Bispecific T-Cell Engagers and Chimeric Antigen Receptor T-Cell Therapies in Glioblastoma: An Update
Abstract
:1. Introduction
2. Bispecific Antibodies
2.1. What Are Bispecific Antibodies
2.1.1. Structure
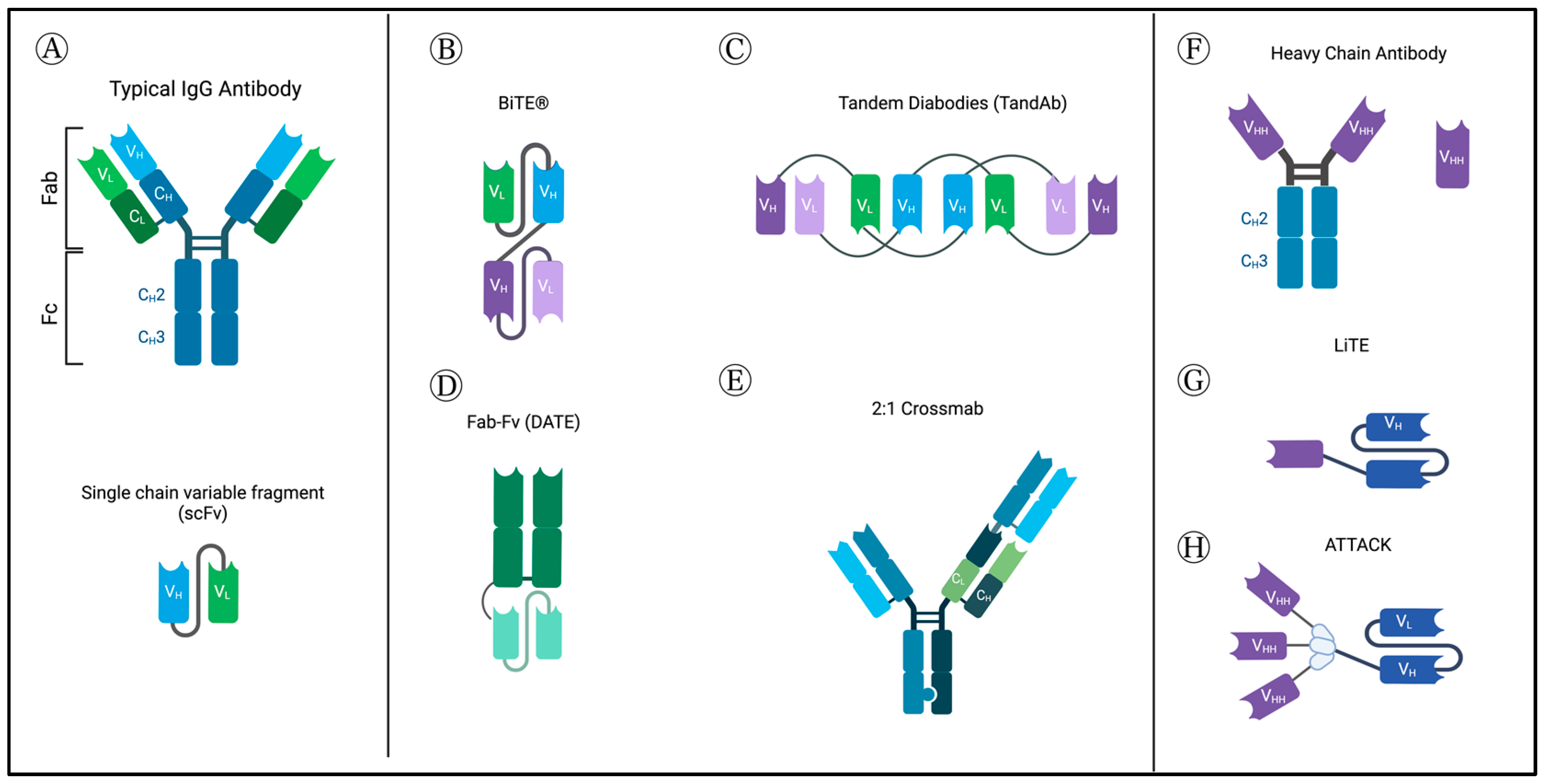
2.1.2. Mechanism of Action
2.2. Advantages
2.2.1. BsAbs Are Easily Manufactured
2.2.2. Smaller Size of BsAbs Fragments May Allow for Better Penetration of Tissue and a Higher Affinity Immune Synapse
2.2.3. BsAbs Are Highly Potent
2.2.4. BiTE Mediated T-Cell Activation Is Independent of TCR-MHC Interaction
2.2.5. BiTEs Initiate Bystander Tumor Cell Killing
2.2.6. BiTEs May Overcome Immunosuppression by Redirecting Tregs
2.3. Targets
2.4. Preclinical Experience
2.4.1. T-Cell-Engaging BsAbs
2.4.2. BsAbs with Targets Other Than CD3e
2.4.3. BsAbs Targeting Dual Signaling Pathways
2.5. Clinical Experience
2.5.1. Clinical Experience in Other Malignancies
2.5.2. Glioblastoma
2.6. Challenges and Novel Approaches
2.6.1. Delivery
2.6.2. Antigen Heterogeneity and Antigen Escape
2.6.3. Immunosuppressive Microenvironment and T-Cell Exhaustion
2.6.4. Toxicity
2.6.5. Novel Designs Utilizing Nanobodies
3. CAR T-Cell Therapy
3.1. What Are CAR T-Cells
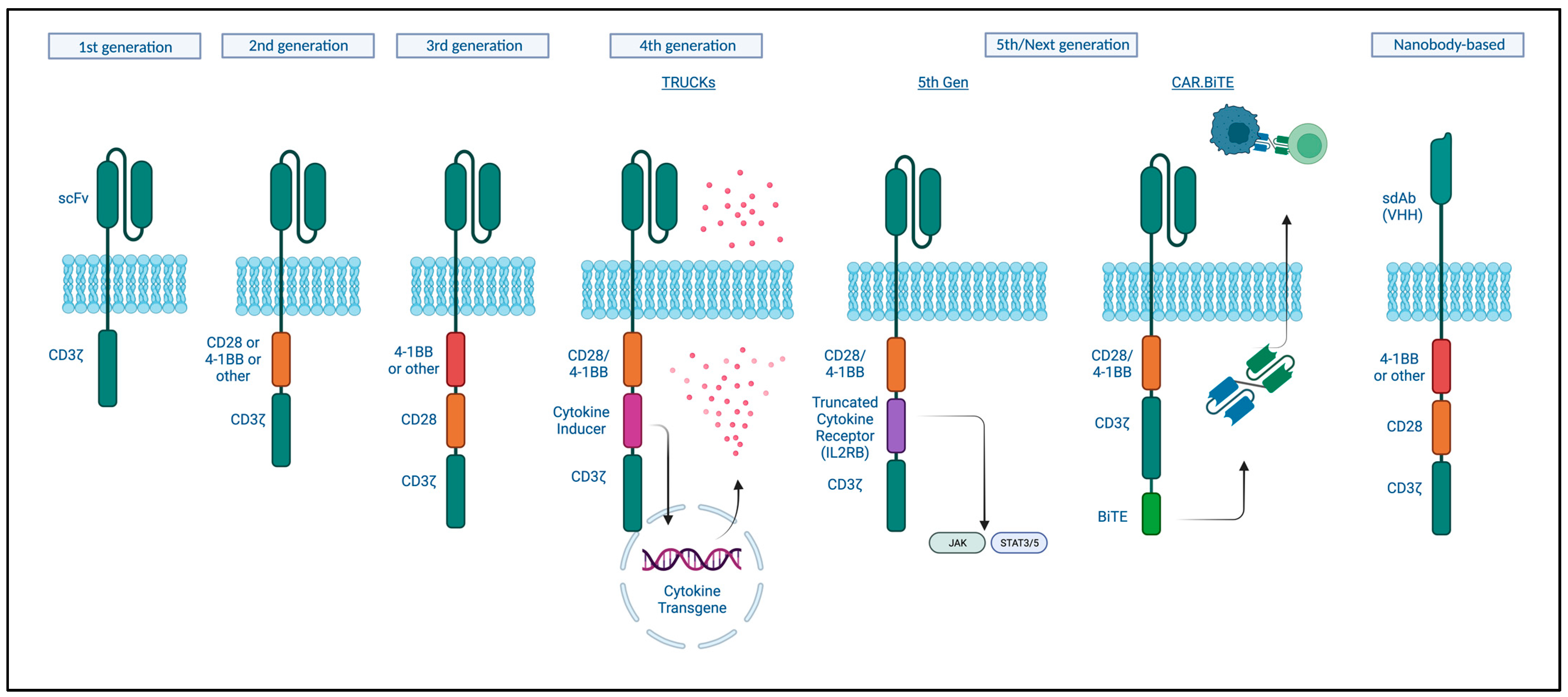
3.1.1. Design
3.1.2. Manufacturing and Mechanism
3.2. Advantages
3.3. Preclinical and Clinical Experience
3.3.1. Preclinical Experience
3.3.2. Clinical Experience in Glioblastoma
3.4. Challenges
3.4.1. Time to Manufacture
3.4.2. Delivery
3.4.3. Antigen Heterogeneity and Antigen Loss
3.4.4. Persistence, T-Cell Exhaustion, and the Microenvironment
3.4.5. Toxicity
4. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stupp, R.; Weller, M.; Belanger, K.; Bogdahn, U.; Ludwin, S.K.; Lacombe, D.; Mirimanoff, R.O. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Ho, V.K.Y.; Reijneveld, J.C.; Enting, R.H.; Bienfait, H.P.; Robe, P.; Baumert, B.G.; Visser, O.; Dutch Society for Neuro-Oncology (LWNO). Changing Incidence and Improved Survival of Gliomas. Eur. J. Cancer 2014, 50, 2309–2318. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, E.; Ermani, M.; Bartolini, S.; Bartolotti, M.; Poggi, R.; Tallini, G.; Marucci, G.; Fioravanti, A.; Tosoni, A.; Agati, R.; et al. Post Progression Survival in Glioblastoma: Where Are We? J. Neurooncol. 2015, 121, 399–404. [Google Scholar] [CrossRef]
- Taal, W.; Oosterkamp, H.M.; Walenkamp, A.M.E.; Dubbink, H.J.; Beerepoot, L.V.; Hanse, M.C.J.; Buter, J.; Honkoop, A.H.; Boerman, D.; de Vos, F.Y.F.; et al. Single-Agent Bevacizumab or Lomustine versus a Combination of Bevacizumab plus Lomustine in Patients with Recurrent Glioblastoma (BELOB Trial): A Randomised Controlled Phase 2 Trial. Lancet Oncol. 2014, 15, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.Q. Immune Checkpoint Inhibitors in GBM. J. Neurooncol. 2021, 155, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chuntova, P.; Chow, F.; Watchmaker, P.B.; Galvez, M.; Heimberger, A.B.; Newell, E.W.; Diaz, A.; Depinho, R.A.; Li, M.O.; Wherry, E.J.; et al. Unique Challenges for Glioblastoma Immunotherapy-Discussions across Neuro-Oncology and Non-Neuro-Oncology Experts in Cancer Immunology. Meeting Report from the 2019 SNO Immuno-Oncology Think Tank. Neuro-Oncol. 2021, 23, 356–375. [Google Scholar] [CrossRef] [PubMed]
- Khasraw, M.; Reardon, D.A.; Weller, M.; Sampson, J.H. PD-1 Inhibitors: Do They Have a Future in the Treatment of Glioblastoma? Clin. Cancer Res. 2020, 26, 5287–5296. [Google Scholar] [CrossRef]
- Janeway, C.A.; Travers, P.; Walport, M.; Shlomchik, M.J. Immunobiology: The Immune System in Health and Disease. In The Structure of a Typical Antibody Molecule; Garland Science: New York, NY, USA, 2001; ISBN 0-8153-3642-X. [Google Scholar]
- Suurs, F.V.; Lub-de Hooge, M.N.; de Vries, E.G.E.; de Groot, D.J.A. A Review of Bispecific Antibodies and Antibody Constructs in Oncology and Clinical Challenges. Pharmacol. Ther. 2019, 201, 103–119. [Google Scholar] [CrossRef]
- Frontiers | Bispecific Antibodies: From Research to Clinical Application. Available online: https://www.frontiersin.org/articles/10.3389/fimmu.2021.626616/full (accessed on 14 March 2023).
