Abstract
The purpose of this meta-analysis was to evaluate the efficacy and safety of celecoxib, a selective cyclooxygenase-2 (COX-2) inhibitor, in addition to standard anticancer therapy. Randomized controlled trials (RCTs) that evaluated the efficacy and safety of celecoxib-combined cancer therapy were systematically searched in PubMed and Embase databases. The endpoints were overall survival (OS), progression-free survival (PFS), disease-free survival (DFS), objective response rate (ORR), disease control rate (DCR), pathological complete response (pCR), and adverse events (AEs). The results of 30 RCTs containing 9655 patients showed limited benefits in celecoxib-combined cancer therapy. However, celecoxib-combined palliative therapy prolonged PFS in epidermal growth factor receptor (EGFR) wild-type patients (HR = 0.57, 95%CI = 0.35–0.94). Moreover, despite a slight increase in thrombocytopenia (RR = 1.35, 95%CI = 1.08–1.69), there was no increase in other toxicities. Celecoxib combined with adjuvant therapy indicated a better OS (HR = 0.850, 95%CI = 0.725–0.996). Furthermore, celecoxib plus neoadjuvant therapy improved the ORR in standard cancer therapy, especially neoadjuvant therapy (overall: RR = 1.13, 95%CI = 1.03–1.23; neoadjuvant therapy: RR = 1.25, 95%CI = 1.09–1.44), but not pCR. Our study indicated that adding celecoxib to palliative therapy prolongs the PFS of EGFR wild-type patients, with good safety profiles. Celecoxib combined with adjuvant therapy prolongs OS, and celecoxib plus neoadjuvant therapy improves the ORR. Thus, celecoxib-combined cancer therapy may be a promising therapy strategy.
1. Introduction
Cancer is a continuing global public health challenge and the second leading cause of death worldwide, closely following cardiovascular (CV) disease [1]. Thus, cancer prevention and therapy are particularly important. Although early diagnosis and therapy strategies of cancer are improving, high mortality and poor prognosis of cancer patients remains a challenge that needs to be addressed [2]. The development of comprehensive therapy strategies may be more beneficial, but the toxicity and side effects of chemotherapy require further consideration. Thus, there remains a need to improve current chemotherapy regimens to maximize the cure rate and minimize chemotherapy-associated toxicity.
Increasing numbers of studies have indicated that tumor occurrence, development, and progression may be promoted by inflammation [3]. The cyclooxygenase-2 (COX-2) enzyme, one of the isoforms of the COX enzyme, catalyzes the production of prostaglandins from arachidonic acid [4]. In addition, COX-2 can be activated by the growth factors, cytokines, and chemokines that are released by the trigger of inflammation [5]. This inflammation-associated metabolic process may play a role in cancer, and COX-2 overexpression is associated with tumor development and progression, tumor characteristics, and poor survival [6,7,8]. Therefore, COX-2 inhibitors are likely to be promising anticancer drugs. Recently, COX-2 inhibitors have been explored in cancer therapy to improve therapeutic efficacy. COX-2 inhibitors may work by antiangiogenic, anti-inflammatory, and proapoptotic mechanisms to promote their antitumor effects [9]. However, there is conflicting data surrounding the efficacy of COX-2 inhibitors combined with anticancer therapy. Furthermore, unlike COX-1 inhibitors, which mainly cause gastrointestinal toxicities due to COX-1 predominating in the gastric mucosa and yielding protective prostaglandins, the COX-2 inhibitors may lead to increased CV thrombotic events by decreasing vasodilatory and antiaggregatory vascular prostacyclin (PGI2) production, which results in the increase of prothrombotic eicosanoids (e.g., thromboxane A2) [10,11]. Therefore, it remains unclear whether the additional COX-2 inhibitors in tumor therapy are clinically beneficial and safe for cancer patients.
Celecoxib, a highly selective COX-2 inhibitor, is an established anti-inflammatory drug that has been used for treating osteoarthritis, rheumatoid arthritis, familial adenomatous polyposis, and acute pain for many years [12]. Numerous studies have reported the chemopreventive efficacy of celecoxib in several precancerous lesions and cancer types, including colon polyp, colorectal adenomas, lung cancer, and prostate cancer [13,14,15,16,17]. Furthermore, celecoxib was found to prevent tumor growth through multiple pathways and targets [18,19,20,21,22,23,24,25,26]. In terms of the molecular mechanism of celecoxib, it can regulate FAK, Cx43, p21, and Ki-67 molecules, and block AKT activation [20,22,24], which can inhibit tumor cell proliferation and induce apoptosis. Moreover, celecoxib is involved in ERK1/2 MAPK and PI3K/AKT pathways, exerting an antiangiogenic effect [18,23]. Celecoxib is also involved in COX-2/PGE2/EP2/p-AKT/p-ERK and PGE2/NF-kB pathways, which prevents the invasion and metastasis of tumor cells [25,26]. Previous animal studies and clinical studies have reported that the administration of celecoxib with chemotherapy could significantly relieve chemotherapy-related toxicities [27,28]. In terms of celecoxib in addition to cancer therapy, some clinical studies reported that additional celecoxib in palliative hormone therapy seemingly contributes to reverse endocrine resistance in breast cancer patients and improves the clinical efficacy of preoperative chemoradiation in rectal cancer [29,30], but no significant clinical benefit was found in celecoxib-combined palliative therapy for lung cancer and colorectal cancer, nor in celecoxib-combined adjuvant hormone therapy for breast cancer [31,32,33]. Since different cancers have varying biological characteristics and exhibit different responses to therapy regimens, the conflicting data may be attributed to cancer type and therapy approach. It is therefore critical to explore whether celecoxib is beneficial to patient prognosis and, if so, which potential patient populations will benefit from celecoxib–targeted anticancer therapy.
In the present study, we collected relevant RCTs and conducted a systematic meta-analysis to assess the efficacy and safety of celecoxib combined with standard anticancer therapy in various cancers and therapy strategies.
2. Materials and Methods
2.1. Literature Search
We systematically searched PubMed and Embase databases up to November 2021 for all relevant RCT studies that evaluated the efficacy and safety of celecoxib in addition to standard therapy for cancer patients, including palliative chemotherapy, adjuvant chemotherapy, and neoadjuvant chemotherapy. The search strategy used the main keywords and MeSH terms of “celecoxib”, “Celebrex”, “cancer”, “tumor”, “neoplasm”, “carcinoma”, “palliative”, “postoperative”, “post-operative”, “adjuvant”, “neoadjuvant”, “preoperative”, “pre-operative”, “chemotherapy”, “chemoradiation”, “hormone therapy”, “randomized trial”, “randomised trial”, “randomized study”, and “randomised study”. In addition, the reference lists of relevant literature were manually screened to identify any potentially eligible literature.
2.2. Eligibility Criteria
This meta-analysis aimed to evaluate whether additional celecoxib in standard therapy improved the prognosis for cancer patients. The inclusion criteria were as follows: (1) Patients: patients were pathologically diagnosed with cancer and treated with standard anticancer therapy. (2) Intervention: cancer patients received standard therapy with celecoxib as opposed to standard therapy alone or with placebo. (3) Comparison: the control group received standard therapy with or without placebo. (4) Outcomes: the study outcomes consisted of overall survival (OS), progression-free survival (PFS), disease-free survival (DFS), objective response rate (ORR), disease control rate (DCR), pathological complete response (pCR), and drug safety; the outcome measures and corresponding 95% confidence intervals (CIs) were reported. (5) Study design: the studies were RCTs. Given that radiotherapy is the local therapy for cancer, and we were focusing on assessing the systemic effects of celecoxib in cancer therapy, we excluded celecoxib-combined radiotherapy studies. If several studies were based on the same patient population, the relevant data of interest were merged into the most informative study, which was enrolled.
2.3. Data Extraction and Assessment of Study Quality
The relevant data of eligible RCTs extracted by two reviewers independently were as follows: first author, country, year of publication, therapy type, cancer type, sample size, age, follow-up period, long-term efficacy, short-term efficacy, and drug safety. The two reviewers resolved all discrepancies through comprehensive discussion in accordance with the PRISMA statement [34]. The study quality was evaluated on the basis of the Jadad criteria [35].
2.4. Statistical Analysis
The primary endpoints were long-term efficacies, including OS, PFS, and DFS. The secondary endpoints were short-term efficacies (ORR, DCR, and pCR) and adverse events (AEs). We extracted the hazard ratios (HRs), relative risks (RRs), and 95%CIs from each trial. In studies that did not obtain direct outcomes, HRs and 95%CIs were calculated according to the method designed by Tierney [36], and the ORR and DCR were calculated from the complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) data according to the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines [37].
The Cochran Q test and I2 index were conducted to evaluate the heterogeneity among RCTs, with I2 < 50% and p > 0.1 indicating no significant heterogeneity [38]. When significant heterogeneity was found, a random-effects model was conducted, otherwise a fixed-effects model was conducted. Overall analyses across all therapy approaches were made based on all the enrolled studies. Then, we focused on analyzing the efficacy and safety profile of celecoxib in addition to anticancer therapy, such as palliative, adjuvant, and neoadjuvant therapies. Simultaneously, subgroup analyses on different therapy methods were performed based on therapy type (chemo/chemoradiation/hormone therapy), cancer type, COX-2 status, the urinary metabolite of prostaglandin E2 (PGEM) status, epidermal growth factor receptor (EGFR) status, use of nonsteroidal anti-inflammatory drugs (NSAIDs), World Health Organization (WHO) performance score, and sample size. Begg’s and Egger’s tests were used to evaluate the publication bias [39,40].
STATA version 12.0 (Stata Corporation, College Station, TX, USA) was used to perform all statistical analyses. When a two-sided p-value was < 0.05, the results were regarded as statistically significant. The protocol of this systematic review was not a routine local research protocol on the PROSPERO registry by the time of completion of the work. Therefore, though the comprehensive web search showed no similar registered study in ROSPERO, we do not have a registration number.
