interval) | |
| Pb | 3.287 | 1.067 | 0.008 | 1.013 | 5.561 |
| Cd | 4.411 | 2.861 | 0.144 | −1.686 | 10.509 |
| Hg | −0.890 | 0.399 | 0.041 | −1.741 | −0.041 |
| Mn | 1.205 | 0.361 | 0.005 | 0.435 | 1.976 |
| Se | 0.010 | 0.030 | 0.738 | −0.053 | 0.074 |
| (e) | |||||
| Total Bilirubin | * Coefficient | Linearized Std. Error | p-Value | (95% Conf. Interval) | |
| Pb | −0.000 | 0.005 | 0.987 | −0.011 | 0.011 |
| Cd | −0.012 | 0.008 | 0.142 | −0.028 | 0.004 |
| Hg | 0.005 | 0.007 | 0.458 | −0.009 | 0.019 |
| Mn | −0.000 | 0.003 | 0.896 | −0.006 | 0.005 |
| Se | 0.001 | 0.000 | 0.006 | 0.000 | 0.002 |
| (f) | |||||
| US-FLI | * Coefficient | Linearized Std. Error | p-Value | (95% Conf. Interval) | |
| Pb | 0.800 | 0.768 | 0.315 | −2.435 | 0.838 |
| Cd | 2.380 | 1.359 | 0.100 | −0.515 | 5.276 |
| Hg | 0.049 | 0.349 | 0.890 | −0.696 | 0.794 |
| Mn | 0.478 | 0.211 | 0.039 | 0.028 | 0.929 |
| Se | 0.035 | 0.025 | 0.179 | −0.018 | 0.087 |
| Variable | PIP |
|---|---|
| Pb | 1.0000 |
| Hg | 0.9512 |
| Cd | 0.5796 |
| Se | 0.5572 |
| Mn | 0.4248 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adetunji, A.G.; Obeng-Gyasi, E. Investigating the Interplay of Toxic Metals and Essential Elements in Liver Disease. Int. J. Environ. Res. Public Health 2024, 21, 762. https://doi.org/10.3390/ijerph21060762
Adetunji AG, Obeng-Gyasi E. Investigating the Interplay of Toxic Metals and Essential Elements in Liver Disease. International Journal of Environmental Research and Public Health. 2024; 21(6):762. https://doi.org/10.3390/ijerph21060762
Chicago/Turabian StyleAdetunji, Aderonke Gbemi, and Emmanuel Obeng-Gyasi. 2024. "Investigating the Interplay of Toxic Metals and Essential Elements in Liver Disease" International Journal of Environmental Research and Public Health 21, no. 6: 762. https://doi.org/10.3390/ijerph21060762
APA StyleAdetunji, A. G., & Obeng-Gyasi, E. (2024). Investigating the Interplay of Toxic Metals and Essential Elements in Liver Disease. International Journal of Environmental Research and Public Health, 21(6), 762. https://doi.org/10.3390/ijerph21060762