- Einsele, H.; Borghaei, H.; Orlowski, R.Z.; Subklewe, M.; Roboz, G.J.; Zugmaier, G.; Kufer, P.; Iskander, K.; Kantarjian, H.M. The BiTE (Bispecific T-Cell Engager) Platform: Development and Future Potential of a Targeted Immuno-Oncology Therapy across Tumor Types. Cancer 2020, 126, 3192–3201. [Google Scholar] [CrossRef]
- Ellwanger, K.; Reusch, U.; Fucek, I.; Knackmuss, S.; Weichel, M.; Gantke, T.; Molkenthin, V.; Zhukovsky, E.A.; Tesar, M.; Treder, M. Highly Specific and Effective Targeting of EGFRvIII-Positive Tumors with TandAb Antibodies. Front. Oncol. 2017, 7, 100. [Google Scholar] [CrossRef]
- Raskov, H.; Orhan, A.; Christensen, J.P.; Gögenur, I. Cytotoxic CD8+ T Cells in Cancer and Cancer Immunotherapy. Br. J. Cancer 2021, 124, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Dreier, T.; Lorenczewski, G.; Brandl, C.; Hoffmann, P.; Syring, U.; Hanakam, F.; Kufer, P.; Riethmuller, G.; Bargou, R.; Baeuerle, P.A. Extremely Potent, Rapid and Costimulation-Independent Cytotoxic T-Cell Response against Lymphoma Cells Catalyzed by a Single-Chain Bispecific Antibody. Int. J. Cancer 2002, 100, 690–697. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Hotchkiss, K.M.; Mohan, A.A.; Reedy, J.L.; Sampson, J.H.; Khasraw, M. For Whom the T Cells Troll? Bispecific T-Cell Engagers in Glioblastoma. J. Immunother. Cancer 2021, 9, e003679. [Google Scholar] [CrossRef] [PubMed]
- Goebeler, M.-E.; Bargou, R.C. T Cell-Engaging Therapies—BiTEs and Beyond. Nat. Rev. Clin. Oncol. 2020, 17, 418–434. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Chen, Y.; Park, J.; Liu, X.; Hu, Y.; Wang, T.; McFarland, K.; Betenbaugh, M.J. Design and Production of Bispecific Antibodies. Antibodies 2019, 8, 43. [Google Scholar] [CrossRef]
- Brown, C.E.; Badie, B.; Barish, M.E.; Weng, L.; Ostberg, J.R.; Chang, W.-C.; Naranjo, A.; Starr, R.; Wagner, J.; Wright, C.; et al. Bioactivity and Safety of IL13Ralpha2-Redirected Chimeric Antigen Receptor CD8+ T Cells in Patients with Recurrent Glioblastoma. Clin. Cancer Res. 2015, 21, 4062–4072. [Google Scholar] [CrossRef]
- Brinkmann, U.; Kontermann, R.E. The Making of Bispecific Antibodies. mAbs 2017, 9, 182–212. [Google Scholar] [CrossRef]
- Wu, Y.; Yi, M.; Zhu, S.; Wang, H.; Wu, K. Recent Advances and Challenges of Bispecific Antibodies in Solid Tumors. Exp. Hematol. Oncol. 2021, 10, 56. [Google Scholar] [CrossRef]
- Wang, L.; Hoseini, S.S.; Xu, H.; Ponomarev, V.; Cheung, N.-K.V. Silencing Fc Domains in T Cell–Engaging Bispecific Antibodies Improves T-Cell Trafficking and Antitumor Potency. Cancer Immunol. Res. 2019, 7, 2013–2024. [Google Scholar] [CrossRef]
- Thurber, G.M.; Schmidt, M.M.; Wittrup, K.D. Antibody Tumor Penetration: Transport Opposed by Systemic and Antigen-Mediated Clearance. Adv. Drug Deliv. Rev. 2008, 60, 1421–1434. [Google Scholar] [CrossRef]
- Weigelin, B.; den Boer, A.T.; Wagena, E.; Broen, K.; Dolstra, H.; de Boer, R.J.; Figdor, C.G.; Textor, J.; Friedl, P. Cytotoxic T Cells Are Able to Efficiently Eliminate Cancer Cells by Additive Cytotoxicity. Nat. Commun. 2021, 12, 5217. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Zhou, Y.; Han, L.; Pan, Z.; Chen, J.; Zong, H.; Bian, Y.; Jiang, H.; Zhang, B.; Zhu, J. A Rational Designed Novel Bispecific Antibody for the Treatment of GBM. Biomedicines 2021, 9, 640. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, P.; Hofmeister, R.; Brischwein, K.; Brandl, C.; Crommer, S.; Bargou, R.; Itin, C.; Prang, N.; Baeuerle, P.A. Serial Killing of Tumor Cells by Cytotoxic T Cells Redirected with a CD19-/CD3-Bispecific Single-Chain Antibody Construct. Int. J. Cancer 2005, 115, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Offner, S.; Hofmeister, R.; Romaniuk, A.; Kufer, P.; Baeuerle, P.A. Induction of Regular Cytolytic T Cell Synapses by Bispecific Single-Chain Antibody Constructs on MHC Class I-Negative Tumor Cells. Mol. Immunol. 2006, 43, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.L.; Sherman, M.; McElroy, P.L.; Lofgren, J.A.; Moody, G.; Baeuerle, P.A.; Coxon, A.; Arvedson, T. Bispecific T Cell Engager (BiTE®) Antibody Constructs Can Mediate Bystander Tumor Cell Killing. PLoS ONE 2017, 12, e0183390. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D. ICAM-1 Overexpression Counteracts Immune-Suppression by Tumour Cell-Derived PGE2 to Restore CTL Function. J. Immunol. Sci. 2018, 2, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Jiang, P.; Wei, S.; Xu, X.; Wang, J. Regulatory T Cells in Tumor Microenvironment: New Mechanisms, Potential Therapeutic Strategies and Future Prospects. Mol. Cancer 2020, 19, 116. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.D.; Gedeon, P.C.; Herndon, J.E.; Archer, G.E.; Reap, E.A.; Sanchez-Perez, L.; Mitchell, D.A.; Bigner, D.D.; Sampson, J.H. Human Regulatory T Cells Kill Tumor Cells through Granzyme-Dependent Cytotoxicity upon Retargeting with a Bispecific Antibody. Cancer Immunol. Res. 2013, 1, 163. [Google Scholar] [CrossRef]
- Choi, B.D.; Yu, X.; Castano, A.P.; Bouffard, A.A.; Schmidts, A.; Larson, R.C.; Bailey, S.R.; Boroughs, A.C.; Frigault, M.J.; Leick, M.B.; et al. CAR-T Cells Secreting BiTEs Circumvent Antigen Escape without Detectable Toxicity. Nat. Biotechnol. 2019, 37, 1049–1058. [Google Scholar] [CrossRef]
- Koristka, S.; Cartellieri, M.; Arndt, C.; Feldmann, A.; Seliger, B.; Ehninger, G.; Bachmann, M.P. Tregs Activated by Bispecific Antibodies: Killers or Suppressors? OncoImmunology 2015, 4, 1–3. [Google Scholar] [CrossRef]
- Duell, J.; Dittrich, M.; Bedke, T.; Mueller, T.; Eisele, F.; Rosenwald, A.; Rasche, L.; Hartmann, E.; Dandekar, T.; Einsele, H.; et al. Frequency of Regulatory T Cells Determines the Outcome of the T-Cell-Engaging Antibody Blinatumomab in Patients with B-Precursor ALL. Leukemia 2017, 31, 2181–2190. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.T.F.; Sharma, V.; Mendelsohn, A.; Wei, Q.; Li, J.; Yu, B.; Larrick, J.W.; Lum, L.G. Broad Reactivity and Enhanced Potency of Recombinant Anti-EGFR x Anti-CD3 Bispecific Antibody-Armed Activated T Cells against Solid Tumours. Ann. Med. 2022, 54, 1047–1057. [Google Scholar] [CrossRef]
- Jiang, H.; Gao, H.; Kong, J.; Song, B.; Wang, P.; Shi, B.; Wang, H.; Li, Z. Selective Targeting of Glioblastoma with EGFRvIII/EGFR Bitargeted Chimeric Antigen Receptor T Cell. Cancer Immunol. Res. 2018, 6, 1314–1326. [Google Scholar] [CrossRef] [PubMed]
- Ravanpay, A.C.; Gust, J.; Johnson, A.J.; Rolczynski, L.S.; Cecchini, M.; Chang, C.A.; Hoglund, V.J.; Mukherjee, R.; Vitanza, N.A.; Orentas, R.J.; et al. EGFR806-CAR T Cells Selectively Target a Tumor-Restricted EGFR Epitope in Glioblastoma. Oncotarget 2019, 10, 7080–7095. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Liu, J.-Y.; Zheng, Z.-Z.; Chen, Y.-J.; Ding, J.-C.; Hu, Y.-H.; Hu, G.-S.; Xia, N.-S.; Liu, W. BRD4 Inhibition Boosts the Therapeutic Effects of Epidermal Growth Factor Receptor-Targeted Chimeric Antigen Receptor T Cells in Glioblastoma. Mol. Ther. 2021, 29, 3011–3026. [Google Scholar] [CrossRef] [PubMed]
- Thokala, R.; Binder, Z.A.; Yin, Y.; Zhang, L.; Zhang, J.V.; Zhang, D.Y.; Milone, M.C.; Ming, G.-L.; Song, H.; O’Rourke, D.M. High-Affinity Chimeric Antigen Receptor with Cross-Reactive scFv to Clinically Relevant EGFR Oncogenic Isoforms. Front. Oncol. 2021, 11, 664236. [Google Scholar] [CrossRef] [PubMed]
- Gan, H.K.; Cvrljevic, A.N.; Johns, T.G. The Epidermal Growth Factor Receptor Variant III (EGFRvIII): Where Wild Things Are Altered. FEBS J. 2013, 280, 5350–5370. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.D.; Kuan, C.-T.; Cai, M.; Archer, G.E.; Mitchell, D.A.; Gedeon, P.C.; Sanchez-Perez, L.; Pastan, I.; Bigner, D.D.; Sampson, J.H. Systemic Administration of a Bispecific Antibody Targeting EGFRvIII Successfully Treats Intracerebral Glioma. Proc. Natl. Acad. Sci. USA 2013, 110, 270–275. [Google Scholar] [CrossRef]
- Gedeon, P.C.; Schaller, T.H.; Chitneni, S.K.; Choi, B.D.; Kuan, C.-T.; Suryadevara, C.M.; Snyder, D.J.; Schmittling, R.J.; Szafranski, S.E.; Cui, X.; et al. A Rationally Designed Fully Human EGFRvIII:CD3-Targeted Bispecific Antibody Redirects Human T Cells to Treat Patient-Derived Intracerebral Malignant Glioma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018, 24, 3611–3631. [Google Scholar] [CrossRef]
- Iurlaro, R.; Waldhauer, I.; Planas-Rigol, E.; Bonfill-Teixidor, E.; Arias, A.; Nicolini, V.; Freimoser-Grundschober, A.; Cuartas, I.; Martinez-Moreno, A.; Martinez-Ricarte, F.; et al. A Novel EGFRvIII T-Cell Bispecific Antibody for the Treatment of Glioblastoma. Mol. Cancer Ther. 2022, 21, 1499–1509. [Google Scholar] [CrossRef]
- Park, D.H.; Perales-Puchalt, A.; Bhojnagarwala, P.; Gary, E.N.; Zhu, X.; Patel, A.; Yun, K.; Smith, T.; Muthumani, K.; Weiner, D.B. EGFRvIII-Targeting DNA-Encoded Immune Cell Engager (DICE) Generates In Vivo Expression of Bispecific Antibody That Induces T Cell-Mediated Cytolytic Activities Against EGFRvIII-Positive Tumors and Controls Tumor Growth in a GBM Mouse Model. Mol. Ther. 2020, 28, 497. [Google Scholar] [CrossRef]
- Park, D.H.; Liaw, K.; Bhojnagarwala, P.; Zhu, X.; Choi, J.; Ali, A.R.; Bordoloi, D.; Gary, E.N.; O’Connell, R.P.; Kulkarni, A.; et al. Multivalent in Vivo Delivery of DNA-Encoded Bispecific T Cell Engagers Effectively Controls Heterogeneous GBM Tumors and Mitigates Immune Escape. Mol. Ther.-Oncolytics 2023, 28, 249–263. [Google Scholar] [CrossRef] [PubMed]
- Gardell, J.L.; Matsumoto, L.R.; Chinn, H.; DeGolier, K.R.; Kreuser, S.A.; Prieskorn, B.; Balcaitis, S.; Davis, A.; Ellenbogen, R.G.; Crane, C.A. Human Macrophages Engineered to Secrete a Bispecific T Cell Engager Support Antigen-Dependent T Cell Responses to Glioblastoma. J. Immunother. Cancer 2020, 8, e001202. [Google Scholar] [CrossRef] [PubMed]
- Bullain, S.S.; Sahin, A.; Szentirmai, O.; Sanchez, C.; Lin, N.; Baratta, E.; Waterman, P.; Weissleder, R.; Mulligan, R.C.; Carter, B.S. Genetically Engineered T Cells to Target EGFRvIII Expressing Glioblastoma. J. Neurooncol. 2009, 94, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Natsume, A.; Ichiro Iwami, K.; Iwamizu, H.; Noritake, K.; Ito, D.; Toi, Y.; Ito, M.; Motomura, K.; Yoshida, J.; et al. Retrovirally Engineered T-Cell-Based Immunotherapy Targeting Type III Variant Epidermal Growth Factor Receptor, a Glioma-Associated Antigen. Cancer Sci. 2010, 101, 2518–2524. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Ohkuri, T.; Kosaka, A.; Tanahashi, K.; June, C.H.; Natsume, A.; Okada, H. Expression of miR-17-92 Enhances Anti-Tumor Activity of T-Cells Transduced with the Anti-EGFRvIII Chimeric Antigen Receptor in Mice Bearing Human GBM Xenografts. J. Immunother. Cancer 2013, 1, 21. [Google Scholar] [CrossRef] [PubMed]
- Sampson, J.H.; Choi, B.D.; Sanchez-Perez, L.; Suryadevara, C.M.; Snyder, D.J.; Flores, C.T.; Schmittling, R.J.; Nair, S.K.; Reap, E.A.; Norberg, P.K.; et al. EGFRvIII mCAR-Modified T-Cell Therapy Cures Mice with Established Intracerebral Glioma and Generates Host Immunity against Tumor-Antigen Loss. Clin. Cancer Res. 2014, 20, 972–984. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.A.; Scholler, J.; Ohkuri, T.; Kosaka, A.; Patel, P.R.; McGettigan, S.E.; Nace, A.K.; Dentchev, T.; Thekkat, P.; Loew, A.; et al. Rational Development and Characterization of Humanized Anti-EGFR Variant III Chimeric Antigen Receptor T Cells for Glioblastoma. Sci. Transl. Med. 2015, 7, 275ra22. [Google Scholar] [CrossRef]
- Ma, W.; Wang, Y.; Zhang, R.; Yang, F.; Zhang, D.; Huang, M.; Zhang, L.; Dorsey, J.F.; Binder, Z.A.; O’Rourke, D.M.; et al. Targeting PAK4 to Reprogram the Vascular Microenvironment and Improve CAR-T Immunotherapy for Glioblastoma. Nat. Cancer 2021, 2, 83–97. [Google Scholar] [CrossRef]
- Dong, X.; Ren, J.; Amoozgar, Z.; Lee, S.; Datta, M.; Roberge, S.; Duquette, M.; Fukumura, D.; Jain, R.K. Anti-VEGF Therapy Improves EGFR-vIII-CAR-T Cell Delivery and Efficacy in Syngeneic Glioblastoma Models in Mice. J. Immunother. Cancer 2023, 11. [Google Scholar] [CrossRef]