3. Results
3.1. Study Selection and Associated Characteristics
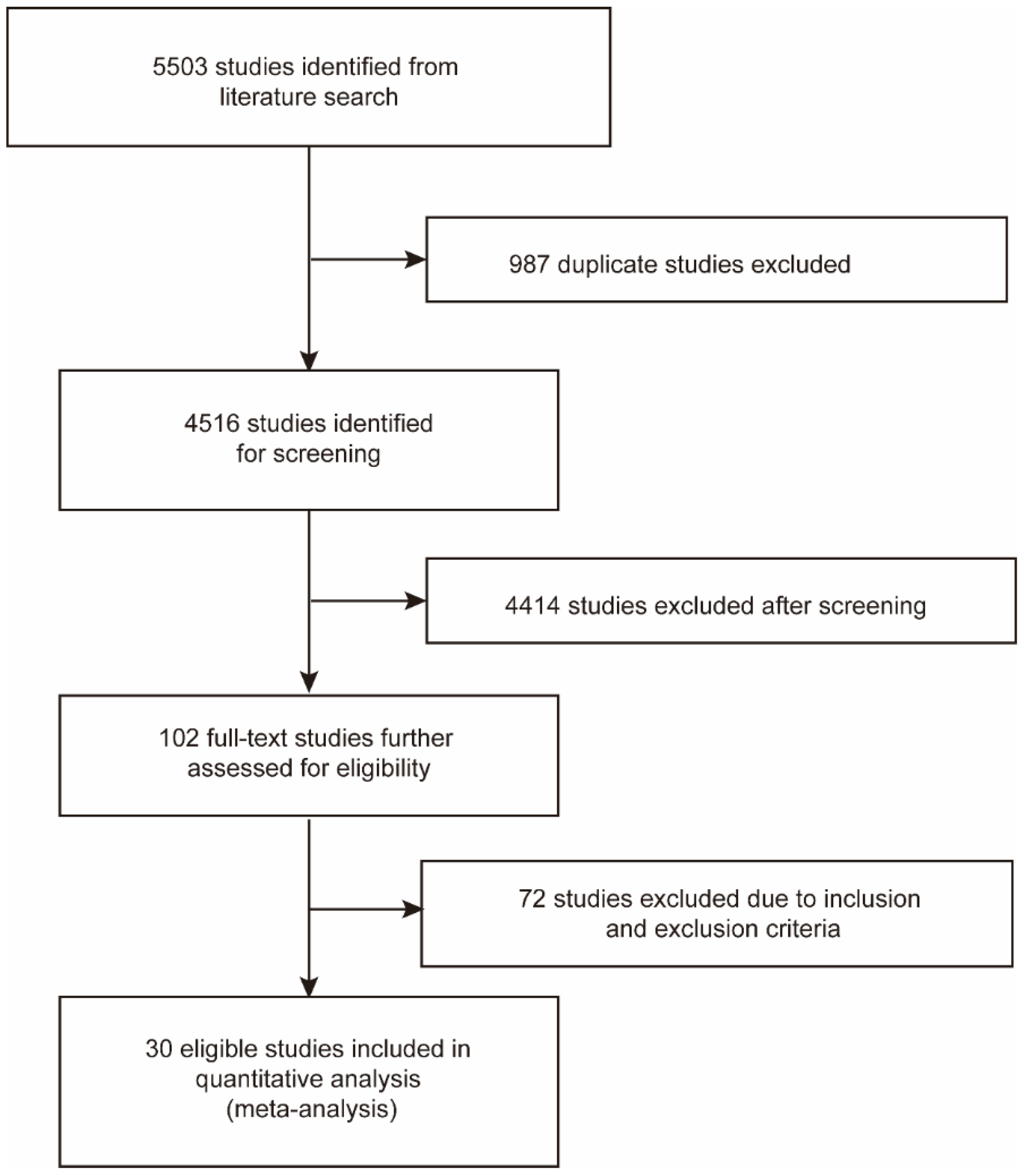
A total of 1368 and 4135 relevant studies were identified from PubMed and Embase databases, respectively. Of these, 987 duplicate studies were excluded. After screening the remaining 4516 studies according to the title and abstract, we excluded 4414 studies on the basis of eligibility criteria. Then we reviewed the full texts of the selected 102 studies and excluded 72 studies due to irrelevant data. Finally, 30 studies were included in this meta-analysis (Figure 1) [29,30,31,32,33,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65].
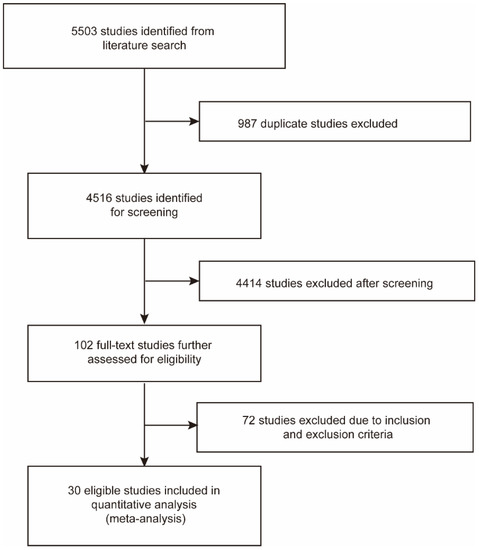
Figure 1.
Flow chart of the literature search and study selection process.
The 30 enrolled studies were published from 2004 to 2021 and contained a total of 9655 patients. Of these studies, Edelman et al. (2017), Koch et al. (2011), and Hamy et al. (2019) had merged the relevant data from Edelman et al. (2014), Gulyas et al. (2018), and Pierga et al. (2010) based on the same patient population, respectively [66,67,68]. Among the included studies, celecoxib was combined with palliative therapy in 20 studies, with adjuvant therapy in three studies and neoadjuvant therapy in seven. The enrolled studies were related to various cancers, including nine breast cancer studies, eight lung cancer studies, six colorectal cancer studies, two gynecological cancer studies, two head and neck cancer studies, one gastric cancer study, one prostate cancer study, and one bladder cancer study. The main characteristics and quality assessments of each included study are shown in Table S1.
3.2. Overall Efficacy of Celecoxib in Standard Anticancer Therapy
In a total of 30 enrolled studies, 17 reported OS, and the pooled result showed no improvement in celecoxib-combined cancer therapy (HR = 1.00, 95%CI = 0.92–1.08). Moreover, the addition of celecoxib in cancer therapy did not improve PFS (HR = 1.02, 95%CI = 0.91–1.13), nor DFS (HR = 1.05, 95%CI = 0.80–1.38). As for the local control, analyses indicated that patients in the celecoxib-combined cancer therapy group obtained a better ORR than the control group (39.3% versus 31.8%; RR = 1.13, 95%CI = 1.03–1.23), but not DCR (71.9% versus 68.9%; RR=1.05, 95%CI = 0.99–1.11). Furthermore, no improvement in pCR was found in the patients who received celecoxib-combined cancer therapy (RR = 1.28, 95%CI = 0.88–1.85). These analysis results are shown in Table 1.

Table 1.
The analysis results across all therapy approaches of celecoxib combined with cancer therapy for cancer patients.
3.3. Celecoxib and Overall Survival (OS) in Palliative Therapy
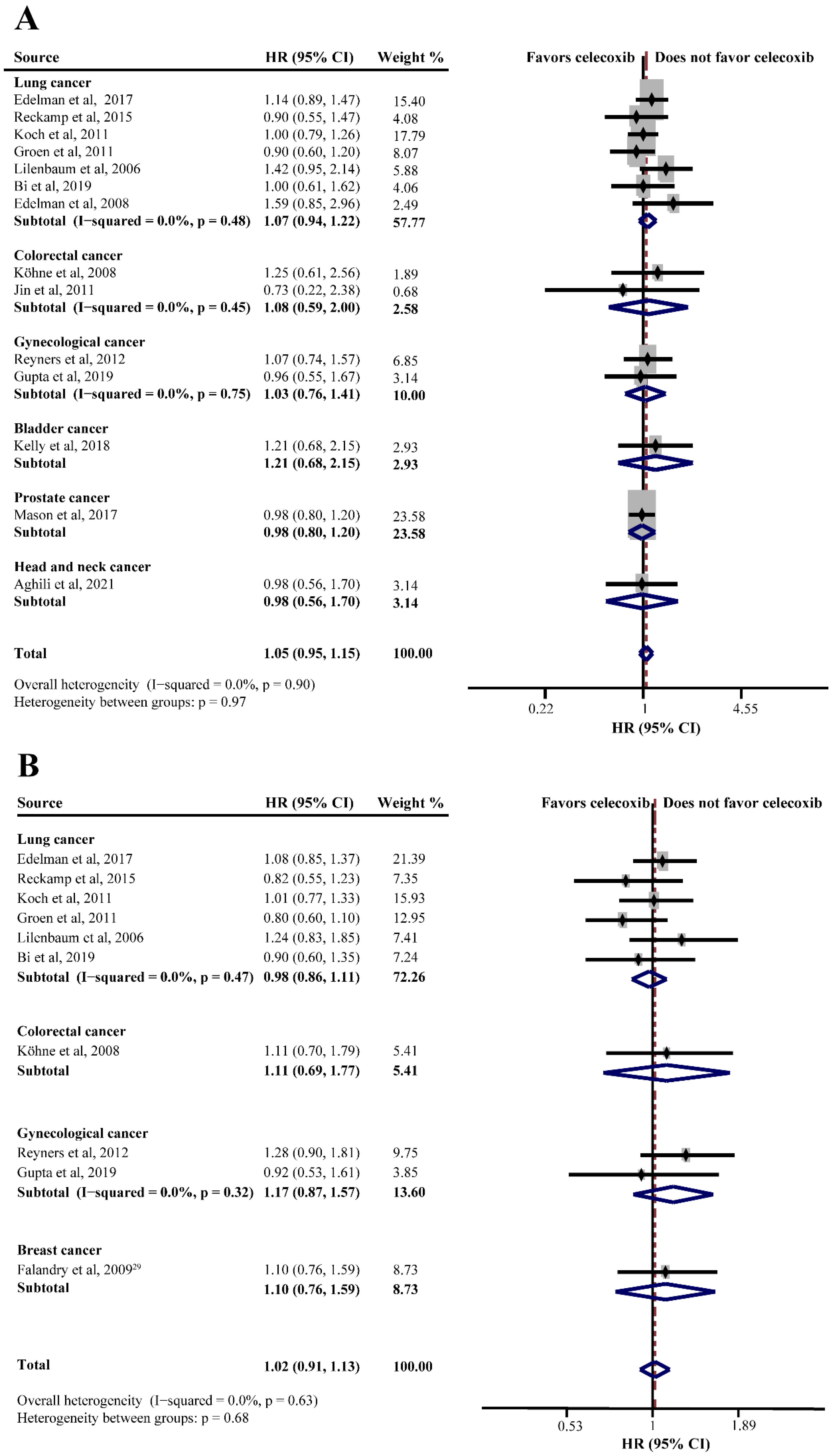
Twenty RCTs consisting of 4688 patients assessed the efficacy of celecoxib in addition to standard palliative therapy. Of these, 14 studies reported the OS of celecoxib-combined therapy versus standard therapy alone or with placebo. No improvement was shown in the OS (HR = 1.05, 95%CI = 0.95–1.15), without significant heterogeneity (I2 = 0.0%) (Figure 2A). Subgroup analysis based on different cancers showed no efficacy of celecoxib combined with palliative therapy on OS (lung cancer: HR = 1.07, 95%CI = 0.94–1.22; colorectal cancer: HR = 1.08, 95%CI = 0.59–2.00; gynecological cancer: HR = 1.03, 95%CI = 0.76–1.41) (Figure 2A). Moreover, the analyses revealed that celecoxib-combined chemotherapy or chemoradiotherapy did not prolong OS (chemotherapy: HR = 1.08, 95%CI = 0.96–1.21; chemoradiotherapy: HR = 0.99, 95%CI = 0.69–1.43). The addition of celecoxib to first-line palliative therapy did not obtain a better efficacy (HR = 1.03, 95%CI = 0.93–1.15), nor to ≥first-line palliative therapy (HR = 0.93, 95%CI = 0.64–1.34). Moreover, our meta-analyses revealed no beneficial effects with respect to COX-2 status, PGEM status, EGFR status, use of NSAIDs, and WHO performance status. Furthermore, we conducted a subgroup analysis after stratifying by sample size, and the results were still insignificant. The detailed results of the subgroup analysis for the OS of celecoxib-combined palliative therapy are shown in Table 2.
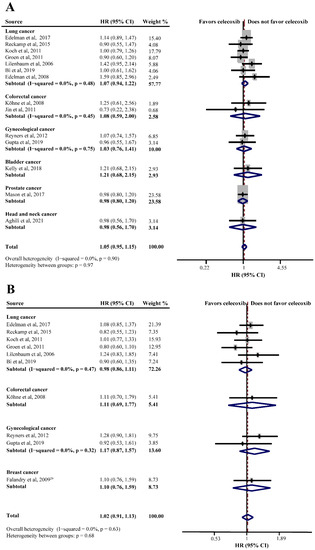
Figure 2.
Efficacy of celecoxib-combined palliative therapy in different cancer types on overall survival (OS) (A) and progression-free survival (PFS) (B). HR: hazard ratio; CI: confidence interval [29,31,41,42,48,50,51,52,56,58,59,60,61,62,63].

Table 2.
The results of subgroup analyses on the survival efficacy of celecoxib combined with palliative therapy.
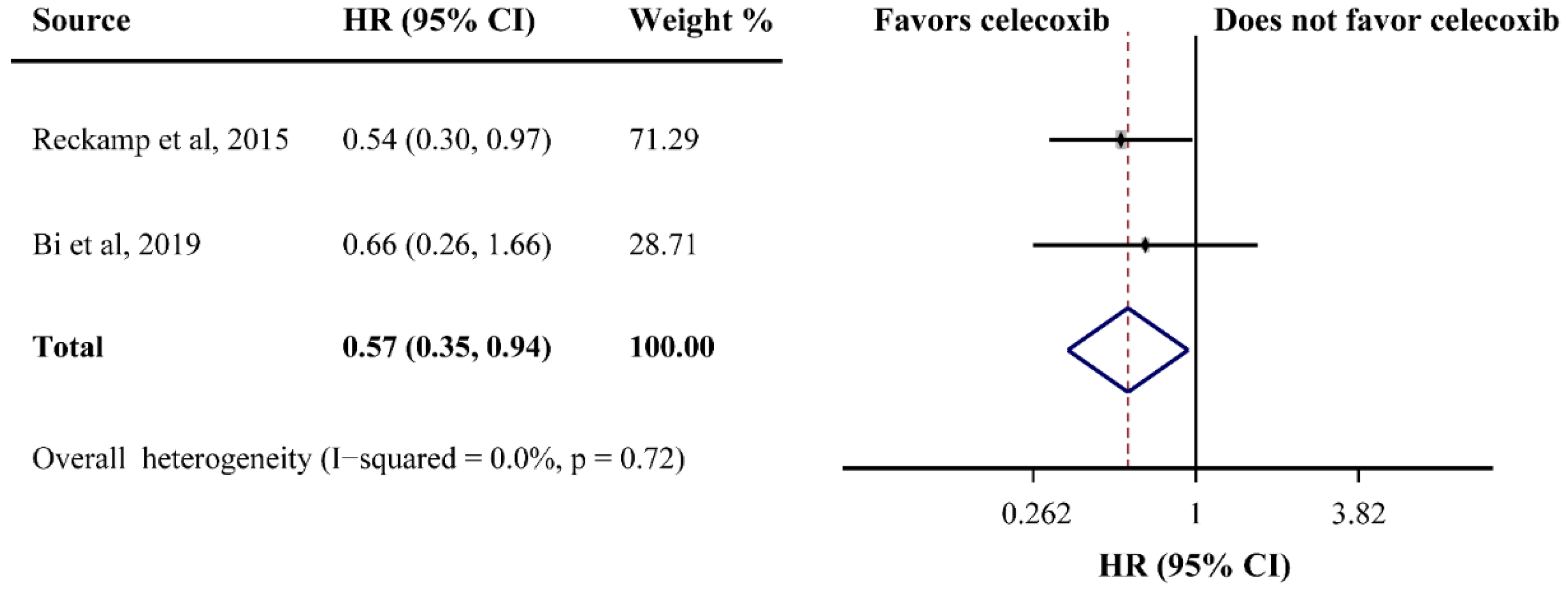
PFS was reported in ten studies and the pooled HR did not indicate a beneficial effect from celecoxib combined with palliative therapy (HR = 1.02, 95%CI = 0.91–1.13), without obvious heterogeneity (I2 = 0.0%) (Figure 2B). Subgroup analyses according to cancer type revealed that celecoxib-combined palliative therapy did not prolong PFS (lung cancer: HR = 0.98, 95%CI = 0.86–1.11; gynecological cancer: HR = 1.17, 95%CI = 0.87–1.57) (Figure 2B). Moreover, celecoxib combined with chemotherapy made no improvement in PFS (HR = 1.02, 95%CI = 0.91–1.15). In addition, celecoxib did not improve PFS when added to first- and ≥ first-line palliative therapies. Likewise, the PFS based on COX-2 status and PGEM status did not significantly differ (high COX-2 status: HR = 1.03, 95%CI = 0.82–1.30; high PGEM status: HR = 0.71, 95%CI = 0.48–1.07; low PGEM status: HR = 1.05, 95%CI = 0.78–1.42). Interestingly, subgroup analysis of EGFR status revealed that EGFR wild-type status was beneficial to PFS (HR = 0.57, 95%CI = 0.35–0.94) (Figure 3). The subgroup analysis results for the PFS of celecoxib-combined palliative therapy are shown in Table 2.
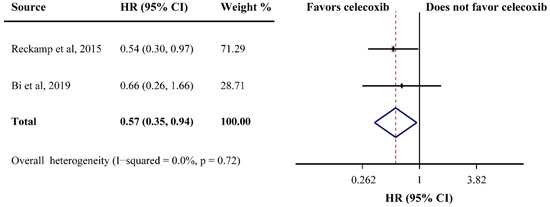
Figure 3.
Efficacy of celecoxib-combined palliative therapy on progression-free survival (PFS) for EGFR wild-type status patients. HR: hazard ratio; CI: confidence interval [48,59].
3.4. Celecoxib and Local Control in Palliative Therapy
In the celecoxib-combined group versus the control group, the ORR was 33.5% versus 27.2%, and the DCR was 71.9% versus 68.9%, respectively. The pooled RRs showed that the addition of celecoxib to palliative therapy made no improvement in the ORR or DCR for patients receiving palliative therapy (ORR:RR = 1.08, 95%CI = 0.96–1.21; DCR:RR = 1.05, 95%CI = 0.99–1.11) (Figure S1A,B). In addition, subgroup analyses based on different cancer types revealed that celecoxib was not beneficial to the local control, no matter which type of cancer (Figure S1A,B). Our subgroup analysis revealed that the local control was not improved when celecoxib was combined with palliative chemotherapy or palliative hormone therapy. Furthermore, analyses according to therapy line revealed similar results. The subgroup analysis results for the local control efficacy of celecoxib combined with palliative therapy are shown in Table S2.
3.5. Celecoxib and Adjuvant Therapy
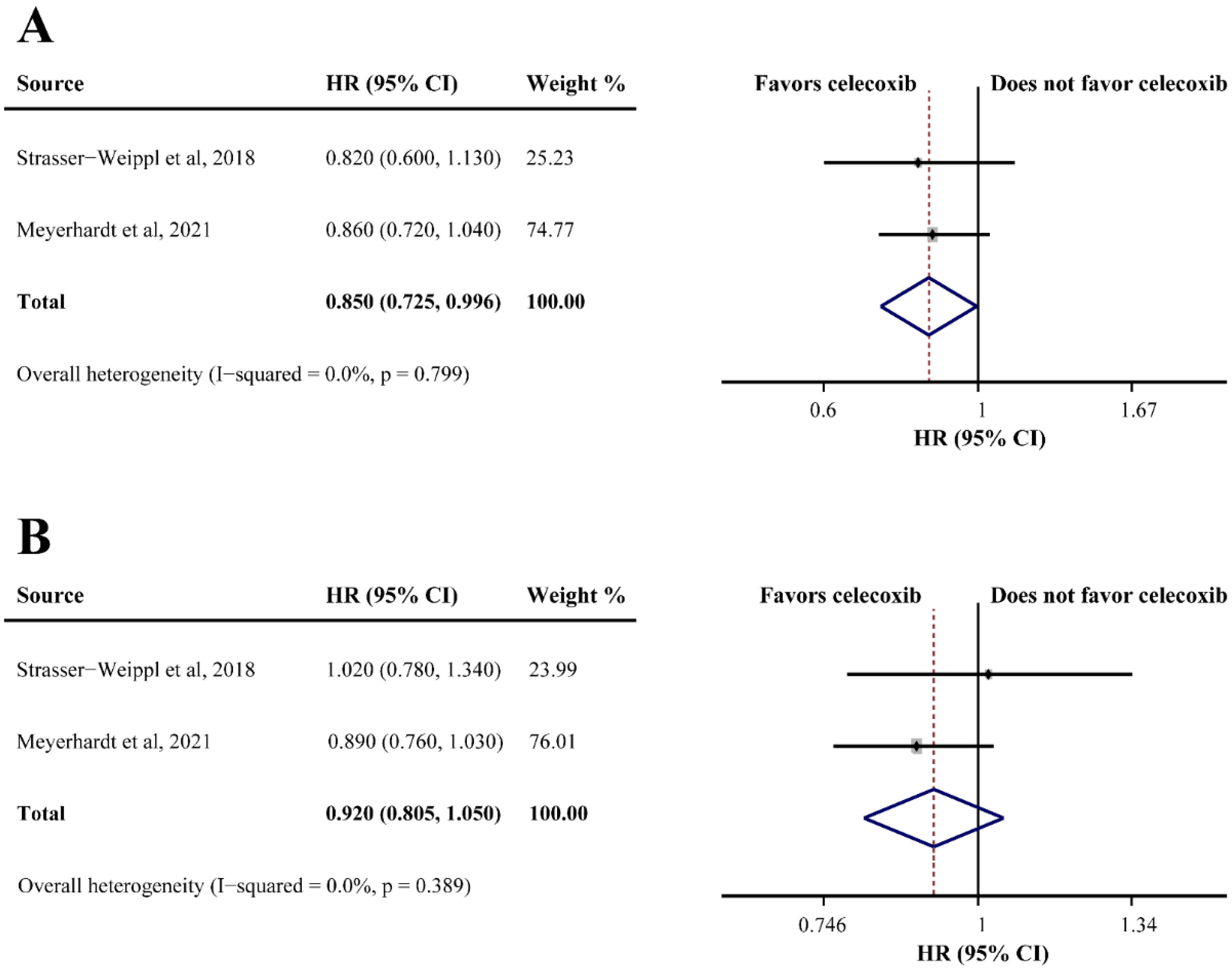
Two studies reported the OS of patients receiving celecoxib-combined adjuvant therapy, and the pooled HR indicated that the addition of celecoxib produced a better OS (HR = 0.850, 95%CI = 0.725–0.996) (Figure 4A). However, celecoxib was found ineffective for DFS (HR = 0.920, 95%CI = 0.805–1.050) (Figure 4B). Non-significant heterogeneities were found in the analyses described above (OS: I2 = 0.0%; DFS: I2 = 0.0%) (Figure 4A,B). One study reported that celecoxib alone was used as maintenance therapy after the completion of adjuvant therapy versus observation without celecoxib in breast cancer, and a significantly improved DFS was observed in the celecoxib group (66% and 41.9%, respectively; HR = 0.17, 95%CI = 0.07–0.40).