- Zhang, D.; Li, A.M.; Hu, G.; Huang, M.; Yang, F.; Zhang, L.; Wellen, K.E.; Xu, X.; Conn, C.S.; Zou, W.; et al. PHGDH-Mediated Endothelial Metabolism Drives Glioblastoma Resistance to Chimeric Antigen Receptor T Cell Immunotherapy. Cell Metab. 2023, 35, 517–534.e8. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Zhang, D.; Jiang, H.; Ye, J.; Zhang, L.; Bagley, S.J.; Winkler, J.; Gong, Y.; Fan, Y. Small-Molecule Toosendanin Reverses Macrophage-Mediated Immunosuppression to Overcome Glioblastoma Resistance to Immunotherapy. Sci. Transl. Med. 2023, 15, eabq3558. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Morello, A.; Tano, Z.; Adusumilli, P.S. CAR T-Cell Intrinsic PD-1 Checkpoint Blockade: A Two-in-One Approach for Solid Tumor Immunotherapy. Oncoimmunology 2016, 6, e1273302. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; You, Y.; Shen, Z.; Shi, L. EGFRvIII-CAR-T Cells with PD-1 Knockout Have Improved Anti-Glioma Activity. Pathol. Oncol. Res. POR 2020, 26, 2135–2141. [Google Scholar] [CrossRef] [PubMed]
- O’Rourke, D.M.; Nasrallah, M.P.; Desai, A.; Melenhorst, J.J.; Mansfield, K.; Morrissette, J.J.D.; Martinez-Lage, M.; Brem, S.; Maloney, E.; Shen, A.; et al. A Single Dose of Peripherally Infused EGFRvIII-Directed CAR T Cells Mediates Antigen Loss and Induces Adaptive Resistance in Patients with Recurrent Glioblastoma. Sci. Transl. Med. 2017, 9, eaaa0984. [Google Scholar] [CrossRef] [PubMed]
- Goff, S.L.; Morgan, R.A.; Yang, J.C.; Sherry, R.M.; Robbins, P.F.; Restifo, N.P.; Feldman, S.A.; Lu, Y.-C.; Lu, L.; Zheng, Z.; et al. Pilot Trial of Adoptive Transfer of Chimeric Antigen Receptor-Transduced t Cells Targeting Egfrviii in Patients with Glioblastoma. J. Immunother. 2019, 42, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Zhang, J.; Yang, Y.-Z.; Wang, F.; Jiang, H.; Chen, H.-D.; Wu, H.-Y.; Sai, K.; Hu, W.-M. IL13RA2 Is Overexpressed in Malignant Gliomas and Related to Clinical Outcome of Patients. Am. J. Transl. Res. 2020, 12, 4702–4714. [Google Scholar] [PubMed]
- Brown, C.E.; Starr, R.; Aguilar, B.; Shami, A.F.; Martinez, C.; D’Apuzzo, M.; Barish, M.E.; Forman, S.J.; Jensen, M.C. Stem-like Tumor-Initiating Cells Isolated from IL13Ralpha2 Expressing Gliomas Are Targeted and Killed by IL13-Zetakine-Redirected T Cells. Clin. Cancer Res. 2012, 18, 2199–2209. [Google Scholar] [CrossRef]
- Debinski, W.; Gibo, D.M.; Hulet, S.W.; Connor, J.R.; Gillespie, G.Y. Receptor for Interleukin 13 Is a Marker and Therapeutic Target for Human High-Grade Gliomas. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 1999, 5, 985–990. [Google Scholar]
- Kawakami, M.; Kawakami, K.; Takahashi, S.; Abe, M.; Puri, R.K. Analysis of Interleukin-13 Receptor Alpha2 Expression in Human Pediatric Brain Tumors. Cancer 2004, 101, 1036–1042. [Google Scholar] [CrossRef]
- Joshi, B.H.; Puri, R.A.; Leland, P.; Varricchio, F.; Gupta, G.; Kocak, M.; Gilbertson, R.J.; Puri, R.K. Identification of Interleukin-13 Receptor A2 Chain Overexpression in Situ in High-Grade Diffusely Infiltrative Pediatric Brainstem Glioma. Neuro-Oncol. 2008, 10, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Jarboe, J.S.; Johnson, K.R.; Choi, Y.; Lonser, R.R.; Park, J.K. Expression of Interleukin-13 Receptor Alpha2 in Glioblastoma Multiforme: Implications for Targeted Therapies. Cancer Res. 2007, 67, 7983–7986. [Google Scholar] [CrossRef] [PubMed]
- Bhojnagarwala, P.S.; O’Connell, R.P.; Park, D.; Liaw, K.; Ali, A.R.; Bordoloi, D.; Cassel, J.; Tursi, N.J.; Gary, E.; Weiner, D.B. In Vivo DNA-Launched Bispecific T Cell Engager Targeting IL-13Ralpha2 Controls Tumor Growth in an Animal Model of Glioblastoma Multiforme. Mol. Ther.-Oncolytics 2022, 26, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Pituch, K.C.; Zannikou, M.; Ilut, L.; Xiao, T.; Chastkofsky, M.; Sukhanova, M.; Bertolino, N.; Procissi, D.; Amidei, C.; Horbinski, C.M.; et al. Neural Stem Cells Secreting Bispecific T Cell Engager to Induce Selective Antiglioma Activity. Proc. Natl. Acad. Sci. USA 2021, 118, e2015800118. [Google Scholar] [CrossRef] [PubMed]
- Kong, S.; Sengupta, S.; Tyler, B.; Bais, A.J.; Ma, Q.; Doucette, S.; Zhou, J.; Sahin, A.; Carter, B.S.; Brem, H.; et al. Suppression of Human Glioma Xenografts with Second-Generation IL13R-Specific Chimeric Antigen Receptor-Modified T Cells. Clin. Cancer Res. 2012, 18, 5949–5960. [Google Scholar] [CrossRef] [PubMed]
- Zannikou, M.; Duffy, J.T.; Levine, R.N.; Seblani, M.; Liu, Q.; Presser, A.; Arrieta, V.A.; Chen, C.J.; Sonabend, A.M.; Horbinski, C.M.; et al. IL15 Modification Enables CAR T Cells to Act as a Dual Targeting Agent against Tumor Cells and Myeloid-Derived Suppressor Cells in GBM. J. Immunother. Cancer 2023, 11, e006239. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.E.; Alizadeh, D.; Starr, R.; Weng, L.; Wagner, J.R.; Naranjo, A.; Ostberg, J.R.; Blanchard, M.S.; Kilpatrick, J.; Simpson, J.; et al. Regression of Glioblastoma after Chimeric Antigen Receptor T-Cell Therapy. N. Engl. J. Med. 2016, 375, 2561–2569. [Google Scholar] [CrossRef]
- Brown, C.E.; Rodriguez, A.; Palmer, J.; Ostberg, J.R.; Naranjo, A.; Wagner, J.R.; Aguilar, B.; Starr, R.; Weng, L.; Synold, T.W.; et al. Off-the-Shelf, Steroid-Resistant, IL13Ralpha2-Specific CAR T Cells for Treatment of Glioblastoma. Neuro-Oncol. 2022, 24, 1318–1330. [Google Scholar] [CrossRef]
- Tran, N.L.; McDonough, W.S.; Savitch, B.A.; Sawyer, T.F.; Winkles, J.A.; Berens, M.E. The Tumor Necrosis Factor-like Weak Inducer of Apoptosis (TWEAK)-Fibroblast Growth Factor-Inducible 14 (Fn14) Signaling System Regulates Glioma Cell Survival via NFκB Pathway Activation and BCL-XL/BCL-W Expression*. J. Biol. Chem. 2005, 280, 3483–3492. [Google Scholar] [CrossRef]
- Tran, N.L.; McDonough, W.S.; Donohue, P.J.; Winkles, J.A.; Berens, T.J.; Ross, K.R.; Hoelzinger, D.B.; Beaudry, C.; Coons, S.W.; Berens, M.E. The Human Fn14 Receptor Gene Is Up-Regulated in Migrating Glioma Cells in Vitro and Overexpressed in Advanced Glial Tumors. Am. J. Pathol. 2003, 162, 1313–1321. [Google Scholar] [CrossRef]
- Li, G.; Zhang, Z.; Cai, L.; Tang, X.; Huang, J.; Yu, L.; Wang, G.; Zhong, K.; Cao, Y.; Liu, C.; et al. Fn14-Targeted BiTE and CAR-T Cells Demonstrate Potent Preclinical Activity against Glioblastoma. OncoImmunology 2021, 10, 1983306. [Google Scholar] [CrossRef] [PubMed]
- Vora, P.; Venugopal, C.; Salim, S.K.; Tatari, N.; Bakhshinyan, D.; Singh, M.; Seyfrid, M.; Upreti, D.; Rentas, S.; Wong, N.; et al. The Rational Development of CD133-Targeting Immunotherapies for Glioblastoma. Cell Stem Cell 2020, 26, 832–844.e6. [Google Scholar] [CrossRef] [PubMed]
- Bao, S.; Wu, Q.; McLendon, R.E.; Hao, Y.; Shi, Q.; Hjelmeland, A.B.; Dewhirst, M.W.; Bigner, D.D.; Rich, J.N. Glioma Stem Cells Promote Radioresistance by Preferential Activation of the DNA Damage Response. Nature 2006, 444, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Yuan, X.; Zeng, Z.; Tunici, P.; Ng, H.; Abdulkadir, I.R.; Lu, L.; Irvin, D.; Black, K.L.; Yu, J.S. Analysis of Gene Expression and Chemoresistance of CD133+ Cancer Stem Cells in Glioblastoma. Mol. Cancer 2006, 5, 67. [Google Scholar] [CrossRef] [PubMed]
- Tatari, N.; Zhang, X.; Chafe, S.C.; McKenna, D.; Lawson, K.A.; Subapanditha, M.; Shaikh, M.V.; Seyfrid, M.; Savage, N.; Venugopal, C.; et al. Dual Antigen T Cell Engagers Targeting CA9 as an Effective Immunotherapeutic Modality for Targeting CA9 in Solid Tumors. Front. Immunol. 2022, 13, 905768. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Ying, H.; Zeng, G.; Wheeler, C.J.; Black, K.L.; Yu, J.S. HER-2, Gp100, and MAGE-1 Are Expressed in Human Glioblastoma and Recognized by Cytotoxic T Cells. Cancer Res. 2004, 64, 4980–4986. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Roder, J.; Scherer, A.; Bodden, M.; Pfeifer Serrahima, J.; Bhatti, A.; Waldmann, A.; Muller, N.; Oberoi, P.; Wels, W.S. Bispecific Antibody-Mediated Redirection of NKG2D-CAR Natural Killer Cells Facilitates Dual Targeting and Enhances Antitumor Activity. J. Immunother. Cancer 2021, 9, e002980. [Google Scholar] [CrossRef]
- Ahmed, N.; Salsman, V.S.; Kew, Y.; Shaffer, D.; Powell, S.; Zhang, Y.J.; Grossman, R.G.; Heslop, H.E.; Gottschalk, S. HER2-Specific T Cells Target Primary Glioblastoma Stem Cells and Induce Regression of Autologous Experimental Tumors. Clin. Cancer Res. 2010, 16, 474–485. [Google Scholar] [CrossRef]
- Ahmed, N.; Brawley, V.; Hegde, M.; Bielamowicz, K.; Kalra, M.; Landi, D.; Robertson, C.; Gray, T.L.; Diouf, O.; Wakefield, A.; et al. HER2-Specific Chimeric Antigen Receptor-Modified Virus-Specific T Cells for Progressive Glioblastoma: A Phase 1 Dose-Escalation Trial. JAMA Oncol. 2017, 3, 1094–1101. [Google Scholar] [CrossRef]
- Vitanza, N.A.; Johnson, A.J.; Wilson, A.L.; Brown, C.; Yokoyama, J.K.; Kunkele, A.; Chang, C.A.; Rawlings-Rhea, S.; Huang, W.; Seidel, K.; et al. Locoregional Infusion of HER2-Specific CAR T Cells in Children and Young Adults with Recurrent or Refractory CNS Tumors: An Interim Analysis. Nat. Med. 2021, 27, 1544–1552. [Google Scholar] [CrossRef]
- Day, B.W.; Stringer, B.W.; Al-Ejeh, F.; Ting, M.J.; Wilson, J.; Ensbey, K.S.; Jamieson, P.R.; Bruce, Z.C.; Lim, Y.C.; Offenhäuser, C.; et al. EphA3 Maintains Tumorigenicity and Is a Therapeutic Target in Glioblastoma Multiforme. Cancer Cell 2013, 23, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Qazi, M.A.; Vora, P.; Venugopal, C.; Adams, J.; Singh, M.; Hu, A.; Gorelik, M.; Subapanditha, M.K.; Savage, N.; Yang, J.; et al. Cotargeting Ephrin Receptor Tyrosine Kinases A2 and A3 in Cancer Stem Cells Reduces Growth of Recurrent Glioblastoma. Cancer Res. 2018, 78, 5023–5037. [Google Scholar] [CrossRef] [PubMed]
- Iwahori, K.; Kakarla, S.; Velasquez, M.P.; Yu, F.; Yi, Z.; Gerken, C.; Song, X.-T.; Gottschalk, S. Engager T Cells: A New Class of Antigen-Specific t Cells That Redirect Bystander T Cells. Mol. Ther. 2015, 23, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Chow, K.K.; Naik, S.; Kakarla, S.; Brawley, V.S.; Shaffer, D.R.; Yi, Z.; Rainusso, N.; Wu, M.-F.; Liu, H.; Kew, Y.; et al. T Cells Redirected to EphA2 for the Immunotherapy of Glioblastoma. Mol. Ther. 2013, 21, 629–637. [Google Scholar] [CrossRef] [PubMed]
- Lin, Q.; Ba, T.; Ho, J.; Chen, D.; Cheng, Y.; Wang, L.; Xu, G.; Xu, L.; Zhou, Y.; Wei, Y.; et al. First-in-Human Trial of EphA2-Redirected CAR T-Cells in Patients with Recurrent Glioblastoma: A Preliminary Report of Three Cases at the Starting Dose. Front. Oncol. 2021, 11, 694941. [Google Scholar] [CrossRef] [PubMed]
- Seaman, S.; Zhu, Z.; Saha, S.; Zhang, X.M.; Yang, M.Y.; Hilton, M.B.; Morris, K.; Szot, C.; Morris, H.; Swing, D.A.; et al. Eradication of Tumors through Simultaneous Ablation of CD276/B7-H3 Positive Tumor Cells and Tumor Vasculature. Cancer Cell 2017, 31, 501–515.e8. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Zhao, S.; Zhang, Y.; Wang, Y.; Zhang, Z.; Yang, M.; Zhu, Y.; Zhang, G.; Guo, G.; Tong, A.; et al. B7-H3 as a Novel CAR-T Therapeutic Target for Glioblastoma. Mol. Ther.-Oncolytics 2019, 14, 279–287. [Google Scholar] [CrossRef]
- Nehama, D.; Di Ianni, N.; Musio, S.; Du, H.; Patane, M.; Pollo, B.; Finocchiaro, G.; Park, J.J.H.; Dunn, D.E.; Edwards, D.S.; et al. B7-H3-Redirected Chimeric Antigen Receptor T Cells Target Glioblastoma and Neurospheres. EBioMedicine 2019, 47, 33–43. [Google Scholar] [CrossRef]
- Haydar, D.; Houke, H.; Chiang, J.; Yi, Z.; Ode, Z.; Caldwell, K.; Zhu, X.; Mercer, K.S.; Stripay, J.L.; Shaw, T.I.; et al. Cell-Surface Antigen Profiling of Pediatric Brain Tumors: B7-H3 Is Consistently Expressed and Can Be Targeted via Local or Systemic CAR T-Cell Delivery. Neuro-Oncol. 2021, 23, 999–1011. [Google Scholar] [CrossRef]
- Tang, X.; Wang, Y.; Huang, J.; Zhang, Z.; Liu, F.; Xu, J.; Guo, G.; Wang, W.; Tong, A.; Zhou, L. Administration of B7-H3 Targeted Chimeric Antigen Receptor-T Cells Induce Regression of Glioblastoma. Signal Transduct. Target. Ther. 2021, 6, 125. [Google Scholar] [CrossRef]
- Prapa, M.; Chiavelli, C.; Golinelli, G.; Grisendi, G.; Bestagno, M.; Di Tinco, R.; Dall’Ora, M.; Neri, G.; Candini, O.; Spano, C.; et al. GD2 CAR T Cells against Human Glioblastoma. Npj Precis. Oncol. 2021, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Gargett, T.; Ebert, L.M.; Truong, N.T.H.; Kollis, P.M.; Sedivakova, K.; Yu, W.; Yeo, E.C.F.; Wittwer, N.L.; Gliddon, B.L.; Tea, M.N.; et al. GD2-Targeting CAR-T Cells Enhanced by Transgenic IL-15 Expression Are an Effective and Clinically Feasible Therapy for Glioblastoma. bioRxiv 2022, 10, e005187. [Google Scholar] [CrossRef] [PubMed]
- MMP-2 Expression and Correlation with Pathology and MRI of Glioma-PMC. Available online: https://www-ncbi-nlm-nih-gov.myaccess.library.utoronto.ca/pmc/articles/PMC6341586/ (accessed on 15 May 2023).