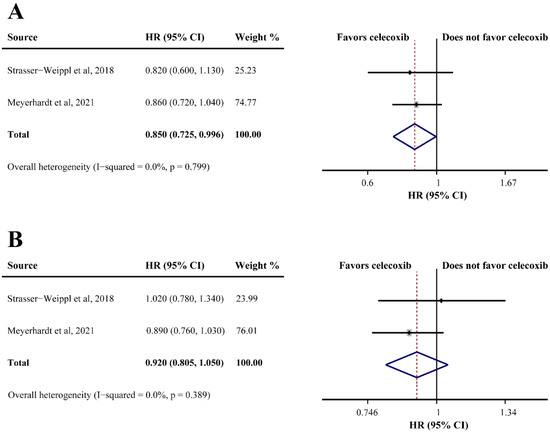
Figure 4.
Efficacy of celecoxib-combined adjuvant therapy on overall survival (OS) (A) and disease-free survival (DFS) (B). HR: hazard ratio; CI: confidence interval [32,47].
3.6. Celecoxib and Neoadjuvant Therapy
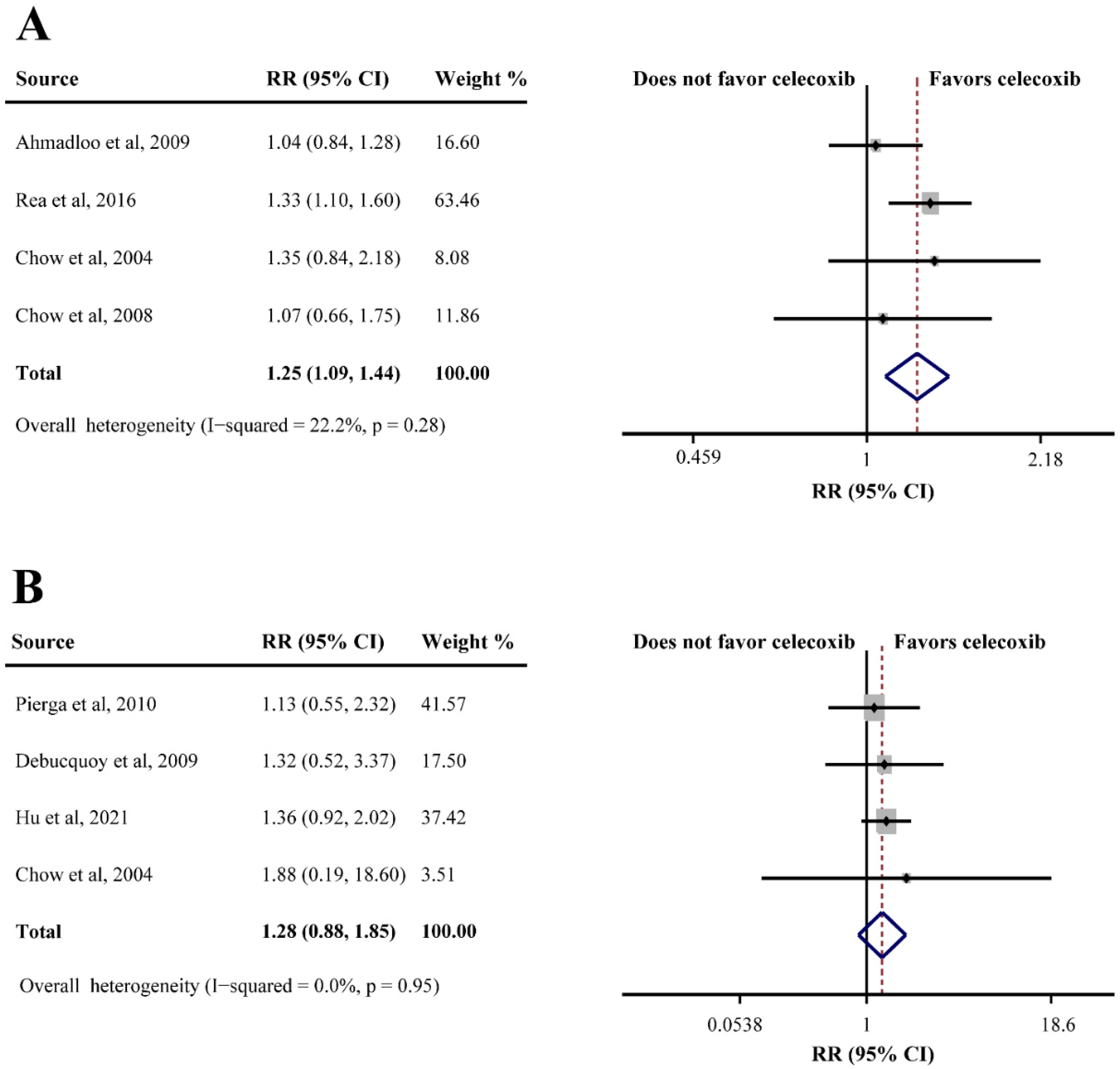
Seven studies consisting of 735 patients evaluated the efficacy of celecoxib plus neoadjuvant therapy (OS, DFS, ORR, and pCR). Only one study reported OS in breast cancer (HR = 1.71, 95%CI = 0.88–3.33), which indicated that celecoxib-combined neoadjuvant therapy had no effect on OS. However, the same study showed that celecoxib combined with neoadjuvant therapy had relative to poor DFS (HR = 1.70, 95%CI = 1.00–2.88). In addition, four studies reporting ORR data found that celecoxib had a beneficial effect when combined with neoadjuvant therapy (75.9% in the celecoxib group compared to 62.5% in the control group; RR = 1.25, 95%CI = 1.09–1.44) (Figure 5A). However, an analysis of four studies based on pCR showed that celecoxib had no significant efficacy in neoadjuvant therapy (RR = 1.28, 95%CI = 0.88–1.85), without substantial heterogeneity (I2 = 0.0%) (Figure 5B).
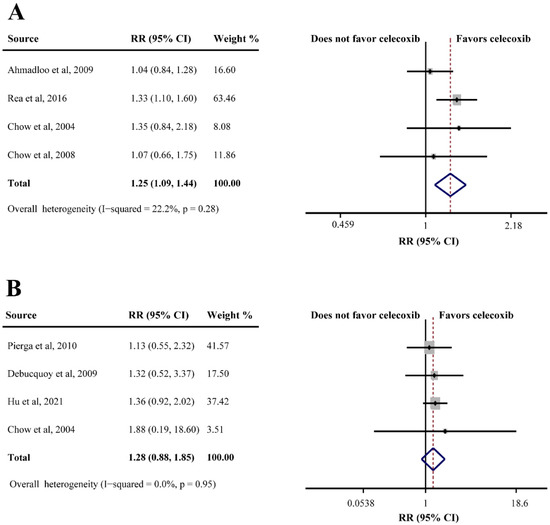
Figure 5.
Efficacy of celecoxib-combined neoadjuvant therapy on objective response rate (ORR) (A) and pathological complete response (pCR) (B). RR: relative risk; CI: confidence interval [30,43,45,53,54,55,68].
3.7. Safety of Celecoxib
Drug safety is an important issue to be taken into consideration for clinical decision-making. Thus, the safety of celecoxib was estimated by analyzing the commonly reported grade ≥ 3 toxicities. Our analysis indicated that celecoxib-combined palliative therapy only increased the incidence of thrombocytopenia (RR = 1.35, 95%CI = 1.08–1.69), and no differences in other common hematological toxicities were observed. Furthermore, our data showed no increase in gastrointestinal toxicities when celecoxib was added to anticancer therapy (overall gastrointestinal toxicities: RR = 1.19, 95%CI = 0.94–1.52), including heartburn/dyspepsia, diarrhea, nausea/vomiting, and constipation. Though celecoxib has been reported to cause CV events, the results in our analyses showed no difference between the celecoxib-combined and control groups, including cardiac ischaemia/infarction, cerebrovascular ischaemia, and thrombosis/thrombus/embolism. Moreover, no increased risk in other common toxicities such as circulatory toxicities, musculoskeletal toxicities, neurotoxicity, fatigue, anorexia, rash, mucositis, infection, and pain was observed after celecoxib therapy (Table S3).
For celecoxib-combined adjuvant therapy, the occurrence rates of grade ≥ 3 AEs for the celecoxib and control groups were 51.7% and 50.8%, respectively Furthermore, for celecoxib-combined neoadjuvant therapy, three RCTs reported no obvious differences in toxicities between the celecoxib and control groups except for skin reaction and increased aspartate aminotransferase levels.