- Wang, D.; Starr, R.; Chang, W.-C.; Aguilar, B.; Alizadeh, D.; Wright, S.L.; Yang, X.; Brito, A.; Sarkissian, A.; Ostberg, J.R.; et al. Chlorotoxin-Directed CAR T Cells for Specific and Effective Targeting of Glioblastoma. Sci. Transl. Med. 2020, 12, eaaw2672. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Ge, H.; Long, Y.; Yang, C.; Chang, Y.E.; Mu, L.; Sayour, E.J.; De Leon, G.; Wang, Q.J.; Yang, J.C.; et al. CD70, a Novel Target of CAR T-Cell Therapy for Gliomas. Neuro-Oncol. 2018, 20, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Weiss, T.; Schneider, H.; Silginer, M.; Steinle, A.; Pruschy, M.; Polić, B.; Weller, M.; Roth, P. NKG2D-Dependent Antitumor Effects of Chemotherapy and Radiotherapy against Glioblastoma. Clin. Cancer Res. 2018, 24, 882–895. [Google Scholar] [CrossRef] [PubMed]
- Weiss, T.; Weller, M.; Guckenberger, M.; Sentman, C.L.; Roth, P. NKG2D-Based CAR T Cells and Radiotherapy Exert Synergistic Efficacy in Glioblastoma. Cancer Res. 2018, 78, 1031–1043. [Google Scholar] [CrossRef]
- Yang, D.; Sun, B.; Dai, H.; Li, W.; Shi, L.; Zhang, P.; Li, S.; Zhao, X. T Cells Expressing NKG2D Chimeric Antigen Receptors Efficiently Eliminate Glioblastoma and Cancer Stem Cells. J. Immunother. Cancer 2019, 7, 171. [Google Scholar] [CrossRef] [PubMed]
- Meister, H.; Look, T.; Roth, P.; Pascolo, S.; Sahin, U.; Lee, S.; Hale, B.D.; Snijder, B.; Regli, L.; Ravi, V.; et al. Multifunctional mRNA-Based CAR T Cells Display Promising Anti-Tumor Activity against Glioblastoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2022, 28, 4747–4756. [Google Scholar] [CrossRef]
- Shiina, S.; Ohno, M.; Ohka, F.; Kuramitsu, S.; Yamamichi, A.; Kato, A.; Motomura, K.; Tanahashi, K.; Yamamoto, T.; Watanabe, R.; et al. CAR T Cells Targeting Podoplanin Reduce Orthotopic Glioblastomas in Mouse Brains. Cancer Immunol. Res. 2016, 4, 259–268. [Google Scholar] [CrossRef]
- Chulpanova, D.S.; Kitaeva, K.V.; Rutland, C.S.; Rizvanov, A.A.; Solovyeva, V.V. Mouse Tumor Models for Advanced Cancer Immunotherapy. Int. J. Mol. Sci. 2020, 21, 4118. [Google Scholar] [CrossRef]
- Scott, A.M.; Lee, F.-T.; Tebbutt, N.; Herbertson, R.; Gill, S.S.; Liu, Z.; Skrinos, E.; Murone, C.; Saunder, T.H.; Chappell, B.; et al. A Phase I Clinical Trial with Monoclonal Antibody Ch806 Targeting Transitional State and Mutant Epidermal Growth Factor Receptors. Proc. Natl. Acad. Sci. USA 2007, 104, 4071–4076. [Google Scholar] [CrossRef] [PubMed]
- Zalutsky, M.R.; Moseley, R.P.; Coakham, H.B.; Coleman, R.E.; Bigner, D.D. Pharmacokinetics and Tumor Localization of 131I-Labeled Anti-Tenascin Monoclonal Antibody 81C6 in Patients with Gliomas and Other Intracranial Malignancies. Cancer Res. 1989, 49, 2807–2813. [Google Scholar] [PubMed]
- Schaller, T.H.; Snyder, D.J.; Spasojevic, I.; Gedeon, P.C.; Sanchez-Perez, L.; Sampson, J.H. First in Human Dose Calculation of a Single-Chain Bispecific Antibody Targeting Glioma Using the MABEL Approach. J. Immunother. Cancer 2020, 8, e000213. [Google Scholar] [CrossRef] [PubMed]
- Haber, L.; Olson, K.; Kelly, M.P.; Crawford, A.; DiLillo, D.J.; Tavaré, R.; Ullman, E.; Mao, S.; Canova, L.; Sineshchekova, O.; et al. Generation of T-Cell-Redirecting Bispecific Antibodies with Differentiated Profiles of Cytokine Release and Biodistribution by CD3 Affinity Tuning. Sci. Rep. 2021, 11, 14397. [Google Scholar] [CrossRef] [PubMed]
- Mandikian, D.; Takahashi, N.; Lo, A.A.; Li, J.; Eastham-Anderson, J.; Slaga, D.; Ho, J.; Hristopoulos, M.; Clark, R.; Totpal, K.; et al. Relative Target Affinities of T-Cell–Dependent Bispecific Antibodies Determine Biodistribution in a Solid Tumor Mouse Model. Mol. Cancer Ther. 2018, 17, 776–785. [Google Scholar] [CrossRef] [PubMed]
- Staflin, K.; de Zafra, C.L.Z.; Schutt, L.K.; Clark, V.; Zhong, F.; Hristopoulos, M.; Clark, R.; Li, J.; Mathieu, M.; Chen, X.; et al. Target Arm Affinities Determine Preclinical Efficacy and Safety of Anti-HER2/CD3 Bispecific Antibody. JCI Insight 2020, 5, e133757. [Google Scholar] [CrossRef] [PubMed]
- Goebeler, M.-E.; Knop, S.; Viardot, A.; Kufer, P.; Topp, M.S.; Einsele, H.; Noppeney, R.; Hess, G.; Kallert, S.; Mackensen, A.; et al. Bispecific T-Cell Engager (BiTE) Antibody Construct Blinatumomab for the Treatment of Patients With Relapsed/Refractory Non-Hodgkin Lymphoma: Final Results From a Phase I Study. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2016, 34, 1104–1111. [Google Scholar] [CrossRef]
- Nathan, P.; Hassel, J.C.; Rutkowski, P.; Baurain, J.-F.; Butler, M.O.; Schlaak, M.; Sullivan, R.J.; Ochsenreither, S.; Dummer, R.; Kirkwood, J.M.; et al. Overall Survival Benefit with Tebentafusp in Metastatic Uveal Melanoma. N. Engl. J. Med. 2021, 385, 1196–1206. [Google Scholar] [CrossRef]
- Simão, D.C.; Zarrabi, K.K.; Mendes, J.L.; Luz, R.; Garcia, J.A.; Kelly, W.K.; Barata, P.C. Bispecific T-Cell Engagers Therapies in Solid Tumors: Focusing on Prostate Cancer. Cancers 2023, 15, 1412. [Google Scholar] [CrossRef]
- Baeuerle, P.A.; Wesche, H. T-Cell-Engaging Antibodies for the Treatment of Solid Tumors: Challenges and Opportunities. Curr. Opin. Oncol. 2022, 34, 552–558. [Google Scholar] [CrossRef]
- Tran, B.; Horvath, L.; Dorff, T.; Rettig, M.; Lolkema, M.P.; Machiels, J.-P.; Rottey, S.; Autio, K.; Greil, R.; Adra, N.; et al. 609O Results from a Phase I Study of AMG 160, a Half-Life Extended (HLE), PSMA-Targeted, Bispecific T-Cell Engager (BiTE®) Immune Therapy for Metastatic Castration-Resistant Prostate Cancer (mCRPC). Ann. Oncol. 2020, 31, S507. [Google Scholar] [CrossRef]
- Hummel, H.-D.; Kufer, P.; Grüllich, C.; Deschler-Baier, B.; Chatterjee, M.; Goebeler, M.-E.; Miller, K.; De Santis, M.; Loidl, W.C.; Buck, A.; et al. Phase I Study of Pasotuxizumab (AMG 212/BAY 2010112), a PSMA-Targeting BiTE (Bispecific T-Cell Engager) Immune Therapy for Metastatic Castration-Resistant Prostate Cancer (mCRPC). J. Clin. Oncol. 2020, 38, 124. [Google Scholar] [CrossRef]
- Bendell, J.C.; Fong, L.; Stein, M.N.; Beer, T.M.; Ross, A.; Gao, X.; Weitzman, A.; Austin, R.; Ganti, V.; Law, C.-L.; et al. First-in-Human Phase I Study of HPN424, a Tri-Specific Half-Life Extended PSMA-Targeting T-Cell Engager in Patients with Metastatic Castration-Resistant Prostate Cancer (mCRPC). J. Clin. Oncol. 2020, 38, 5552. [Google Scholar] [CrossRef]
- Pishvaian, M.J.; Morse, M.; McDevitt, J.T.; Ren, S.; Robbie, G.; Ryan, P.C.; Soukharev, S.; Bao, H.; Denlinger, C.S. Phase 1 Dose Escalation Study of MEDI-565, a Bispecific T-Cell Engager That Targets Human Carcinoembryonic Antigen (CEA), in Patients with Advanced Gastrointestinal (GI) Adenocarcinomas. J. Clin. Oncol. 2016, 34, 320. [Google Scholar] [CrossRef]
- Tabernero, J.; Melero, I.; Ros, W.; Argiles, G.; Marabelle, A.; Rodriguez-Ruiz, M.E.; Albanell, J.; Calvo, E.; Moreno, V.; Cleary, J.M.; et al. Phase Ia and Ib Studies of the Novel Carcinoembryonic Antigen (CEA) T-Cell Bispecific (CEA CD3 TCB) Antibody as a Single Agent and in Combination with Atezolizumab: Preliminary Efficacy and Safety in Patients with Metastatic Colorectal Cancer (mCRC). J. Clin. Oncol. 2017, 35, 3002. [Google Scholar] [CrossRef]
- Johnson, M.L.; Solomon, B.J.; Awad, M.M.; Cho, B.C.; Gainor, J.F.; Goldberg, S.B.; Keam, B.; Lee, D.H.; Huang, C.; Helms, H.-J.; et al. MORPHEUS: A Phase Ib/II Multi-Trial Platform Evaluating the Safety and Efficacy of Cancer Immunotherapy (CIT)-Based Combinations in Patients (Pts) with Non-Small Cell Lung Cancer (NSCLC). J. Clin. Oncol. 2018, 36, TPS9105. [Google Scholar] [CrossRef]
- Kebenko, M.; Goebeler, M.-E.; Wolf, M.; Hasenburg, A.; Seggewiss-Bernhardt, R.; Ritter, B.; Rautenberg, B.; Atanackovic, D.; Kratzer, A.; Rottman, J.B.; et al. A Multicenter Phase 1 Study of Solitomab (MT110, AMG 110), a Bispecific EpCAM/CD3 T-Cell Engager (BiTE®) Antibody Construct, in Patients with Refractory Solid Tumors. Oncoimmunology 2018, 7, e1450710. [Google Scholar] [CrossRef]
- Sternjak, A.; Lee, F.; Thomas, O.; Balazs, M.; Wahl, J.; Lorenczewski, G.; Ullrich, I.; Muenz, M.; Rattel, B.; Bailis, J.M.; et al. Preclinical Assessment of AMG 596, a Bispecific T-Cell Engager (BiTE) Immunotherapy Targeting the Tumor-Specific Antigen EGFRvIII. Mol. Cancer Ther. 2021, 20, 925–933. [Google Scholar] [CrossRef]
- Rosenthal, M.A.; Balana, C.; van Linde, M.E.; Sayehli, C.; Fiedler, W.M.; Wermke, M.; Massard, C.; Mellinghoff, I.K.; Khasraw, M.; Ang, A.; et al. ATIM-49 (LTBK-01). AMG 596, a novel anti-EGFRvIII bispecific T cell engager (BiTE®) molecule for the treatment of glioblastoma (GBM): Planned interim analysis in recurrent GBM (rGBM). Neuro-Oncol. 2019, 21, vi283. [Google Scholar] [CrossRef]
- Kast, J.; Bose, M.; Mehta, K.; Stienen, S.; Dutta, S.; Upreti, V. ACCP Abstract Booklet. Clin. Pharmacol. Drug Dev. 2020, 9, 1–75. [Google Scholar] [CrossRef]
- Schaller, T.H.; Foster, M.W.; Thompson, J.W.; Spasojevic, I.; Normantaite, D.; Moseley, M.A.; Sanchez-Perez, L.; Sampson, J.H. Pharmacokinetic Analysis of a Novel Human EGFRvIII:CD3 Bispecific Antibody in Plasma and Whole Blood Using a High-Resolution Targeted Mass Spectrometry Approach. J. Proteome Res. 2019, 18, 3032–3041. [Google Scholar] [CrossRef] [PubMed]
- Perales-Puchalt, A.; Duperret, E.K.; Yang, X.; Hernandez, P.; Wojtak, K.; Zhu, X.; Jung, S.-H.; Tello-Ruiz, E.; Wise, M.C.; Montaner, L.J.; et al. DNA-Encoded Bispecific T Cell Engagers and Antibodies Present Long-Term Antitumor Activity. JCI Insight 2019, 4, e126086. [Google Scholar] [CrossRef] [PubMed]
- Full Article: Clinical and Immune Responses to Anti-CD3 x Anti-EGFR Bispecific Antibody Armed Activated T Cells (EGFR BATs) in Pancreatic Cancer Patients. Available online: https://www.tandfonline.com/doi/full/10.1080/2162402X.2020.1773201 (accessed on 2 April 2023).
- Asano, K.; Nabeyama, A.; Miyake, Y.; Qiu, C.-H.; Kurita, A.; Tomura, M.; Kanagawa, O.; Fujii, S.; Tanaka, M. CD169-Positive Macrophages Dominate Antitumor Immunity by Crosspresenting Dead Cell-Associated Antigens. Immunity 2011, 34, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Heidbuechel, J.P.W.; Engeland, C.E. Oncolytic Viruses Encoding Bispecific T Cell Engagers: A Blueprint for Emerging Immunovirotherapies. J. Hematol. Oncol. 2021, 14, 63. [Google Scholar] [CrossRef] [PubMed]
- Arnone, C.M.; Polito, V.A.; Mastronuzzi, A.; Carai, A.; Diomedi, F.C.; Antonucci, L.; Petrilli, L.L.; Vinci, M.; Ferrari, F.; Salviato, E.; et al. Oncolytic Adenovirus and Gene Therapy with EphA2-BiTE for the Treatment of Pediatric High-Grade Gliomas. J. Immunother. Cancer 2021, 9, e001930. [Google Scholar] [CrossRef] [PubMed]
- Ruella, M.; Maus, M.V. Catch Me If You Can: Leukemia Escape after CD19-Directed T Cell Immunotherapies. Comput. Struct. Biotechnol. J. 2016, 14, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Huffman, A.P.; Lin, J.H.; Kim, S.I.; Byrne, K.T.; Vonderheide, R.H. CCL5 Mediates CD40-Driven CD4+ T Cell Tumor Infiltration and Immunity. JCI Insight 2020, 5, e137263. [Google Scholar] [CrossRef]
- Tian, L.; Xu, B.; Chen, Y.; Li, Z.; Wang, J.; Zhang, J.; Ma, R.; Cao, S.; Hu, W.; Chiocca, E.A.; et al. Specific Targeting of Glioblastoma with an Oncolytic Virus Expressing a Cetuximab-CCL5 Fusion Protein via Innate and Adaptive Immunity. Nat. Cancer 2022, 3, 1318–1335. [Google Scholar] [CrossRef]
- Bifunctional PD-1 × αCD3 × αCD33 Fusion Protein Reverses Adaptive Immune Escape in Acute Myeloid Leukemia-ScienceDirect. Available online: https://www-sciencedirect-com.myaccess.library.utoronto.ca/science/article/pii/S0006497120429544?via%3Dihub (accessed on 7 April 2023).