3.8. Publication Bias
Begg’s and Egger’s tests showed no significant publication bias in our meta-analysis (Figures S2–S5, Table S4).
4. Discussion
Cancer remains a life-threatening disease and the second leading cause of death worldwide [69]. As early as 46 years ago, chemoprevention was introduced as an approach to prevent cancer occurrence [70], and researchers gradually discovered that some medicines have additional effects of cancer prevention in the precancer period, including the precancerous lesions [71,72]. Limitations in current anticancer therapeutic strategies highlight the importance of developing more efficient regimens to improve prognosis. As inflammation is closely connected to the development of cancer, COX-2, a proinflammatory enzyme, may be a novel target to improve the therapeutic effect. Recent studies have demonstrated that COX-2 plays an essential role in cancer initiation, progression, invasion, and metastasis [73]. Overexpressed COX-2 was found in several cancer types and promotes the malignant biological behavior of tumor cells though various cytokines and signature pathways [5,8]. COX-2 inhibitors have been found to have cancer prevention and anticancer activities [71,74]. Among them, rofecoxib has been found to suppress the growth of tumor cells [75]. Similarly, valdecoxib exhibited potent growth inhibition and cytotoxic effects against cancer cells [75]. A study has reported that apricoxib plus erlotinib improved the survival of non-small cell lung cancer (NSCLC) patients younger than 65 years old [76]. As a widespread COX-2 inhibitor, celecoxib has cancer preventive effects across primary, secondary, and tertiary prevention [77]. For primary chemoprevention, celecoxib can effectively prevent the occurrence of lung cancer in former smokers [17]. As to second chemoprevention, celecoxib is an effective agent for the prevention of colorectal adenomas and familial adenomatous polyposis [14,77]. In terms of tertiary chemoprevention, also known as the clinical therapy phase, clinical studies reported that celecoxib improved the clinical efficacy of breast cancer, gastric cancer, rectal cancer, colorectal cancer, and head and neck cancer [29,30,46,50,56]. Celecoxib may exert its anticancer effects by reducing the production of COX-2-dependent PGE2 and then inhibiting the COX-2/PGE2/EP2/p-AKT/p-ERK and PGE2/NF-kB pathways [25,26]. However, the evidence remains conflicting in the clinical effects of celecoxib-combined therapy. Here, we aimed to assess the efficacy and safety profile of celecoxib in addition to anticancer therapy, such as palliative, adjuvant, and neoadjuvant therapies. Unlike the previous studies that used the epidemiological information of medication use to explore the prognosis, this study is the first meta-analysis to include RCTs that compared celecoxib-combined therapy and placebo-combined therapy or therapy alone. As far as we know, this is also the first meta-analysis to evaluate the efficacy and safety of celecoxib combined with standard cancer therapy based on different cancer types, concomitant therapy strategies, and therapy stages.
The current meta-analysis consisted of 30 RCTs and included data from 9655 cancer patients. Results showed that there were limited benefits in celecoxib-combined cancer therapy for OS, PFS, DFS, DCR, and pCR, but there was a better ORR (RR = 1.13, 95%CI = 1.03–1.23). Although celecoxib combined with palliative therapy showed no improvement in patient survival and the local control of the tumor, EGFR wild-type patients had a prolonged PFS with celecoxib-combined therapy (HR = 0.57, 95%CI = 0.35–0.94). In addition, celecoxib produced no increase in hematological, gastrointestinal, or other AEs, except for a slight increase in thrombocytopenia (RR = 1.35, 95%CI = 1.08–1.69). In celecoxib-combined adjuvant therapy studies, a better OS was reported in the celecoxib group compared to the control group (HR = 0.850, 95%CI = 0.725–0.996). Interestingly, one study reported that celecoxib alone administered as maintenance therapy to patients with non-metastatic triple-negative breast cancer after adjuvant therapy could prolong DFS (HR = 0.17, 95%CI = 0.07–0.40) [64]. Furthermore, celecoxib improved the ORR for patients receiving celecoxib-combined neoadjuvant therapy for breast cancer (RR = 1.25, 95%CI = 1.09–1.44).
Our meta-analysis indicated limited benefits of OS, PFS, DFS, DCR, and pCR, but a better ORR in celecoxib-combined cancer therapy. Based on the more detailed analyses, there were no survival and local control benefits associated with celecoxib-combined palliative therapy. This insignificant result may be because patients receiving palliative therapy usually had advanced or metastatic cancer, and thus the shorter survival contributed to insufficient intake of celecoxib and a subsequent reduction in its potential anticancer effects [78]. Unlike COX-1 inhibitors which cause gastrointestinal toxicities, the COX-2 inhibitors mainly cause CV toxicities, and the potential risk for CV events needs to be considered during clinical decision-making. Previous studies have reported that celecoxib has the preventive effect of colorectal adenomas, but it cannot be routinely recommended for this indication due to the increased risk of CV events [14]. Fortunately, based on the analyses in this study, there was no significant increase in CV events including cardiac ischaemia/infarction, cerebrovascular ischaemia, and thrombosis/thrombus/embolism. Moreover, in the five studies used to analyze CV toxicity, there was only one that stopped because of a small increased risk of CV events [42]. There is still a need for further studies involving large and representative samples to confirm the celecoxib safety in combined cancer therapy due to the limited data. Further, the pooled results showed no difference between the toxicities of celecoxib and control groups, except for a slightly higher occurrence of thrombocytopenia when celecoxib was combined with palliative therapy. Since high COX-2 expression has been found in bone marrow stromal cells in cancer patients, that the addition of a COX-2 inhibitor delays the recovery of hematopoietic progenitor cells after chemotherapy may be the cause of thrombocytopenia [79,80]. Our findings showed that celecoxib prolonged PFS in patients with EGFR wild-type NSCLC, suggesting that celecoxib-combined palliative therapy is a promising therapy strategy for this population. The EGFR-activated-type patients with advanced NSCLC were reported to benefit from the EGFR tyrosine kinase inhibitor (TKI) therapy [81,82], but the EGFR wild-type patients had no effective targeted therapies other than conventional chemotherapy. Therefore, EGFR may be a potential biomarker to identify the efficacy of celecoxib-combined therapy.
Moreover, though the high level of prostaglandin E2 (PGE2), induced by the overexpressed COX-2, can cause the proliferation of tumor cells, stimulate Bcl-2 protein expression that inhibits apoptosis, and promote the malignant biological behavior of tumors [83], several studies have revealed that COX-2 inhibitors may exert anticarcinogenic effects, not only through COX-2-dependent but also COX-2-independent mechanisms. Moreover, the synthesis of COX-2-dependent PGEM was not the main cause of COX-2 inhibitor efficacy [74]. These findings may explain why celecoxib could not improve the survival and local control of the tumor, independent of COX-2 status or PGEM status. However, further studies are needed to confirm the relationship between celecoxib-combined cancer therapy and the status of COX-2 and PGEM, due to the limited data from the relevant studies. Moreover, early use of celecoxib may be beneficial, since a better OS was observed in celecoxib-combined adjuvant therapy, and a better ORR was reported in celecoxib-combined neoadjuvant therapy. Clinicians should carefully weigh the clinical benefits against the potential increase in CV events during clinical decision-making. In addition, the better ORR in neoadjuvant therapy was obtained by pooling data from breast cancer studies, as breast cancer generally has high long-term survival rates. Furthermore, the development of bone marrow micro-metastasis and subsequent osteolytic bone metastases in breast cancer patients may be due to the overexpression of COX-2 in primary breast cancer cells. Since studies have shown that the COX-2 inhibitor, celecoxib, inhibited this metastatic process [84], these findings may explain why the celecoxib-combined neoadjuvant therapy is beneficial for breast cancer patients.
Our meta-analysis study has many strengths. Firstly, the present study included all relevant RCTs, and therefore the heterogeneities were found to be low. Secondly, we systematically evaluated all kinds of tumors, and we enhanced the stability and reliability of our results and conclusions by an in-depth subgroup analysis based on different therapy stages, cancer types, and more detailed subgroups. Thirdly, the addition of celecoxib to adjuvant therapy prolonged OS.
The present study also had several limitations. Firstly, subgroup analyses were not performed on detailed therapeutic regimens and celecoxib dosage due to limited data, which may influence the evaluation of the efficacy of celecoxib. Secondly, we made the subgroup according to cancer molecular type, but the limited data from enrolled studies only supported the analyses based on COX-2, EGFR, and PEGM status, and we found EGFR wild-type patients obtain a prolonged PFS in celecoxib-combined palliative therapy. Therefore, further studies are needed to explore the efficacy of celecoxib in cancer patients with other molecular types based on different cancers. Thirdly, we were unable to obtain detailed individual data since this study was based on previously published data from the enrolled RCTs. Thus, we could not perform an in-depth subgroup analysis according to prognosis-related factors such as age, sex, ethnic group, and TNM stage. In addition, the efficacy of celecoxib may have been underestimated since we could not completely control these inherently confounding factors in the included studies.
5. Conclusions
The addition of celecoxib to palliative therapy cannot improve survival and local control rates except for the PFS of EGFR wild-type patients, without obvious toxicities and AEs. In terms of adjuvant therapy, the addition of celecoxib can prolong the OS but not the DFS. Moreover, the combination of celecoxib with neoadjuvant therapy can improve the ORR for breast cancer. In general, further studies evaluating the celecoxib efficacy on cancer therapy should be conducted with caution in certain populations to reduce high clinical trial costs and use of medical recourses.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/curroncol29090482/s1. Figure S1: local control efficacy of celecoxib-combined palliative therapy in different cancer types on objective response rate (ORR) (A) and disease control rate (DCR) (B); RR: relative risk; CI: confidence interval. Figure S2: Begg’s funnel plot of publication bias analysis in celecoxib-combined standard cancer therapy on overall survival (OS) (A), progression-free survival (PFS) (B), disease-free survival (DFS) (C), objective response rate (ORR) (D), disease control rate (DCR) (E), and pathological complete response (pCR) (F). Figure S3: Begg’s funnel plot of publication bias analysis in celecoxib-combined palliative therapy on overall survival (OS) (A), progression-free survival (PFS) (B), objective response rate (ORR) (C), and disease control rate (DCR) (D). Figure S4: Begg’s funnel plot of publication bias analysis in celecoxib-combined adjuvant therapy on overall survival (OS) (A) and disease-free survival (DFS) (B). Figure S5: Begg’s funnel plot of publication bias analysis in celecoxib-combined neoadjuvant therapy on objective response rate (ORR) (A) and pathological complete response (pCR) (B). Table S1: the baseline patient characteristics of the included RCTs. Table S2: the subgroup analysis results for the local control efficacy of celecoxib-combined palliative therapy. Table S3: the analysis results of toxicities in celecoxib combined with palliative therapy for cancer patients. Table S4: the analysis results of publication bias in celecoxib combined with standard cancer therapy.