- Suntharalingam, G.; Perry, M.R.; Ward, S.; Brett, S.J.; Castello-Cortes, A.; Brunner, M.D.; Panoskaltsis, N. Cytokine Storm in a Phase 1 Trial of the Anti-CD28 Monoclonal Antibody TGN1412. N. Engl. J. Med. 2006, 355, 1018–1028. [Google Scholar] [CrossRef]
- Correnti, C.E.; Laszlo, G.S.; de van der Schueren, W.J.; Godwin, C.D.; Bandaranayake, A.; Busch, M.A.; Gudgeon, C.J.; Bates, O.M.; Olson, J.M.; Mehlin, C.; et al. Simultaneous Multiple Interaction T-Cell Engaging (SMITE) Bispecific Antibodies Overcome Bispecific T-Cell Engager (BiTE) Resistance via CD28 Co-Stimulation. Leukemia 2018, 32, 1239–1243. [Google Scholar] [CrossRef]
- Wu, L.; Seung, E.; Xu, L.; Rao, E.; Lord, D.M.; Wei, R.R.; Cortez-Retamozo, V.; Ospina, B.; Posternak, V.; Ulinski, G.; et al. Trispecific Antibodies Enhance the Therapeutic Efficacy of Tumor-Directed T Cells through T Cell Receptor Co-Stimulation. Nat. Cancer 2020, 1, 86–98. [Google Scholar] [CrossRef] [PubMed]
- Porter, C.E.; Shaw, A.R.; Jung, Y.; Yip, T.; Castro, P.D.; Sandulache, V.C.; Sikora, A.; Gottschalk, S.; Ittman, M.M.; Brenner, M.K.; et al. Oncolytic Adenovirus Armed with BiTE, Cytokine, and Checkpoint Inhibitor Enables CAR T Cells to Control the Growth of Heterogeneous Tumors. Mol. Ther. 2020, 28, 1251–1262. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, D.; Chandra, R.; Karalis, J.; Teke, M.; Aguilera, T.; Maddipati, R.; Wachsmann, M.B.; Ghersi, D.; Siravegna, G.; Zeh, H.J.; et al. Cancer-Associated Fibroblasts: Versatile Players in the Tumor Microenvironment. Cancers 2020, 12, 2652. [Google Scholar] [CrossRef] [PubMed]
- Simon, G.; Subbiah, V.; Rosen, L.; Lenz, H.-J.; Park, H.; Patel, M.; Miles, D.; Wallis, S.; Evilevitch, V.; Krige, D.; et al. 762 First-in-Human Phase 1a Study of NG-641, a Tumour-Selective Vector Expressing a FAP-TAc Bispecific Antibody and Immune Enhancer Module, in Patients with Metastatic/Advanced Epithelial Tumours (STAR). J. Immunother. Cancer 2022, 10, A794. [Google Scholar] [CrossRef]
- Scott, E.M.; Jacobus, E.J.; Lyons, B.; Frost, S.; Freedman, J.D.; Dyer, A.; Khalique, H.; Taverner, W.K.; Carr, A.; Champion, B.R.; et al. Bi- and Tri-Valent T Cell Engagers Deplete Tumour-Associated Macrophages in Cancer Patient Samples. J. Immunother. Cancer 2019, 7, 320. [Google Scholar] [CrossRef] [PubMed]
- Arvedson, T.; Bailis, J.M.; Britten, C.D.; Klinger, M.; Nagorsen, D.; Coxon, A.; Egen, J.G.; Martin, F. Targeting Solid Tumors with Bispecific T Cell Engager Immune Therapy. Annu. Rev. Cancer Biol. 2022, 6, 17–34. [Google Scholar] [CrossRef]
- Panchal, A.; Seto, P.; Wall, R.; Hillier, B.J.; Zhu, Y.; Krakow, J.; Datt, A.; Pongo, E.; Bagheri, A.; Chen, T.-H.T.; et al. COBRATM: A Highly Potent Conditionally Active T Cell Engager Engineered for the Treatment of Solid Tumors. mAbs 2020, 12, 1792130. [Google Scholar] [CrossRef]
- Lin, J.; Rocha, S.; Kwant, K.; Dayao, M.; Ng, T.; Aaron, W.; Callihan, E.; Gamez-Guerrero, M.; Hemmati, G.; Wright, K.; et al. 632 HPN601 Is a Protease-Activated EpCAM-Targeting T Cell Engager with an Improved Safety Profile for the Treatment of Solid Tumors. J. Immunother. Cancer 2020, 8, A379. [Google Scholar] [CrossRef]
- Revitope Oncology, Inc. Revitope—Third-Generation T Cell Engager Immunotherapies. Available online: https://www.nature.com/articles/d43747-021-00131-w (accessed on 6 April 2023).
- Cattaruzza, F.; Nazeer, A.; To, M.; Hammond, M.; Koski, C.; Liu, L.Y.; Pete Yeung, V.; Rennerfeldt, D.A.; Henkensiefken, A.; Fox, M.; et al. Precision-Activated T-Cell Engagers Targeting HER2 or EGFR and CD3 Mitigate on-Target, off-Tumor Toxicity for Immunotherapy in Solid Tumors. Nat. Cancer 2023, 4, 485–501. [Google Scholar] [CrossRef]
- Cattaruza, F.; Koski, C.; Nazeer, Å.; Lange, Z.; Henkensiefken, A.; Hammond, M.; Derynck, M.; Schellenberger, V.; Irving, B. HER2-XPAT, a Novel Protease-Activatable pro-Drug T Cell Engager (TCE), with Potent T-Cell Activation and Efficacy in Solid Tumors and Large Predicted Safety Margins in Non-Human Primate (NHP). Eur. J. Cancer 2020, 138, S10. [Google Scholar] [CrossRef]
- Deshaies, R.J. Multispecific Drugs Herald a New Era of Biopharmaceutical Innovation. Nature 2020, 580, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Bannas, P.; Hambach, J.; Koch-Nolte, F. Nanobodies and Nanobody-Based Human Heavy Chain Antibodies As Antitumor Therapeutics. Front. Immunol. 2017, 8, 1603. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.; van Dongen, G.A.M.S.; Walsum, M.S.; Roovers, R.C.; Stam, J.C.; Mali, W.; van Diest, P.J.; van Bergen en Henegouwen, P.M.P. Rapid Visualization of Human Tumor Xenografts through Optical Imaging with a Near-Infrared Fluorescent Anti–Epidermal Growth Factor Receptor Nanobody. Mol. Imaging 2012, 11, 7290-2011. [Google Scholar] [CrossRef]
- Schmitz, K.R.; Bagchi, A.; Roovers, R.C.; van Bergen en Henegouwen, P.M.P.; Ferguson, K.M. Structural Evaluation of EGFR Inhibition Mechanisms for Nanobodies/VHH Domains. Structure 2013, 21, 1214–1224. [Google Scholar] [CrossRef] [PubMed]
- Roovers, R.C.; Vosjan, M.J.W.D.; Laeremans, T.; el Khoulati, R.; de Bruin, R.C.G.; Ferguson, K.M.; Verkleij, A.J.; van Dongen, G.A.M.S.; van Bergen en Henegouwen, P.M.P. A Biparatopic Anti-EGFR Nanobody Efficiently Inhibits Solid Tumour Growth. Int. J. Cancer 2011, 129, 2013–2024. [Google Scholar] [CrossRef] [PubMed]
- Lamers, C.H.J.; Willemsen, R.; van Elzakker, P.; van Steenbergen-Langeveld, S.; Broertjes, M.; Oosterwijk-Wakka, J.; Oosterwijk, E.; Sleijfer, S.; Debets, R.; Gratama, J.W. Immune Responses to Transgene and Retroviral Vector in Patients Treated with Ex Vivo-Engineered T Cells. Blood 2011, 117, 72–82. [Google Scholar] [CrossRef]
- Ackaert, C.; Smiejkowska, N.; Xavier, C.; Sterckx, Y.G.J.; Denies, S.; Stijlemans, B.; Elkrim, Y.; Devoogdt, N.; Caveliers, V.; Lahoutte, T.; et al. Immunogenicity Risk Profile of Nanobodies. Front. Immunol. 2021, 12, 632687. [Google Scholar] [CrossRef]
- Xing, J.; Lin, L.; Li, J.; Liu, J.; Zhou, C.; Pan, H.; Shu, R.; Dong, B.; Cao, D.; Li, Q.; et al. BiHC, a T-Cell–Engaging Bispecific Recombinant Antibody, Has Potent Cytotoxic Activity Against Her2 Tumor Cells. Transl. Oncol. 2017, 10, 780–785. [Google Scholar] [CrossRef]
- Mølgaard, K.; Harwood, S.L.; Compte, M.; Merino, N.; Bonet, J.; Alvarez-Cienfuegos, A.; Mikkelsen, K.; Nuñez-Prado, N.; Alvarez-Mendez, A.; Sanz, L.; et al. Bispecific Light T-Cell Engagers for Gene-Based Immunotherapy of Epidermal Growth Factor Receptor (EGFR)-Positive Malignancies. Cancer Immunol. Immunother. 2018, 67, 1251–1260. [Google Scholar] [CrossRef]
- Harwood, S.L.; Alvarez-Cienfuegos, A.; Nuñez-Prado, N.; Compte, M.; Hernández-Pérez, S.; Merino, N.; Bonet, J.; Navarro, R.; Van Bergen en Henegouwen, P.M.P.; Lykkemark, S.; et al. ATTACK, a Novel Bispecific T Cell-Recruiting Antibody with Trivalent EGFR Binding and Monovalent CD3 Binding for Cancer Immunotherapy. OncoImmunology 2018, 7, e1377874. [Google Scholar] [CrossRef]
- Subklewe, M.; von Bergwelt-Baildon, M.; Humpe, A. Chimeric Antigen Receptor T Cells: A Race to Revolutionize Cancer Therapy. Transfus. Med. Hemotherapy 2019, 46, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Brocker, T.; Karjalainen, K. Signals through T Cell Receptor-Zeta Chain Alone Are Insufficient to Prime Resting T Lymphocytes. J. Exp. Med. 1995, 181, 1653–1659. [Google Scholar] [CrossRef] [PubMed]
- Chimeric Antigen Receptor–Modified T Cells in Chronic Lymphoid Leukemia-PMC. Available online: https://www-ncbi-nlm-nih-gov.myaccess.library.utoronto.ca/pmc/articles/PMC3387277/ (accessed on 12 April 2023).
- Gong, M.C.; Latouche, J.-B.; Krause, A.; Heston, W.D.; Bander, N.H.; Sadelain, M. Cancer Patient T Cells Genetically Targeted to Prostate-Specific Membrane Antigen Specifically Lyse Prostate Cancer Cells and Release Cytokines in Response to Prostate-Specific Membrane Antigen. Neoplasia 1999, 1, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Tomasik, J.; Jasiński, M.; Basak, G.W. Next Generations of CAR-T Cells-New Therapeutic Opportunities in Hematology? Front. Immunol. 2022, 13, 1034707. [Google Scholar] [CrossRef] [PubMed]
- Brentjens, R.J.; Davila, M.L.; Riviere, I.; Park, J.; Wang, X.; Cowell, L.G.; Bartido, S.; Stefanski, J.; Taylor, C.; Olszewska, M.; et al. CD19-Targeted T Cells Rapidly Induce Molecular Remissions in Adults with Chemotherapy-Refractory Acute Lymphoblastic Leukemia. Sci. Transl. Med. 2013, 5, 177ra38. [Google Scholar] [CrossRef] [PubMed]
- Silveira, C.R.F.; Corveloni, A.C.; Caruso, S.R.; Macêdo, N.A.; Brussolo, N.M.; Haddad, F.; Fernandes, T.R.; de Andrade, P.V.; Orellana, M.D.; Guerino-Cunha, R.L. Cytokines as an Important Player in the Context of CAR-T Cell Therapy for Cancer: Their Role in Tumor Immunomodulation, Manufacture, and Clinical Implications. Front. Immunol. 2022, 13, 947648. [Google Scholar] [CrossRef] [PubMed]
- Kagoya, Y.; Tanaka, S.; Guo, T.; Anczurowski, M.; Wang, C.-H.; Saso, K.; Butler, M.O.; Minden, M.D.; Hirano, N. A Novel Chimeric Antigen Receptor Containing a JAK-STAT Signaling Domain Mediates Superior Antitumor Effects. Nat. Med. 2018, 24, 352–359. [Google Scholar] [CrossRef]
- Yeku, O.O.; Purdon, T.J.; Koneru, M.; Spriggs, D.; Brentjens, R.J. Armored CAR T Cells Enhance Antitumor Efficacy and Overcome the Tumor Microenvironment. Sci. Rep. 2017, 7, 10541. [Google Scholar] [CrossRef]
- Moghanloo, E.; Mollanoori, H.; Talebi, M.; Pashangzadeh, S.; Faraji, F.; Hadjilooei, F.; Mahmoodzadeh, H. Remote Controlling of CAR-T Cells and Toxicity Management: Molecular Switches and next Generation CARs. Transl. Oncol. 2021, 14, 101070. [Google Scholar] [CrossRef]
- Wang, X.; Rivière, I. Clinical Manufacturing of CAR T Cells: Foundation of a Promising Therapy. Mol. Ther. Oncolytics 2016, 3, 16015. [Google Scholar] [CrossRef]
- Yaghoubi, S.S.; Jensen, M.C.; Satyamurthy, N.; Budhiraja, S.; Paik, D.; Czernin, J.; Gambhir, S.S. Noninvasive Detection of Therapeutic Cytolytic T Cells with 18 F-FHBG PET in a Patient with Glioma. Nat. Rev. Clin. Oncol. 2009, 6, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Kalos, M.; Levine, B.L.; Porter, D.L.; Katz, S.; Grupp, S.A.; Bagg, A.; June, C.H. T Cells with Chimeric Antigen Receptors Have Potent Antitumor Effects and Can Establish Memory in Patients with Advanced Leukemia. Sci. Transl. Med. 2011, 3, 95ra73. [Google Scholar] [CrossRef] [PubMed]
- Melenhorst, J.J.; Chen, G.M.; Wang, M.; Porter, D.L.; Chen, C.; Collins, M.A.; Gao, P.; Bandyopadhyay, S.; Sun, H.; Zhao, Z.; et al. Decade-Long Leukaemia Remissions with Persistence of CD4+ CAR T Cells. Nature 2022, 602, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Tantalo, D.G.M.; Oliver, A.J.; Von Scheidt, B.; Harrison, A.J.; Mueller, S.N.; Kershaw, M.H.; Slaney, C.Y. Understanding T Cell Phenotype for the Design of Effective Chimeric Antigen Receptor T Cell Therapies. J. Immunother. Cancer 2021, 9, e002555. [Google Scholar] [CrossRef] [PubMed]
- Arcangeli, S.; Bove, C.; Mezzanotte, C.; Camisa, B.; Falcone, L.; Manfredi, F.; Bezzecchi, E.; Khoury, R.E.; Norata, R.; Sanvito, F.; et al. CAR T Cell Manufacturing from Naive/Stem Memory T Lymphocytes Enhances Antitumor Responses While Curtailing Cytokine Release Syndrome. J. Clin. Investig. 2022, 132, e150807. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Chen, H.; Li, F.; Huang, S.; Chen, F.; Li, Y. Bright Future or Blind Alley? CAR-T Cell Therapy for Solid Tumors. Front. Immunol. 2023, 14, 1045024. [Google Scholar] [CrossRef] [PubMed]
- Durgin, J.S.; Henderson, F.; Nasrallah, M.P.; Mohan, S.; Wang, S.; Lacey, S.F.; Melenhorst, J.J.; Desai, A.S.; Lee, J.Y.K.; Maus, M.V.; et al. Case Report: Prolonged Survival Following EGFRvIII CAR T Cell Treatment for Recurrent Glioblastoma. Front. Oncol. 2021, 11, 669071. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhou, J.; Yang, X.; Liu, Y.; Zou, C.; Lv, W.; Chen, C.; Cheng, K.K.-Y.; Chen, T.; Chang, L.-J.; et al. Safety and Antitumor Activity of GD2-Specific 4SCAR-T Cells in Patients with Glioblastoma. Mol. Cancer 2023, 22, 3. [Google Scholar] [CrossRef]
- Dual-Target IL7Ra-Modified Autologous T Lymphocytes Correlated Inversely. in Recurrent Glioblastoma-Clinical Trials Registry-ICH GCP. Available online: https://ichgcp.net/pt/clinical-trials-registry/NCT05577091#google_vignette (accessed on 18 April 2023).
- Rutkowska, A.; Stoczynska-Fidelus, E.; Janik, K.; Wlodarczyk, A.; Rieske, P. EGFRvIII: An Oncogene with Ambiguous Role. J. Oncol. 2019, 2019, 1092587. [Google Scholar] [CrossRef]
- Rindopepimut with Temozolomide for Patients with Newly Diagnosed, EGFRvIII-Expressing Glioblastoma (ACT IV): A Randomised, Double-Blind, International Phase 3 Trial-ScienceDirect. Available online: https://www-sciencedirect-com.myaccess.library.utoronto.ca/science/article/pii/S147020451730517X (accessed on 16 April 2023).
- Morgan, R.A.; Yang, J.C.; Kitano, M.; Dudley, M.E.; Laurencot, C.M.; Rosenberg, S.A. Case Report of a Serious Adverse Event Following the Administration of T Cells Transduced with a Chimeric Antigen Receptor Recognizing ERBB2. Mol. Ther. J. Am. Soc. Gene Ther. 2010, 18, 843–851. [Google Scholar] [CrossRef]
- Pule, M.A.; Savoldo, B.; Myers, G.D.; Rossig, C.; Russell, H.V.; Dotti, G.; Huls, M.H.; Liu, E.; Gee, A.P.; Mei, Z.; et al. Virus-Specific T Cells Engineered to Coexpress Tumor-Specific Receptors: Persistence and Antitumor Activity in Individuals with Neuroblastoma. Nat. Med. 2008, 14, 1264–1270. [Google Scholar] [CrossRef] [PubMed]
- Schuessler, A.; Smith, C.; Beagley, L.; Boyle, G.M.; Rehan, S.; Matthews, K.; Jones, L.; Crough, T.; Dasari, V.; Klein, K.; et al. Autologous T-Cell Therapy for Cytomegalovirus as a Consolidative Treatment for Recurrent Glioblastoma. Cancer Res. 2014, 74, 3466–3476. [Google Scholar] [CrossRef] [PubMed]
- Louis, C.U.; Straathof, K.; Bollard, C.M.; Ennamuri, S.; Gerken, C.; Lopez, T.T.; Huls, M.H.; Sheehan, A.; Wu, M.-F.; Liu, H.; et al. Adoptive Transfer of EBV-Specific T Cells Results in Sustained Clinical Responses in Patients with Locoregional Nasopharyngeal Carcinoma. J. Immunother. (Hagerstown Md 1997) 2010, 33, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Leen, A.M.; Myers, G.D.; Sili, U.; Huls, M.H.; Weiss, H.; Leung, K.S.; Carrum, G.; Krance, R.A.; Chang, C.-C.; Molldrem, J.J.; et al. Monoculture-Derived T Lymphocytes Specific for Multiple Viruses Expand and Produce Clinically Relevant Effects in Immunocompromised Individuals. Nat. Med. 2006, 12, 1160–1166. [Google Scholar] [CrossRef] [PubMed]
- Heslop, H.E.; Slobod, K.S.; Pule, M.A.; Hale, G.A.; Rousseau, A.; Smith, C.A.; Bollard, C.M.; Liu, H.; Wu, M.-F.; Rochester, R.J.; et al. Long-Term Outcome of EBV-Specific T-Cell Infusions to Prevent or Treat EBV-Related Lymphoproliferative Disease in Transplant Recipients. Blood 2010, 115, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Donovan, L.K.; Delaidelli, A.; Joseph, S.K.; Bielamowicz, K.; Fousek, K.; Holgado, B.L.; Manno, A.; Srikanthan, D.; Gad, A.Z.; Van Ommeren, R.; et al. Locoregional Delivery of CAR T Cells to the Cerebrospinal Fluid for Treatment of Metastatic Medulloblastoma and Ependymoma. Nat. Med. 2020, 26, 720–731. [Google Scholar] [CrossRef] [PubMed]
- Theruvath, J.; Sotillo, E.; Mount, C.W.; Graef, C.M.; Delaidelli, A.; Heitzeneder, S.; Labanieh, L.; Dhingra, S.; Leruste, A.; Majzner, R.G.; et al. Locoregionally Administered B7-H3-Targeted CAR T Cells for Treatment of Atypical Teratoid/Rhabdoid Tumors. Nat. Med. 2020, 26, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Tumor-Derived Chemokine MCP-1/CCL2 Is Sufficient for Mediating Tumor Tropism of Adoptively Transferred T Cells-PubMed. Available online: https://pubmed-ncbi-nlm-nih-gov.myaccess.library.utoronto.ca/17709550/ (accessed on 18 April 2023).
- Fujita, M.; Zhu, X.; Ueda, R.; Sasaki, K.; Kohanbash, G.; Kastenhuber, E.R.; McDonald, H.A.; Gibson, G.A.; Watkins, S.C.; Muthuswamy, R.; et al. Effective Immunotherapy against Murine Gliomas Using Type 1 Polarizing Dendritic Cells--Significant Roles of CXCL10. Cancer Res. 2009, 69, 1587–1595. [Google Scholar] [CrossRef]
- Poly-ICLC Promotes the Infiltration of Effector T Cells into Intracranial Gliomas via Induction of CXCL10 in IFN-Alpha and IFN-Gamma Dependent Manners-PubMed. Available online: https://pubmed-ncbi-nlm-nih-gov.myaccess.library.utoronto.ca/20549206/ (accessed on 18 April 2023).
- Hsu, K.; Middlemiss, S.; Saletta, F.; Gottschalk, S.; McCowage, G.B.; Kramer, B. Chimeric Antigen Receptor-Modified T Cells Targeting EphA2 for the Immunotherapy of Paediatric Bone Tumours. Cancer Gene Ther. 2021, 28, 321–334. [Google Scholar] [CrossRef]
- Okada, H.; Weller, M.; Huang, R.; Finocchiaro, G.; Gilbert, M.R.; Wick, W.; Ellingson, B.M.; Hashimoto, N.; Pollack, I.F.; Brandes, A.A.; et al. Immunotherapy Response Assessment in Neuro-Oncology (iRANO): A Report of the RANO Working Group. Lancet Oncol. 2015, 16, e534–e542. [Google Scholar] [CrossRef]
- Wang, S.; O’Rourke, D.M.; Chawla, S.; Verma, G.; Nasrallah, M.P.; Morrissette, J.J.D.; Plesa, G.; June, C.H.; Brem, S.; Maloney, E.; et al. Multiparametric Magnetic Resonance Imaging in the Assessment of Anti-EGFRvIII Chimeric Antigen Receptor T Cell Therapy in Patients with Recurrent Glioblastoma. Br. J. Cancer 2019, 120, 54–56. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.K.; Pyo, J.; Suh, C.H.; Park, H.S.; Chae, Y.K.; Kim, K.W. Safety and Efficacy of Chimeric Antigen Receptor T-Cell Therapy for Glioblastoma: A Systemic Review and Meta-Analysis. Front. Oncol. 2022, 12, 851877. [Google Scholar] [CrossRef] [PubMed]
- Ghassemi, S.; Durgin, J.S.; Nunez-Cruz, S.; Patel, J.; Leferovich, J.; Pinzone, M.; Shen, F.; Cummins, K.D.; Plesa, G.; Cantu, V.A.; et al. Rapid Manufacturing of Non-Activated Potent CAR T Cells. Nat. Biomed. Eng. 2022, 6, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Newick, K.; Moon, E.; Albelda, S.M. Chimeric Antigen Receptor T-Cell Therapy for Solid Tumors. Mol. Ther. Oncolytics 2016, 3, 16006. [Google Scholar] [CrossRef] [PubMed]
- Treda, C.; Wlodarczyk, A.; Pacholczyk, M.; Rutkowska, A.; Stoczynska-Fidelus, E.; Kierasinska, A.; Rieske, P. Increased EGFRvIII Epitope Accessibility after Tyrosine Kinase Inhibitor Treatment of Glioblastoma Cells Creates More Opportunities for Immunotherapy. Int. J. Mol. Sci. 2023, 24, 4350. [Google Scholar] [CrossRef] [PubMed]
- Jamnani, F.R.; Rahbarizadeh, F.; Shokrgozar, M.A.; Mahboudi, F.; Ahmadvand, D.; Sharifzadeh, Z.; Parhamifar, L.; Moghimi, S.M. T Cells Expressing VHH-Directed Oligoclonal Chimeric HER2 Antigen Receptors: Towards Tumor-Directed Oligoclonal T Cell Therapy. Biochim. Biophys. Acta BBA-Gen. Subj. 2014, 1840, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Hegde, M.; Mukherjee, M.; Grada, Z.; Pignata, A.; Landi, D.; Navai, S.A.; Wakefield, A.; Fousek, K.; Bielamowicz, K.; Chow, K.K.H.; et al. Tandem CAR T Cells Targeting HER2 and IL13Ralpha2 Mitigate Tumor Antigen Escape. J. Clin. Investig. 2016, 126, 3036–3052. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Tang, X.; Zhang, Z.; Gu, L.; Wei, H.; Zhao, S.; Zhong, K.; Mu, M.; Huang, C.; Jiang, C.; et al. Tandem CAR-T Cells Targeting CD70 and B7-H3 Exhibit Potent Preclinical Activity against Multiple Solid Tumors. Theranostics 2020, 10, 7622–7634. [Google Scholar] [CrossRef]
- Schmidts, A.; Srivastava, A.A.; Ramapriyan, R.; Bailey, S.R.; Bouffard, A.A.; Cahill, D.P.; Carter, B.S.; Curry, W.T.; Dunn, G.P.; Frigault, M.J.; et al. Tandem Chimeric Antigen Receptor (CAR) T Cells Targeting EGFRvIII and IL-13Rα2 Are Effective against Heterogeneous Glioblastoma. Neuro-Oncol. Adv. 2023, 5, vdac185. [Google Scholar] [CrossRef]
- Muhammad, N.; Wang, R.; Li, W.; Zhang, Z.; Chang, Y.; Hu, Y.; Zhao, J.; Zheng, X.; Mao, Q.; Xia, H. A Novel TanCAR Targeting IL13Ralpha2 and EphA2 for Enhanced Glioblastoma Therapy. Mol. Ther.-Oncolytics 2022, 24, 729–741. [Google Scholar] [CrossRef]
- Bielamowicz, K.; Fousek, K.; Byrd, T.T.; Samaha, H.; Mukherjee, M.; Aware, N.; Wu, M.-F.; Orange, J.S.; Sumazin, P.; Man, T.-K.; et al. Trivalent CAR T Cells Overcome Interpatient Antigenic Variability in Glioblastoma. Neuro-Oncol. 2018, 20, 506–518. [Google Scholar] [CrossRef] [PubMed]
- Bos, T.J.; De Bruyne, E.; Van Lint, S.; Heirman, C.; Vanderkerken, K. Large Double Copy Vectors Are Functional but Show a Size-Dependent Decline in Transduction Efficiency. J. Biotechnol. 2010, 150, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.; Li, J.; Sun, C.; Gao, K.; Hirabayashi, K.; Li, H.; Savoldo, B.; Liu, R.; Dotti, G. Cancer Immunotherapy with T Cells Carrying Bispecific Receptors That Mimic Antibodies. Cancer Immunol. Res. 2019, 7, 773–783. [Google Scholar] [CrossRef] [PubMed]
- Choe, J.H.; Watchmaker, P.B.; Simic, M.S.; Gilbert, R.D.; Li, A.W.; Krasnow, N.A.; Downey, K.M.; Yu, W.; Carrera, D.A.; Celli, A.; et al. SynNotch-CAR T Cells Overcome Challenges of Specificity, Heterogeneity, and Persistence in Treating Glioblastoma. Sci. Transl. Med. 2021, 13, eabe7378. [Google Scholar] [CrossRef] [PubMed]
- Song, E.Z.; Wang, X.; Philipson, B.I.; Zhang, Q.; Thokala, R.; Zhang, L.; Assenmacher, C.-A.; Binder, Z.A.; Ming, G.-L.; O’Rourke, D.M.; et al. The IAP Antagonist Birinapant Enhances Chimeric Antigen Receptor T Cell Therapy for Glioblastoma by Overcoming Antigen Heterogeneity. Mol. Ther.-Oncolytics 2022, 27, 288–304. [Google Scholar] [CrossRef] [PubMed]
- Suryadevara, C.M.; Desai, R.; Abel, M.L.; Riccione, K.A.; Batich, K.A.; Shen, S.H.; Chongsathidkiet, P.; Gedeon, P.C.; Elsamadicy, A.A.; Snyder, D.J.; et al. Temozolomide Lymphodepletion Enhances CAR Abundance and Correlates with Antitumor Efficacy against Established Glioblastoma. OncoImmunology 2018, 7, e1434464. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, Z.; Zhong, K.; Wang, Z.; Yang, N.; Tang, X.; Li, H.; Lu, Q.; Wu, Z.; Yuan, B.; et al. CXCL11-Armed Oncolytic Adenoviruses Enhance CAR-T Cells Therapeutic Efficacy and Reprogram Tumor Microenvironment in Glioblastoma. Mol. Ther. J. Am. Soc. Gene Ther. 2022. [Google Scholar] [CrossRef] [PubMed]
- Daneshimehr, F.; Barabadi, Z.; Abdolahi, S.; Soleimani, M.; Verdi, J.; Ebrahimi-Barough, S.; Ai, J. Angiogenesis and Its Targeting in Glioblastoma with Focus on Clinical Approaches. Cell J. 2022, 24, 555–568. [Google Scholar] [CrossRef] [PubMed]
- Conlon, K.C.; Miljkovic, M.D.; Waldmann, T.A. Cytokines in the Treatment of Cancer. J. Interferon Cytokine Res. 2019, 39, 6–21. [Google Scholar] [CrossRef]
- Agliardi, G.; Liuzzi, A.R.; Hotblack, A.; De Feo, D.; Nunez, N.; Stowe, C.L.; Friebel, E.; Nannini, F.; Rindlisbacher, L.; Roberts, T.A.; et al. Intratumoral IL-12 Delivery Empowers CAR-T Cell Immunotherapy in a Pre-Clinical Model of Glioblastoma. Nat. Commun. 2021, 12, 444. [Google Scholar] [CrossRef]
- Swan, S.L.; Mehta, N.; Ilich, E.; Shen, S.H.; Wilkinson, D.S.; Anderson, A.R.; Segura, T.; Sanchez-Perez, L.; Sampson, J.H.; Bellamkonda, R.V. IL7 and IL7 Flt3L Co-Expressing CAR T Cells Improve Therapeutic Efficacy in Mouse EGFRvIII Heterogeneous Glioblastoma. Front. Immunol. 2023, 14, 1085547. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zheng, M.; Zhang, Z.; Tang, X.; Chen, Y.; Peng, A.; Peng, X.; Tong, A.; Zhou, L. Interleukin-7-Loaded Oncolytic Adenovirus Improves CAR-T Cell Therapy for Glioblastoma. Cancer Immunol. Immunother. 2021, 70, 2453–2465. [Google Scholar] [CrossRef] [PubMed]
- Shum, T.; Omer, B.; Tashiro, H.; Kruse, R.L.; Wagner, D.L.; Parikh, K.; Yi, Z.; Sauer, T.; Liu, D.; Parihar, R.; et al. Constitutive Signaling from an Engineered IL7 Receptor Promotes Durable Tumor Elimination by Tumor-Redirected T Cells. Cancer Discov. 2017, 7, 1238–1247. [Google Scholar] [CrossRef] [PubMed]