Author Contributions
Conceptualization, S.-Y.Y., J.-Y.L. and X.-Z.H.; methodology, S.-Y.Y., T.-H.L. and X.-Z.H.; software, S.-Y.Y., J.-Y.L. and X.-W.C.; validation, S.-Y.Y., J.-X.S. and J.-H.Z.; formal analysis, S.-Y.Y., J.-Y.L. and Z.-H.W.; investigation, S.-Y.Y. and Y.L.; resources, S.-Y.Y., Y.-X.S. and P.G.; data curation, S.-Y.Y.; writing—original draft preparation, S.-Y.Y.; writing—review and editing, S.-Y.Y., J.-Y.L. and X.-Z.H.; visualization, S.-Y.Y. and X.-W.C.; supervision, X.-Z.H.; project administration, P.G. and X.-Z.H.; funding acquisition, P.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China, grant number 82002599.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
COX-2: cyclooxygenase-2; RCTs: randomized controlled trials; OS: overall survival; PFS: progression-free survival; DFS: disease-free survival; ORR: objective response rate; DCR: disease control rate; pCR: pathological complete response; AEs: adverse events; EGFR: epidermal growth factor receptor; CIs: confidence intervals; HRs: hazard ratios; RRs: the relative risks; CR: complete response; PR: partial response; SD: stable disease; PD: progressive disease; CV: cardiovascular; RECIST: Response Evaluation Criteria in Solid Tumors; PGEM: the urinary metabolite of prostaglandin E2; NSAIDs: nonsteroidal anti-inflammatory drugs; WHO: World Health Organization; NSCLC: non-small cell lung cancer; TKI: tyrosine kinase inhibitor.
References
- Bray, F.; Laversanne, M.; Weiderpass, E.; Soerjomataram, I. The ever-increasing importance of cancer as a leading cause of premature death worldwide. Cancer 2021, 127, 3029–3030. [Google Scholar] [CrossRef] [PubMed]
- Krysan, K.; Cui, X.; Gardner, B.K.; Reckamp, K.L.; Wang, X.; Hong, L.; Walser, T.C.; Rodriguez, N.L.; Pagano, P.C.; Garon, E.B.; et al. Elevated neutrophil gelatinase-associated lipocalin contributes to erlotinib resistance in non-small cell lung cancer. Am. J. Transl. Res. 2013, 5, 481–496. [Google Scholar] [PubMed]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Dannenberg, A.J.; Lippman, S.M.; Mann, J.R.; Subbaramaiah, K.; DuBois, R.N. Cyclooxygenase-2 and epidermal growth factor receptor: Pharmacologic targets for chemoprevention. J. Clin. Oncol. 2005, 23, 254–266. [Google Scholar] [CrossRef]
- Smith, W.L.; Langenbach, R. Why there are two cyclooxygenase isozymes. J. Clin. Investig. 2001, 107, 1491–1495. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, X.F. Overexpression of cyclooxygenase-2 correlates with advanced stages of colorectal cancer. Am. J. Gastroenterol. 2002, 97, 1037–1041. [Google Scholar] [CrossRef]
- Ristimäki, A.; Sivula, A.; Lundin, J.; Lundin, M.; Salminen, T.; Haglund, C.; Joensuu, H.; Isola, J. Prognostic significance of elevated cyclooxygenase-2 expression in breast cancer. Cancer Res. 2002, 62, 632–635. [Google Scholar]
- Saba, N.F.; Choi, M.; Muller, S.; Shin, H.J.; Tighiouart, M.; Papadimitrakopoulou, V.A.; El-Naggar, A.K.; Khuri, F.R.; Chen, Z.G.; Shin, D.M. Role of cyclooxygenase-2 in tumor progression and survival of head and neck squamous cell carcinoma. Cancer Prev. Res. 2009, 2, 823–829. [Google Scholar] [CrossRef]
- Masferrer, J.L.; Leahy, K.M.; Koki, A.T.; Zweifel, B.S.; Settle, S.L.; Woerner, B.M.; Edwards, D.A.; Flickinger, A.G.; Moore, R.J.; Seibert, K. Antiangiogenic and antitumor activities of cyclooxygenase-2 inhibitors. Cancer Res. 2000, 60, 1306–1311. [Google Scholar]
- Belton, O.; Byrne, D.; Kearney, D.; Leahy, A.; Fitzgerald, D.J. Cyclooxygenase-1 and -2-dependent prostacyclin formation in patients with atherosclerosis. Circulation 2000, 102, 840–845. [Google Scholar] [CrossRef]
- Mukherjee, D.; Nissen, S.E.; Topol, E.J. Risk of cardiovascular events associated with selective COX-2 inhibitors. JAMA 2001, 286, 954–959. [Google Scholar] [CrossRef] [PubMed]
- El-Malah, A.A.; Gineinah, M.M.; Deb, P.K.; Khayyat, A.N.; Bansal, M.; Venugopala, K.N.; Aljahdali, A.S. Selective COX-2 Inhibitors: Road from Success to Controversy and the Quest for Repurposing. Pharmaceuticals 2022, 15, 827. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.A.; Ashbeck, E.L.; Roe, D.J.; Fales, L.; Buckmeier, J.; Wang, F.; Bhattacharyya, A.; Hsu, C.H.; Chow, S.H.; Ahnen, D.J.; et al. Celecoxib for the Prevention of Colorectal Adenomas: Results of a Suspended Randomized Controlled Trial. J. Natl. Cancer Inst. 2016, 108, djw151. [Google Scholar] [CrossRef] [PubMed]
- Bertagnolli, M.M.; Eagle, C.J.; Zauber, A.G.; Redston, M.; Solomon, S.D.; Kim, K.; Tang, J.; Rosenstein, R.B.; Wittes, J.; Corle, D.; et al. Celecoxib for the prevention of sporadic colorectal adenomas. N. Engl. J. Med. 2006, 355, 873–884. [Google Scholar] [CrossRef]
- Flamiatos, J.F.; Beer, T.M.; Graff, J.N.; Eilers, K.M.; Tian, W.; Sekhon, H.S.; Garzotto, M. Cyclooxygenase-2 (COX-2) inhibition for prostate cancer chemoprevention: Double-blind randomised study of pre-prostatectomy celecoxib or placebo. BJU Int. 2017, 119, 709–716. [Google Scholar] [CrossRef]
- Thompson, P.A.; Martinez, J.A. The Importance of Drug Concentration at the Site of Action: Celecoxib and Colon Polyp Prevention as a Case Study. Cancer Prev. Res. 2022, 15, 205–208. [Google Scholar] [CrossRef]
- Mao, J.T.; Roth, M.D.; Fishbein, M.C.; Aberle, D.R.; Zhang, Z.F.; Rao, J.Y.; Tashkin, D.P.; Goodglick, L.; Holmes, E.C.; Cameron, R.B.; et al. Lung cancer chemoprevention with celecoxib in former smokers. Cancer Prev. Res. 2011, 4, 984–993. [Google Scholar] [CrossRef]
- Tudor, D.V.; Baldea, I.; Olteanu, D.E.; Fischer-Fodor, E.; Piroska, V.; Lupu, M.; Calinici, T.; Decea, R.M.; Filip, G.A. Celecoxib as a Valuable Adjuvant in Cutaneous Melanoma Treated with Trametinib. Int. J. Mol. Sci. 2021, 22, 4387. [Google Scholar] [CrossRef]
- Toloczko-Iwaniuk, N.; Dziemianczyk-Pakiela, D.; Nowaszewska, B.K.; Celinska-Janowicz, K.; Miltyk, W. Celecoxib in Cancer Therapy and Prevention—Review. Curr. Drug Targets 2019, 20, 302–315. [Google Scholar] [CrossRef]
- Hsu, A.L.; Ching, T.T.; Wang, D.S.; Song, X.; Rangnekar, V.M.; Chen, C.S. The cyclooxygenase-2 inhibitor celecoxib induces apoptosis by blocking Akt activation in human prostate cancer cells independently of Bcl-2. J. Biol. Chem. 2000, 275, 11397–11403. [Google Scholar] [CrossRef]
- Lin, E.; Morris, J.S.; Ayers, G.D. Effect of celecoxib on capecitabine-induced hand-foot syndrome and antitumor activity. Oncology 2002, 16, 31–37. [Google Scholar] [PubMed]
- Shan, B.Z.; Guo, B.; Li, Y.S.; Sun, X.F. Effect of celecoxib on protein expression of FAK and Cx43 in DMBA induced rat tongue carcinoma cells. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 9454–9463. [Google Scholar] [CrossRef] [PubMed]
- Medina, M.V.; D’Agostino, A.; Ma, Q.; Eroles, P.; Cavallin, L.; Chiozzini, C.; Sapochnik, D.; Cymeryng, C.; Hyjek, E.; Cesarman, E.; et al. KSHV G-protein coupled receptor vGPCR oncogenic signaling upregulation of Cyclooxygenase-2 expression mediates angiogenesis and tumorigenesis in Kaposi’s sarcoma. PLoS Pathog. 2020, 16, e1009006. [Google Scholar] [CrossRef]
- Yoshida, H.; Yoshimura, H.; Matsuda, S.; Yamamoto, S.; Ohmori, M.; Ohta, K.; Ryoke, T.; Itoi, H.; Kiyoshima, T.; Kobayashi, M.; et al. Celecoxib suppresses lipopolysaccharide-stimulated oral squamous cell carcinoma proliferation in vitro and in vivo. Oncol. Lett. 2019, 18, 5793–5800. [Google Scholar] [CrossRef]
- Zhang, X.; Yan, K.; Deng, L.; Liang, J.; Liang, H.; Feng, D.; Ling, B. Cyclooxygenase 2 Promotes Proliferation and Invasion in Ovarian Cancer Cells via the PGE2/NF-kappaB Pathway. Cell Transplant. 2019, 28, 1S–13S. [Google Scholar] [CrossRef]
- Tai, Y.; Zhang, L.H.; Gao, J.H.; Zhao, C.; Tong, H.; Ye, C.; Huang, Z.Y.; Liu, R.; Tang, C.W. Suppressing growth and invasion of human hepatocellular carcinoma cells by celecoxib through inhibition of cyclooxygenase-2. Cancer Manag. Res. 2019, 11, 2831–2848. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.X.; Wu, X.J.; Wan, D.S.; Lu, Z.H.; Kong, L.H.; Pan, Z.Z.; Chen, G. Celecoxib can prevent capecitabine-related hand-foot syndrome in stage II and III colorectal cancer patients: Result of a single-center, prospective randomized phase III trial. Ann. Oncol. 2012, 23, 1348–1353. [Google Scholar] [CrossRef]
- Suddek, G.M.; El-Kenawi, A.E.; Abdel-Aziz, A.; El-Kashef, H.A. Celecoxib, a selective cyclooxygenase-2 inhibitor, attenuates renal injury in a rat model of Cisplatin-induced nephrotoxicity. Chemotherapy 2011, 57, 321–326. [Google Scholar] [CrossRef]