- Wick, A.; Desjardins, A.; Suarez, C.; Forsyth, P.; Gueorguieva, I.; Burkholder, T.; Cleverly, A.L.; Estrem, S.T.; Wang, S.; Lahn, M.M.; et al. Phase 1b/2a Study of Galunisertib, a Small Molecule Inhibitor of Transforming Growth Factor-Beta Receptor I, in Combination with Standard Temozolomide-Based Radiochemotherapy in Patients with Newly Diagnosed Malignant Glioma. Investig. New Drugs 2020, 38, 1570–1579. [Google Scholar] [CrossRef]
- Capper, D.; von Deimling, A.; Brandes, A.A.; Carpentier, A.F.; Kesari, S.; Sepulveda-Sanchez, J.M.; Wheeler, H.R.; Chinot, O.; Cher, L.; Steinbach, J.P.; et al. Biomarker and Histopathology Evaluation of Patients with Recurrent Glioblastoma Treated with Galunisertib, Lomustine, or the Combination of Galunisertib and Lomustine. Int. J. Mol. Sci. 2017, 18, 995. [Google Scholar] [CrossRef] [PubMed]
- Narayan, V.; Barber-Rotenberg, J.S.; Jung, I.-Y.; Lacey, S.F.; Rech, A.J.; Davis, M.M.; Hwang, W.-T.; Lal, P.; Carpenter, E.L.; Maude, S.L.; et al. PSMA-Targeting TGFβ-Insensitive Armored CAR T-Cells in Metastatic Castration Resistant Prostate Cancer: A Phase 1 Trial. Nat. Med. 2022, 28, 724–734. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Liu, L.; Su, H.; Liu, Q.; Shen, J.; Dai, H.; Zheng, W.; Lu, Y.; Zhang, W.; Bei, Y.; et al. Chimeric Antigen Receptor Macrophage Therapy for Breast Tumours Mediated by Targeting the Tumour Extracellular Matrix. Br. J. Cancer 2019, 121, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.J.; Dougan, M.; Jailkhani, N.; Ingram, J.; Fang, T.; Kummer, L.; Momin, N.; Pishesha, N.; Rickelt, S.; Hynes, R.O.; et al. Nanobody-Based CAR T Cells That Target the Tumor Microenvironment Inhibit the Growth of Solid Tumors in Immunocompetent Mice. Proc. Natl. Acad. Sci. USA 2019, 116, 7624–7631. [Google Scholar] [CrossRef]
- Xie, Y.J.; Dougan, M.; Ingram, J.R.; Pishesha, N.; Fang, T.; Momin, N.; Ploegh, H.L. Improved Antitumor Efficacy of Chimeric Antigen Receptor T Cells That Secrete Single-Domain Antibody Fragments. Cancer Immunol. Res. 2020, 8, 518–529. [Google Scholar] [CrossRef]
- Yin, X.; He, L.; Guo, Z. T-Cell Exhaustion in CAR-T-Cell Therapy and Strategies to Overcome It. Immunology 2023, 169, 400–411. [Google Scholar] [CrossRef]
- Long, A.H.; Haso, W.M.; Shern, J.F.; Wanhainen, K.M.; Murgai, M.; Ingaramo, M.; Smith, J.P.; Walker, A.J.; Kohler, M.E.; Venkateshwara, V.R.; et al. 4-1BB Costimulation Ameliorates T Cell Exhaustion Induced by Tonic Signaling of Chimeric Antigen Receptors. Nat. Med. 2015, 21, 581–590. [Google Scholar] [CrossRef]
- Weber, E.W.; Parker, K.R.; Sotillo, E.; Lynn, R.C.; Anbunathan, H.; Lattin, J.; Good, Z.; Belk, J.A.; Daniel, B.; Klysz, D.; et al. Transient Rest Restores Functionality in Exhausted CAR-T Cells through Epigenetic Remodeling. Science 2021, 372, eaba1786. [Google Scholar] [CrossRef] [PubMed]
- Beavis, P.A.; Henderson, M.A.; Giuffrida, L.; Mills, J.K.; Sek, K.; Cross, R.S.; Davenport, A.J.; John, L.B.; Mardiana, S.; Slaney, C.Y.; et al. Targeting the Adenosine 2A Receptor Enhances Chimeric Antigen Receptor T Cell Efficacy. J. Clin. Investig. 2017, 127, 929–941. [Google Scholar] [CrossRef] [PubMed]
- Sitkovsky, M.V.; Hatfield, S.; Abbott, R.; Belikoff, B.; Lukashev, D.; Ohta, A. Hostile, Hypoxia-A2-Adenosinergic Tumor Biology as the Next Barrier to the Tumor Immunologists. Cancer Immunol. Res. 2014, 2, 598–605. [Google Scholar] [CrossRef]
- Giuffrida, L.; Sek, K.; Henderson, M.A.; Lai, J.; Chen, A.X.Y.; Meyran, D.; Todd, K.L.; Petley, E.V.; Mardiana, S.; Mølck, C.; et al. CRISPR/Cas9 Mediated Deletion of the Adenosine A2A Receptor Enhances CAR T Cell Efficacy. Nat. Commun. 2021, 12, 3236. [Google Scholar] [CrossRef] [PubMed]
- Ligtenberg, M.A.; Mougiakakos, D.; Mukhopadhyay, M.; Witt, K.; Lladser, A.; Chmielewski, M.; Riet, T.; Abken, H.; Kiessling, R. Coexpressed Catalase Protects Chimeric Antigen Receptor–Redirected T Cells as Well as Bystander Cells from Oxidative Stress–Induced Loss of Antitumor Activity. J. Immunol. Author Choice 2016, 196, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Seo, H.; Chen, J.; González-Avalos, E.; Samaniego-Castruita, D.; Das, A.; Wang, Y.H.; López-Moyado, I.F.; Georges, R.O.; Zhang, W.; Onodera, A.; et al. TOX and TOX2 Transcription Factors Cooperate with NR4A Transcription Factors to Impose CD8+ T Cell Exhaustion. Proc. Natl. Acad. Sci. USA 2019, 116, 12410–12415. [Google Scholar] [CrossRef]
- Yin, Y.; Boesteanu, A.C.; Binder, Z.A.; Xu, C.; Reid, R.A.; Rodriguez, J.L.; Cook, D.R.; Thokala, R.; Blouch, K.; McGettigan-Croce, B.; et al. Checkpoint Blockade Reverses Anergy in IL-13Ralpha2 Humanized scFv-Based CAR T Cells to Treat Murine and Canine Gliomas. Mol. Ther. Oncolytics 2018, 11, 20–38. [Google Scholar] [CrossRef]
- Song, Y.; Liu, Q.; Zuo, T.; Wei, G.; Jiao, S. Combined Antitumor Effects of Anti-EGFR Variant III CAR-T Cell Therapy and PD-1 Checkpoint Blockade on Glioblastoma in Mouse Model. Cell. Immunol. 2020, 352, 104112. [Google Scholar] [CrossRef]
- Conroy, M.; Naidoo, J. Immune-Related Adverse Events and the Balancing Act of Immunotherapy. Nat. Commun. 2022, 13, 392. [Google Scholar] [CrossRef]
- Liu, X.; Ranganathan, R.; Jiang, S.; Fang, C.; Sun, J.; Kim, S.; Newick, K.; Lo, A.; June, C.H.; Zhao, Y.; et al. A Chimeric Switch-Receptor Targeting PD1 Augments the Efficacy of Second-Generation CAR T Cells in Advanced Solid Tumors. Cancer Res. 2016, 76, 1578–1590. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; He, X.; Zhang, B.; Guo, F.; Ou, X.; Yang, Q.; Shu, P.; Chen, Y.; Li, K.; Gao, G.; et al. A PD-L1-Targeting Chimeric Switch Receptor Enhances Efficacy of CAR-T Cell for Pleural and Peritoneal Metastasis. Signal Transduct. Target. Ther. 2022, 7, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Wang, J. Therapeutic Effect of Dual CAR-T Targeting PDL1 and MUC16 Antigens on Ovarian Cancer Cells in Mice. BMC Cancer 2020, 20, 678. [Google Scholar] [CrossRef] [PubMed]
- Santomasso, B.; Bachier, C.; Westin, J.; Rezvani, K.; Shpall, E.J. The Other Side of CAR T-Cell Therapy: Cytokine Release Syndrome, Neurologic Toxicity, and Financial Burden. Am. Soc. Clin. Oncol. Educ. Book 2019, 39, 433–444. [Google Scholar] [CrossRef] [PubMed]
- McComb, S.; Nguyen, T.; Shepherd, A.; Henry, K.A.; Bloemberg, D.; Marcil, A.; Maclean, S.; Zafer, A.; Gilbert, R.; Gadoury, C.; et al. Programmable Attenuation of Antigenic Sensitivity for a Nanobody-Based EGFR Chimeric Antigen Receptor Through Hinge Domain Truncation. Front. Immunol. 2022, 13, 864868. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Rodriguez, J.L.; Li, N.; Thokala, R.; Nasrallah, M.P.; Hu, L.; Zhang, L.; Zhang, J.V.; Logun, M.T.; Kainth, D.; et al. Locally Secreted BiTEs Complement CAR T Cells by Enhancing Killing of Antigen Heterogeneous Solid Tumors. Mol. Ther. 2022, 30, 2537–2553. [Google Scholar] [CrossRef] [PubMed]
- Liao, Q.; He, H.; Mao, Y.; Ding, X.; Zhang, X.; Xu, J. Engineering T Cells with Hypoxia-Inducible Chimeric Antigen Receptor (HiCAR) for Selective Tumor Killing. Biomark. Res. 2020, 8, 56. [Google Scholar] [CrossRef] [PubMed]
- Labanieh, L.; Majzner, R.G.; Klysz, D.; Sotillo, E.; Fisher, C.J.; Vilches-Moure, J.G.; Pacheco, K.Z.B.; Malipatlolla, M.; Xu, P.; Hui, J.H.; et al. Enhanced Safety and Efficacy of Protease-Regulated CAR-T Cell Receptors. Cell 2022, 185, 1745–1763.e22. [Google Scholar] [CrossRef]
- Celichowski, P.; Turi, M.; Charvátová, S.; Radhakrishnan, D.; Feizi, N.; Chyra, Z.; Šimíček, M.; Jelínek, T.; Bago, J.R.; Hájek, R.; et al. Tuning CARs: Recent Advances in Modulating Chimeric Antigen Receptor (CAR) T Cell Activity for Improved Safety, Efficacy, and Flexibility. J. Transl. Med. 2023, 21, 197. [Google Scholar] [CrossRef]
- Liu, D.; Zhao, J.; Song, Y. Engineering Switchable and Programmable Universal CARs for CAR T Therapy. J. Hematol. Oncol. 2019, 12, 69. [Google Scholar] [CrossRef]



| Antigen | Properties | BsAbs | CART | |
|---|---|---|---|---|
| Preclinical | Clinical | |||
| EGFR |
|
| ||
| EGFRvIII |
| Clinical:
| ||
| IL13Rα2 |
| |||
| Fn14 |
|
|
|
|
| CD133 |
|
|
|
|
| CA9 |
|
|
|
|
| HER2 |
|
| ||
| EphA2 |
|
|
| |
| B7-H3 |
|
|
| |
| GD2 |
|
|
| |
| Chlorotoxin/MMP2 |
|
|
|
|
| CD147 |
|
|
|
|
| CD70 |
|
|
|
|
| NKG2D |
|
|
| |
| Podoplanin |
|
|
|
|
| Population | Intervention | Outcome | Toxicity | Other Findings | ||
|---|---|---|---|---|---|---|
| IL13Ra2 | ||||||
| Brown et al., 2015 [18] (NCT00730613) | 3 patients with recurrent, unifocal, resectable grade 3 or 4 glioma | CAR: 1st generation (IL13-zetakine) Route: Repeated IC injection following resection Dosing: 107 to 108 in 12 doses over 5 weeks LD: no | Mean survival 11 mo Longest survival 14 mo | Grade 3 neurological A/E at 108 1 pt with 2 headache episodes 1 pt with tongue deviation and gait disturbance | Transient MRI worsening correlated with highest antigen expression Transient response Antigen loss Time to manufacture limited enrollment Persistence: up to 14 weeks | |
| Brown et al., 2016 [69] (NCT02208362) | 1 patient with MGMT unmethylated GBM and multifocal/LM recurrence after standard Rx and Infigratinib trial | CAR: 2nd generation, 4-1BB, enriched for TCM Route: IC + IVT Dosing: 5 IC infusions of 2 mil then 10 mil cells followed by 10 additional IVT treatments for recurrence LD: no | Stability of treated tumor cavity Decrease of 77–100% in lesions’ size after 5th IVT Rx Response maintained for 7.5 months from the first Rx | Grade 2, within 72 h: headache, fatigue, myalgia, olfactory aura | Antigen loss CAR T detected in CSF at all points but not in PB Decreased number of CAR T cells with decreasing tumor burden Increase in endogenous cells; recruitment of immune system Cytokines increase by 10-fold correlating with A/E. | |
| Brown et al., 2022 [70] (NCT01082926) | 6 patients with non-resectable recurrent grade 3 or 4 on steroids | CAR: Glucocorticoid receptor negative allogenic IL13 zetakine CTLs infused Route: IC Dosing: 4 cycles, twice weekly × 2 weeks, followed by IC IL-12 d2-5 and then d1-5 LD: no | Median OS 2.9 mo Longest 11.5 mo | Grade 1: injection site reaction, fever Grade 1/2: H/A, confusion, fatigue, tachycardia, distant stroke 2 weeks later | Manufacturing: 5 months Use of dexamethasone did not decrease CAR cytotoxicity but may have abrogated endogenous response | |
| Ongoing | Criteria | Intervention | Status | Start | Completion | |
| NCT02208362 City of Hope | 82 pts with Recurrent/Refractory any grade glioma | CAR: Autologous IL13(EQ)BBzeta/CD19t+ TCM-enriched or naïve/memory (TN/MEM) T Cells Route: ITu, IC, IVT, or 2 locations | May 2018 | June 2023 | ||
| NCT04661384 City of Hope | Leptomeningeal mets from Ependymoma, Glioblastoma, or Medulloblastoma | CAR: Autologous IL13Ralpha2-specific Hinge-optimized 41BB-co-stimulatory Route: IVT q1 week × 4 | Recruiting | December 2020 | November 2025 | |
| NCT04003649 City of Hope | Recurrent glioblastoma | CAR: Autologous IL13Ralpha2-specific Hinge-optimized 4-1BB-co-stimulatory With or without nivolumab + ipilimumab or nivolumab alone | Recruiting | December 2019 | November 2023 | |
| NCT05540873 (MAGIC-I) CellabMED | Recurrent or refractory grade 3 or 4 glioma | CAR: IL13Ra2 CART Route: IV | Recruiting | July 2022 | April 2024 | |
| EGFRvIII | ||||||