- Falandry, C.; Debled, M.; Bachelot, T.; Delozier, T.; Crétin, J.; Romestaing, P.; Mille, D.; You, B.; Mauriac, L.; Pujade-Lauraine, E.; et al. Celecoxib and exemestane versus placebo and exemestane in postmenopausal metastatic breast cancer patients: A double-blind phase III GINECO study. Breast Cancer Res. Treat. 2009, 116, 501–508. [Google Scholar] [CrossRef]
- Debucquoy, A.; Roels, S.; Goethals, L.; Libbrecht, L.; Cutsem, E.V.; Geboes, K.; Penninckx, F.; D’Hoore, A.; McBride, W.H.; Haustermans, K. Double blind randomized phase II study with radiation + 5-fluorouracil ± celecoxib for resectable rectal cancer. Radiother. Oncol. 2009, 93, 273–278. [Google Scholar] [CrossRef]
- Koch, A.; Bergman, B.; Holmberg, E.; Sederholm, C.; Ek, L.; Kosieradzki, J.; Lamberg, K.; Thaning, L.; Ydreborg, S.O.; Sörenson, S. Effect of celecoxib on survival in patients with advanced non-small cell lung cancer: A double blind randomised clinical phase III trial (CYCLUS study) by the Swedish Lung Cancer Study Group. Eur. J. Cancer 2011, 47, 1546–1555. [Google Scholar] [CrossRef] [PubMed]
- Strasser-Weippl, K.; Higgins, M.J.; Chapman, J.W.; Ingle, J.N.; Sledge, G.W.; Budd, G.T.; Ellis, M.J.; Pritchard, K.I.; Clemons, M.J.; Badovinac-Crnjevic, T.; et al. Effects of Celecoxib and Low-dose Aspirin on Outcomes in Adjuvant Aromatase Inhibitor-Treated Patients: CCTG MA.27. J. Natl. Cancer Inst. 2018, 110, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Maiello, E.; Giuliani, F.; Gebbia, V.; Di Renzo, N.; Pezzella, G.; Romito, S.; Mallamaci, R.; Lopez, M.; Colucci, G. FOLFIRI with or without celecoxib in advanced colorectal cancer: A randomized phase II study of the Gruppo Oncologico dell’Italia Meridionale (GOIM). Ann. Oncol. 2006, 17 (Suppl. 7), 55–59. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control. Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Tierney, J.F.; Stewart, L.A.; Ghersi, D.; Burdett, S.; Sydes, M.R. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials 2007, 8, 16. [Google Scholar] [CrossRef]
- Therasse, P.; Arbuck, S.G.; Eisenhauer, E.A.; Wanders, J.; Kaplan, R.S.; Rubinstein, L.; Verweij, J.; Van Glabbeke, M.; van Oosterom, A.T.; Christian, M.C.; et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J. Natl. Cancer Inst. 2000, 92, 205–216. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Begg, C.B.; Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994, 50, 1088–1101. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Mason, M.D.; Clarke, N.W.; James, N.D.; Dearnaley, D.P.; Spears, M.R.; Ritchie, A.W.S.; Attard, G.; Cross, W.; Jones, R.J.; Parker, C.C.; et al. Adding Celecoxib With or Without Zoledronic Acid for Hormone-Naïve Prostate Cancer: Long-Term Survival Results From an Adaptive, Multiarm, Multistage, Platform, Randomized Controlled Trial. J. Clin. Oncol. 2017, 35, 1530–1541. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.D.; Tan, W.S.; Porta, N.; Mostafid, H.; Huddart, R.; Protheroe, A.; Bogle, R.; Blazeby, J.; Palmer, A.; Cresswell, J.; et al. BOXIT—A Randomised Phase III Placebo-controlled Trial Evaluating the Addition of Celecoxib to Standard Treatment of Transitional Cell Carcinoma of the Bladder (CRUK/07/004). Eur. Urol. 2019, 75, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Chow, L.W.; Yip, A.Y.; Loo, W.T.; Lam, C.K.; Toi, M. Celecoxib anti-aromatase neoadjuvant (CAAN) trial for locally advanced breast cancer. J. Steroid Biochem. Mol. Biol. 2008, 111, 13–17. [Google Scholar] [CrossRef]
- Hamy, A.S.; Tury, S.; Wang, X.; Gao, J.; Pierga, J.Y.; Giacchetti, S.; Brain, E.; Pistilli, B.; Marty, M.; Espié, M.; et al. Celecoxib With Neoadjuvant Chemotherapy for Breast Cancer Might Worsen Outcomes Differentially by COX-2 Expression and ER Status: Exploratory Analysis of the REMAGUS02 Trial. J. Clin. Oncol. 2019, 37, 624–635. [Google Scholar] [CrossRef] [PubMed]
- Ahmadloo, N.; Nazer Mozaffari, M.A.; Mohammadianpanah, M.; Omidvari, S.H.; Mosalaei, A.; Mosleh Shirazi, M.A. Combined neoadjuvant chemotherapy and celecoxib in locally advanced breast cancer. Iran. Red Crescent Med. J. 2009, 11, 419–424. [Google Scholar]
- Guo, Q.; Li, Q.; Wang, J.; Liu, M.; Wang, Y.; Chen, Z.; Ye, Y.; Guan, Q.; Zhou, Y. A comprehensive evaluation of clinical efficacy and safety of celecoxib in combination with chemotherapy in metastatic or postoperative recurrent gastric cancer patients: A preliminary, three-center, clinical trial study. Medicine 2019, 98, e16234. [Google Scholar] [CrossRef]
- Meyerhardt, J.A.; Shi, Q.; Fuchs, C.S.; Meyer, J.; Niedzwiecki, D.; Zemla, T.; Kumthekar, P.; Guthrie, K.A.; Couture, F.; Kuebler, P.; et al. Effect of Celecoxib vs Placebo Added to Standard Adjuvant Therapy on Disease-Free Survival among Patients with Stage III Colon Cancer: The CALGB/SWOG 80702 (Alliance) Randomized Clinical Trial. JAMA 2021, 325, 1277–1286. [Google Scholar] [CrossRef]
- Bi, N.; Liang, J.; Zhou, Z.; Chen, D.; Fu, Z.; Yang, X.; Feng, Q.; Hui, Z.; Xiao, Z.; Lv, J.; et al. Effect of Concurrent Chemoradiation With Celecoxib vs Concurrent Chemoradiation Alone on Survival Among Patients With Non-Small Cell Lung Cancer With and Without Cyclooxygenase 2 Genetic Variants: A Phase 2 Randomized Clinical Trial. JAMA Netw. Open 2019, 2, e1918070. [Google Scholar] [CrossRef]
- Mohammadianpanah, M.; Razmjou-Ghalaei, S.; Shafizad, A.; Ashouri-Taziani, Y.; Khademi, B.; Ahmadloo, N.; Ansari, M.; Omidvari, S.; Mosalaei, A.; Mosleh-Shirazi, M.A. Efficacy and safety of concurrent chemoradiation with weekly cisplatin ± low-dose celecoxib in locally advanced undifferentiated nasopharyngeal carcinoma: A phase II-III clinical trial. J. Cancer Res. Ther. 2011, 7, 442–447. [Google Scholar] [CrossRef]
- Aghili, M.; Ghalehtaki, R.; Rayzan, E.; Farzin, M.; Mojahed, M.M.; Izadi, S.; Kazemian, A. The efficacy of celecoxib during chemoradiation in locally advanced head and neck carcinoma; a phase 2 randomized placebo control clinical trial. Int. J. Cancer Manag. 2021, 14, e103653. [Google Scholar] [CrossRef]
- Edelman, M.J.; Watson, D.; Wang, X.; Morrison, C.; Kratzke, R.A.; Jewell, S.; Hodgson, L.; Mauer, A.M.; Gajra, A.; Masters, G.A.; et al. Eicosanoid modulation in advanced lung cancer: Cyclooxygenase-2 expression is a positive predictive factor for celecoxib + chemotherapy—Cancer and leukemia group B trial 30203. J. Clin. Oncol. 2008, 26, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Köhne, C.H.; De Greve, J.; Hartmann, J.T.; Lang, I.; Vergauwe, P.; Becker, K.; Braumann, D.; Joosens, E.; Müller, L.; Janssens, J.; et al. Irinotecan combined with infusional 5-fluorouracil/folinic acid or capecitabine plus celecoxib or placebo in the first-line treatment of patients with metastatic colorectal cancer. EORTC study 40015. Ann. Oncol. 2008, 19, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Rea, D.; Francis, A.; Poole, C.; Brookes, C.; Stein, R.; Bartlett, J.; Dunn, J.; Canney, P.; Sutton, R.; Daoud, R.; et al. NEO-EXCEL phase III neoadjuvant trial of pre-operative exemestane or letrozole +/− celecoxib in the treatment of ER positive postmenopausal early breast cancer. Cancer Res. 2016, 76, PD2–02. [Google Scholar] [CrossRef]
- Hu, H.; Kang, L.; Zhang, J.; Wu, Z.; Wang, H.; Huang, M.; Lan, P.; Wu, X.; Wang, C.; Cao, W.; et al. Neoadjuvant PD-1 blockade with toripalimab, with or without celecoxib, in mismatch repair-deficient or microsatellite instability-high, locally advanced, colorectal cancer (PICC): A single-centre, parallel-group, non-comparative, randomised, phase 2 trial. Lancet Gastroenterol. Hepatol. 2022, 7, 38–48. [Google Scholar] [CrossRef]
- Chow, L.W.; Toi, M.; Takebayashi, Y. Neoadjuvant use of 5-fluorouracil, epirubicin and cyclophosphamide with and without celecoxib for locally advanced breast cancer: A phase II study correlating response to celecoxib with high levels of COX-2 gene expression in the tumor. J. Clin. Oncol. 2004, 22, 9603. [Google Scholar] [CrossRef]
- Jin, C.H.; Wang, A.H.; Chen, J.M.; Li, R.X.; Liu, X.M.; Wang, G.P.; Xing, L.Q. Observation of curative efficacy and prognosis following combination chemotherapy with celecoxib in the treatment of advanced colorectal cancer. J. Int. Med. Res. 2011, 39, 2129–2140. [Google Scholar] [CrossRef]