| O’Rourke et al., 2017 [57] (NCT02209376) Terminated to pursue combination Rx | 10 pts with recurrent GBM | CAR: CART-EGFRvIII, 2nd-generation, 4-1BB costimulatory domain Route: IV, single infusion LD: no | Median OS 10 mo | No DLT, no CRS 3 pt received Siltuximab for neuro Sx | Antigen loss, heterogeneity Increase immune suppression in the TME following infusion CAR T cells present up to 2 mo T-cell expansion and infiltration in some areas of tumor | |
| Goff et al., 2019 [58] (NCT01454596) | 18 pts with recurrent GBM | CAR: 3rd-generation CART Route: IV Dosing: 6.3 mil to 23 bil cells LD: yes +Post-infusion IL-2 | Median PFS 1.3 mo, one outlier at 12.5 mo Median OS 6.9 mo, 1 pt alive at 59 mo, 2 for 13 mo No OR’s defined by serial MRI | 1 mortality at highest dose 2 respiratory symptoms Grade 2 neurological A/E in 10 pts | Median time between biopsy proving EGFRvIII+ and infusion was 11 mo Dose correlated with persistence but not survival CAR T present at 3 mo | |
| Durgin et al., 2021 [174] (NCT02209376) | 1 pt | See O’Rourke et al. [57] above | OS 34 mo | Post-infusion D7: flu-like symptoms, including arthralgia, myalgia, and headache | CAR T persistence > 29 mo | |
| Ongoing | Criteria | Intervention | Status | Start | Completion | |
| NCT03296696 Amgen | 30 pts with newly diagnosed or recurrent GBM | TCE: AMG 596—EGFRvIII × CD3 BiTE +/− Pembrolizumab Route/dosing: IV continuous | Completed | August 2018 | August 2021 | |
| NCT04903795 Duke University | 18 pts with newly diagnosed GBM or first recurrence | TCE: hEGFRvIII-CD3 Bi-scFv Dosing: 57.0 ng/kg, 570.0 ng/kg, 5700.0 ng/kg, and 57,000.0 ng/kg. | Not yet Recruiting | August 2023 | December 2023 | |
| NCT05187624 Hoffman-LaRoche | Est 200 pts with newly diagnosed GBM | TCE: RO7428731—EGFRvIIIxCD3 IgG-like Route/dosing: IV Q3 weeks | Recruiting | April 2022 | February 2025 | |
| NCT03726515 University of Pennsylvania | 7 pts with newly diagnosed MGMT-unmethylated GBM | CAR: 2nd-generation, 4-1BB costimulation with Pembrolizumab | Completed—no results | March 2019 | February 2021 | |
| NCT05063682 (CARTREMENDOUS) Chembrain LTD | 10 pts Leptomeningeal disease from EGFRvIII+ GBM | CAR: 2nd-generation EGFRvIII-specific hinge-optimized CD3 ζ-stimulatory/41BB-co-stimulatory Route: IVT x1 +/− additional cycles | Active, not recruiting | May 2020 | October 2023 | |
| NCT03283631 (INTERCEPT) Duke Uni./NCI | 2 pts with recurrent glioblastoma | CAR: EGFRvIII CAR T cells radiolabeled with 111Indium (111In) Route: ITu delivery by CED post-SRS. SPECT on day 1 and 2 to visualize cells Dosing: 250 mil cells | Suspended April 2020 to amend for enrollment of fewer pts Terminated June 2021 to shift to next iteration of a CAR T cell | May 2018 | June 2020 | |
| NCT02844062 Beijing Sanbo Brain Hospital | Est. 10 pts | CAR: EGFRvIII CAR T cells with truncated EGFR (for tracking/ablation) Route: IV LD: yes | Unknown | July 2016 | July 2019 Last updated July 2016 | |
| NCT02664363 (ExCeL) Duke University | 3 pts | CAR: EGFRvIII CAR radiolabeled with 111Indium Dosing: after resection, SOC and up to 3 cycles of dose-intensified TMZ | Terminated—study funding ended | February 2017 | September 2019 | |
| NCT05660369 (INCIPIENT) Massachusetts General Hospital | Est. 21 pts with newly diagnosed or recurrent glioblastoma, supratentorial | CAR: CARv3-TEAM-E; T cells transduced with lentiviral vector to express EGFRvIII-CAR and EGFRwt-TEAM Route: IVT (Ommaya reservoir) Dosing: Safety run of one infusion then dose escalation × 3 arms, weekly infusions × 6 | Recruiting | March 2023 | June 2026 | |
| NCT05024175 Massachusetts General Hospital | Est. 18 pts who have completed 24 mo since CARv3-TEAM-E T-cell infusion or <24 mo if they discontinued due to progression or other | Observational study Long-term safety and efficacy of CARv3-TEAM-E T-cell therapy | Not yet recruiting | December 2021 | August 2039 | |
| NCT05802693 Beijing Tsinghua Chang Gung Hospital | Est 22 pts with recurrent glioblastoma | Ommaya reservoir | Not yet recruiting | April 2023 | April 2025 | |
| HER2 | ||||||
| Ahmed et al., 2017 [81] (NCT01109095) | 17 adult and pediatric pts with rGBM, CMV seropositive | CAR: Autologous 2nd-generation (CD28) HER 2 CAR VST Route: IV Dosing: Dose escalation up to 1 × 108 cells/m2, up to 6 doses at 6–12 week intervals at the same dose level LD: no | Median OS 11.1 mo post-CART 3 pts stable at 29, 28.8 and 24 mo. 18 m-survival 29.4% For 7 pts who failed only 1st line Rx: Median OS 27.2 mo 18 m-survival 43% | No DLTs Grade 2 headache/seizure in 2 pts | Median time to infusion 12.5 months 59% had failed 1–5 lines of Rx other than initial SOC No expansion of CAR T but persistence up to 1 year MRI inflammatory responses mimicking progression Survival correlated with lack of previous salvage therapy | |
| Vitanza et al., 2021 [82] (NCT03500991) | 3 pts aged 15–26 with recurrent CNS tumors (1 grade 3 astrocytoma, 2 ependymoma) | CAR: 2nd-generation (4-1BB) Route: IC or IVT Dosing: 3 doses per month for a max of 18 doses, dose 10 mil to 25 mil LD: no | PD in 2/3 pts | No DLTs H/A, pain at spinal met site, worsening neurologic deficit IV route: fever | High CRP with symptoms | |
| Ongoing | Criteria/enrollment | CAR and administration | Status | Start | Completion | |
| NCT03389230 City of Hope | Est. 42 pts with recurrent/refractory grade 3 or 4 glioma | CAR: 2nd gen. HER2-Specific, Hinge-Optimized, 41BB-Costimulatory and Truncated CD19 Route/dosing: IC or ITu or both weekly for 3 weeks | Recruiting | August 2018 | December 2023 | |
| EphA2 | ||||||
| Lin et al., 2021 [87] (NCT03423992) | 3 pts (initial cohort) | CAR: 2nd gen EphA2-CAR, 4-1BB costimulatory domain and truncated EGFR Route: IV Dosing: single infusion, starting at 1 mil cells/kg LD: yes (Flu/cyclo) | SD in 1 pt, PD in 2 pts OS 81-186d | Grade 2 CRS and pulmonary edema in 2/3 patients | Expansion of CAR T cells peripherally persisting for 28d+, with peak at 7–10 d | |
| NKG2D | ||||||
| Ongoing | Criteria/enrollment | CAR and administration | Status | Start | Completion | |
| NCT05131763 Fudan University | Est 3 pts with relapsed HCC, GBM, medulloblastoma, or colon Ca | CAR: 2nd gen CAR with 4-1BB costim Route: IV | Recruiting | March 2021 | December 2023 | |
| NCT04717999 UWELL Biopharma | 20 pts with 1st or 2nd GBM relapse | NKG2D CAR-T Route: Ommaya reservoir | Not yet recruiting | September 2021 | December 2023 | |
| B7-H3 | ||||||
| Tang et al., 2021 [92] | 1 patient with rGBM and 50% heterogeneous B7-H3 expression | CAR: B7-H3 CAR-T Route: IC Dosing: 4 mil to 20 mil cells Weekly | PR after first cycle Symptomatic and MRI PD after cycle 6 | No DLT Grade 2 headache related to infusions worse in the first 4 cycles | Expansion of T cells and CART in the CSF, peak cycle 3, and decline in later cycles | |
| Ongoing | Criteria/enrollment | CAR and administration | Status | Start | Completion | |
| NCT04385173 Zhejiang University | 12 pts with recurrent or refractory GBM | Route: ITu, IVT Dosing: 3 injections, 1–2 week intervals In between temozolomide cycles | Recruiting | December 2022 | May 2024 | |
| NCT04077866 Zhejiang University | 40 pts with recurrent or refractory GBM | 2 arms, B7H3 CART + temozolomide vs. temozolomide alone Route: ITu, IVT Dosing: 3 injections, 1–2 week intervals In between temozolomide cycles | Recruiting | June 2023 | August 2025 | |
| GD2 | ||||||
| Liu et al., 2023 [175] NCT03170141 | 8 adult and pediatric pts with recurrent IDH-WT, MGMT unmethylated, GBM | CAR: 4th gen CAR-T cells (CD28 transmembrane and cytoplasmic domains, co-stimulatory 4-1BB, CD3z, IC9) Route: Single IV (n = 5) or IV + ICT if surgical candidates (n = 3) Dose: 2.5 mil cells/kg (IV), 100,000 cells/Kg (ICT) LD: yes (cyclo + fludara) | PR in 4/8, SD in 1/8 and PD in 3/8 on 28-day MRI Median OS 10 mo (3–24 mo) | No DLT 1 grade 3 headache 1 grade 2 seizure | CART expansion, peak at 1–3 weeks. CART detected in all pts at 4 weeks. Pts with PD were alive at 6–24 mo post-infusion; 1 confirmed treatment effect rather than TP at Bx. Antigen loss Increased CD8+ T cells, decreased M2 macrophages | |
| Ongoing | Criteria/enrollment | CAR and administration | Status | Start | Completion | |
| NCT04099797 (GAIL-B) Baylor College of Medicine | 34 pts with High grade glioma, DIPG, embryonal or ependymal tumors | CAR: C7R-GD2.CART Route: Ommaya reservoir/VP shunt Dose: 10 mil to 30 mil cells/m2 LD: yes (cyclo/fludara) | Recruiting | February 2020 | Primary: February 2025 Final: February 2039 | |
| Ongoing | Target and Criteria/enrollment | CAR and administration | Status | Start | Completion | |
| NCT05577091 Beijing Tiantan Hospital | CD133 CD44 [176] | Est. 10 patients with recurrent glioblastoma | CAR: 4th-gen dual-target truncated IL7Ra modified CAR T cells Route: Ommaya reservoir ITu Dosing: low-dose group and high-dose group receiving 1 dose, multidose-group receiving weekly infusions × 8 weeks max | Not yet Recruiting | May 2023 | Primary: November 2024 Final: November 2032 |
| NCT03423992 Xuanwu Hospital, Beijing | EGFRVIII, IL13Rα2, Her-2, EphA2, CD133, GD2 | Est 100 pts | CAR: Autologous T cells expressing CAR ± PD-L1 antibody | Recruiting | March 2018 | January 2023 Last updated June 2021 |
| NCT04214392 City of Hope | Chlorotoxin | 36 pts with MMP2+ recurrent for progressive GBM | CAR: Chlorotoxin (EQ)-CD28-CD3zeta-CD19t-expressing CAR T Route/dosing: Arm 1: single delivery, 3 weekly cycles: begins with 1 infusion intracranial intratumoral or intracavitary (ICT) and lasts for 1 week. Arm 2: dual delivery ICT and intraventricular | Recruiting | February 2020 | December 2024 |
| NCT04045847 Xijing Hospital | CD147 | 31 pts with Recurrent GBM | CAR: CD147-CART Route: Ommaya reservoir Dosing: 3 doses at weekly intervals | Unknown | May 2019 | May 2022 Last updated May 2022 |
| NCT05353530 (IMPACT) University of Florida | CD70 | Newly diagnosed MGMT unmethylated GBM | CAR: Autologous IL-8 receptor (CXCR2) modified CD70 CAR (8R-70CAR) Route: IV Dose: Single infusion 2 weeks after RT of 1 mil–100 mil cells LD: in 1 cohort | Not yet recruiting | October 2022 | December 2025 (primary completion) December 2040 |
| NCT03170141 Shenzhen Geno-Immune Medical Institute | Multiple (GD2, EGFRvIII, CD70) | Est 20 pts with recurrent GBM | CAR: EGFRvIII targeting CAR-T cells modified with immune modulatory genes (IgT) i.e., ICI Route: IV or ITu in 3 days (split dose) Dose: 50,000/kg to 25 mil/kg LD: yes (fludara and/or cyclo) | Enrolling by invitation | May 2020 | December 2023 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alsajjan, R.; Mason, W.P. Bispecific T-Cell Engagers and Chimeric Antigen Receptor T-Cell Therapies in Glioblastoma: An Update. Curr. Oncol. 2023, 30, 8501-8549. https://doi.org/10.3390/curroncol30090619
Alsajjan R, Mason WP. Bispecific T-Cell Engagers and Chimeric Antigen Receptor T-Cell Therapies in Glioblastoma: An Update. Current Oncology. 2023; 30(9):8501-8549. https://doi.org/10.3390/curroncol30090619
Chicago/Turabian StyleAlsajjan, Roa, and Warren P. Mason. 2023. "Bispecific T-Cell Engagers and Chimeric Antigen Receptor T-Cell Therapies in Glioblastoma: An Update" Current Oncology 30, no. 9: 8501-8549. https://doi.org/10.3390/curroncol30090619
APA StyleAlsajjan, R., & Mason, W. P. (2023). Bispecific T-Cell Engagers and Chimeric Antigen Receptor T-Cell Therapies in Glioblastoma: An Update. Current Oncology, 30(9), 8501-8549. https://doi.org/10.3390/curroncol30090619