- Zhou, S.; Zhou, C.; Xu, J.; Lv, M. P2-327: First-line chemotherapy of vinorelbine/cisplatin (NP) combined with cyclooxygenase-2 inhibitor celecoxib in advanced non-small-cell lung cancer (NSCLC). J. Thorac. Oncol. 2007, 2, S702–S703. [Google Scholar] [CrossRef]
- Edelman, M.J.; Wang, X.; Hodgson, L.; Cheney, R.T.; Baggstrom, M.Q.; Thomas, S.P.; Gajra, A.; Bertino, E.; Reckamp, K.L.; Molina, J.; et al. Phase III Randomized, Placebo-Controlled, Double-Blind Trial of Celecoxib in Addition to Standard Chemotherapy for Advanced Non-Small-Cell Lung Cancer With Cyclooxygenase-2 Overexpression: CALGB 30801 (Alliance). J. Clin. Oncol. 2017, 35, 2184–2192. [Google Scholar] [CrossRef]
- Reckamp, K.L.; Koczywas, M.; Cristea, M.C.; Dowell, J.E.; Wang, H.J.; Gardner, B.K.; Milne, G.L.; Figlin, R.A.; Fishbein, M.C.; Elashoff, R.M.; et al. Randomized phase 2 trial of erlotinib in combination with high-dose celecoxib or placebo in patients with advanced non-small cell lung cancer. Cancer 2015, 121, 3298–3306. [Google Scholar] [CrossRef]
- Reyners, A.K.; de Munck, L.; Erdkamp, F.L.; Smit, W.M.; Hoekman, K.; Lalisang, R.I.; de Graaf, H.; Wymenga, A.N.; Polee, M.; Hollema, H.; et al. A randomized phase II study investigating the addition of the specific COX-2 inhibitor celecoxib to docetaxel plus carboplatin as first-line chemotherapy for stage IC to IV epithelial ovarian cancer, Fallopian tube or primary peritoneal carcinomas: The DoCaCel study. Ann. Oncol. 2012, 23, 2896–2902. [Google Scholar] [CrossRef]
- Lilenbaum, R.; Socinski, M.A.; Altorki, N.K.; Hart, L.L.; Keresztes, R.S.; Hariharan, S.; Morrison, M.E.; Fayyad, R.; Bonomi, P. Randomized phase II trial of docetaxel/irinotecan and gemcitabine/irinotecan with or without celecoxib in the second-line treatment of non-small-cell lung cancer. J. Clin. Oncol. 2006, 24, 4825–4832. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Cristea, M.; Frankel, P.; Ruel, C.; Chen, C.; Wang, Y.; Morgan, R.; Leong, L.; Chow, W.; Koczywas, M.; et al. Randomized trial of oral cyclophosphamide versus oral cyclophosphamide with celecoxib for recurrent epithelial ovarian, fallopian tube, and primary peritoneal cancer. Cancer Treat. Res. Commun. 2019, 21, 100155. [Google Scholar] [CrossRef] [PubMed]
- Groen, H.J.; Sietsma, H.; Vincent, A.; Hochstenbag, M.M.; van Putten, J.W.; van den Berg, A.; Dalesio, O.; Biesma, B.; Smit, H.J.; Termeer, A.; et al. Randomized, placebo-controlled phase III study of docetaxel plus carboplatin with celecoxib and cyclooxygenase-2 expression as a biomarker for patients with advanced non-small-cell lung cancer: The NVALT-4 study. J. Clin. Oncol. 2011, 29, 4320–4326. [Google Scholar] [CrossRef] [PubMed]
- Gharib, F.; Zamzam, Y.; Sad, L.M. Role of COX-2 inhibitors as maintenance therapy in non-metastatic triple negative breast cancer Egyptian patients, single institution study. Oncol. Radiother. 2020, 1, 1–6. [Google Scholar]
- Dirix, L.Y.; Ignacio, J.; Nag, S.; Bapsy, P.; Gomez, H.; Raghunadharao, D.; Paridaens, R.; Jones, S.; Falcon, S.; Carpentieri, M.; et al. Treatment of advanced hormone-sensitive breast cancer in postmenopausal women with exemestane alone or in combination with celecoxib. J. Clin. Oncol. 2008, 26, 1253–1259. [Google Scholar] [CrossRef]
- Edelman, M.J.; Wang, X.; Hodgson, L.; Cheney, R.T.; Baggstrom, M.; Sachdev, T.; Gajra, A.; Bertino, E.; Reckamp, K.; Molina, J.; et al. Phase III randomized, placebo controlled trial of COX-2 inhibition in addition to standard chemotherapy for advanced non-small cell lung cancer (NSCLC):CALGB 30801 (Alliance). Cancer Res. 2014, 74, CT238. [Google Scholar] [CrossRef]
- Gulyas, M.; Mattsson, J.S.M.; Lindgren, A.; Ek, L.; Lamberg Lundström, K.; Behndig, A.; Holmberg, E.; Micke, P.; Bergman, B. COX-2 expression and effects of celecoxib in addition to standard chemotherapy in advanced non-small cell lung cancer. Acta Oncol. 2018, 57, 244–250. [Google Scholar] [CrossRef]
- Pierga, J.Y.; Delaloge, S.; Espié, M.; Brain, E.; Sigal-Zafrani, B.; Mathieu, M.C.; Bertheau, P.; Guinebretière, J.M.; Spielmann, M.; Savignoni, A.; et al. A multicenter randomized phase II study of sequential epirubicin/cyclophosphamide followed by docetaxel with or without celecoxib or trastuzumab according to HER2 status, as primary chemotherapy for localized invasive breast cancer patients. Breast Cancer Res. Treat. 2010, 122, 429–437. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Sporn, M.B. Approaches to prevention of epithelial cancer during the preneoplastic period. Cancer Res. 1976, 36, 2699–2702. [Google Scholar] [PubMed]
- Sporn, M.B.; Suh, N. Chemoprevention: An essential approach to controlling cancer. Nat. Rev. Cancer 2002, 2, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Brenner, H.; Chen, C. The colorectal cancer epidemic: Challenges and opportunities for primary, secondary and tertiary prevention. Br. J. Cancer 2018, 119, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Sobolewski, C.; Cerella, C.; Dicato, M.; Ghibelli, L.; Diederich, M. The role of cyclooxygenase-2 in cell proliferation and cell death in human malignancies. Int. J. Cell Biol. 2010, 2010, 215158. [Google Scholar] [CrossRef] [PubMed]
- Grösch, S.; Maier, T.J.; Schiffmann, S.; Geisslinger, G. Cyclooxygenase-2 (COX-2)-independent anticarcinogenic effects of selective COX-2 inhibitors. J. Natl. Cancer Inst. 2006, 98, 736–747. [Google Scholar] [CrossRef]
- Jeyamogan, S.; Khan, N.A.; Siddiqui, R. Antitumour Activities of Selected Pure Compounds Identified from the Serum of Crocodylus porosus, Malayopython reticulatus, Varanus salvator and Cuora kamaroma amboinensis. Asian Pac. J. Cancer Prev. 2021, 22, 97–106. [Google Scholar] [CrossRef]
- Gitlitz, B.J.; Bernstein, E.; Santos, E.S.; Otterson, G.A.; Milne, G.; Syto, M.; Burrows, F.; Zaknoen, S. A randomized, placebo-controlled, multicenter, biomarker-selected, phase 2 study of apricoxib in combination with erlotinib in patients with advanced non-small-cell lung cancer. J. Thorac. Oncol. 2014, 9, 577–582. [Google Scholar] [CrossRef]
- Reddy, B.S.; Hirose, Y.; Lubet, R.; Steele, V.; Kelloff, G.; Paulson, S.; Seibert, K.; Rao, C.V. Chemoprevention of colon cancer by specific cyclooxygenase-2 inhibitor, celecoxib, administered during different stages of carcinogenesis. Cancer Res. 2000, 60, 293–297. [Google Scholar]
- Wu, Z.; Qu, B.; Huang, X.; Song, Y.; Gao, P.; Shi, J.; Zhou, C.; Wang, Z. The potential adjunctive benefit of adding metformin to standard treatment in inoperable cancer patients: A meta-analysis of randomized controlled trials. Ann. Transl. Med. 2020, 8, 1404. [Google Scholar] [CrossRef]
- Lorenz, M.; Slaughter, H.S.; Wescott, D.M.; Carter, S.I.; Schnyder, B.; Dinchuk, J.E.; Car, B.D. Cyclooxygenase-2 is essential for normal recovery from 5-fluorouracil-induced myelotoxicity in mice. Exp. Hematol. 1999, 27, 1494–1502. [Google Scholar] [CrossRef]
- Ono, K.; Akatsu, T.; Murakami, T.; Kitamura, R.; Yamamoto, M.; Shinomiya, N.; Rokutanda, M.; Sasaki, T.; Amizuka, N.; Ozawa, H.; et al. Involvement of cyclo-oxygenase-2 in osteoclast formation and bone destruction in bone metastasis of mammary carcinoma cell lines. J. Bone. Miner. Res. 2002, 17, 774–781. [Google Scholar] [CrossRef]
- Rosell, R.; Carcereny, E.; Gervais, R.; Vergnenegre, A.; Massuti, B.; Felip, E.; Palmero, R.; Garcia-Gomez, R.; Pallares, C.; Sanchez, J.M.; et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): A multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012, 13, 239–246. [Google Scholar] [CrossRef]
- Mok, T.S.; Wu, Y.L.; Thongprasert, S.; Yang, C.H.; Chu, D.T.; Saijo, N.; Sunpaweravong, P.; Han, B.; Margono, B.; Ichinose, Y.; et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N. Engl. J. Med. 2009, 361, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Yang, Y.; Yuan, H.; Wu, M.; Li, S.; Gong, W.; Yu, J.; Xia, W.; Zhang, Y.; Ding, G.; et al. Inhibition of COX-2/PGE2 cascade ameliorates cisplatin-induced mesangial cell apoptosis. Am. J. Transl. Res. 2017, 9, 1222–1229. [Google Scholar] [PubMed]
- Lucci, A.; Krishnamurthy, S.; Singh, B.; Bedrosian, I.; Meric-Bernstam, F.; Reuben, J.; Broglio, K.; Mosalpuria, K.; Lodhi, A.; Vincent, L.; et al. Cyclooxygenase-2 expression in primary breast cancers predicts dissemination of cancer cells to the bone marrow. Breast Cancer Res. Treat. 2009, 117, 61–68. [Google Scholar] [CrossRef][Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).